Abstract
HIV-infected women have higher rates of persistence of human papillomavirus (HPV) infection, of abnormal cervical cytology results, and of cervical cancer than uninfected women. It is currently recommended that HIV-infected, sexually active women have a Papanicolaou (Pap) test performed at the time of initial diagnosis of HIV infection, followed by annual Pap testing if the previous test result is normal. Women whose test results show abnormalities greater than atypical squamous cells of undetermined significance (ASCUS) should be referred for colposcopy. Those with ASCUS should undergo immediate colposcopy or repeat cervical cytology in 6 months to 12 months, and those whose repeat cervical cytology results show ASCUS or greater abnormalities should undergo colposcopy. Recent findings indicate that screening intervals can be lengthened for HIV-infected women whose Pap test results are persistently normal and who are engaged in routine care, and that HPV DNA testing may have a role in screening. This article summarizes a presentation by Marla J. Keller, MD, at the IAS–USA continuing education program, Improving the Management of HIV Disease, held in Atlanta, Georgia, in March 2015.
Keywords: HIV, human papillomavirus, HPV, cervical cancer screening, cervical cytology, oncogenic HPV DNA testing
More than 150 types of human papillomavirus (HPV) have been identified, including approximately 40 that can infect the cervix and approximately a dozen that are carcinogenic. HPV-16 accounts for 50% to 55% of all cases of invasive cervical cancer worldwide, and HPV-18 accounts for an additional 10% to 15%. Cervical cancer represents approximately 10% of all cancers that affect women worldwide, and virtually all are attributable to HPV.
Cervical cancer is the third most common type of cancer among women worldwide, with approximately 530,000 new cases and 275,000 related deaths occurring each year. Approximately 12,000 new cases of cervical cancer occur each year in the United States. The risk of cervical cancer, an AIDS-defining malignancy, is increased several fold in HIV-infected women.
Human Papillomavirus Infection in HIV-Infected Women
Clearance of HPV infection is extremely common. Typically, it takes decades for a high-grade squamous intraepithelial lesion (HSIL) to grow and become invasive. Before cervical precancer or cancer can develop, there must be persistent oncogenic HPV infection and an accumulation of genetic changes over time. Thus, a woman with a normal Papanicolaou (Pap) test result who does not have oncogenic HPV infection should have a low risk of developing cervical precancer or cancer for several years, regardless of her HIV serostatus. However, HIV-infected women have greater persistence of HPV infection compared to HIV-uninfected women. Persistence rates are 2- to 6-fold higher for any HPV type and 6-fold higher for HPV-16 and HPV-18.1,2
In the WIHS (Women’s Interagency HIV Study), HIV-infected and uninfected women have semiannual Pap testing, polymerase chain reaction (PCR) testing for more than 40 types of HPV, and colposcopy when indicated. Data on approximately 1900 HIV-infected women and 500 uninfected women who were observed for at least 10 years showed a cumulative risk of 77% and 50%, respectively, of having an abnormal cytology result and a cumulative risk of 4% and 1%, respectively, of having HSILs.3 Overall, HIV-infected women have greater diversity of HPV types; greater prevalence of numerous types of HPV; greater preponderance of types of HPV other than HPV-16 and HPV-18; higher prevalence of low-grade squamous intraepithelial lesions (LSILs) and HSILs; rates of progression of HPV infection that are more rapid; lower rates of spontaneous regression of HPV infection; and higher rates of persistent or recurrent HPV infection following treatment.
Overall, HIV-infected women have a 5.4-fold higher risk for cervical cancer and a 6.8-fold higher risk for anal cancer than uninfected women. Longer duration of HPV persistence, longer life expectancy owing to effective antiretroviral therapy, and greater proportions of women entering age groups in which cervical cancer rates reach their peak contribute to the excess risk of cervical cancer.4 Increased immunosuppression in HIV-infected women is associated with increased risk for cervical cancer. As shown in the Figure, the cervical cancer rate is higher in HIV-infected women with CD4+ cell counts below 200/μL than in HIV-infected women with CD4+ cell counts above 200/μL and uninfected women.5,6
Figure.
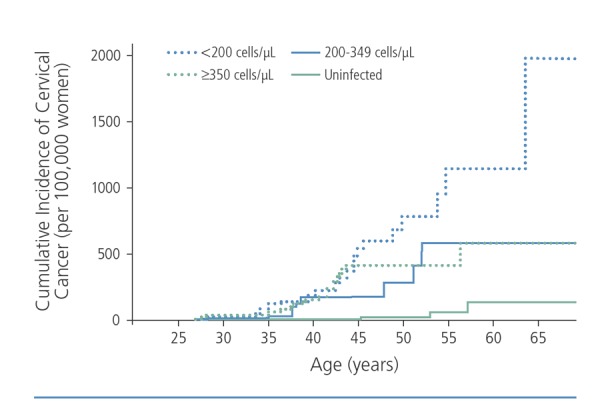
Risk for invasive cervical cancer according to CD4+ cell count in HIV-infected women. Adapted from Abraham et al.5
The incidence of cervical cancer in HIV-infected women has not decreased appreciably since the introduction of antiretroviral therapy. However, data indicate that HIV-infected women who are adherent to antiretroviral therapy experience substantial reductions in the burdens of HPV infection and SILs. A study in the WIHS cohort showed a 40% reduction in prevalence of oncogenic HPV infections, a 50% reduction in incidence of oncogenic HPV infections, and faster clearance of oncogenic HPV-related SILs after initiation of antiretroviral therapy.7 Benefits of antiretroviral therapy were reduced in women who were less adherent.
Cervical Cancer Screening Guidelines
Guidelines for screening for HPV-related cervical disease in HIV-uninfected women include those by the US Preventive Services Task Force (USPSTF)8; those endorsed by the American Cancer Society (ACS), the American Society for Colposcopy and Cervical Pathology (ASCCP), and the American Society for Clinical Pathology (ASCP)9; and those by the ASCCP that address the management of abnormal cervical cancer screening tests and cancer precursors.10 For HIV-infected women, the guidelines most often referred to are those for the prevention and treatment of opportunistic infections (OIs) in HIV-infected adults and adolescents endorsed by the Centers for Disease Control and Prevention (CDC), the National Institutes of Health (NIH), and the HIV Medicine Association (HIVMA) of the Infectious Diseases Society of America.11 A goal of each of these guidelines is to aid in the prevention of cervical cancer by screening, by evaluation of women with positive test results, and by treatment of biopsy-confirmed high-grade cancer precursors.
General Guidelines
Guidelines for screening of HIV-seronegative and -seropositive women for cervical cancer are compared in the Table.11,12 For HIV-seronegative women, it is recommended that screening for cervical cancer begin at age 21 years. Young women without HIV infection have a high rate of regression of HPV infection, and intervention with colposcopy or treatment is not generally recommended except in cases of high-grade disease.
However, HIV-infected women have higher rates of HPV infection and cervical intraepithelial neoplasia (CIN) and lower rates of regression of HPV infection. Thus, it is recommended that HIV-infected women who are younger than 21 years and who are sexually active be screened for cervical cancer within 1 year of onset of sexual activity but by no later than age 21 years. HIV-infected women who are aged 21 years to 29 years should have a Pap test performed at the time of initial diagnosis of HIV infection and then annually
if the previous Pap test result is normal. If the initial Pap test result shows an abnormality greater than atypical squamous cells of undetermined significance (ASCUS), then colposcopy should be performed; treatment for HIV-infected adolescents should be reserved for CIN of grade 3 or worse. Adult women with ASCUS should have a colposcopy performed immediately or should undergo repeat cytology in 6 months to 12 months. A finding of ASCUS or higher on repeat cytology results should prompt colposcopy. Screening for cervical cancer in HIV-infected women should continue throughout a woman’s lifetime and not, as for uninfected women, end at age 65 years. As with HIV-seronegative women, HIV-infected women with a history of high-grade cervical disease or invasive cervical cancer should continue to undergo annual Pap testing after a hysterectomy.
It is currently recommended that HIV-infected women undergo annual screening for cervical cancer, although new data suggest that women with serially negative Pap test results may consider a longer screening interval. A study in the WIHS cohort showed that among 942 HIV-infected women with normal Pap test results at baseline, high-grade CIN developed in 1% within 15 months and in 4% within 39 months.13 However, among women with normal results on 3 consecutive Pap tests, there were no cases of precancer after 15 months and 2% developed precancer after 39 months. None of the women developed precancer or cancer within 39 months after having normal results on 10 consecutive Pap tests. These findings indicate that a 3-year screening interval may be appropriate for HIV-infected women whose Pap test results are persistently normal and who are adherent to routine care.
Role of Human Papillomavirus DNA Testing
Current USPSTF, ACS, ASCCP, and ASCP recommendations for HIV-seronegative women indicate that a Pap test and HPV DNA test should be performed for women aged 30 years or older and, if the results are normal and negative, respectively, that screening should be performed again in 5 years.8 HPVDNA testing is appropriate for use in the triage of HIV-uninfected women with ASCUS to determine the need for colposcopy, in the management of postmenopausal women with LSILs, and as follow-up after a colposcopy or treatment procedure (eg, a loop electrosurgical excision procedure or cone biopsy). Current ASCCP guidelines recommend HPV DNA testing to triage ASCUS for HIV-infected and uninfected women. If Pap test results reveal ASCUS and reflex HPV DNA testing results are positive, then a colposcopy is recommended.
In a study in the WIHS cohort, HPV DNA testing was performed when ASCUS was first detected via a Pap test during semiannual follow-up over an 8-year period.14 Among evaluable cases (successful PCR amplification), HPV test results were positive in 16 of 17 (94%) patients with CIN of grade 2 or worse and in 35 of 97 (36%) patients with CIN of less than grade 2, yielding an overall sensitivity rate of 94% and an overall specificity rate of 64%. These findings suggest that HPV testing to triage ASCUS would detect most cases of CIN of grade 2 or worse and would allow individuals with CIN of less than grade 2, approximately two-thirds of patients in this study, to avoid colposcopy.
The current CDC/NIH/HIVMA OI guidelines recommend that the ASCCP guidelines be followed for the management of HIV-infected women with abnormal cervical screening test results. With regard to whether Pap and HPV DNA testing can be used to lengthen the screening interval in HIV-infected women with normal Pap test results and negative results on HPV DNA testing, a study in the WIHS cohort reported in 2005 showed a low 5-year risk of cervical precancer and cancer among HIV-seropositive women with normal Pap test results and negative HPV test results.15 However, this study was based on cytology data, lacked histology data, and included data from before the widespread use of potent antiretroviral therapy.
More recently, the incidence of precancer and cancer was assessed among 420 HIV-infected women and 279 uninfected women in the WIHS population.16 HIV-infected women with a normal Pap test result and a negative oncogenic HPV test result who were in long-term follow-up had a similar risk of cervical precancer and cancer to uninfected women through at least 5 years of follow-up. The median age was 33 years in the HIV-infected group and 29 years in the uninfected group, and 56% of the HIV-infected women had CD4+ cell counts of 500/μL or greater, 47% were taking antiretroviral therapy, and 88% tested negative for oncogenic HPV DNA. Analysis of outcomes in HIV-infected and uninfected women who had normal Pap test results and negative results on HPV DNA testing showed that HSIL occurred in 1 HIV-infected woman (CD4+ cell count > 500/μL) and 1 uninfected woman. The cumulative incidence of CIN of grade 2 or worse among HIV-infected women was 2% in those with a CD4+ cell count less than 350/μL, 2% in those with a CD4+ cell count of 350/μL to 499/μL, and 6% in those with a CD4+ cell count of 500/μL or greater, compared with 5% in uninfected women. Overall, the 5-year cumulative incidence of CIN of grade 2 or worse was 5% in HIV-infected and uninfected women, and the 5-year cumulative incidence of CIN of grade 3 or worse was 0.5% in HIV-infected women and 0.7% in uninfected women; no cases of cervical cancer were identified. Over 9 years of follow-up, the incidence of CIN of grade 3 or worse was 2% in HIV-infected women and 0.7% in uninfected women, with no cases of cervical cancer identified. These data led the CDC/NIH/HIVMA Panel on OIs in HIV-Infected Adults and Adolescents to recommend cotesting (Pap testing and HPV testing) as an acceptable screening approach for HIV-infected women aged 30 years or older.
Table 1.
Cervical Cancer Screening Guidelines
| USPSTF/ACS/ASCCP/ASCP Guidelines for Women Without HIV Infection | CDC/NIH/HIVMA Guidelines for Women With HIV Infection | |
|---|---|---|
| Age at initiation of screening | Age 21 y, regardless of risk factors | Within 1 y of onset of sexual activity but by no later than age 21 y |
| Frequency of screening | ||
| Age 21-29 y | Pap test every 3 y | Pap test every 3 y after 3 consecutive Pap test results are normal |
| Age ≥30 y | Pap test every 3 y or Pap test and HPV DNA test (cotesting) every 5 y | Pap test every 3 y after 3 consecutive Pap test results are normal, or Pap test and HPV DNA test (cotesting) every 3 y |
| Discontinuation of screening | Age 65 y | Never |
| Screening after hysterectomy | Discontinue for benign reasons and no history of CIN of grade 2 or worse for 20 y, otherwise rou- tine screening for at least 20 y | Discontinue for benign reasons and no history of CIN of grade 2 or worse, otherwise annual screening |
| HPV vaccinated | No change | No change |
Abbreviations: ACS, American Cancer Society; ASCCP, American Society for Colposcopy and Cervical Pathology; ASCP, American Society for Clinical Pathology; CDC, Centers for Disease Control and Prevention; CIN, cervical intraepithelial neoplasia; HIVMA, HIV Medicine Association; HPV, human papillomavirus; NIH, National Institutes of Health; Pap, Papanicolaou; USPSTF, US Preventive Services Task Force. Adapted from Moyer12 and Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents.11
Similarly, data from Kaiser Permanente Northern California from 245 HIV-infected women aged 30 years or older, who had normal Pap test results and negative HPV DNA test results, showed no cases of CIN of grade 2 or worse and 1 case of HSIL during 42 months of follow-up.17 These findings suggest that it may be acceptable to extend cervical cancer screening intervals by performing HPV testing for HIV-infected women who are in routine care, thereby minimizing the potential harms of cytologic screening, including psychological distress (anxiety, concern) related to positive results and risk of adverse effects with additional procedures (colposcopies and biopsies).
Pregnancy
Pregnant women with HIV infection should undergo similar screening to women with HIV infection who are not pregnant. The only finding that would affect the management, timing, and route of delivery of screening is invasive cancer. Pap testing is recommended at the initial prenatal visit unless a normal test result was obtained within the past year. Colposcopy to detect ASCUS or LSILs can be delayed until 6 weeks postpartum, with immediate colposcopy recommended for women with HSILs or atypical glandular cells. Treatment for CIN is not recommended unless invasive disease is expected. HPV vaccination is not recommended during pregnancy, although in 5 trials, vaccination in women who became pregnant did not appear to negatively affect pregnancy outcomes.18
Conclusion
Abnormal cervical cytology is common among HIV-infected women. At this time, annual Pap tests are the standard of care. For ASCUS, colposcopy or repeat cytology in 6 months to 12 months is recommended (colposcopy can be deferred until 6 weeks postpartum for pregnant women). Referral for colposcopy should occur if Pap test results reveal ASCUS or worse. There appears to be a role for cotesting for HPV infection to determine screening intervals in HIV-infected women, and a negative result on an oncogenic HPV test has a strong negative predictive value for precancer and cancer, similar to that seen in uninfected women.
The risk of cervical precancer in HIV-infected women with a positive oncogenic HPV test result despite normal cervical cytology is also important for setting screening practices. A recently completed study in the WIHS cohort showed that HIV-infected women with a normal Pap test result who test positive for HPV-16 have a high risk of cervical precancer that may warrant immediate colposcopy, whereas those who test positive for other oncogenic HPV types are at moderate risk that may warrant repeat cotesting in 1 year.19 Current data suggest that HIV-infected women may benefit from cotesting, as is recommended for the general female population. Future studies should incorporate HPV tests with greater specificity. Examples of molecular assays currently under investigation include the p16/Ki-67 cytology and E6/E7 messenger RNA assays. Currently, there are no national guidelines for routine anal Pap screening in HIV-infected women. At present, many specialists recommend anal cytologic screening, with test results that reveal abnormalities greater than ASCUS prompting a high-resolution anoscopy.
References
- 1.Sun XW, Kuhn L, Ellerbrock TV, Chiasson MA, Bush TJ, Wright TC, Jr. Human papillomavirus infection in women infected with the human immunodeficiency virus. N Engl J Med. 1997;337(19):1343-1349. [DOI] [PubMed] [Google Scholar]
- 2.Minkoff H, Feldman J, Dehovitz J, Landesman S, Burk R. A longitudinal study of human papillomavirus carriage in human immunodeficiency virus-infected and human immunodeficiency virus uninfected women. Am J Obstet Gynecol. 1998;178(5):982-986. [DOI] [PubMed] [Google Scholar]
- 3.Massad LS, Seaberg EC, Wright RL, et al. Squamous cervical lesions in women with human immunodeficiency virus: long-term follow-up. Obstet Gynecol. 2008;111(6):1388-1393. [DOI] [PubMed] [Google Scholar]
- 4.Frisch M, Biggar RJ, Goedert JJ. Human papillomavirus-associated cancers in patients with human immunodeficiency virus infection and acquired immunodeficiency syndrome. J Natl Cancer Inst. 2000; 92(18):1500-1510. [DOI] [PubMed] [Google Scholar]
- 5.Abraham AG, D'Souza G, Jing Y, et al. Invasive cervical cancer risk among HIV-infected women: a North American multicohort collaboration prospective study. JAIDS. 2013;62(4):405-413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Guiguet M, Boue F, Cadranel J, Lang JM, Rosenthal E, Costagliola D. Effect of immunodeficiency, HIV viral load, and antiretroviral therapy on the risk of individual malignancies (FHDH-ANRS CO4): a prospective cohort study. Lancet Oncol. 2009;10(12):1152-1159. [DOI] [PubMed] [Google Scholar]
- 7.Minkoff H, Zhong Y, Burk RD, et al. Influence of adherent and effective antiretroviral therapy use on human papillomavirus infection and squamous intraepithelial lesions in human immunodeficiency virus-positive women. J Infect Dis. 2010;201(5):681-690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. US Preventive Services Task Force. Cervical cancer: screening. http://www.uspreventiveservicestaskforce.org/Page/Document/UpdateSummaryFinal/cervical-cancer-screening. Accessed on November 5, 2015.
- 9.Saslow D, Solomon D, Lawson HW, et al. American Cancer Society, American Society for Colposcopy and Cervical Pathology, and American Society for Clinical Pathology screening guidelines for the prevention and early detection of cervical cancer. CA Cancer J Clin. 2012;62(3):147-172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Massad LS, Einstein MH, Huh WK, et al. 2012 updated consensus guidelines for the management of abnormal cervical cancer screening tests and cancer precursors. Obstet Gynecol. 2013;121(4):829-846. [DOI] [PubMed] [Google Scholar]
- 11.Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. http://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf. Accessed on November 5, 2015.
- 12.Moyer VA. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012; 156(12):880-891, W312. [DOI] [PubMed] [Google Scholar]
- 13.Massad LS, D'Souza G, Tian F, et al. Negative predictive value of pap testing: implications for screening intervals for women with human immunodeficiency virus. Obstet Gynecol. 2012;120(4):791-797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.D'Souza G, Burk RD, Palefsky JM, Massad LS, Strickler HD. Cervical human papillomavirus testing to triage borderline abnormal pap tests in HIV-coinfected women. AIDS. 2014;28(11):1696-1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Harris TG, Burk RD, Palefsky JM, et al. Incidence of cervical squamous intraepithelial lesions associated with HIV serostatus, CD4 cell counts, and human papillomavirus test results. JAMA. 2005; 293(12):1471-1476. [DOI] [PubMed] [Google Scholar]
- 16.Keller MJ, Burk RD, Xie X, et al. Risk of cervical precancer and cancer among HIV-infected women with normal cervical cytology and no evidence of oncogenic HPV infection. JAMA. 2012;308(4): 362-369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Castle PE, Fetterman B, Poitras N, Lorey T, Kinney W. Safety against cervical precancer and cancer following negative human papillomavirus and Papanicolaou test results in human immunodeficiency virus-infected women. Arch Intern Med. 2012;172(13):1041-1043. [DOI] [PubMed] [Google Scholar]
- 18.Garland SM, Ault KA, Gall SA, et al. Pregnancy and infant outcomes in the clinical trials of a human papillomavirus type 6/11/16/18 vaccine: a combined analysis of five randomized controlled trials. Obstet Gynecol. 2009;114(6):1179-1188 [DOI] [PubMed] [Google Scholar]
- 19.Keller MJ, Burk RD, Massad LS, et al. Cervical precancer risk in HIV-infected women who test positive for oncogenic human papillomavirus despite a normal Pap test. Clin Infect Dis. 2015;61(10):1573-1581. [DOI] [PMC free article] [PubMed] [Google Scholar]


