Abstract
During our investigation of the neuroprotective activity of Platycodi radix we found that an aqueous extract of this folk medicine exhibited significant protection against glutamate-induced toxicity in primary cultured rat cortical cells. In order to clarify the neuroprotective mechanism(s) of this observed effect, activity-guided isolation was performed to seek and identify active fractions and components. By such fractionation, four known triterpene saponin compounds - platycodins A, C and D and deapioplatycodin D - were isolated from the n-butanol fraction. Among these four compounds, platycodin A exhibited significant neuroprotective activities against glutamate-induced toxicity, exhibiting cell viability of about 50 %, at concentrations ranging from 0.1 µM to 10 µM. Therefore, the neuroprotective effect of Platycodi radix might be due to the inhibition of glutamate-induced toxicity by the saponin compounds it contains.
Keywords: Platycodi radix, neuroprotective, triterpenes saponin, glutamate-induced toxicity
Introduction
Neuronal death is an important feature of both acute and chronic neurodegenerative diseases. Alzheimer’s disease (AD) is a progressive neurodegenerative disorder, the most common form of dementia in the elderly [1] and a major cause of morbidity and disability in the adult population. AD is associated with the accumulation of L-glutamate (Glu) deposits in senile plaques and neurofibrillary tangle lesions in specific areas of brain. L-Glutamate is the major excitatory neurotransmitter in the CNS, involved in fast synaptic transmission, neuronal plasticity, outgrowth and survival, memory, learning and behaviour [2]. However, glutamate-mediated excitotoxicity appears to play a crucial role in neurodegenerative disorders, particularly in Parkinson’s disease, Alzheimer’s disease, epilepsy, spinal cord trauma, and ischemic stroke [3]. Thus, neuroprotection against glutamate-induced toxicity has been a therapeutic strategy for preventing and/or treating both acute and chronic forms of neurodegeneration [4].
The roots of Platycodon grandiflorum (Jacq.) A.DC. (Campanulaceae) have been used in the traditional Chinese folk medicine ‘‘Platycodi radix’’, which is used as an antiphlogistic, antitussive and expectorant agent in China, Korea, and Japan. Modern pharmacological research suggests that extracts from P. radix possess wide-ranging health benefits and this plant is presently a hot research topic in South Korea and Japan because of its potential health care uses. Many novel and remarkable activities have been discovered in succession. Previously, the fresh roots of P. grandiflorum have been eaten as pickles for preventing NF-kappaB activity [5]. However, the component(s) responsible for the neuroprotective effects of P. radix still remain unidentified. In the context of our natural product chemistry program dealing with the development of new potent neuroprotection agents, we have examined the isolation of triterpenes saponin compounds as leads for novel glutamate-induced toxicity inhibitors. As part of our continued study of the neuroprotective effects of P. radix, we have now isolated single compounds (platycodin A, C, D and deapioplatycodin D) from the methanol extract of P. radix and examined their inhibitory effects on glutamate-induced toxicity in primary cultured rat cortical cells.
Results and Discussion
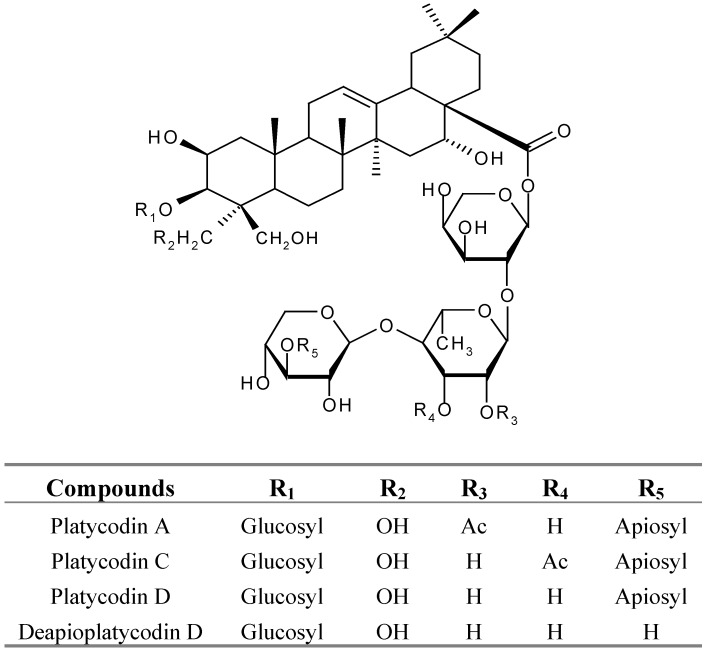
In the present experiments, we found that an aqueous extract of P. radix might inhibit glutamate-induced toxicity in primary cultured rat cortical cells. In order to clarify the neuroprotective components of P. radix, as part of a continued study of neuroprotection effects of P. radix, activity-guided isolation was performed to seek active fractions and components. After solvent fractionation, we compared the inhibiting effects of various fractions on neuroprotective activity and the aqueous extract of P. radix was found to inhibit the this activity in a dose-dependence manner in the assay system using glutamate-induced toxicity in primary cultured rat cortical cells. Total saponins were found to have significant inhibitory activity on neuroprotection, while the ether and EtOAC fractions have no such activity towards glutamate-induced toxicity in primary cultured rat cortical cells. The above-mentioned results suggested that the neuroprotective actions of the aqueous extract of P. radix may be attributed in part to its total saponin content. To clarify the active substances of P. radix, we examined the effects of the major secondary metabolites from P. radix, the total saponin fractions, on neuroprotective activity. Activity-guided isolation was further performed to yield single compounds. Four known triterpenoid saponin compounds were isolated from total saponin fraction by repeated column chromatography. These compounds were identified as platycodin A, C, D and deapioplatycodin D (Figure 1) by comparing physicochemical and spectroscopic data with literature values [6,7].
Figure 1.
Triterpene saponins isolated from P. radix.
Furthermore, to investigate and compare the neuroprotective activities of these triterpene saponins isolated from the total saponin fraction of P. radix, the activities of platycodin A, C, D and deapioplatycodin D were evaluated in glutamate-injured primary cultured rat cortical cells at concentrations ranging from 0.1 to 10 μM (Table 1). It is notable that the neuroprotective activities of both platycodin C, deapioplatycodin D and platycodin D were comparable to those of MK-801, APV and CNOX, all ineffective, and among the tested four triterpene saponins, only platycodin A displayed activity, significantly attenuating glutamate-induced toxicity at concentrations ranging from 0.1 to 10 μM and exhibiting cell viabilities of 50-60 %. Although more extensive structure-activity study might be needed, these results indicate that substitution of a Ac-group at R3 of triterpenes seems to increase the neuroprotective activity. Although the cellular and molecular mechanisms that underlie the neuroprotective action of several effective triterpenes saponin revealed in the present study should be studied further, these findings about triterpenes saponin of P. radix containing neuroprotective activities may provide some ideas for the development of agents to ameliorate the cellular dysfunction due to glutamate-induced toxicity.
Table 1.
Neuroprotective effects of 4 triterpene saponin from P. radix against glutamate-induced toxicity in primary cultured rat cortical cells a.
| Compounds | Cell viability b (%) | ||
| 0.1 | 1 | 10 | |
| Control c | 100 | ||
| Glutamate-treated c, e | 0 | ||
| Platycodin A | 13.6 ± 0.4* | 32.5 ± 4.8* | 52.4 ± 3.8*** |
| Platycodin C | 11.6 ± 2.8 | 20.3± 2.5 | 16.3 ± 2.1 |
| Platycodin D | 16.5 ± 2.7 | 21.6 ± 2.1 | 26.5 ± 3.7 |
| Deapioplatycodin D | 22.3 ± 3.8 | 21.2 ± 1.3 | 26.5 ± 2.0 |
| APV f | 10.9 ± 2.2 | 23.9 ± 2.8 | 40.0 ± 3.8* |
| MK-801 g | 51.8 ± 4.8** | 63.8 ± 5.4*** | 72.8 ± 4.9*** |
| CNQX h | 28.3 ± 3.4* | 41.5 ± 3.6* | 51.5 ± 4.8*** |
-
a Rat cortical cell cultures were incubated with test compounds for 1h. The cultures were then exposed to 100 μM glutamate for 24 hrs. After the incubation, the cultures were assessed for the extent of neuronal damage;
-
b Cell viability was measured by the LDH assay;
-
c LDH released from control and glutamate-treated cultures were 11.9 ± 1.2 and 48.3 ± 4.1 units/mL, respectively;
-
d Cell viability was calculated as 100 x (LDH released from glutamate-treated-LDH released from glutamate+test compound-treated)/(LDH released from glutamate-treated-LDH released from control). The values shown are the mean ± STD of three experiments (3-4 cultures per experiment). Results differ significantly from the glutamate-treated: * p < 0.05, ** p < 0.01, *** p < 0.001;
-
e Glutamate-treated value differed significantly from the untreated control at the level of p < 0.001;
-
f APV: DL-2-amino-5-phosphonovaleric acid, a competitive NMDA receptor antagonist;
-
g MK-801: dizocilpine maleate, a noncompetitive NMDA receptor;
-
h CNQX: 6-cyano-7-nitroquinoxaline-2,3-dione, non-NMDA receptor antagonist.
Experimental
General
UV: UV-2200 UV–VIS recording spectrophotometer (Shimadzu, Japan); IR: Jasco Report-100 spectrophotometer; NMR: Bruker AMX 400 spectrometer (Bruker, USA), the chemical shifts being represented as ppm with tetramethylsilane as a internal standard; GC-MS (HP 5890 series II plus GC, HP 5972 series Mass Selective Detector, Column: HP-1MS); column chromatography: silica gel 60 (70~230 and 230~400 mesh, Merck), Sephadex LH-20 (Pharmacia, Sweden) and YMC-GEL ODS-A (12 nm, S-75 mm, YMC); TLC: pre-coated silica gel 60 F254. Dizocilpine maleate(MK-801), DL-2-amino-5-phosphonovaleric acid(APV) and 6-cyano-7-nitroquinoxaline-2,3-dione (CNOX) were purchased from Sigma Chemical Company (St. Louis, MO, USA).
Plant material
The P. radix was purchased in a local Oriental medicine market in Seoul, South Korea and voucher specimens (WKU-0705) have been deposited in the Herbarium of the Inam Neuroscience Research Center, Sanbon, Wonkwang University.
Plant material extraction, the total saponin from P. radix.
The crushed dried P. radix (5 kg) were extracted with MeOH five times under reflux for 6 h yielding 512 g of a brown solid extract, 500 g of which was suspended in MeOH (2 L) and precipitated by adding a 5-fold volume of acetone. The supernatant was concentrated in vacuo to yield a brown gum, which was suspended in H2O (1 L) and defatted with Et2O. The aqueous layer was extracted with aqueous saturated n-BuOH. The resulting n-BuOH solution was concentrated in vacuo to yield the n-BuOH fraction (132 g). A portion of the n-BuOH fraction (100 g) was suspended in water (1 L), the soluble fraction was subjected to a macroreticular resin column chromatography and eluted first using water for desugarizing and decoloring, then with an EtOH-water gradient. The various eluted fractions were combined and concentrated in vacuo until the smell of EtOH had disappeared, then the concentrated solution was freezed to dryness to yield the total saponin powder (82.3 g).
Isolation of platycodins from total saponins
The above-mentioned total saponin lyophilized powder (80 g) was chromatographed on a silica gel column and eluted using a CHCl3-MeOH-water elution system (65:35:10, v/v/v, lower layer) to yield 10 fractions; these fractions were further separated by repetitive silica gel, Sephadex LH-20, ODS column chromatography. Four known triterpenoid saponin compounds were isolated. The structures of these compounds were identified by comparing their physicochemical and spectroscopic data with previous reported results.
Cortical cell culture and Cell viability assessment
Primary cultures of mixed cortical cells containing both neurons and glia were prepared from 17~19-day-old fetal rats (Sprague-Dawley) as described previously [8]. Cultures were allowed to mature for at least 2 weeks before being used for experiments. Test fraction and compounds were dissolved in DMSO (final concentration in culture, 0.1%). Cortical cell cultures were washed with DMEM and incubated with test compounds for 1 hr. The cultures were then exposed to 100 μM glutamate and maintained for 24 hr. After the incubation, the cultures were assessed for the extent of neuronal damage by measuring the efflux of LDH (lactic dehydrogenase) which reflects the integrity of cellular membrane.
Statistical analysis
The results are expressed as means ± standard errors (S.E.). The data were statistically analyzed by one way ANOVA. Differences with P < 0.05 were considered significant.
Acknowledgments
This work has been supported by Wonkwang University (2005). Dr Moon is thankful to INRC for start-up funds and for partial support of this work.
Footnotes
Sample Availability: Samples of the compounds are available from authors.
References
- 1.Wang R., Zhou J., Tang X.C. Tacrine attenuates hydrogen peroxide-induced apoptosis by regulating expression of apoptosisrelated genes in rat PC12 cells. Brain Res. Mol. Brain Res. 2002;107:1–8. doi: 10.1016/S0169-328X(02)00402-3. [DOI] [PubMed] [Google Scholar]
- 2.Sucher N. J., Awobuluyi M., Choi Y. B., Lipton S. A. NMDA receptors: from genes to channels. Trends Pharmacol. Sci. 1996;17:348–355. [PubMed] [Google Scholar]
- 3.Cacabelos R., Takeda M., Winblad B. The glutamatergic system and neurodegeneration in dementia: preventive strategies in Alzheimer’s disease. Int. J. Geriatr. Psychiatry. 1999;14:3–47. doi: 10.1002/(sici)1099-1166(199901)14:1<3::aid-gps897>3.0.co;2-7. [DOI] [PubMed] [Google Scholar]
- 4.Trist D. G. Excitatory aminoacid agonists and antagonists: pharmacology and therapeutic applications. Pharm. Acta Helv. 2000;74:221–229. doi: 10.1016/s0031-6865(99)00053-9. [DOI] [PubMed] [Google Scholar]
- 5.Ahn K.S., Hahn B.S., Kwack K., Lee E.B., Kim Y.S. Platycodin D-induced apoptosis through nuclear factor-kappaB activation in immortalized keratinocytes. Eur. J. Pharmacol. 2006;537:1–11. doi: 10.1016/j.ejphar.2006.03.012. [DOI] [PubMed] [Google Scholar]
- 6.Tada A., Kaneiwa Y., Shoji J., Shibata S. Studies on the saponins from roots of Platycodon grandiflorum A. De Candolle. I. Isolation and the structure of platycodin-D. Chem. Pharm. Bull. 1975;23:2965–2972. doi: 10.1248/cpb.23.2965. [DOI] [PubMed] [Google Scholar]
- 7.Ishii H., Tori K., Tozyo T., Yoshimura Y. Saponins from roots of Platycodon grandiflorum. Part 2. Isolation and structure of new triterpene glycosides. J. Chem. Soc. Perkin Trans. I. 1984:661–668. [Google Scholar]
- 8.Kang S.Y., Lee K.Y., Sung S.H., Kim Y.C. Four new neuroprotective dihydropyranocoumarins from Angelica gigas. J. Nat. Prod. 2005;68:56–69. doi: 10.1021/np049705v. [DOI] [PubMed] [Google Scholar]