ABSTRACT
Carboplatin is a platinum-based chemotherapeutic agent used for the treatment of many solid tumors. Peripheral neuropathy is a common side effect; but, to our knowledge, ocular motor cranial neuropathies have not been reported in the literature. We describe a case of persistent third and fourth nerve palsies after systemic administration of intra-arterial carboplatin for glioblastoma multiforme. Neither nerve regained function after carboplatin was stopped.
KEYWORDS: Carboplatin, glioblastoma, cranial neuropathy
Introduction
The Food and Drug Administration first approved platinum-containing drugs in 1978 as chemotherapeutic agents for various types of cancer. Their effectiveness in crosslinking DNA, interfering with DNA repair mechanisms, and subsequently inducing apoptosis in tumor cells gives them a broad spectrum of activity against many solid tumor types. Carboplatin is typically used for ovarian, lung, head, and neck cancers, but it is also efficacious for primary intracranial tumors.1 Common side effects of carboplatin include bone marrow suppression, renal insufficiency, electrolyte abnormalities, and peripheral neuropathy. Approximately 1% of patients experience visual disturbances such as diplopia.2 Peripheral neuropathy occurs in approximately 5% of patients, and neurotoxicity is often the dose-limiting factor in treatment. It typically occurs after cumulative dosing in a sensory, length-dependent pattern, appearing symmetrically in the most distal parts of the extremities and moving proximally.3 While symptoms such as numbness, tingling, weakness, and tremor are relatively common,4 to our knowledge, ocular motor cranial neuropathies have not been reported. This report describes a patient with glioblastoma multiforme who developed persistent third and fourth nerve palsies after intra-arterial (IA) administration of carboplatin.
Case description
A 38-year-old man diagnosed with glioblastoma multiforme of the left parietal lobe in 2013 presented with double vision in April 2017. He was initially treated with chemoradiation followed by two cycles of adjuvant temozolomide. After continued tumor growth, he underwent 12 cycles of uncomplicated IA carboplatin in the left internal carotid artery with intravenous (IV) bevacizumab, which he completed in December 2014. He was subsequently managed conservatively until March 2016 when his tumor burden increased again. He resumed IA carboplatin 880 mg/m2 with IV bevacizumab every 5 weeks. Within a few days of completing the 11th cycle, he developed left ptosis and ophthalmoplegia. Brain MRI was negative for new tumor growth, radiation necrosis, cranial nerve enhancement, or stroke. Brain CT angiogram did not show an aneurysm. He did not improve with dexamethasone treatment.
On examination, his visual acuities were 20/20, right eye and 20/40 improving to 20/25 with pinhole, left eye. His right pupil was normal but his left showed a complete efferent defect without afferent defect. There was complete ptosis of the left upper eyelid and a complete left third nerve palsy with lack of torsional movements on attempted vertical ductions indicating a concomitant left 4th nerve palsy (Figure 1). The remainder of the ophthalmologic (including color vision) and neurologic exam was unremarkable. He did not develop peripheral neuropathy in his extremities. Repeat MRI brain did not show a lesion to explain the cranial neuropathies. Seven months later, there was no change in his eye movements. He continued IV bevacizumab without complication.
Figure 1.
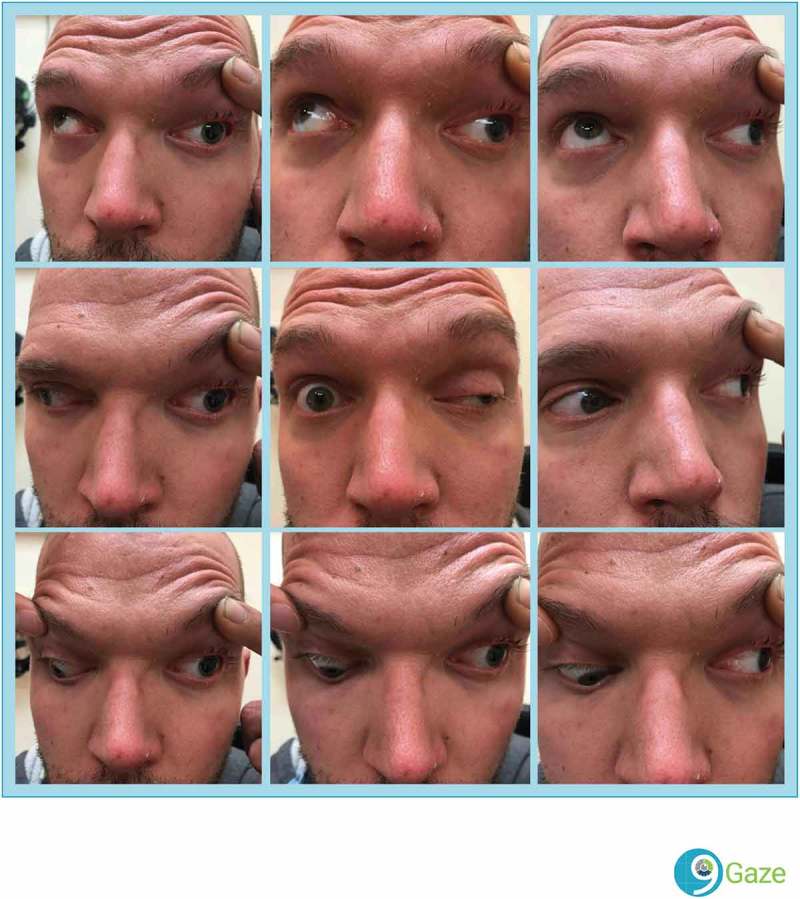
Nine gaze montage demonstrates significant ptosis of the left upper eyelid along with poor elevation, adduction, and depression of the left eye consistent with a third nerve palsy. The lack of torsion indicating a fourth nerve palsy is not discernible on this figure.
Discussion
Carboplatin is a commonly used, platinum-based chemotherapeutic agent, and peripheral neuropathy is a well-known side effect of treatment, affecting 4–6% of patients.4 Neurotoxicity often hinders the administration of efficacious dosing and results in treatment withdrawal.5 Most cases of neuropathy are length- and dose- dependent, and occur after 3 to 4 cycles of treatment. Symptom onset often occurs after cumulative doses of 600 mg/m2. Early symptoms include paresthesias, tingling, and numbness. Continued dosing can eventually affect proprioception, resulting in gait ataxia. The neuropathy is usually irreversible, and patients report a significant decrease in quality of life.5
The postulated pathogenesis of neurotoxicity relates to the affinity of carboplatin for the dorsal root ganglia (DRG).3,5,6 Two mechanisms of injury have been suggested. The formation of platinum inter-strand DNA crosslinks may alter cell cycle kinetics. Alternatively, increased mitochondrial oxidative stress leads to DRG apoptosis.3
Cranial neuropathy induced by carboplatin has not been noted in the literature to our knowledge. Since carboplatin has a known association with peripheral neuropathy, it is highly likely that it could produce a peripheral cranial neuropathy. Our patient developed persistent third and fourth nerve palsies after his second round of 11 cycles of carboplatin and bevacizumab. In our experience (JET), MRI abnormalities do not occur in carboplatin-induced peripheral neuropathy. While laser Doppler imaging and high-resolution ultrasound may be useful for evaluation of carboplatin-induced peripheral neuropathy, neither of these were performed on this patient.7 No causative lesion was identified as a source of his symptoms on neuroimaging.
Carboplatin is most often delivered via IV administration; however, IA administration is indicated for primary intracranial tumors. Current evidence suggests that IA administration of chemotherapy for primary CNS tumors leads to higher concentrations of the drug reaching the tumor and may help explain why our patient developed cranial neuropathies. The IA administration in the left carotid artery would comport with the left-sided neuropathies as well. This provides superior tumor control and leads to improved outcomes.8–10 Specifically for glioblastoma, IA chemotherapy has been shown to prolong survival and reduce the risk of adverse events.9 The patient herein did not develop peripheral neuropathy.
The patient herein was also on bevacizumab. Two cases of transient sixth nerve palsies after administration of bevacizumab have been reported. Common adverse events from bevacizumab include hypertension, poor wound healing, and arterial thrombosis,8 but peripheral neuropathy has not. Both cases of bevacizumab included patients in their sixties who enjoyed complete resolution of the sixth nerve palsy within 3 months. It is much more likely that these represented microvascular insults, which typically affect patients in this age group and resolve spontaneously in 3–4 months.11,12, It seem unlikely that our patient’s persistent ocular motor cranial neuropathies are the result of bevacizumab based on previous reports. His young age would argue against this as a microvascular palsy. The lack of improvement consistent with permanent peripheral neuropathies from carboplatin would suggest this is an adverse event from the chemotherapy.
Tumor recurrence was considered as another alternative explanation for this patient’s symptoms; however, the lack of radiographic evidence on serial MRI makes this unlikely. CSF seeding of intracranial tumors is most often preceded by direct invasion of at least one ventricle, which was not seen on follow-up MRI of this patient.13 CSF seeding typically presents with spinal symptoms over an average of 4 weeks, and post-seeding survival averages 3–4 months.14,15 Leptomeningeal carcinomatosis would typically show progressive neurologic and MRI findings over the 6 months of follow-up. This was not the case in our patient, and therefore, a lumbar puncture was not performed. Radiation-related injury was also considered, but the radiation fields did not include the brainstem or ocular motor nerves. There was also no evidence of radiation necrosis or cranial nerve enhancement to support this.
To our knowledge, this is the first report of ocular motor cranial neuropathy secondary to intra-arterial carboplatin. Because platinum drugs are used in approximately half of all patients receiving anti-cancer chemotherapy,16 it is imperative to increase awareness of rare side effects to differentiate these effects from symptoms of the tumor itself and to allow for appropriate dosing and management. Unfortunately, the cranial neuropathies may not improve similar to what occurs with carboplatin-related peripheral neuropathies.
Declaration of interest
The authors report no conflicts of interest.
References
- 1.Dasari S, Tchounwou PB.. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014;740:364–378. doi: 10.1016/j.ejphar.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Truven Health Analytics DynaMed Plus [Internet]. Ipswich, MA: EBSCO Information Services; 1995. Record No. 233477, Carboplatin http://www.dynamed.com/login.aspx?direct=true&site=DynaMed&id=233477. Published April 04, 2017. Accessed July 28, 2017 [Google Scholar]
- 3.Griswold W, Cavaletti G, Windebank A. Peripheral neuropathies from chemotherapeutics and targeted agents: diagnosis, treatment, and prevention. Neuro Oncol. 2012;14:iv45–iv54. doi: 10.1093/neuonc/nos203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Amptoulach S, Tsavaris N. Neurotoxicity caused by the treatment with platinum analogues. Chemother Res Pract. 2011:843019. doi: 10.1155/2011/843019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Avan A, Postma T, Ceresa C, et al. Platinum-induced neurotoxicity and preventive strategies: Past, present, and future. Oncologist. 2014;20(4):411–432. doi: 10.1634/theoncologist.2014-0044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Argyriou A. Chemotherapy-Induced Peripheral Neurotoxicity (CIPN): An update. Crit Rev Oncol/ Hematol. 2017;82(1):51–77. doi: 10.1016/j.critrevonc.2011.04.012. [DOI] [PubMed] [Google Scholar]
- 7.Addington J, Freimer M. Chemotherapy-induced peripheral neuropathy: An update on the current understanding. F1000Res. 12 November 2017;5(2016):F1000 Faculty Rev–1466. PMC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.M, Veras L, Zakashansky K. Bevacizumab-induced transient sixth nerve palsy in ovarian cancer: a case report. Asia Pac J Clin Oncol. 2016;12(1):e196–e198. doi: 10.1111/ajco.12129. [DOI] [PubMed] [Google Scholar]
- 9.Theodotou C, Shah A. The role of intra-arterial chemotherapy as an adjuvant treatment for glioblastoma. Br J Neurosurg. 2013;28(4): 438–446. [DOI] [PubMed] [Google Scholar]
- 10.Angelov L, Doolittle N, Kraemer D, et al. Blood-brain barrier disruption and intra-arterial methotrexate-based therapy for newly diagnosed primary CNS lymphoma: A multi-institutional experience. J Clin Oncol. 2009;27(21):3503–3509. doi: 10.1200/JCO.2008.19.3789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Momeni M, Veras L, Zakashansky K. Bevacizumab-induced transient sixth nerve palsy in ovarian cancer: A case report. Asia Pac J Clin Oncol. 2016;12(1):e196–e198. doi: 10.1111/ajco.12129. [DOI] [PubMed] [Google Scholar]
- 12.Park H, Guy J. Sixth nerve palsy post intravitreal bevacizumab for AMD: A new possibly causal relationship and complication? Binocul Vis Strabismus Q. 2007;22(4):209. [PubMed] [Google Scholar]
- 13.Bryan P. CSF seeding of intra-cranial tumours: A study of 96 cases. Clin Radiol.1984;25.3:355–360. [DOI] [PubMed] [Google Scholar]
- 14.Schwaninger M. Spinal canal metastases: A late complication of glioblastoma. J Neurooncol.1992;12(1):93–98. [DOI] [PubMed] [Google Scholar]
- 15.Fakhrai N. Glioblastoma with spinal seeding. Strahlentherapie Und Onkologie.2004;180(7):455–457. doi: 10.1007/s00066-004-1293-3. [DOI] [PubMed] [Google Scholar]
- 16.Johnstone T, Park G, Lippard S. Understanding and improving platinum anticancer drugs – phenanthriplatin. Anticancer Res. 2014;34(1):471–476. [PMC free article] [PubMed] [Google Scholar]


