Abstract
Objective
To describe our experience using patient-specific tissue-like kidney models created with advanced three-dimensional (3D)-printing technology for preoperative planning and surgical rehearsal prior to robot-assisted laparoscopic partial nephrectomy (RALPN).
Patients and Methods
A feasibility study of 10 patients with solid renal masses who underwent RALPN after preoperative rehearsal using 3D-print kidney models. A single surgeon performed all surgical rehearsals and procedures. Using standard preoperative imaging and 3D reconstruction, we generated pre-surgical models using a silicone-based material. All surgical rehearsals were performed using the da Vinci® robotic system (Intuitive Surgical Inc., Sunnyvale, CA, USA) before the actual procedure. To determine construct validity, we compared resection times between the model and actual tumour in a patient-specific manner. Using 3D laser scanning in the operating room, we quantified and compared the shape and tumour volume resected for each model and patient tumour.
Results
We generated patient-specific models for 10 patients with complex tumour anatomy. R.E.N.A.L. nephrometry scores were between 7 and 11, with a mean maximal tumour diameter of 40.6 mm. The mean resection times between model and patient (6:58 vs 8:22 min, P = 0.162) and tumour volumes between the computer model, excised model, and excised tumour (38.88 vs 38.50 vs 41.79 mm3, P = 0.98) were not significantly different.
Conclusions
We have developed a patient-specific pre-surgical simulation protocol for RALPN. We demonstrated construct validity and provided accurate representation of enucleation time and resected tissue volume. This simulation platform can assist in surgical decision-making, provide preoperative rehearsals, and improve surgical training.
Keywords: robotic partial nephrectomy, RALPN, surgical simulation, 3D printing, #KidneyCancer, #KCSM
Introduction
Partial nephrectomy (PN) has become an established standard for the nephron-sparing treatment of T1 renal parenchymal tumours. Technological advances over the past two decades have fuelled a paradigm shift from open to minimally invasive techniques for PN 1. Robotic-assisted laparoscopic PN (RALPN) has had a major impact on patient management by significantly reducing morbidity, while providing equivalent oncological outcomes to open PN 2, 3. The most important phases of this surgery are (a) exposure of the tumour-bearing kidney, (b) vascular control of the renal hilum, (c) identification and exposure of the renal mass, (d) excision of the tumour and (e) reconstruction by closure of medullary vascular and collecting system structures and the parenchymal defect. The complexity of RALPN is highly variable and depends on patient factors, as well as the location and size of the mass and its relationship to vital kidney structures. There is ongoing debate about the optimal technique for tumour removal: either wedge resection of the tumour bearing segment or tumour enucleation 4. Many surgeons now favour an enucleation approach that minimises unnecessary removal of normal functional nephrons and minimises positive margin rates. Tumour enucleation can be technically more difficult as the integrity of the tumour capsule should not be compromised to achieve a negative margin.
The R.E.N.A.L. nephrometry score, which is based on tumour size and location, is a standardised scoring system designed to capture the difficulty of surgical resection for a specific tumour 5, 6 Preoperative CT or MRI, as well as intraoperative ultrasonography, facilitate tumour resection with negative surgical margins and maximal preservation of normal renal parenchyma. Three-dimensional (3D) reconstruction and printing of kidneys containing renal masses has been reported, but has been limited to aiding anatomical visualization and preoperative planning 7, 8 Conventional 3D-printing techniques have limited capability to generate a high-fidelity model with tissue-like properties suitable for ‘hands-on’ surgical simulation and preoperative case rehearsal. In the present study, we investigate a novel rapid prototyping protocol for generating patient-specific customized 3D models with soft tissue properties and investigate whether these models have construct validity in simulating actual RALPN.
Patients and Methods
We prospectively enrolled 10 patients with complex renal masses (nephrometry score ≥7) that were amenable to RALPN. Patient demographics and tumour characteristics are listed in Table 1. A patient-specific 3D digital model was reconstructed based on the available preoperative axial imaging (CT or MRI) and then converted into a cast silicone soft-tissue model as described below. Patients were not subjected to any additional cross-sectional imaging to facilitate model development. Robot-assisted laparoscopic enucleation of the kidney tumour was rehearsed for all patients using the 3D-printed model, with the da Vinci®Si robotic surgical system in a three-arm configuration (Intuitive Surgical Inc., Sunnyvale, CA, USA), before performing the RALPN on each individual patient. Informed consent was obtained from all patients for the surgical procedure. The clinical characteristics for each tumour including volume, location, final pathology, margin status, and resection time were all prospectively collected. This study has been approved under an Institutional Review Board protocol at Baylor College of Medicine (protocol number H-37409 ‘Validation of 3D printed models in robotic urologic surgery’).
Table 1.
Tumour characteristics included in preoperative rehearsal
| Patient identification number |
Maximal tumour diameter on imaging, mm |
R.E.N.A.L. nephrometry score |
Final pathology |
Pathological tumour stage |
|---|---|---|---|---|
| 1 | 35 | 7a | ccRCC | pT1a |
| 2 | 15 | 8a | ccRCC | pT1a |
| 3 | 31 | 9p | ccRCC | pT1a |
| 4 | 61 | 7x | ccRCC | pT1b |
| 5 | 25 | 9a | ccRCC | pT1a |
| 6 | 28 | 9a | chRCC | pT1a |
| 7 | 29 | 11x | ccRCC | pT3a |
| 8 | 50 | 9xh | ccRCC | pT1b |
| 9 | 60 | 10a | ccRCC | pT1b |
| 10 | 72 | 10p | ccRCC | pT2 |
cc, clear cell; ch, chromophobe.
Creation of Silicone Kidney Models Based on Preoperative CT or MRI
Using 3D Slicer (https://www.slicer.org/) editing software, a label map was created for each axial slice that designated the mass with one label and renal parenchyma with a second label. For quality control, this label map was overlapped with the sagittal and coronal slices, and any necessary changes were made. If a second scan series was available for the patient, the label map was then compared to the second series to ensure consistency. Once the label map was completed, a 3D reconstruction of the tumour and parenchyma was created using 3D Slicer Model Maker. Models were created using the Joint Smoothing, Point Normals and Pad options with a decimation of 0% and smoothing of 40%. The resulting in silico 3D model was saved in .stl format and then subjected to final processing and refinement. Upon receiving the 3D model of the patient’s kidney, rapid prototyping of the physical patient kidney model was performed by Lazarus 3D (Houston, TX, USA). The physical kidney models were compared to the 3D reconstruction and the patient radiology data to confirm coherence in the spatial relationship between kidney and renal mass.
Models were manufactured using mixtures of silicone rubber [shore hardness 10A, Die B tear strength of 17.863 N/mm (102 pounds/linear inch), tensile strength 3.2750 MPa (475 psi)] and silicone oil as a thinner. In a series of preliminary studies, different mixes of these materials were tested to formulate the mixture that most realistically simulated normal kidney and tumour tissue properties. The best performance was found using a parenchyma consisting of 70% by volume silicone rubber and 30% by volume thinner. Colorants were included in the mix to achieve a distinct colour. The tumour was created using 85% silicone rubber and 15% thinner and was coloured orange. These mixes result in a tumour with slightly higher shore hardness, tensile strength, and tear strength when compared with the renal parenchyma.
Preoperative Rehearsal on the Robotic Platform
All cases were rehearsed with the patient-specific 3D model using the da Vinci Si robotic surgical system before performing the corresponding patient’s surgery. All cases were performed by the same surgeon (R.E.L.) who later performed the RALPN. We used a standard laparoscopic trainer box and robotic instruments for the simulation. The 3D model was aligned in the box to reflect the anatomical position expected for the in vivo surgical procedure, allowing for similar angles and a realistic configuration. In posterior tumours, in which we anticipated flipping the kidney over medially to access the posterior surface from a transperitoneal anterior approach, the model was positioned such that it would reflect the most likely position at the time of the enucleation. The tumour resection was timed and video recorded. The surgical margin on the model was determined to be negative if there was no silicone of the distinct tumour colour visible in the resection bed, indicating complete excision of the mass. The excised tumour model was then laser scanned using the Matter and Form 3D Scanner (Toronto, ON, Canada).
RALPN
All patients underwent a RAPLN by one surgeon (R.E.L.) at our institution. Patients were positioned in the modified flank position with their side elevated ~30 °. The camera port was placed in a lateral position and a three-arm technique was used (robotic camera + two working arms). Bipolar forceps were placed in the left robotic hand and monopolar scissors in the right hand. A contoured resection technique using cold scissors, following the contour of the tumour was used rather than a true enucleation. We used intraoperative US in all cases to aid in planning the site of entry into the parenchyma, to calibrate the depth of tumour penetration into the medulla, and to establish the relationship of the tumour to the renal collecting system and major vascular branches. We clamped the renal artery or arteries but not the renal vein in all cases. The assistant operated a laparoscopic 5-mm suction device to clear the field and provide gentle traction. Renal reconstruction was performed with sliding-clip renorrhaphy using a 10-mm Weck Hem-o-Lock (Teleflex, Research Triangle Park, NC, USA) and LapraTy (Ethicon, Cincinnati, OH,USA) on a 2-0 polyglactin suture to provide closing tension. In deep resections, a 3-0 polyglactin suture was run at the base of the resection bed for haemostasis and closure of the collecting system.
3D Laser Scan Process and Volume Reconstruction Process
Both the resected model tumour and the patient’s actual resected tumour were 3D laser scanned in the operating room for volumetric measurement. Bright lights were placed surrounding the sample to ensure rapid and accurate colour scanning. The resulting in silicomodel was exported as a stereolithography (.stl) file. The base structure was removed from the resulting 3D model (Autodesk, San Rafael, CA, USA), leaving only the sample. This process generated models with accuracy within ±0.25 mm. All scans were then reprocessed and each pair (specimen scan from 3D-kidney model and patient) was digitally overlaid to visualise the concordance in mass contour.
Statistical Analysis
Resection times and tumour volumes are reported as means (±SD). P values were calculated using a paired t-test or ANOVA and a P < 0.05 was considered statistically significant. Statistical analysis was performed using R version 3.2.3 (https://www.r-project.org).
Results
We generated patient specific pre-surgical renal models for 10 patients based on preoperative imaging. R.E.N.A.L. nephrometry scores were between 7 and 11; with a mean maximal tumour diameter on imaging of 40.6 mm. Pathological stages and tumour histology are shown in Table 1. There was no difference in resection times between the patient-specific pre-surgical models and the actual surgery (6:58 vs 8:22 min, P = 0.162; Table 2, Fig. 2).
Table 2.
Comparison of enucleation times in preoperative rehearsal and in vivo surgical removal
| Patient identification number |
Simulation, min |
In vivo, min |
P |
|---|---|---|---|
| All cases, mean (SD) |
6.61 (0.13) | 7.93 (0.15) |
0.162 |
| 1 | 6.4 | 5.97 | |
| 2 | 4.92 | 6.08 | |
| 3 | 9.67 | 6.13 | |
| 4 | 5.55 | 12 | |
| 5 | 5.5 | 9.92 | |
| 6 | 3.32 | 7.28 | |
| 7 | 14.02 | 15.34 | |
| 8 | 3.58 | 5 | |
| 9 | 10.32 | 10.32 | |
| 10 | 6.45 | 5.67 |
Figure 2.
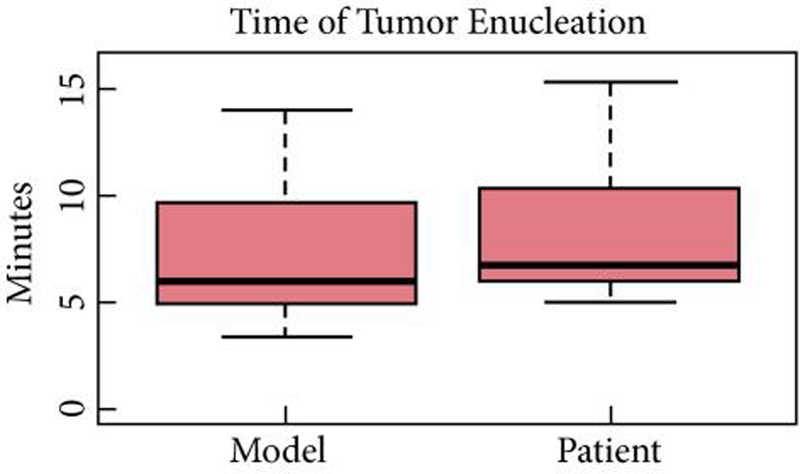
Comparison of enucleation times in preoperative rehearsal and in vivo surgical removal.
Total tumour volume was calculated from the original digital model (3D-reconstructed tumour volume). Volumes were then measured through laser scanning of the resected pre-surgical model and the resected patient specimen. There was no significant difference in volumes between the original 3D reconstruction from the CT scan, the resected pre-surgical model, and the resected tumour specimen (P = 0.98; Table 3, Fig. 3).
Table 3.
Comparison of computer-generated model volumes to enucleated model and surgical tumour volumes
| Patient identification number |
Computer tumour volume, mm3 |
Model enucleation volume, mm3 |
Tumour enucleation volume, mm3 |
P |
|---|---|---|---|---|
| All cases, mean |
38.88 | 38.5 | 41.79 | 0.976 |
| 1 | 20.4 | 21.12 | 20.31 | |
| 2 | 5.93 | 6.47 | 12.83 | |
| 3 | 25.79 | 24.94 | 18.71 | |
| 4 | 77.29 | 68 | 65.63 | |
| 5 | 9.24 | 10.31 | 4.47 | |
| 6 | 5.08 | 6.91 | 5.65 | |
| 7 | 30.36 | 36.05 | 31.21 | |
| 8 | 43.17 | 42.37 | 26.29 | |
| 9 | 60.76 | 68.9 | 106.87 | |
| 10 | 110.8 | 99.94 | 125.89 |
Figure 3.
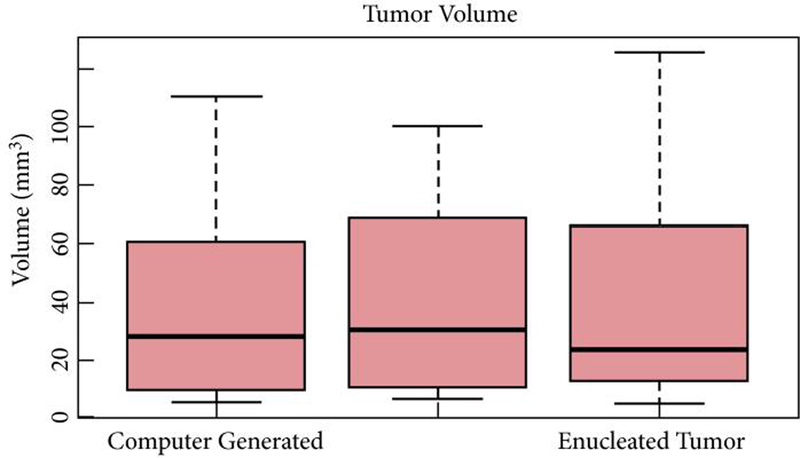
Comparison of enucleation volumes in preoperative rehearsal and in vivo surgical removal.
For a visual comparison of the scanned tumour volumes we created 3D-reconstructed overlays of the scan images of the resected model and the resected tumour (Figure 4). These images provide a more qualitative measure and document the similarity in shape between the excised model tumour and the actual patient’s specimen (accompanying video).
Figure 4.
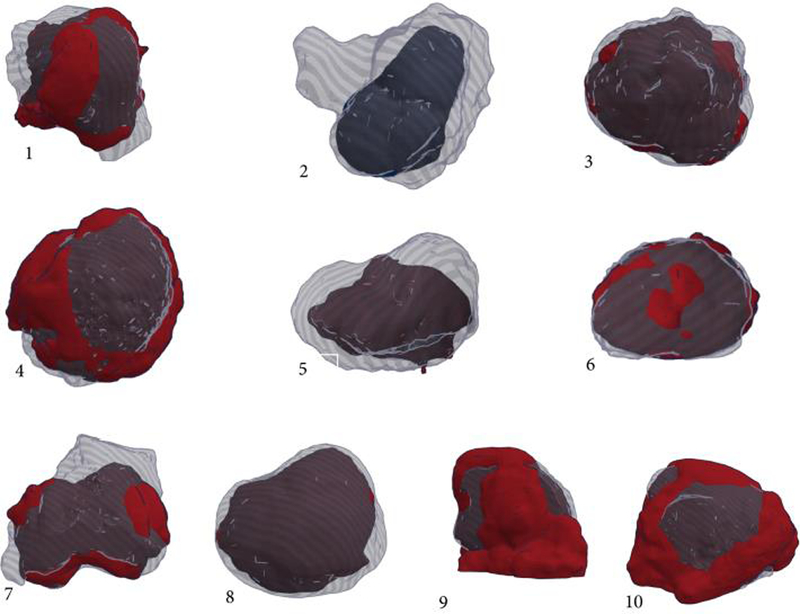
3D-reconstructed overlays of the scan images of the resected model and the resected tumor for all ten patients.
There were no positive surgical margins in the preoperative surgical models. This was evaluated by gross inspection of the kidney evaluating for residual orange silicone (tumour colour) in the resection bed. One of 10 patients had a positive margin on final pathology, with tumour only focally present at the resection margin.
Although not quantitative, side-by-side video comparisons of tumour resections in the model and the patient’s kidney were almost superimposable in time and morphology. These results confirm that a very similar sequence of manoeuvres was used to excise the same tumour in the model and the real kidney (accompanying video).
Discussion
To our knowledge, the present study is the first evaluating the construct validity of a patient-specific tissue-like renal model for pre-surgical simulation of RALPN. Here, we demonstrate striking similarity in tumour resection time, resected tumour volume and morphology for these models when compared to the patient’s actual resected tumour. These results are encouraging and suggest that this system may prove valuable for surgical planning, pre-surgical rehearsal, and RALPN training.
Operative time is a limited surrogate marker for construct validity in simulating a real surgical experience. However, in RALPN tumour excision time is a critical endpoint, as this manoeuvre is performed under warm ischaemia with the renal artery clamped. Total warm ischaemia time can significantly impact the degree of renal preservation during RALPN 9. Total ischaemia time is a combination of the time spent excising the tumour and the time invested in reconstructing the kidney defect after tumour removal. With experience, the reconstructive portion of the procedure can be done consistently rapidly. Excision time contributes the most variability to total ischaemia time, as it is dependent on tumour complexity. Our present model system allows an accurate prediction of tumour excision time, which can be valuable in predicting the feasibility of RALPN within an acceptable total ischaemia time. Moreover, this prediction is surgeon-specific, as the same surgeon resects both the model and the patient’s tumour.
3D models have been previously used in the education of patients and trainees to enhance their understanding of tumour anatomy 8, 10. Our present study extends this theme by generating patient-specific soft-tissue models that provide not only a 3D representation of tumour anatomy but also allow tumour excision to be performed on the model with high fidelity. Although difficult to quantify, we found that performing these surgical rehearsals significantly altered our approach to the actual tumour in several cases. In particular, unanticipated difficulty enucleating the deep margin in the model prompted us to alter our entry point into the renal parenchyma in several patients to provide better exposure. Even in this pilot study, we perceive that pre-surgical rehearsal on the model significantly improved our resection strategy in the corresponding patient in complex cases.
The application of laser scanning to quantify volume and provide overlay comparisons of morphology is a novel method to assess construct validity. It should be clearly stated that there are limitations to this approach. Tumour resection in the patient kidney can include overlying perinephric fat, which can increase the scanned tumour excised tissue volume as compared to the model that does not have perinephric fat. On the contrary, the loss of blood flow to the tumour after excision could potentially decrease the objective volume of the excised specimen as compared to the silicone model. Moreover, the tumour volume of the actual pathological specimen may vary, as it is placed on the scanning platform without any suspension. This can compress the lower portion of the specimen especially in softer tumours, as it sits flatly on the surface. We tried to adjust for that by orienting the tumour in such a way that the parenchymal side would face the scanner plate.
Finally, and particularly in endophytic tumours with similar densities as compared to adjacent parenchyma, there may be some uncertainty about the exact tumour contour during segmentation and 3D reconstruction. This may create a degree of discordance between the predicted model and the actual patient tumour with respect to volume. Despite these factors, the concordance in volume was excellent between the excised specimen and the excised model in the present pilot study.
The present series is a feasibility study and is limited by its small sample size. Simulation of surgical procedures can only address certain aspects of the operation. Tumour enucleation in a PN can be made more difficult by bleeding and a challenging exposure. Importantly, in vivo tissue may show less tensile strength, especially in tumours without a dense capsule. However, the present series is, to our knowledge, the first prospective study using 3D printing for patient-specific surgical rehearsal prior to RALPN.
Conclusion
Our model represents an innovative patient-specific platform for the simulation of RALPN, using a novel 3D printing technology. Further comparative studies are needed to evaluate the potential of 3D model-based surgery for resident training and its role in enhancing surgical performance in complex renal surgery.
Supplementary Material
Figure 1.
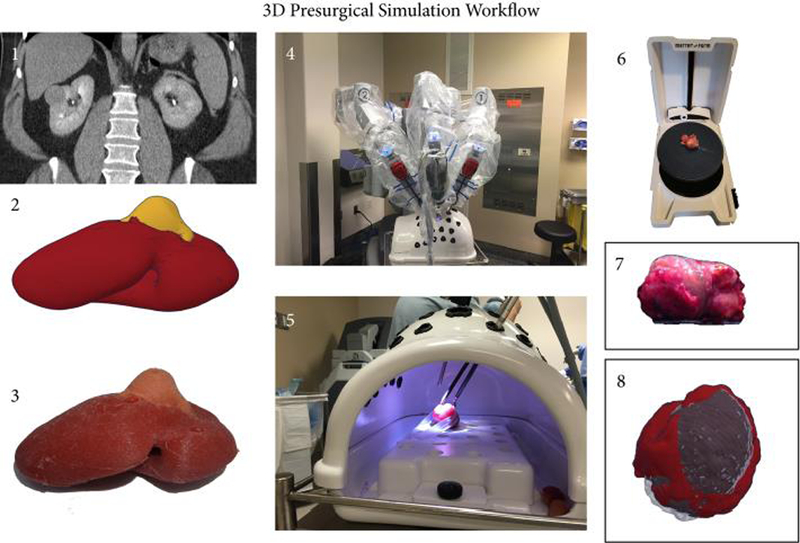
Workflow for the Presurgical Simulation. A patient specific 3D digital model is reconstructed on the basis of the available preoperative axial imaging (CT or MRI) (1-2) and then converted into a cast silicone soft tissue model as described below (3). Robotic-assisted laparoscopic enucleation of the kidney tumor is rehearsed for all patients using the 3D printed model in a three-arm configuration (4-5). Both the resected model tumor and the patient’s actual resected tumor are 3D laser scanned in the operating room for volumetric measurement (6-7). All scans are then reprocessed and each pair (specimen scan from 3D kidney model and patient) is digitally overlaid to visualize the concordance in mass contour.
Footnotes
Conflicts of Interest: Jacques Zaneveld is the president of Lazarus 3D LLC, a corporation that produces 3D printed anatomic models. All other authors report no conflict of interest.
References
- 1.Patel HD, Mullins JK, Pierorazio PM et al. Trends in renal surgery: robotic technology is associated with increased use of partial nephrectomy. J Urol 2013; 189: 1229–35 [DOI] [PubMed] [Google Scholar]
- 2.Wu Z, Li M, Liu B et al. Robotic versus open partial nephrectomy: a systematic review and meta-analysis. PLoS ONE 2014; 9: e94878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sprenkle PC, Power N, Ghoneim T et al. Comparison of open and minimally invasive partial nephrectomy for renal tumors 4–7 centimeters. Eur Urol 2012; 61: 593–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mukkamala A, Allam CL, Ellison JS et al. Tumor enucleation vs sharp excision in minimally invasive partial nephrectomy: technical benefit without impact on functional or oncologic outcomes. Urology 2014; 83: 1294–9 [DOI] [PubMed] [Google Scholar]
- 5.Kutikov A, Uzzo RG. The R.E.N.A.L. nephrometry score: a comprehensive standardized system for quantitating renal tumor size, location and depth. J Urol 2009; 182: 844–53 [DOI] [PubMed] [Google Scholar]
- 6.Mayer WA, Godoy G, Choi JM, Goh AC, Bian SX, Link RE. Higher RENAL Nephrometry Score is predictive of longer warm ischemia time and collecting system entry during laparoscopic and robotic-assisted partial nephrectomy. Urology 2012; 79: 1052–6 [DOI] [PubMed] [Google Scholar]
- 7.Silberstein JL, Maddox MM, Dorsey P, Feibus A, Thomas R, Lee BR. Physical models of renal malignancies using standard cross-sectional imaging and 3-dimensional printers: a pilot study. Urology 2014; 84: 268–72 [DOI] [PubMed] [Google Scholar]
- 8.Knoedler M, Feibus AH, Lange A et al. Individualized physical 3-dimensional kidney tumor models constructed from 3-dimensional printers result in improved trainee anatomic understanding. Urology 2015; 85: 1257–61 [DOI] [PubMed] [Google Scholar]
- 9.Volpe A, Blute ML, Ficarra V et al. Renal ischemia and function after partial nephrectomy: a collaborative review of the literature. Eur Urol 2015; 68: 61–74 [DOI] [PubMed] [Google Scholar]
- 10.Bernhard JC, Isotani S, Matsugasumi T et al. Personalized 3D printed model of kidney and tumor anatomy: a useful tool for patient education. World J Urol 2016; 34: 337–45 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


