We describe the first report of a clinical colistin-resistant ST84 Enterobacter cloacae isolate coharboring mcr-4.3 (previously named mcr-4.2) and blaNDM-1 from a patient in China. The blaNDM-1-harboring IncX3 plasmid and the novel mcr-4.3-harboring ColE plasmid were completely sequenced.
KEYWORDS: colistin, mcr-4, NDM-1, plasmid, carbapenem resistant
ABSTRACT
We describe the first report of a clinical colistin-resistant ST84 Enterobacter cloacae isolate coharboring mcr-4.3 (previously named mcr-4.2) and blaNDM-1 from a patient in China. The blaNDM-1-harboring IncX3 plasmid and the novel mcr-4.3-harboring ColE plasmid were completely sequenced. Although this isolate showed a high level of resistance to colistin, mcr-4.3 plasmid transformation, gene subcloning, susceptibility testing, and lipid A matrix-assisted laser desorption ionization mass spectrometry analysis indicated that mcr-4.3 itself does not confer resistance to colistin.
TEXT
Colistin, a cationic antimicrobial peptide, is one of the last-resort antibiotics used to treat infections caused by multidrug-resistant Gram-negative organisms (1, 2). The major mechanism for resistance to colistin is associated with chromosomal gene-mediated lipopolysaccharide (LPS) modification, e.g., transfer of the phosphoethanolamine (PEA) moiety to the suggestive 4′-phosphate position of LPS-bound lipid A (3); however, in 2016, the first report was published describing a plasmid carrying a gene (mcr-1) responsible for colistin resistance in Escherichia coli and Klebsiella pneumoniae isolates recovered from animals and patients in China (4). Since that report, >10 distinct alleles of mcr-1 have been identified in E. coli, Klebsiella, and Salmonella isolates (5, 6). In 2016, a second colistin resistance gene, mcr-2, was discovered in porcine and bovine E. coli isolates in Belgium (7), and the list now includes mcr-3, mcr-4, mcr-5, mcr-6, mcr-7, and mcr-8 isolated from Enterobacteriaceae (8–13).
Similar to the various alleles found in mcr-1, different alleles of mcr-4 have been reported. Recently, a new variant of the mcr-4 gene, mcr-4.2, was identified in two Salmonella isolates of human origin collected in 2013 in Italy (14). Compared to the prototype mcr-4.1 gene (GenBank accession no. MF543359), the mcr-4.2 gene (GenBank accession no. MG581979) contains a mutation at position 331, resulting in amino acid transition (Q→R) (Table 1) (14). A second study, published around the same time, described another mcr-4 gene variant that was also named mcr-4.2 (but later renamed mcr-4.3; see below). This mutant was described in six Enterobacter cloacae clinical isolates from Singapore that coharbored blaKPC-2 (15). Compared to the prototype mcr-4.1 gene, this mcr-4.2 gene (GenBank accession no. MG026621) contains two missense mutations at positions 179 (V→G) and 236 (V→F) (Table 1). In a recent proposal to streamline the nomenclature of mcr genes, this mcr-4.2 has been renamed mcr-4.3 (31). In addition, mcr-4.4 and mcr-4.5 gene variants were identified from E. coli isolates obtained from the feces of pigs in Spain. The mcr-4.4 gene contains missense mutations at positions 205 (H→N) and 331 (Q→R), whereas the mcr-4.5 gene harbors mutations at positions 110 (P→L) and 331 (Q→R) (17). Moreover, an mcr-4.6 gene (originally named mcr-4.3) was identified from Salmonella enterica serovar Kedougou strain 151570, and this mutant contains a missense mutation at position 236 (V→F) (Table 1) (16, 31). Here, we report the identification of another example of the mcr-4.3 gene, and in this instance, it is uniquely found with blaNDM-1 in an Enterobacter strain recovered from a patient in China.
TABLE 1.
Variants of mcr-4 gene
| mcr-4 variant | Mutation |
Accession no. | Reference | ||||
|---|---|---|---|---|---|---|---|
| P110L | V179G | H205N | V236F | Q331R | |||
| mcr-4.1 | – | – | – | – | – | MF543359 | 9 |
| mcr-4.2 | – | – | – | – | + | MG822663 | 14 |
| mcr-4.3a | – | + | – | + | – | MG026621 | 15 |
| mcr-4.4 | – | – | + | – | + | MG822665 | 17 |
| mcr-4.5 | + | – | – | – | + | MG822664 | 17 |
| mcr-4.6b | – | – | – | + | – | MH423812 | 16 |
Originally named mcr-4.2.
Originally named mcr-4.3.
A 75-year-old man was injured in a traffic accident in March 2013 and presented to a tertiary hospital in eastern China. The patient was a local farmer with no travel history for >2 years. He was diagnosed with bronchiectasis with severe aspiration pneumonia, and sputum samples grew E. cloacae. Susceptibility testing by Vitek 2 Compact (bioMérieux) showed that the E. cloacae isolate (named En_MCR4) exhibited resistance to multiple antimicrobial agents, including ceftazidime (MIC, ≥64 μg/ml), ceftriaxone (MIC, ≥64 μg/ml), cefepime (MIC, ≥64 μg/ml), imipenem (MIC, ≥16 μg/ml), meropenem (MIC, ≥16 μg/ml), aztreonam (MIC, >32 μg/ml), ciprofloxacin (MIC, >2 μg/ml), gentamicin (MIC, >8 μg/ml), tobramycin (MIC, >8 μg/ml), piperacillin-tazobactam (MIC, >64/4 μg/ml), and nitrofurantoin (MIC, ≥512 μg/ml). Additional broth microdilution testing showed that it also had high-level resistance to colistin (MIC, >256 μg/ml) (18).
To genotype the resistance mechanisms underlying the strain's multidrug resistance phenotype, whole-genome sequencing was performed using an Illumina NextSeq platform with 150-bp paired-end reads. In silico multilocus sequence typing (MLST) analysis revealed that En_MCR4 belongs to ST84 (allele profile, 60-1-61-1-36-22-1) (19), and the mining of acquired resistance genes showed that En_MCR4 harbors 10 antimicrobial resistance genes encoding resistance to β-lactams (blaNDM-1, blaCTX-M-9, and ampC), aminoglycosides [aadA2 and ant(2″)-Ia], colistin (mcr-4.3), fluoroquinolones (qnrA), phenicol (catA1), sulfonamide (sul1), and trimethoprim (dfrA16) (20).
Sequencing and characterization of the blaNDM-1- and mcr-4.3-harboring plasmids were achieved by first segregating the plasmids into an E. coli host. Conjugation experiments were performed using the E. coli J53Azr strain as a recipient as described previously (21). The transconjugants were selected on lysogeny broth (LB) agar plates with 100 μg/ml sodium azide in combination with 2 μg/ml imipenem or colistin. Multiple attempts to transfer mcr-4.3 plasmid failed; however, the blaNDM-1-bearing plasmid was successfully transferred to E. coli J53 by conjugation. Plasmid DNA was isolated from En_MCR-4, electroporated into E. coli DH10B (Invitrogen) as described previously (21), and selected on LB agar plates with 0.5, 1, and 2 μg/ml colistin. Growth was observed only on plates with colistin 0.5 μg/ml, and the transformants were screened for the presence of mcr-4.3 by PCR using primers described previously (9). Plasmid DNA from blaNDM-1-harboring E. coli J53 transconjugants and from mcr-4.3-harboring E. coli DH10B transformant was extracted using the Qiagen Plasmid Midi kit (Qiagen, Valencia, CA) and sequenced using an Illumina NextSeq system as described previously (22).
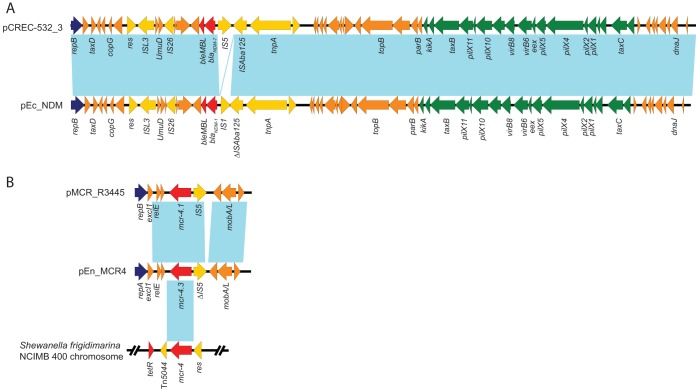
The blaNDM-1-harboring plasmid pEn_NDM is 45,739 bp in size and has an average G+C content of 46.6%. The plasmid contains 61 predicted open reading frames (ORFs) and belongs to the IncX3 incompatibility group (Fig. 1A). A BLAST search of the plasmid sequences against the GenBank database showed that pEn_NDM is highly similar to several blaNDM-harboring IncX3 plasmids, such as the blaNDM-7-harboring plasmid pCREC-532_3 from E. coli (GenBank accession no. CP024833), blaNDM-4-harboring plasmids pJEG027 from K. pneumoniae (23) and pM216_X3 from E. coli (24), and blaNDM-5-harboring plasmid pNDM5_IncX3 from K. pneumoniae (25). Plasmid pEn_NDM harbored an intact set of conjugative transfer genes to facilitate the transfer of plasmids among different members of Enterobacteriaceae, which is consistent with its ability to conjugate into E. coli J53Azr as described above (Fig. 1A).
FIG 1.
The plasmid structures. (A) Comparison of IncX3 plasmids pCREC-532_3 (CP024833) and pEn_NDM (MH061381). (B) Comparison of mcr-4-harboring plasmids pMCR_R3445 (MF543359), pEn_MCR4 (MH061380), and S. frigidimarina NCIMB 400 chromosome (CP000447). Colored arrows indicate ORFs. Dark blue, yellow, green, red, and orange arrows represent replication genes, mobile elements, plasmid transfer genes, the resistance gene, and plasmid backbone genes, respectively. Blue shading denotes regions of shared homology among different plasmids or chromosome sequences.
Plasmid pEn_MCR4 is 8,639 bp in size with a G+C content of 45.3%. It harbors eight predicted ORFs and belongs to the ColE incompatibility group (Fig. 1B). A BLAST search of all plasmid sequences against the GenBank database showed the highest 75% query coverage and 99% identity to the mcr-4.1 prototype plasmid pMCR_R3445 (GenBank accession no. MF543359) from Salmonella species (9). Unlike pMCR_R3445, plasmid pEn_MCR4 carries a different replication gene, repA, identical to plasmid pPSP-b98 (CP009870) from the Pantoea species (26). Interestingly, the mcr-4.3 gene in plasmid pEn_MCR4 showed 100% nucleotide identity with the genome of Shewanella frigidimarina NCIMB 400 (CP000447) and encoded for a putative member of phosphoethanolamine transferases (9). pEn_MCR4, pMCR_R3445, and NCIMB 400 contain conserved 59-bp mcr-4 upstream sequences, encompassing the predicted −35 (TTATTT) and −10 (AGCTAGTAT) promoter regions. In addition, the mcr-4.3 gene was identical to the mcr-4-like gene located on a 7.7-kb contig and found in six blaKPC-2-harboring E. cloacae isolates from Singapore (previously also named mcr-4.2) (Table 1) (15). Only an ∼7.7-kb mcr-4.3-harboring contig was obtained in that study; however, because the contig sequence is currently not available for comparison, it is not clear whether mcr-4.3 was carried by the same ColE plasmids as pEn_MCR4. Moreover, all six mcr-4.3-harboring E. cloacae isolates from Singapore belonged to ST54 (allele profile, 41-3-54-37-3-15-17), which is distinct from the ST84 found in our study, suggesting the horizontal transfer of mcr-4.3 into different E. cloacae strains.
The mcr-4.3-harboring E. coli DH10B transformant was subjected to susceptibility testing for colistin using the broth microdilution method according to Clinical and Laboratory Standards Institute (CLSI) guidelines (27). The transformant showed a colistin MIC of 0.5 μg/ml, which is only 2-fold higher than that of native E. coli DH10B cells (MIC, 0.25 μg/ml). Consistent with this finding, the entire coding sequence of mcr-4.3 cloned with its native promoter into plasmid pET-28a (+) in E. coli DH10B cells also had a colistin MIC of 0.5 μg/ml. Our results are in agreement with those of Teo et al. (15), which show that the mcr-4.3 gene does not confer resistance to colistin. In contrast, in the original mcr-4.1 study, plasmid pMCR_R3445 was transformed into DH5α E. coli and had an MIC of 2 μg/ml for colistin, which is an 8-fold increase compared with the MIC of the DH5α E. coli host (MIC, 0.25 μg/ml) (9).
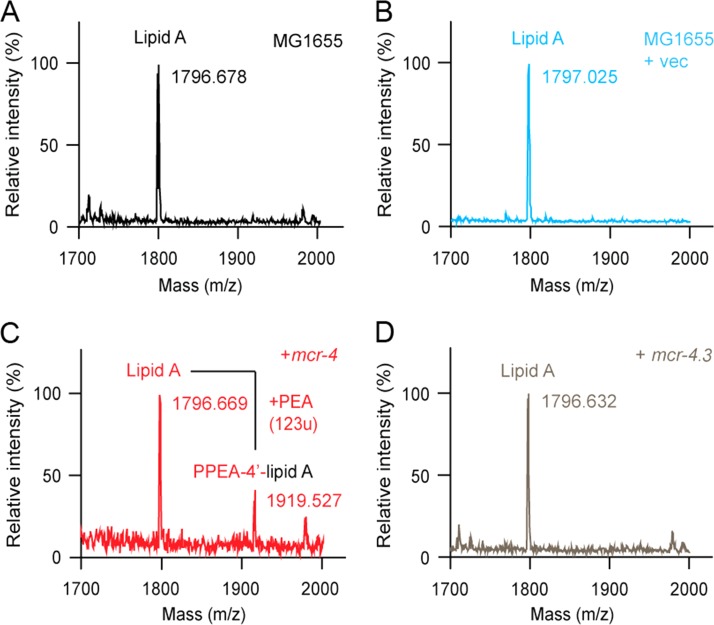
We therefore hypothesize that two missense mutations at positions 179 (V→G) and 236 (V→F) might cause a decrease in the colistin MIC in mcr-4.3-harboring strains. Subsequently, to investigate structural changes in lipid A, we applied matrix-assisted laser desorption ionization (MALDI) mass spectrometry (MS) to an array of E. coli strains with and without mcr-4 genes (Fig. 2). Briefly, the entire coding sequence of the mcr-4.3 gene from En_MCR4 and mcr-4.1 (generated by site-directed mutagenesis from mcr-4.3) was cloned into an arabinose-inducible plasmid pBAD24 and expressed into the E. coli MG1655 strain. LPS-lipid A was extracted and purified as described previously (28), and the structure of lipid A was analyzed by MALDI–time of flight mass (TOF) MS (Bruker, ultrafleXtreme) in negative-ion mode (28). As expected, a single MS spectrum of lipid A (m/z, ∼1,796.6) appears in colistin-susceptible E. coli MG1655 (Fig. 2A) and strain MG1655 carrying the empty vector pBAD24 (Fig. 2B). In contrast, the MS peak of PEA-4′-lipid A, a chemically decorated lipid A with addition of PEA, is detected in strain MG1655 harboring mcr-4.1 (Fig. 2C). Interestingly, the presence of mcr-4.3 in E. coli MG1655 fails to modify the LPS-lipid A (Fig. 2D). Collectively, we present comprehensive evidence that mcr-4.3 is a variant of mcr-4 without a lipid A modification function and, consequently, does not confer resistance to colistin.
FIG 2.
MS evidence that no addition of PEA to lipid A occurs in the mcr-4.3 E. coli MG1655 constructs. MALDI-TOF MS profile of LPS-lipid A from MG1655 alone (A) or with the empty vector pBAD24 (B). (C) MALDI-TOF MS profile of LPS-lipid A from MG1655 expressing the wild-type mcr-4.1. Two distinct MS spectra of LPS-lipid A are present. The unmodified lipid A appears at m/z 1,796.669, while the PEA-4′-lipid A (lipid A with addition of PEA) is present at m/z 1,919.527. (D) The presence of mcr-4.3 in E. coli MG1655 fails to modify the LPS-lipid A species.
Our studies indicate that the presence of mcr-4.3 cannot explain the high colistin resistance level (>256 μg/ml) observed in En_MCR4. It is known that chromosomes encoding resistance mechanisms, such as mutations in the PmrAB or PhoPQ two-component regulatory system and MgrB inactivation, are usually associated with high-level colistin resistance (29). Consequently, we mined the mgrB, phoP, phoQ, pmrA, and pmrB gene variations in En_MCR4 and compared them with 80 colistin-susceptible strains (all MICs, <1 μg/ml) from our previous Enterobacter spp. genomic study (30). No insertion, deletion, or stop codons were identified in these genes in En_MCR4; however, a number of unique (found only in En_MCR4 but not in the susceptible strains) missense mutations, such as I10V in MgrB; R2K, I4L, L5M, R69Q, I102V, and L168P in PhoQ; G21S, N72D, and Q143C in PmrA; and K91Q, T173S, N233P, L276R, and G331A in PmrB, were found. We suspect that some of these missense mutations may contribute to the colistin resistance in En_MCR4, and we are currently investigating the correlation of colistin resistance and these mutations.
In summary, this study describes the first report of an mcr-4.3-positive bacterial isolate coharboring blaNDM-1 of human origin from China. We completely characterized the blaNDM-1-harboring IncX3 plasmid and the novel mcr-4.3-harboring ColE plasmid from a clinical ST84 Enterobacter isolate. Although this isolate showed high-level resistance to colistin, mcr-4.3 does not appear to contribute to this resistant phenotype, suggesting that the two amino acid substitutions (i.e., V179G and V236F) in mcr-4.3 significantly alter the mcr-4 function. Further studies comparing additional amino acid substitutions in mcr-4 (Table 1) are necessary to understand the correlation of enzyme changes and colistin resistance and the molecular evolution of mcr-4.
Accession number(s).
The complete nucleotide plasmid sequences were deposited in GenBank as accession numbers MH061380 and MH061381. The raw whole-genome sequencing data from this study were submitted to the NCBI Sequence Read Archive (SRA) under accession no. SRR6833776.
ACKNOWLEDGMENTS
This work was supported in part by grants from the National Institute of Allergy and Infectious Diseases (R01AI090155 and R21AI135250 to B.N.K. and R21AI117338 to L.C.) and from the National Key R&D Program of China (2017YFD0500202 to Y.F.).
Y.F. is a recipient of the national “Young 1000 Talents” Award of China.
REFERENCES
- 1.Falagas ME, Rafailidis PI. 2008. Re-emergence of colistin in today's world of multidrug-resistant organisms: personal perspectives. Expert Opin Investig Drugs 17:973–981. doi: 10.1517/13543784.17.7.973. [DOI] [PubMed] [Google Scholar]
- 2.Velkov T, Thompson PE, Nation RL, Li J. 2010. Structure–activity relationships of polymyxin antibiotics. J Med Chem 53:1898–1916. doi: 10.1021/jm900999h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xu Y, Wei W, Lei S, Lin J, Srinivas S, Feng Y. 2018. An evolutionarily conserved mechanism for intrinsic and transferable polymyxin resistance. mBio 9:e02317-. doi: 10.1128/mBio.02317-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liu YY, Wang Y, Walsh TR, Yi LX, Zhang R, Spencer J, Doi Y, Tian G, Dong B, Huang X, Yu LF, Gu D, Ren H, Chen X, Lv L, He D, Zhou H, Liang Z, Liu JH, Shen J. 2016. Emergence of plasmid-mediated colistin resistance mechanism MCR-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect Dis 16:161–168. doi: 10.1016/S1473-3099(15)00424-7. [DOI] [PubMed] [Google Scholar]
- 5.Yang YQ, Li YX, Song T, Yang YX, Jiang W, Zhang AY, Guo XY, Liu BH, Wang YX, Lei CW, Xiang R, Wang HN. 2017. Colistin resistance gene mcr-1 and its variant in Escherichia coli isolates from chickens in China. Antimicrob Agents Chemother 61:e01204-. doi: 10.1128/AAC.01204-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Di Pilato V, Arena F, Tascini C, Cannatelli A, Henrici De Angelis L, Fortunato S, Giani T, Menichetti F, Rossolini GM. 2016. mcr-1.2, a new mcr variant carried on a transferable plasmid from a colistin-resistant KPC carbapenemase-producing Klebsiella pneumoniae strain of sequence type 512. Antimicrob Agents Chemother 60:5612–5615. doi: 10.1128/AAC.01075-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xavier BB, Lammens C, Ruhal R, Kumar-Singh S, Butaye P, Goossens H, Malhotra-Kumar S. 2016. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, Belgium, June 2016. Euro Surveill 21. doi: 10.2807/1560-7917.ES.2016.21.27.30280. [DOI] [PubMed] [Google Scholar]
- 8.Yin W, Li H, Shen Y, Liu Z, Wang S, Shen Z, Zhang R, Walsh TR, Shen J, Wang Y. 2017. Erratum for Yin et al. “Novel plasmid-mediated colistin resistance gene mcr-3 in Escherichia coli.” mBio 8:e01166-. doi: 10.1128/mBio.01166-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carattoli A, Villa L, Feudi C, Curcio L, Orsini S, Luppi A, Pezzotti G, Magistrali CF. 2017. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Euro Surveill 22:30589. doi: 10.2807/1560-7917.ES.2017.22.31.30589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Borowiak M, Fischer J, Hammerl JA, Hendriksen RS, Szabo I, Malorny B. 2017. Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar Paratyphi B. J Antimicrob Chemother 72:3317–3324. doi: 10.1093/jac/dkx327. [DOI] [PubMed] [Google Scholar]
- 11.AbuOun M, Stubberfield EJ, Duggett NA, Kirchner M, Dormer L, Nunez-Garcia J, Randall LP, Lemma F, Crook DW, Teale C, Smith RP, Anjum MF. 2017. mcr-1 and mcr-2 variant genes identified in Moraxella species isolated from pigs in Great Britain from 2014 to 2015. J Antimicrob Chemother 72:2745–2749. doi: 10.1093/jac/dkx286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yang YQ, Li YX, Lei CW, Zhang AY, Wang HN. 2018. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J Antimicrob Chemother. doi: 10.1093/jac/dky111. [DOI] [PubMed] [Google Scholar]
- 13.Wang X, Wang Y, Zhou Y, Li J, Yin W, Wang S, Zhang S, Shen J, Shen Z, Wang Y. 2018. Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae. Emerg Microbes Infect 7:122. doi: 10.1038/s41426-018-0124-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carretto E, Brovarone F, Nardini P, Russello G, Barbarini D, Pongolini S, Gagliotti C, Carattoli A, Sarti M. 2018. Detection of mcr-4 positive Salmonella enterica serovar Typhimurium in clinical isolates of human origin, Italy, October to November 2016. Euro Surveill 23. doi: 10.2807/1560-7917.ES.2018.23.2.17-00821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Teo JWP, Kalisvar M, Venkatachalam I, Tek NO, Lin RTP, Octavia S. 2017. mcr-3 and mcr-4 variants in carbapenemase-producing clinical Enterobacteriaceae do not confer phenotypic polymyxin resistance. J Clin Microbiol 56:e01562-. doi: 10.1128/JCM.01562-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rebelo AR, Bortolaia V, Kjeldgaard JS, Pedersen SK, Leekitcharoenphon P, Hansen IM, Guerra B, Malorny B, Borowiak M, Hammerl JA, Battisti A, Franco A, Alba P, Perrin-Guyomard A, Granier SA, De Frutos Escobar C, Malhotra-Kumar S, Villa L, Carattoli A, Hendriksen RS. 2018. Multiplex PCR for detection of plasmid-mediated colistin resistance determinants, mcr-1, mcr-2, mcr-3, mcr-4 and mcr-5 for surveillance purposes. Euro Surveill 23. doi: 10.2807/1560-7917.ES.2018.23.6.17-00672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garcia V, Garcia-Menino I, Mora A, Flament-Simon SC, Diaz-Jimenez D, Blanco JE, Alonso MP, Blanco J. 2018. Co-occurrence of mcr-1, mcr-4 and mcr-5 genes in multidrug-resistant ST10 Enterotoxigenic and Shiga toxin-producing Escherichia coli in Spain (2006-2017). Int J Antimicrob Agents 52:104–108. doi: 10.1016/j.ijantimicag.2018.03.022. [DOI] [PubMed] [Google Scholar]
- 18.Clinical and Laboratory Standards Institute. 2018. Performance standards for antimicrobial susceptibility testing. CLSI document M100-S28. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 19.Miyoshi-Akiyama T, Hayakawa K, Ohmagari N, Shimojima M, Kirikae T. 2013. Multilocus sequence typing (MLST) for characterization of Enterobacter cloacae. PLoS One 8:e66358. doi: 10.1371/journal.pone.0066358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zankari E, Hasman H, Cosentino S, Vestergaard M, Rasmussen S, Lund O, Aarestrup FM, Larsen MV. 2012. Identification of acquired antimicrobial resistance genes. J Antimicrob Chemother 67:2640–2644. doi: 10.1093/jac/dks261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen L, Chavda KD, Fraimow HS, Mediavilla JR, Melano RG, Jacobs MR, Bonomo RA, Kreiswirth BN. 2013. Complete nucleotide sequences of blaKPC-4- and blaKPC-5-harboring IncN and IncX plasmids from Klebsiella pneumoniae strains isolated in New Jersey. Antimicrob Agents Chemother 57:269–276. doi: 10.1128/AAC.01648-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen L, Hu H, Chavda KD, Zhao S, Liu R, Liang H, Zhang W, Wang X, Jacobs MR, Bonomo RA, Kreiswirth BN. 2014. Complete sequence of a KPC-producing IncN multidrug-resistant plasmid from an epidemic Escherichia coli sequence type 131 strain in China. Antimicrob Agents Chemother 58:2422–2425. doi: 10.1128/AAC.02587-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Espedido BA, Dimitrijovski B, van Hal SJ, Jensen SO. 2015. The use of whole-genome sequencing for molecular epidemiology and antimicrobial surveillance: identifying the role of IncX3 plasmids and the spread of blaNDM-4-like genes in the Enterobacteriaceae. J Clin Pathol 68:835–838. doi: 10.1136/jclinpath-2015-203044. [DOI] [PubMed] [Google Scholar]
- 24.Sugawara Y, Akeda Y, Sakamoto N, Takeuchi D, Motooka D, Nakamura S, Hagiya H, Yamamoto N, Nishi I, Yoshida H, Okada K, Zin KN, Aye MM, Tonomo K, Hamada S. 2017. Genetic characterization of blaNDM-harboring plasmids in carbapenem-resistant Escherichia coli from Myanmar. PLoS One 12:e0184720. doi: 10.1371/journal.pone.0184720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Du H, Chen L, Tang YW, Kreiswirth BN. 2016. Emergence of the mcr-1 colistin resistance gene in carbapenem-resistant Enterobacteriaceae. Lancet Infect Dis 16:287–288. doi: 10.1016/S1473-3099(16)00056-6. [DOI] [PubMed] [Google Scholar]
- 26.Conlan S, Thomas PJ, Deming C, Park M, Lau AF, Dekker JP, Snitkin ES, Clark TA, Luong K, Song Y, Tsai YC, Boitano M, Dayal J, Brooks SY, Schmidt B, Young AC, Thomas JW, Bouffard GG, Blakesley RW, Mullikin JC, Korlach J, Henderson DK, Frank KM, Palmore TN, Segre JA. 2014. Single-molecule sequencing to track plasmid diversity of hospital-associated carbapenemase-producing Enterobacteriaceae. Sci Transl Med 6:254ra126. doi: 10.1126/scitranslmed.3009845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chavda KD, Chen L, Jacobs MR, Bonomo RA, Kreiswirth BN. 2016. Molecular diversity and plasmid analysis of KPC-producing Escherichia coli. Antimicrob Agents Chemother 60:4073–4081. doi: 10.1128/AAC.00452-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xu Y, Lin J, Cui T, Srinivas S, Feng Y. 2018. Mechanistic insights into transferable polymyxin resistance among gut bacteria. J Biol Chem 293:4350–4365. doi: 10.1074/jbc.RA117.000924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wright MS, Suzuki Y, Jones MB, Marshall SH, Rudin SD, van Duin D, Kaye K, Jacobs MR, Bonomo RA, Adams MD. 2015. Genomic and transcriptomic analyses of colistin-resistant clinical isolates of Klebsiella pneumoniae reveal multiple pathways of resistance. Antimicrob Agents Chemother 59:536–543. doi: 10.1128/AAC.04037-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chavda KD, Chen L, Fouts DE, Sutton G, Brinkac L, Jenkins SG, Bonomo RA, Adams MD, Kreiswirth BN. 2016. Comprehensive genome analysis of carbapenemase-producing Enterobacter spp.: new insights into phylogeny, population structure, and resistance mechanisms. mBio 7:e02093-16. doi: 10.1128/mBio.02093-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Partridge SR, Di Pilato V, Doi Y, Feldgarden M, Haft DH, Klimke W, Kumar-Singh S, Liu J-H, Malhotra-Kumar S, Prasad A, Rossolini GM, Schwarz S, Shen J, Walsh T, Wang Y, Xavier BB. 2018. Proposal for assignment of allele numbers for mobile colistin resistance (mcr) genes. J Antimicrob Chemother doi: 10.1093/jac/dky262. [DOI] [PMC free article] [PubMed] [Google Scholar]