Abstract
Purpose
In Traditional Persian Medicine (TPM), different natural treatments have been suggested for skin damages such as Narcissus tazetta L. bulb application. New drug delivery systems such as niosomes have shown considerable increase transdermal drug delivery through stratum corneum, the main barrier against substances transport into skin. The aim of this study is preparation of niosomal formulations from N. tazetta bulb extract and evaluation of its in vitro wound healing effect.
Materials and methods
Non-ionic surfactant vesicles (NSVs or niosomes) were prepared by film hydration method from percolated extract of N. tazetta bulb. A number of 12 niosomal formulations (F1-F12) were prepared using different proportions of Span 60/Tween 60/cholesterol and 80% methanol-dissolved/aqueous PEN (percolation extract of N. tazetta) (30 and 50 mg/ml). Their morphology, particle size, physical and chemical stability and encapsulation efficiency was studied. In vitro wound healing effect of various concentrations of the best PEN niosomal formulation (F9) was evaluated in comparison to PEN on human dermal fibroblasts (HDFs).
Results
Increasing the aqueous/methanolic PEN concentration from 3 to 5% resulted size reduction of NSVs with statistically significant difference (p < 0.05). F9 showed the most physicochemical stability and was chosen for in vitro wound healing effect. This formulation exhibited significantly effects (p < 0.05) on cell proliferation in HDF cells at 1.562 and 3.125 μg/ml compared with the untreated cells using neutral red assay.
Conclusion
Formulation of PEN in niosome carrier significantly decreased the gap width on human dermal fibroblasts.
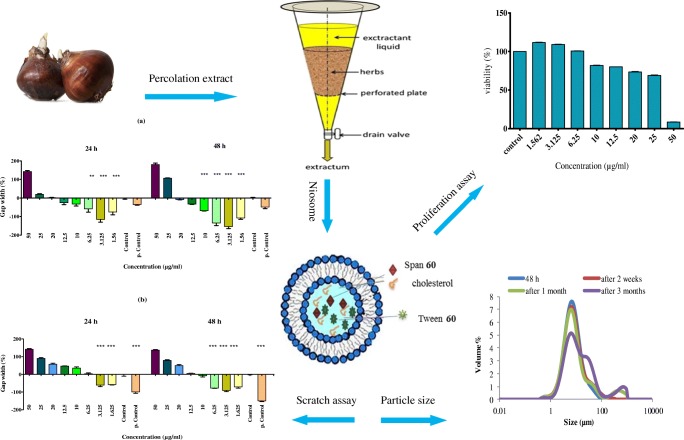
Graphical abstract.
Schematic processes of proliferation effect of narcisus tazetta bulb on fibroblast cells.
Keywords: Narcissus tazetta, Niosome, Wound healing, Human dermal fibroblast, Traditional Persian medicine
Introduction
Skin damage, like various wounds, can occur as intentional or accidental trauma. Considering the skin as the largest vital organ in the human body, skin damage can disturb its efficiency. The wound healing involves four different but overlapping phases: hemostasis, inflammation, proliferation and remodeling [1]. To enhance the transport and efficacy of many materials through stratum corneum (SC), one of various methods expanded nowadays is the usage of new drug delivery systems such as niosomal formulations. Niosomes are self-assemblies of non-ionic surfactants with or without cholesterol, which can be used for encapsulation of both hydrophilic substances such as insulin [2] or hydrophobic ones such as minoxidil [3].
Niosomes have been shown considerably increased transdermal drug delivery through SC as the main barrier against drug transport through skin. So, niosomes can be used in targeted drug delivery [4, 5] via two mechanisms: firstly, niosomes increase SC hydration and change the attributes of the SC by reducing trans epidermal water loss (TEWL). Second, niosomes increase drug transfer through the SC by acting such surfactants as the matrix and the micro scale size of niosomes [6]. Niosome can prolong the release of drug in skin damage, thereby reducing the need for dressing change during the day [4, 5, 7]. Curcumin loaded sodium hyaluronate immobilized vesicles (hyalurosomes) were used successfully in healing wounds and showed good effectiveness on skin inflammation [8]. Motaal et al. showed the superior properties of Hypericum perforatum L. niosomal gel in healing rat’s skin wound model [9].
In Traditional Persian Medicine (TPM), different natural treatments have been suggested for skin damage, which mostly contain medicinal plants. Narcissus tazetta L. bulb, from Amaryllidaceae family, has been used to treat many diseases, including wounds [10–14]. This ornamental plant has white and yellow flowers, with either uniform or contrasting colored sepals and corona. Flowering time is from January to April [15]. In general, although Amaryllidaceae plants are often tropical or subtropical, N. tazetta occurs firstly in the Mediterranean region and Southwestern Europe, with a center of variety in the Iberian Peninsula (Spain and Portugal). Few species expand the range into southern France and Italy, and even the Balkans (N. poeticus, N. serotinus and N. tazetta) and the Eastern Mediterranean (N. serotinus). N. tazetta occurs as far east as Kashmir and Iran [16]. Narcissus bulb mostly contained alkaloids like galantamine, [17] lycorine [18] and flavonoids such as rutin, quercetin and kaempferol, phenolic acids [19] and a glycoprotein called lectin [20].
N. tazetta is named “Nargis” in Persian and “Narjes” and “Abhar” in TPM [13, 21]. According to several sources of TPM, N. tazetta bulb has different therapeutic properties such as anthelmintic, uterus cleaning and wound healing. Regarding TPM, N. tazetta bulb is hot and dry in third, strong solvent, absorbent, purifier, astringent, improves the internal and external ulcers. The form of Zaroor has been used to stop the topical bleeding and wound healing. In TPM, Zaroor is one of dosage forms that mean: the fine powder of dried plants sprinkled on an organ for treatment [10, 11, 13]. A combination of bulb powder with honey is recommended for burns, nerve injury and internal ulcers and removes skin stain, especially with vinegar [12, 14]. In TCM (Traditional Chinese Medicine), traditional medicine of Turkish and Jordan, the Narcissus bulb is suggested for healing ulcer and improving inflammation [22, 23].
Since in vitro wound healing effect of Zaroor extract of N. tazetta was evaluated in pervious study [24], the aim of this study was preparation niosomal formulation, as a new drug delivery, of N. tazetta bulb and evaluation of particle size, encapsulation percentage, in vitro release study of encapsulated material, and stability of it along three months and effectiveness of this formulation on human dermal fibroblasts. To our knowledge, there is no previous report on the production and usage of niosomal formulation of N. tazetta bulb on wound healing.
Materials and methods
Materials
Sorbitan monostearate 60 (Span 60), its polyxylated derivative i.e. polysorbate 60 (Tween 60), cholesterol (Chol), isopropyl alcohol, dimethyl sulfoxide (DMSO), standard rutin were purchased from Merck, Germany, aluminum chloride and Sodium nitrite were in analytical grade. Normal saline was purchased from Exir Pharmaceutical Company. Primary human dermal fibroblasts (HDF) were purchased from Afzalipour Medicine School of Kerman University of Medical Sciences (KMU). Penicillin-G, streptomycin and amphotericin B (bioser) and phosphate buffered saline (PBS) were supplied by Sigma–Aldrich. Dulbecco’s modified eagle’s medium (DMEM) and fetal bovine serum (FBS) were used of grade Gibco (Germany). The other chemicals and solvents were in analytical grade, and bought from Merck, Germany.
Plant materials
Fresh bulbs of N. tazetta L. (Amaryllidaceae) were purchased from the local market in Kerman (Iran) in December of 2015. For authentication, the bulbs of N. tazetta were planted in December in the Botanic Garden of School of Pharmacy, Kerman, Iran. The plant was authenticated by Dr. Mirtadzadini, after flowering in March, 2016 and a voucher specimen was deposited at the Herbarium Center of Faculty of Pharmacy, Kerman University of Medical Sciences (voucher specimen no. KF1624).
Extraction method
The fresh bulbs were skinned, sliced and dried in the shade at room temperature then grinded and passed through a sieve (mesh 60). Extraction of powder of plant was carried out with methanol 80% by percolation method and dried by rotary evaporator [25]. The extraction stopped when the percolate had no reaction with alkaloid and polyphenol reagents.
Determination of Total flavonoids
Procedure with limited modifications
An aliquot (1 ml) of extract or standard solution of rutin (10–200 μg/ml) with 1 ml of distilled water was added to test tubes. After that 100 μl of nitrite sodium 5% were added and shaked for 6 min. Two hundred μl of aluminum chloride 10% and after 5 min, 1 ml sodium hydroxide 1 mol litre −1 were added to the mixture. The solution was mixed well and the absorbance was measured against prepared reagent blank at 510 nm with ELISA reader (Biotek, USA) [26]. All quantitative experiments were performed triplicate and the results were shown as mean ± SD.
Niosomes preparation
As described previously, non-ionic surfactant vesicles (NSVs or niosomes) were prepared by film hydration method [27]. Two different methods were utilized; first method, different molar ratio of Span 60/Tween 60/cholesterol and methanolic PEN (30 and 50 mg/ml) were dissolved in chloroform in a round-bottomed flask. The organic solvent was evaporated by a rotary evaporator (Heidolf, Germany) and the trace residue of chloroform was removed by a vacuum oven for 12 h. The dried thin layer of lipids/PEN was hydrated by normal saline at 55 °C for 30 min. Second method of preparation, aqueous solution of PEN (30 and 50 mg/ml) was used for hydration of dried thin lipid film. Blank niosomes were prepared in the same way without using PEN extract. Generally, 12 niosomal formulations were prepared as presented in Table 1.
Table 1.
Different niosomal formulations prepared for PEN encapsulation
| Formulation code | ST60 35:35 | ST60 30:30 | ST60 25:25 | Chol | Amount of extract (μg/ml) | W/M | Mean volume diameter (μm) ± SD |
|---|---|---|---|---|---|---|---|
| F1 | ✓ | – | – | 30 | 3% | W | 12.06 ± 4.45 |
| F2 | – | ✓ | – | 40 | 3% | W | 8.18 ± 0.06 |
| F3 | – | – | ✓ | 50 | 3% | W | 8.37 ± 0.01 |
| F4 | ✓ | – | – | 30 | 3% | M | 7.51 ± 0.14 |
| F5 | – | ✓ | – | 40 | 3% | M | 21.82 ± 10.98 |
| F6 | – | – | ✓ | 50 | 3% | M | 14.25 ± 0.16 |
| F7 | ✓ | – | – | 30 | 5% | W | 5.71 ± 0.2 |
| F8 | – | ✓ | – | 40 | 5% | W | 8.01 ± 0.11 |
| F9 | – | – | ✓ | 50 | 5% | W | 7.52 ± 0.03 |
| F10 | ✓ | – | – | 30 | 5% | M | 8.31 ± 0.42 |
| F11 | – | ✓ | – | 40 | 5% | M | 10.4 ± 1.75 |
| F12 (LS-HC-H/M) | – | – | ✓ | 50 | 5% | M | 9.84 ± 0.35 |
S, Span; T, Tween; Chol, Cholesterol; PEN, Percolated extract of N. tazetta bulb; W, PEN in distilled water solution; M, methanolic PEN
Niosomal suspensions were maintained at room temperature for 24 h and then placed in refrigerator for further studies.
Niosome characterization
Vesicle morphology
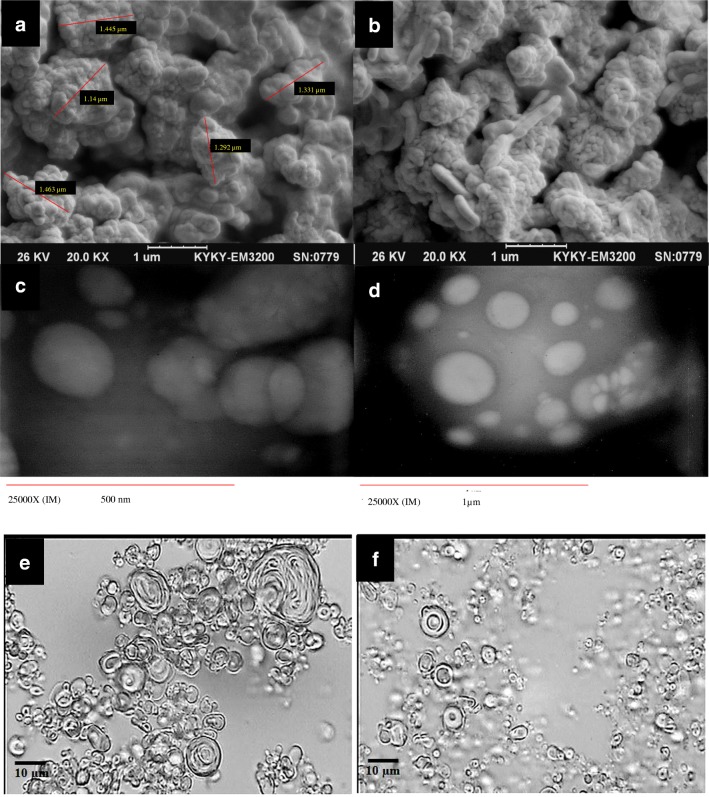
To evaluate the formation of niosomes, possible vesicular aggregation and/or separation of vesicles constituent, niosomal samples were observed by optical microscopy (Hfxdx, Nikon, Japan) equipped by a picture transferring loop and software (DinoCapture 20, Taiwan). Scanning Electron Microscopy (KYKY SEM China; EM-3200) was used to obtain some photomicrographs and Transmission Electron Microscopy (Zeiss EM 10; Germany) was also performed in freshly sonicated formulation.
Vesicle size measurement
Size analysis of non-ionic surfactant vesicles (NSVs) was conducted by using static laser light diffraction method (Malvern Instruments, Master Sizer 2000E, UK). Micromeritics parameters, including preparation of size distribution curve, mean volume diameter, (dv50) determination and other statistically derived volume diameters, were calculated by Master Sizer 2000E software.
Encapsulation efficiency measurement
For separating the entrapped and non-entrapped PEN, ultra-centrifuge (Vision/VS-35SMTi, South Korea), 59,000×g was used for 30 min at 6∘C. The total flavonoids in the supernatant and in the pellet were analyzed using ELISA reader (Biotek, USA) after disrupting niosomal lipid bilayers by isopropyl alcohol. The total flavonoids content was utilized for encapsulation efficiency calculation of niosomes using the following equation:
Where, Cp and Cs are the total flavonoids in a centrifuged pellet and supernatant, respectively.
Physicochemical stability of vesicles
The ability of niosomes to retain the drug (drug retention behavior) was studied by maintaining the niosomal formulations at three different temperatures of refrigeration temperature (4–8 °C), room temperature (25 ± 2 °C/30% RH) and 40 ± 2 °C/70% RH. Samples were stored in glass vials for 3 months and were withdrawn at regular time intervals (48 h, 15 days, 1 month and 3 months). For making sure the integrity maintenance of niosomal lipid bilayers, EE% was measured during 3 months storage. Furthermore, the size analysis was conducted as an indicator for physical stability assessment of vesicular formulations.
Cell culture and condition
Primary human dermal fibroblasts (HDFs) isolated from the adult human skin of a single donor were prepared from Kerman Afzalipour Medicine School and were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 20% fetal bovine serum (FBS), and 100 U/ml penicillin-G, 100 μg/ml streptomycin and 1 μg/ml amphotericin B and incubated at 37 °C in 5% CO2.
Fibroblast proliferation assay
HDF (approximately 1 × 104 cells) were seeded on a 96-well microtiter plate. After 24 h of incubation, the growth medium was removed and was replaced with exposure medium. Different concentrations of selected formulation (F9) were prepared in culture medium and added to each well. After 48 h, added 0.33% Neutral Red (NR) solution in an amount equal to 10% of the culture medium volume and incubated for 4 h. After incubation, the medium was carefully removed from the wells by slow aspiration, and the cells were quickly rinsed with NR Assay fixator. The fixator was removed, and the incorporated dye was dissolved in NR assay solubilization solution equal to the original volume of culture medium [28]. Empty niosomes and untreated cells were used as blank and control, respectively. Absorbance was measured at 540 nm with Eliza reader (Biotech, USA). The assay was made in triplicate.
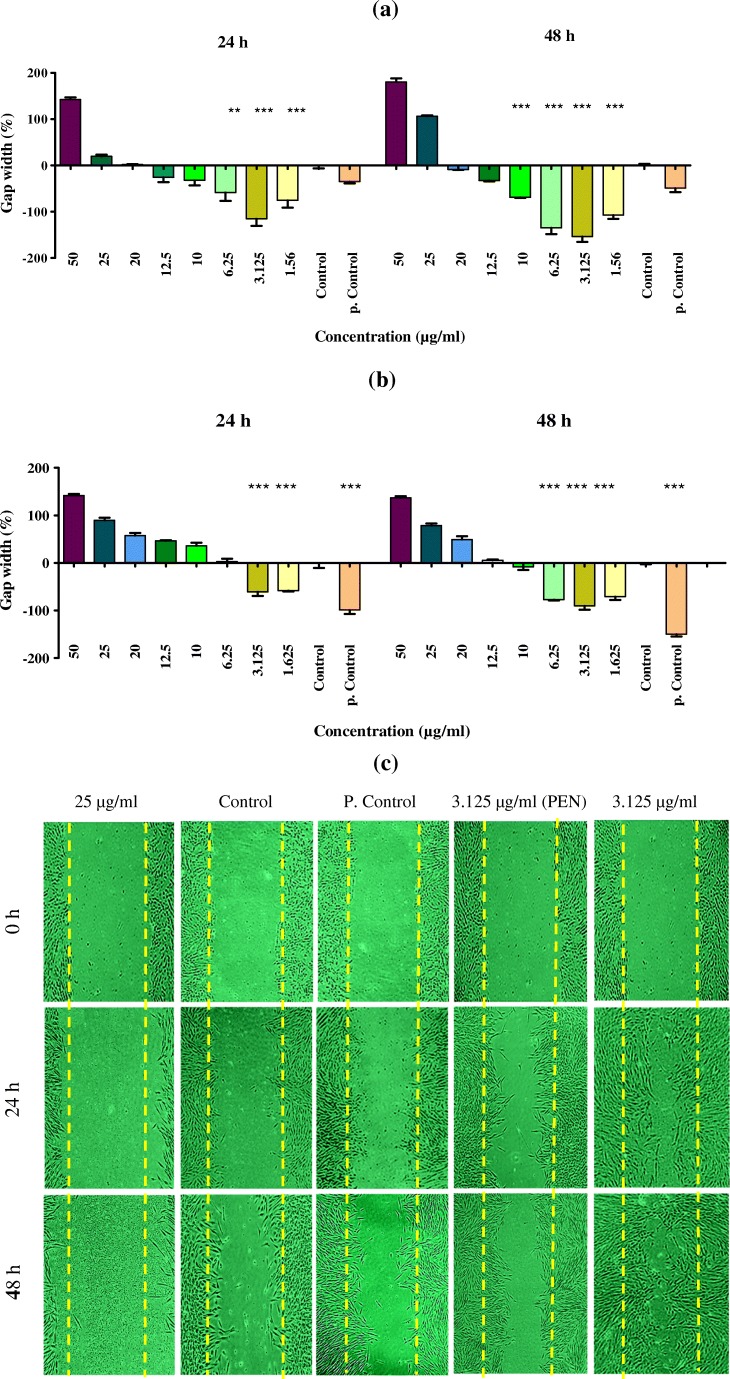
Scratch assay
The wound was created on a confluent HDF monolayer on a 6-well dish by making a horizontal and vertical scratch with a sterile 100-μL plastic pipette tip. Wound margins were photographed immediately after wounding. The cells were treated with various concentrations of PEN and its niosomal formulation (F9) in DMEM containing 5% FBS, incubated at 37 °C with 5% CO2, and the same fields of the wound margin were photographed after 24 and 48-h time points. Untreated cells were used as control, cells with FBS 10% as positive control and empty niosome as blank. The average extent of gap width was evaluated using ImageJ software [29].
Ethical considerations
The Ethics Committee of Kerman University of Medical Sciences approved this study by ethical code number: IR.KMU.REC.1394.742. Single fibroblast donor also signed the related ethical agreement form.
Statistical analysis
For continuous quantified data, one-way ANOVA was conducted with Tukey’s multiple comparison post-test using SPSS 16 software. p values <0.05 were considered for statistically significant difference.
Results
Amount of extractable compounds and flavonoid contents
The Yield of extraction was 25.78% w/w. Total flavonoid content was estimated 27.04 ± 0.93 mg rutin equivalent/g dried extract. The results calculated from regression equation of the calibration curve (A = 0.003B-0.008, R2 = 0.999) of rutin.
Niosomal characterization
Morphology and size
As shown in Fig. 1. Multilamellar vesicles (MLVs) were formed in the presence of 30, 40 and 50 M percentage of cholesterol.
Fig. 1.
Optical micrographs of 12 niosomes formulations (×400 magnification). (Scale bar: 10 μm)
Stability
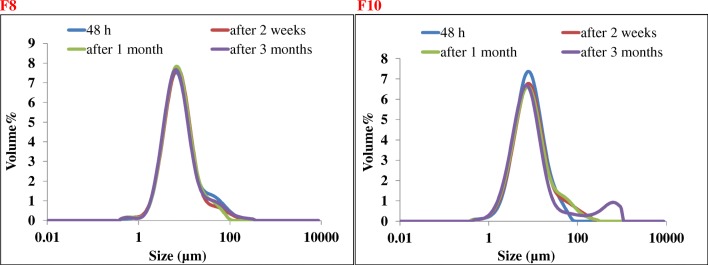
High physical stability was observed for F8 and F10 formulations that were chosen for physical stability of NSVs at refrigerator temperature (Fig. 2).
Fig. 2.
F8 and F10 formulations were chosen for physical stability of NSVs at refrigerator temperature (4–8 °C) for 3-month storage duration. High physical stability was observed for both formulations
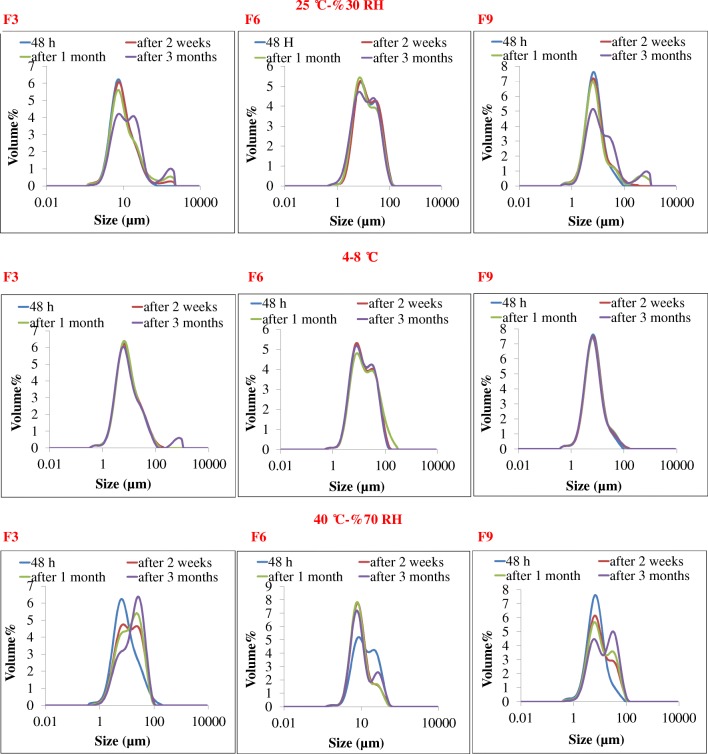
Tables 2 and 3 presented the effect of storage temperature on physical (size analysis) and chemical (EE%) stability of selected formulations (F3, F6 and F9).
Table 2.
The mean volume diameter of vesicles in 2 formulations (F8 & F10) at different time intervals after preparation and storage at 4–8 °C and the mean of volume diameter of vesicles in 3 formulations (F3, F6, F9) at different time and temperature intervals after preparation
| Formulation code | Mean volume diameter (μm) ± SD/4–8 °C | Mean volume diameter (μm) ± SD/25 ± 2 °C-30% RH | Mean volume diameter (μm) ± SD/40 ± °C-70% RH | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 48 h | 15 days | 1 month | 3 months | 48 h | 15 days | 1 month | 3 months | 48 h | 15 days | 1 month | 3 months | |
| F3 | 8.37 ± 0.01 | 8.48 ± 0.11 | 8.41 ± 0.05 | 8.87 ± 0.49 | 8.37 ± 0.01 | 9.06 ± 0.14 | 9.81 ± 0.28 | 15.28 ± 0.60 | 8.37 ± 0.01 | 12.74 ± 0.20 | 14.09 ± 0.26 | 19.16 ± 0.14 |
| F6 | 14.25 ± 0.16 | 14.43 ± 0.47 | 15.83 ± 1.00 | 14.31 ± 0.46 | 14.25 ± 0.16 | 14.07 ± 0.36 | 12.49 ± 0.46 | 12.52 ± 0.20 | 14.25 ± 0.16 | 8.63 ± 0.21 | 8.36 ± 0.14 | 8.85 ± 0.14 |
| F8 | 8.01 ± 0.11 | 7.90 ± 0.06 | 7.88 ± 0.28 | 7.63 ± 0.06 | – | – | – | – | – | – | – | – |
| F9 | 7.52 ± 0.03 | 7.57 ± 0.15 | 7.55 ± 0.06 | 7.27 ± 0.14 | 7.52 ± 0.03 | 7.81 ± 0.08 | 7.68 ± 0.12 | 12.02 ± 0.59 | 7.52 ± 0.03 | 9.33 ± 0.32 | 10 ± 0.28 | 14.80 ± 0.83 |
| F10 | 8.31 ± 0.42 | 8.87 ± 0.0.01 | 8.76 ± 0.17 | 8.36 ± 0.27 | – | – | – | – | – | – | – | – |
| Empty niosome ST60-(S:25, T:25, Chol:50) | 8.4 ± 0.4 | 9.00 ± 0.19 | 8.88 ± 006 | 8.38 ± 0.31 | 8.4 ± 0.4 | 8.86 ± 0.23 | 9.36 ± 0.8 | 7.67 ± 0.31 | 8.4 ± 0.4 | 8.65 ± 0.07 | 8.80 ± 0.02 | 7.65 ± 0.33 |
Table 3.
Comparison the encapsulation efficiency (%) of various formulations (F3, F6, and F9) in refrigerator, room temperature and 40 °C
| Formulation code | Encapsulation efficiency (%) ± SD/4–8 °C | Encapsulation efficiency (%) ± SD/25 ± 2 °C-30% RH | Encapsulation efficiency (%) ± SD/40 ± °C-70% RH | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 48 h | 15 days | 1 month | 3 month | 48 h | 15 days | 1 month | 3 month | 48 h | 15 days | 1 month | 3 month | |
| F3 | 80.2 ± 0.4 | 76.9 ± 2.4 | 74.4 ± 1.1 | 70.4 ± 1.7 | 80.2 ± 0.4 | 75.2 ± 1.7 | 70.5 ± 1.0 | 64.8 ± 1.2 | 80.2 ± 0.4 | 71.6 ± 1.0 | 63.2 ± 16.5 | 52.1 ± 0.7 |
| F6 | 70.1 ± 6.6 | 65.8 ± 1.7 | 59.8 ± 4.1 | 58.9 ± 5.7 | 70.1 ± 6.6 | 60.2 ± 11.8 | 58.4 ± 4.8 | 56.3 ± 3.2 | 70.1 ± 6.6 | 63.5 ± 0.2 | 62.8 ± 3.0 | 55.7 ± 1.7 |
| F9 | 88.8 ± 1.1 | 86.2 ± 2.7 | 83.2 ± 1.2 | 75.7 ± 3.1 | 88.8 ± 1.1 | 81.8 ± 3.4 | 75.2 ± 1.2 | 68.1 ± 1.6 | 88.8 ± 1.1 | 76.4 ± 4.6 | 68.5 ± 1.1 | 54.8 ± 1.0 |
Encapsulation efficiency percent (EE %)
As shown in Table 3, in the presence of 50% cholesterol, aqueous PEN (F3) showed more EE% (p < 0.05) than methanolic one (F6), and also increasing aqueous PEN concentration from 3 (F3) to 5% (F9) led to more EE%. F9 showed the most physicochemical stability characterizations depicted as the least volume diameter and encapsulation efficiencies changes as indicated in Fig. 3, Tables 2 and 3. So, F9 was selected for next step of this study. SEM photo of F9, showed the smooth surface of niosome formed in Fig. 4a and b and c, d, e & f showed the multilamilar vesicles of F9.
Fig. 3.
Comparison of physical stability of selected formulations including F3, F6 and F9 in refrigerator, room temperature and 40 °C. F9 showed the most physical stability characterizations depicted as the least volume diameter changes
Fig. 4.
a and b SEM photomicrograph images, c and d TEM photomicrograph images, e and f Morphological micrographs (×400 magnification) of F9 formulation
Fibroblast proliferation and scratch assay
As shown in Fig. 5, F9 at 1.562 and 3.125 μg/ml significantly (p < 0.05) increased cell proliferation compared with the control (untreated cells) while decreased the proliferation in concentrations higher than 10 μg/ml (p < 0.05).
Fig. 5.
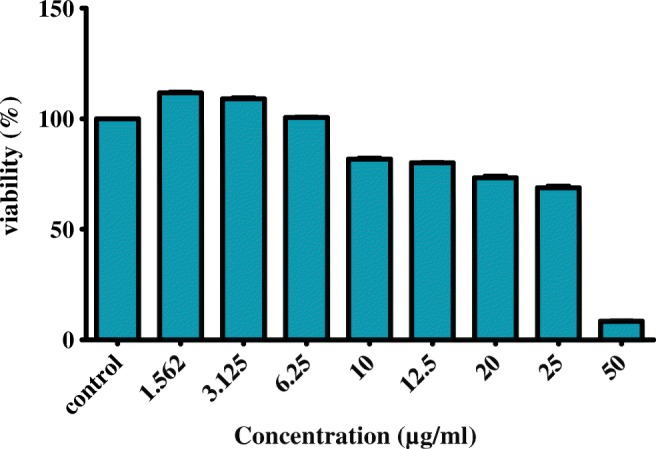
Proliferating effect of different concentrations of F9 (the best niosomal formulation of PEN) on human dermal fibroblasts (HDF) using Neutral Red assay. 1 × 104 (HDF) were seeded in 96 wells plate. After incubation in 37 °C and 5% CO2 for 24 h, treated with various concentrations of F9 and untreated cells used as control. Proliferation effect was determined using Neutral Red assay after 48 h and absorbance was measured at 540 nm with Eliza reader. The value from the baseline control group was set at 100%. (***) significantly increased cell proliferation (p < 0.05). The results were expressed as mean ± S.D.
In all, the niosomal formulation of PEN showed more strong effects on gap width compared to the free PEN extract (Fig. 6a, b and c).
Fig. 6.
Gap widths of scratch of different concentrations of F9 (a) and PEN (b) were analyzed with ImageJ software in comparison of untreated cells as the control and cells with FBS 10% as the positive control. (***) significantly reduced the gap width compared to the control group (p < 0.05). c Comparison of wound healing effect of F9, PEN, control and positive control, using scratch assay on human dermal fibroblast (HDF)
Discussion
There are many reports using Span/Tween/Chol composition for niosome preparation [30, 31]. Due to C18 hydrocarbon chain length of Span/Tween 60, multilamellar vesicles (MLVs) were formed in the presence of 30, 40 and 50 M percentag of cholesterol. Increment the amount of cholesterol from 30 to 40 M percent often resulted in larger niosomes and maybe increased in the number of lipid bilayers [32]. An increase in the aqueous PEN concentration from 3 to 5% leads to significant size reduction of NSVs in prepared ST60/Chol niosomes (p < 0.05). The same result was observed for methanolic PEN in the presence of 40 and 50 mol percentage of cholesterol. It has been reported that tretinoin-loaded niosomes and liposomes were smaller than empty lipid vesicles [33]. On the other hand, larger niosomes were obtained following incorporation of bacterial antigens into lipid vesicles due to possible influence of encapsulated material on vesicle fusion or packing arrangements of the surfactants [34].
Incorporation of PEGylated surfactants such as polysorbates (Tweens) in niosomal formulations alters lipid bilayer elasticity, [35] vesicular stability [36] and pharmacokinetic of encapsulated active pharmaceutical ingredients [37]. F8 and F10 formulations were selected for physical stability of NSVs at refrigerator temperature (4–8 °C) for 3-months storage duration. High physical stability was observed for both formulations depicted as negligible changes in size distribution curves. This could be attributed to steric stabilization of lipid vesicles containing PEGylated surfactants [38].
The proper amount of cholesterol for achieving the most stable lipid vesicular formulations is different and is related to their composition, entrapped compound and method of preparation. For example, Briuglia et al. [39] reported that phospholipid/cholesterol molar ratio of 70:30 can form the most stable liposomal formulations. On the other hand, Span 60 tail group orientation of the niosome bilayers with 50% cholesterol exhibits less tilt as well as an increase in the hydrogen bond number between Span 60 and water, which means more stable lipid bilayers [32]. Our recent study also showed incorporation of cholesterol led to more stable lipid bilayers formed by Span 80/Tween 80 niosomes [40]. F3, F6 and F9 formulations were chosen to evaluation the effect of storage temperature on physical (size analysis) and chemical (encapsulation efficiency) stability because, the presence of 50 mol% of cholesterol stabilized lipid bilayers, according to the mentioned studies.
Entrapment of different both hydrophilic and hydrophobic compounds in lipid vesicles is one of the specific characterizations of vesicular systems turning these types of new drug delivery systems into vast use and administration. We used aqueous and methanolic PEN (3 and 5%) for niosomal encapsulation and in the presence of 50% cholesterol, aqueous PEN (F3) was encapsulated more than methanolic one (F6) with significant difference (p < 0.05). This finding could be related to more hydrogen bonds formation between hydrophilic materials in aqueous PEN and bilayer components in the presence of 50% cholesterol [32] and PEGylated surfactants [31]. Increasing the concentration of both lipophilic compounds such as lacidipine [41] or alpha-tocopherol [42] and hydrophilic ones, for example, metformin [43] resulted in more entrapment of an active agent. Similar result was observed in present study, increasing aqueous PEN concentration from 3% F3 to 5% F9 led to more EE%.
In the final step of this study, we compared the effect of PEN and F9 on gap width and proliferation of fibroblast cells. In the previous study, PEN had no significant effect on proliferation of fibroblast cells [24]. Results of Kern study [44] revealed that cosmetic creams containing processed N. tazetta bulb extract caused cell proliferation reduction and cell growth inhibition of human fibroblasts and keratinocytes in primary cultures. Another study showed that Chamomile had no effect on fibroblasts proliferation [45] in spite of Chamomile application as an anti-inflammatory drug in hemorrhoids, gastrointestinal ulcers, oral ulcers and acne [46]. Narcissus bulb has also relatively good antibacterial, anti-inflammatory [47], antiviral [16], and anti-oxidant effects [44]. That seems these factors might be responsible for its effect on the ulcer healing. On the other hand, F9 at 1.562 and 3.125 μg/ml significantly (p < 0.05) increased cell proliferation compared with the control. Results of scratch assay indicated that after 24 h F9 at 1.562, 3.126, and 6.25 μg/ml concentrations and after 48 h, at 1.562 to 10 μg/ml concentrations reduced the gap width which was significantly different from control (p < 0.05). PEN after 24 h at 1.562, 3.125 μg/ml concentrations and after 48 h at 1.562 to 6.25 μg/ml concentrations significantly (p < 0.05) decreased the gap width compared to the control. The results of the effectiveness of PEN on gap width were approximately agreement with the results of the previous study in this field [24]. In comparison, F9 caused significant reduction in gap width to the PEN. However, both of PEN and F9 caused cell death in higher concentrations (especially at 25 and 50 μg/ml). Plant bulbs contain a variety of phytochemicals such as alkaloids, flavonoids, saponins and tannins, which may be responsible for inhibitory effects on cell proliferation [48, 49].
We found that its entering into the niosome carrier significantly increased the activity of the N. tazetta. Rabia Nur Un., et al. compared the effect of the extract and niosomal formulation of Calendula officinalis flowers, on external wound healing and reported the similar results. The encapsulation of plant extracts into niosomal carriers is a way to enhance bioavailability, to protect them from environmental conditions, to cover their pungent taste and to enable targeted delivery [50]. For example, niosomal formulation of diclofenac enhanced the permeation capability of drug through rat skin [51]. AbouGhaly [52] showed targeted delivery might be achieved using topically applied methotrexate niosomes for treatment of psoriasis. The healing duration in injured guinea pig skin for niosomal gel formulations containing phenytoin sodium was also less than placebo [53].
New formulations of drug delivery such as lipid vesicles play an important role in increasing the bioavailability of water-soluble plant compounds, such as flavonoids, tannins, glycosides, aglycones, etc. which is poorly absorbed and due to the large molecular size and poor lipid solubility, their ability to transfer from biological membranes is severely restricted. Thus, with using the new drug delivery technology, desirable results such as increase in bioavailability and solubility, and generally pharmacokinetic and therapeutic indices, as well as reducing the side effects of natural active compounds, can be achieved and improved their treatment performance [50, 54, 55].
The results of this study and the previous study confirmed the effect of Narcissus on gap width. Although niosomes may be suitable carriers for the PEN due to the simplicity of the production method, animal studies should be carried out to evaluate the toxicity and pharmacokinetic properties of the PEN. In this regard, clinical studies are also recommended to evaluate its effect on wound healing.
Conclusion
The in vitro wound healing effect results showed that the effectiveness of PEN was significantly increased, when entrapped in niosomal carrier and would be a good option for medicinal and cosmetic uses.
Compliance with ethical standards
Conflict of interest
The authors declare no conflict of interest.
Contributor Information
Maryam Rameshk, Phone: +983433122023, Email: mrameshk77@gmail.com.
Fariba Sharififar, Phone: +983431325001, Email: fa.sharififar@gmail.com.
Mitra Mehrabani, Phone: +983431325001, Email: mmehrabani@hotmail.com.
Abbas Pardakhty, Phone: +983431325001, Email: abpardakhty@kmu.ac.ir.
Alireza Farsinejad, Phone: +983431325374, Email: farsinezhad239@yahoo.com.
Mehrnaz Mehrabani, Phone: +983432263855, Email: mehrnaz.mehrabani@yahoo.com.
References
- 1.Hess Ct. Clinical guide to skin and wound care. Philadelphia: Lippincott Williams & Wilkins; 2012.
- 2.Pardakhty A, Moazeni E, Varshosaz J, Hajhashemi V, Rouholamini Najafabadi A. Pharmacokinetic study of niosome-loaded insulin in diabetic rats. Daru J Pharm Sci. 2011;19(6):404–411. [PMC free article] [PubMed] [Google Scholar]
- 3.Pardakhty A. Pharmaceutical sciences: breakthrough in research and practice. United States of American: IGI Global; 2016. Non-ionic surfactant vesicles (Niosomes) as new drug delivery systems; pp. 154–183. [Google Scholar]
- 4.Makeshwar KB, Wasankar SR. Niosome: a novel drug delivery system. Asian J Pharm Res. 2013;3(1):16–20. [Google Scholar]
- 5.Hamishehkar H, Rahimpour Y, Kouhsoltani M. Niosomes as a propitious carrier for topical drug delivery. Expert Opin Drug Deliv. 2013;10(2):261–272. doi: 10.1517/17425247.2013.746310. [DOI] [PubMed] [Google Scholar]
- 6.Zhang Y, Zhang K, Wu Z, Guo T, Ye B, Lu M, Zhao J, Zhu C, Feng N. Evaluation of transdermal salidroside delivery using niosomes via in vitro cellular uptake. Int J Pharm. 2015;478(1):138–146. doi: 10.1016/j.ijpharm.2014.11.018. [DOI] [PubMed] [Google Scholar]
- 7.Aqil F, Munagala R, Jeyabalan J, Vadhanam MV. Bioavailability of phytochemicals and its enhancement by drug delivery systems. Cancer Lett. 2013;334(1):133–141. doi: 10.1016/j.canlet.2013.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Manca ML, Castangia I, Marco Z, Nacher A, Valenti D, Fernandez-Busquets X, Fadda AM, Manconi M. Development of curcumin loaded sodium hyaluronate immobilized vesicles (hyalurosomes) and their potential on skin inflammation and wound restoring. Biomaterials. 2015;71:100–109. doi: 10.1016/j.biomaterials.2015.08.034. [DOI] [PubMed] [Google Scholar]
- 9.Motaal AAA, Ali M, El-Gazayerly ON. An in vivo study of Hypericum perforatum in topical drug delivery systems. Planta Med. 2016;82(S01):934. [DOI] [PMC free article] [PubMed]
- 10.Azam Khan M. Exir azam (persian). Tehran: Institute of medical history, research institute for Islamic and complementary Medicine; 2008.
- 11.Razi MEZ. Al-Havi fil Tibb. Beirut: Dar Ehia Al-Turath al-‘Arabi; 2001.
- 12.Jorjani SI. Zakhire Kharazmshahi. Qom: Institute for natural medicine recovery; 2012.
- 13.Tonekaboni Mazandarani MM. Tohfat al momenin. Qom: Institute for Natural Medicine recovery; 2011.
- 14.Avicenna (Ibn Sina). the persian translation of Al-Qanun fi al-Tibb (The canon of medicine). Translated from Arabic to Persian by Abdurrahman Sharaf-kandi. Tehran, Iran: Soroush publication; 1983.
- 15.Mazhari N. Flora of Iran. Iran: research institute of forests and rangelands; 2004.
- 16.Hanks GR. Narcissus and Daffodil: the genus narcissus. United States: CRC Press; 2003.
- 17.Wang X, Dubois R, Adams D., Jr Alzheimer's disease prevention and use of traditional plant medicines. Lett Drug Des Discov. 2015;12(2):140–151. doi: 10.2174/1570180811666140819223819. [DOI] [Google Scholar]
- 18.McNulty J, Nair JJ, Bastida J, Pandey S, Griffin C. Structure-activity studies on the lycorine pharmacophore: a potent inducer of apoptosis in human leukemia cells. Phytochemistry. 2009;70(7):913–919. doi: 10.1016/j.phytochem.2009.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lubbe A, Gude H, Verpoorte R, Choi YH. Seasonal accumulation of major alkaloids in organs of pharmaceutical crop Narcissus Carlton. Phytochemistry. 2013;88:43–53. doi: 10.1016/j.phytochem.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 20.Ooi LS, Ng TB, Geng Y, Ooi VE. Lectins from bulbs of the Chinese daffodil Narcissus tazetta (family Amaryllidaceae) Biochem Cell Biol. 2000;78(4):463–468. doi: 10.1139/o00-052. [DOI] [PubMed] [Google Scholar]
- 21.Aghili Shirazi M. Makhzan-al-advia Tehran: Tehran university of medical sciences; 2009.
- 22.Gürdal B, Kültür Ş. An ethnobotanical study of medicinal plants in Marmaris (Muğla, Turkey) J Ethnopharmacol. 2013;146(1):113–126. doi: 10.1016/j.jep.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 23.Talib WH, Mahasneh AM. Antimicrobial, cytotoxicity and phytochemical screening of Jordanian plants used in traditional medicine. Molecules. 2010;15(3):1811–1824. doi: 10.3390/molecules15031811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rameshk M, Sharififar F, Mehrabani M, Pardakhty A, Farsinejad A. In vitro proliferation and wound healing effects of Narcissus tazetta L. bulb on primary human dermal fibroblasts. J Pharm Res Int. 2018;20(6):1–11. doi: 10.9734/JPRI/2017/39090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.pharmacopoeia EcoIh. Iranian herbal pharmacopoeia. Tehran: Ministry of health and medical education/food and drug administration; 2002.
- 26.Ansari M, Sharififar F, Kazemipour M, Sarhadinejad Z, Mahdavi H. Teucrium polium L. extract adsorbed on zinc oxide nanoparticles as a fortified sunscreen. Int J Pharm Investig. 2013;3(4):188–193. doi: 10.4103/2230-973X.121289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pardakhty A, Shakibaie M, Daneshvar H, Khamesipour A, Mohammadi-Khorsand T, Forootanfar H. Preparation and evaluation of niosomes containing autoclaved Leishmania major: a preliminary study. J Microencapsul. 2012;29(3):219–224. doi: 10.3109/02652048.2011.642016. [DOI] [PubMed] [Google Scholar]
- 28.Borenfreund E, Babich H, Martin-Alguacil N. Comparisons of two in vitro cytotoxicity assays—the neutral red (NR) and tetrazolium MTT tests. Toxicol in Vitro. 1988;2(1):1–6. doi: 10.1016/0887-2333(88)90030-6. [DOI] [PubMed] [Google Scholar]
- 29.Dinda M, Mazumdar S, Das S, Ganguly D, Dasgupta UB, Dutta A, Jana K, Karmakar P. The water fraction of Calendula officinalis hydroethanol extract stimulates in vitro and in vivo proliferation of dermal fibroblasts in wound healing. Phytother Res. 2016;30(10):1696–1707. doi: 10.1002/ptr.5678. [DOI] [PubMed] [Google Scholar]
- 30.Junyaprasert VB, Singhsa P, Suksiriworapong J, Chantasart D. Physicochemical properties and skin permeation of span 60/tween 60 niosomes of ellagic acid. Int J Pharm. 2012;423(2):303–311. doi: 10.1016/j.ijpharm.2011.11.032. [DOI] [PubMed] [Google Scholar]
- 31.Varshosaz Jaleh, Taymouri Somayeh, Pardakhty Abbas, Asadi-Shekaari Majid, Babaee Abodolreza. Niosomes of Ascorbic Acid and α-Tocopherol in the Cerebral Ischemia-Reperfusion Model in Male Rats. BioMed Research International. 2014;2014:1–9. doi: 10.1155/2014/816103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ritwiset A, Krongsuk S, Johns JR. Molecular structure and dynamical properties of niosome bilayers with and without cholesterol incorporation: a molecular dynamics simulation study. Appl Surf Sci. 2016;380:23–31. doi: 10.1016/j.apsusc.2016.02.092. [DOI] [Google Scholar]
- 33.Manconi M, Sinico C, Valenti D, Loy G, Fadda AM. Niosomes as carriers for tretinoin. I. Preparation and properties. Int J Pharm. 2002;234(1–2):237–248. doi: 10.1016/S0378-5173(01)00971-1. [DOI] [PubMed] [Google Scholar]
- 34.Vangala A, Kirby D, Rosenkrands I, Agger EM, Andersen P, Perrie Y. A comparative study of cationic liposome and niosome-based adjuvant systems for protein subunit vaccines: characterisation, environmental scanning electron microscopy and immunisation studies in mice. J Pharm Pharmacol. 2006;58(6):787–799. doi: 10.1211/jpp.58.6.0009. [DOI] [PubMed] [Google Scholar]
- 35.Shalini M, Ali MH, Lakshmi P. Formulation and evaluation of elastic niosomes of eletriptan hydrobromide. Int J Pharm Sci Res. 2016;7(4):1679. [Google Scholar]
- 36.Moazeni E, Gilani K, Sotoudegan F, Pardakhty A, Najafabadi AR, Ghalandari R, Fazeli MR, Jamalifar H. Formulation and in vitro evaluation of ciprofloxacin containing niosomes for pulmonary delivery. J Microencapsul. 2010;27(7):618–627. doi: 10.3109/02652048.2010.506579. [DOI] [PubMed] [Google Scholar]
- 37.Shehata T, Kimura T, Higaki K, Ogawara K-i. In-vivo disposition characteristics of PEG niosome and its interaction with serum proteins. Int J Pharm. 2016;512(1):322–328. doi: 10.1016/j.ijpharm.2016.08.058. [DOI] [PubMed] [Google Scholar]
- 38.Lin T, Fang Q, Peng D, Huang X, Zhu T, Luo Q, Zhou K, Chen W. PEGylated non-ionic surfactant vesicles as drug delivery systems for Gambogenic acid. Drug Deliv. 2013;20(7):277–284. doi: 10.3109/10717544.2013.836618. [DOI] [PubMed] [Google Scholar]
- 39.Briuglia M-L, Rotella C, McFarlane A, Lamprou DA. Influence of cholesterol on liposome stability and on in vitro drug release. Drug Deliv Transl Res. 2015;5(3):231–242. doi: 10.1007/s13346-015-0220-8. [DOI] [PubMed] [Google Scholar]
- 40.Nematollahi MH, Pardakhty A, Torkzadeh-Mahanai M, Mehrabani M, Asadikaram G. Changes in physical and chemical properties of niosome membrane induced by cholesterol: a promising approach for niosome bilayer intervention. RSC Adv. 2017;7(78):49463–49472. doi: 10.1039/C7RA07834J. [DOI] [Google Scholar]
- 41.Qumbar M, Imam SS, Ali J, Ahmad J, Ali A. Formulation and optimization of lacidipine loaded niosomal gel for transdermal delivery: in-vitro characterization and in-vivo activity. Biomed Pharmacother. 2017;93:255–266. doi: 10.1016/j.biopha.2017.06.043. [DOI] [PubMed] [Google Scholar]
- 42.Basiri L, Rajabzadeh G, Bostan A. α-Tocopherol-loaded niosome prepared by heating method and its release behavior. Food Chem. 2017;221:620–628. doi: 10.1016/j.foodchem.2016.11.129. [DOI] [PubMed] [Google Scholar]
- 43.Samed Noufel, Sharma Varsha, Sundaramurthy Anandhakumar. Hydrogen bonded niosomes for encapsulation and release of hydrophilic and hydrophobic anti-diabetic drugs: An efficient system for oral anti-diabetic formulation. Applied Surface Science. 2018;449:567–573. doi: 10.1016/j.apsusc.2017.11.055. [DOI] [Google Scholar]
- 44.Kern D, Method to treat skin conditions with narcissus tazetta bulb extract. US patent App. 12/019163, 2008; 1–8.
- 45.Wyganowska-Swiatkowska M, Urbaniak P, Szkaradkiewicz A, Jankun J, Kotwicka M. Effects of chlorhexidine, essential oils and herbal medicines (Salvia, chamomile, Calendula) on human fibroblast in vitro. Cent Eur J Immunol. 2016;41(2):125. doi: 10.5114/ceji.2016.59739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Singh O, Khanam Z, Misra N, Srivastava MK. Chamomile (Matricaria chamomilla L.): an overview. Pharmacogn Rev. 2011;5(9):82–95. doi: 10.4103/0973-7847.79103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bouchareb D, Bouraoui NK, Fernandez X, Michel T. Phytochemical characterization of Narcissus tazetta and evaluation of its bioactive potent. Planta Med. 2016;81(S 01):P59. [Google Scholar]
- 48.Arabski M, Węgierek-Ciuk A, Czerwonka G, Lankoff A, Kaca W. Effects of Saponins against clinical E. coli strains and eukaryotic cell line. J Biomed Biotechnol. 2012;2012:286216. doi: 10.1155/2012/286216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kang TH, Pae HO, Yoo JC, Kim NY, Kim YC, Ko GI, Chung HT. Antiproliferative effects of alkaloids from Sedum sarmentosum on murine and human hepatoma cell lines. J Ethnopharmacol. 2000;70(2):177–182. doi: 10.1016/S0378-8741(99)00164-6. [DOI] [PubMed] [Google Scholar]
- 50.Un RN, Barlas FB, Yavuz M, Ag Seleci D, Seleci M, Gumus ZP, Guler E, Demir B, Can M, Coskunol H. Phyto-niosomes: in vitro assessment of the novel nanovesicles containing marigold extract. Int J Polym Mater. 2015;64(17):927–937. doi: 10.1080/00914037.2015.1030663. [DOI] [Google Scholar]
- 51.Ioele G, Tavano L, De Luca M, Ragno G, Picci N, Muzzalupo R. Photostability and ex-vivo permeation studies on diclofenac in topical niosomal formulations. Int J Pharm. 2015;494(1):490–497. doi: 10.1016/j.ijpharm.2015.08.053. [DOI] [PubMed] [Google Scholar]
- 52.Abdelbary AA, AbouGhaly MHH. Design and optimization of topical methotrexate loaded niosomes for enhanced management of psoriasis: application of box–Behnken design, in-vitro evaluation and in-vivo skin deposition study. Int J Pharm. 2015;485(1):235–243. doi: 10.1016/j.ijpharm.2015.03.020. [DOI] [PubMed] [Google Scholar]
- 53.Ali A, Sarhan HA, Magdy T. Preparation and characterization of phenytoin sodium niosomes for enhanced closure of skin injuries. Int J Pharm Pharm Sci. 2014;6:542–546. [Google Scholar]
- 54.Puglia C, Lauro MR, Tirendi GG, Fassari GE, Carbone C, Bonina F, Puglisi G. Modern drug delivery strategies applied to natural active compounds. Expert Opin Drug Deliv. 2017;14(6):755–768. doi: 10.1080/17425247.2017.1234452. [DOI] [PubMed] [Google Scholar]
- 55.Alexander A, Patel RJ, Saraf S, Saraf S. Recent expansion of pharmaceutical nanotechnologies and targeting strategies in the field of phytopharmaceuticals for the delivery of herbal extracts and bioactives. J Control Release. 2016;241:110–124. doi: 10.1016/j.jconrel.2016.09.017. [DOI] [PubMed] [Google Scholar]