Abstract
Background
Insight is impaired in the majority of schizophrenia patients. The exact neural correlates of impaired insight remain unclear. We assume that the ability to regulate emotions contributes to having good clinical insight, as patients should be able to regulate their emotional state in such a way that they can adapt adequately in order to cope with impaired functioning and negative stigma associated with a diagnosis of schizophrenia. Numerous studies have shown emotional dysregulation in schizophrenia. We investigated the association between insight and brain activation and connectivity during emotion regulation.
Methods
Brain activation during emotion regulation was measured with functional MRI in 30 individuals with schizophrenia. Two emotion regulation strategies were examined: cognitive reappraisal and expressive suppression. Clinical insight was measured with the Schedule for the Assessment of Insight – Expanded, and cognitive insight was measured with the Beck Cognitive Insight Scale. Whole brain random effects multiple regression analyses were conducted to assess the relation between brain activation during emotion regulation and insight. Generalized psychophysiological interaction (gPPI) was used to investigate the relation between task-related connectivity and insight.
Results
No significant associations were found between insight and neural correlates of cognitive reappraisal. For clinical insight and suppression, significant positive associations were found between symptom relabeling and activation in the left striatum, thalamus and insula, right insula and caudate, right pre- and postcentral gyrus, left superior occipital gyrus and cuneus and right middle and superior occipital gyrus and cuneus. Furthermore, reduced clinical insight was associated with more connectivity between midline medial frontal gyrus and right middle occipital gyrus. For cognitive insight and suppression, significant positive associations were found between self-reflectiveness and activation in pre- and postcentral gyrus and left middle cingulate gyrus.
Conclusions
Our results suggest an association between the capacity to relabel symptoms and activation of brain systems involved in cognitive-emotional control and visual processing of negative stimuli. Furthermore, poorer self-reflectiveness may be associated with brain systems subserving control and execution.
Keywords: Insight, Cognitive reappraisal, Expressive suppression, fMRI, gPPI, Emotion regulation
Highlights
-
•
We examined the association between emotion regulation and insight in schizophrenia.
-
•
Patients with poorer insight engage different neural pathways during suppression.
-
•
This finding was strengthened by connectivity measures of corresponding brain areas.
-
•
No relationship between cognitive reappraisal and insight was found.
1. Introduction
Clinical insight is impaired in the majority of schizophrenia patients (Dam, 2006). It includes the following dimensions: (i) illness awareness, (ii) attribution of symptoms to the illness, and (iii) awareness of need for treatment (David, 1990). Impaired clinical insight is one of the most common reasons for poor treatment adherence, and a strong association between impaired clinical insight and poorer outcome of the disorder has been shown (Lincoln et al., 2006). Clinical insight is separated from cognitive insight, which relates to patients' attributive metacognitive ability. Cognitive insight is defined as the ability to evaluate and reflect upon one's own aberrant views and interpretations (Beck et al., 2004; Cooke et al., 2010). Clinical insight requires the ability to reflect upon oneself (i.e. cognitive insight), but also the abilities to inhibit one's own perspective, to take someone else's perspective and to switch between perspectives until the perspective is found that matches reality best. Consequently, it has been suggested that social cognitive functions such as self-reflectiveness and perspective taking, as well as cognitive functions such as cognitive (inhibitory) control and cognitive flexibility (Pijnenborg et al., 2011) may play an important role in clinical insight. We assume that the ability to regulate emotions contributes to having good clinical insight, as patients should be able to regulate their emotional state in such a way that they can adapt adequately in order to cope with impaired functioning and negative stigma associated with a diagnosis of schizophrenia (Pijnenborg et al., 2011). Better emotion regulation skills may also be associated with being more open to considering the possibility of having a mental disorder. The association between emotion regulation and insight has not been studied before. Results of an earlier study suggested that stigma resistance was associated with emotion regulation in patients with schizophrenia (Raij et al., 2014). A model illustrating the relationship between several processes that may be involved in impaired insight can be seen in Fig. 1.
Fig. 1.
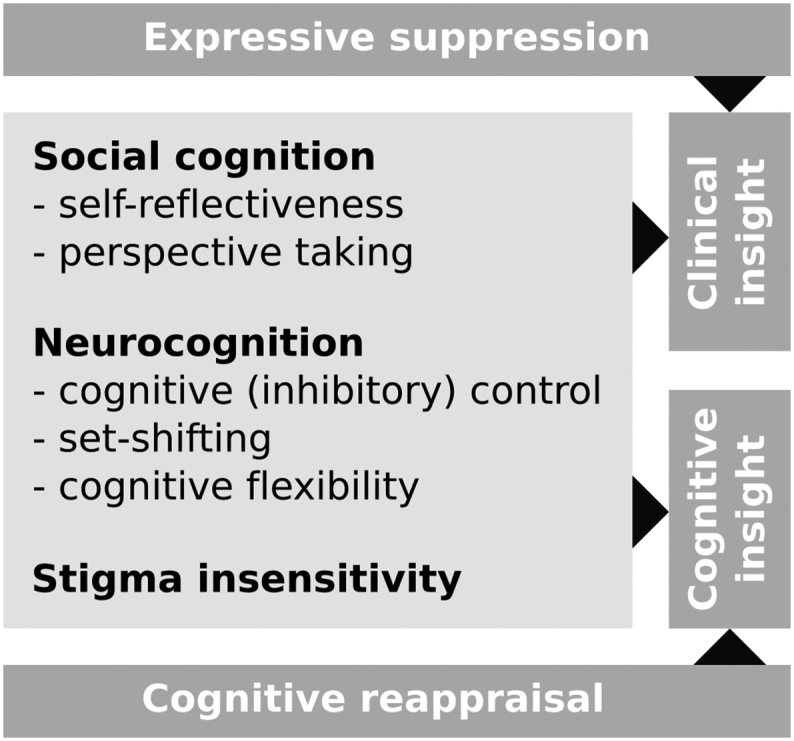
Model of impaired insight in schizophrenia. Adapted from Pijnenborg et al. (2011).
Numerous studies have shown emotional dysregulation in schizophrenia (Henry et al., 2007; Horan et al., 2013; Morris et al., 2012; Perry et al., 2012; Van der Meer et al., 2014). Emotion regulation refers to an individual's ability to manage their emotional states (Gross, 1998). Several emotion regulation strategies exist and individuals differ in their use of them (Gross, 1998). In this study, we focus on the two most-applied emotion regulation strategies, namely cognitive reappraisal and expressive suppression. Cognitive reappraisal is antecedent-focused (i.e. focused on processes that precede an emotional response) and expressive suppression is response-focused (i.e., focused on response that is already under way). Several studies have shown that schizophrenia patients use reappraisal less frequently and suppression more frequently compared to healthy individuals (Kimhy et al., 2012; Livingstone et al., 2009; van der Meer et al., 2009), while other studies did not find significant differences (Badcock et al., 2011; Henry et al., 2008; Perry et al., 2011).
During cognitive reappraisal, individuals control negative emotions by changing their way of thinking. The neural correlates of cognitive reappraisal of emotional stimuli have been investigated extensively in healthy subjects using functional neuroimaging. These studies found increased activation of the prefrontal cortex (PFC; including the dorsolateral prefrontal cortex (DLPFC), ventrolateral PFC (VLPFC), the dorsomedial PFC (DMPFC) and the posterior prefrontal cortex), inferior parietal cortex, dorsal anterior cingulate cortex (dACC) and reduced activation of the amygdala, ventral striatum, insula and ventromedial PFC (VMPFC) during reappraisal (see Diekhof et al. (2011) and Buhle et al. (2014) for meta-analyses). These studies suggest top-down control of the prefrontal cortex on areas involved in emotion processing (e.g. the amygdala). In schizophrenia, neuroimaging studies have reported hypo-activation of the VLPFC compared to healthy controls (Morris et al., 2012; Van der Meer et al., 2014). During expressive suppression, emotion expression is inhibited. Two neuroimaging studies in healthy participants found increased activation of the DLPFC, VLPFC, and insula during suppression (Goldin et al., 2008; Ohira et al., 2006). Findings of these studies on amygdala activation were mixed, with one study finding increased and the other study finding decreased activation during suppression (Goldin et al., 2008; Ohira et al., 2006). No neuroimaging studies on suppression have been conducted in schizophrenia yet.
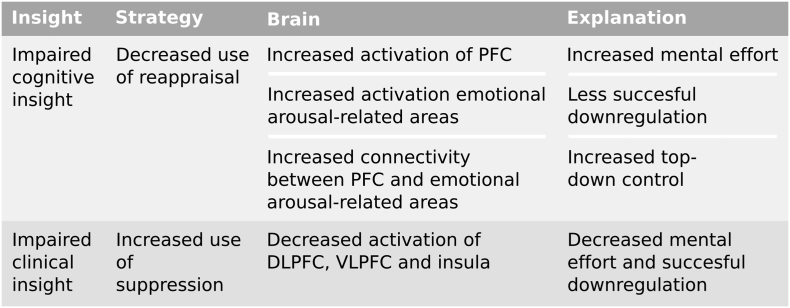
We assume that emotion regulation by means of cognitive reappraisal requires insight and awareness, as it entails conscious effort in order to initiate it and monitor emotions during its execution. Therefore, we hypothesize that patients with impaired insight are less able to regulate their negative emotions through reappraisal and will make more use of suppression. Specifically, we expect a relationship between reappraisal and cognitive insight since reappraisal is a predominantly cognitive process. We hypothesize that patients with poorer cognitive insight show increased activation of prefrontal and emotional arousal-related areas, as well as more connectivity between prefrontal and emotional arousal-related areas suggesting increased mental effort and top-down control to exert cognitive reappraisal. In addition, we hypothesize that patients with poorer clinical insight will make more use of suppression and, therefore, will show less brain activation of and connectivity between relevant areas (DLPFC, VLPF and insula) during suppression compared to patients with better insight. A visualization of our hypotheses can be seen in Fig. 2.
Fig. 2.
Visualization of hypotheses.
2. Methods
2.1. Participants
35 individuals with schizophrenia (SZ; 73% men) and 16 healthy controls (HC; 67% men) were included in this study. All patients were diagnosed with schizophrenia by a psychiatrist according to DSM-IV-TR (American Psychiatric Association, 2000) and ICD-10 criteria (World Health Organisation, 2012), which was confirmed with the Mini International Neuropsychiatric Interview (MINI-plus) (Sheehan et al., 1998). Patients were recruited from several mental health institutions in the Netherlands in a consecutive manner. All healthy controls were assessed with the MINI-plus (Sheehan et al., 1998) to confirm lack of personal history of psychiatric, somatic and neurological illnesses. They were matched to the patient group on age, handedness, sex and education. All participants were of ages 18 and above and were able to give informed consent. Exclusion criteria for this study were having an acute psychosis, having a co-morbid psychiatric, somatic and/or neurological disorder, drug use, change of medication within the last week, use of a benzodiazepine equivalent to >3 mg lorazepam, electroconvulsive therapy within the last year and MRI contra-indications (i.e. metal implants, red ink tattoos, pregnancy or possibility thereof and claustrophobia). All participants provided informed consent and received 45 euros for participation. The study protocol was approved by the medical ethical board of the University Medical Center Groningen and was in accordance with the latest version of the Declaration of Helsinki.
Five patients and one healthy individual were excluded from analyses for different reasons: lack of understanding of fMRI task (2 SZ), MRI artefact (1 SZ), lack of logging of onset times of different conditions fMRI task (1 SZ) and excessive head motion (1 SZ and 1 HC). This left a group of 30 SZ patients and 15 HC for analyses; their clinical and demographic characteristics can be seen in Table 1.
Table 1.
Clinical and demographic characteristics of all participants.
| Variable | Schizophrenia patients (mean (SD)) | Healthy controls (mean (SD)) | Significance |
|---|---|---|---|
| Age (years)a | 35.00 (10.16) (range: 20–57) | 33.60 (11.11) (range: 21–53) | F(1,43) = 0.178, p = 0.675 |
| Sex (percentage male)a | 73% | 67% | χ(1) = 0.216, p = 0.642 |
| Level of educationa,b | 5.37 (1.16) | 5.60 (0.91) | U = 202.5, p = 0.571 |
| Self-reported handedness (percentage right-handed)a | 87% | 87% | χ(1) = 0, p = 1 |
| Estimate of premorbid intelligencea,c | 98.00 (16.30) | 93.1 (10.05) | F(1,42) = 1.508, p = 0.226 |
| Age of illness onset (years) | 23.83 (7.73) | ||
| Illness duration (years)d | 11.10 (8.60) | ||
| Use of antipsychotic medicatione | |||
| None | 2 | ||
| Olanzapine | 14 | ||
| Aripiprazole | 12 | ||
| Quetiapine | 5 | ||
| Clozapine | 3 | ||
| Haloperidol | 1 | ||
| Perphenazine | 1 | ||
| Pimozide | 1 | ||
| Risperidone | 1 | ||
| PANSS | |||
| Negative symptoms | 14.27 (4.61) | ||
| Positive symptoms | 14.47 (5.47) | ||
| General psychopathology | 29.17 (7.73) | ||
| Total | 57.90 (14.71) | ||
| SAI-E | |||
| Awareness of illness | 8.84 (3.49) | ||
| Relabeling of symptoms | 3.57 (2.14) | ||
| Need for treatment | 1.70 (0.70) | ||
| Subtotal | 15.93 (7.21) | ||
| BCIS | |||
| Self-reflection | 9.83 (4.20) | ||
| Self-certainty | 15.87 (5.16) | ||
| Composite score | 6.03 (5.01) |
Abbreviations: PANSS = Positive and Negative Syndrome Scale; SAI-E = Schedule for the Assessment of Insight – Expanded; BCIS = Beck Cognitive Insight Scale.
No significant differences were found between groups in age (tested with ANOVA), sex (tested with Chi-Square test), level of education (tested with Mann-Whitney U Test), handedness (tested with Chi-Square test) and estimate of premorbid intelligence (tested with ANOVA).
According to Verhage (1964).
Measured with the Dutch Adult Reading Test (DART). DART-scores were missing for 1 healthy control.
Illness duration information was missing for 1 patient.
Some patients were using multiple antipsychotic medications.
2.2. Clinical measures
2.2.1. Clinical characteristics
Severity and frequency of last week's symptoms were assessed with the Positive and Negative Syndrome Scale (PANSS) (Kay et al., 1987), which is a semi-structured interview scored by a trained interviewer. The PANSS was used for the assessment of general psychopathology as well as positive and negative symptoms.
2.2.2. Clinical insight
Clinical insight was measured with the Schedule for the Assessment of Insight – Expanded (SAI-E) (Kemp and David, 1997). The SAI-E is a 12-item researcher-reported semi-structured interview measuring three subdimensions of insight: (1) awareness of illness, (2) relabeling of symptoms and (3) awareness of need for treatment (David, 1990). Three subscale scores can be calculated, as well as a subtotal score (items 1–9), with higher scores indicating better clinical insight. These subscales have been confirmed with factor analysis in several studies that found three similar factors (Dantas and Banzato, 2007; David et al., 2003; Konstantakopoulos et al., 2013). Subscale scores as well as SAI-E subtotal score were used for fMRI analyses.
2.2.3. Cognitive insight
Cognitive insight was measured with the Beck Cognitive Insight Scale (BCIS) (Beck et al., 2004). The BCIS is a self-report 15-item questionnaire that consists of two subscales: (1) self-reflection (SR; e.g., ‘I am sometimes wrong about how people feel and think about me’; 9 items) and (2) self-certainty (SC; e.g., ‘my interpretations of my experiences are absolutely right’; 6 items). These two factors have been confirmed by multiple studies (Buchy et al., 2012; Favrod et al., 2008; Gutiérrez-Zotes et al., 2012; Kao and Liu, 2010; Uchida et al., 2009). Two subscales scores and a composite index score (self-reflection score minus self-certainty score) were computed. All three scores were used for subsequent fMRI analyses. Poor cognitive insight is reflected by high scores on self-certainty, low scores on self-reflection and a low composite index score.
2.2.4. Emotion regulation questionnaire
Emotion regulation strategies were assessed with the Dutch version of the Emotion Regulation Questionnaire (ERQ) (Gross and John, 2003). This is a 10-item self-report questionnaire that measures the use of reappraisal and suppression as emotion regulation strategies in daily life. This questionnaire consists of 6 items measuring reappraisal (e.g. ‘I control my emotions by changing the way I think about the situation I am in’) and 4 items measuring suppression (e.g. ‘I control my emotions by not expressing them’). These subscales have been confirmed with factor analysis (Melka et al., 2011). All items were measured on a 7-point scale (strongly disagree - strongly agree). Subscale scores were calculated by dividing the total subscale score by the number of subscale items.
2.2.5. Affect
The Positive and Negative Affect Schedule (PANAS) (Watson et al., 1988) is a 20-item self-report measure assessing positive and negative affect at this moment. Responses were measured on a 5-point scale (very slightly or not at all - extremely). The PANAS has been shown to be a reliable and valid measure of affective state (Crawford and Henry, 2004).
2.3. Task and stimuli
The emotion regulation task of event-related design that is used in this study is based on the theoretical framework of Gross (Gross, 1998), and was designed and published by Ochsner et al. (2002). The task was programmed using E-Prime software version 1.0.2 (Psychology Software Tools, Pittsburg, PA, USA), run in Windows, which also recorded participants' responses. This task was validated in a previous study by our group examining healthy individuals, patients with schizophrenia and non-affected siblings (Van der Meer et al., 2014). Before scanning, all participants received training to make sure they completely understood the task and what was asked of them. They told the researcher how they would regulate their emotions during the different conditions in order to practice all different emotion regulation strategies until complete understanding of the task. The emotional pictures, extracted from the International Affective Picture System (IAPS), which were used as stimuli during training, were different from pictures used during the experiment.
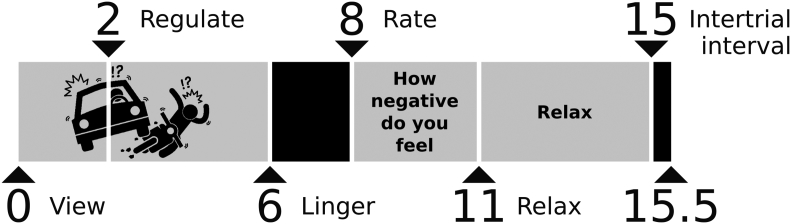
During the experiment, 88 stimuli were shown in total, of which 66 were negative (mean valence: 2.6; mean arousal: 5.7) and 22 were neutral (mean valence: 1.3, mean arousal: 1.9). Stimuli were selected in a randomized way. Each trial lasted 15.5 s and consisted of six parts: (1) view (2 s), (2) regulation (4 s), (3) lingering (2 s), (4) rating (3 s), (5) relax (4 s) and (6) intertrial interval (0.5 s) (see Fig. 3). Only the regulation part differed between trials, and could be one of five conditions: attend neutral, attend negative, reappraise, suppress and increase. During View, participants were shown the emotional picture with the instruction ‘view’ asking participants to look at the picture. After that, instructions were presented on the screen below the picture instructing the participants how to regulate their emotional reaction to the picture (Regulation: attend, reappraise, suppress or increase). Instructions were in accordance with (Ochsner et al., 2002) and (Goldin et al., 2008), but translated into Dutch. During ‘attend’ the participants were asked to just look at the picture, while the participants were asked to regulate their emotions during ‘reappraise’, ‘suppress’ and ‘increase’. During ‘reappraisal’ the participant was instructed to reinterpret the picture in a way that decreases negative feelings. The instructions for ‘suppression’ asked the participant to suppress the expressive emotional reaction provoked by the picture. They were instructed that no one should be able to read their emotions from their face. The ‘increase’ condition entailed the opposite of the ‘reappraise’ condition, so participants had to increase the negative emotions caused by the stimulus. Stimuli were shown for 6 s in total (during View and Regulation). After the emotion regulation part of this task, the participants were asked to let their emotions linger while a black screen was shown to them (Lingering). Consequently, participants had to rate how negatively they felt at that moment (Rating; score 1–4 from ‘not negative’ to ‘very negative’). At last, the word ‘relax’ was presented and participants could relax for 4 s (Relax). The intertrial interval lasted 0.5 s and consisted of a black screen to indicate start of a new trial. Each regulation condition occurred 22 times; hence, the experimental paradigm consisted of 110 trials (22 trials per condition) of 15.5 s. After every tenth trial, a rest block was included in which a fixation cross was presented for 20 s (Fixation; baseline). Total fMRI scan duration for this task was around 32 min and the task was scanned in two sessions to prevent fatigue.
Fig. 3.
Overview of task.
2.4. Image acquisition
All scans were made in the Neuroimaging Center of the University Medical Center Groningen (UMCG) in Groningen. Scans were acquired using a 3T Phillips Intera Quaser (Philips Intera, Best, the Netherlands) equipped with a synergy SENSE eight-channel head coil. Functional images were acquired using a T2*-weighted echo-planar sequence producing 37 interleaved axial slices tilted approximately 30°, a thickness of 3.5 mm and no slice gap to cover the entire cortex (TR = 2 s; TE = 30 ms; flip angle = 70°; FOV = 224, 129.5, 224; 64 × 62 matrix of 3.5 × 3.5 × 3.5 voxels). In addition, a T1-weighted 3D fast field echo (FFE) anatomical image (voxel size 1 × 1 × 1 mm) containing 170 slices (TR = 9 ms; TE = 3.54 ms; slice thickness = 1 mm; 256 × 256 matrix; FOV 232, 170, 256 mm) was acquired parallel to the bicommissural plane. Every run started with several dummy scans to ensure steady state magnetization before acquiring the data used for analyses.
2.5. Method of analysis
2.5.1. Demographic and clinical characteristics and insight
SPSS 20 (SPSS Inc., Chicago, IL, USA) was used for all behavioral data analyses. Methods and results of group comparisons (SZ vs HC) as well as correlations between insight and demographic or clinical variables are described in Supplementary Materials 1.1 and 2.1.1, respectively. Haloperidol equivalents were calculated based on antipsychotic dose (Andreasen et al., 2010). Haloperidol equivalents of pimozide were calculated by first converting to olanzapine according to (Gardner et al., 2010), followed by conversion to haloperidol according to (Andreasen et al., 2010).
2.5.2. Emotion regulation questionnaire
We calculated Pearson correlations between a priori emotion regulation strategies and insight, results of which were evaluated at an FDR-corrected level corrected for 4 tests. Methods and results of group comparisons are described in Supplementary Materials 1.2 and 2.1.2, respectively.
2.5.3. Affect
Pearson correlations between a priori (negative and positive) affect and insight were calculated. A threshold of p < 0.05, two-tailed, was used as the standard for statistical significance and all correlations between PANAS subscales and insight were evaluated at an FDR-corrected level corrected for 4 tests. Methods and results of group comparisons are described in Supplementary Materials 1.3 and 2.1.3, respectively.
2.5.4. Emotion regulation task
Pearson correlations between negative affect ratings during the task and insight were calculated. A threshold of p < 0.05, two-tailed, was used as the standard for statistical significance and all correlations between negative affect ratings and RTs (after reappraise and suppress) and (clinical and cognitive) insight were evaluated at an FDR-corrected level corrected for 8 tests. Methods and results of group comparisons are described in Supplementary Materials 1.4 and 2.1.4, respectively.
2.5.5. fMRI analyses
We analyzed fMRI-data using Statistical Parametric Mapping (SPM12 – version r6223) (www.fil.ion.ucl.ac.uk/spm) run in Matlab 8.1 (The MathWorks Inc., Natick, MA, USA). First, all images were checked for artefacts (1 patient was excluded, as mentioned in 2.1 Participants subsection). Functional as well as anatomical images were reoriented parallel to the AC-PC plane. Functional images were corrected for slice timing (reference slice: middle slice) and realigned to the first volume of the first run. The details of the transformation were checked and participants who showed >6 mm movement across the session were dropped from analyses (1 HC and 1 SZ, as mentioned in 2.1 Participants subsection). The mean functional scans created during realignment were coregistered to the anatomical scans (4th degree B-spline interpolation). Functional images were spatially normalized based on the Montreal Neurological Image (MNI) T1 template and smoothed with a 6 mm full-width half maximum (FWHM) Gaussian smoothing kernel. Smoothing was done with a 6 mm kernel given our a priori interest in the amygdala. Coregistration and normalization were checked after coregistration and/or normalization and, if necessary, adjusted manually (adjustment after coregistration, n = 31; additional adjustment after normalization, n = 21). In case of manual adjustment after normalization, functional images were resliced after this adjustment.
At first level, a general linear model (GLM) was created which included 14 condition-regressors (7 condition-regressors per session) modelled with a boxcar function convolved with a canonical hemodynamic response function (HRF): (1) view, (2) attend neutral, (3) attend negative, (4) reappraisal, (5) suppression, (6) increase, (7) after. The regressor ‘after’ included the Lingering, Rating and Relax parts of the task. Fixation crosses and intertrial intervals were seen as baseline brain activity. The six estimated motion parameters and their derivatives were added to the model to correct for motion effects. A high-pass filter cut-off of 128 s was used, and serial correlations were accounted for using an autoregressive (AR (1)) model during classical (ReML) parameter estimation. Three contrasts were defined for each participant: (1) reappraise versus attend negative, (2) suppress versus attend negative, and (3) increase versus attend negative. These contrasts examine which brain regions are activated more during emotion regulation compared to attending to a negative picture, to isolate regions that are important for these emotion regulation strategies. The increase condition was not of our interest, but activation during this condition and differences between groups in activation during this condition will be described in Supplementary Materials 2.2 and 2.3, respectively.
At second level, first, main task effects were examined in all participants with one sample t-tests using contrasts reappraise > attend negative, suppress > attend negative and increase > attend negative. Second, between group differences were examined with two sample t-tests using the same contrasts. Between group analyses were conducted to examine whether patients engaged additional or different neural resources compared to HC. Third, second-level whole brain random effects multiple regression analyses were conducted to assess the relation between brain activation during emotion regulation and insight. Only patients were included in these analyses (n = 30), and whole brain as well as region of interest (ROI) analyses were conducted. Contrasts reappraisal > attend negative and suppression > attend negative were used for further analyses, as there was no rationale for examining the association between insight and use of reappraisal to increase negative emotions. Thus, the contrast reappraisal > attend negative or suppression > attend negative was entered, with demeaned clinical insight scores (SAI-E subtotal score) or demeaned cognitive insight scores (BCIS composite index score) as covariate of interest. The same analysis was done for the subscale scores, with either the three demeaned SAI-E subscale scores as covariates of interest or the demeaned BCIS subscale scores as covariates of interest. All of these analyses were thresholded with family-wise error (FWE) cluster-level correction at p < 0.05 (using an initial threshold of p < 0.001, uncorrected). No extra correction was applied to correct for the number of contrasts tested. For the ROI random effects multiple regression analyses, masks of the IFG were made using Marsbar (Brett et al., 2002), based on group level activation during either reappraise > attend negative or suppress > attend negative. For reappraisal > attend negative, the mask included the right inferior frontal gyrus, insula and superior temporal gyrus. Two masks were made for suppression > attend negative; the first mask included the left inferior frontal gyrus and insula, while the second mask included the right inferior frontal gyrus and insula. Results of these ROI random effects multiple regression analyses were viewed with voxel-level pFWE < 0.05 and small volume correction (SVC).
In order to investigate task-related connectivity, we used generalized psychophysiological interaction version 13.1 (gPPI) (McLaren et al., 2012). One seed region of interest, with a 6 mm-radius, was defined functionally per contrast based on peak activation of the group: (1) left IFG activated during reappraisal > attend negative (MNI coordinates -52 20 12) and (2) midline MFG activated during suppression > attend negative (MNI coordinates 2 2 60). The seed ROIs were made using Marsbar (Brett et al., 2002). First, the time course representing activation in the seed region (first eigenvariate from the timeseries) was extracted from these seed regions for each subject, and neural activation in these seed regions was estimated by hemodynamic deconvolution (physiological term). The estimated neural activation in these seed regions was multiplied with task vectors and reconvolved with a canonical HRF (PPI term). The individual task vectors were also convolved with a canonical HRF (psychological term). Separate task regressors were formed for each condition, and a separate interaction term was formed for each condition. For each seed region, a first level (individual) GLM analysis was performed with the PPI terms, the task regressors (psychological term), the seed region timecourse (physiological term) and a constant. Two contrasts were created: reappraise versus attend negative and suppress versus attend negative. For each seed region, random effects multiple regression analyses with insight were computed at group level. Results were thresholded at FWE-corrected p < 0.05 at cluster-level using an initial threshold of p < 0.001 (uncorrected).
3. Results
3.1. Behavioral results
No significant correlations were found between insight and emotion regulation strategies, a priori affect, nor affect ratings and RTs during fMRI emotion regulation task after FDR-correction.
3.2. fMRI results
3.2.1. Main task effects and group differences
Results of main task effects and group difference analyses are shown in Supplementary Materials 2.2 and 2.3, respectively.
3.2.2. Activation during emotion regulation and insight
No significant associations were found between activation during reappraisal and (clinical or cognitive) insight.
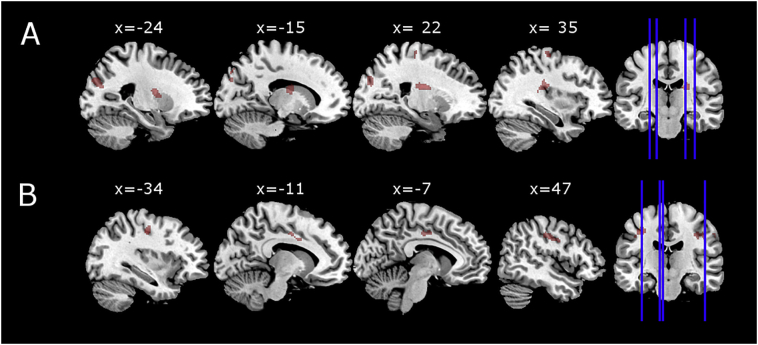
We found a positive association between scores on SAI-E Relabeling of symptoms subscale and activation during suppression > attend negative in left striatum, thalamus and insula, right insula and caudate, right pre- and postcentral gyrus, left superior occipital gyrus and cuneus, and the right middle and superior occipital gyrus and cuneus. These results can be seen in Fig. 4A and Table 2. ROI-analyses did not show additional activations that survived voxel-level FWE-correction (p < 0.05). We checked overlap between these areas and areas that were found in main task effects. We firstly made ROIs of areas that were associated with insight in Marsbar (Brett et al., 2002). Consequently, we checked overlap between these areas and main task activation by entering these ROIs with main task activation t-maps in MRIcroGL (Rorden and Brett, 2000). With FWE-correction at cluster level to correct for multiple testing, the left striatum, thalamus and insula, the right insula and caudate, and the right precentral and postcentral gyrus regions did not overlap with regions observed in the main effect of suppression > attend negative, suggesting that these regions may be important for insight but making it uncertain which role they play in expressive suppression. Most of these regions were activated during suppression when viewing results with lower statistical thresholds, however (punc < 0.01 or < 0.05).
Fig. 4.
A. Association between clinical insight (SAI-E subscale relabeling of symptoms) and brain activation during suppression.
B. Association between cognitive insight (BCIS subscale self-reflectiveness) and brain activation during suppression.
Table 2.
Associations between insight and activation or connectivity during emotion regulation.
| Hemisphere | k voxels | MNI coordinates |
Z | |||
|---|---|---|---|---|---|---|
| x | y | z | ||||
| Activation | ||||||
| Clinical insight | ||||||
| Suppression > attend negative and SAI-E relabeling of symptoms | ||||||
| Positive correlation | ||||||
| Caudate, putamen, thalamus, insula | L | 211 | −26 | 0 | 10 | 4.61 |
| −24 | −6 | 16 | 4.14 | |||
| −16 | −8 | 22 | 4.04 | |||
| Insula, caudate | R | 251 | 22 | −16 | 20 | 4.59 |
| 36 | −24 | 24 | 3.91 | |||
| 32 | −32 | 20 | 3.87 | |||
| Precentral gyrus, postcentral gyrus | R | 162 | 28 | −32 | 68 | 4.12 |
| 34 | −20 | 62 | 4.02 | |||
| 20 | −24 | 64 | 3.63 | |||
| Superior occipital gyrus, cuneus | L | 228 | −14 | −82 | 42 | 4.11 |
| −22 | −82 | 30 | 4.09 | |||
| −28 | −72 | 18 | 3.67 | |||
| Middle occipital gyrus, superior occipital gyrus, cuneus | R | 209 | 28 | −76 | 30 | 3.83 |
| 14 | −78 | 28 | 3.74 | |||
| 20 | −84 | 26 | 3.52 | |||
| Cognitive insight | ||||||
| Suppression > attend negative and BCIS self-reflection | ||||||
| Positive correlation | ||||||
| Precentral gyrus, postcentral gyrus | L | 113 | −34 | −18 | 44 | 4.85 |
| −46 | −12 | 38 | 3.49 | |||
| Middle cingulate gyrus | L | 121 | −12 | −6 | 40 | 4.38 |
| −10 | 2 | 34 | 3.73 | |||
| −6 | −14 | 40 | 3.68 | |||
| Precentral gyrus, postcentral gyrus | R | 224 | 46 | −16 | 36 | 4.32 |
| 36 | −18 | 40 | 4.09 | |||
| 48 | −4 | 32 | 4.05 | |||
| Connectivity | ||||||
| Clinical insight | ||||||
| Suppression > attend negative and SAI-E subtotal (seed: midline medial frontal gyrus) | ||||||
| Negative correlation | ||||||
| Middle occipital gyrus (lingual gyrus, fusiform gyrus) | R | 104 | 30 | −80 | 0 | 4.16 |
| 28 | −68 | 2 | 3.73 | |||
| 26 | −86 | −8 | 3.45 | |||
All results of regression analyses are shown with an initial threshold of p < 0.001 (uncorrected) and cluster-level FWE-corrected p < 0.05.
Abbreviations: L = left; R = right.
Whole-brain regression analyses with cognitive insight showed significant positive correlations between scores on SR and activation during suppression > attend negative in bilateral pre- and postcentral gyrus, and the left middle cingulate gyrus. These results can be seen in Fig. 4B and Table 2. ROI-analyses did not show additional activations that survived FWE-correction at voxel-level (p < 0.05).
3.2.3. Connectivity during emotion regulation and insight
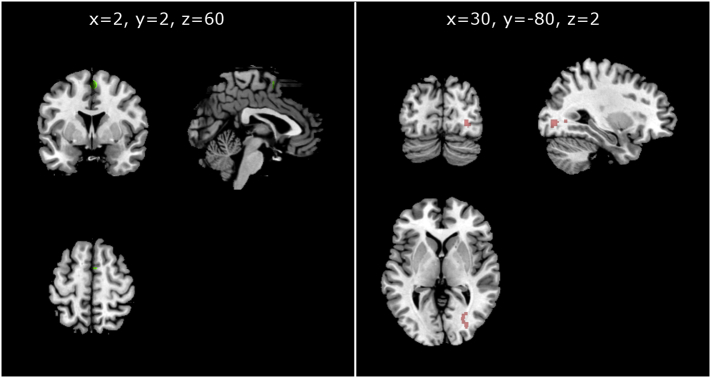
Whole brain gPPI analyses for suppression > attend negative showed more connectivity between midline medial frontal gyrus and the right middle occipital gyrus (lingual gyrus and fusiform gyrus) in patients with lower SAI-E subtotal scores. This result can be seen in Fig. 5 and Table 2. No other associations with (clinical or cognitive) insight survived correction for multiple testing at cluster-level (pFWE < 0.05).
Fig. 5.
Association between clinical insight (SAI-E subtotal scores) and brain connectivity during suppression. Left: seed region in the midline medial frontal gyrus. Right: association between clinical insight (SAI-E subtotal scores) and connectivity with seed region during suppression.
4. Discussion
The aim of this study was to examine the relationship between insight and brain activation as well as connectivity during emotion regulation in schizophrenia. For reappraisal, we did not find any significant associations between activation or connectivity and insight. A possible explanation for this could be that this study was conducted in a structured laboratory setting which does not resemble real life, as patients were explicitly cued and instructed to reappraise. This could result in patients being abler to regulate their emotions compared to real life, since in more complex social settings, cognitive control and working memory may be more challenged, especially in patients with impaired insight. For expressive suppression, we hypothesized that patients with poorer clinical insight would show less brain activation of relevant areas (i.e. DLPFC, VLPC and insula) during expressive suppression. We indeed found that patients with poorer ability to relabel symptoms showed less brain activation in the left striatum, thalamus and insula and the right insula and caudate during expressive suppression. In addition, they showed less brain activation in areas involved in visual processing of negative stimuli (i.e. left superior occipital gyrus and cuneus and right middle and superior occipital gyrus and cuneus). The latter is strengthened by the finding that connectivity between the midline medial frontal gyrus, an important area for execution of emotion regulation (Kohn et al., 2014; Vanderhasselt et al., 2013), and the right middle occipital gyrus, showed to be increased in patients with poorer clinical insight. Our results suggest that patients with poorer ability to relabel symptoms engage different neural pathways during expressive suppression, which are implicated in cognitive-emotional control and visual processing of negative stimuli, implying a role of emotion regulation in clinical insight. Decreased activation of the insula during suppression might imply decreased self-focus, as individuals with poorer ability to relabel symptoms might direct their attention less inward to monitor their expressions (Hayes et al., 2010; Richards and Gross, 2000). In addition, less activation of areas involved in visual processing of negative stimuli could indicate that patients with poorer ability to relabel symptoms implicitly reduce processing of emotion-evoking aspects of negative stimuli during expressive suppression. Other studies showed attentional shifts and reduced viewing of negative stimuli during emotion regulation (Dillon et al., 2007; Gross, 1998), even without instruction to do so (Bebko et al., 2011; Hayes et al., 2010; van Reekum et al., 2007). Reduction of negative affect by use of expressive suppression appeared to be as successful in patients with poorer insight, as we did not find a significant correlation between (clinical or cognitive) insight and negative affect rating after suppression.
Other studies also found associations between clinical insight and brain activation of areas that were shown to be involved in expressive suppression in our study. Sapara and colleagues, for example, found that patients with poorer insight (based on Birchwood Insight Scale scores) showed less activation in the left putamen, extending to the caudate, insula and IFG, compared to patients with preserved insight during a self-monitoring task (Sapara et al., 2015). They noted that these areas are known to be involved in self-monitoring, specifically in the appraisal and attribution of self-generated stimuli (Kumari et al., 2010; McGuire et al., 1996; Shergill et al., 2001). In addition, an earlier study of our group found an association between poorer clinical insight (lower SAI-E subtotal scores) and less activation in the left anterior insula, among other areas, during a self-reflection task (van der Meer et al., 2013). Less insula activation was interpreted as weaker emotional and interoceptive response (Modinos et al., 2011). It was suggested that insula activation may result from an emotional response evoked by self-reflection (Fossati et al., 2003), which may fail to occur in patients with impaired insight in whom these self-reflective processes are hampered to begin with. Other studies have also suggested that the representation of interoceptive information mediated by the insula plays an important role in higher order processes such as self-awareness and insight (Palaniyappan et al., 2011). Lastly, Shad and colleagues found a correlation between unawareness of symptoms (as measured with the Scale to Assess Unawareness of Mental Disorder) and activation in the left frontal inferior operculum (including parts of the insula, caudate and putamen) and left lingual gyrus, as well as between misattribution of symptoms (SUMD) and activation of the left frontal inferior triangle (including parts of the insula), right putamen and left lingual gyrus during a self-awareness task (Shad and Keshavan, 2015). They noted that several studies have shown involvement of the basal ganglia in integrative and cognitive processes influencing not only sensory-motor control, but also different types of cognitive and limbic affective functions (Middleton and Strick, 2000), which underlie complex and integrative processes such as self-awareness, introspective perspective of one's own self and consciousness (Kircher and Leube, 2003). However, it is difficult to disentangle the neural substrate of impaired insight, as underlying (social) cognitive and emotional processes (and their neural substrates) are related, in addition to schizophrenia being a heterogeneous disorder.
With regard to cognitive insight, we found that patients with poorer self-reflectiveness abilities had lower activation of brain systems subserving control and execution of emotion regulation (i.e. left and right pre- and postcentral gyrus and the left middle cingulate gyrus) during suppression. Few fMRI studies have studied the association between brain activation during a task and cognitive insight, and associations between higher self-reflection abilities or higher cognitive insight (composite index scores) and higher activation of prefrontal areas (VMPFC, VLPFC, DLPFC) has been found most frequently (Buchy et al., 2015; Ćurčić-Blake et al., 2015; Lee et al., 2015; van der Meer et al., 2013). No associations between cognitive insight and activation in the pre- and postcentral gyrus and middle cingulate gyrus have been found before. The midcingulate cortex (MCC) has been suggested to be involved in cognitive control and intentional motor control and selection (Hoffstaedter et al., 2014), while the pre- and postcentral gyri have been associated with movement but also with motor imagery and task execution (Stippich et al., 2002). Kohn et al. (2014) also showed coactivation of the MCC with several emotion regulation areas such as the insula, VLPFC, SMA and thalamus suggesting a role of this area in emotion regulation (Kohn et al., 2014). Our results could suggest less control and execution of suppression in patients with poorer self-reflectiveness. These results are in line with our suggestion that patients with poorer insight may implicitly reduce processing of emotion-evoking aspects of negative stimuli during expressive suppression and are therefore less involved in expressive suppression. Following this line of thought – the avoidance of emotion-evoking aspects of negative stimuli – is in line with the psychological denial model, which is one of several models attempting to explain the etiology of insight. The psychological denial model suggests that poor insight is caused by the use of denial as a coping strategy in order to reduce distress caused by stigma associated with diagnosis of schizophrenia (Cooke et al., 2005). Patients who use denial as a coping strategy, therefore, may have impaired insight but suffer less distress as has been shown in several cross-sectional studies (Mintz et al., 2003). We did not directly test whether poor insight is related to the use of denial as a coping strategy, however. No significant relationship was found between self-reflectiveness abilities and negative affect after suppression, suggesting that patients with poorer self-reflectiveness abilities were as successful in reducing negative affect. No significant relationships were found between the other SAI-subscale scores and activation or connectivity during emotion regulation. This is in line with the idea that insight is a multidimensional construct, in which different brain areas are involved in separate dimensions of insight (Antonius et al., 2011; Shad et al., 2006). In addition, no significant relationships were found between activation or connectivity and the BCIS self-certainty score. Other studies investigating the association between insight and activation during tasks also found mixed results with regard to the BCIS composite index score and subscale scores. One study found significant associations with SR only (van der Meer et al., 2013), while another study found significant associations with both the composite index score as well as SR (Lee et al., 2015). No fMRI-studies found significant associations with the SC-subscale thus far.
We did not find significant correlations between insight and other variables such as demographic or clinical characteristics, self-reported use of suppression and reappraisal, and self-reported negative affect after correction for multiple testing. The lack of significant correlations between self-reported use of emotion regulation strategy and insight could be explained by our measure of emotion regulation strategy (i.e. ERQ), which is a relatively simple self-report questionnaire. This measure may not be the most optimal way to measure emotion regulation strategies in patients with poor insight, also given that emotion regulation often occurs implicitly.
4.1. Limitations
First, this study was cross-sectional so no conclusions about the direction of causality can be drawn. Second, antipsychotic medication may have influenced brain activation. However, several studies have shown that there is no common effect of antipsychotics on BOLD-signal (Roder et al., 2013). Also, a high percentage of patients with schizophrenia used antipsychotic medication in this study, which makes this group of participants a good representation of the whole patient population. Third, the ability to regulate emotions was difficult to monitor. The effectiveness of reduction of negative affect was measured with self-reported affect and neural indices in this study. Future studies should verify effectiveness of emotion regulation by measuring facial expression and/or physiological measures of emotional reactivity such as heart rate, skin conductance, pupil dilation and startle eye blink magnitude. Fourth, in this study, a late-cueing design was used in which the regulation instruction was presented 2 s after stimulus presentation. Therefore, we did not find activation in the amygdala during regulation as this happened before. A design in which there is simultaneous presentation of the stimulus and instructions for regulation could be more sensitive to amygdala activation. On the other hand, our late-cueing design is more similar to real life in which a negative stimulus presents before an individual starts regulating their emotions.
4.2. Conclusions
To summarize, our results suggest that patients with poorer ability to relabel symptoms engage different neural pathways during expressive suppression, which are implicated in cognitive-emotional control and visual processing of negative stimuli. This may be explained by implicit reduction of processing of emotion-evoking aspects of negative stimuli during expressive suppression. In addition, our results suggest that patients with poorer self-reflectiveness abilities engage less in control and execution of the task during suppression. Our results are in line with the denial model that suggests that poor insight is caused by the use of denial as a coping strategy in order to reduce distress caused by stigma associated with diagnosis of schizophrenia. Future studies could investigate whether incorporating emotion regulation aspects into interventions that try to improve insight helps in improving insight as well as prognosis, as there is still a great need for improvement (Pijnenborg et al., 2013).
The following are the supplementary data related to this article.
Supplementary Fig. S1.
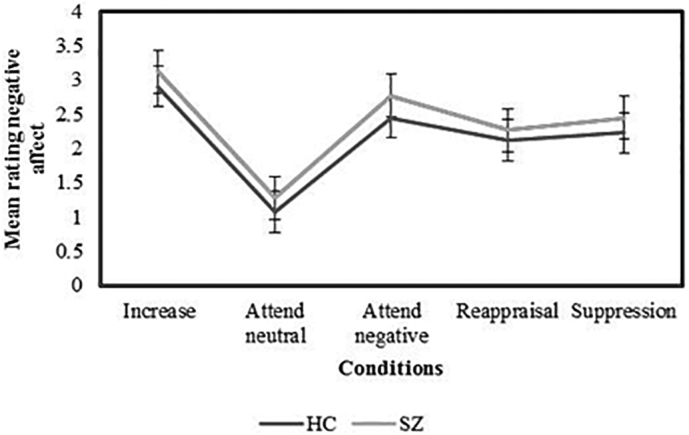
Affect ratings during emotion regulation task.
Supplementary material
Declaration of interest
None.
Acknowledgments
Acknowledgements
The authors would like to thank all participants for their participation, and involved mental health care organizations for their cooperation. We would also like to thank Anita Sibeijn-Kuiper and Judith Streurman for support in scanning the participants, and Dr. Jan-Bernard Marsman and Dr. Remco Renken for their support in data analysis.
Funding
This work was supported by a European Young Investigator Award of the European Science Foundation (no. 044035001) awarded to A.A.
References
- American Psychiatric Association . 2000. DSM-IV-TR, Diagnostic and Statistical Manual of Mental Disorders 4th edition TR. [Google Scholar]
- Andreasen N.C., Pressler M., Nopoulos P., Miller D., Ho B.C. Antipsychotic dose equivalents and dose-years: a standardized method for comparing exposure to different drugs. Biol. Psychiatry. 2010;67:255–262. doi: 10.1016/j.biopsych.2009.08.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antonius D., Prudent V., Rebani Y., D'Angelo D., Ardekani B.A., Malaspina D., Hoptman M.J. White matter integrity and lack of insight in schizophrenia and schizoaffective disorder. Schizophr. Res. 2011;128:76–82. doi: 10.1016/j.schres.2011.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badcock J.C., Paulik G., Maybery M.T. The role of emotion regulation in auditory hallucinations. Psychiatry Res. 2011;185:303–308. doi: 10.1016/j.psychres.2010.07.011. [DOI] [PubMed] [Google Scholar]
- Bebko G.M., Franconeri S.L., Ochsner K.N., Chiao J.Y. Look before you regulate: differential perceptual strategies underlying expressive suppression and cognitive reappraisal. Emotion. 2011;11:732–742. doi: 10.1037/a0024009. [DOI] [PubMed] [Google Scholar]
- Beck A.T., Baruch E., Balter J.M., Steer R.A., Warman D.M. A new instrument for measuring insight: the beck cognitive insight scale. Schizophr. Res. 2004;68:319–329. doi: 10.1016/S0920-9964(03)00189-0. [DOI] [PubMed] [Google Scholar]
- Brett M., Anton J.L., Valabregue R., Poline J.B. Available on CD-ROM in NeuroImage. Vol. 16. 2002. Region of interest analysis using an SPM toolbox [abstract] Presented at the 8th international conference on functional mapping of the human brain, June 2-6, 2002, Sendai, Japan; p. 2. [Google Scholar]
- Buchy L., Brodeur M.B., Lepage M. The beck cognitive insight scale: psychometric properties in a Canadian community sample. Schizophr. Res. 2012;137:254–255. doi: 10.1016/j.schres.2012.02.020. [DOI] [PubMed] [Google Scholar]
- Buchy L., Hawco C., Joober R., Malla A., Lepage M. Cognitive insight in first-episode schizophrenia: further evidence for a role of the ventrolateral prefrontal cortex. Schizophr. Res. 2015;166:65–68. doi: 10.1016/j.schres.2015.05.009. [DOI] [PubMed] [Google Scholar]
- Buhle J.T., Silvers J.A., Wager T.D., Lopez R., Onyemekwu C., Kober H., Weber J., Ochsner K.N. Cognitive reappraisal of emotion: a meta-analysis of human neuroimaging studies. Cereb. Cortex. 2014;24:2981–2990. doi: 10.1093/cercor/bht154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooke M.A., Peters E.R., Kuipers E., Kumari V. Disease, deficit or denial? Models of poor insight in psychosis. Acta Psychiatr. Scand. 2005;112:4–17. doi: 10.1111/j.1600-0447.2005.00537.x. [DOI] [PubMed] [Google Scholar]
- Cooke M.A., Peters E.R., Fannon D., Aasen I., Kuipers E., Kumari V. Cognitive insight in psychosis: the relationship between self-certainty and self-reflection dimensions and neuropsychological measures. Psychiatry Res. 2010;178:284–289. doi: 10.1016/j.psychres.2009.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford J.R., Henry J.D. The Positive and Negative Affect Schedule (PANAS): Construct validity, measurement properties and normative data in a large non-clinical sample. Br. J. Clin. Psychol. 2004;43:245–265. doi: 10.1348/0144665031752934. [DOI] [PubMed] [Google Scholar]
- Ćurčić-Blake B., van der Meer L., Pijnenborg G.H.M., David A.S., Aleman A. Insight and psychosis: functional and anatomical brain connectivity and self-reflection in Schizophrenia. Hum. Brain Mapp. 2015;36:4859–4868. doi: 10.1002/hbm.22955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dam J. Insight in schizophrenia: a review. Nord. J. Psychiatr. 2006;60:114–120. doi: 10.1080/08039480600600185. [DOI] [PubMed] [Google Scholar]
- Dantas C. de R., Banzato C.E.M. Inter-rater reliability and factor analysis of the Brazilian version of the Schedule for the Assessment of Insight - Expanded Version (SAI-E) Rev. Bras. Psiquiatr. 2007;29:359–362. doi: 10.1590/s1516-44462006005000041. [DOI] [PubMed] [Google Scholar]
- David A.S. Insight and psychosis. Br. J. Psychiatry. 1990;156:798–808. doi: 10.1192/bjp.156.6.798. [DOI] [PubMed] [Google Scholar]
- David A.S., Morgan K.D., Mallet J.L., Left J., Murray R.M. Presented at International Congress on Schizophrenia Research 2003, in: Schizophrenia Research. 2003. Insight: unitary or multi-dimensional phenomenon? p. 14. [Google Scholar]
- Diekhof E.K., Geier K., Falkai P., Gruber O. Fear is only as deep as the mind allows. NeuroImage. 2011;58:275–285. doi: 10.1016/j.neuroimage.2011.05.073. [DOI] [PubMed] [Google Scholar]
- Dillon D.G., Ritchey M., Johnson B.D., Labar K.S. Dissociable effects of conscious emotion regulation strategies on explicit and implicit memory. Emotion. 2007;7:354–365. doi: 10.1037/1528-3542.7.2.354. [DOI] [PubMed] [Google Scholar]
- Favrod J., Zimmermann G., Raffard S., Pomini V., Khazaal Y. The Beck Cognitive Insight Scale in outpatients with psychotic disorders: further evidence from a French-speaking sample. Can. J. Psychiatr. 2008;53:783–787. doi: 10.1177/070674370805301111. [DOI] [PubMed] [Google Scholar]
- Fossati P., Hevenor S.J., Graham S.J., Grady C., Keightley M.L., Craik F., Mayberg H. In search of the emotional self: an FMRI study using positive and negative emotional words. Am. J. Psychiatry. 2003;160:1938–1945. doi: 10.1176/appi.ajp.160.11.1938. [DOI] [PubMed] [Google Scholar]
- Gardner D.M., Murphy A.L., O'Donnell H., Centorrino F., Baldessarini R.J. International consensus study of antipsychotic dosing. Am. J. Psychiatry. 2010;167:686–693. doi: 10.1176/appi.ajp.2009.09060802. [DOI] [PubMed] [Google Scholar]
- Goldin P.R., McRae K., Ramel W., Gross J.J. The neural bases of emotion regulation: reappraisal and suppression of negative emotion. Biol. Psychiatry. 2008;63:577–586. doi: 10.1016/j.biopsych.2007.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross J.J. Antecedent- and response-focused emotion regulation: divergent consequences for experience, expression, and physiology. J. Pers. Soc. Psychol. 1998;74:224–237. doi: 10.1037//0022-3514.74.1.224. [DOI] [PubMed] [Google Scholar]
- Gross J.J., John O.P. Individual differences in two emotion regulation processes: implications for affect, relationships, and well-being. J. Pers. Soc. Psychol. 2003;85:348–362. doi: 10.1037/0022-3514.85.2.348. [DOI] [PubMed] [Google Scholar]
- Gutiérrez-Zotes J.A., Valero J., Cortés M.J., Labad A., Ochoa S., Ahuir M., Carlson J., Bernardo M., Cañizares S., Escartin G., Cañete J., Gallo P., Salamero M. Spanish adaptation of the Beck Cognitive Insight Scale (BCIS) for schizophrenia. Actas Esp. Psiquiatr. 2012;40:2–9. [PubMed] [Google Scholar]
- Hayes J.P., Morey R.A., Petty C.M., Seth S., Smoski M.J., McCarthy G., Labar K.S. Staying cool when things get hot: emotion regulation modulates neural mechanisms of memory encoding. Front. Hum. Neurosci. 2010;4:1–10. doi: 10.3389/fnhum.2010.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry J.D., Green M.J., de Lucia A., Restuccia C., McDonald S., O'Donnell M. Emotion dysregulation in schizophrenia: reduced amplification of emotional expression is associated with emotional blunting. Schizophr. Res. 2007;95:197–204. doi: 10.1016/j.schres.2007.06.002. [DOI] [PubMed] [Google Scholar]
- Henry J.D., Rendell P.G., Green M.J., McDonald S., O'Donnell M. Emotion regulation in schizophrenia: affective, social, and clinical correlates of suppression and reappraisal. J. Abnorm. Psychol. 2008;117:473–478. doi: 10.1037/0021-843X.117.2.473. [DOI] [PubMed] [Google Scholar]
- Hoffstaedter F., Grefkes C., Caspers S., Roski C., Palomero-Gallagher N., Laird A.R., Fox P.T., Eickhoff S.B. The role of anterior midcingulate cortex in cognitive motor control: evidence from functional connectivity analyses. Hum. Brain Mapp. 2014;35:2741–2753. doi: 10.1002/hbm.22363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horan W.P., Hajcak G., Wynn J.K., Green M.F. Impaired emotion regulation in schizophrenia: evidence from event-related potentials. Psychol. Med. 2013;43:2377–2391. doi: 10.1017/S0033291713000019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kao Y.-C., Liu Y.-P. The Beck Cognitive Insight Scale (BCIS): translation and validation of the Taiwanese version. BMC Psychiatr. 2010;10(27) doi: 10.1186/1471-244X-10-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kay S.R., Fiszbein A., Opler L.A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 1987;13:261–276. doi: 10.1093/schbul/13.2.261. [DOI] [PubMed] [Google Scholar]
- Kemp R., David A.S. Insight and compliance. In: Blackwell B., editor. Treatment Compliance and the Therapeutic Alliance in Serious Mental Illness. Hardwood Academic Publishers; Amsterdam: 1997. pp. 61–84. [Google Scholar]
- Kimhy D., Vakhrusheva J., Jobson-Ahmed L., Tarrier N., Malaspina D., Gross J.J. Emotion awareness and regulation in individuals with schizophrenia: implications for social functioning. Psychiatry Res. 2012;200:193–201. doi: 10.1016/j.psychres.2012.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kircher T.T.J., Leube D.T. Self-consciousness, self-agency, and schizophrenia. Conscious. Cogn. 2003;12:656–669. doi: 10.1016/s1053-8100(03)00071-0. [DOI] [PubMed] [Google Scholar]
- Kohn N., Eickhoff S.B., Scheller M., Laird A.R., Fox P.T., Habel U. Neural network of cognitive emotion regulation - an ALE meta-analysis and MACM analysis. NeuroImage. 2014;87:345–355. doi: 10.1016/j.neuroimage.2013.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konstantakopoulos G., Ploumpidis D., Oulis P., Soumani A., Nikitopoulou S., Pappa K., Papadimitriou G.N., David A.S. Is insight in schizophrenia multidimensional? Internal structure and associations of the Greek version of the schedule for the assessment of insight-expanded. Psychiatry Res. 2013;209:346–352. doi: 10.1016/j.psychres.2013.02.016. [DOI] [PubMed] [Google Scholar]
- Kumari V., Fannon D., Ffytche D.H., Raveendran V., Antonova E., Premkumar P., Cooke M.A., Anilkumar A.P.P., Williams S.C.R., Andrew C., Johns L.C., Fu C.H.Y., McGuire P.K., Kuipers E. Functional MRI of verbal self-monitoring in schizophrenia: performance and illness-specific effects. Schizophr. Bull. 2010;36:740–755. doi: 10.1093/schbul/sbn148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J.S., Chun J.W., Lee S.H., Kim E., Lee S.K., Kim J.J. Altered neural basis of the reality processing and its relation to cognitive insight in schizophrenia. PLoS ONE. 2015;10:1–15. doi: 10.1371/journal.pone.0120478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lincoln T.M., Lullmann E., Rief W. Correlates and long-term consequences of poor insight in patients with schizophrenia. A systematic review. Schizophr. Bull. 2006;33:1324–1342. doi: 10.1093/schbul/sbm002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Livingstone K., Harper S., Gillanders D. An exploration of emotion regulation in psychosis. Clin. Psychol. Psychother. 2009;16:418–430. doi: 10.1002/cpp.635. [DOI] [PubMed] [Google Scholar]
- McGuire P.K., Silbersweig D.A., Frith C.D. Functional neuroanatomy of verbal self-monitoring. Brain. 1996;119:907–917. doi: 10.1093/brain/119.3.907. [DOI] [PubMed] [Google Scholar]
- McLaren D.G., Ries M.L., Xu G., Johnson S.C. A generalized form of context-dependent psychophysiological interactions (gPPI): a comparison to standard approaches. NeuroImage. 2012;61:1277–1286. doi: 10.1016/j.neuroimage.2012.03.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melka S.E., Lancaster S.L., Bryant A.R., Rodriguez B.F. Confirmatory factor and measurement invariance analyses of the emotion regulation questionnaire. J. Clin. Psychol. 2011;67:1283–1293. doi: 10.1002/jclp.20836. [DOI] [PubMed] [Google Scholar]
- Middleton F.A., Strick P.L. Basal ganglia and cerebellar loops: motor and cognitive circuits. Brain Res. Rev. 2000;31:236–250. doi: 10.1016/s0165-0173(99)00040-5. [DOI] [PubMed] [Google Scholar]
- Mintz A.R., Dobson K.S., Romney D.M. Insight in schizophrenia: a meta-analysis. Schizophr. Res. 2003;61:75–88. doi: 10.1016/s0920-9964(02)00316-x. [DOI] [PubMed] [Google Scholar]
- Modinos G., Renken R., Ormel J., Aleman A. Self-reflection and the psychosis-prone brain: an fMRI study. Neuropsychology. 2011;25:295–305. doi: 10.1037/a0021747. [DOI] [PubMed] [Google Scholar]
- Morris R.W., Sparks A., Mitchell P.B., Weickert C.S., Green M.J. Lack of cortico-limbic coupling in bipolar disorder and schizophrenia during emotion regulation. Transl. Psychiatry. 2012;2:e90. doi: 10.1038/tp.2012.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochsner K.N., Bunge S.A., Gross J.J., Gabrieli J.D.E. Rethinking feelings: an FMRI study of the cognitive regulation of emotion. J. Cogn. Neurosci. 2002;14:1215–1229. doi: 10.1162/089892902760807212. [DOI] [PubMed] [Google Scholar]
- Ohira H., Nomura M., Ichikawa N., Isowa T., Iidaka T., Sato A., Fukuyama S., Nakajima T., Yamada J. Association of neural and physiological responses during voluntary emotion suppression. NeuroImage. 2006;29:721–733. doi: 10.1016/j.neuroimage.2005.08.047. [DOI] [PubMed] [Google Scholar]
- Palaniyappan L., Mallikarjun P., Joseph V., Liddle P.F. Appreciating symptoms and deficits in schizophrenia: right posterior insula and poor insight. Prog. Neuro-Psychopharmacol. Biol. Psychiatr. 2011;35:523–527. doi: 10.1016/j.pnpbp.2010.12.008. [DOI] [PubMed] [Google Scholar]
- Perry Y., Henry J.D., Grisham J.R. The habitual use of emotion regulation strategies in schizophrenia. Br. J. Clin. Psychol. 2011;50:217–222. doi: 10.1111/j.2044-8260.2010.02001.x. [DOI] [PubMed] [Google Scholar]
- Perry Y., Henry J.D., Nangle M.R., Grisham J.R. Regulation of negative affect in schizophrenia: the effectiveness of acceptance versus reappraisal and suppression. J. Clin. Exp. Neuropsychol. 2012;34:497–508. doi: 10.1080/13803395.2012.661405. [DOI] [PubMed] [Google Scholar]
- Pijnenborg G.H.M., Van der Gaag M., Bockting C.L.H., Van der Meer L., Aleman A. REFLEX, a social-cognitive group treatment to improve insight in schizophrenia: study protocol of a multi-center RCT. BMC Psychiatr. 2011;11(161) doi: 10.1186/1471-244X-11-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pijnenborg G.H.M., Van Donkersgoed R.J.M., David A.S., Aleman A. Changes in insight during treatment for psychotic disorders: a meta-analysis. Schizophr. Res. 2013;144:109–117. doi: 10.1016/j.schres.2012.11.018. [DOI] [PubMed] [Google Scholar]
- Raij T.T., Korkeila J., Joutsenniemi K., Saarni S.I., Riekki T.J.J. Association of stigma resistance with emotion regulation - functional magnetic resonance imaging and neuropsychological findings. Compr. Psychiatry. 2014;55:727–735. doi: 10.1016/j.comppsych.2013.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards J.M., Gross J.J. Emotion regulation and memory: the cognitive costs of keeping one's cool. J. Pers. Soc. Psychol. 2000;79:410–424. doi: 10.1037//0022-3514.79.3.410. [DOI] [PubMed] [Google Scholar]
- Roder C.H., Dieleman S., van der Veen F.M., Linden D. Systematic review of the influence of antipsychotics on the blood oxygenation level-dependent signal of functional magnetic resonance imaging. Curr. Med. Chem. 2013;20:448–461. doi: 10.2174/092986713804870891. [DOI] [PubMed] [Google Scholar]
- Rorden C., Brett M. Stereotaxic display of brain lesions. Behav. Neurol. 2000;12:191–200. doi: 10.1155/2000/421719. [DOI] [PubMed] [Google Scholar]
- Sapara A., Ffytche D.H., Cooke M.A., Williams S.C.R., Kumari V. Is it me? Verbal self-monitoring neural network and clinical insight in schizophrenia. Psychiatry Res. Neuroimaging. 2015;234:328–335. doi: 10.1016/j.pscychresns.2015.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shad M.U., Keshavan M.S. Neurobiology of insight deficits in schizophrenia: an fMRI study. Schizophr. Res. 2015;165:220–226. doi: 10.1016/j.schres.2015.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shad M.U., Muddasani S., Keshavan M.S. Prefrontal subregions and dimensions of insight in first-episode schizophrenia - a pilot study. Psychiatry Res. Neuroimaging. 2006;146:35–42. doi: 10.1016/j.pscychresns.2005.11.001. [DOI] [PubMed] [Google Scholar]
- Sheehan D.V., Lecrubier Y., Sheehan K.H., Amorim P., Janavs J., Weiller E., Hergueta T., Baker R., Dunbar G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatr. 1998;59:22–33. [PubMed] [Google Scholar]
- Shergill S.S., Bullmore E.T., Brammer M.J., Williams S.C., Murray R.M., McGuire P.K. A functional study of auditory verbal imagery. Psychol. Med. 2001;31:241–253. doi: 10.1017/s003329170100335x. [DOI] [PubMed] [Google Scholar]
- Stippich C., Ochmann H., Sartor K. Somatotopic mapping of the human primary sensorimotor cortex during motor imagery and motor execution by functional magnetic resonance imaging. Neurosci. Lett. 2002;331:50–54. doi: 10.1016/s0304-3940(02)00826-1. [DOI] [PubMed] [Google Scholar]
- Uchida T., Matsumoto K., Kikuchi A., Miyakoshi T., Ito F., Ueno T., Matsuoka H. Psychometric properties of the Japanese version of the beck cognitive insight scale: Relation of cognitive insight to clinical insight. Psychiatry Clin. Neurosci. 2009;63:291–297. doi: 10.1111/j.1440-1819.2009.01946.x. [DOI] [PubMed] [Google Scholar]
- van der Meer L., Van'T Wout M., Aleman A. Emotion regulation strategies in patients with schizophrenia. Psychiatry Res. 2009;170:108–113. doi: 10.1016/j.psychres.2009.07.010. [DOI] [PubMed] [Google Scholar]
- van der Meer L., de Vos A.E., Stiekema A.P.M., Pijnenborg G.H.M., Van Tol M.J., Nolen W.A., David A.S., Aleman A. Insight in schizophrenia: involvement of self-reflection networks? Schizophr. Bull. 2013;39:1288–1295. doi: 10.1093/schbul/sbs122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Meer L., Swart M., Van Der Velde J., Pijnenborg G., Wiersma D., Bruggeman R., Aleman A. Neural correlates of emotion regulation in patients with schizophrenia and non-affected siblings. PLoS ONE. 2014;9 doi: 10.1371/journal.pone.0099667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Reekum C.M., Johnstone T., Urry H.L., Thurow M.E., Schaefer H.S., Alexander A.L., Davidson R.J. Gaze fixations predict brain activation during the voluntary regulation of picture-induced negative affect. NeuroImage. 2007;36:1041–1055. doi: 10.1016/j.neuroimage.2007.03.052. [DOI] [PubMed] [Google Scholar]
- Vanderhasselt M.A., Kühn S., De Raedt R. “Put on your poker face”: neural systems supporting the anticipation for expressive suppression and cognitive reappraisal. Soc. Cogn. Affect. Neurosci. 2013;8:903–910. doi: 10.1093/scan/nss090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verhage . Van Gorcum; Assen: 1964. Intelligentie en leeftijd: Onderzoek bij Nederlanders van twaalf tot zevenenzeventig jaar [Intelligence and age: Research study in Dutch individuals age twelve to seventy-seven] [Google Scholar]
- Watson D., Clark L.A., Tellegen A. Development and validation of brief measures of positive and negative affect: the PANAS scales. J. Pers. Soc. Psychol. 1988;54:1063–1070. doi: 10.1037//0022-3514.54.6.1063. [DOI] [PubMed] [Google Scholar]
- World Health Organisation . 2012. ICD 10 [WWW Document]. WHO. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material