Abstract
Diabetic kidney disease (DKD) is characterized by progressive glomerulosclerosis (GS). ROP mice have a sclerosis-prone phenotype. However, they develop severe, rapidly progressive GS when rendered diabetic. Since GS also develops in aged C57Bl6 mice, and can be reversed using bone marrow from young mice which have lower oxidative stress and inflammation (OS/Infl), we postulated that this might also apply to DKD. Therefore, this pilot study asked whether reducing OS/Infl in young adult sclerosis-prone (ROP) diabetic mice leads to resolution of existing GS in early DKD using safe, FDA-approved drugs.After 4 weeks of stable streptozotocin-induced hyperglycemia 8–12 week-old female mice were randomized and treated for 22 weeks as follows: 1) enalapril (EN) (n = 8); 2) pyridoxamine (PYR)+EN (n = 8); 3) pentosan polysulfate (PPS)+EN (n = 7) and 4) PPS+PYR+EN (n = 7). Controls were untreated (non-DB, n = 7) and hyperglycemic (DB, n = 8) littermates. PPS+PYR+EN reduced albuminuria and reversed GS in DB. Treatment effects: 1) Anti-OS/Infl defenses: a) PPS+PYR+EN increased the levels of SIRT1, Nrf2, estrogen receptor α (ERα) and advanced glycation endproduct-receptor1 (AGER1) levels; and b) PYR+EN increased ERα and AGER1 levels. 2) Pro-OS/Infl factors: a) PPS+PYR+EN reduced sTNFR1, b) all except EN reduced MCP1, c) RAGE was reduced by all treatments. In summary, PYR+PPS+EN modulated GS in sclerosis-prone hyperglycemic mice. PYR+PPS+EN also decreased albuminuria, OS/Infl and the sclerosis-prone phenotype. Thus, reducing OS/Infl may reverse GS in early diabetes in patients, and albuminuria may allow early detection of the sclerosis-prone phenotype.
Introduction
While the pathogenesis and causes of glomerulosclerosis (GS) are unclear, it has been thought to be inevitably progressive. Diabetic nephropathy (DKD), the most common form of GS in adults, is restricted to a subset of diabetic patients (~20–40%) suggesting a role for a sclerosis-prone genetic background, similar to that described in animal models [1–3]. Factors in addition to hyperglycemia [4] and genetic susceptibility [5] that may influence the development of DKD include gender, age and dietary intake [6–8].
The first successful attempt to identify components of the sclerosis-prone phenotype was a comparison of young adult ROP Os+ (sclerosis-prone) and C57 Os/+ (sclerosis-resistant) mice by transcriptomes [6]. This study “highlighted the roles for oxidative stress” and “provided evidence for activation of the pro-fibrotic and pro-inflammatory transforming growth factor β1 (TGFβ1) signal” in the genetic susceptibility to GS. These data complemented other data suggesting that the pathogenesis of DKD includes both environmental [9–11] and genetic factors [5, 12] in which inflammation and oxidative stress (OS/Infl) may play pivotal roles [6, 13–17].
The modern human diet and most pelleted experimental animal food contain a ubiquitous group of environmental factors, including glycation endproducts (AGEs), which are strong inducers of OS/Infl and are a risk factor for DKD and chronic kidney disease (CKD) [7, 8, 10, 11, 16, 17]. We found that GS develops as OS/Infl increases in normal aging, and that GS can be reduced by controlling the oral intake of agents that increase OS/Infl, such as AGEs [18].
Additionally, GS can be reduced by the transplantation of bone marrow from young mice, which have intact OS/Infl defenses, into old mice, which have lowered OS/Infl defenses [19, 20]. In addition GS is induced in young mice after administration of bone marrow from old mice.
These studies support a role for OS/Infl in the development as well as the regression of GS independently of hyperglycemia. However, the effect of OS/Infl on the sclerosis-prone phenotype and to gender has not been further tested.
A search for drugs that control OS/Infl revealed that pentosan polysulfate (PPS) blocks certain cell surface pro-inflammatory receptors, such as tumor necrosis factor- α (TNF-α). In addition, pyridoxamine (PYR) blocks intracellular pro-OS/Infl factors, such as methylglyoxal (a very reactive AGE) [21, 22]. Recently, we found that PYR treatment of anestrus (21 months-old) female C57Bl6 mice resulted in increased renal expression of anti-oxidant defenses such as estrogen receptor α (ERα), Sirtuin 1 (SIRT1) and AGER1 (a receptor for AGEs), while decreasing TGFβ1 in aged mice [23]. We postulated that the combination of enalapril (EN), PPS and PYR might provide protection against the development of GS in hyperglycemic sclerosis-prone mice. In this pilot study we chose young hyperglycemic adult, female ROP Os/+ mice at an age at which they were known to developed, progressive GS and albuminuria, but not renal failure [2, 24].
Our aim was to determine if reducing OS/Infl modulated established GS and modified sclerosis-prone phenotype using combinations of FDA-approved generic drugs (PPS, PYR and EN).
Materials and methods
Experimental design
Female ROP Os/+mice were obtained from Jackson Laboratories (Bar Harbor, ME, USA). The protocol was approved by the Icahn School of Medicine at Mount Sinai Institutional Animal Care and Use Committee (Protocol # 080328). Animals were housed in groups of two to four in plastic cages with corncob bedding, maintained on a 12-hour light:dark cycle, and provided with standard laboratory animal chow and water ad lib. Stable hyperglycemia (DB) was induced at 8–12 weeks of age by 7 to 14 intraperitoneal injections of STZ(50 μg/g body wt), or vehicle (citrate buffer) as a control, administered within a two week period [24]. Mice were fed with standard laboratory animal chow. While we have found that standard laboratory contain large amounts of AGEs, we decided to utilize this chow so that the results of these experiments could be more easily reproduced in other laboratories [25]. Mice were weighed and fasting blood glucose levels were determined by tail vein nicking at baseline and weekly thereafter. After 4 weeks of stable hyperglycemia (>200 mg/dl), mice (DB) were randomized to the following treatment groups: EN (10 mg/kg/day, n = 8), PYR+EN (PYR: 200mg/kg/day, n = 8), PPS+EN (PPS: 25mg/kg/day, n = 7), and PPS+PYR+EN (n = 7). Untreated non-DB (non-DB, n = 7) and untreated DB mice (DB, n = 8) served as controls. The estimated number of animals was based on our prior laboratory experience in this ROP/Os + mouse strain, with and without the induction of hyperglycemia [26, 27].
Intraperitoneal NPH insulin was given to maintain blood glucose levels below 300 mg/dl. EN was given to mimic the standard treatment now extant for patients with DKD [28]. Urine albumin excretion was measured weekly by ELISA (Bethyl Laboratories Inc., Houston, TX) [29]. Urine creatinine levels were measured by a colorimetric assay (Cayman Chemical Company, Ann Arbor, MI), and the urine albumin excretion rate was expressed as the ratio of albumin to creatinine (ACR) [9, 29]. Blood urea nitrogen (BUN) levels were evaluated by a colorimetric method (TECO Diagnostics, California, USA) [29, 30]. After 22 weeks of follow-up, mice were terminally anesthetized with ketamine/xylazine according to Guidelines established by the National Institutes of Health (NIH). Level of anesthesia was assessed by pedal reflex and all efforts were made to minimize suffering.
The study was reviewed and approved by the Institutional Animal Care and Use Committee of the Icahn School of Medicine at Mount Sinai, as a prerequisite for submission of the original grant [Juvenile Diabetes Research Foundation (Grant number 17-2008-1041)], which supported this pilot study.
Histology
Kidneys were flushed with phosphate-buffered saline and then fixed in 4% paraformaldehyde [9, 29]. Forty eight hours after fixation, tissue samples were embedded in glycol methacrylate and 4μm sections were stained with periodic acid Schiff.
Glomerular morphometry
Morphologic analysis was performed in periodic acid–Schiff (PAS) stained sections [31]. A total of 30 glomeruli were randomly selected from each kidney by moving the slide from the outer to the inner cortex in a random fashion to obtain non-overlapping sample fields. Glomerular images were recorded using a CCD camera (Sony, Tokyo, Japan) mounted on a light microscope (Zeiss, Gottingen, Germany). Glomerular tuft surface area was obtained using the MetaMorph image analysis computer program (Universal Imaging Co., West Chester, PA). From the glomerular tuft image, the amount of PAS–positive material was selected automatically by use of the color recognition properties of the software. The number of pixels in the PAS-positive area was considered to represent the area of sclerosis and was expressed as a fraction of the tuft surface area [30].
Plasma sTNFR1
Plasma soluble tumor necrosis factor receptor-1 (sTNFR1) levels were determined by ELISA kits following the manufacturers’ protocols (eBioscience, Inc. San Diego, Ca, USA and R&D Systems Inc., Minneapolis, MN, USA respectively).
Circulating and tissue AGEs
The amount of AGEs in the tissue (liver), and plasma was determined by a competitive enzyme-linked immunosorbent assay using a monoclonal antibody reacting with N-carboxymethyl-lysine (CML, 4G9 mAb) or Methylglyoxal (MG, 3D11 mAb) [25]. Tissue values were corrected to the protein sample concentrations. N-carboxymethyl-lysine-bovine or Methylglyoxal serum albumin were used as standards for quantitation.
Oxyblot
An oxyblot protein oxidation detection kit (Chemicon International, Temecula, CA) was used to assess overall carbonyl groups introduced into the protein side chain by oxidative modification in renal tissues [9]. 2,4-Dinitrophenylhydrazine derivatization was performed for 15 minutes following the manufacturer's instructions on total 10 μg of protein obtained from the kidney tissue lysate. The dinitrophenylhydrazine-derivatized protein samples were separated by 12%SDS-polyacrylamide gel electrophoresis. Proteins were transferred to polyvinylidenedifluoride membranes, stained by Ponceau Red, and then probed with an anti-dinitrophenylhydrazine antibody. Blots were developed using a chemiluminescence detection system. No visible bands were seen in samples without reaction with 2,4-dinitrophenylhydrazine before Western blots (data not shown).
Real time PCR studies
Total RNA was isolated from the renal cortex using a PureYield RNA Midiprep kit (Promega, Madison, WI) and the preparation was freed of DNA contamination by incubating with DNase I. Equal amounts of RNA from the samples were then reverse-transcribed as described previously [32]. The mRNA levels of TGFβ1, a receptor for AGEs (RAGE), monocyte chemotactic protein-1 (MCP1), nuclear factor (erythroid-derived 2)-like 2 (Nrf2) and estrogen receptor-α (ERα) were determined by real-time PCR under standard conditions using the SYBR Green PCR Master Mix (Applied Biosystems; Warrington, UK). The primer sequences were: TGFβ1, forward, 5’-GTGCGGCAGCTGTACATTGACTTT and reverse, 5’-TGTACTGTGTGTCCAGGCTCCAAA; RAGE, forward 5’-CCTGGGAAGCCAGAAATT and reverse 5’-ACACAGGTCAAGGTCACA; MCP1, forward, 5’-AGGTCCCTGTCATGCTTCTC and reverse, 5’-TCATTGGGATCATCTTGCT: Nrf2, forward, 5’-TCCATTCCCGAATTACAGTGTC and reverse, 5’-CACAGTTGCCCACTTCTTTTT; ERα, forward, 5’-ATTGGTCTCGTCTGGCGCT and reverse, 5’-CTCCACCATGCCCTCTACACA. The mRNA levels in each sample were normalized by β-actin mRNA levels [9].
Western blots
Proteins were isolated from the renal cortex after homogenization and sonication in a lysis buffer containing protease and phosphatase inhibitors at 4° C (Pierce, Rockford, IL). Samples were centrifuged at 4° C at 14,000 rpm for 15 minutes, the supernatant was recovered and the protein concentration was measured by a colorimetric assay according to the manufacturer’s instructions (BCA Protein Assay Kit, Pierce). The same amount of protein obtained from three mice was pooled from each group and a total of fifteen micrograms of protein were loaded onto 8 or 10% SDS-PAGE gels. After separation, proteins were transferred to PVDF membranes. For Western blots, membranes were pretreated with a blocking buffer (Thermo Fisher Scientific, Waltham, MA) at room temperature for 30 min before incubating with the following antibodies with appropriate dilution: anti-OST48 (AGER1) (Santa Cruz Biotechnology, Santa Cruz, CA, USA) and anti-SIRT1 (Santa Cruz). After overnight incubation with primary antibody at 4°C, membranes were washed and incubated with a horseradish peroxidase-labeled secondary antibody and immunoreactivity was detected using the enhanced chemiluminescence assay (GE Healthcare, Buckinghamshire, UK). Membranes were then washed 30 min with a stripping buffer (Thermo Fisher Scientific) and re-probed with an anti-β-actin antibody (1:5000; Sigma, St. Louis, MO) to ensure that protein loading was similar for each sample. These experiments were at least repeated twice [25].
Statistical analysis
Data were expressed as mean value ± SE. Differences in mean were analyzed by ANOVA and a p value of less than 0.05 was considered significant. Graph Pad Prism 6.0 software was used for data analysis. Outliers were removed from the data set according Moore et al [33]. Backward multiple linear regression analysis were performed to estimate the independent contribution of blood glucose, serum and intracellular CML (sCML and iCML, respectively) and MG (sMG and iMG, respectively) levels, sTNRF1, AGER1 and SIRT-1 protein levels and TGFβ1, MCP1, RAGE, Nrf2 and ERα mRNA levels to ACR and GS, independently, both considered as clinical markers in the prediction of both the presence and the progression of DKD.
Results
General characteristics
Insulin was used to control hyperglycemia (mean = 265±81mg/dl). Blood glucose area under the curve (AUC), calculated after randomization and weekly until sacrifice, was significantly higher in all diabetic groups compared to non-DB controls (p<0.001) but was similar in all diabetic groups (Table 1). Body weight was significantly reduced only in the PPS+EN group compared to non-DB mice (<10%) (p<0.05). BUN remained in the normal range in all groups and kidney weight (unlike that found in sclerosis-resistant strains) was not altered by diabetes or treatment.
Table 1. Characteristics at sacrifice.
| Treatment | Body weight (gr) | Kidney weight/ body weight |
Blood glucose (mg/dl) |
Blood glucose AUC |
BUN (mg/dl) |
|---|---|---|---|---|---|
| Non-DB (n 7) | 23.7 ±0.4 | 0.0051±0.0004 | 124.8±4.0 | 7022±202 | 26.22±1.36 |
| DB (n 8) | 21.8±0.8 | 0.0047±0.0002 | 289.7±37.2 *** | 40318±4787*** | 25.74±2.36 |
| DB EN (n 8) | 21.9 ±0.6 | 0.0042±0.0001 | 269.7±30.2 ** | 38230±4047*** | 24.66±1.32 |
| DB EN+PYR (n 8) | 22.8±0.7 | 0.0043±0.0001 | 224.5±18.3 * | 32556±2144*** | 28.19±2.06 |
| DB EN+PPS (n 7) | 20.9±0.5* | 0.0044±0.0002 | 253.9±23.9** | 37079±3493*** | 32.09±2.76 |
| DB EN+PYR+PPS (n 7) | 21.4±0.6 | 0.0044±0.0003 | 234.4±24.7 * | 33298±2824*** | 30.08±2.31 |
BUN: blood urea nitrogen; AUC: area under the curve.
Data are expressed as mean value ± SE.
*p<0.05
**p<0.01
***p<0.001 vs. Non-DB
Glomerulosclerosis and albuminuria
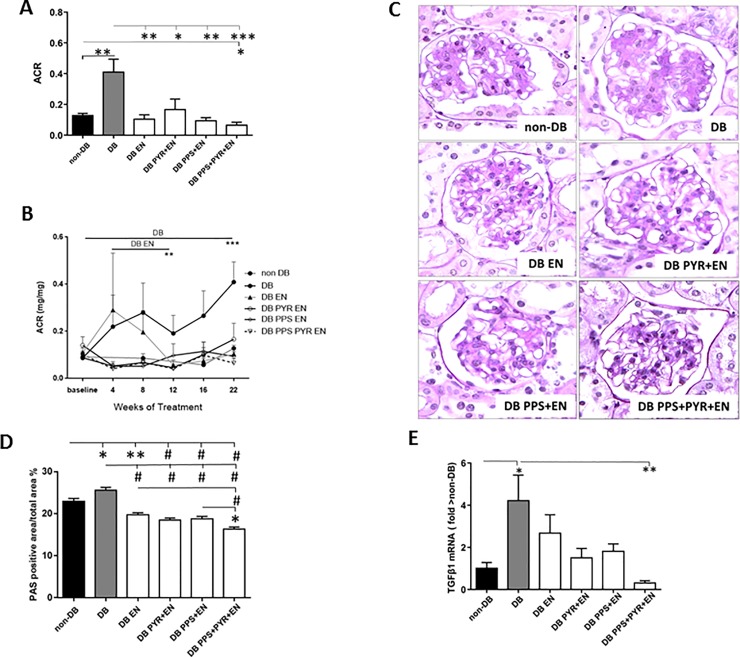
ROP Os/+ mice spontaneously develop proteinuria, glomerulosclerosis, glomerular hypertrophy and are prone to develop slowly progressive DKD [24]. ACR remained stable in non-DB mice, continued to increase in DB mice to reach nearly two-fold higher levels than non-DB at 22 weeks, but was reduced in all treatment groups (Fig 1A and 1B). ACR was most reduced by PPS+PYR+EN (p<0.005) (Fig 1A). PPS+PYR and PPS+PYR+EN treatment reduced albuminuria below DB at 4 weeks (p<0.05 and p<0.001, respectively), an effect that continued to study end. While ACR in EN treated mice was increased at 4 weeks, it returned to non-DB levels by 12 weeks. PPS+PYR+EN reduced ACR below non-DB controls at 22 weeks (p<0.05) (Fig 1A).
Fig 1.
A) Albumin to creatinine ratio (ACR) at sacrifice (22 weeks). B) ACR follow-up for 22 weeks. C) Periodic acid–Schiff (PAS) staining: a) non-DB, b) DB, c) DB EN, d) DB PYR+EN, e) DB PPS+EN, f) DB PPS+PYR+EN. D) Morphometric evaluation of glomerular PAS positive areas. E) Kidney TGFβ1 mRNA levels. *p<0.05; **p<0.01; ***p<0.005; #p<0.001. DB, diabetics; EN, enalapril; PPS, pentosan polysulfate; PYR, pyridoxamine; PAS, periodic acid–Schiff; TGFβ1, transforming growth factor β1.
GS, as evaluated by PAS staining (Fig 1C), was evident in non-DB littermates and increased in DB (p<0.05), and was improved by all treatments (p<0.001) (Fig 1C and 1D). GS was more reduced by PPS+PYR+EN than by EN (p<0.001), or DB PPS+EN (p<0.05). The treatment regimens reduced GS below that in non-DB (EN p<0.01; PYR+EN, PPS+EN, PPS+PYR+EN p<0.001) (Fig 1C and 1D). None of the DB mice had tubulo-interstitial inflammatory cell infiltrates or glomerular hypertrophy compared to non-DB, consistent with previous data on ROP Os+ mice at this age [2, 3].
The increased renal TGFβ1 mRNA levels, often associated with GS[34], were increased in DB, compared to non-DB (p<0.05) and reduced only by PPS+PYR+EN (p<0.01) (Fig 1E).
Oxidative stress and inflammation
Pro-OS/Infl factors
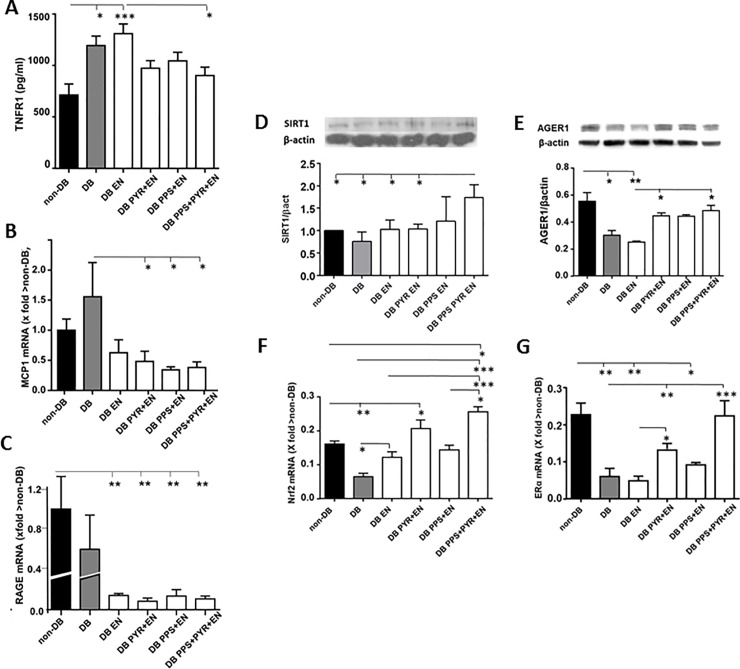
In comparison to non-DB, sTNFR1 levels were higher in DB and DB EN (p<0.05 and p<0.005, respectively) (Fig 2A). PPS+PYR+EN was the only treatment that significantly reduced sTNFR1 levels in DB mice (p<0.05), while there was only a trend in DB PYR+EN and DB PPS+EN.
Fig 2.
A) Plasma sTNFR1 levels. B) Kidney MCP1 mRNA levels. C) Kidney RAGE mRNA levels. D) Kidney SIRT1 protein levels. E) Kidney AGER1 protein levels. F) Kidney Nrf2 mRNA levels G) Kidney ERα mRNA levels. *p<0.05; **p<0.01; ***p<0.005; #p<0.001. DB, diabetics; EN, enalapril; PPS, pentosan polysulfate; PYR, pyridoxamine; sTNFR1, tumor necrosis factor receptor-1; MCP1, monocyte chemotactic protein-1; RAGE and AGER1, receptors for AGEs; SIRT1, Sirtuin-1; Nrf2, nuclear factor (erythroid-derived 2)-like 2; ERα, Estrogen receptor α.
MCP1 mRNA levels [35] were not different between DB and non-DB (Fig 2B), but were significantly reduced in all treatment groups compared to DB (p<0.05) except in DB EN in which only a trend was present. There were no differences between the treatment groups and non-DB.
RAGE mRNA levels were not different between DB and non-DB, but were decreased in all treatment groups (p<0.01) (Fig 2C).
Anti-Os/Infl factors
SIRT1 protein levels were increased in DB PPS+PYR+EN, compared to both non-DB and all DB groups except PPS+EN treated animals (p<0.05) (Fig 2D).
AGER1 protein levels were reduced in both DB (p<0.05) and DB+EN compared to non-DB (p<0.01), but were increased to approximate non-DB levels in DB mice treated with either DB PYR+EN or DB PPS+PYR+EN (p<0.05) (Fig 2E). The levels in DB PYR+EN and DB PPS+PYR+EN were higher than DB EN (p<0.05)
Nrf2 mRNA levels were higher in non-DB mice compared to DB (p<0.01). Nrf2 mRNA was increased in DB+EN compared to DB (p<0.05) (Fig 2F). Nrf2 mRNA levels were also increased in DB PPS+PYR+EN compared to DB PPS+EN (p<0.05), DB EN (p<0.005), DB (p<0.005) and non-DB (p<0.05).
ERα mRNA levels were lower in DB compared to non-DB (p<0.01), DB+EN (p<0.01), DB PYR+EN and DB PPS+EN (p<0.05). ERα expression was also higher in DB+EN compared to DB EN+PYR (p< 0.05). However, ERα expression was increased by PYR+EN compared to DB (p<0.01). ERα expression was returned to non-DB control levels by PPS+PYR+EN (p<0.005) (Fig 2G).
There were no significant differences in liver tissue (intracellular) or circulating (serum) levels of CML or MG (data not shown). As in most previous studies of ROP mice at this “young/adult” age we found little histological evidence of interstitial inflammatory cells [2, 3, 24, 36].
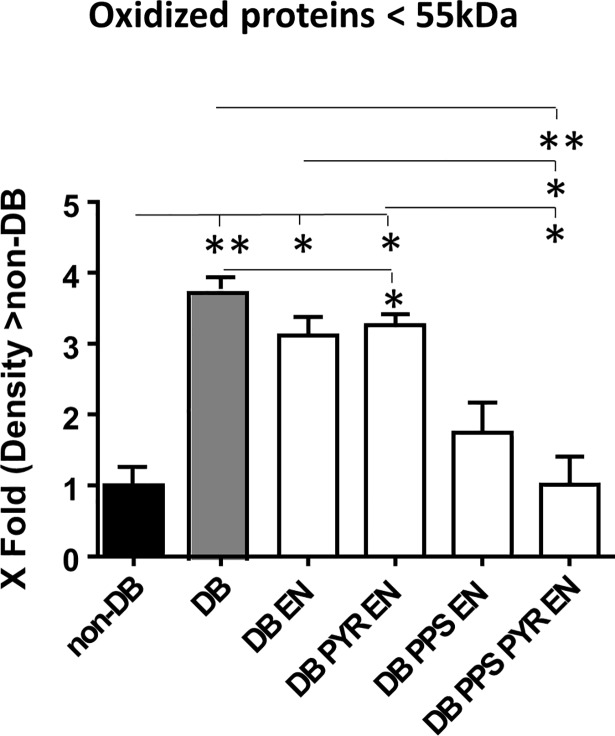
Oxidized total kidney protein
The levels of oxidized proteins with a molecular weight <55 kDa were significantly increased in DB kidneys (p<0.01) (Fig 3). PYR+EN and PPS+PYR+EN reduced the levels of these proteins in DB kidneys (p<0.05 and p<0.01 respectively), reaching levels similar to non-DB controls. The levels of oxidized kidney proteins with a MW >55Kd were not increased in DB, but were lower than DB in the PPS+PYR+EN group (p<0.05) (S1 Fig).
Fig 3. Oxidized kidney proteins < 55kDa, densitometric analyses.
*p<0.05; **p<0.01. DB, diabetics; EN, enalapril; PPS, pentosan polysulfate; PYR, pyridoxamine.
Independent association of inflammation and oxidative stress-related parameters and the albumin/creatinine ratio (ACR)
In a multiple linear regression analysis with ACR as the dependent variable, sMG and sTNFR1 levels as well as TGFβ1, RAGE and MCP1 mRNA levels appeared as significant predictors of ACR (all p<0.05) (Table 2). However, there was no independent link of ACR with sCML, iCML, iMG levels and AGER1 protein levels, and Nrf2 and ERα mRNA levels. ACR levels were reduced to values comparable to non-DB controls only by PPS+PYR+EN (p<0.01). There were no associations with any of the other analyzed parameters when GS was used as the dependent variable.
Table 2. Multiple linear regression coefficients* to predict ACR.
| Variables | Unstandardized Coefficients | Standardized Coefficients | p value | |
|---|---|---|---|---|
| B | Standard error | Beta | ||
| sMG | 0.164 | 0.066 | 0.306 | 0.019 |
| sTNFR1 | 3.18 x 10−4 | 9.21 x 10−5 | 0.322 | 0.007 |
| TGFβ1 mRNA | 0.038 | 0.016 | 0.277 | 0.036 |
| RAGE mRNA | 0.153 | 0.047 | 0.449 | 0.008 |
| MCP1 mRNA | 0.222 | 0.048 | 0.517 | 0.001 |
Dependent Variable: ACR (units) Predictive variables tested by backward method: blood glucose (mg/dl), sCML (U/mL), sMG (nmol/mL), iCML (U/mg protein), iMG (nmol/mg protein), sTNFR1 (pg/mL), AGER1 and SIRT1 protein levels, and TGFβ1, MCP1, RAGE, Nrf2 and ERα mRNA levels.
*(Constant) = 0.333. (R) = 0.945.
Discussion
These results show that progressive GS in adult sclerosis-prone mice with controlled hyperglycemia can be reversed using a combination of FDA-approved drugs that are safe, readily available and inexpensive. Pentosan polysulfate+pyridoxamine+enalapril (PPS+PYR+EN) treatment: 1) reduced established GS and albuminuria; 2) increased anti-OS/Infl defenses and decreased pro-OS/Infl components, and 3) reduced elements of the sclerosis-prone phenotype in non-DB controls. Other combinations of these drugs had less marked results, suggesting that effective control of DKD may require addressing both extracellular and intracellular OS/Infl.
PPS and PYR are extensively utilized in the clinics and have good a safety profile. PPS is a hyper-sulfated, natural heparinoid with minimal anticoagulant effect (1/15th that of heparin), and has significant anti-inflammatory properties [37]. It reduces CKD in several experimental mouse and rat models [22, 31, 38], and reduces inflammation and/or sclerosis in vascular grafts in animals and humans [26, 39]. PYR is a biguanide that inhibits intracellular OS/Infl by scavenging carbonyl compounds and hydroxyl radicals, that have been shown to reduce cellular anti-OS/Infl defenses [21]. PYR decreases intracellular OS/Infl in part by binding intracellular methylglyoxal (an AGE precursor), which is a very active oxidant formed during normal metabolism, but in excess amounts in diabetes [16, 17, 21, 40]. PYR has be shown to reduce DKD in animal models [40–42].
EN, an established standard in the clinical care for DKD with albuminuria [43]. When given as a single agent, it reduced GS and albuminuria less significantly than did PPS+PYR+EN. EN as a single drug or in combination with PPS or PYR significantly reduced RAGE below DB and non-DB levels. These data suggest that the RAAS system may be involved in the sclerosis-prone phenotype of ROP mice [44].
The fact that PPS+PYR+EN reduced GS and albuminuria below those in non-treated control ROP/Os+ controls provides critical support for a role for OS/Infl in the development of GS in mice and, perhaps, with the sclerosis-prone phenotype. Further support for the hypothesis that increased OS/Infl is a significant component of the development of GS is provided by two related studies. First, pre-anestrus hyperglycemic C57Bl6 mice have robust OS/Infl defenses and do not develop significant glomerular changes [2]. However, hyperglycemia induces a rapidly progressive inflammatory glomerular disease after menopause [9], a time at which OS/Infl defenses, including ER, have been compromised [22, 24, 25, 45, 46]. Secondly, C57Bl6 mice transgenic for SV40Tag, which induces inflammation, develop progressive glomerulosclerosis at a young age [47]. Thus, intact OS/Infl defenses may play a role in the resistance to glomerulosclerosis and CKD in mice. Finally, impaired OS/Infl defenses are part of the sclerosis-prone phenotype in mice.
SIRT1, a key member of the OS/Infl defense system, may protect against GS by suppressing downstream TGFβ1 signaling [48, 49]. Sirt1 mRNA levels are reduced in DB and aged C57Bl6 mice [50] and humans [8]. The current study provides evidence that SIRT1, while it is reduced in hyperglycemic ROP Os/+ mice, their levels can be increased with drugs. Since SIRT1 levels in the PPS+PYR+EN group exceeded those in the non-treated litter-mate controls (non-DB), suggests that reduced SIRT1 levels may be an important part of the sclerosis phenotype and that intact SIRT1 levels may provide protect against hyperglycemia-induced GS. Finally, it also suggests that intact SIRT1 levels may modify the sclerosis-prone phenotype.
AGER1, another important member of the anti-oxidant defense system [51], is present both on the cell surface where it serves as an AGE receptor, and in the endoplasmic reticulum, where it is a part of the oligosaccharide transferase complex (OST) [52]. Cell surface AGER1 acts in synergism with SIRT1. While AGER1 protein levels were decreased in DB, all treatments except EN increased AGER1 to non-DB levels. This may have contributed to decreased OS/Infl and GS.
Nrf2 and ERα mRNA levels, both of which are redox-sensitive transcription factors, were reduced in both non-DB and DB ROP OS/+ mice. Nrf2 is an essential component in the transactivation of genes containing antioxidant-response elements in their regulatory regions, which allows the coordinated expression of genes associated with protection against OS/Infl [53, 54]. PPS+PYR+EN enhanced the expression of Nrf2 in the DB kidney to levels above those in non-DB. ERα mRNA levels were markedly suppressed in DB and while they were increased by EN+PYR, they were restored to non-DB levels only by EN+PYR+PPS. The potential clinical relevance of this finding is that reduced ER function in diabetic patients with CKD was restored by reducing inflammation [55–57]. Thus, decreased OS/Infl defenses, a component of the sclerosis-prone phenotype, appear to be modifiable.
The multiple linear regression analysis corroborated the contribution of OS/Infl factors (sMG and sTNFR1 levels and TGFβ1, RAGE and MCP1 mRNA levels) as being contributing factors in the pathogenesis of albuminuria. It also suggests that these factors might prove useful in assessing the utility of interventions in early DKD.
This pilot study has several limitations including the relatively short follow-up period, the absence of a treated non-DB control or male mice, the presence of tissue and OS/Infl marker studies only at study end, and the small size of the study groups. Since the relatively large amount of oxidant AGEs in the usual animal chow, was not modified in this study [17, 18], a more pronounced effect of dietary oxidants on OS/Infl might have been seen if their restriction, by either low AGE food or drugs that bind AGEs in food had been utilized [55, 56]. These pilot data suggest that additional drugs that might preserve OS/Infl defenses should be explored.
In summary, the present pilot study in hyperglycemic and naive ROP/Os+ mice demonstrates that control of OS/Infl with currently available and relatively inexpensive and safe agents can modulate established GS, and DKD, decrease the sclerosis-prone phenotype in mice. The relevance of these data to humans with progressive DKD and a detailed study of the mechanisms involved, or other types of genetically determined CKD remains to be determined.
Supporting information
(PPTX)
Data Availability
All relevant data are within the paper.
Funding Statement
The author(s) received no specific funding for this work.
References
- 1.Sun JK, Keenan HA, Cavallerano JD, Asztalos BF, Schaefer EJ, Sell DR, et al. Protection from retinopathy and other complications in patients with type 1 diabetes of extreme duration: the joslin 50-year medalist study. Diabetes Care. 2011;34(4):968–74. 10.2337/dc10-1675 ; PubMed Central PMCID: PMCPMC3064059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Esposito C, He CJ, Striker GE, Zalups RK, Striker LJ. Nature and severity of the glomerular response to nephron reduction is strain-dependent in mice. Am J Pathol. 1999;154(3):891–7. 10.1016/S0002-9440(10)65336-9 ; PubMed Central PMCID: PMCPMC1866424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.He C, Esposito C, Phillips C, Zalups RK, Henderson DA, Striker GE, et al. Dissociation of glomerular hypertrophy, cell proliferation, and glomerulosclerosis in mouse strains heterozygous for a mutation (Os) which induces a 50% reduction in nephron number. J Clin Invest. 1996;97(5):1242–9. 10.1172/JCI118539 ; PubMed Central PMCID: PMCPMC507177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Skupien J, Warram JH, Smiles A, Galecki A, Stanton RC, Krolewski AS. Improved glycemic control and risk of ESRD in patients with type 1 diabetes and proteinuria. J Am Soc Nephrol. 2014;25(12):2916–25. 10.1681/ASN.2013091002 ; PubMed Central PMCID: PMCPMC4243344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Igo RP Jr., Iyengar SK, Nicholas SB, Goddard KA, Langefeld CD, Hanson RL, et al. Genomewide linkage scan for diabetic renal failure and albuminuria: the FIND study. Am J Nephrol. 2011;33(5):381–9. 10.1159/000326763 ; PubMed Central PMCID: PMCPMC3078269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.El-Meanawy A, Schelling JR, Iyengar SK, Hayden P, Barathan S, Goddard K, et al. Identification of nephropathy candidate genes by comparing sclerosis-prone and sclerosis-resistant mouse strain kidney transcriptomes. BMC Nephrol. 2012;13:61 10.1186/1471-2369-13-61 ; PubMed Central PMCID: PMCPMC3502501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stinghen AE, Massy ZA, Vlassara H, Striker GE, Boullier A. Uremic Toxicity of Advanced Glycation End Products in CKD. J Am Soc Nephrol. 2016;27(2):354–70. 10.1681/ASN.2014101047 ; PubMed Central PMCID: PMCPMC4731113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vlassara H, Striker GE. AGE restriction in diabetes mellitus: a paradigm shift. Nat Rev Endocrinol. 2011;7(9):526–39. 10.1038/nrendo.2011.74 ; PubMed Central PMCID: PMCPMC3708644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wu J, Zhang R, Torreggiani M, Ting A, Xiong H, Striker GE, et al. Induction of diabetes in aged C57B6 mice results in severe nephropathy: an association with oxidative stress, endoplasmic reticulum stress, and inflammation. Am J Pathol. 2010;176(5):2163–76. 10.2353/ajpath.2010.090386 ; PubMed Central PMCID: PMCPMC2861082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Koschinsky T, He CJ, Mitsuhashi T, Bucala R, Liu C, Buenting C, et al. Orally absorbed reactive glycation products (glycotoxins): an environmental risk factor in diabetic nephropathy. Proc Natl Acad Sci U S A. 1997;94(12):6474–9. ; PubMed Central PMCID: PMCPMC21074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vlassara H, Torreggiani M, Post JB, Zheng F, Uribarri J, Striker GE. Role of oxidants/inflammation in declining renal function in chronic kidney disease and normal aging. Kidney Int Suppl. 2009;(114):S3–11. 10.1038/ki.2009.401 . [DOI] [PubMed] [Google Scholar]
- 12.Freedman BI, Bostrom M, Daeihagh P, Bowden DW. Genetic factors in diabetic nephropathy. Clin J Am Soc Nephrol. 2007;2(6):1306–16. 10.2215/CJN.02560607 . [DOI] [PubMed] [Google Scholar]
- 13.Lee HB, Yu MR, Yang Y, Jiang Z, Ha H. Reactive oxygen species-regulated signaling pathways in diabetic nephropathy. J Am Soc Nephrol. 2003;14(8 Suppl 3):S241–5. . [DOI] [PubMed] [Google Scholar]
- 14.Navarro-Gonzalez JF, Mora-Fernandez C, Muros de Fuentes M, Garcia-Perez J. Inflammatory molecules and pathways in the pathogenesis of diabetic nephropathy. Nat Rev Nephrol. 2011;7(6):327–40. 10.1038/nrneph.2011.51 . [DOI] [PubMed] [Google Scholar]
- 15.Singh DK, Winocour P, Farrington K. Oxidative stress in early diabetic nephropathy: fueling the fire. Nat Rev Endocrinol. 2011;7(3):176–84. 10.1038/nrendo.2010.212 . [DOI] [PubMed] [Google Scholar]
- 16.Yang CW, Vlassara H, Peten EP, He CJ, Striker GE, Striker LJ. Advanced glycation end products up-regulate gene expression found in diabetic glomerular disease. Proc Natl Acad Sci U S A. 1994;91(20):9436–40. ; PubMed Central PMCID: PMCPMC44827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zheng F, He C, Cai W, Hattori M, Steffes M, Vlassara H. Prevention of diabetic nephropathy in mice by a diet low in glycoxidation products. Diabetes Metab Res Rev. 2002;18(3):224–37. 10.1002/dmrr.283 . [DOI] [PubMed] [Google Scholar]
- 18.Cai W, He JC, Zhu L, Chen X, Zheng F, Striker GE, et al. Oral glycotoxins determine the effects of calorie restriction on oxidant stress, age-related diseases, and lifespan. Am J Pathol. 2008;173(2):327–36. 10.2353/ajpath.2008.080152 ; PubMed Central PMCID: PMCPMC2475771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cornacchia F, Fornoni A, Plati AR, Thomas A, Wang Y, Inverardi L, et al. Glomerulosclerosis is transmitted by bone marrow-derived mesangial cell progenitors. J Clin Invest. 2001;108(11):1649–56. 10.1172/JCI12916 ; PubMed Central PMCID: PMCPMC200982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yang HC, Rossini M, Ma LJ, Zuo Y, Ma J, Fogo AB. Cells derived from young bone marrow alleviate renal aging. J Am Soc Nephrol. 2011;22(11):2028–36. 10.1681/ASN.2010090982 ; PubMed Central PMCID: PMCPMC3231782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Voziyan PA, Hudson BG. Pyridoxamine as a multifunctional pharmaceutical: targeting pathogenic glycation and oxidative damage. Cell Mol Life Sci. 2005;62(15):1671–81. 10.1007/s00018-005-5082-7 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu J, Guan TJ, Zheng S, Grosjean F, Liu W, Xiong H, et al. Inhibition of inflammation by pentosan polysulfate impedes the development and progression of severe diabetic nephropathy in aging C57B6 mice. Lab Invest. 2011;91(10):1459–71. 10.1038/labinvest.2011.93 . [DOI] [PubMed] [Google Scholar]
- 23.Pereira-Simon S, Rubio GA, Xia X, Cai W, Choi R, Striker GE, et al. Inhibition of Advanced Glycation End Products (AGEs) Accumulation by Pyridoxamine Modulates Glomerular and Mesangial Cell Estrogen Receptor alpha Expression in Aged Female Mice. PLoS One. 2016;11(7):e0159666 10.1371/journal.pone.0159666 ; PubMed Central PMCID: PMCPMC4948910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zheng F, Striker GE, Esposito C, Lupia E, Striker LJ. Strain differences rather than hyperglycemia determine the severity of glomerulosclerosis in mice. Kidney Int. 1998;54(6):1999–2007. 10.1046/j.1523-1755.1998.00219.x . [DOI] [PubMed] [Google Scholar]
- 25.Cai W, He JC, Zhu L, Chen X, Wallenstein S, Striker GE, et al. Reduced oxidant stress and extended lifespan in mice exposed to a low glycotoxin diet: association with increased AGER1 expression. Am J Pathol. 2007;170(6):1893–902. 10.2353/ajpath.2007.061281 ; PubMed Central PMCID: PMCPMC1899464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Illien-Junger S, Grosjean F, Laudier DM, Vlassara H, Striker GE, Iatridis JC. Combined anti-inflammatory and anti-AGE drug treatments have a protective effect on intervertebral discs in mice with diabetes. PLoS One. 2013;8(5):e64302 10.1371/journal.pone.0064302 ; PubMed Central PMCID: PMCPMC3656842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schwedler SB, Gilbert T, Moreau E, Striker LJ, Merlet-Benichou C, Striker GE. Nephrotoxin exposure in utero reduces glomerular number in sclerosis-prone but not sclerosis-resistant mice. Kidney Int. 1999;56(5):1683–90. 10.1046/j.1523-1755.1999.00749.x . [DOI] [PubMed] [Google Scholar]
- 28.Ruggenenti P, Cravedi P, Remuzzi G. The RAAS in the pathogenesis and treatment of diabetic nephropathy. Nat Rev Nephrol. 2010;6(6):319–30. 10.1038/nrneph.2010.58 . [DOI] [PubMed] [Google Scholar]
- 29.Zheng F, Cornacchia F, Schulman I, Banerjee A, Cheng QL, Potier M, et al. Development of albuminuria and glomerular lesions in normoglycemic B6 recipients of db/db mice bone marrow: the role of mesangial cell progenitors. Diabetes. 2004;53(9):2420–7. . [DOI] [PubMed] [Google Scholar]
- 30.Zheng F, Plati AR, Potier M, Schulman Y, Berho M, Banerjee A, et al. Resistance to glomerulosclerosis in B6 mice disappears after menopause. Am J Pathol. 2003;162(4):1339–48. 10.1016/S0002-9440(10)63929-6 ; PubMed Central PMCID: PMCPMC1851217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bobadilla NA, Tack I, Tapia E, Sanchez-Lozada LG, Santamaria J, Jimenez F, et al. Pentosan polysulfate prevents glomerular hypertension and structural injury despite persisting hypertension in 5/6 nephrectomy rats. J Am Soc Nephrol. 2001;12(10):2080–7. . [DOI] [PubMed] [Google Scholar]
- 32.Wu J, Guan TJ, Zheng S, Grosjean F, Liu W, Xiong H, et al. Inhibition of inflammation by pentosan polysulfate impedes the development and progression of severe diabetic nephropathy in aging C57B6 mice. Lab Invest. 2011. 10.1038/labinvest.2011.93 . [DOI] [PubMed] [Google Scholar]
- 33.Moore DS, McCabe GP. The Introduction to the Practice of Statistics (3rd ed.). Company; WHFa, editor. New York: 1999. [Google Scholar]
- 34.Chen S, Jim B, Ziyadeh FN. Diabetic nephropathy and transforming growth factor-beta: transforming our view of glomerulosclerosis and fibrosis build-up. Semin Nephrol. 2003;23(6):532–43. . [DOI] [PubMed] [Google Scholar]
- 35.Chow FY, Nikolic-Paterson DJ, Ozols E, Atkins RC, Rollin BJ, Tesch GH. Monocyte chemoattractant protein-1 promotes the development of diabetic renal injury in streptozotocin-treated mice. Kidney Int. 2006;69(1):73–80. 10.1038/sj.ki.5000014 . [DOI] [PubMed] [Google Scholar]
- 36.He C, Zalups RK, Henderson DA, Striker GE, Striker LJ. Molecular analysis of spontaneous glomerulosclerosis in Os/+ mice, a model with reduced nephron mass. Am J Physiol. 1995;269(2 Pt 2):F266–73. 10.1152/ajprenal.1995.269.2.F266 . [DOI] [PubMed] [Google Scholar]
- 37.Striker GE, Lupia E, Elliot S, Zheng F, McQuinn C, Blagg C, et al. Glomerulosclerosis, arteriosclerosis, and vascular graft stenosis: treatment with oral heparinoids. Kidney Int Suppl. 1997;63:S120–3. . [PubMed] [Google Scholar]
- 38.Schwedler SB, Bobadilla N, Striker LJ, Vaamonde CA, Herrera-Acosta J, Striker GE. Pentosan polysulfate treatment reduces cyclosporine-induced nephropathy in salt-depleted rats. Transplantation. 1999;68(10):1583–8. . [DOI] [PubMed] [Google Scholar]
- 39.Schuchman EH, Ge Y, Lai A, Borisov Y, Faillace M, Eliyahu E, et al. Pentosan polysulfate: a novel therapy for the mucopolysaccharidoses. PLoS One. 2013;8(1):e54459 10.1371/journal.pone.0054459 ; PubMed Central PMCID: PMCPMC3554761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zheng F, Zeng YJ, Plati AR, Elliot SJ, Berho M, Potier M, et al. Combined AGE inhibition and ACEi decreases the progression of established diabetic nephropathy in B6 db/db mice. Kidney Int. 2006;70(3):507–14. 10.1038/sj.ki.5001578 . [DOI] [PubMed] [Google Scholar]
- 41.Nagaraj RH, Sarkar P, Mally A, Biemel KM, Lederer MO, Padayatti PS. Effect of pyridoxamine on chemical modification of proteins by carbonyls in diabetic rats: characterization of a major product from the reaction of pyridoxamine and methylglyoxal. Arch Biochem Biophys. 2002;402(1):110–9. 10.1016/S0003-9861(02)00067-X . [DOI] [PubMed] [Google Scholar]
- 42.Brouwers O, Niessen PM, Miyata T, Ostergaard JA, Flyvbjerg A, Peutz-Kootstra CJ, et al. Glyoxalase-1 overexpression reduces endothelial dysfunction and attenuates early renal impairment in a rat model of diabetes. Diabetologia. 2014;57(1):224–35. 10.1007/s00125-013-3088-5 . [DOI] [PubMed] [Google Scholar]
- 43.Nagle RB, Kohnen PW, Bulger RE, Striker GE, Benditt EP. Ultrastructure of human renal obsolescent glomeruli. Lab Invest. 1969;21(6):519–26. . [PubMed] [Google Scholar]
- 44.Jefferson JA, Shankland SJ, Pichler RH. Proteinuria in diabetic kidney disease: a mechanistic viewpoint. Kidney Int. 2008;74(1):22–36. 10.1038/ki.2008.128 . [DOI] [PubMed] [Google Scholar]
- 45.He CJ, Striker LJ, Tsokos M, Yang CW, Peten EP, Striker GE. Relationships between mesangial cell proliferation and types I and IV collagen mRNA levels in vitro. Am J Physiol. 1995;269(3 Pt 1):C554–62. 10.1152/ajpcell.1995.269.3.C554 . [DOI] [PubMed] [Google Scholar]
- 46.Elliot SJ, Berho M, Korach K, Doublier S, Lupia E, Striker GE, et al. Gender-specific effects of endogenous testosterone: female alpha-estrogen receptor-deficient C57Bl/6J mice develop glomerulosclerosis. Kidney Int. 2007;72(4):464–72. 10.1038/sj.ki.5002328 . [DOI] [PubMed] [Google Scholar]
- 47.MacKay K, Striker LJ, Stauffer JW, Agodoa LY, Striker GE. Relationship of glomerular hypertrophy and sclerosis: studies in SV40 transgenic mice. Kidney Int. 1990;37(2):741–8. . [DOI] [PubMed] [Google Scholar]
- 48.Li J, Qu X, Ricardo SD, Bertram JF, Nikolic-Paterson DJ. Resveratrol inhibits renal fibrosis in the obstructed kidney: potential role in deacetylation of Smad3. Am J Pathol. 2007;177(3):1065–71. 10.2353/ajpath.2010.090923 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kume S, Haneda M, Kanasaki K, Sugimoto T, Araki S, Isshiki K, et al. SIRT1 inhibits transforming growth factor beta-induced apoptosis in glomerular mesangial cells via Smad7 deacetylation. J Biol Chem. 2007;282(1):151–8. 10.1074/jbc.M605904200 . [DOI] [PubMed] [Google Scholar]
- 50.Tikoo K, Tripathi DN, Kabra DG, Sharma V, Gaikwad AB. Intermittent fasting prevents the progression of type I diabetic nephropathy in rats and changes the expression of Sir2 and p53. FEBS Lett. 2007;581(5):1071–8. 10.1016/j.febslet.2007.02.006 . [DOI] [PubMed] [Google Scholar]
- 51.Cai W, He JC, Zhu L, Lu C, Vlassara H. Advanced glycation end product (AGE) receptor 1 suppresses cell oxidant stress and activation signaling via EGF receptor. Proc Natl Acad Sci U S A. 2006;103(37):13801–6. 10.1073/pnas.0600362103 ; PubMed Central PMCID: PMCPMC1564251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vlassara H, Striker GE. Advanced glycation endproducts in diabetes and diabetic complications. Endocrinol Metab Clin North Am. 2013;42(4):697–719. 10.1016/j.ecl.2013.07.005 . [DOI] [PubMed] [Google Scholar]
- 53.Kobayashi M, Yamamoto M. Molecular mechanisms activating the Nrf2-Keap1 pathway of antioxidant gene regulation. Antioxid Redox Signal. 2005;7(3–4):385–94. 10.1089/ars.2005.7.385 . [DOI] [PubMed] [Google Scholar]
- 54.Xu W, Hellerbrand C, Kohler UA, Bugnon P, Kan YW, Werner S, et al. The Nrf2 transcription factor protects from toxin-induced liver injury and fibrosis. Lab Invest. 2008;88(10):1068–78. 10.1038/labinvest.2008.75 . [DOI] [PubMed] [Google Scholar]
- 55.Vlassara H, Cai W, Chen X, Serrano EJ, Shobha MS, Uribarri J, et al. Managing chronic inflammation in the aging diabetic patient with CKD by diet or sevelamer carbonate: a modern paradigm shift. J Gerontol A Biol Sci Med Sci. 2012;67(12):1410–6. 10.1093/gerona/gls195 ; PubMed Central PMCID: PMCPMC3636675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yubero-Serrano EM, Woodward M, Poretsky L, Vlassara H, Striker GE, Group AG-lS. Effects of sevelamer carbonate on advanced glycation end products and antioxidant/pro-oxidant status in patients with diabetic kidney disease. Clin J Am Soc Nephrol. 2015;10(5):759–66. 10.2215/CJN.07750814 ; PubMed Central PMCID: PMCPMC4422240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Vlassara H, Uribarri J, Cai W, Goodman S, Pyzik R, Post J, et al. Effects of sevelamer on HbA1c, inflammation, and advanced glycation end products in diabetic kidney disease. Clin J Am Soc Nephrol. 2012;7(6):934–42. 10.2215/CJN.12891211 ; PubMed Central PMCID: PMCPMC3362316. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PPTX)
Data Availability Statement
All relevant data are within the paper.