Abstract
Allergic rhinitis is a common disorder that is strongly linked to asthma and conjunctivitis. It is usually a long-standing condition that often goes undetected in the primary-care setting. The classic symptoms of the disorder are nasal congestion, nasal itch, rhinorrhea and sneezing. A thorough history, physical examination and allergen skin testing are important for establishing the diagnosis of allergic rhinitis. Second-generation oral antihistamines and intranasal corticosteroids are the mainstay of treatment. Allergen immunotherapy is an effective immune-modulating treatment that should be recommended if pharmacologic therapy for allergic rhinitis is not effective or is not tolerated, or if chosen by the patient. This article provides an overview of the pathophysiology, diagnosis, and appropriate management of this disorder.
Background
Rhinitis is broadly defined as inflammation of the nasal mucosa. It is a common disorder that affects up to 40% of the population [1]. Allergic rhinitis is the most common type of chronic rhinitis, affecting 10–20% of the population, and evidence suggests that the prevalence of the disorder is increasing [2]. Severe allergic rhinitis has been associated with significant impairments in quality of life, sleep and work performance [2].
In the past, allergic rhinitis was considered to be a disorder localized to the nose and nasal passages, but current evidence indicates that it may represent a component of a systemic airway disease involving the entire respiratory tract. There are a number of physiological, functional and immunological relationships between the upper (nose, nasal cavity, paranasal sinuses, Eustachian tube, pharynx and larynx) and lower (trachea, bronchial tubes, bronchioles and lungs) respiratory tracts. For example, both tracts contain a ciliated epithelium consisting of goblet cells that secrete mucous, which serves to filter the incoming air and protect structures within the airways. Furthermore, the submucosa of both the upper and lower airways includes a collection of blood vessels, mucous glands, supporting cells, nerves and inflammatory cells. Evidence has shown that allergen provocation of the upper airways not only leads to a local inflammatory response, but may also lead to inflammatory processes in the lower airways, and this is supported by the fact that rhinitis and asthma frequently coexist. Therefore, allergic rhinitis and asthma appear to represent a combined airway inflammatory disease, and this needs to be considered to ensure the optimal assessment and management of patients with allergic rhinitis [1, 3].
Comprehensive and widely-accepted Canadian guidelines for the diagnosis and treatment of allergic rhinitis were published in 2007 [1]. This article provides an overview and update of the recommendations provided in these guidelines as well as a review of current literature related to the pathophysiology, diagnosis, and appropriate management of allergic rhinitis.
Pathophysiology
In allergic rhinitis, numerous inflammatory cells, including mast cells, CD4-positive T cells, B cells, macrophages, and eosinophils, infiltrate the nasal lining upon exposure to an inciting allergen (most commonly airborne dust mite fecal particles, cockroach residues, animal dander, moulds, and pollens). In allergic individuals, the T cells infiltrating the nasal mucosa are predominantly T helper 2 (Th2) in nature and release cytokines (e.g., interleukin [IL]-3, IL-4, IL-5, and IL-13) that promote immunoglobulin E (IgE) production by plasma cells. Crosslinking of IgE bound to mast cells by allergens, in turn, triggers the release of mediators, such as histamine and leukotrienes, that are responsible for arteriolar dilation, increased vascular permeability, itching, rhinorrhea, mucous secretion, and smooth muscle contraction in the lung [1, 2]. The mediators and cytokines released during the early phase of an immune response to an inciting allergen trigger a further cellular inflammatory response over the next 4–8 h (late-phase inflammatory response) which results in recurrent symptoms (usually nasal congestion) that often persist [1, 4].
Classification
Rhinitis is classified into one of the following categories according to etiology: IgE-mediated (allergic), autonomic, infectious and idiopathic (unknown). Although the focus of this article is allergic rhinitis, a brief description of the other forms of rhinitis is provided in Table 1.
Table 1.
Etiological classification of rhinitis [1]
| Description | |
|---|---|
| IgE-mediated (allergic) | • IgE-mediated inflammation of the nasal mucosa, resulting in eosinophilic and Th2-cell infiltration of the nasal lining • Further classified as intermittent or persistent |
| Autonomic | • Vasomotor • Drug-induced (rhinitis medicamentosa) • Hypothyroidism • Hormonal • Non-allergic rhinitis with eosinophilia syndrome (NARES) |
| Infectious | • Precipitated by viral (most common), bacterial, or fungal infection |
| Idiopathic | • Etiology cannot be determined |
Traditionally, allergic rhinitis has been categorized as seasonal (occurs during a specific season) or perennial (occurs throughout the year). However, not all patients fit into this classification scheme. For example, some allergic triggers, such as pollen, may be seasonal in cooler climates, but perennial in warmer climates, and patients with multiple “seasonal” allergies may have symptoms throughout most of the year [4]. Therefore, allergic rhinitis is now classified according to symptom duration (intermittent or persistent) and severity (mild, moderate or severe) (see Fig. 1) [1, 5]. The Allergic Rhinitis and its Impact on Asthma (ARIA) guidelines have classified “intermittent” allergic rhinitis as symptoms that are present less than 4 days per week or for less than 4 consecutive weeks, and “persistent” allergic rhinitis as symptoms that are present more than 4 days/week and for more than 4 consecutive weeks [5]. Symptoms are classified as mild when patients have no impairment in sleep and are able to perform normal activities (including work or school). Symptoms are categorized as moderate/severe if they significantly affect sleep or activities of daily living, and/or if they are considered bothersome. It is important to classify the severity and duration of symptoms as this will guide the management approach for individual patients [1].
Fig. 1.
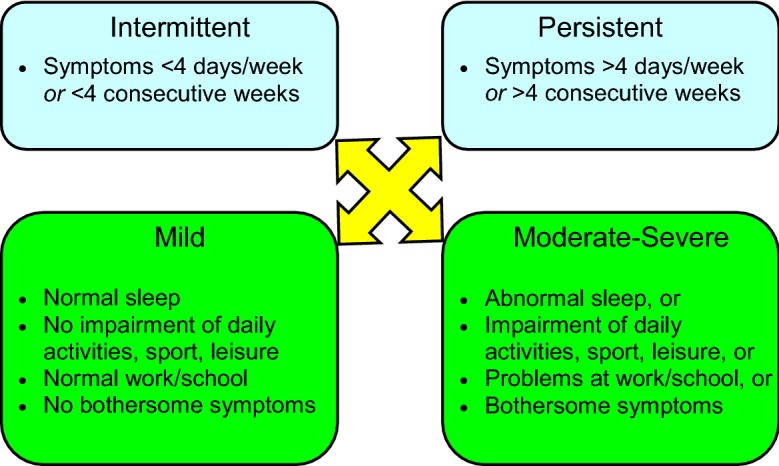
Classification of allergic rhinitis according to symptom duration and severity.
In recent years, two additional types of rhinitis have been classified that deserve some mention here—occupational rhinitis and local allergic rhinitis.
Occupational rhinitis
Occupational rhinitis is defined as an inflammatory disease of the nose characterized by intermittent or persistent symptoms that include airflow limitation, hypersecretion, sneezing and pruritus that are attributable to a particular work environment and not to stimuli encountered outside the workplace [6]. Although the overall prevalence of occupational rhinitis is unknown, high-risk professions include laboratory or food-processing workers, veterinarians, farmers and workers in various manufacturing industries [6–8]. Occupational rhinitis usually develops within the first 2 years of employment. The condition may be IgE-mediated due to allergen sensitization, or due to exposure to respiratory irritants. Symptoms may develop immediately or several hours after exposure to the inciting stimuli. Often there are associated ocular and pulmonary symptoms. An evaluation of the patient suspected of having occupational rhinitis should include the usual history and physical examination (discussed later), as well as a site visit and skin testing or in vitro testing to inhalants. Treatment primarily involves avoiding exposure to the causative agent and pharmacotherapy as needed. There is little evidence to suggest that occupational rhinitis will progress to occupational asthma with ongoing exposure [6, 8], although this is possible. Therefore, patients are generally not advised to leave their jobs if exposure cannot be eliminated but symptoms are adequately controlled.
Local allergic rhinitis
Local allergic rhinitis (LAR) is a clinical entity characterized by a localized allergic response in the nasal mucosa in the absence of evidence of systemic atopy [9–11]. By definition, patients with LAR have negative skin tests and/or in vitro tests for IgE, but have evidence of local IgE production in the nasal mucosa; these patients also react to nasal challenges with specific allergens [12].
The symptoms of LAR are similar to those provoked in patients with allergic rhinitis, and the assumption is that LAR is an IgE-mediated disease based on both clinical findings and the detection of specific IgE in the nasal mucosa. To date, there is no evidence to suggest that LAR is a precursor to allergic rhinitis since follow-up does not show the evolution to typical allergic rhinitis in these patients [13]; however, patient follow-up may not have been long enough to detect this disease evolution. The implications for treatment of LAR are not well understood at this time, although some evidence suggests that allergen immunotherapy may be effective in this type of rhinitis [9, 11].
Diagnosis and investigations
Allergic rhinitis is usually a long-standing condition that often goes undetected in the primary-care setting. Patients suffering from the disorder often fail to recognize the impact of the disorder on quality of life and functioning and, therefore, do not frequently seek medical attention. In addition, physicians fail to regularly question patients about the disorder during routine visits [1, 14]. Therefore, screening for rhinitis is recommended, particularly in asthmatic patients since studies have shown that rhinitis is present in up to 95% of patients with asthma [15–18].
A thorough history and physical examination are the cornerstones of establishing the diagnosis of allergic rhinitis (see Table 2). Allergy testing is also important for confirming that underlying allergies cause the rhinitis [1]. Referral to an allergist should be considered if the diagnosis of allergic rhinitis is in question.
Table 2.
Components of a complete history and physical examination for suspected rhinitis [1]
| History | Physical examination |
|---|---|
|
Personal
• Congestion • Nasal itch • Rhinorrhea • Sneezing • Eye involvement • Seasonality • Triggers Family • Allergy • Asthma Environmental • Pollens • Animals • Flooring/upholstery • Mould • Humidity • Tobacco exposure Medication/drug use • Beta-blockers • ASA • NSAIDs • ACE inhibitors • Hormone therapy • Recreational cocaine use Quality of life • Rhinitis-specific questionnaire Comorbidities • Asthma • Mouth breathing • Snoring ± apnea • Impaired smell or taste • Sinus involvement • Otitis media • Nasal polyps • Conjunctivitis Response to previous interventions • Avoidance measures • Saline nasal rinses • Second-generation oral antihistamines • Intranasal corticosteroids |
Outward signs
• Mouth breathing • Rubbing the nose/transverse nasal crease • Frequent sniffling and/or throat clearing • Allergic shiners (dark circles under eyes) Nose • Mucosal swelling, bleeding • Pale, thin secretions • Polyps or other structural abnormalities Ears • Generally normal • Pneumatic otoscopy to assess for Eustachian tube dysfunction • Valsalva’s maneuver to assess for fluid behind the ear drum Sinuses • Palpation of sinuses for signs of tenderness • Maxillary tooth sensitivity Posterior oropharynx • Postnasal drip • Lymphoid hyperplasia (“cobblestoning”) • Tonsillar hypertrophy Chest and skin • Atopic disease • Wheezing |
Adapted from Small et al. [1]
ASA acetylsalicylic acid, NSAIDs non-steroidal anti-inflammatory drugs, ACE angiotensin-converting enzyme
History
During the history, patients will often describe the following classic symptoms of allergic rhinitis: nasal congestion, nasal itch, rhinorrhea and sneezing. Allergic conjunctivitis (inflammation of the membrane covering the white part of the eye) is also frequently associated with allergic rhinitis and symptoms generally include redness, tearing and itching of the eyes [1].
An evaluation of the patient’s home and work/school environments is recommended to determine potential triggers of allergic rhinitis. The environmental history should focus on common and potentially relevant allergens including pollens, furred animals, textile flooring/upholstery, tobacco smoke, humidity levels at home, as well as other potential noxious substances that the patient may be exposed to at work or at home. The use of certain medications (e.g., beta-blockers, acetylsalicylic acid [ASA], non-steroidal anti-inflammatory drugs [NSAIDs], angiotensin-converting enzyme [ACE] inhibitors, and hormone therapy) as well as the recreational use of cocaine can lead to symptoms of rhinitis and, therefore, patients should be asked about current or recent medication and drug use [1].
The history should also include patient questioning regarding a family history of atopic disease, the impact of symptoms on quality of life and the presence of comorbidities such as asthma, mouth breathing, snoring, sleep apnea, sinus involvement, otitis media (inflammation of the middle ear), or nasal polyps. Patients may attribute persistent nasal symptoms to a “constant cold” and, therefore, it is also important to document the frequency and duration of “colds” [1].
Before seeking medical attention, patients often attempt using over-the-counter or other medications to manage their symptoms. Assessing patient response to such treatments may provide information that can aid in the diagnosis and subsequent management of allergic rhinitis. For example, symptom improvement with newer, second-generation antihistamines (e.g., desloratadine [Aerius], fexofenadine [Allegra], loratadine [Claritin], cetirizine [Reactine]) is strongly suggestive of an allergic etiology. However, it is important to note that response to first-generation antihistamines (e.g., brompheniramine maleate [Dimetane], chlorpheniramine maleate [Chlor-Tripolon], clemastine [Tavist-1]) does not imply an allergic etiology since the anticholinergic and sedative properties of these agents reduce rhinorrhea and may improve sleep quality regardless of whether the inflammation is allergic. Previous response to intranasal corticosteroids may also be suggestive of an allergic etiology, and likely indicates that such treatment will continue to be beneficial in the future [1].
Important elements of the history for patients with suspected allergic rhinitis are summarized in Table 2.
Physical examination
The physical examination of patients with suspected allergic rhinitis should include an assessment of outward signs, the nose, ears, sinuses, posterior oropharynx (area of the throat that is at the back of the mouth), chest and skin (see Table 2). Outward signs that may be suggestive of allergic rhinitis include: persistent mouth breathing, rubbing at the nose or an obvious transverse nasal crease, frequent sniffling or throat clearing, and allergic shiners (dark circles under the eyes that are due to nasal congestion). Examination of the nose typically reveals swelling of the nasal mucosa and pale, thin secretions. An internal endoscopic examination of the nose should also be considered to assess for structural abnormalities including septal deviation, nasal ulcerations, and nasal polyps [1].
The ears generally appear normal in patients with allergic rhinitis; however, assessment for Eustachian tube dysfunction using a pneumatic otoscope should be considered. Valsalva’s maneuver (increasing the pressure in the nasal cavity by attempting to blow out the nose while holding it shut) can also be used to assess for fluid behind the ear drum [1].
The sinus examination should include palpation of the sinuses for evidence of tenderness or tapping of the maxillary teeth with a tongue depressor for evidence of sensitivity. The posterior oropharynx should also be examined for signs of post nasal drip (mucous accumulation in the back of the nose and throat), and the chest and skin should be examined carefully for signs of concurrent asthma (e.g., wheezing) or dermatitis [1].
Diagnostic tests
Although a thorough history and physical examination are required to establish the clinical diagnosis of rhinitis, further diagnostic testing is necessary to confirm that underlying allergies cause the rhinitis. Skin-prick testing is considered the primary method for identifying specific allergic triggers of rhinitis. Skin prick testing involves placing a drop of a commercial extract of a specific allergen on the skin of the forearms or back, then pricking the skin through the drop to introduce the extract into the epidermis. Within 15–20 min, a wheal-and-flare response (an irregular blanched wheal surrounded by an area of redness) will occur if the test is positive. Testing is typically performed using the allergens relevant to the patient’s environment (e.g., pollen, animal dander, moulds and house dust mites). A reasonable alternative to skin prick testing is the use of allergen-specific IgE tests (e.g., performed by immunosorbent assay—previously performed by radioallergosorbent tests [RASTs]) that provide an in vitro measure of a patient’s specific IgE levels against particular allergens. These in vitro tests can be performed when eczema is extensive, or if the patient cannot stop antihistamine therapy to allow for testing. However, skin prick tests are generally considered to be more sensitive and cost effective than allergen-specific serum IgE tests, and have the further advantage of providing physicians and patients with immediate results [1, 14].
Treatment
The treatment goal for allergic rhinitis is relief of symptoms. Therapeutic options available to achieve this goal include avoidance measures, nasal saline irrigation, oral antihistamines, intranasal corticosteroids, combination intranasal corticosteroid/antihistamine sprays; leukotriene receptor antagonists (LTRAs), and allergen immunotherapy (see Fig. 2). Other therapies that may be useful in select patients include decongestants and oral corticosteroids. If the patient’s symptoms persist despite appropriate treatment, referral to an allergist should be considered. As mentioned earlier, allergic rhinitis and asthma appear to represent a combined airway inflammatory disease and, therefore, treatment of asthma is also an important consideration in patients with allergic rhinitis.
Fig. 2.
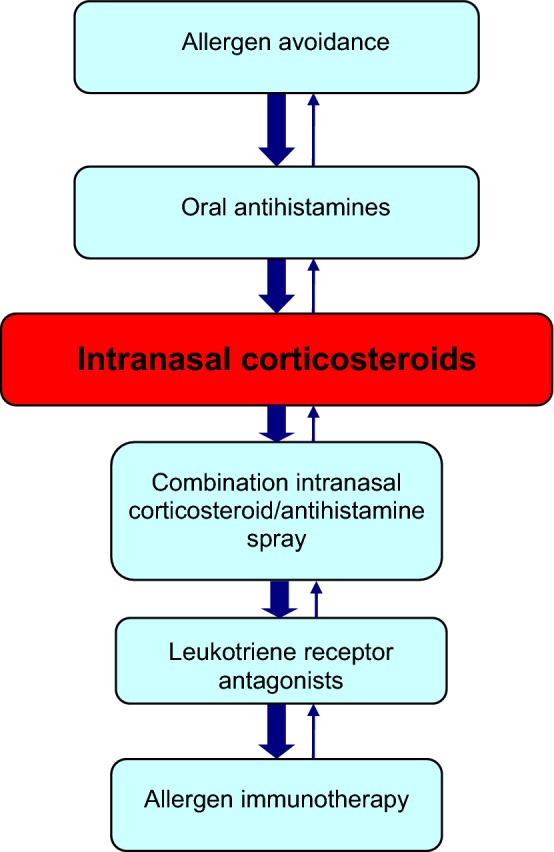
A simplified, stepwise algorithm for the treatment of allergic rhinitis. Treatments can be used individually or in any combination
Allergen avoidance
The first-line treatment of allergic rhinitis involves the avoidance of relevant allergens (e.g., house dust mites, moulds, pets, pollens) and irritants (e.g., tobacco smoke). Patients allergic to house dust mites should be instructed to use allergen-impermeable covers for bedding and to keep the relative humidity in the home below 50% (to inhibit mite growth). Pollen and outdoor mould exposure can be reduced by keeping windows closed, using window screen filters, using an air conditioner, and limiting the amount of time spent outdoors during peak pollen seasons. For patients allergic to animal dander, removal of the animal from the home is recommended and usually results in a significant reduction in symptoms within 4–6 months. However, compliance with this recommendation is poor and, therefore, the use of high-efficiency particulate air (HEPA) filters and restricting the animal from the bedroom or to the outdoors may be needed to attempt to decrease allergen levels. Measures for reducing exposure to mould allergens include cleaning with fungicides, dehumidification to less than 50%, remediation of any water damage, and HEPA filtration. These avoidance strategies can effectively improve the symptoms of allergic rhinitis, and patients should be advised to use a combination of measures for optimal results [1].
Antihistamines
The second-generation oral anti-histamines (e.g., desloratadine [Aerius], fexofenadine [Allegra], loratadine [Claritin], cetirizine [Reactine]) are the first-line pharmacological treatments recommended for all patients with allergic rhinitis. Recently, two new second-generation antihistamines—Bilastine (Blexten) and rupatadine (Rupall)—have been introduced in Canada. At present, these antihistamines are available by prescription only (see Table 3 for a list of second-generation antihistamines and their recommended dosing regimens).
Table 3.
Overview of pharmacologic treatment options for allergic rhinitis
| Usual adult dose | Usual pediatric dose | |
|---|---|---|
| Oral antihistamines (second generation) | ||
| Bilastine (Blexten) | 1 tablet (20 mg) once daily | For children ≥ 12 years of age: 1 tablet (20 mg) once daily Not recommended for children < 12 years of age |
| Cetirizine (Reactine) | 1–2 tablets (5 mg) once daily 1 tablet (10 mg) once daily |
5–10 mL (1–2 teaspoons) once daily (children’s formulation) |
| Desloratadine (Aerius) | 1 tablet (5 mg) once daily | 2.5–5 mL (0.5–1.0 teaspoon) once daily (children’s formulation) |
| Fexofenadine (Allegra) | 1 tablet (60 mg) every 12 h (12-h formulation) 1 tablet (120 mg), once daily (24-h formulation) |
Not currently indicated for children < 12 years of age |
| Loratadine (Claritin) | 1 tablet (10 mg), once daily | 5–10 mL (1–2 teaspoons) once daily (children’s formulation) |
| Rupatadine (Rupall) | 1 tablet (10 mg) once daily | For children ≥ 12 years: 1 tablet (10 mg) once daily For children 2–11 years and body weight 10–25 kg: 2.5 mL (0.5 teaspoon) once daily For children 2–11 years and body weight > 25 kg: 5 mL (1.0 teaspoon) once daily |
| Intranasal corticosteroids | ||
|---|---|---|
| Beclomethasone (Beconase) | 1–2 sprays (50 µg/spray) EN, twice daily | 1 spray (50 µg/spray) EN, twice daily |
| Budesonide (Rhinocort) | 2 sprays (64 μg/spray) EN, once daily or 1 spray EN, twice daily | 2 sprays (64 μg/spray) EN, once daily or 1 spray EN, twice daily (do not exceed 256 μg) |
| Ciclesonide (Omnaris) | 2 sprays (50 µg/spray) EN, once daily | Not indicated for children < 12 years of age |
| Fluticasone furoate (Avamys) | 2 sprays (27.5 µg/spray) EN, once daily | 1 spray (27.5 µg/spray) EN, once daily |
| Fluticasone propionate (Flonase) | 2 sprays (50 µg/spray) EN, once daily or every 12 h (for severe rhinitis) | 1–2 sprays (50 µg/spray) EN, once daily |
| Mometasone furoate (Nasonex) | 2 sprays (50 µg/spray) EN, once daily | 1 spray (50 µg/spray) EN, once daily |
| Triamcinolone acetonide (Nasacort) | 2 sprays (55 µg/spray) EN, once daily | 1 spray (55 µg/spray) EN, once daily |
| Combination intranasal corticosteroid/antihistamine nasal spray | ||
|---|---|---|
| Fluticasone propionate/azelastine hydrochloride (Dymista) | 1 spray EN, twice daily | For children ≥ 12 years of age: 1 spray EN, twice daily Not recommended for children < 12 years of age |
| Leukotriene receptor antagonists | ||
|---|---|---|
| Montelukast | 1 tablet (10 mg), once daily | Not currently approved for patients < 15 years of age |
EN each nostril
The second-generation oral anti-histamines have been found to effectively reduce sneezing, itching and rhinorrhea when taken regularly at the time of maximal symptoms or before exposure to an allergen. Although the older (first-generation) sedating antihistamines (e.g., diphenhydramine, chlorpheniramine) are also effective in relieving symptoms, they have been shown to negatively impact cognition and functioning and, therefore, they are not routinely recommended for the treatment of allergic rhinitis [1, 14].
Intranasal corticosteroids
Intranasal corticosteroids are also first-line therapeutic options for patients with mild persistent or moderate/severe symptoms and they can be used alone or in combination with oral antihistamines. When used regularly and correctly, intranasal corticosteroids effectively reduce inflammation of the nasal mucosa and improve mucosal pathology. Studies and meta-analyses have shown that intranasal corticosteroids are superior to antihistamines and leukotriene receptor antagonists in controlling the symptoms of allergic rhinitis, including nasal congestion, and rhinorrhea [19–22]. They have also been shown to improve ocular symptoms and reduce lower airway symptoms in patients with concurrent asthma and allergic rhinitis [23–25].
The intranasal corticosteroids available in Canada are shown in Table 3 and include fluticasone furoate (Avamys), beclomethasone (Beconase), fluticasone propionate (Flonase), triamcinolone acetonide (Nasacort), mometasone furoate (Nasonex), ciclesonide (Omnaris) and budesonide (Rhinocort). Since proper application of the nasal spray is required for optimal clinical response, patients should be counseled on the appropriate use of these intranasal devices. Ideally, intranasal corticosteroids are best started just prior to exposure to relevant allergens and, because their peak effect may take several days to develop, they should be used regularly [4].
The most common side effects of intranasal corticosteroids are nasal irritation and stinging. However, these side effects can usually be prevented by aiming the spray slightly away from the nasal septum [1]. Evidence suggests that intranasal beclomethasone and triamcinolone, but not other intranasal corticosteroids, may slow growth in children compared to placebo. However, long-term studies examining the impact of usual doses of intranasal beclomethasone on growth are lacking [26–29].
It is important to note that most patients with allergic rhinitis presenting to their primary-care physician have moderate-to-severe symptoms and will require an intranasal corticosteroid. Bousquet et al. [30] noted improved outcomes in patients with moderate-to-severe symptoms treated with a combination of these agents.
Combination intranasal corticosteroid and antihistamine nasal spray
If intranasal corticosteroids are not effective, a combination corticosteroid/antihistamine spray can be tried. Combination fluticasone propionate/azelastine hydrochloride (Dymista) is now available in Canada. This combination spray has been shown to be more effective than the individual components with a safety profile similar to intranasal corticosteroids [31–34].
Leukotriene receptor antagonists (LTRAs)
The LTRAs montelukast and zafirlukast are also effective in the treatment of allergic rhinitis; however, they do not appear to be as effective as intranasal corticosteroids [35–37]. Although one short-term study found the combination of LTRAs and antihistamines to be as effective as intranasal corticosteroids [38], longer-term studies have found intranasal corticosteroids to be more effective than the combination for reducing nighttime and nasal symptoms [20, 39]. It is important to note that in Canada, montelukast is the only LTRA indicated for the treatment of allergic rhinitis in adults.
LTRAs should be considered when oral antihistamines, intranasal corticosteroids and/or combination corticosteroid/antihistamine sprays are not well tolerated or are ineffective in controlling the symptoms of allergic rhinitis. If combination pharmacological therapy with oral antihistamines, intranasal corticosteroids, combination corticosteroid/antihistamine sprays and LTRAs is not effective or is not tolerated, then allergen immunotherapy should be considered [1, 14].
Allergen immunotherapy
Allergen immunotherapy involves the subcutaneous administration of gradually increasing quantities of the patient’s relevant allergens until a dose is reached that is effective in inducing immunologic tolerance to the allergen (see Allergen-specific Immunotherapy article in this supplement). Allergen immunotherapy is an effective treatment for allergic rhinitis, particularly for patients with intermittent (seasonal) allergic rhinitis caused by pollens, including tree, grass and ragweed pollens [40–43]. It has also been shown to be effective for the treatment of allergic rhinitis caused by house dust mites, Alternaria, cockroach, and cat and dog dander (although it should be noted that therapeutic doses of dog allergen are difficult to attain with the allergen extracts available in Canada). Allergen immunotherapy should be reserved for patients in whom optimal avoidance measures and pharmacotherapy are insufficient to control symptoms or are not well tolerated. Since this form of therapy carries the risk of anaphylactic reactions, it should only be prescribed by physicians who are adequately trained in the treatment of allergy and who are equipped to manage possible life-threatening anaphylaxis [1].
Evidence suggests that at least 3 years of allergen-specific immunotherapy provides beneficial effects in patients with allergic rhinitis that can persist for several years after discontinuation of therapy [44, 45]. In Canada, most allergists consider stopping immunotherapy after 5 years of adequate treatment. Immunotherapy may also reduce the risk for the future development of asthma in children with allergic rhinitis [41].
Typically, allergen immunotherapy is given on a perennial basis with weekly incremental increases in dose over the course of 6–8 months, followed by maintenance injections of the maximum tolerated dose every 3–4 weeks for 3–5 years. After this period, many patients experience a prolonged, protective effect and, therefore, consideration can be given to stopping therapy. Pre-seasonal preparations that are administered on an annual basis are also available [1, 14].
Sublingual immunotherapy is a way of desensitizing patients and involves placing a tablet of allergen extract under the tongue until it is dissolved. It is currently available for the treatment of grass and ragweed allergy, as well as house dust mite-induced allergic rhinitis (with or without conjunctivitis). At present, four sublingual tablet immunotherapy products are available in Canada: Oralair®, Grastek®, Ragwitek® and Acarizax™ [46–49]. The sublingual route of immunotherapy offers multiple potential benefits over the subcutaneous route including the comfort of avoiding injections, the convenience of home administration, and a favourable safety profile. Like subcutaneous immunotherapy, sublingual immunotherapy is indicated for those with allergic rhinitis who have not responded to or tolerated conventional pharmacotherapy, or who are adverse to the use of these conventional treatments.
The most common side effects of sublingual immunotherapy are local reactions such as oral pruritus, throat irritation, and ear pruritus [42]. These symptoms typically resolve after the 1st week of therapy. There is a very small risk of more severe systemic allergic reactions with this type of immunotherapy and, therefore, some allergists may offer the patient an epinephrine auto-injector in case a reaction occurs at home. The risk of systemic allergic reactions is much lower with sublingual immunotherapy compared to traditional injections [42].
Similar to subcutaneous immunotherapy, sublingual immunotherapy is contraindicated in patients with severe, unstable or uncontrolled asthma. It should ideally be avoided in patients on beta-blocker therapy as well as in those with active oral inflammation or sores [46–50]. Sublingual immunotherapy should only be administered using the Health Canada approved products discussed above.
A simplified, stepwise algorithm for the treatment of allergic rhinitis is provided in Fig. 2. Note that mild, intermittent allergic rhinitis can generally be managed effectively with avoidance measures and oral antihistamines. However, as mentioned earlier, most patients presenting with allergic rhinitis have moderate-to-severe symptoms and, therefore, will require a trial of intranasal corticosteroids.
Other therapeutic options
Oral and intranasal decongestants (e.g., pseudoephedrine, phenylephrine) are useful for relieving nasal congestion in patients with allergic rhinitis. However, the side-effect profile associated with oral decongestants (i.e., agitation, insomnia, headache, palpitations) may limit their long-term use. Furthermore, these agents are contraindicated in patients with uncontrolled hypertension and severe coronary artery disease. Prolonged use of intranasal decongestants carries the risk of rhinitis medicamentosa (rebound nasal congestion) and, therefore, these agents should not be used for more than 3–5 days [51]. Oral corticosteroids have also been shown to be effective in patients with severe allergic rhinitis that is refractory to treatment with oral antihistamines and intranasal corticosteroids [1, 4].
Although not as effective as intranasal corticosteroids, intranasal sodium cromoglycate (Cromolyn) has been shown to reduce sneezing, rhinorrhea and nasal itching and is, therefore, a reasonable therapeutic option for some patients. The anti-IgE antibody, omalizumab, has also been shown to be effective in seasonal allergic rhinitis and asthma [1], however, it is not currently approved for the treatment of allergic rhinitis.
Surgical therapy may be helpful for select patients with rhinitis, polyposis, or chronic sinus disease that is refractory to medical treatment. Most surgical interventions can be performed under local anesthesia in an office or outpatient setting [1].
It is important to note that allergic rhinitis may worsen during pregnancy and, as a result, may necessitate pharmacologic treatment. The benefit-to-risk ratio of pharmacological agents for allergic rhinitis needs to be considered before recommending any medical therapy to pregnant women. Intranasal sodium cromoglycate can be used as a first-line therapy for allergic rhinitis in pregnancy since no teratogenic effects have been noted with the cromones in humans or animals. Antihistamines may also be considered for allergic rhinitis in pregnancy. Starting or increasing allergen immunotherapy during pregnancy is not recommended because of the risk of anaphylaxis to the fetus. However, maintenance doses are considered to be safe and effective during pregnancy [1].
Complementary and alternative medicines (CAM)
Given the popularity of complementary and alternative medicines (CAM) in the general population, it is reasonable for physicians to ask patients about their use of CAM in a nonjudgmental manner. Given the limited number of well-designed clinical trials examining the efficacy of CAM in allergic rhinitis, it is difficult for clinicians to evaluate these therapies and provide guidance. Nonetheless, since there will be patients who wish to pursue CAM for the management of allergic rhinitis, it is advisable to provide some information about these therapies including a discussion of the lack of high-quality studies evaluating some of these therapies.
Various CAM have been used for the management of allergic rhinitis, including traditional Chinese medicines, acupuncture, homeopathy, and herbal therapies [52]. In a number of studies, acupuncture has been shown to provide modest benefits for patients with allergic rhinitis [52, 53]. However, acupuncture is time consuming.
Conclusions
Allergic rhinitis is a common disorder that can significantly impact patient quality of life. The diagnosis is made through a comprehensive history and physical examination. Further diagnostic testing using skin-prick tests or allergen-specific IgE tests is usually required to confirm that underlying allergies cause the rhinitis. The therapeutic options available for the treatment of allergic rhinitis are effective in managing symptoms and are generally safe and well-tolerated. Second-generation oral antihistamines and intranasal corticosteroids are the mainstay of treatment for the disorder. Allergen immunotherapy as well as other medications such as decongestants and oral corticosteroids may be useful in select cases.
Key take-home messages
Allergic rhinitis is linked strongly with asthma and conjunctivitis.
Allergen skin testing is the best diagnostic test to confirm allergic rhinitis.
Intranasal corticosteroids are the mainstay of treatment for most patients that present to physicians with allergic rhinitis.
Allergen immunotherapy is an effective immune-modulating treatment that should be recommended if pharmacologic therapy for allergic rhinitis is not effective or is not tolerated.
Declarations
Authors’ contributions All authors wrote and/or edited sections of the manuscript. All authors read and approved the final manuscript.
Acknowledgements
This article is an update to the Allergic Rhinitis article that originally appeared in the supplement entitled, Practical Guide to Allergy and Immunology in Canada, which was published in Allergy, Asthma & Clinical Immunology in 2011 (available at: https://aacijournal.biomedcentral.com/articles/supplements/volume-7-supplement-1).
The authors would like to thank Julie Tasso for her editorial services and assistance in the preparation of this manuscript.
Competing interests
Dr. Peter Small has received consulting fees or honoraria from AstraZeneca, Teva and GlaxoSmithKline.
Dr. Paul Keith has received consulting fees or honoraria from ALK, Aralez, AstraZeneca, Boehringer Ingelheim, CSL Behring, GlaxoSmithKline, Meda, Merck, Mylan, Novartis, Nycomed, Pediapharm, Sanofi, Shire, Stallergenes Greer and Teva. He has also participated in clinical trials as an investigator with AstraZeneca, CSL Behring, Novartis, Shire, and Merck.
Dr. Harold Kim is Vice President of the Canadian Society of Allergy and Clinical Immunology, Past President of the Canadian Network for Respiratory Care, and Co-chief Editor of Allergy, Asthma and Clinical Immunology. He has received consulting fees and honoraria for continuing medical education from AstraZeneca, Aralez, Boehringer Ingelheim, CSL Behring, Kaleo, Merck, Novartis, Pediapharm, Sanofi, Shire and Teva.
Availability of data and materials
Data sharing not applicable to this article as no datasets were generated or analyzed during the development of this review.
Consent for publication
Not applicable.
Ethics approval and consent to participate
Ethics approval and consent to participate are not applicable to this review article.
Funding
Publication of this supplement has been supported by AstraZeneca, Boehringer Ingelheim, CSL Behring Canada Inc., MEDA Pharmaceuticals Ltd., Merck Canada Inc., Pfizer Canada Inc., Shire Pharma Canada ULC, Stallergenes Greer Canada, Takeda Canada, Teva Canada Innovation, Aralez Tribute and Pediapharm.
About this supplement
This article has been published as part of Allergy, Asthma & Clinical Immunology Volume 14 Supplement 2, 2018: Practical guide for allergy and immunology in Canada 2018. The full contents of the supplement are available online at https://aacijournal.biomedcentral.com/articles/supplements/volume-14-supplement-2.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviations
- Th2
T helper 2
- IL
interleukin
- IgE
immunoglobulin E
- LAR
local allergic rhinitis
- ACE
angiotensin-converting enzyme
- ASA
acetylsalicylic acid
- NSAIDs
non-steroidal anti-inflammatory drugs
- LTRAs
leukotriene receptor antagonists
- CAM
complementary and alternative medicines
- ARIA
Allergic Rhinitis and its Impact on Asthma
- EN
each nostril
References
- 1.Small P, Frenkiel S, Becker A, Boisvert P, Bouchard J, Carr S, Cockcroft D, Denburg J, Desrosiers M, Gall R, Hamid Q, Hébert J, Javer A, Keith P, Kim H, Lavigne F, Lemièr C, Massoud E, Payton K, Schellenberg B, Sussman G, Tannenbaum D, Watson W, Witterick I, Wright E, The Canadian Rhinitis Working Group Rhinitis: a practical and comprehensive approach to assessment and therapy. J Otolaryngol. 2007;36(Suppl 1):S5–S27. doi: 10.2310/7070.2006.X002. [DOI] [Google Scholar]
- 2.Dykewicz MS, Hamilos DL. Rhinitis and sinusitis. J Allergy Clin Immunol. 2010;125:S103–S115. doi: 10.1016/j.jaci.2009.12.989. [DOI] [PubMed] [Google Scholar]
- 3.Bourdin A, Gras D, Vachier I, Chanez P. Upper airway 1: allergic rhinitis and asthma: united disease through epithelial cells. Thorax. 2009;64(11):999–1004. doi: 10.1136/thx.2008.112862. [DOI] [PubMed] [Google Scholar]
- 4.Lee P, Mace S. An approach to allergic rhinitis. Allergy Rounds. 2009;1:1. [Google Scholar]
- 5.Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, Togias A, Zuberbier T, Baena-Cagnani CE, Canonica GW, van Weel C, Agache I, Aït-Khaled N, Bachert C, Blaiss MS, Bonini S, Boulet LP, Bousquet PJ, Camargos P, Carlsen KH, Chen Y, Custovic A, Dahl R, Demoly P, Douagui H, Durham SR, van Wijk RG, Kalayci O, Kaliner MA, Kim YY, Kowalski ML, Kuna P, Le LT, Lemiere C, Li J, Lockey RF, Mavale-Manuel S, Meltzer EO, Mohammad Y, Mullol J, Naclerio R, O’Hehir RE, Ohta K, Ouedraogo S, Palkonen S, Papadopoulos N, Passalacqua G, Pawankar R, Popov TA, Rabe KF, Rosado-Pinto J, Scadding GK, Simons FE, Toskala E, Valovirta E, van Cauwenberge P, Wang DY, Wickman M, Yawn BP, Yorgancioglu A, Yusuf OM, Zar H, Annesi-Maesano I, Bateman ED, Ben Kheder A, Boakye DA, Bouchard J, Burney P, Busse WW, Chan-Yeung M, Chavannes NH, Chuchalin A, Dolen WK, Emuzyte R, Grouse L, Humbert M, Jackson C, Johnston SL, Keith PK, Kemp JP, Klossek JM, Larenas-Linnemann D, Lipworth B, Malo JL, Marshall GD, Naspitz C, Nekam K, Niggemann B, Nizankowska-Mogilnicka E, Okamoto Y, Orru MP, Potter P, Price D, Stoloff SW, Vandenplas O, Viegi G, Williams D, World Health Organization; GA(2)LEN; AllerGen Allergic rhinitis and its impact on asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2)LEN and AllerGen) Allergy. 2008;63(Suppl 86):8–160. doi: 10.1111/j.1398-9995.2007.01620.x. [DOI] [PubMed] [Google Scholar]
- 6.Moscato G, Vandenplas O, Van Wijk RG, Malo JL, Perfetti L, Quirce S, Walusiak J, Castano R, Pala G, Gautrin D, De Groot H, Folletti I, Yacoub MR, A Siracusa, European Academy of Allergology and Clinical Immunolgy EAACI position paper on occupational rhinitis. Respir Res. 2009;10:16. doi: 10.1186/1465-9921-10-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jang JH, Kim DW, Kim SW, Kim DY, Seong WK, Son TJ, Rhee CS. Allergic rhinitis in laboratory animal workers and its risk factors. Ann Allergy Asthma Immunol. 2009;102(5):373–377. doi: 10.1016/S1081-1206(10)60507-8. [DOI] [PubMed] [Google Scholar]
- 8.Moscato G, Siracusa A. Rhinitis guidelines and implications for occupational rhinitis. Curr Opin Allergy Clin Immunol. 2009;9(2):110–115. doi: 10.1097/ACI.0b013e328328cfe7. [DOI] [PubMed] [Google Scholar]
- 9.Rondón C, Campo P, Togias A, Fokkens WJ, Durham SR, Powe DG, Mullol J, Blanca M. Local allergic rhinitis: concept, pathophysiology, and management. J Allergy Clin Immunol. 2012;129(6):1460–1467. doi: 10.1016/j.jaci.2012.02.032. [DOI] [PubMed] [Google Scholar]
- 10.Campo P, Rondón C, Gould HJ, Barrionuevo E, Gevaert P, Blanca M. Local IgE in non-allergic rhinitis. Clin Exp Allergy. 2015;45(5):872–881. doi: 10.1111/cea.12476. [DOI] [PubMed] [Google Scholar]
- 11.Campo P, Salas M, Blanca-López N, Rondón C. Local allergic rhinitis. Immunol Allergy Clin North Am. 2016;36(2):321–332. doi: 10.1016/j.iac.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 12.Rondón C, Romero JJ, López S, Antúnez C, Martín-Casañez E, Torres MJ, Mayorga C, R-Pena R, Blanca M. Local IgE production and positive nasal provocation test in patients with persistent nonallergic rhinitis. J Allergy Clin Immunol. 2007;119(4):899–905. doi: 10.1016/j.jaci.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 13.Rondón C, Campo P, Zambonino MA, Blanca-Lopez N, Torres MJ, Melendez L, Herrera R, Guéant-Rodriguez RM, Guéant JL, Canto G, Blanca M. Follow-up study in local allergic rhinitis shows a consistent entity not evolving to systemic allergic rhinitis. J Allergy Clin Immunol. 2014;133(4):1026–1031. doi: 10.1016/j.jaci.2013.10.034. [DOI] [PubMed] [Google Scholar]
- 14.Kim H, Kaplan A. Treatment and management of allergic rhinitis [feature]. Clin Focus. 2008;1–4.
- 15.Guerra S, Sherrill D, Martinez F, Barbee RA. Rhinitis as an independent risk factor for adult-onset asthma. J Allergy Clin Immunol. 2002;109(3):419–425. doi: 10.1067/mai.2002.121701. [DOI] [PubMed] [Google Scholar]
- 16.Horowitz E, Diemer FB, Poyser J, Rice V, Jean LG, Britt V. Asthma and rhinosinusitis prevalence in a Baltimore city public housing complex. J Allergy Clin Immunol. 2001;107:S280. [Google Scholar]
- 17.Kapsali T, Horowitz E, Togias A. Rhinitis is ubiquitous in allergic asthmatics. J Allergy Clin Immunol. 1997;99:S138. [Google Scholar]
- 18.Leynaert B, Bousquet J, Neukirch C, Liard R, Neukirch F. Perennial rhinitis: an independent risk factor for asthma in nonatopic subjects: results from the European community respiratory health survey. J Allergy Clin Immunol. 1999;104(2 Pt 1):301–304. doi: 10.1016/S0091-6749(99)70370-2. [DOI] [PubMed] [Google Scholar]
- 19.Yanez A, Rodrigo GJ. Intranasal corticosteroids versus topical H1 receptor antagonists for the treatment of allergic rhinitis: a systematic review with meta-analysis. Ann Allergy Asthma Immunol. 2002;89(5):479–484. doi: 10.1016/S1081-1206(10)62085-6. [DOI] [PubMed] [Google Scholar]
- 20.Pullerits T, Praks L, Ristioja V, Lötvall J. Comparison of a nasal glucocorticoid, antileukotriene, and a combination of antileukotriene and antihistamine in the treatment of seasonal allergic rhinitis. J Allergy Clin Immunol. 2002;109(6):949–955. doi: 10.1067/mai.2002.124467. [DOI] [PubMed] [Google Scholar]
- 21.Wilson AM, O’Byrne PM, Parameswaran K. Leukotriene receptor antagonists for allergic rhinitis: a systematic review and meta-analysis. Am J Med. 2004;116(5):338–344. doi: 10.1016/j.amjmed.2003.10.030. [DOI] [PubMed] [Google Scholar]
- 22.Weiner JM, Abramson MJ, Puy RM. Intranasal corticosteroids versus oral H1 receptor antagonists in allergic rhinitis: systematic review of randomised controlled trials. BMJ. 1998;317(7173):1624–1629. doi: 10.1136/bmj.317.7173.1624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.DeWester J, Philpot EE, Westlund RE, Cook CK, Rickard KA. The efficacy of intranasal fluticasone propionate in the relief of ocular symptoms associated with seasonal allergic rhinitis. Allergy Asthma Proc. 2003;24(5):331–337. [PubMed] [Google Scholar]
- 24.Bernstein DI, Levy AL, Hampel FC, Baidoo CA, Cook CK, Philpot EE, Rickard KA. Treatment with intranasal fluticasone propionate significantly improves ocular symptoms in patients with seasonal allergic rhinitis. Clin Exp Allergy. 2004;34(6):952–957. doi: 10.1111/j.1365-2222.2004.01952.x. [DOI] [PubMed] [Google Scholar]
- 25.Watson WT, Becker AB, Simons FER. Treatment of allergic rhinitis with intranasal corticosteroids in patients with mild asthma: effect on lower airway hyperresponsiveness. J Allergy Clin Immunol. 1993;91(1 Pt 1):97–101. doi: 10.1016/0091-6749(93)90301-U. [DOI] [PubMed] [Google Scholar]
- 26.Skoner DP, Rachelefsky GS, Meltzer EO, Chervinsky P, Morris RM, Seltzer JM, Storms WW, Wood RA. Detection of growth suppression in children during treatment with intranasal beclomethasone dipropionate. Pediatrics. 2000;105:E23. doi: 10.1542/peds.105.2.e23. [DOI] [PubMed] [Google Scholar]
- 27.Allen DB, Meltzer EO, Lemanske RF, Jr, Philpot EE, Faris MA, Kral KM, Prillaman BA, Rickard KA. No growth suppression in children treated with the maximum recommended dose of fluticasone propionate aqueous nasal spray for one year. Allergy Asthma Proc. 2002;23(6):407–413. [PubMed] [Google Scholar]
- 28.Agertoft L, Pedersen S. Effect of long-term treatment with inhaled budesonide on adult height in children with asthma. N Engl J Med. 2000;343(15):1064–1069. doi: 10.1056/NEJM200010123431502. [DOI] [PubMed] [Google Scholar]
- 29.Schenkel EJ, Skoner DP, Bronsky EA, Miller SD, Pearlman DS, Rooklin A, Rosen JP, Ruff ME, Vandewalker ML, Wanderer A, Damaraju CV, Nolop KB, Mesarina-Wicki B. Absence of growth retardation in children with perennial allergic rhinitis after one year of treatment with mometasone furoate aqueous nasal spray. Pediatrics. 2000;105:E22. doi: 10.1542/peds.105.2.e22. [DOI] [PubMed] [Google Scholar]
- 30.Bousquet J, Lund VJ, van Cauwenberge P, Bremard-Oury C, Mounedji N, Stevens MT, El-Akkad T. Implementation of guidelines for seasonal allergic rhinitis: a randomized controlled trial. Allergy. 2003;58(8):733–741. doi: 10.1034/j.1398-9995.2003.00076.x. [DOI] [PubMed] [Google Scholar]
- 31.Carr W, Bernstein J, Lieberman P, Meltzer E, Bachert C, Price D, Munzel U, Bousquet J. A novel intranasal therapy of azelastine with fluticasone for the treatment of allergic rhinitis. J Allergy Clin Immunol. 2012;129(5):1282–1289. doi: 10.1016/j.jaci.2012.01.077. [DOI] [PubMed] [Google Scholar]
- 32.Meltzer E, Ratner P, Bachert C, Carr W, Berger W, Canonica GW, Hadley J, Lieberman P, Hampel FC, Mullol J, Munzel U, Price D, Scadding G, Virchow JC, Wahn U, Murray R, Bousquet J. Clinically relevant effect of a new intranasal therapy (MP29-02) in allergic rhinitis assessed by responder analysis. Int Arch Allergy Immunol. 2013;161(4):369–377. doi: 10.1159/000351404. [DOI] [PubMed] [Google Scholar]
- 33.Price D, Shah S, Bhatia S, Bachert C, Berger W, Bousquet J, Carr W, Hellings P, Munzel U, Scadding G, Lieberman P. A new therapy (MP29-02) is effective for the long-term treatment of chronic rhinitis. J Invest Allergol Clin Immunol. 2013;23(7):495–503. [PubMed] [Google Scholar]
- 34.Berger WE, Shah S, Lieberman P, Hadley J, Price D, Munzel U, Bhatia S. Long-term, randomized safety study of MP29-02 (a novel intranasal formulation of azelastine hydrochloride and fluticasone propionate in an advanced delivery system) in subjects with chronic rhinitis. J Allergy Clin Immunol Pract. 2014;2(2):179–185. doi: 10.1016/j.jaip.2013.09.019. [DOI] [PubMed] [Google Scholar]
- 35.Pullerits T, Praks L, Skoogh BE, Ani R, Lötvall J. Randomized placebo-controlled study comparing a leukotriene receptor antagonist and a nasal glucocorticoid in seasonal allergic rhinitis. Am J Respir Crit Care Med. 1999;159(6):1814–1818. doi: 10.1164/ajrccm.159.6.9810016. [DOI] [PubMed] [Google Scholar]
- 36.Ratner PH, Howland WC, 3rd, Arastu R, Philpot EE, Klein KC, Baidoo CA, Faris MA, Rickard KA. Fluticasone propionate aqueous nasal spray provided significantly greater improvement in daytime and nighttime nasal symptoms of seasonal allergic rhinitis compared with montelukast. Ann Allergy Asthma Immunol. 2003;90(5):536–542. doi: 10.1016/S1081-1206(10)61847-9. [DOI] [PubMed] [Google Scholar]
- 37.Wilson AM, Dempsey OJ, Sims EJ, Lipworth BJ. A comparison of topical budesonide and oral montelukast in seasonal allergic rhinitis and asthma. Clin Exp Allergy. 2001;31(4):616–624. doi: 10.1046/j.1365-2222.2001.01088.x. [DOI] [PubMed] [Google Scholar]
- 38.Wilson AM, Orr LC, Sims EJ, Lipworth BJ. Effects of monotherapy with intra-nasal corticosteroid or combined oral histamine and leukotriene receptor antagonists in seasonal allergic rhinitis. Clin Exp Allergy. 2001;31(1):61–68. doi: 10.1111/j.1365-2222.2001.00964.x. [DOI] [PubMed] [Google Scholar]
- 39.Di Lorenzo G, Pacor ML, Pellitteri ME, Morici G, Di Gregoli A, Lo Bianco C, Ditta V, Martinelli N, Candore G, Mansueto P, Rini GB, Corrocher R, Caruso C. Randomized placebo-controlled trial comparing fluticasone aqueous nasal spray in mono-therapy, fluticasone plus cetirizine, fluticasone plus montelukast and cetirizine plus montelukast for seasonal allergic rhinitis. Clin Exp Allergy. 2004;34(2):259–267. doi: 10.1111/j.1365-2222.2004.01877.x. [DOI] [PubMed] [Google Scholar]
- 40.Walker SM, Durham SR, Till SJ, Roberts G, Corrigan CJ, Leech SC, Krishna MT, Rajakulasingham RK, Williams A, Chantrell J, Dixon L, Frew AJ, Nasser SM, British Society for Allergy and Clinical Immunology Immunotherapy for allergic rhinitis. Clin Exp Allergy. 2011;41(9):1177–1200. doi: 10.1111/j.1365-2222.2011.03794.x. [DOI] [PubMed] [Google Scholar]
- 41.Frew AJ. Allergen immunotherapy. J Allergy Clin Immunol. 2010;125(2 Suppl 2):S306–S313. doi: 10.1016/j.jaci.2009.10.064. [DOI] [PubMed] [Google Scholar]
- 42.Canadian Society of Allergy and Clinical Immunology. Immunotherapy manual. 2016. http://csaci.ca/wp-content/uploads/2017/12/IT-Manual-2016-5-July-2017-rev.pdf. Accessed 12 July 2018.
- 43.Calderon MA, Alves B, Jacobson M, Hurwitz B, Sheikh A, Durham S. Allergen injection immunotherapy for seasonal allergic rhinitis. Cochrane Database Syst Rev. 2007;1:CD001936. doi: 10.1002/14651858.CD001936.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Durham SR, Walker SM, Varga EM, Jacobson MR, O’Brien F, Noble W, Till SJ, Hamid QA, Nouri-Aria KT. Long-term clinical efficacy of grass pollen immunotherapy. N Engl J Med. 1999;341(7):468–475. doi: 10.1056/NEJM199908123410702. [DOI] [PubMed] [Google Scholar]
- 45.Eng PA, Borer-Reinhold M, Heijnen IA, Gnehm HP. Twelve-year follow-up after discontinuation of preseasonal grass pollen immunotherapy in childhood. Allergy. 2006;61(2):198–201. doi: 10.1111/j.1398-9995.2006.01011.x. [DOI] [PubMed] [Google Scholar]
- 46.Merck Canada Inc. GRASTEK product monograph; 2017.
- 47.Stallergenes Canada Inc. ORALAIR product monograph; 2015.
- 48.Merck Canada Inc. RAGWITEK product monograph; 2017.
- 49.ALK-Abelló A/S. ACARIZAX product monograph; 2017.
- 50.Canonica G, Cox L, Pawankar R, Baena-Cagnani CE, Blaiss M, Bonini S, Bousquet J, Calderón M, Compalati E, Durham SR, van Wijk RG, Larenas-Linnemann D, Nelson H, Passalacqua G, Pfaar O, Rosário N, Ryan D, Rosenwasser L, Schmid-Grendelmeier P, Senna G, Valovirta E, Van Bever H, Vichyanond P, Wahn U, Yusuf O. Sublingual immunotherapy: World Allergy Organization position paper 2013 update. World Allergy Organ J. 2014;7(1):6. doi: 10.1186/1939-4551-7-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yoo JK, Seikaly H, Calhoun KH. Extended use of topical nasal decongestants. Laryngoscope. 1997;107(1):40–43. doi: 10.1097/00005537-199701000-00010. [DOI] [PubMed] [Google Scholar]
- 52.Kern J, Bielory L. Complementary and alternative therapy (CAM) in the treatment of allergic rhinitis in the treatment of allergic rhinitis. Curr Allergy Asthma Rep. 2014;14(12):479. doi: 10.1007/s11882-014-0479-8. [DOI] [PubMed] [Google Scholar]
- 53.Brinkhaus B, Ortiz M, Witt CM, Roll S, Linde K, Pfab F, Niggemann B, Hummelsberger J, Treszl A, Ring J, Zuberbier T, Wegscheider K, Willich SN. Acupuncture in patients with seasonal allergic rhinitis: a randomized trial. Ann Intern Med. 2013;158(4):225–234. doi: 10.7326/0003-4819-158-4-201302190-00002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the development of this review.


