Abstract
Conventional techniques for detecting rare DNA sequences require many cycles of PCR amplification for high sensitivity and specificity, potentially introducing significant biases and errors. While amplification-free methods exist, they rarely achieve the ability to detect single molecules, and their ability to discriminate between single-nucleotide variants is often dictated by the specificity limits of hybridization thermodynamics. Here we show that a direct detection approach using single-molecule kinetic fingerprinting can surpass the thermodynamic discrimination limit by three orders of magnitude, with a dynamic range of up to five orders of magnitude with optional super-resolution analysis. This approach detects mutations as subtle as the drug resistance-conferring cancer mutation EGFR T790M (a single C→T substitution) with an estimated specificity of 99.99999%, surpassing even the leading PCR-based methods and enabling detection of 1 mutant molecule in a background of at least 1 million wild-type molecules. This level of specificity revealed rare, heat-induced cytosine deamination events that introduce false positives in PCR-based detection, but which can be overcome in our approach through milder thermal denaturation and enzymatic removal of damaged nucleobases.
Keywords: Single Molecule Fluorescence, Super-Resolution Microscopy, Nucleic Acid Hybridization, T790M Mutation, Cytosine Deamination
Graphical Abstract

INTRODUCTION
Detection and quantification of rare mutant DNA alleles is increasingly important for a broad set of applications in biological and clinical research, including tracking the emergence of rare alleles responsible for drug resistance in populations of microbes, viruses or cancer cells1–2. For example, rare fragments of circulating tumor DNA (ctDNA) are present in biofluids (e.g., urine and blood) of cancer patients and can be used to track tumor evolution or detect minimal residual disease after treatment2. However, the high-specificity detection of such rare DNA mutant alleles in a large wild-type background remains a critical challenge, which is further complicated by the low (and highly-variable) DNA content of different biofluids (Table S1). While a wide variety of innovative methods for detection of nucleic acids have been developed3–8, PCR-based methods continue to dominate. Quantitative PCR-based assays primarily exploit differential hybridization thermodynamics to distinguish homologous sequences; however, such thermodynamic discrimination is necessarily finite9, frequently resulting in poor specificity for challenging targets such as point mutations. Next-generation sequencing (NGS)-based protocols have been optimized to increase specificity, but in practice still have difficulty detecting rare DNA alleles with <10−4 relative abundance without significant increases in sequencing depth10–11.
A significant and perhaps underappreciated confounder in all workflows involving PCR amplification—including NGS and droplet digital PCR (ddPCR)—are errors caused by heat-induced chemical damage (e.g., nucleotide deamination or oxidation) and imperfect copying fidelity of DNA polymerases12–14. Once present, these errors can be exponentially amplified over many cycles, yielding significant false positives in assays for rare point mutations. While amplification-free methods can in principle avoid these errors, they typically struggle to detect single-nucleotide variants at relative abundances below ~0.1%15–17. This limitation is fundamental because hybridization reactions exhibit a maximal discrimination factor , where is the difference in the Gibbs free energy of hybridization of a detection probe to a target sequence and of the same probe to a related but spurious target sequence; in practice, this translates to Qmax,therm values ranging from about 20 to 20,000 for single-nucleotide variants9. Moreover, in contrast to PCR-based methods, amplification-free methods rarely achieve the ability to detect single-molecules.
Previously, we reported a method for microRNA detection18 using a PCR-free detection principle—termed SiMREPS, or single-molecule recognition through equilibrium Poisson sampling—wherein the direct visualization of the repeated binding of fluorescent probes to surface-captured single-stranded microRNAs yields a sequence-specific “kinetic fingerprint” that permits high-confidence identification and counting of single target molecules. While the theoretical potential for high specificity was discussed, the detection of single-nucleotide variants at very low abundance (i.e., < 0.01%) was not experimentally realized in prior work. Here, we show that SiMREPS can detect very rare mutations in double-stranded (ds)DNA with extremely high single-base discrimination. Using a straightforward, gentle denaturation combined with enzymatic removal of DNA damage, we achieve direct, amplification-free detection of single target DNA molecules with a specificity (99.99999%) that exceeds the most advanced versions of digital PCR and NGS by at least an order of magnitude10,19, and an apparent discrimination factor (Qapp > 106) exceeding Qmax,therm by greater than two orders of magnitude. Using time-encoded kinetic information, SiMREPS thus realizes the ultra-high specificity required for rare mutant DNA allele quantification and, consequently, is able to reveal spontaneous, heat-induced cytosine deamination events that render PCR-based detection of C→T mutations particularly challenging.
EXPERIMENTAL SECTION
Oligonucleotides and DNA substrates.
All unmodified DNA oligonucleotides used in this study were purchased from Integrated DNA Technologies (IDT) with standard desalting purification. Fluorescent probe oligonucleotides were purchased from IDT with either a 5′ or 3′ fluorophore modification and high-performance liquid chromatography (HPLC) purification. Biotinylated LNA capture probes were purchased from Exiqon with HPLC purification. See Table S2 for all oligonucleotide sequences. Fluorescent probes 5’-/Cy5/ATG TCT TG-3’ (FP2) specific for Exon 19 deletion and 5’-CTG CAT GA/Cy5/−3’ (FP1) specific for T790M were used in all experiments unless otherwise noted. Double-stranded DNA substrates were prepared by combining complementary oligonucleotides (20 µM each strand) in annealing buffer (10 mM Tris-HCl [pH 8.0 at 25 °C], 50 mM NaCl, 1 mM EDTA), heating at 95 °C for 5 min, followed by slow cooling at room temperature for 15 min. Details for DNA substrates prepared by restriction enzyme digestion from plasmid DNA are provided in the Supporting Information. For additional information regarding the design of auxiliary competitor probes, see the Supplementary Note.
Prediction of Fluorescent Probe discrimination properties using NUPACK.
To determine the predicted discrimination between mutant (MUT) and wild-type (WT) DNA with a specific FP design, the equilibrium molar concentration of FP-MUT and FP-WT complexes were calculated using the NUPACK web application20 (http://www.nupack.org/partition/new) using the following parameters: Target DNA concentration = 1 nM, FP concentration = 25 nM, 25 °C, 600 mM NaCl, number of strand species = 2, maximum complex size = 2, histogram filter minimum concentration = 1E-7. The predicted discrimination was then taken as the ratio of [FP-MUT]/[FP-WT] for each specific FP design. To determine the predicted FP dimer mole fraction, the FP dimer molar concentration for each FP design was calculated, and normalized by the total FP concentration [FP-FP]/ 25 nM.
The theoretical Qmax,therm was calculated from9 i.e., , where .
and are the free energies of hybridization of the MUT fluorescent probe (FP) to the MUT and WT targets, respectively, and were estimated using NUPACK with the following parameters: Nucleic acid type = DNA, DNA energy parameters = SantaLucia, 1998, Number of strand species = 2, Maximum complex size = 2, Temperature = 20 °C, [Na+] = 0.6 M, 1 µM each strand, Dangle treatment = Some. Additional guidelines for SiMREPS probe and assay design can be found elsewhere21.
Single-molecule fluorescence microscopy.
SiMREPS experiments were performed on an Olympus IX-81 objective-type TIRF microscope equipped with a 60X oil-immersion objective (APON 60XOTIRF, 1.49 NA) with both Cell^TIRF and z-drift control modules, and an EMCCD camera (IXon 897, Andor, EM gain 150). For an expanded discussion of the general instrumentation requirements, we refer the reader to a recent publication by Johnson-Buck et al.21 Sample cells were constructed as previously described18. Briefly, glass coverslips were passivated with a 1:10 mixture of biotin-PEG-5000 and mPEG-5000 (Laysan Bio, Inc.), and stored for up to two weeks in the dark under nitrogen (see Supplementary Protocol). Directly prior to single-molecule experiments, 20 µL pipette tips (Thermo Scientific #2149P05HR, low retention) were cut to an ~1 cm length as measured from the wide end, and the non-cut base was adhered to the coated glass coverslip via epoxy (Hardman DOUBLE/BUBBLE #04001) to form the sample cell.
SiMREPS assay protocol.
The SiMREPS protocol used in this study was based on our previously described method18, with three main modifications: (1) the LNA concentration was increased from 20 nM to 100 nM during surface coating, (2) a thermal denaturing step was added directly before analyte capture to increase access to the target DNA strand, and (3) two additional oligonucleotides including an LNA blocker and WT competitor (WTC) were added to the imaging solution when probing T790M samples. All DNA handling was performed in GeneMate low-adhesion 1.7-mL micro centrifuge tubes, and all dilutions and denaturing steps were performed in the presence of 2 µM poly-T oligodeoxyribonucleotide (dT10). The updated protocol was performed as follows. first, sample cells were washed with T50 buffer (10 mM Tris-HCl [pH 8.0 at 25 °C], 1 mM EDTA), and then incubated with 40 µL of 1 mg/mL streptavidin for 10 min. Cells were then washed with T50 buffer to remove excess streptavidin, incubated with 40 µL of 100 nM biotinylated LNA for 10 min, and washed with 1X PBS. Target DNA oligonucleotide was denatured at 95 °C for 3 min (unless otherwise specified) in a thermocycler, cooled in room temperature water for 5 min (via partial submersion), and immediately added to the LNA coated surface. DNA capture was performed for 1 hour at room temperature in a humidified chamber. After capture, excess DNA was removed by washing with 4X PBS. Directly before imaging, imaging buffer containing 25 nM fluorescent probe, 4X PBS, 5 mM 3,4-dihydroxybenzoate, 50 nM protocatechuate dioxygenase, and 1 mM Trolox was added to the sample cell. When probing T790M, 25 nM LNA blocker and 1 µM WTC were also added to the imaging solution (refer to Supplementary Note). Transient binding of the fluorescent probe was monitored for 10 min under TIRF illumination by 640 nM laser light with a 500 ms exposure time (1200 total frames).
Analysis of SiMREPS data.
Two different analysis pipelines were used to investigate kinetic data in this study. The first analysis pipeline is our previously optimized method, termed “diffraction limited,” which employs custom Matlab code to identify sites of fluorescent probe binding and dissociation in a 14,024 µm2 field of view and calculate intensity-versus-time trajectories at each site18. Hidden Markov modeling (HMM) is then applied using the QuB software suite (State University of New York at Buffalo) to determine the number of binding and dissociation events (Nb+d) and the median fluorescent probe bound (τbound,median) and unbound (τunbound,median) time for each candidate molecule22. Based on (i) no DNA, (ii) WT DNA only, and (iii) MUT DNA only control experiments, kinetic thresholds were defined to achieve nominal false positive frequency while retaining high sensitivity for true MUT candidates. Thresholds applied for all DNA targets include a signal-to-noise ratio >3, mean bound state intensity >500 counts above the mean background intensity, maximum τbound,median of 20 s, maximum τunbound,median of 30 s, and Nb+d ≥20. For Exon 19 deletion quantification, the minimum τbound,median and τunbound,median thresholds were set to 2 s and 2.5 s, respectively, while for T790M quantification the minimum τbound,median and τunbound,median thresholds were set to 3 s. Although τunbound,median is in principle a function of solution probe concentration, in practice we have found that minimum and maximum thresholds for τunbound,median can be used to eliminate molecule candidates that display uncharacteristically transient interactions (e.g., rapid and repeated photoblinking of the Cy5) or irreversible binding followed by photobleaching (e.g., probe interactions with surface-defects) and thus can reasonably be assumed to be artefactual. The diffraction limited analysis pipeline was used for all experiments in this study except for experiments presented in Figure 3b-c (“super-resolution” curves) and Figure 3d, where a new super-resolution analysis pipeline, which also incorporates subpixel image registration23, was used (see Supporting Information).
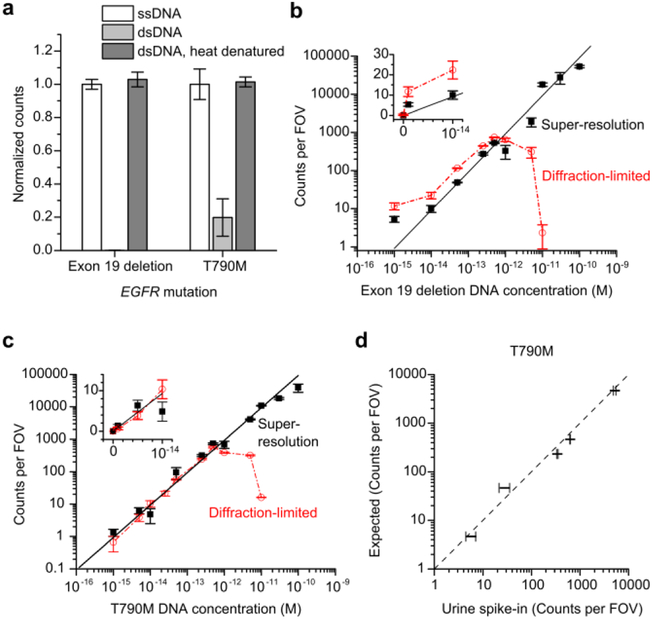
Figure 3.
Dynamic range of mutant dsDNA detection and quantification in buffer and urine matrices is enhanced by super-resolution analysis. (a) Impact of DNA strandedness and brief heat denaturation on the number of detected molecules (Counts) for 1 pM of the two MUT target DNA alleles used in this study. Data are presented as means ± s.e.m. of at least 3 independent measurements and are normalized to the mean counts detected using ssDNA as input. (b) Performance of super-resolution and diffraction-limited analysis methods over a wide concentration range for Exon 19 deletion. Data are presented as the mean ± s.e.m of n = 3 independent measurements. The red dashed line connected points from diffraction-limited analysis is included to show the reduction in detected counts at concentrations > 1 pM. Error-weighted linear fits of the super-resolution analysis (black) were constrained to a y-intercept of 0; R2 = 0.948. (c) Same as in (b), but for T790M. R2 = 0.935. (d) Correlation of observed counts from a urine spike-in sample versus the number of counts expected at a given concentration of spiked-in MUT DNA allele substrate fragment. Expected counts at a given concentration are based on the standard curve constructed with super-resolution analysis of MUT DNA captured from 1X PBS. Points that appear exactly on the diagonal (dashed line) have perfect correlation. Data are presented as the mean ± s.e.m. (x-error bars) of n = 3 independent measurements, y-error bars represent the error in the number of expected counts given the error of the linear fit.
Statistical analysis.
Statistical significance of the differences between mean counts per field of view shown in Figure 4f was determined using a using 2-tailed, unpaired t test (GraphPad QuickCalcs, www.graphpad.com). The discrimination factor Qapp from LOD experiments was calculated as described in the Supplementary Note. Curve fitting was performed using OriginPro 8 software, and values were weighted using instrumental weighting by the respective error value during linear regression.
Figure 4. High MUT versus WT discrimination and mitigation of DNA damage leads to ultra-low LOD.
(a) Plot of diagnostic kinetic parameters used to identify genuine Exon 19 deletion (MUT) DNA allele molecules based on their kinetic fingerprints. Plots are generated from one representative movie from each condition, 500 nM WT (WT only) and a mixture of 500 fM MUT with 500 nM WT (1:106 MUT:WT), after applying thresholds for fluorescence intensity, signal-to-noise, and median unbound dwell time. Each point represents an individual candidate molecule, plotted by its median bound dwell time (τbound,median) and number of binding and dissociation events (Nb+d) during a 10 min observation. Dashed lines indicate threshold values used to discriminate genuine MUT molecules (red points, upper right quadrant) from non-specific binding (grey points) (b) Limit of detection (LOD) experimental analysis for EGFR Exon 19 deletion. 500 fM MUT DNA was mixed with increasing amounts of WT DNA (open circles), or a scrambled DNA sequence that is not complementary to the capture LNA (filled circle), to achieve various MUT fractions. Data are presented as the mean ± s.d. of n = 3 independent measurements except for 10_1 and 10−4 MUT fractions (n = 6). (c) Effect of DNA preparation method, enzymatic removal of damaged DNA bases, and heat denaturation protocol on the number of observed counts for T790 (WT, blue) and T790M (MUT, red) alleles. DNA prepared by chemical synthesis, Synthetic, open bars; DNA prepared by restriction digestion from plasmid DNA, Cloned, filled bars; mild heat denaturation at 80 °C for 1 min, hashed bars. Enzyme cocktail treatment: combination of uracil-DNA glycosylase (UDG), formamidopyrimidine-DNA glycosylase (FpG), and endonuclease VIII (EndoVIII); in situ UDG treatment: treatment of surface-immobilized DNA with UDG prior to observation via the TIRF microscope. Note the difference in concentration used between WT (5 nM) and MUT (500 fM). Data are presented as the mean ± s.e.m. of n = 3 independent measurements. Tabulated values are presented in Table S3. (d) Plot of diagnostic kinetic parameters as in (a), but for the T790M-specific fluorescent probe and LNA capture strand. 5 nM WT (WT only) and a mixture of 50 fM MUT with 5 nM WT (1:106 MUT:WT). (e) Limit of detection (LOD) experimental analysis for T790M. 50 fM MUT DNA was mixed with increasing amounts of WT DNA to achieve various MUT fractions. Data are presented as the mean ± s.d. of n = 3 independent measurements except for 10−4 MUT fraction (n = 4). (f) Comparison of counts from low MUT allelic fraction and WT only conditions. Exon 19/deletion: 15 ± 2 counts (1:106 MUT:WT), 0.7 ± 0.6 counts (WT only), P = 0.0003; T790/M: 12.7 ± 1.5 counts (1:106 MUT:WT), 1.1 ± 1.1 counts (WT only), P < 0.0001. Data are presented as the mean ± s.d. of n = 3 independent measurements, except for T790 WT only (n = 8). Significance was assessed using a 2-tailed, unpaired t test.
Code availability.
The custom Matlab scripts used in this study are available upon request.
Data availability.
The microscopy data (e.g., movie files) that support the findings of this study are available in the Deep Blue Data Repository from the University of Michigan library (https://deepblue.lib.umich.edu/).
RESULTS
Optimized fluorescent probe design for improved discrimination between wild-type and mutant alleles.
To adapt the SiMREPS kinetic fingerprinting approach to rare mutant DNA allele detection, we began by optimizing the design of the fluorescent probe (FP) through a combination of theoretical and empirical methods. The two EGFR mutations chosen for use in this study were an in-frame deletion in exon 19 (COSMIC ID: COSM6225; c.2236_2250del15 [p.E746_A750delELREA]), and a single point mutation in exon 20 that results in the missense mutation T790M (COSMIC ID: COSM6240, c.2369C>T [p.T790M]).
First, the effect of FP length on DNA detection was examined. The differences in the kinetics of FP binding to, and dissociating from, genuine target molecules versus those that are closely-related in sequence (i.e., off-target molecules) form the basis of the SiMREPS assay’s ability to distinguish between target and off-target molecules18,21. Therefore, it is advantageous to design FP sequences that undergo many cycles of binding and dissociation within the experimental observation time. Although 9-nt DNA FPs were used predominantly in our previous work on miRNA detection18, we found the bound residence time to be undesirably long for a 9 nt FP binding to a DNA target, whereas an 8 nt FP (Figure S1) yielded a reliable kinetic fingerprint in a 10-minute experiment. We hypothesize that this is due to the difference in binding energetics between RNA-DNA and DNA-DNA hybrids. The modeling software NUPACK20 was then used to assess 8 nt FP sequences having different registers with respect to the mutation site (Figure 1a). In addition, FP registers were chosen that would minimize the number of possible contiguous base pairs with the wild-type sequence, which has been shown to be a key determinant of binding stability, and thus dissociation rate, for DNA duplexes on this length scale24–25. For Exon 19 deletion, for example, the FP was chosen to span the junction of the 15 bp deletion, thus limiting the number of contiguous complementary base pairs with the wild-type sequence. FPs were evaluated based on predictions of i) theoretical discrimination between mutant and wild-type and ii) probability of FP dimer formation (Figure 1b).
Figure 1.
Fluorescent probe design. (a) Fluorescent probe (FP) sequences in having various registers with respect to the EGFR mutations used in this study. A 3’ exogenous barcode sequence (TAGGAC) present on the DNA analytes was not considered while designing the probe sequences and is omitted for clarity. Lower case letters represent the mutation site. CY5, fluorophore. (b) Plot of mutant/wild-type discrimination and FP dimer formation predicted using NUPACK.
High-confidence detection of mutant dsDNA with single-molecule kinetic fingerprinting.
Whereas microRNAs are easily captured from solution because they are single-stranded, extending SiMREPS to DNA required development of an effective protocol for denaturation of the targeted dsDNA. We found that thermal denaturation with addition of oligonucleotide dT10 as a carrier in high molar excess substantially reduced re-annealing (Figure S2), thus permitting efficient surface capture of the single-stranded (ss)DNA target strand. We thus incorporated this short thermal denaturation step into the SiMREPS protocol, followed by surface capture and visualization of single target molecules via the repeated transient binding of an 8-nucleotide fluorescent probe (FP) using total internal reflection fluorescence (TIRF) microscopy (Figure 2a).
Figure 2.
Direct DNA visualization and counting of mutant molecules by amplification-free single-molecule kinetic fingerprinting. (a) Schematic depicting the experimental SiMREPS approach adapted for DNA detection. Double-stranded (duplex) DNA is converted to single-stranded DNA with brief heat-denaturation in the presence of high concentrations of single-stranded dT10 carrier to disfavor re-annealing of complementary analyte strands. Single-stranded target DNA is captured by target gene-specific LNA capture strands immobilized on the slide surface, and remaining unbound DNA is washed away prior to kinetic fingerprinting with a mutant (MUT)-specific fluorescent probe (FP) with optimized binding and dissociation kinetics. Surface immobilized molecules are seen as bright points in the accompanying 50-frame averaged image showing a subset of the entire field-of-view. Scale bar = 3 microns. (b) Representative kinetic traces using a MUT-specific FP with MUT DNA, with wild-type (WT) DNA, or without DNA (No DNA control) for EGFR Exon 19/deletion. Fluorescence intensity trace, black; 2-state idealization from hidden Markov modeling, magenta line. (c) Histograms of the number of candidate molecules per field-of-view (FOV) showing a given number of intensity transitions (Nb+d), after applying thresholds for fluorescence intensity and signal-to-noise (see Experimental Section), for experiments in which 500 fM EGFR Exon 19 deletion MUT, 500 nM WT or no DNA was used. (d) Plot for the same dataset shown in (c) depicting the number of candidate molecules as a function of filtering thresholds applied to arrive at the final accepted molecule count.
Since some FP binding to the imaging surface is observed even in absence of the mutant (MUT) target—in this case, an EGFR exon 19 deletion (c.2236_2250del15)—simply counting fluorescent spots would typically yield hundreds of false positives per field of view (Figure 2). However, analysis of fluorescence-versus-time traces revealed clear differences between the repetitive binding of the FP to a single immobilized MUT molecule and the more sporadic binding observed in the presence of only wild-type (WT) DNA, or in no-DNA controls (NDC) (Figure 2b). By requiring a minimum number of FP binding and dissociation events (Nb+d) for a candidate to be counted as a MUT molecule, the majority of nonspecific binding events from WT and NDC conditions could be excluded (Figure 2c).
The few remaining false positives could be excluded by applying filters based on the apparent lifetimes in the FP-bound (τbound,median) and FP-unbound (τunbound,median) states, resulting in essentially zero background in typical measurements (Figure 2d and Experimental Section).
Assay validation for clinically-relevant mutant DNA targets and optional dynamic range expansion by super-resolution data analysis.
We applied the above kinetic analysis to two clinically relevant EGFR tyrosine kinase domain mutations used to validate the assay: c.2236_2250del15 (Exon 19 deletion) and c.2369C>T (T790M). First, we compared the number of counts detected for dsDNA, either with or without heat denaturation, relative to the same concentration (1 pM) of ssDNA; we observed a similar number of counts only when the dsDNA had been heat denatured, confirming that the denaturation protocol enabled the same detection efficiency for dsDNA as ssDNA (Figure 3a). Next, we found that the kinetic fingerprinting assays exhibit a linear dependence on MUT dsDNA concentration over more than two orders of magnitude (Figure 3b-c, Figure S3) with diffraction-limited imaging. We note that at high densities of captured MUT molecules, deviations from linearity occurred (> 1 molecule per 25 µm2) where neighboring single-molecule kinetic fingerprints could no longer be resolved from one another.
To date, single-molecule kinetic fingerprinting has been diffraction-limited and therefore unable to separate single-molecule binding events above a critical analyte density, where signal saturation artifacts arise. For cases where a broader dynamic range of analyte concentrations without sample dilution is desired, we developed a super-resolution analytic pipeline, inspired by stochastic reconstruction microscopy techniques26–27. Here, fluctuations in fluorescence intensity due to FP binding and dissociation are localized with sub-pixel accuracy, clustered according to position, and the resulting kinetic fingerprints analyzed analogously to the standard diffraction-limited SiMREPS approach (Figure S4). This super-resolution analysis increased the dynamic range of SiMREPS to >5 orders of magnitude for both Exon 19 deletion (Figure 3b) and T790M (Figure 3c), rivaling digital PCR assays28.
A common application of current PCR-based assays for mutant DNA detection is for quantification of MUT ctDNA extracted from human biofluids1–2,29–30. In order to examine matrix effects on our assay, we tested the robustness of our super-resolution kinetic fingerprinting assay in urine—a compositionally complex biological sample matrix in which trans-renal ctDNA can be detected31. As proof-of-principle, we quantified 28-bp synthetic T790M DNA molecules spiked into healthy donor urine and observed a linear response that showed strong concordance with counts measured in buffer alone (R2 = 0.919) (Figure 3d), supporting the potential for clinical development of our SiMREPS assay.
Enhancing the specificity for rare mutant alleles amid excess wild-type.
Using our standard diffraction-limited imaging, we sought to characterize the specificity for MUT DNA in the presence of a large excess of WT sequence. Using the optimized FP for Exon 19 deletion (FP2, Figure 1a), a specificity of >99.99999% was achieved between the Exon 19 deletion MUT and WT DNA, based on evaluation of single-molecule kinetic fingerprints of binding event frequency and duration (Figure 4a, Supplementary Note). This approach enabled a limit of detection (LOD), defined here as the lowest MUT allele fraction showing >3 s.e.m. above background, of ≤ 0.0001% (10−6; Figure 4b, Figure S5). Interestingly, we found that this LOD was limited not by FP specificity, but by saturation of surface capture sites with excess WT DNA, as was revealed by replacing the Exon 19 WT sequence with a scrambled DNA sequence incapable of binding the capture probe (Figure 4b, filled circle).
We next aimed to achieve similarly robust detection of the T790M point mutation, a clinically actionable biomarker for which there is a specific therapy available32 and which represents an especially challenging analytic target for current DNA detection technology33. Despite using the optimized FP-sequence for T790M (FP1, Figure 1), we observed that our initial sensitivity for T790M detection was lower than expected. To test the hypothesis that this decreased sensitivity arose in part from sequestration of the FP due to spurious binding to the partially complementary LNA, we introduced an additional “blocker” strand and indeed observed a ~3-fold improvement in observed molecule count and homogeneity of the kinetic fingerprints (Figure S6, Supplementary Note). Moreover, in negative control experiments containing only T790 (WT) DNA, we observed appreciable background noise in the fluorescence microscopy images, which we hypothesized was due to extremely transient off-target binding between the MUT-specific FP and surface-bound WT molecules that becomes significant when the WT is present at very high concentrations. Consistent with this hypothesis, inclusion of an auxiliary competitor probe designed to suppress such interactions resulted in significantly improved signal-to-noise (Figure S7, Supplementary Note).
Effects of molecular heterogeneity and DNA damage on specificity.
In contrast to the Exon 19 WT allele, for which false positive signal was independent of WT concentration and indistinguishable from no-DNA controls, we noticed that the chemically synthesized T790 (WT) DNA exhibited a significant false positive frequency of about 1-in-3,870 (Table S3).
While the resulting apparent discrimination factor Qapp was already comparable to the theoretical thermodynamic maximum Qmax,therm of 4,080 (Table 1), it was surprising to observe any significant MUT-like kinetic fingerprints in the WT sample. We hypothesized that errors during solid-phase oligonucleotide synthesis could result in a minute fraction of MUT sequence in the WT sample. Therefore, high-fidelity T790 (WT) and T790M (MUT) DNA fragments were prepared by enzymatic digestion from a multi-insert bacterial plasmid (Figure S8). The sequence fidelity of substrates prepared in this way is expected to be primarily dictated by DNA replication error rates in E. coli, which is on the order of 10−9-10−11 errors per base pair34. This high-fidelity material exhibited ~30% fewer false positives than the corresponding synthetic DNA (Figure 4c) suggesting that, although chemical synthesis errors may contribute, they cannot account for the majority of the false positive signal.
Table 1.
Apparent specificity and discrimination factor for EGFR T790M MUT vs. WT as a function of input material and assay protocol.
| Denaturation Protocol | Enzyme Cocktail | in situ UDG | Specificity | Qapp | Qapp/Qmax,therm | |
|---|---|---|---|---|---|---|
| 5 nM synthetic WTa | 95 °C / 3 min | − | − | 99.975±0.001% | 3860 | 0.95 |
| 5 nM cloned WTa | 95 °C / 3 min | − | − | 99.9821±0.0003% | 5580 | 1.37 |
| + | − | 99.9960±0.0003% | 24,700 | 6.1 | ||
| + | + | 99.99970±0.00007% | 340,000 | 83 | ||
| 80 °C / 1 min | + | + | 99.99996±0.00001% | 2,600,000 | 640 | |
| 50 nM cloned WTb | 80 °C / 1 min | + | + | 99.99999±0.00001% | 11,000,000 | 2600 |
Calculated from data shown in Figure 4f and Figure S12
Because T790M is a cytosine (C) to thymine (T) point mutation, we hypothesized that spontaneous deamination of cytosine to uracil (U) may cause a MUT-like kinetic fingerprint in ostensibly WT DNA. We therefore analyzed synthetic DNA bearing a C→U mutation as a mimic of deamination, and found this target to have a sufficiently similar, though not identical, kinetic fingerprint to that of MUT DNA (Figure S9, Supporting Information). To remove such false positives from WT material, we pre-treated WT DNA with a combination of commercially available enzymes that recognize and remove damaged DNA bases prior to SiMREPS analysis: uracil-DNA glycosylase (UDG), formamidopyrimidine-DNA glycosylase (FpG) and endonuclease VIII (EndoVIII) (Supporting Information). Consistent with our hypothesis, this “enzyme cocktail” pre-treatment (designed to remove uracil as well as uracil-like products arising from oxidation of cytosine, such as uracil glycol and 5-hydroxy uracil35–36) decreased false positives by >80% compared to the synthetic DNA (Figure 4c), with separate experiments indicating UDG as the dominant factor in the reduction (Figure S10).
In light of literature evidence that prolonged heating catalyzes C→U deamination for T79012, we reasoned that the remaining false positives (about 1-in-24,700) may be caused by the thermal denaturation used to liberate ssDNA in the SiMREPS assay. Therefore, we also treated DNA with UDG in situ (i.e., after denaturation and surface capture), which reduced the rate of false positives to about 1-in-340,000. Next, we examined multiple denaturation temperatures and durations for their effect on false positive signal (Figure S11). Using a shorter and lower-temperature denaturation protocol (80 °C for 1 min) led to a reduction of false positives by nearly 8-fold compared to the standard protocol of 95 °C for 3 min, yielding a false positive frequency of approximately 1-in-2.6 million (Figure 4c, Table S3). An LOD experiment using assay conditions revised to limit the influence of DNA damage (Figure 4d-e and Figure S12) measured reliable detection down to 10−6 MUT allele fraction, nearly matching the result achieved for the Exon 19 deletion (Figure 4f) and resulting in an estimated specificity of 99.99999% and a Qapp of 1.1 × 107.
DISCUSSION
Exponential amplification of input nucleic acids by PCR is integral to most assays for rare mutant alleles due to its multiplicative improvement of both sensitivity and specificity. Yet PCR itself imposes limitations on the specificity for very rare sequences, especially point mutations such as EGFR T790M, by introducing and amplifying errors such as single-base substitutions and deamination of cytosine. Methods utilizing isothermal amplification in lieu of PCR have also been reported, and while they show promise for point-of-care detection with limited or no instrumentation, residual signal from WT-only samples still places a lower limit on the fraction of MUT allele that can be detected37. Conversely, most amplification-free assays forego the multiplicative improvements provided by PCR, resulting in lower sensitivity and a sequence-dependent maximal specificity dictated by hybridization thermodynamics. Recently, simulation-guided design of isothermal exchange probes has permitted the consistent realization of specificity close to the thermodynamic limit, but a sufficiently large excess of WT sequence (200- to 3000-fold) will nonetheless give the same signal as a given concentration of MUT sequence15.
In contrast, the kinetic fingerprinting assay developed here exhibits both the ability to directly observe single molecules and extremely high specificity by essentially integrating over multiple, temporally-resolved probing events. Our amplification-free SiMREPS assay can detect both Exon 19 deletion and T790M mutations with an LOD ≤ 1-in-1 million; in the case of T790M, this is 10-fold lower than even the highest-performing ddPCR assay for that mutation19. The apparent discrimination factor Qapp for T790M (Figure 4f, Table 1) is ~11 million in our optimized assay, which is approximately 2600 times greater than the maximum thermodynamic discrimination factor under our imaging conditions as estimated by NUPACK38. While kinetic fingerprinting has been shown to be capable of single-nucleotide discrimination18,39, the demonstrated specificity in prior studies was 2–4 orders of magnitude lower than that achieved here, and in one case required asymmetric PCR amplification39. The performance achieved in the present work suggests that arbitrarily high specificity in an amplification-free assay can indeed be achieved by single-molecule kinetic fingerprinting, given (1) suitable assay optimization and (2) treatment of samples to remove damage that would otherwise be read as false positives. Furthermore, the >100-fold increase of assay dynamic range by super-resolution analysis should significantly broaden the potential applications of this technique.
The absolute LOD – defined in terms of absolute concentration of MUT analyte, rather than abundance relative to the WT sequence that was discussed above – of our kinetic fingerprinting assays for EGFR mutations is currently ~1 fM (Figure 3b-c). While similar to or better than the that of leading amplification-free methods16,40, this absolute LOD is significantly higher than that of ddPCR, the highest-sensitivity method for nucleic acid quantification. Our assay’s sensitivity is mainly a function of the efficiency of capturing the target nucleic acids within a small enough region of the coverslip to easily detect, which depends in turn on diffusion rates and both the thermodynamics and kinetics of analyte capture. However, as a single-molecule technique, there is no reason SiMREPS should not be capable of much lower absolute LODs in the future after including suitable mass transfer or other optimizations of the capture protocol.
There is a growing need for accurate, reproducible quantification of ultra-rare DNA alleles for multiple applications, including, for example, identification of residual disease or emerging drug-resistant clones after cancer therapy based on detection of ctDNA in blood31. In addition, detection of specific DNA alleles in urine is an important approach for analyzing fetal DNA in pregnant women41–44 and in patients with solid tumors45–46. Based on the advances described here that overcome the barriers of specificity, LOD, and ease of workflow faced by current gold standard PCR and NGS approaches, we envision that single-molecule kinetic fingerprinting by SiMREPS will be enabling for a range of fields that require highly specific, rare sequence detection and quantification. Furthermore, since the detection principle underlying single-molecule kinetic fingerprinting does not rely on any nucleic acid-specific enzymatic processing, it is likely that this technique will find application in the detection of analytes other than DNA and RNA with specificity surpassing the thermodynamic limit.
Supplementary Material
ACKNOWLEDGMENTS
The authors acknowledge support from a Michigan Economic Development Corporation MTRAC for Life Sciences grant to M.T., N.G.W., and A.J-B.; pilot grants from the University of Michigan MCubed 2.0 program, the James Selleck Bower Permanently Endowed Innovative Promise Funds for Cancer Research of the University of Michigan Rogel Cancer Center, and Fast Forward GI Innovation Fund to N.G.W. and M.T.; and NIH grant R21 CA204560 to N.G.W. and M.T. The authors also wish to thank the Single Molecule Analysis in Real-Time (SMART) Center of the University of Michigan, seeded by NSF MRI-R2-ID award DBI-0959823 to N.G.W., as well as J.D. Hoff for training, technical advice and use of the objective-type TIRF microscope.
Footnotes
Supporting Information. Additional experimental details, supporting figures, tables, notes and protocol. This material is available free of charge via the Internet at http://pubs.acs.org.
NotesA.J.-B., M.T., and N.G.W. are inventors on multiple patent applications related to SiMREPS, and equity holders of Alight Sciences LLC, a startup company aiming to commercialize the presented technology.
REFERENCES
- 1.Rybicka M; Stalke P; Bielawski KP, Current molecular methods for the detection of hepatitis B virus quasispecies. Rev. Med. Virol. 2016, 26 (5), 369–81. [DOI] [PubMed] [Google Scholar]
- 2.Diaz LA Jr.; Bardelli A, Liquid biopsies: genotyping circulating tumor DNA. J. Clin. Oncol 2014, 32 (6), 579–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Li Z; Hayman RB; Walt DR, Detection of single-molecule DNA hybridization using enzymatic amplification in an array of femtoliter-sized reaction vessels. J. Am. Chem. Soc 2008, 130 (38), 12622–3. [DOI] [PubMed] [Google Scholar]
- 4.Johnson RP; Richardson JA; Brown T; Bartlett PN, A label-free, electrochemical SERS-based assay for detection of DNA hybridization and discrimination of mutations. J. Am. Chem. Soc. 2012, 134 (34), 14099–107. [DOI] [PubMed] [Google Scholar]
- 5.Ge Z; Lin M; Wang P; Pei H; Yan J; Shi J; Huang Q; He D; Fan C; Zuo X, Hybridization chain reaction amplification of microRNA detection with a tetrahedral DNA nanostructure-based electrochemical biosensor. Anal. Chem. 2014, 86 (4), 2124–30. [DOI] [PubMed] [Google Scholar]
- 6.Das J; Ivanov I; Sargent EH; Kelley SO, DNA Clutch Probes for Circulating Tumor DNA Analysis. J. Am. Chem. Soc. 2016, 138 (34), 11009–16. [DOI] [PubMed] [Google Scholar]
- 7.Chen SX; Seelig G, An Engineered Kinetic Amplification Mechanism for Single Nucleotide Variant Discrimination by DNA Hybridization Probes. J. Am. Chem. Soc. 2016, 138 (15), 5076–86. [DOI] [PubMed] [Google Scholar]
- 8.Smith SJ; Nemr CR; Kelley SO, Chemistry-Driven Approaches for Ultrasensitive Nucleic Acid Detection. J. Am. Chem. Soc. 2017, 139 (3), 1020–1028. [DOI] [PubMed] [Google Scholar]
- 9.Zhang DY; Chen SX; Yin P, Optimizing the specificity of nucleic acid hybridization. Nat. Chem 2012, 4 (3), 208–214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Newman AM; Lovejoy AF; Klass DM; Kurtz DM; Chabon JJ; Scherer F; Stehr H; Liu CL; Bratman SV; Say C; Zhou L; Carter JN; West RB; Sledge GW Jr; Shrager JB; Loo BW Jr; Neal JW; Wakelee HA; Diehn M; Alizadeh AA, Integrated digital error suppression for improved detection of circulating tumor DNA. Nat. Biotechnol. 2016, 34 (5), 547–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aravanis AM; Lee M; Klausner RD, Next-Generation Sequencing of Circulating Tumor DNA for Early Cancer Detection. Cell 2017, 168 (4), 571–574. [DOI] [PubMed] [Google Scholar]
- 12.Chen G; Mosier S; Gocke CD; Lin MT; Eshleman JR, Cytosine deamination is a major cause of baseline noise in next-generation sequencing. Mol. Diagn. Ther. 2014, 18 (5), 587–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Potapov V; Ong JL, Examining Sources of Error in PCR by Single-Molecule Sequencing. PLoS ONE 2017, 12 (1), e0169774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen L; Liu P; Evans TC Jr.; Ettwiller LM, DNA damage is a pervasive cause of sequencing errors, directly confounding variant identification. Science 2017, 355 (6326), 752–756. [DOI] [PubMed] [Google Scholar]
- 15.Wang JS; Zhang DY, Simulation-guided DNA probe design for consistently ultraspecific hybridization. Nat. Chem. 2015, 7 (7), 545–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cohen L; Hartman MR; Amardey-Wellington A; Walt DR, Digital direct detection of microRNAs using single molecule arrays. Nucleic Acids Res. 2017, 45 (14), e137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li Z; Zhou X; Li L; Liu S; Wang C; Yu C; Su X, Probing DNA Hybridization Equilibrium by Cationic Conjugated Polymer for Highly Selective Detection and Imaging of Single-Nucleotide Mutation. Anal. Chem. 2018, 90 (11), 6804–6810. [DOI] [PubMed] [Google Scholar]
- 18.Johnson-Buck A; Su X; Giraldez MD; Zhao M; Tewari M; Walter NG, Kinetic fingerprinting to identify and count single nucleic acids. Nat. Biotechnol 2015, 33 (7), 730–732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Watanabe M; Kawaguchi T; Isa S; Ando M; Tamiya A; Kubo A; Saka H; Takeo S; Adachi H; Tagawa T; Kakegawa S; Yamashita M; Kataoka K; Ichinose Y; Takeuchi Y; Sakamoto K; Matsumura A; Koh Y, Ultra-Sensitive Detection of the Pretreatment EGFR T790M Mutation in Non-Small Cell Lung Cancer Patients with an EGFR-Activating Mutation Using Droplet Digital PCR. Clin. Cancer Res 2015, 21 (15), 3552–60. [DOI] [PubMed] [Google Scholar]
- 20.Zadeh JN; Steenberg CD; Bois JS; Wolfe BR; Pierce MB; Khan AR; Dirks RM; Pierce NA, NUPACK: Analysis and design of nucleic acid systems. J. Comput. Chem 2011, 32 (1), 170–3. [DOI] [PubMed] [Google Scholar]
- 21.Johnson-Buck A; Li J; Tewari M; Walter NG, A guide to nucleic acid detection by single-molecule kinetic fingerprinting. Methods 2018, (In Press, Accepted Manuscript), DOI: 10.1016/j.ymeth.2018.08.002. Available Online: Aug 10, 2018. 10.1016/j.ymeth.2018.08.002 (accessed Aug 11, 2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Blanco M; Walter NG, Analysis of complex single-molecule FRET time trajectories. Methods Enzymol. 2010, 472, 153–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guizar-Sicairos M; Thurman ST; Fienup JR, Efficient subpixel image registration algorithms. Opt. Lett 2008, 33 (2), 156–8. [DOI] [PubMed] [Google Scholar]
- 24.Cisse II; Kim H; Ha T, A rule of seven in Watson-Crick base-pairing of mismatched sequences. Nat. Struct. Mol. Biol 2012, 19 (6), 623–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dupuis NF; Holmstrom ED; Nesbitt DJ, Single-molecule kinetics reveal cation-promoted DNA duplex formation through ordering of single-stranded helices. Biophys. J 2013, 105 (3), 756–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hess ST; Girirajan TP; Mason MD, Ultra-high resolution imaging by fluorescence photoactivation localization microscopy. Biophys. J 2006, 91 (11), 4258–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rust MJ; Bates M; Zhuang X, Sub-diffraction-limit imaging by stochastic optical reconstruction microscopy (STORM). Nat. Meth 2006, 3 (10), 793–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jones GM; Busby E; Garson JA; Grant PR; Nastouli E; Devonshire AS; Whale AS, Digital PCR dynamic range is approaching that of real-time quantitative PCR. Biomol. Detect. Quantif 2016, 10, 31–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cohen JD; Li L; Wang Y; Thoburn C; Afsari B; Danilova L; Douville C; Javed AA; Wong F; Mattox A; Hruban RH; Wolfgang CL; Goggins MG; Dal Molin M; Wang TL; Roden R; Klein AP; Ptak J; Dobbyn L; Schaefer J; Silliman N; Popoli M; Vogelstein JT; Browne JD; Schoen RE; Brand RE; Tie J; Gibbs P; Wong HL; Mansfield AS; Jen J; Hanash SM; Falconi M; Allen PJ; Zhou S; Bettegowda C; Diaz LA Jr.; Tomasetti C; Kinzler KW; Vogelstein B; Lennon AM; Papadopoulos N, Detection and localization of surgically resectable cancers with a multi-analyte blood test. Science 2018, 359 (6378), 926–930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Heitzer E; Ulz P; Geigl JB, Circulating tumor DNA as a liquid biopsy for cancer. Clin. Chem 2015, 61 (1), 112–23. [DOI] [PubMed] [Google Scholar]
- 31.Husain H; Melnikova VO; Kosco K; Woodward B; More S; Pingle SC; Weihe E; Park BH; Tewari M; Erlander MG; Cohen E; Lippman SM; Kurzrock R, Monitoring Daily Dynamics of Early Tumor Response to Targeted Therapy by Detecting Circulating Tumor DNA in Urine. Clin. Cancer Res 2017, 23 (16), 4716–4723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang S; Cang S; Liu D, Third-generation inhibitors targeting EGFR T790M mutation in advanced non-small cell lung cancer. J. Hematol. Oncol 2016, 9, 34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Milbury CA; Zhong Q; Lin J; Williams M; Olson J; Link DR; Hutchison B, Determining lower limits of detection of digital PCR assays for cancer-related gene mutations. Biomol. Detect. Quantif 2014, 1 (1), 8–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fijalkowska IJ; Schaaper RM; Jonczyk P, DNA replication fidelity in Escherichia coli: a multi-DNA polymerase affair. FEMS Microbiol. Rev 2012, 36 (6), 1105–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kreutzer DA; Essigmann JM, Oxidized, deaminated cytosines are a source of C --> T transitions in vivo. Proc. Natl. Acad. Sci. U.S.A. 1998, 95 (7), 3578–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Purmal AA; Lampman GW; Bond JP; Hatahet Z; Wallace SS, Enzymatic processing of uracil glycol, a major oxidative product of DNA cytosine. J. Biol. Chem 1998, 273 (16), 10026–35. [DOI] [PubMed] [Google Scholar]
- 37.Gootenberg JS; Abudayyeh OO; Kellner MJ; Joung J; Collins JJ; Zhang F, Multiplexed and portable nucleic acid detection platform with Cas13, Cas12a, and Csm6. Science 2018, 360 (6387), 439–444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wolfe BR; Porubsky NJ; Zadeh JN; Dirks RM; Pierce NA, Constrained Multistate Sequence Design for Nucleic Acid Reaction Pathway Engineering. J. Am. Chem. Soc 2017, 139 (8), 3134–3144. [DOI] [PubMed] [Google Scholar]
- 39.Su X; Li L; Wang S; Hao D; Wang L; Yu C, Single-Molecule Counting of Point Mutations by Transient DNA Binding. Sci Rep 2017, 7, 43824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Geiss GK; Bumgarner RE; Birditt B; Dahl T; Dowidar N; Dunaway DL; Fell HP; Ferree S; George RD; Grogan T; James JJ; Maysuria M; Mitton JD; Oliveri P; Osborn JL; Peng T; Ratcliffe AL; Webster PJ; Davidson EH; Hood L; Dimitrov K, Direct multiplexed measurement of gene expression with color-coded probe pairs. Nat. Biotechnol 2008, 26 (3), 317–25. [DOI] [PubMed] [Google Scholar]
- 41.Koide K; Sekizawa A; Iwasaki M; Matsuoka R; Honma S; Farina A; Saito H; Okai T, Fragmentation of cell-free fetal DNA in plasma and urine of pregnant women. Prenat. Diagn 2005, 25 (7), 604–607. [DOI] [PubMed] [Google Scholar]
- 42.Stephanie C; Lee SW; Jiang P; Leung TY; Chan KA; Chiu RW; Lo YD, High-resolution profiling of fetal DNA clearance from maternal plasma by massively parallel sequencing. Clin. Chem 2013, 59 (8), 1228–1237. [DOI] [PubMed] [Google Scholar]
- 43.Shekhtman EM; Anne K; Melkonyan HS; Robbins DJ; Warsof SL; Umansky SR, Optimization of transrenal DNA analysis: detection of fetal DNA in maternal urine. Clin. Chem 2009, 55 (4), 723–729. [DOI] [PubMed] [Google Scholar]
- 44.Tsui NB; Jiang P; Chow KC; Su X; Leung TY; Sun H; Chan KA; Chiu RW; Lo YD, High resolution size analysis of fetal DNA in the urine of pregnant women by paired-end massively parallel sequencing. PLoS ONE 2012, 7 (10), e48319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Su Y-H; Wang M; Brenner DE; Norton PA; Block TM, Detection of Mutated K-ras DNA in Urine, Plasma, and Serum of Patients with Colorectal Carcinoma or Adenomatous Polyps. Ann. N. Y. Acad. Sci 2008, 1137 (1), 197–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Su YH; Wang M; Block TM; Landt O; Botezatu I; Serdyuk O; Lichtenstein A; Melkonyan H; Tomei LD; Umansky S, Transrenal DNA as a diagnostic tool: important technical notes. Ann. N. Y. Acad. Sci 2004, 1022 (1), 81–89. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The microscopy data (e.g., movie files) that support the findings of this study are available in the Deep Blue Data Repository from the University of Michigan library (https://deepblue.lib.umich.edu/).