Abstract
Purpose
This paper aimed to evaluate the intraocular pressure (IOP)-lowering effects of prostaglandin analogs (PGAs) in Japanese patients with normal-tension glaucoma (NTG) by reviewing the current literature.
Methods
In February 2018, database searches were performed in PubMed, Embase, ProQuest, and the Japanese databases JAPICDOC and JMEDPlus. Studies were sorted into two categories: Category 1 consisted of studies of patients with NTG who reported reduced IOP values and Category 2 consisted of studies of patients with NTG who had IOP values at predosing and a final evaluation point. Search terms included ([unoprostone or latanoprost or travoprost or bimatoprost or tafluprost] and [glaucoma] and [Japan or Japanese]). The weighted ocular hypotensive efficacy was calculated. A scatter plot analysis was performed and a regression equation was calculated for each medication. The fitting of each regression equation was evaluated by the least squares method.
Results
Eleven articles were eligible for Category 1 and 25 articles for Category 2. In the rank order of IOP-lowering efficacy of PGAs, bimatoprost was the strongest and latanoprost the weakest. Travoprost and tafluprost had almost the same level of ocular hypotensive effect, and both were stronger than latanoprost. The scatter plot analysis showed that all PGAs reduced IOP by 15%–20%. At higher IOP (17–18 mmHg), the ocular hypotensive effect was almost the same with latanoprost, travoprost, and tafluprost. In contrast, at lower IOP (12–15 mmHg), the IOP reduction with latanoprost was weaker than with travoprost or tafluprost.
Conclusion
This literature review of the ocular hypotensive effects of PGAs in Japanese patients with NTG highlighted that PGAs had different ocular hypotensive effects. Ophthalmologists should understand the IOP-lowering profiles of various PGAs and apply them to patients with NTG on a case-by-case basis.
Trial registration
UMIN Clinical Trials Registry UMIN000032344.
Keywords: Japanese, normal-tension glaucoma, ocular hypotensive effects, prostaglandin analogs
Introduction
Ocular hypotensive therapy is the only evidence-based therapy for glaucoma.1 In clinical practice, patients with glaucoma have variable responses to medication, depending on stage and severity of disease, intraocular pressure (IOP) before treatment, age, life expectancy, progression of visual field defect, family history, conditions of another eye, etc.1 Therefore, ophthalmologists should consider these factors before initiating IOP-lowering medication.1,2 Due to the large number of studies conducted to evaluate drugs used to treat glaucoma, a high-quality review of real-world studies comparing the efficacy of different medications may be helpful for an ophthalmologist. Currently, Japanese ophthalmologists select medications for patients with normal-tension glaucoma (NTG) based on data from small or poorly designed studies. Meta-analyses, including network meta-analyses, of randomized controlled trials (RCTs) have the highest evidence levels.3 Several meta-analyses or network meta-analyses of treatments for open-angle glaucoma (OAG) have emerged in this decade;4–12 however, there have been no meta-analyses of ocular hypotensive efficacy with prostaglandin analogs (PGAs) in Japanese patients with NTG.
Two epidemiological studies conducted by the Japan Glaucoma Society found that the main type of glaucoma in Japan was NTG.13,14 However, the majority of studies of treatments for glaucoma investigated their ocular hypotensive effects in patients of primary OAG or ocular hypertension, while comparative studies of ocular hypotensive effects in patients with NTG were scarce. An integrated analysis of single-arm studies of patients with NTG may be useful to assess the IOP-lowering profiles in this population. Furthermore, to our knowledge, no meta-analyses of IOP-lowering medications have been conducted in Japanese patients with NTG.
PGAs are the first-line therapy for the treatment of glaucoma due to their potent ocular hypotensive effect and a favorable safety profile, particularly in terms of systemic side effects.1 Currently, PGAs available in Japan include unoprostone (launched in October 1995), latanoprost (May 1999), travoprost (October 2007), tafluprost (December 2008), and bimatoprost (October 2009). As these PGAs have been on the market for over 8 years, the number of research articles regarding PGA monotherapy has decreased in the last few years compared with when the medications were first launched. There is a need to evaluate PGAs in patients with NTG.
In this paper, the literature surrounding the use of PGAs in Japanese patients with NTG was evaluated to provide evidence for ophthalmologists treating these patients.
Ethics approval and informed consent
This article was based on previously conducted studies and does not contain any studies with human participants or animals performed by any of the authors. Thus, informed consent was not needed.
Materials and methods
Study selection
Identification
In February 2018, the databases PubMed, Embase, Pro-Quest, JAPICDOC (Japan Pharmaceutical Information Center, Tokyo, Japan), and JMEDPlus (Japan Science and Technology Agency, Kawaguchi, Japan) were searched for English and Japanese publications of ocular hypotensive therapies in Japanese patients with NTG who were naïve to medication therapy or newly started on medications, with a comprehensive washout period. Search terms in English included ([unoprostone or latanoprost or travoprost or bimatoprost or tafluprost] and [glaucoma] and [Japan or Japanese]). Search terms in Japanese were slightly modified (“glaucoma” was restricted in “disease” field in JAPICDOC, and material designation was restricted to “article” or “short article” in JMEDPlus).
Screening
Articles were screened by their abstract and eliminated using the following criteria: abstract absent, nonclinical study (animal, cell, or organ), patients with NTG not included, Japanese patients not included, review article or case report with small sample size (n<10), abstracts of conferences or meetings, IOP not measured, absence of PGA monotherapy, PGA used as the switching therapy with no washout period. To eliminate overestimation of the efficacy of PGAs, long-term studies (exceeding 7 months) were excluded as patients who completed extended studies may be good responders to their medications.
Eligibility
Two categories were defined, and full-text articles were assessed for eligibility accordingly. Category 1 included patients with NTG who had reduced IOP values; Category 2 described patients with NTG who had IOP values at predosing and postdosing of the final evaluation points. If patients with OAG were included, the data of the stratified NTG group was needed.
Statistical analyses were conducted using JMP version 13.2 (SAS Institute Inc., Cary, NC, USA).
Category 1
The value of IOP reduction at postdosing final evaluation point was extracted from all eligible articles. From the statistical data (mean, SD or standard error of the mean [SEM]), the total mean, and variance for each medication was calculated. The weighted ocular hypotensive efficacy was calculated.
Category 2
IOP data at predosing and postdosing final evaluation points were extracted from eligible articles. If IOP values were reported at multiple time points postdosing, the longest time point was chosen. A scatter plot analysis was performed and the regression equation was calculated for each medication. The fitting of each regression equation was evaluated by the least squares method.
Results
Study selection
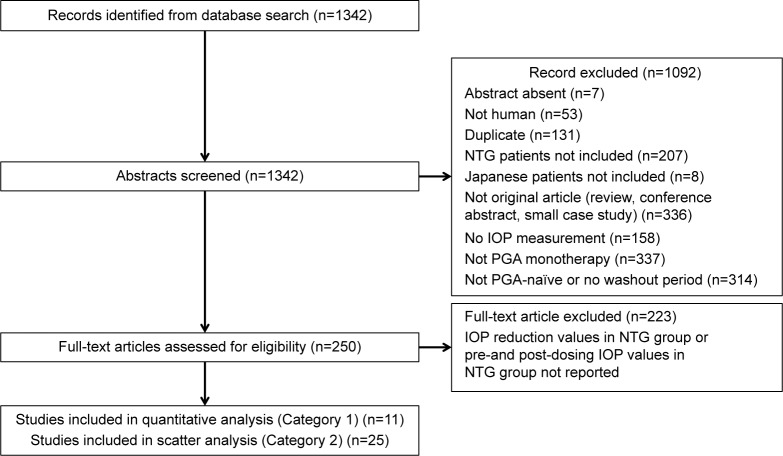
The literature search resulted in 470 hits in Embase, 268 in PubMed, 303 in JDream III/JAPICDOC, and 574 in JDream III/JMEDPlus (1,342 articles were defined as the original data set). After excluding duplicate data and articles with no abstract, 250 articles, which fit the eligibility criteria, remained. Twenty-seven articles were included in the final analysis (11 articles in Category 1 and 25 articles in Category 2; Figure 1). In Category 1, there were three articles for latanoprost,15–17 two articles for travoprost,18,19 one article for bimatoprost,20 five articles for tafluprost21–25 (Table 1), and 0 articles for unoprostone. In Category 2, the number of articles for each PGA was as follows: 14 for latanoprost,15,16,26–37 seven for travoprost,18,19,26,38–41 three for bimatoprost,20,26,40 seven for tafluprost,21–23,25,26,39,40 and 0 articles for unoprostone (Table 2).
Figure 1.
Selection of articles outlined according to PRISMA guidelines.
Notes: Some articles covered multiple topics that were excluded.
Abbreviations: IOP, intraocular pressure; NTG, normal-tension glaucoma; PGA, prostaglandin analogs.
Table 1.
List of studies included in Category 1
| Study | PGA | Number of patients | Evaluation period | Study design | Endpoint(s) |
|---|---|---|---|---|---|
| Naito et al18 | Travoprost | 30 | 12 weeks | Single-arm | IOP |
| Inoue et al15 | Latanoprost | 18 | 3 months | Single-arm | IOP |
| Okada et al21 | Tafluprost | 42 | 6 months | Single-arm | IOP |
| Sone et al22 | Tafluprost | 38 | 12 weeks | Single-arm | IOP |
| Arai et al20 | Bimatoprost | 54 | 6 months | Single-arm | IOP |
| Nakano et al23 | Tafluprost | 41 | 12 weeks | Single-arm | IOP |
| Miyagawa et al24 | Tafluprost | 30 | 12 weeks | Single-arm | IOP |
| Kuwayama and Komemushi25 | Tafluprost | 48 | 4 weeks | RCT | IOP |
| Nagashima et al19 | Travoprost | 66 | 6 months | Single-arm | IOP |
| Kimura et al16 | Latanoprost | 43 | 3 months | Single-arm | IOP |
| Iwata et al17 | Latanoprost | 42 | 12 weeks | Single-arm | IOP |
Abbreviations: IOP, intraocular pressure; PGA, prostaglandin analog; RCT, randomized controlled trial.
Table 2.
List of studies included in Category 2
| Study | PGA | Number of patients | Evaluation period | Study design | Endpoint(s) |
|---|---|---|---|---|---|
| Naito et al18 | Travoprost | 30 | 12 weeks | Single-arm | IOP reduction |
| Inoue et al26 | Latanoprost | 40 | 3 months | Four-arm | IOP |
| Travoprost | 64 | ||||
| Tafluprost | 52 | ||||
| Bimatoprost | 53 | ||||
| Mizoue et al38 | Travoprost | 103 | 12 weeks | Single-arm | IOP |
| Ko et al35 | Latanoprost | 60 | 3 months | Single-arm | IOP |
| Mizoguchi et al39 | Tafluprost | 90 | 12 weeks | Crossover | Diurnal IOP |
| Travoprost | 90 | ||||
| Tanabe et al40 | Travoprost | 49 | 3 months | Non-RCT | IOP |
| Tafluprost | 38 | ||||
| Bimatoprost | 27 | ||||
| Arai et al20 | Bimatoprost | 54 | 6 months | Single-arm | IOP |
| Nakano et al23 | Tafluprost | 41 | 12 weeks | Single-arm | IOP |
| Inoue et al15 | Latanoprost | 18 | 3 months | Single-arm | IOP |
| Okada et al21 | Tafluprost | 42 | 6 months | Single-arm | IOP |
| Sone et al22 | Tafluprost | 38 | 12 weeks | Single-arm | IOP |
| Nagashima et al19 | Travoprost | 66 | 6 months | Single-arm | IOP |
| Sunanaga and Kawano36 | Latanoprost | 52 | 6 months | Single-arm | IOP |
| Nomura et al41 | Travoprost | 17 | 1 or 2 months | Single-arm | Diurnal IOP |
| Kuwayama and Komemushi25 | Tafluprost | 48 | 4 weeks | RCT (2 arms) | IOP |
| Sugiyama et al37 | Latanoprost | 15 | 2 months | Run-in of non-RCT | ONH blood flow |
| Nakamoto and Yasuda27 | Latanoprost | 20 | 8 weeks | Run-in of RCT | Diurnal IOP |
| Ishibashi et al32 | Latanoprost | 22 | More than 12 weeks | Single-arm | Diurnal IOP |
| Hashimoto et al33 | Latanoprost | 32 | 4 weeks | Single-arm | IOP |
| Ishibashi et al31 | Latanoprost | 16 | More than 3 months | Single-arm | IOP |
| Hashimoto et al34 | Latanoprost | 21 | 6 months | Single-arm | IOP |
| Nakamoto et al30 | Latanoprost | 25 | More than 8 weeks | RCT (2 arm) | Diurnal IOP |
| Nakamoto et al29 | Latanoprost | 23 | More than 8 weeks | Single-arm | Diurnal IOP |
| Kimura et al16 | Latanoprost | 43 | 3 months | Single-arm | IOP |
| Nakamoto et al28 | Latanoprost | 25 | 4 weeks | Single-arm | IOP and ONH blood flow |
Abbreviations: IOP, intraocular pressure; ONH, optic nerve head; PGA, prostaglandin analog; RCT, randomized controlled trial.
Ocular hypotensive efficacy
In Category 1, bimatoprost had the strongest ocular hypotensive efficacy and latanoprost the weakest (Figure 2). The mean ± SD change in IOP was −3.03±2.18 mmHg with latanoprost, −3.33±1.79 mmHg with travoprost, −3.05±1.99 mmHg with tafluprost, and −4.00±1.90 mmHg with bimatoprost. Latanoprost, travoprost, and tafluprost had similar levels of ocular hypotensive efficacy and all three were weaker than bimatoprost.
Figure 2.
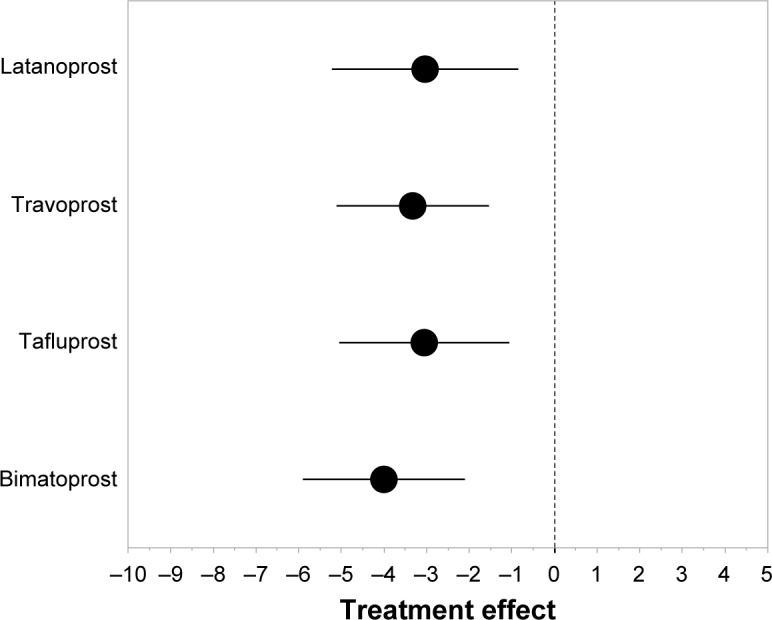
Weighted ocular hypotensive efficacy of prostaglandin analogs calculated using data from the studies in Category 1.
Note: Data are presented as weighted mean ± SD.
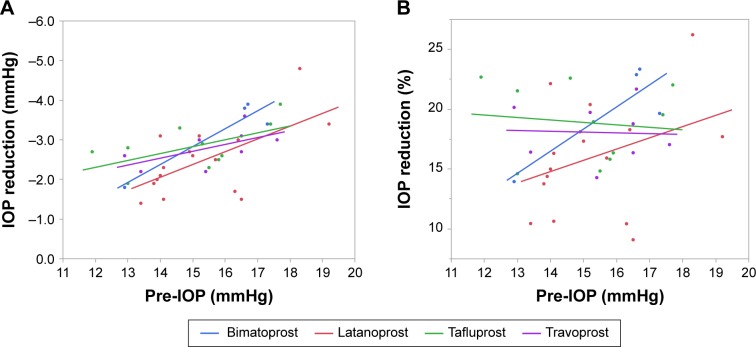
In Category 2, the ocular hypotensive efficacy of all PGAs was dependent on pretreatment IOP values (Figure 3). The r2 values from linear regression analysis ranged between 0.85 (latanoprost) and 0.93 (travoprost). The slopes of linear regression of all PGAs were similar.
Figure 3.
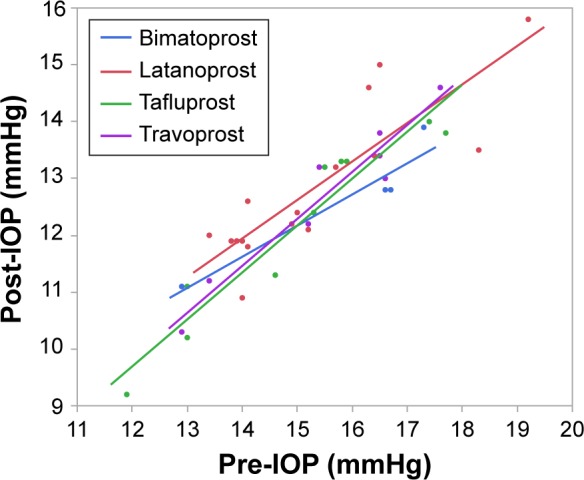
Scatter plot analysis of studies in Category 2 as a function of predosing IOP values.
Abbreviation: IOP, intraocular pressure.
While the slopes were similar for all PGAs, their ocular hypotensive effects had different profiles (Figure 4A and B). The ocular hypotensive efficacy of latanoprost was weaker than that of other drugs, both in terms of absolute and percentage IOP reductions. The slope was steepest with bimatoprost, while the slopes for travoprost and tafluprost were shallower. The ocular hypotensive efficacies of tafluprost and travoprost were stronger than that of latanoprost, especially at lower predosing IOP values. Furthermore, the ocular hypotensive efficacy profiles of tafluprost and travoprost were almost identical.
Figure 4.
Scatter plot analysis of studies in Category 2 as a function of (A) IOP reduction and (B) percentage IOP reduction.
Abbreviation: IOP, intraocular pressure.
The percentage IOP reductions were between 15% and 20% with different PGAs in patients with NTG (Figure 4B). The ocular hypotensive efficacies of travoprost and tafluprost followed an almost flat pattern, while those of bimatoprost and latanoprost were dependent on predosing IOP. Bimatoprost was associated with a steeper slope than latanoprost. The ocular hypotensive efficacies of tafluprost and travoprost were greater than that of latanoprost, especially at lower predosing IOP values. In the study in which predosing IOP was higher (17–18 mmHg), the IOP reduction was almost same with latanoprost, travoprost, and tafluprost. On the other hand, in the study in which predosing IOP was lower (12–15 mmHg), the IOP reduction with latanoprost was weaker than with travoprost or tafluprost.
Discussion
To our knowledge, this is the first literature review of studies using PGAs in Japanese patients with NTG. All PGAs appeared to lower IOP in this population; the magnitude of IOP reduction had a positive relationship with predosing IOP values; and the IOP reduction profiles of the PGAs were slightly different in terms of their IOP-lowering ratio.
The IOP-lowering effect profiles were different for the investigated PGAs. Latanoprost, travoprost, and tafluprost were effective in patients with NTG, while travoprost and tafluprost were more effective in patients with lower baseline IOP (≤12–14 mmHg). The reason for this difference is unclear as it was revealed from the literature review and not the evaluating study. Among the five PGAs, there are differences in structure and binding affinity for prostanoid FP and other receptors.42 This is most likely due to the difference in the chemical structure (-prost or -prostone), functional group at position 15 (ketone, hydroxyl, difluorine), and the nature of the alpha chain (ester or amide). Bimatoprost has a different receptor affinity compared with other PGAs in that it binds to chimeric receptors of ordinary FP receptor and splice-variant FP receptor,43 while other prost-type PGAs bind to FP receptors.44 These differences may have contributed to the different IOP-reducing profiles of PGAs. Some genetic polymorphisms in prostanoid receptors correlate to the difference in the efficacy of latanoprost.45–49 These polymorphisms may cause the differences in the 3D-structure of receptors and affinity of each PGA to those receptors. Thus, these genetic polymorphisms have also contributed to the different IOP-reducing profiles of PGAs.
The main limitation of this literature review was that the study designs of the chosen articles were not identical and comprised of non-RCTs. The study designs varied in each study, especially for evaluation time points. For the purpose of evaluating the IOP-lowering profiles of PGAs, a large number of studies were required; therefore, there were no restrictions on study design. In the real-world practice of glaucoma management, the disease profiles of patients often vary dramatically. Clinical trials during a medication’s pre-launch period were often conducted with restrictive criteria and many patients, such as those with no serious complicating diseases, were excluded. While meta-analyses of RCTs provide the highest level of evidence, information based on these analyses may be insufficient for daily practice. In this literature review, patients who may not typically fit the inclusion criteria of RCTs were also included, and therefore it may help ophthalmologists translate the conclusions to real-world patients with NTG. Furthermore, the weighting of the number of patients was not considered in scatter plot and regression analyses, rather the averages of the data were used. Despite the critical nature of sample size weighting in meta-analyses, this was not considered in this literature review. This was due to the lack of estimation of data variance in the scatter plot analysis. Pooled analyses of selected studies in this review were not performed as original data sets for each study were not accessible; therefore, only averages were used for all data.
Conclusion
The five PGAs evaluated in this review had different ocular hypotensive efficacy. Ophthalmologists should understand and consider individual profiles of the available PGAs in order to select the optimal medication for patients with NTG.
Acknowledgments
We would like to thank Yuko Noda of Medical Tribune Inc. for article screening and data collection from each article. This article screening and data collection was funded by Santen Pharmaceutical Co., Ltd. We would like to thank Mimi Chan, PhD, of inScience Communications, Springer Healthcare, for English and technical editing of the manuscript. This medical writing assistance was funded by Santen Pharmaceutical Co., Ltd. The editing of this review and all article processing charges was funded by Santen Pharmaceutical Co., Ltd. All authors interpreted the data and wrote the manuscript together with the sponsor’s medical writing services. Tafluprost is a proprietary compound of Santen Pharmaceutical Co., Ltd.
Footnotes
Author contributions
All authors contributed to data analysis, drafting and revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
YT, KS, and MH are employees of Santen Pharmaceutical Co., Ltd. TF has received an advisor fee from Santen Pharmaceutical Co., Ltd. The authors report no other conflicts in this work.
References
- 1.Japan Glaucoma Society . Guideline for Glaucoma Practice. 3rd ed. 1. Vol. 116. Tokyo: Japan Glaucoma Society; 2012. [Accessed March 09, 2018]. pp. 3–46. Available from: http://www.ryokunaisho.jp/guidelines/guidelines_all.pdf. [PubMed] [Google Scholar]
- 2.Lee DA, Higginbotham EJ. Glaucoma and its treatment: a review. Am J Health Syst Pharm. 2005;62(7):691–699. doi: 10.1093/ajhp/62.7.691. [DOI] [PubMed] [Google Scholar]
- 3.OCEBM Levels of Evidence Working Group . The Oxford 2011 Levels of Evidence. Oxford: Oxford Centre for Evidence-Based Medicine; 2011. [Google Scholar]
- 4.Denis P, Lafuma A, Khoshnood B, Mimaud V, Berdeaux G. A meta-analysis of topical prostaglandin analogues intra-ocular pressure lowering in glaucoma therapy. Curr Med Res Opin. 2007;23(3):601–608. doi: 10.1185/030079907X178720. [DOI] [PubMed] [Google Scholar]
- 5.Aptel F, Cucherat M, Denis P. Efficacy and tolerability of prostaglandin-timolol fixed combinations: a meta-analysis of randomized clinical trials. Eur J Ophthalmol. 2012;22(1):5–18. doi: 10.5301/ejo.5000009. [DOI] [PubMed] [Google Scholar]
- 6.Cheng JW, Cheng SW, Gao LD, Lu GC, Wei RL. Intraocular pressure-lowering effects of commonly used fixed-combination drugs with timolol: a systematic review and meta-analysis. PLoS One. 2012;7(9):e45079. doi: 10.1371/journal.pone.0045079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Quaranta L, Biagioli E, Riva I, et al. Prostaglandin analogs and timolol-fixed versus unfixed combinations or monotherapy for open-angle glaucoma: a systematic review and meta-analysis. J Ocul Pharmacol Ther. 2013;29(4):382–389. doi: 10.1089/jop.2012.0186. [DOI] [PubMed] [Google Scholar]
- 8.Wang YQ, Wang X, Liu P. Meta analysis about the efficacy and safety of anti-ocular hypertension eye drops without benzalkonium chloride. Asian Pac J Trop Med. 2013;6(12):1004–1008. doi: 10.1016/S1995-7645(13)60181-9. [DOI] [PubMed] [Google Scholar]
- 9.Lou H, Wang H, Zong Y, Cheng JW, Wei RL. Efficacy and tolerability of prostaglandin-timolol fixed combinations: an updated systematic review and meta-analysis. Curr Med Res Opin. 2015;31(6):1139–1147. doi: 10.1185/03007995.2015.1039504. [DOI] [PubMed] [Google Scholar]
- 10.Li T, Lindsley K, Rouse B, et al. Comparative effectiveness of first-line medications for primary open-angle glaucoma: a systematic review and network meta-analysis. Ophthalmology. 2016;123(1):129–140. doi: 10.1016/j.ophtha.2015.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li J, Lin X, Yu M. Meta-analysis of randomized controlled trials comparing latanoprost with other glaucoma medications in chronic angle-closure glaucoma. Eur J Ophthalmol. 2015;25(1):18–26. doi: 10.5301/ejo.5000506. [DOI] [PubMed] [Google Scholar]
- 12.Stewart WC, Konstas AG, Nelson LA, Kruft B. Meta-analysis of 24-hour intraocular pressure studies evaluating the efficacy of glaucoma medicines. Ophthalmology. 2008;115(7):1117–1122. doi: 10.1016/j.ophtha.2007.10.004. [DOI] [PubMed] [Google Scholar]
- 13.Iwase A, Suzuki Y, Araie M, et al. Tajimi Study Group, Japan Glaucoma Society The prevalence of primary open-angle glaucoma in Japanese: the Tajimi Study. Ophthalmology. 2004;111(9):1641–1648. doi: 10.1016/j.ophtha.2004.03.029. [DOI] [PubMed] [Google Scholar]
- 14.Yamamoto S, Sawaguchi S, Iwase A, et al. Primary open-angle glaucoma in a population associated with high prevalence of primary angle-closure glaucoma: the Kumejima Study. Ophthalmology. 2014;121(8):1558–1565. doi: 10.1016/j.ophtha.2014.03.003. [DOI] [PubMed] [Google Scholar]
- 15.Inoue K, Masumoto M, Wakakura M, Tomita G. Ocular hypotensive effects and safety of latanoprost without benzalkonium hydrochloride (article in Japanese) Atarashii Ganka. 2011;28(11):1635–1639. [Google Scholar]
- 16.Kimura H, Nozaki M, Ogura Y, Kimura A, Arimura K, Tanihara H. Ocular hypotensive effect of latanoprost monotherapy for untreated glaucomas (article in Japanese) Rinsho Ganka. 2003;57(5):700–704. [Google Scholar]
- 17.Iwata S, Endou Y, Saitou H, et al. Efficacy of latanoprost 0.005% once daily in normal-tension glaucoma (article in Japanese) Atarashii Ganka. 2003;20(5):709–711. [Google Scholar]
- 18.Naito T, Okuma S, Nagayama M, et al. Sustainability of intraocular pressure reduction of travoprost ophthalmic solution in subjects with normal tension glaucoma. Adv Ther. 2016;33(3):435–446. doi: 10.1007/s12325-016-0297-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Nagashima S, Inoue K, Shiokawa M, et al. Hypotensive effect of travoprost ophthalmic solution for normal-tension glaucoma (article in Japanese) Rinsho Ganka. 2010;64(6):911–914. [Google Scholar]
- 20.Arai K, Inoue K, Shiokawa M, Masumoto M, Wakakura M, Tomita G. Short-term effect of bimatoprost ophtalmic solution for normal-tension glaucoma (article in Japanese) Rinsho Ganka. 2011;65(7):1083–1087. [Google Scholar]
- 21.Okada F, Inoue K, Wakakura M, Tomita G. Ocular hypotensive effects and safety of tafluprost in normal-tension glaucoma (article in Japanese) Atarashii Ganka. 2011;28(7):1043–1046. [Google Scholar]
- 22.Sone A, Katsushima H, Funahashi K, Nishino K, Takeda A. Efficacy and safety of 0.0015% tafluprost ophthalmic solution in normal-tension glaucoma (article in Japanese) Atarashii Ganka. 2011;28(4):568–570. [Google Scholar]
- 23.Nakano T, Yoshikawa K, Kimura T, Suzumura H, Nanno M, Noro T. Efficacy and safety of tafluprost in normal-tension glaucoma with intraocular pressure of 16 mmHg or less. Jpn J Ophthalmol. 2011;55(6):605–613. doi: 10.1007/s10384-011-0082-7. [DOI] [PubMed] [Google Scholar]
- 24.Miyagawa Y, Yamazaki H, Nakazawa M. Short-term efficacy of tafluprost monotherapy for untreated glaucoma (article in Japanese) Atarashii Ganka. 2010;27(7):967–969. [Google Scholar]
- 25.Kuwayama Y, Komemushi S, Tafluprost Multi-center Study Group Intraocular pressure lowering effect of 0.0015% tafluprost as compared to placebo in patients with normal tension glaucoma: randomized, double-blind, multicenter, phase III study. Nippon Ganka Gakkai Zasshi. 2010;114(5):436–443. [PubMed] [Google Scholar]
- 26.Inoue K, Setogawa A, Tomita G. Nonresponders to prostaglandin analogs among normal-tension glaucoma patients. J Ocul Pharmacol Ther. 2016;32(2):90–96. doi: 10.1089/jop.2015.0086. [DOI] [PubMed] [Google Scholar]
- 27.Nakamoto K, Yasuda N. Effect of concomitant use of latanoprost and brinzolamide on 24-hour variation of IOP in normal-tension glaucoma. J Glaucoma. 2007;16(4):352–357. doi: 10.1097/IJG.0b013e318033b491. [DOI] [PubMed] [Google Scholar]
- 28.Nakamoto K, Nanno M, Kinohira Y, Murai K, Yasuda N. Intraocular pressure reduction and optic disc changes with latanoprost treatment in normal-tension glaucoma (article in Japanese) Atarashii Ganka. 2001;18(11):1417–1419. [Google Scholar]
- 29.Nakamoto K, Yasuda N, Nanno M, Kinohira Y, Murai K, Fukuda T. Effect of latanoprost on diurnal variations of intraocular pressure in normal-tension glaucoma. Nippon Ganka Gakkai Zasshi. 2003;107(9):530–534. [PubMed] [Google Scholar]
- 30.Nakamoto K, Yasuda N, Nanno M, Fukuda T. Comparison of the effects of latanoprost and timolol gel-forming solution on diurnal variation of intraocular pressure in normal-tension glaucoma. Nippon Ganka Gakkai Zasshi. 2004;108(7):401–407. [PubMed] [Google Scholar]
- 31.Ishibashi S, Hirose N, Tawara A. Effect of latanoprost on diurnal variations in intraocular and ocular perfusion pressure in normal-tension glaucoma (article in Japanese) Atarashii Ganka. 2004;21(12):1693–1696. [Google Scholar]
- 32.Ishibashi S, Hirose N, Tawara A, Kubota T. Effect of latanoprost on the diurnal variations in the intraocular and ocular perfusion pressure in normal tension glaucoma. J Glaucoma. 2006;15(5):354–357. doi: 10.1097/01.ijg.0000212264.96864.3e. [DOI] [PubMed] [Google Scholar]
- 33.Hashimoto N, Hara T, Tsuru T. Selection of therapeutic modality by four-week latanoprost followed by additional timolol-gel ophthalmic solution (article in Japanese) Rinsho Ganka. 2006;60(5):757–759. [Google Scholar]
- 34.Hashimoto T, Hara T, Takahashi Y, Kubota S, Kubota M, Tsuru T. Short and long-term hypotensive effect of timolol-gel and latanoprost instillation in normal-tension glaucoma. Nippon Ganka Gakkai Zasshi. 2004;108(8):477–481. [PubMed] [Google Scholar]
- 35.Ko E, Okamoto Y, Inoue T, Tanihara H. Efficacy of latanoprost as first-line drug and comparison of timolol and dorzolamide in the additive effect for normal tension glaucoma (article in Japanese) Ganka Rinsho Kiyou. 2013;6(7):533–537. [Google Scholar]
- 36.Sunanaga T, Kawano S. Effect of topical latanoprost as the initial treatment for glaucoma (article in Japanese) Rinsho Ganka. 2010;64(2):207–211. [Google Scholar]
- 37.Sugiyama T, Kojima S, Ishida O, Ikeda T. Changes in optic nerve head blood flow induced by the combined therapy of latanoprost and beta blockers. Acta Ophthalmol. 2009;87(7):797–800. doi: 10.1111/j.1755-3768.2008.01460.x. [DOI] [PubMed] [Google Scholar]
- 38.Mizoue S, Nakano T, Fuse N, Iwase A, Matsumoto S, Yoshikawa K. Travoprost with sofZia® preservative system lowered intraocular pressure of Japanese normal tension glaucoma with minimal side effects. Clin Ophthalmol. 2014;8:347–354. doi: 10.2147/OPTH.S57640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mizoguchi T, Ozaki M, Unoki K, Dake Y, Eto T, Arai M. A randomized crossover study comparing tafluprost 0.0015% with travoprost 0.004% in patients with normal-tension glaucoma. Clin Ophthalmol. 2012;6:1579–1584. doi: 10.2147/OPTH.S33414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tanabe Y, Kanno M, Yamashita H. Intraocular pressure-lowering effect of travoprost, tafluprost and bimatoprost in normal tension glaucoma (article in Japanese) Atarashii Ganka. 2012;29(8):1131–1135. [Google Scholar]
- 41.Nomura Y, Nakakura S, Moriwaki M, Takahashi Y, Shiraki K. Effect of travoprost on 24-hour intraocular pressure in normal tension glaucoma. Clin Ophthalmol. 2010;4:643–647. doi: 10.2147/opth.s10521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Aihara M. Prostaglandin analogues (article in Japanese) Atarashii Ganka. 2010;27(10):1347–1356. [Google Scholar]
- 43.Liang Y, Woodward DF, Guzman VM, et al. Identification and pharmacological characterization of the prostaglandin FP receptor and FP receptor variant complexes. Br J Pharmacol. 2008;154(5):1079–1093. doi: 10.1038/bjp.2008.142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bean GW, Camras CB. Commercially available prostaglandin analogs for the reduction of intraocular pressure: similarities and differences. Surv Ophthalmol. 2008;53(Suppl 1):S69–S84. doi: 10.1016/j.survophthal.2008.08.012. [DOI] [PubMed] [Google Scholar]
- 45.Sakurai M, Higashide T, Takahashi M, Sugiyama K. Association between genetic polymorphisms of the prostaglandin F2alpha receptor gene and response to latanoprost. Ophthalmology. 2007;114(6):1039–1045. doi: 10.1016/j.ophtha.2007.03.025. [DOI] [PubMed] [Google Scholar]
- 46.Sakurai M, Higashide T, Ohkubo S, Takeda H, Sugiyama K. Association between genetic polymorphisms of the prostaglandin F2α receptor gene, and response to latanoprost in patients with glaucoma and ocular hypertension. Br J Ophthalmol. 2014;98(4):469–473. doi: 10.1136/bjophthalmol-2013-304267. [DOI] [PubMed] [Google Scholar]
- 47.Gao LC, Wang D, Liu FQ, et al. Influence of PTGS1, PTGFR, and MRP4 genetic variants on intraocular pressure response to latanoprost in Chinese primary open-angle glaucoma patients. Eur J Clin Pharmacol. 2015;71(1):43–50. doi: 10.1007/s00228-014-1769-8. [DOI] [PubMed] [Google Scholar]
- 48.Ussa F, Fernandez I, Brion M, et al. Association between SNPs of metalloproteinases and prostaglandin F2α receptor genes and latanoprost response in open-angle glaucoma. Ophthalmology. 2015;122(5):1040–1048. doi: 10.1016/j.ophtha.2014.12.038. [DOI] [PubMed] [Google Scholar]
- 49.Liu H, Yang ZK, Li Y, Zhang WJ, Wang YT, Duan XC. ABCB1 variants confer susceptibility to primary open-angle glaucoma and predict individual differences to latanoprost treatment. Biomed Pharmacother. 2016;80:115–120. doi: 10.1016/j.biopha.2016.02.028. [DOI] [PubMed] [Google Scholar]