Original citation: J Clin Invest. 2011;121(3):941–955. https://doi.org/10.1172/JCI43584
Citation for this corrigendum: J Clin Invest. 2018;128(10):4742–4743. https://doi.org/10.1172/JCI123039
The Editors recently posted an Expression of Concern for this article following notification from an investigative committee at Duke University that a research technician in the animal pulmonary physiology laboratory fabricated and/or falsified flexiVent data reported in Figures 1A, 2A, and 7D of this paper. The authors have now repeated these experiments, and the journal has published an updated online version of the article with new experimental data in Figures 1A and 2A. The airway hyperresponsiveness data in the original version of Figure 7D were not reliable, and the corrected version of the figure does not include these data. In addition, the journal has published an online version of the original article with the unreliable data crossed out (Supplemental File, Redaction). In that version, Supplemental Figure 3, which supported the original Figure 1A, is no longer referenced. The authors have provided a detailed description of the new experiments below.
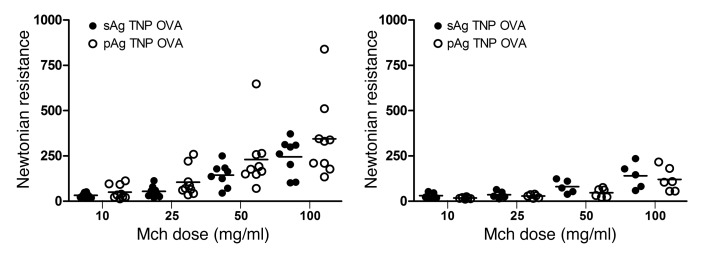
Figure 1A.
Figure 2A.
In our published work, we had reported that inhalable particulate allergens are more potent in causing allergic asthma than soluble allergens in sensitized mice when challenged via the airways. We based our conclusions on three independent readouts, two of which, eosinophil recruitment and histopathological examination of the lung cross sections, were undertaken in the Abraham laboratory by then–graduate student Cong Jin and are not in question. We believe that these two studies, which represent standard assays to assess lung inflammation, closely correlate with each other and therefore are sufficient to support the conclusions of this paper. The third assay, involving assessment of airway hyperresponsiveness using a flexiVent apparatus, was undertaken in a separate laboratory, that of Mike Foster, and was performed by Erin Potts.
Over a year ago, Duke University became aware that mouse data generated in the Foster lab was questionable because Erin Potts, the technician who undertook all of the flexiVent experiments, was found to have been falsifying data. Upon hearing of the possibility that our data may have been compromised, we informed the JCI and also indicated that we would attempt to redo the flexiVent studies to ensure the veracity of our published work. The Board gave us permission to commence these studies and also published an Expression of Concern in the journal.
With the help of a technician and the use of a flexiVent apparatus supplied by Duke University, we undertook many studies in an attempt to recapitulate our original flexiVent data. On many occasions, we found the flexiVent instrument to be erratic, producing inconsistent results. For example, while we were able to reproduce some of the published data, we could not reproduce others, even though the only change we made was altering the inhalable allergen employed for the study. In those cases where we were unable to reproduce our published studies, we could not get the positive and negative controls to work (even after numerous iterations), so we are unable to draw any conclusions regarding whether or not our original hypothesis was valid. The corrected Figure 1A, which appears below, includes flexiVent data showing sensitized mice challenged with soluble and particulate house dust mite (HDM) allergen. We show that at the highest concentration of methacholine, we were able to observe a significant difference between particulate and soluble allergens. These data support the original conclusions drawn from Figure 1A of our published manuscript. The corrected Figure 2A, which appears below, shows the lack of airway responses to particulate and soluble TNP OVA allergen in mast cell–deficient (Sash) mice. These data also confirm results in Figure 2A of the published manuscript. However, when we substituted HDM allergen with ragweed allergen (RWD), we could not reproduce any of the RWD airway hyperresponses reported in Figure 1A of the published paper, as in many cases, the airway responses to the negative controls were comparable to those to the positive controls (data not shown).
We ascribe some of our difficulties to the notoriously poor airway responses exhibited by C57BL/6 mice (1, 2) and our inability to switch mouse strains, as all of the other data were generated in this mouse strain. Another contributing factor is our inexperience with the flexiVent instrument, and we were unable to successfully troubleshoot problems we faced. As a result of this finding, we are correcting our article to remove the airway hyperresponsiveness data.
Supplementary Material
Version 1. 10/01/2018
Print issue publication
Footnotes
See the related article at Particulate allergens potentiate allergic asthma in mice through sustained IgE-mediated mast cell activation.
References
- 1.Takeda K, Haczku A, Lee JJ, Irvin CG, Gelfand EW. Strain dependence of airway hyperresponsiveness reflects differences in eosinophil localization in the lung. Am J Physiol Lung Cell Mol Physiol. 2001;281(2):L394–L402. doi: 10.1152/ajplung.2001.281.2.L394. [DOI] [PubMed] [Google Scholar]
- 2.Zhang Y, Lamm WJ, Albert RK, Chi EY, Henderson WR, Jr, Lewis DB. Influence of the route of allergen administration and genetic background on the murine allergic pulmonary response. Am J Respir Crit Car Med. 1997;155(2):661–669. doi: 10.1164/ajrccm.155.2.9032210. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.