Abstract
To reduce costs of biological disease-modifying antirheumatic drugs (bDMARDs), we evaluated the efficacy of repeated etanercept (ETN) discontinuation and restarting in rheumatoid arthritis (RA) patients in a case–control study.
Thirty-one bDMARD-naive RA patients with moderate to high disease activity received ETN until low disease activity (LDA) was achieved, after which ETN was discontinued. Upon flaring, ETN was readministered with observation every 2 months for 2 years, and radiographically evaluated in comparison with a historical control group treated continuously with ETN. Statistical methods including Fisher exact test, analysis of variance (ANOVA), Kruskal–Wallis test, multiple regression analysis, and Student t test were conducted as appropriate.
Thirteen patients with inadequate response to ETN were withdrawn from the study, and 5 had no flare-up after ETN discontinuation. In the remaining 13 patients, ETN was used on-demand to maintain LDA. Multivariate analysis revealed that MTX was significantly correlated with ETN. All 13 patients achieved LDA at final follow-up. Although joint damage progressed in patients using ETN on-demand, structural damage progression in the on-demand group was not significantly different from that in controls.
On-demand use of ETN for flaring reduced disease activity but not structural damage in 50% of patients (though not significantly). However, inhibition of joint damage was achieved in 50% of patients after 2 years, supporting on-demand use of ETN as a treatment option for patients with RA who cannot afford bDMARD or targeted synthetic DMARD therapy.
Keywords: discontinuation, disease flare, etanercept, rheumatoid arthritis, structural damage
1. Introduction
Successful treatment with synthetic and biological disease-modifying antirheumatic drugs (DMARDs) in line with the concept of “treat to target” has been shown to prevent joint damage and physical disability in patients with rheumatoid arthritis (RA).[1–3] To achieve treatment targets, use of biological DMARDs (bDMARDs) should be considered, as significant evidence of their efficacy has been demonstrated in recent years.[2,3] Upon achievement of treatment targets and persistent remission, tapering of bDMARDs or dose reduction of conventional synthetic DMARDs (csDMARDs) should be considered according to European League Against Rheumatism (EULAR) recommendations.[3]
One reason to consider tapering bDMARDs is their high cost, both at a national level as well as for patients. In the United States, the total annual cost for the treatment of RA has increased threefold since the introduction of bDMARDs.[4] Among other arguments for the tapering of bDMARDs, Sokka et al[5] reported that the treatment target of remission/low disease activity (LDA) and good functional status can be reached in RA patients using expensive as well as less-expensive antirheumatic drugs. However, in the BeSt trial, most aggressive treatment arm with use of infliximab (IFX) achieved significantly better quality of life, although treatment costs were prohibitive. However, the authors suggested that these costs would be compensated by reduced losses in patient productivity in the long term.[6] In this regard, discontinuation of bDMARDs in RA patients achieving the treatment target is not warranted.[6] Moreover, in the most aggressive IFX treatment arm, a high rate of bDMARD tapering was achieved.[7] Several studies have reported the outcomes of bDMARD discontinuation in RA patients with sustained clinical remission or LDA.[7,8] In these studies, disease activity flared in some patients who discontinued bDMARDs and it was necessary to restart treatment. It is therefore necessary to determine the efficacy of readministration of bDMARDs in this patient population. Conversely, if readministration of discontinued bDMARDs is shown to be as effective as treatment before discontinuation, bDMARDs could be administered only when flares occur in RA patients who cannot afford the cost of continuous bDMARD treatment.
In clinical practice, patients with RA are reluctant to use bDMARDs because of their high cost, even when administered under the Japanese health insurance system.[9] Patients with RA who cannot afford bDMARDs at the scene of clinics will lose the therapeutic window of opportunity, and joint damage will progress irreversibly.
We conducted a prospective study with historical control patients to examine the sustained efficacy of the bDMARD etanercept (ETN), following its discontinuation when LDA was reached and subsequent readministration in patients with RA. Joint damage was evaluated in these patients at a 2-year follow-up visit.
2. Methods
2.1. Patients
RA patients included in this study were aged 20 years or over with no history of bDMRAD treatment, who met the 1987 American Rheumatism Association classification criteria for RA,[10] and whose disease activity was moderate to high (Disease Activity Score-28 [DAS28]–erythrocyte sedimentation rate [ESR] ≥ 3.2)[11] after receiving methotrexate (MTX) for more than 3 months. Patients with severe joint destruction of the hands and feet were excluded because of the limited ability to obtain radiological measurements for these patients. Patients receiving tacrolimus and/or glucocorticoid at a dose >5 mg/day were also excluded. A total of 32 patients (23 women) were enrolled in this study protocol and provided written informed consent. One patient subsequently discontinued from the study following hospitalization for cerebral infarction before ETN administration. The remaining 31 patients were analyzed as the full analysis set, defined as those who received ETN for at least 4 months.
To compare the extent structural damage among this study population, a series of 16 RA patients treated with full use of ETN was used as a historical control. These patients were consecutively enrolled at our institute from 2008 to 2010 and have been partially described in our previous paper.[12]
Approval for this study was received from the institutional review board or ethics committee at Osaka City University Medical School (date of issue: December 6, 2010, Registration number: #192), and this clinical trial was registered by UMIN-CTR (http://www.umin.ac.jp/ctr/index-j.htm, UMIN000008164). The institutional review board or ethics committee at each participating institute approved the study protocol, and the study was conducted in accordance with the ethical standards laid down in an appropriate version of the Declaration of Helsinki (as revised in Brazil 2013).
2.2. Study design
This was a prospective, single-arm, multiinstitutional, 2-year study which used historical control patients. The primary endpoint of the study was radiographic nonprogression at 2 years. This prospective study was conducted between February 1, 2011 and December 31, 2013 (final follow-up date was October 23, 2014). Patients were administered ETN at 50 mg/week, and treatment was discontinued when LDA (DAS28-ESR < 3.2) was achieved. If disease activity increased over LDA, ETN was restarted at the same dose, with observation every 2 months. This strategy was maintained for 2 years (Fig. 1). If patients were unable to achieve LDA within 6 months of ETN administration, they were considered to be discontinued from the on-demand use study, although data were collected from these patients thereafter (ETN-on group).
Figure 1.
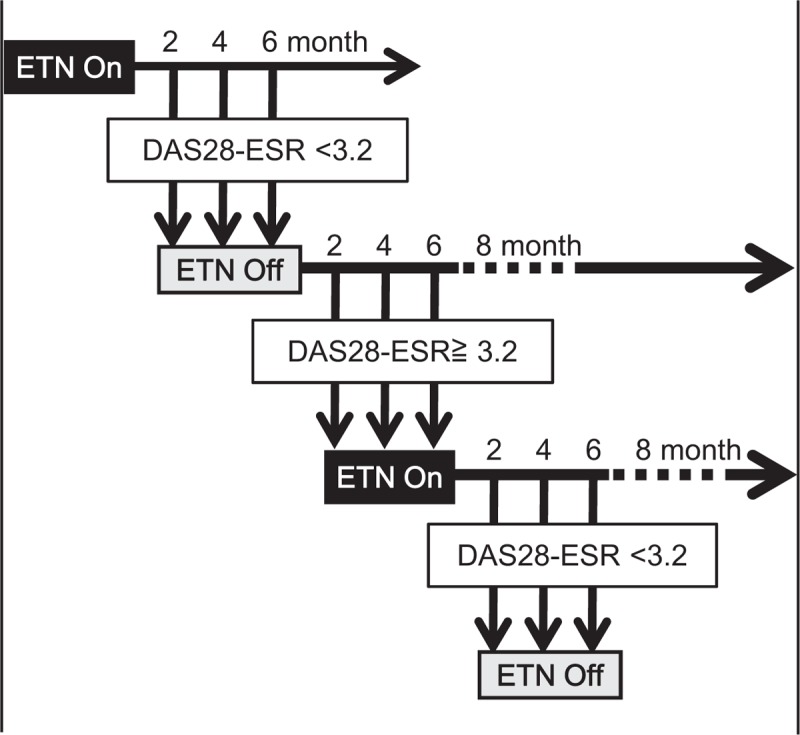
Study protocol. When disease activity score reached low disease activity or remission evaluated by disease activity score-28–erythrocyte sedimentation rate (DAS28-ESR), administration of etanercept (ETN) was discontinued (ETN-Off). If DAS28-ESR became moderate or higher in these patients, ETN was readministered with observation every 2 months (ETN-On). Patients who could not achieve low disease activity within 6 months were considered to be discontinued from the study.
2.3. Evaluation
Every 2 months, we evaluated the efficacy of restarting ETN following a flare in disease activity. Radiographs were obtained at baseline and at the 1- and 2-year visits. Radiographic evaluation was performed by 2 of 3 experienced rheumatologists (KI, MT, YS) using van der Heijde's modified total Sharp score (mTSS; 0–448 points)[13] in a blinded manner. Two physicians who were described in previous sentence masked to the treatment regimens and imaging sequence separately read the digitized radiographic images. Lack of progression was defined as change in mTSS ≤ 0.5 from baseline, and the nonprogression (structural remission) rate was calculated.
2.4. Statistical analysis
Univariate analysis of baseline characteristics was conducted using Fisher exact test for nominal variables, one-way analysis of variance (ANOVA) for continuous variables with normal distribution, and the Kruskal–Wallis test for continuous variables with nonnormal distribution. The prognostic factors for discontinuation of ETN more than once were identified with multiple regression analysis, using the variables of gender, age, disease duration, anticitrullinated protein antibody positivity, and use of MTX at baseline. Statistical analysis of structural damage from baseline was determined by Fisher exact test. Logistic regression analysis for structural remission at 1 or 2 years was performed with variables of disease duration, DAS28-ESR, MTX dose, and baseline mTSS. The difference of erosion score and joint narrowing score between on-demand group and historical control group was analyzed by Student t test. All statistical analyses were performed using EZR (version 1.28), a graphical user interface for R (version 3.2.0, The R Foundation for Statistical Computing). More precisely, it is a modified version of R commander (version 2.1-7) designed to provide statistical functions frequently used in biostatistics.[14] All statistical analyses were 2-sided and values of P < .05 were considered significant.
3. Results
3.1. Baseline characteristics of patients
Of the full analysis set of 31 patients, 13 patients did not discontinue ETN dosage because LDA was never achieved (ETN-on group). Of the remaining 18 patients, 5 maintained LDA throughout the study period after the first instance of ETN stoppage, indicating that successful discontinuation of ETN was achieved (ETN-off group). Among the other 13 patients, ETN was administered repeatedly according to the protocol (on-demand group) (Table 1).
Table 1.
Baseline patient characteristics.
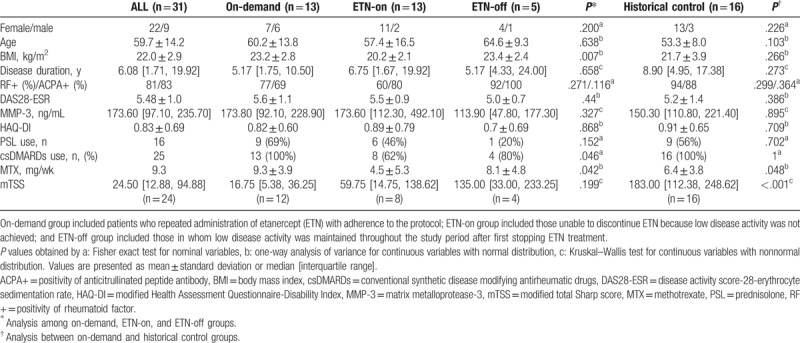
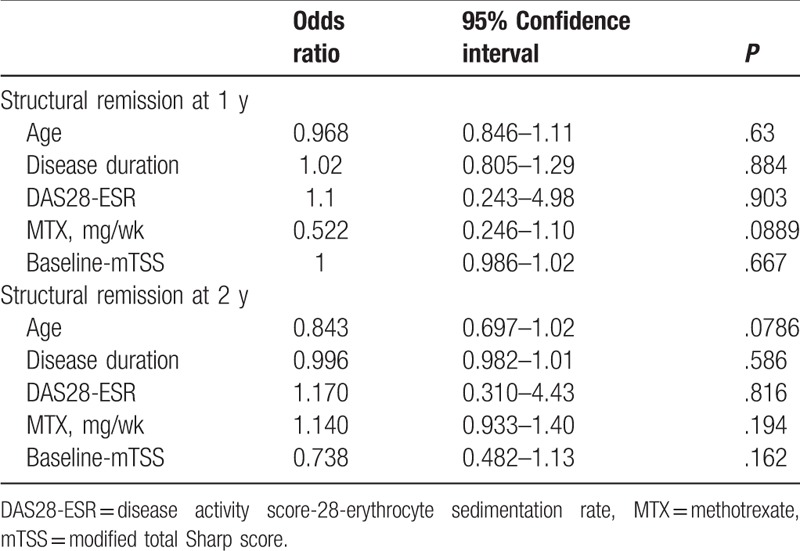
Univariate analysis of the baseline characteristics between the ETN-on group and the other 2 groups revealed that dose of MTX (P = .042), use of any csDMARDs (P = .046), and body mass index (P = .007) were significantly different (Table 1). Multivariate association for the ETN-on group compared to the other 2 groups was performed by logistic regression analysis using 4 factors as independent variables (DAS28-ESR, disease duration, dose of MTX, and ACPA). MTX was significantly correlated with ETN (odds ratio [OR]: 1.29; P = .017), that is, RA patients taking a lower dose of MTX were less likely to discontinue ETN at least once (Table 2). Binomial logistic regression analysis for structural remission at 1 or 2 years was conducted among the on-demand group and the historical control group together. The model including age, baseline mTSS, DAS28-ESR, disease duration, and dose of MTX as explanatory variables, and no independent factors were found to exist (Table 3).
Table 2.
Logistic regression analysis for stop of ETN at least once.
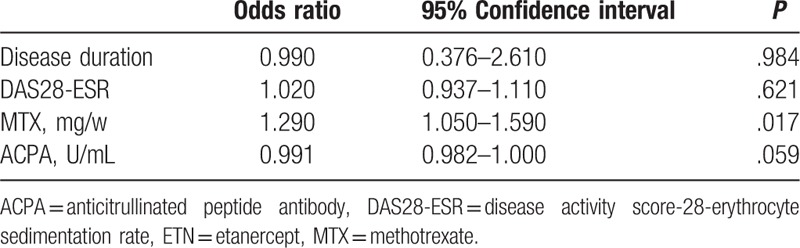
Table 3.
Logistic regression analysis for structural remission at 1 or 2 years.
3.2. Disease activity in on-demand group
The DAS28-ESR courses for all 13 patients in the on-demand group are shown in Fig. 2A. One solid line indicates one DAS28-ESR course of one patient, which contained at least one set of a peak value >3.2 and a trough value <3.2. During the observation period, 21 sets of peak and trough values in 13 patients (one set: 7 patients; 2 sets: 4 patients; 3 sets: 2 patients) were recorded in the on-demand group (Fig. 2B). All patients in the on-demand group had achieved LDA at the end of final ON period (Fig. 2B). Mean duration of recurrence from LDA after discontinuation of ETN was 2.7 ± 1.2 months (median value 2 months; max 6 months, min 2 months) and mean duration of discontinuation of ETN after recurrence was 3.7 ± 2.9 months (median value 2 months; max 14 months, min 2 months). Total duration of ETN withdrawn in the ETN-off group was 62 person-months in the observation period of 88 person-months, and was 102 person-months in a period of 306 person-months in the on-demand group. Thus, ETN was halted for 164 person-months out of the 394 person-months of the entire observation period, corresponding to a 42% reduction in ETN use in this study.
Figure 2.
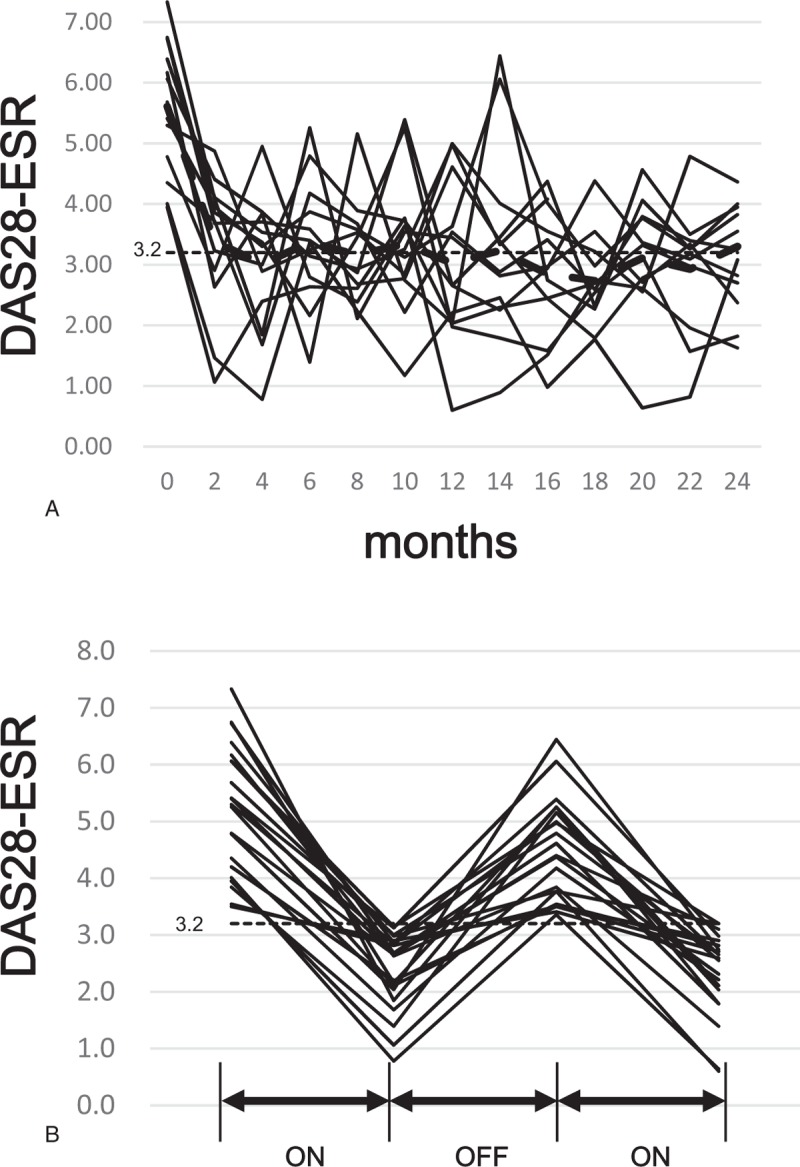
(A) Courses of all individual cases of disease activity score-28–erythrocyte sedimentation rate (DAS28-ESR) in the on-demand group. Thick dotted line indicates the mean DAS28-ESR score. Thin dotted straight transverse line indicates DAS28-ESR = 3.2. (B) Disease activity score-28–erythrocyte sedimentation rate (DAS28-ESR) course for 21 sets in 13 patients in the on-demand group of “start-stop-start-stop,” “ON” indicates the time when etanercept was administered, and “OFF” indicates the time when etanercept was discontinued. All 21 sets achieved low disease activity. During the observation period, 1 set of peak and trough levels was observed in 7 patients, 2 sets in 4 patients, and 3 sets in 2 patients.
3.3. Radiographic evaluation
The interobserver error for the radiographic evaluation was 0.896 (P < .0001), as assessed by Spearman's rank-correlation coefficient. Radiographs of 11 patients in the on-demand group at both baseline and 1 year and of 12 patients at both baseline and 2 years were obtained. At 1 year, 82% of patients in the on-demand group had achieved radiographic remission, a percentage equivalent to that observed in the data from the historical control group, which used a full ETN regimen (Table 4). Change in erosion score for the on-demand group and the historical control group at 1 year was an average of 0.32 ± 9.3 (±SD) and −1.09 ± 1.93 (P = .027; Student t test), and this difference was statistically significant (Table 5). At the 2-year final follow up, the structural remission rate was lower in the on-demand group (Table 4) than in the historical control group, but the difference was not statistically significant (P = .114).
Table 4.
Number of structural remission evaluated by mTSS (difference from baseline).

Table 5.
Erosion and joint narrowing score (difference from baseline).

4. Discussion
Recent RA treatment strategies, such as treat-to-target,[1] have emphasized the achievement of at least LDA as soon as possible in order to avoid further joint damage, especially in patients with poor prognosis, to avoid permanent disabilities.[15] For achievement of LDA at an earlier stage of the disease, it is clear that bDMARDs represent the most potent of the DMARDs.[16] In patients with an inadequate response to MTX and/or other csDMARD strategies, with or without glucocorticoid treatment, bDMARDs administered with MTX is the recommended treatment for RA.[2,3] Therefore, it is appropriate that bDMARDs are used in all patients with RA insofar as they can be afforded.
For patients who cannot afford bDMARDs or targeted synthetic DMARDs (tsDMARDs), an alternative treatment target is required which accepts some degree of structural damage, although this contradicts the principles of treat-to-target. Recent reports of triple therapy with MTX plus sulfasalazine and hydroxychloroquine demonstrated results that were equivalent to combination therapy with bDMARDs.[17–20] However, data on long-term results with triple therapy are required because adhesion to this regiment appears to be suboptimal.[21] Biosimilar DMARDs (bsDMARDs) represent another option for these patients, although even bsDMARDs are more expensive than csDMARDs and most patients may discontinue their use. This study shows that the use of ETN at a reduced dose or at spaced intervals may be a realistic option for patients who cannot afford their continuous use.
Emery et al reported sustained efficacy with ETN tapering in patients with early RA. Furthermore, no significant differences in radiographic progression were observed between a discontinuation group and a reduced ETN dose group after a 52-week observation.[22] Other studies have shown that a reduced dose of ETN was effective in improving clinical measurements in patients with RA throughout the ETN treatment period[12] and after achievement of LDA,[23] although joint destruction progressed.[12,23] Henaux et al[24] mentioned that the risk of losing remission or LDA and radiographic progression on discontinuation of bDMARDs, however, dose reduction of bDMARDs does not increase such risk by the meta-analysis.
In this study, all 13 patients with RA in the on-demand group achieved LDA at the end of final ON period, implying that ETN efficacy was maintained after several instances of discontinuation and restart over at least 2 years. Therefore, on-demand use of ETN seems a suitable choice for treating disease flare only in patients with RA, for the purpose of reducing the total ETN dose.
Full dose, on-demand use of TNF inhibitors raises concerns about the risk of anti-TNF inhibitors antibody production. Readministration of IFX in patients with ankylosing spondylitis after long-term continuous therapy and withdrawal was reported to be generally safe and efficacious.[25] Another report[26] indicated that episodic IFX retreatment in Crohn's disease—specifically, a distant second infusion—was associated with high rates of severe systemic reaction in adults, probably caused by the development of antibodies to IFX.[27] One reason that the retention rate of ETN is longer than that of other TNF inhibitors[28] is that neutralizing antibodies to ETN are less likely to develop because of its receptor-based structure.[27,29] Therefore, ETN is an ideal candidate for on-demand use in patients with RA.[30] Furthermore, given its short half-life compared with other TNF inhibitors, its administration is notably easier to control.[31]
Such characteristics of ETN are highly convenient in clinical practice, as some physicians are willing to use lower doses in patients with RA for reasons of safety and economy.
The structural damage analysis in this study showed that the radiographic nonprogression rate was not inferior to that of ETN 50 mg/week at 1 year (P = 1; Fisher exact probability test), and at 2 years (P = .114; Fisher exact probability test) (Table 4). However, we assumed that patients with RA could not afford to pay the full cost of bDMARDs in the first place. If the on-demand use of ETN for disease flares was not available to these patients, joint damage would be worse in the absence of any biologics. Thus, approximately half of the patients in the on-demand group were deemed to have been rescued from joint destruction.
Our findings must be interpreted in the context of the study limitations. First, the number of patients enrolled in this study was small, although our primary objective was to investigate the efficacy of ETN after several instances of treatment discontinuation and restart. In this regard, the results obtained achieved the aim of this study. Second, the period of this study was limited to 2 years. It is not clear whether the efficacy of ETN in the on-demand group would be maintained in the long term compared with the full ETN regimen group, as the overall retention rate for ETN treatment was reported as approximately 55% at 5 years of treatment.[28] Finally, we used a historical control group, which may have introduced bias.
In conclusion, the on-demand use of ETN for RA flares reduced the disease activity but did not reduce structural damage in half of the patients over 2 years. However, inhibition of joint damage was achieved in half of the patients at a lower cost, supporting the on-demand use of ETN as an option for the treatment of patients with RA who cannot afford expensive bDMARD or tsDMARD therapy. Therefore, those patients who can afford the cost should continue ETN use, while those who cannot should use ETN on-demand.
Acknowledgments
We thank Atsuko Kamiyama and Tomoko Nakatsuka for serving as research coordinators in terms of recruiting participants, collecting data and managing the quality of the data.
Author contributions
Conceptualization: Kentaro Inui, Tatsuya Koike, Tadashi Okano.
Data curation: Kentaro Inui, Tatsuya Koike, Masahiro Tada, Yuko Sugioka, Tadashi Okano, Kenji Mamoto, Akira Sakawa, Kenzo Fukushima, Hiroaki Nakamura.
Formal analysis: Kentaro Inui.
Investigation: Kentaro Inui.
Methodology: Kentaro Inui.
Project administration: Kentaro Inui.
Supervision: Hiroaki Nakamura.
Validation: Kentaro Inui.
Writing – original draft: Kentaro Inui.
Writing – review & editing: Tatsuya Koike.
Kentaro Inui orcid: 0000-0002-8747-7279
Footnotes
Abbreviations: ANOVA = analysis of variance, bDMARDs = biological DMARDs, csDMARDs = conventional synthetic DMARDs, DAS28-ESR = Disease Activity Score-28–erythrocyte sedimentation rate, DMARDs = disease-modifying antirheumatic drugs, ETN = etanercept, EULAR = European League Against Rheumatism, IFX = infliximab, LDA = low disease activity, mTSS = modified total Sharp score, MTX = methotrexate, OR = odds ratio, RA = rheumatoid arthritis, tsDMARDs = targeted synthetic DMARDs.
Conflicts of interest: K.I. has received speakers fee and/or research grant from Chugai Pharmaceutical Co., Ltd, Mitsubishi Tanabe Pharma Co., Astellas Pharma, Inc., Abbvie GK, Eisai Co., Ltd, Takeda Pharmaceutical Co., Ltd, Daiichi Sankyo Co., Ltd, Ono Pharmaceutical Co., Ltd, Janssen Pharmaceutical K.K., Pfizer Japan, Inc., Eisai Co., Ltd, Sanofi K.K., Qol Co., Ltd.
T.K. has received grant fees, research fees, consulting fees, or other remuneration from AbbVie, Astellas Pharma, Inc., Bristol-Myers Squibb, Chugai Pharmaceutical, Eisai, Janssen, Lilly, Mitsubishi Tanabe Pharma Corporation, MSD, Ono Pharmaceutical, Pfizer, Roche, Takeda Pharmaceutical, Teijin Pharma, and UCB.
T.O. received speaking fees from AbbVie.
N.H. has received speakers fee and/or research grant from Chugai Pharmaceutical Co., Ltd, Astellas Pharma, Inc., Nippon Zoki Pharmaceutical Co., Ltd, Daiichi Sankyo Co., Ltd, Eisai Co., Ltd, Takeda Pharmaceutical Co., Ltd, Hisamitsu Pharmaceutical Co., Inc., Ono Pharmaceutical Co., Ltd, Osteopharma, Inc., Teijin Pharma Ltd, Asahi Kasei Pharma Co., Taisho Toyama Pharmaceutical Co., Ltd, Eli Lilly Japan K.K., Pfizer Japan, Inc., and Janssen Pharmaceutical K.K.
All other authors have declared no conflicts of interest.
No specific funding was received from any bodies for this work.
References
- [1].Smolen JS, Breedveld FC, Burmester GR, et al. Treating rheumatoid arthritis to target: 2014 update of the recommendations of an international task force. Ann Rheum Dis 2016;75:3–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Singh JA, Saag KG, Bridges SL, Jr, et al. 2015 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Care Res (Hoboken) 2016;68:1–25. [DOI] [PubMed] [Google Scholar]
- [3].Smolen JS, Landewe R, Bijlsma J, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Ann Rheum Dis 2017;76:960–77. [DOI] [PubMed] [Google Scholar]
- [4].Michaud K, Messer J, Choi HK, et al. Direct medical costs and their predictors in patients with rheumatoid arthritis: a three-year study of 7,527 patients. Arthritis Rheum 2003;48:2750–62. [DOI] [PubMed] [Google Scholar]
- [5].Sokka T, Haugeberg G, Asikainen J, et al. Similar clinical outcomes in rheumatoid arthritis with more versus less expensive treatment strategies. Observational data from two rheumatology clinics. Clin Exp Rheumatol 2013;31:409–14. [PubMed] [Google Scholar]
- [6].van den Hout WB, Goekoop-Ruiterman YP, Allaart CF, et al. Cost-utility analysis of treatment strategies in patients with recent-onset rheumatoid arthritis. Arthritis Rheum 2009;61:291–9. [DOI] [PubMed] [Google Scholar]
- [7].Klarenbeek NB, van der Kooij SM, Guler-Yuksel M, et al. Discontinuing treatment in patients with rheumatoid arthritis in sustained clinical remission: exploratory analyses from the BeSt study. Ann Rheum Dis 2011;70:315–9. [DOI] [PubMed] [Google Scholar]
- [8].Tanaka Y, Takeuchi T, Mimori T, et al. Discontinuation of infliximab after attaining low disease activity in patients with rheumatoid arthritis: RRR (remission induction by Remicade in RA) study. Ann Rheum Dis 2010;69:1286–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Koike T. Treatment of rheumatoid arthritis by molecular-targeted agents: efficacy and limitations. J Orthop Sci 2015;20:951–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Arnett FC, Edworthy SM, Bloch DA, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 1988;31:315–24. [DOI] [PubMed] [Google Scholar]
- [11].Prevoo ML, van’t Hof MA, Kuper HH, et al. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum 1995;38:44–8. [DOI] [PubMed] [Google Scholar]
- [12].Tada M, Koike T, Okano T, et al. Comparison of joint destruction between standard- and low-dose etanercept in rheumatoid arthritis from the Prevention of Cartilage Destruction by Etanercept (PRECEPT) study. Rheumatology (Oxford, England) 2012;51:2164–9. [DOI] [PubMed] [Google Scholar]
- [13].van der Heijde D. How to read radiographs according to the Sharp/van der Heijde method. J Rheumatol 2000;27:261–3. [PubMed] [Google Scholar]
- [14].Kanda Y. Investigation of the freely available easy-to-use software ’EZR’ for medical statistics. Bone Marrow Transplant 2013;48:452–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Moura CS, Abrahamowicz M, Beauchamp ME, et al. Early medication use in new-onset rheumatoid arthritis may delay joint replacement: results of a large population-based study. Arthritis Res Ther 2015;17:197–205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Visser K, Goekoop-Ruiterman YP, de Vries-Bouwstra JK, et al. A matrix risk model for the prediction of rapid radiographic progression in patients with rheumatoid arthritis receiving different dynamic treatment strategies: post hoc analyses from the BeSt study. Ann Rheum Dis 2010;69:1333–7. [DOI] [PubMed] [Google Scholar]
- [17].Moreland LW, O’Dell JR, Paulus HE, et al. A randomized comparative effectiveness study of oral triple therapy versus etanercept plus methotrexate in early aggressive rheumatoid arthritis: the treatment of Early Aggressive Rheumatoid Arthritis Trial. Arthritis Rheum 2012;64:2824–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Karlsson JA, Neovius M, Nilsson JA, et al. Addition of infliximab compared with addition of sulfasalazine and hydroxychloroquine to methotrexate in early rheumatoid arthritis: 2-year quality-of-life results of the randomised, controlled, SWEFOT trial. Ann Rheum Dis 2013;72:1927–33. [DOI] [PubMed] [Google Scholar]
- [19].O’Dell JR, Mikuls TR, Taylor TH, et al. Therapies for active rheumatoid arthritis after methotrexate failure. N Engl J Med 2013;369:307–18. [DOI] [PubMed] [Google Scholar]
- [20].de Jong PH, Hazes JM, Han HK, et al. Randomised comparison of initial triple DMARD therapy with methotrexate monotherapy in combination with low-dose glucocorticoid bridging therapy; 1-year data of the tREACH trial. Ann Rheum Dis 2014;73:1331–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Sauer BC, Teng CC, Tang D, et al. Persistence with conventional triple therapy versus a tumor necrosis factor inhibitor and methotrexate in US veterans with rheumatoid arthritis. Arthritis Care Res (Hoboken) 2017;69:313–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Emery P, Hammoudeh M, FitzGerald O, et al. Sustained remission with etanercept tapering in early rheumatoid arthritis. N Engl J Med 2014;371:1781–92. [DOI] [PubMed] [Google Scholar]
- [23].Smolen JS, Nash P, Durez P, et al. Maintenance, reduction, or withdrawal of etanercept after treatment with etanercept and methotrexate in patients with moderate rheumatoid arthritis (PRESERVE): a randomised controlled trial. Lancet (London, England) 2013;381:918–29. [DOI] [PubMed] [Google Scholar]
- [24].Henaux S, Ruyssen-Witrand A, Cantagrel A, et al. Risk of losing remission, low disease activity or radiographic progression in case of bDMARD discontinuation or tapering in rheumatoid arthritis: systematic analysis of the literature and meta-analysis. Ann Rheum Dis 2018;77:515–22. [DOI] [PubMed] [Google Scholar]
- [25].Baraliakos X, Listing J, Rudwaleit M, et al. Safety and efficacy of readministration of infliximab after longterm continuous therapy and withdrawal in patients with ankylosing spondylitis. J Rheumatol 2007;34:510–5. [PubMed] [Google Scholar]
- [26].Kugathasan S, Levy MB, Saeian K, et al. Infliximab retreatment in adults and children with Crohn's disease: risk factors for the development of delayed severe systemic reaction. Am J Gastroenterol 2002;97:1408–14. [DOI] [PubMed] [Google Scholar]
- [27].Toki H, Momohara S, Tsukahara S, et al. Infusion reaction to infliximab in a patient with rheumatoid arthritis after discontinuation over 1 year and readministration. J Rheumatol 2008;35:1896–7. author reply 7. [PubMed] [Google Scholar]
- [28].Neovius M, Arkema EV, Olsson H, et al. Drug survival on TNF inhibitors in patients with rheumatoid arthritis comparison of adalimumab, etanercept and infliximab. Ann Rheum Dis 2015;74:354–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Russell E, Zeihen M, Wergin S, et al. Patients receiving etanercept may develop antibodies that interfere with monoclonal antibody laboratory assays. Arthritis Rheum 2000;43:944. [DOI] [PubMed] [Google Scholar]
- [30].Sugiura F, Kojima T, Oba M, et al. Anaphylactic reaction to infliximab in two rheumatoid arthritis patients who had previously received infliximab and resumed. Mod Rheumatol 2005;15:201–3. [DOI] [PubMed] [Google Scholar]
- [31].Mittal M, Raychaudhuri SP. Golimumab and certolizumab: the two new anti-tumor necrosis factor kids on the block. Indian J Dermatol Venereol Leprol 2010;76:602–8. quiz 9. [DOI] [PubMed] [Google Scholar]


