Abstract
Purpose
The purpose of this study was to investigate the effects of bilberry extract with its anthocyanins on retinal photoreceptor cell damage and on the endoplasmic reticulum (ER) stress induced by exposure to blue light-emitting diode (LED) light.
Methods
Cultured murine photoreceptor cells (661W) were exposed to blue LED light with or without bilberry extract or its anthocyanins in the culture media. Aggregated short-wavelength opsin (S-opsin) in murine photoreceptor cells was observed with immunostaining. The expression of factors involved in the unfolded protein response was examined with immunoblot analysis and quantitative real-time reverse transcription (RT)-PCR. Furthermore, cell death was observed with double staining with Hoechst 33342 and propidium iodide after dithiothreitol (DTT) treatment.
Results
Bilberry extract and anthocyanins suppressed the aggregation of S-opsin, activation of ATF4, and expression of the mRNA of the factors associated with the unfolded protein response (UPR). In addition, bilberry extract and the anthocyanins inhibited the death of photoreceptor cells induced by DTT, an ER stress inducer.
Conclusions
These findings suggest that bilberry extract containing anthocyanins can alter the effects of blue LED light and DTT-induced retinal photoreceptor cell damage. These effects were achieved by modulating the activation of ATF4 and through the suppression of the abnormal aggregation of S-opsin.
Introduction
Blue light, 450 to 495 nm, is part of the high-energy end of the visible light spectrum, and prolonged exposure to blue light can cause damage to retinal photoreceptor cells in in vivo experiments [1-5]. It has been found that exposure to blue light causes oxidative stress, aggregation of short wavelength opsin (S-opsin), and activation of transcription factor 4 (ATF4) in the retina [1,6]. ATF4 is a protein of the unfolded protein response (UPR), and is one of the causes of the aggregation of S-opsin, a photoreceptor protein, in murine photoreceptor cells [6]. It is important to prevent aggregation of S-opsin and suppress oxidative stress in retinal photoreceptor cells to prevent damage from exposure to blue light.
Bilberry (Vaccinium myrtillus L.) is a member of the Ericaceous family of plants that grow in northern European forests. Bilberry extract contains 15 types of anthocyanins [7,8] and has antioxidant activity [9], promotes collagen biosynthesis [10], inhibits platelet aggregation [11], and improves vascular tone, blood flow, and vasculoprotective activity [12,13]. Furthermore, results from animal experiments showed that bilberry improves the visual function [14,15], and clinical research has shown that bilberry can alleviate visual fatigue [16]. The results of our previous studies showed that bilberry extract had protective effects against retinal neuronal damage induced by N-methyl-D-aspartate [17] and had antiangiogenic effects in a mouse model of oxygen-induced retinopathy [18]. In addition, natural foods that contain anthocyanins (e.g., bilberry, maqui berry, and purple rice) have been shown to have protective effects against light-induced retinal damage [19-22]. We have reported that bilberry extract and its constituent anthocyanidins protect retinal photoreceptor cell against the damage induced by exposure to blue light-emitting diode (LED) light (λmax 464 nm) mainly through inhibition of the production of reactive oxygen species (ROS) [20]. However, exposure to blue LED light can also induce aggregation of S-opsin that can then cause rapid cone degeneration [23]. To protect photoreceptor cells from damage by exposure to blue LED light, it is necessary to reduce the level of ROS and inhibit S-opsin aggregation.
Because bilberry extract was reported to suppress amyloid fibril formation [24], we reasoned that bilberry extract with its anthocyanins might be able to suppress abnormal protein aggregation and inhibit the activation of ATF4 and endoplasmic reticulum (ER) stress responses by suppressing the aggregation of S-opsin induced by exposure to blue LED light. Thus, the purpose of this study was to determine the effects of bilberry extract and its anthocyanin components, delphinidin-3-glucoside (Dp3G), cyanidin-3-glucoside (Cy3G), and malvidin-3-glucoside (Mv3G), on the damage of cultured retinal photoreceptor cells induced by exposure to blue LED light. In addition, we examined the effects of bilberry extract on ER stress.
Methods
Materials
Bilberry extract was supplied by Wakasa Seikatsu Co., Ltd (Kyoto, Japan). The anthocyanin content in this bilberry extract was 38.35% as determined with high performance liquid chromatography (HPLC). The anthocyanins consisted of 15.03% delphinidin, 9.25% cyanidin, and 5.02% malvidin. We confirmed the bilberry extract concentration used in this experiment did not affect the absorption of blue LED light (464 nm; data not shown). Dp3G, Cy3G, and Mv3G were purchased from Tokiwa Phytochemical Co., Ltd. (Chiba, Japan). Mouse antibodies against ATF4 and against ubiquitin were purchased from Cell Signaling Technology (Beverly, MA). Rabbit opsin antibody was purchased from Merck Millipore (Darmstadt, Germany). Antibody against mouse β-actin was purchased from Sigma-Aldrich (St. Louis, MO). Dithiothreitol (DTT) was purchased from Wako Pure Chemical Industries, Ltd. Osaka, Japan. Hoechst 33342 and propidium iodide (PI) were purchased from Thermo Fisher Scientific (Waltham, MA). Blue LED light with λmax at 464 nm was purchased from M-Trust Co, Ltd. (Hyogo, Japan).
The mouse cell line, 661W, derived from retinal tumors in transgenic mice was a kind gift from Dr. Muayyad R. Al-Ubaidi of the University of Houston, Department of Biomedical Engineering, Houston, TK. The 661W cells have cellular and biochemical characteristics of cone photoreceptor cells and have been used as an in vitro model of cone photoreceptors.
Cell cultures
The murine photoreceptor cell line was maintained in Dulbecco’s modified Eagle’s medium (DMEM) containing 10% fetal bovine serum (FBS), 100 U/ml penicillin, and 100 μg/ml streptomycin under a humidified atmosphere of 5% CO2 at 37 °C. Cultured cells were released by trypsin and passaged every 2 to 3 days.
STR analysis
The murine photoreceptor cell line was confirmed with short tandem repeats (STR) profile and interspecies contamination testing (IDEXX BioResearch, Ludwigsburg, Germany). The sample was verified to be of mouse origin, and no mammalian interspecies contamination was detected. The genetic profile was proven to be consistent with a mixed FVB and C57BL/6 mouse strain of origin, and to carry the HIT1 transgene, which is unique to this cell line (Appendix 1) [25].
Exposure to LED light
A blue LED light device aligns many LED light sources. In this report, murine photoreceptor cells were exposed with blue LED light at 450 lux illuminance (equal to 0.38 mW/cm2), below the cell culturing plates. Exposure to blue LED light was performed in a CO2 incubator, in a humidified atmosphere of 5% CO2 at 37 °C.
Western blot analysis
Murine photoreceptor cells (3 × 104 cell/well) were seeded in 12-well plates and cultured at 37 °C for 24 h in a humidified atmosphere of 5% CO2. After 24 h, the medium was replaced with 1% FBS-DMEM, and after 30 min, bilberry extract containing Dp3G, Cy3G, and Mv3G or each anthocyanin was added. The cells were incubated for 1 h, and then the cells were exposed to 450 lux of blue LED light for 6 h. After the light exposure, the photoreceptor cells were washed with PBS (1X; 136.9 mM NaCl, 2.68 mM KCl, 10.14 mM Na2HPO4•12H2O, 1.76 mM KH2PO4, pH 7.3) and lysed in radioimmunoprecipitation assay (RIPA) buffer supplemented with 1% protease inhibitor and phosphatase inhibitor cocktail 2 and 3 (Sigma Aldrich). Lysates were centrifuged at 12,000 ×g for 20 min, and the protein concentration in the supernatant was measured with the BCA protein assay kit (Thermo Fisher Scientific) with bovine serum albumin (BSA) as the standard. The protein sample and the sample buffer with 2-mercaptoethanol were mixed and then separated on 5–20% sodium dodecyl sulfate (SDS) polyacrylamide gels. The separated proteins were transferred to polyvinylidene difluoride (PVDF) membranes (Immobilon-P; EMD Millipore Corporation, Billerica, MA). The following primary antibodies were used for immunoblotting; rabbit anti-ATF4, mouse anti-ubiquitin (1:1,000), and mouse anti-β-actin (1:5,000). Then, the membranes were incubated with secondary antibodies horseradish peroxidase (HRP)–conjugated goat anti-rabbit or goat anti-mouse (1:2,000; Thermo Fisher Scientific). The immunoreactive bands were made visible with ImmnoStar®LD (Wako Pure Chemical Industries, Osaka, Japan), and the LAS-4000 Luminescent Image Analyzer (Fuji Film, Tokyo, Japan). β-actin was used as the loading control.
Immunostaining
Murine photoreceptor cells (1.5 × 104 cell/500 µl) were seeded in glass chamber slides (Laboratory-Tek; Life Technologies) and incubated for 24 h. After 24 h, the medium was replaced with 1% FBS-DMEM and incubated for 30 min, and then bilberry extract with Dp3G, Cy3G, and Mv3G or each anthocyanin was added and incubated for 1 h. Subsequently, the cells were exposed to 450 lux blue LED for 6 h. After the exposure, the cells were immunostained according to Kuse et al.’s method [1]. The stained slides were photographed with a confocal fluorescence microscope (Olympus), and the perinuclear S-opsin aggregated cells were counted in a 212 µm2 area with ImageJ software. At least five sites in each well were photographed, and the average value was used for the statistical analyses. Three to four wells were counted for each group.
Real-time RT–PCR
Murine photoreceptor cells (1.5 × 104 cell/well) were seeded in 24-well plates and incubated for 24 h. After 24 h, the medium was replaced with 1% FBS-DMEM and incubated for 30 min, and then bilberry extract, Dp3G, Cy3G, and Mv3G were added and incubated for 1 h. Subsequently, the cells were exposed to 450 lux blue LED for 6 h. Six hours after exposure to blue LED light, RNA was extracted with NucleoSpin® RNA (Takara Bio Inc., Shiga, Japan). Real-time reverse transcription (RT)-PCR was performed with a Thermal Cycler Real Time System with SYBR Premix Ex TaqII according to the manufacturer’s protocol. The PCR primer sequences used were for GRP94, 5′-TTT GAA CCT CTG CTC AAC TGG AT-3′ (forward) and 5′-CTG ACT GGC CAC AAG AGC ACA-3′ (reverse); for BiP/GRP78, 5′-CTC CAC GGC TTC CGA TAA TCA-3′ (forward) and 5′-TCC AGT CAG ATC AAA TGT ACC CAG A-3′ (reverse); and for GAPDH, 5′-TGT GTC CGT CGT GGA TCT GA-3′ (forward) and 5′-TTG CTG TTG AAG TCG CAG GAG-3′ (reverse).The results are expressed relative to the GAPDH internal control.
DTT-induced cell death assay
Murine photoreceptor cells (3 × 103 cells/well) were seeded into 96-well plates and cultured at 37 °C. The medium was exchanged for 1% FBS-DMEM, and after 30 min, bilberry extract, or Dp3G, Cy3G, and Mv3G were individually added. The cells were incubated for 1 h. Then 3 mM of DTT was added, and the cells were further cultured for 24 h. At the end of the incubation period, Hoechst 33342 and PI were added to the culture medium (8.1 and 1.5 μM, respectively) and then incubated for 15 min. The cells were photographed through fluorescence filters for Hoechst 33342 (U-MWU, Olympus, Tokyo, Japan) and PI (U-MWIG, Olympus) with a charge-coupled device camera (DP30BW, Olympus). Finally, the cell mortality rate was calculated from the number of PI-positive cells as a percentage of the number of Hoechst 33342–positive cells.
Statistical analyses
Data are presented as the mean ± standard error of the mean (SEM). Statistical comparisons were made with one-way ANOVA followed by Student t tests, Tukey’s tests, or Dunnett’s multiple-comparison tests. A p value of less than 0.05 was considered statistically significant.
Results
Effects of bilberry extract, Dp3G, Cy3G, and Mv3G on blue LED light–induced aggregation of S-opsin
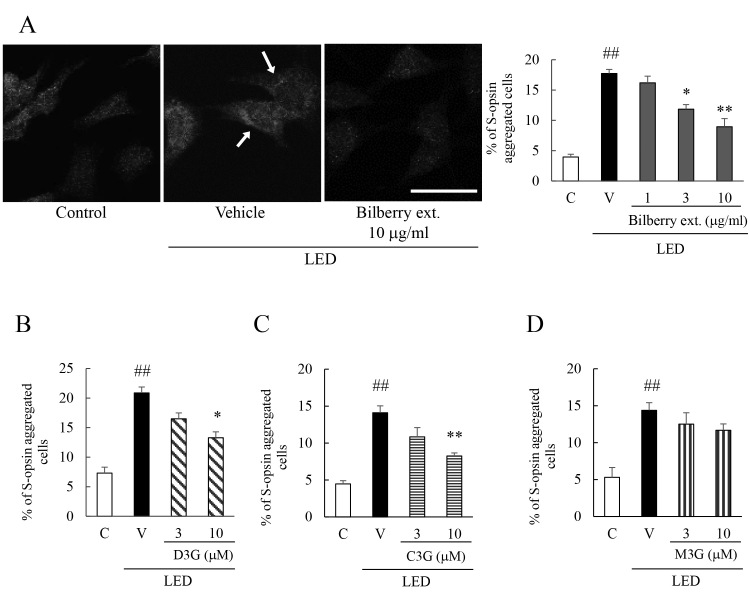
Exposure of 661W cells to 450 lux of blue light for 6 h induced aggregation of S-opsin, a protein specific for photoreceptor cells [1,26]. Exposure to blue LED light caused aggregation of S-opsin time dependency [1]. S-opsin tended to aggregate from 3 h of LED exposure, and statistically significantly increased with exposure to LED light for 6 h [1]. Addition of bilberry extract, or Dp3G, or Cy3G inhibited the aggregation of S-opsin in a concentration-dependent manner. However, when Mv3G was added, only a slight inhibition of the aggregation of S-opsin occurred, and the changes were not statistically significant (Figure 1). However, exposure to blue LED light and/or the addition of bilberry extract showed no statistically significant effect on the amounts of S-opsin mRNA and translation proteins (Appendix 2). In addition, we performed immunoblotting using cycloheximide, a translation inhibitor, but as there was no change in the amount of S-opsin protein, it is inferred that cycloheximide did not affect the decomposition amount of the protein (Appendix 2). These experiments were performed under exposure to blue LED light for 6 h. Under these conditions, bilberry extract had no effect on the transcription and translation of S-opsin. Moreover, bilberry extract did not affect the degradation rate of S-opsin, because bilberry extract did not change the amount of the S-opsin protein even with cycloheximide.
Figure 1.
Effect of bilberry extract, and its constituent anthocyanins, or Dp3G, Cy3G, and Mv3G individually, on the aggregation of S-opsin induced by exposure to blue LED light. Representative immunostained images of S-opsin after exposure to blue light-emitting diode (LED) light for 6 h and quantitative data after (A) bilberry extract, (B) delphinidin-3-glucoside (Dp3G), (C) cyanidin-3-glucoside (Cy3G), and (D) malvidin-3-glucoside (Mv3G). The quantitative analysis of the immunostained images is the ratio of the aggregated S-opsin cells. Data are the mean ± standard error of the mean (SEM; n=3 or 4). ##p<0.01 versus control; **p<0.01, *p<0.05 versus the vehicle (Dunnett’s multiple comparison tests or Student t tests). The scale bar represents 50 µm. Arrowheads indicate the aggregation of S-opsin.
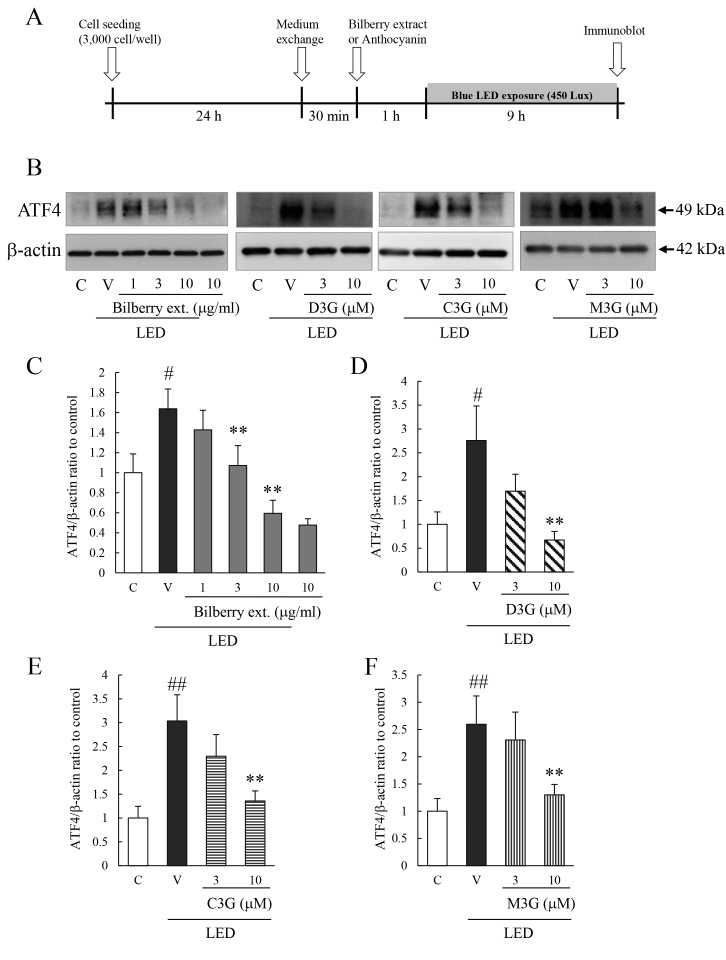
Effects of bilberry extract, Dp3G, Cy3G, and Mv3G on activation of ATF4 by exposure to blue LED light
The effect of bilberry extract or Dp3G, Cy3G, or Mv3G on the activation of ATF4 was determined with western blot analyses. The exposure duration was determined based on a previous report [6]. Exposure to blue LED light statistically significantly increased the expression of the ATF4 protein. Exposure to bilberry extract or its active components (Dp3G, Cy3G, and Mv3G) statistically significantly decreased the level of ATF4 expression induced by exposure to blue LED light (Figure 2).
Figure 2.
Effect of bilberry extract, or Dp3G, Cy3G, and Mv3G individually on blue LED light–induced ATF4 activation in 661W cells, a murine photoreceptor cell line, in culture. Immunoblotting shows the level of the ATF4 protein. Cells were pretreated with bilberry extract, or delphinidin-3-glucoside (Dp3G), cyanidin-3-glucoside (Cy3G), and malvidin-3-glucoside (Mv3G) for 1 h. After 1 h, exposure to 450 lux blue light-emitting diode (LED) light for 9 h. A: Experimental protocol in vitro. B: Typical photomicrograph and quantitative data of ATF4 protein levels using (C) bilberry extract (C), (D) delphinidin 3-glucoside, (E) cyanidin 3-glucoside, and (F) malvidin 3-glucoside. Data are the mean ± standard error of the mean (SEM; n=5 to 6). #p<0.05, ##p<0.01 versus control; **p<0.01 versus vehicle (Dunnett’s multiple comparison tests or Student t tests).
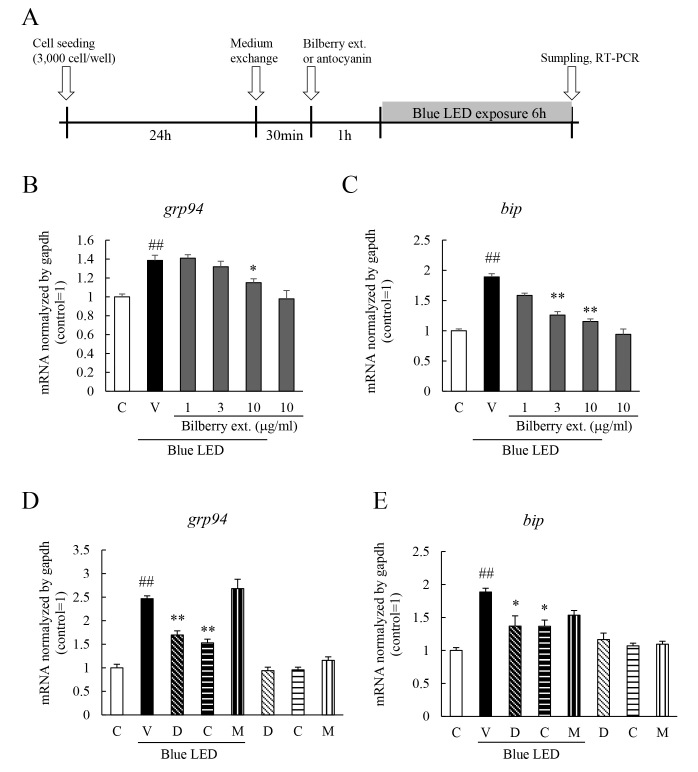
Effects of bilberry extract, Dp3G, Cy3G, and Mv3G on the level of expression of UPR factors induced by blue LED light determined with RT–PCR
RT–PCR was performed to investigate the effects of bilberry extract and the anthocyanins on the expression of the UPR factors. Exposure to blue LED light activated ATF4 and increased the expression of Grp94 and bip/grp78, UPR factors, in the murine photoreceptor cell line [6]. Grp94 and bip/grp78 are UPR factors present downstream of ATF4. Exposure to blue LED light upregulated the expression of the mRNA of grp94 and bip, and bilberry extract inhibited the increase of these factors (Figure 3B,C). Dp3G and Cy3G also inhibited the expression of the mRNA of grp94 and bip/grp78, but Mv3G had no statistically significant effect on this expression (Figure 3D,E).
Figure 3.
Effect of bilberry extract, or Dp3G, Cy3G, and Mv3G individually on the expression of UPR factors induced by exposure to blue LED light. Unfolded protein response-related mRNAs, bip and grp94, in murine photoreceptor cells induced by exposure to blue light-emitting diode (LED) light. The levels of mRNA after 9 h exposure to blue LED light with real-time reverse transcriptase (RT)–PCR. A: Experimental protocol. Induction of the mRNA of (A) bip, (B) grp94 treated with bilberry extract of 1 to 10 μg/ml. Induction of (C) bip and (D) grp94 mRNA treated with anthocyanins at 10 µM. Gapdh mRNA was used as the control. Data are the mean ± standard error of the mean (SEM; n=4). ##p<0.01 versus control; *p<0.05, **p<0.01 versus vehicle (Student t tests, Dunnett’s multiple comparison test or Tukey's test).
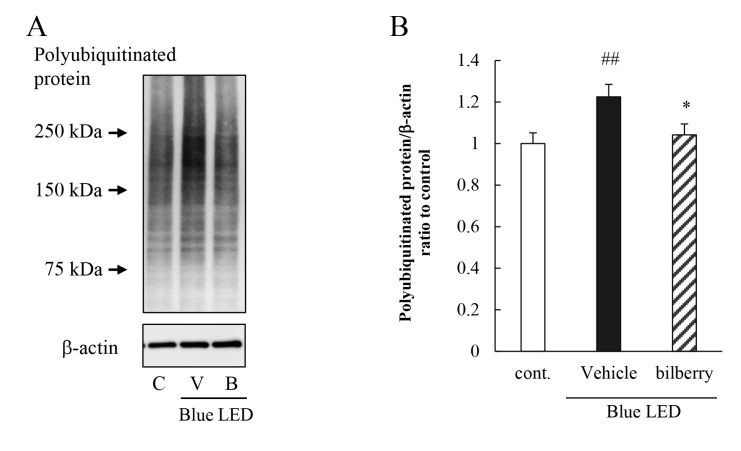
Effect of bilberry extract on blue LED light–induced induction of protein polyubiquitination
Short exposure to blue LED light increased the polyubiquitination of proteins in the murine photoreceptor cell line [6]. We performed western blot analysis to show that 6 h of exposure to blue LED light increased the expression of polyubiquitinated proteins. Addition of bilberry extract to the medium suppressed the degree of protein polyubiquitination (Figure 4).
Figure 4.
Effect of bilberry extract on ubiquitinated protein level induced by exposure to blue LED light. Cells were pretreated with bilberry extract for 1 h and then exposed to 450 lux blue light-emitting diode (LED) light for 6 h. Immunoblotting show ubiquitinated protein levels. A: Representative image and (B) quantitative data of the immunoblotting. Data are the mean ± standard error of the mean (SEM; n=20). ##p<0.01 versus control; *p<0.05 versus vehicle (Student t test).
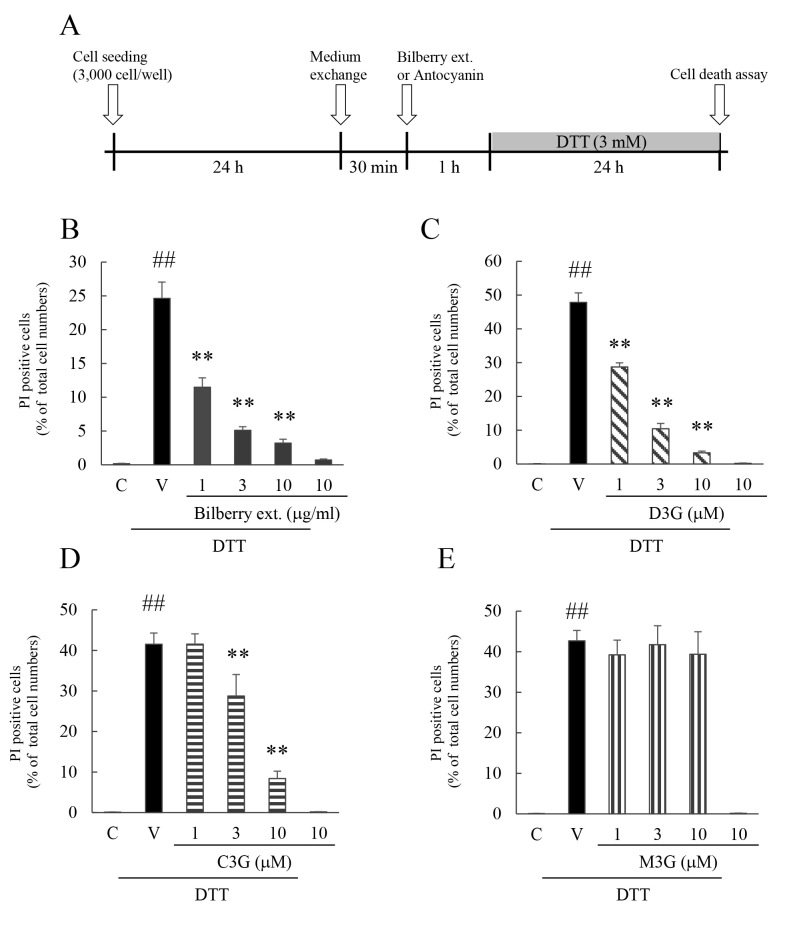
Effects of bilberry extract, Dp3G, Cy3G, and Mv3G on DTT-induced death of murine photoreceptor cells
Exposure to blue LED light induced S-opsin aggregation, ATF4 activation, and upregulation of the downstream factors of ATF4. Thus, we reasoned that exposure to blue LED light should induce ER stress [6], and we investigated the effect of bilberry extract on DTT-induced cell death. DTT, a reducing agent, is one of the ER stress inducers, and it can induce death of murine photoreceptor cells. Cell damage induced by DTT was protected by the addition of positive control drug tauroursodeoxycholic acid (TUDCA; data not shown). Bilberry extract, Dp3G, and Cy3G inhibited DTT-induced cell death, but Mv3G had no statistically significant effect (Figure 5).
Figure 5.
Effect of bilberry extract, and Dp3G, Cy3G, or Mv3G individually on DTT-induced cell death in murine photoreceptor cell line. A: Experimental protocol in vitro. Effect of (B) bilberry extract, (C) delphinidin-3-glucoside (Dp3G), (D) cyanidin-3-glucoside (Cy3G), and (E) malvidin-3-glucoside (Mv3G) on dithiothreitol (DTT)-induced cell death in murine photoreceptor cells. The number of cells exhibiting propidium iodide (PI) fluorescence was counted and expressed as a percentage of Hoechst 33342–positive cells. Data are the means ± standard error of the mean (SEM; n=5 to 6). ##p<0.01 versus control; **p<0.01 versus the vehicle (Dunnett’s multiple comparison test or Student t test).
Discussion
Our previous results showed that bilberry extract had protective effects against ultraviolet A (UV-A) irradiation and exposure to blue LED light for the murine photoreceptor cell line. This was accomplished by reducing the production of ROS [19,20]. In the present study, we focused on the effects of bilberry extract and its anthocyanins against retinal photoreceptor cell damage induced by exposure to blue LED light that aggregated S-opsin and stimulated the UPR response in an in vitro model. Bilberry extract contains 15 kinds of anthocyanins (including delphinidin, cyanidin, malvidin, petunidin, and peonidin) and three types of sugars (glucose, galactose, and arabinose). The absorption and metabolism mechanisms of action of anthocyanin have not been determined. Previously, anthocyanin was detected in the form of glycosides and not as aglycone in an anthocyanin absorption test using rats [27]. Therefore, we used glycosides to determine the active components of the bilberry extract.
S-opsin is more likely to aggregate than mouse M-opsin [28]. It was previously reported that S-opsin aggregated around the nucleus after exposure to blue LED light and after exposure to the ER stress inducers DTT and tunicamycin [1,6]. Exposure to blue LED light causes aggregation of S-opsin resulting in an increase in ATF4 [6]. In this study, we investigated the effects of bilberry extract and its active components on ATF4 activation and aggregation of S-opsin, which was reported in a previous report [6]. Bilberry extract, Dp3G, and Cy3G suppressed ATF4 activation and aggregation of S-opsin induced by exposure to blue LED light in the murine photoreceptor cell line in vitro. Mv3G suppressed blue LED light–induced ATF4 activation but had only a slight effect on aggregation of S-opsin. It has been reported that exposure to blue LED light activates Nrf2 in retinal photoreceptor cells [29]. Nrf2 is a factor related to the oxidative stress response, and Nrf2 can protect photoreceptor cells from photooxidative stress [29]. It was reported that Nrf2 binds to the transcriptional region of ATF4, and Nrf2 and ATF4 act in a coordinated manner [30,31]. The cause of the activation of ATF4 is not only the aggregation of S-opsin but also the activation of Nrf2 [30]. Mv3G did not have a statistically significant effect on the aggregation of S-opsin; however, Mv3G may have suppressed the activation of ATF4 by suppressing oxidative stress.
In this report, there was no influence of exposure to blue LED light and addition of bilberry extract on the translation, synthesis, and degradation of S-opsin. Therefore, it was suspected that the aggregation of S-opsin observed this experiment was not changed in the amount of S-opsin protein but in the suppression of aggregation or refolding of aggregated S-opsin. It has been reported that the bilberry extract suppresses abnormal aggregation of Aβ [24]. Anthocyanins have three-ring structures with many hydroxyl groups [17]. The chemical chaperones TUDCA and ursodeoxycholic acid (UDCA) that correct protein folding abnormalities also have ring structures and hydroxyl groups [32]. We suggest that suppression of the aggregation of S-opsin by anthocyanins is related to the structural properties of anthocyanin.
We also investigated the effects of bilberry extract and individual anthocyanins on the expression level of the mRNAs of bip/grp78 and grp94 that were increased by exposure to blue LED light [6]. The Bip protein tended to increase with a short period of blue LED light irradiation, but the increase was not statistically significant [6]. In this study, we investigated at the time point where the change in mRNA levels was observed with exposure to blue LED light in a previous report [6]. Bilberry extract, Dp3G, and Cy3G inhibited the activation of ATF4 and the expression of the mRNA of bip/grp78 and grp 94. However, Mv3G suppressed the activation of ATF4 but did not alter the level of expression of the mRNA of bip/grp78 and grp 94. It appears that Mv3G suppressed the activation of ATF4 only through the Nrf2 pathway due to the antioxidant effect but did not affect the aggregation of S-opsin.
Previously, there are some reports that polyubiquitinated proteins increase by exposure to light, and ER stress rises accompanied by exposure to light in murine photoreceptor cells [6,33]. ER stress is caused by an accumulation of unfolded proteins that are rapidly polyubiquitinated and degraded through the ubiquitin-proteasome pathway [34]. Accumulation of polyubiquitinated protein levels has been observed in neurodegenerative diseases associated with ER stress [35,36]. In addition, there are reports that M-opsin is difficult to aggregate [23], or the ubiquitinated M-opsin is quickly degraded by the proteasome system [37]. In this study, S-opsin and M-opsin were not separately examined, and all ubiquitinated proteins were detected. The present study showed that bilberry extract suppresses the increase in a misfolded or unfolded protein after exposure to blue LED light, and thus, it is reasonable to believe that bilberry extract suppresses ER stress.
Moreover, bilberry extract and Dp3G and Cy3G individually had protective effects against cell death caused by DTT-induced ER stress. DTT causes ER stress through the disruption of nonspecific thioester bonds. It has been shown that bilberry extract has a protective effect against the DTT-induced structural failure of the protein. In addition, N-acetyl-L-cysteine (NAC), an antioxidant, had no protective effect against the cell damage caused by DTT (data not shown). Therefore, we suggest that the protective effect exhibited by bilberry extract was not due to an antioxidant effect but inhibition of the intracellular stress caused by DTT-induced destruction of the thioester bonds. Furthermore, bilberry extract had a protective effect against DTT-induced cell death, but no protective effect against tunicamycin-induced cell death (Appendix 3). Tunicamycin inhibits N-linked glycosylation that causes protein misfolding [38,39]. The effect of tunicamycin depends on protein biosynthesis, and tunicamycin also has an adverse effect on the synthesis of normal proteins [40]. In contrast, DTT prevents disulfide bonds and induces misfolded protein independently of protein synthesis [39,41]. It has been suggested that the active components of bilberry extract are involved in the protection of cysteine residues or thioester bonds of aggregated proteins because bilberry extract has protective effects against the aggregation of proteins caused by protein reduction. S-opsin has one thioester bond and 15 cysteine residues [42]. It is suspected that bilberry extract and its components inhibited the aggregation of S-opsin by protecting the cysteine residues or thioester bonds. It has been reported that bilberry extract could divert peptide Aβ aggregation to an alternate non-toxic form [24]. These findings suggest that bilberry extract has similar effects against protein homeostasis.
The concentrations of delphinidin and cyanidin in the bilberry extract were relatively high in the present study. We suggest that the inhibition of ATF4 activation, S-opsin aggregation after exposure to blue LED light, and protection of DTT-induced cell death were largely due to the function of the glycosides of delphinidin and cyanidin contained in bilberry extract. The results of previous studies showed that the plasma concentration of anthocyanins after oral intake of cranberry juice containing 94.47 mg of anthocyanin ranges from 0.56 to 4.46 nmol/l in humans and animals [43]. Another study reported that the maximum rate of 13C elimination (32.53±14.24 μg/h) from blood after consumption of 500 mg of isotopically labeled Cy3G was observed at 30 min [44]. In a previous study, Cy3G was detected in the plasma at 30 min after oral administration of Cy3G in rats [27]. However, in pigs whose diet was supplemented with 0% to 4% w/w blueberry for 4 weeks, 11 intact anthocyanins were detected in the liver, eye, cortex, and cerebellum [45]. Another study reported that anthocyanin could be detected in the plasma and the eye after oral and intraperitoneal administration in rats [46]. The intraperitoneal administration of black currant anthocyanin at 108 mg/kg bodyweight in rats resulted in a concentration of 6.72 µg/ml in the vitreous, 6.89 µg/g in the retina, and 2.30 μg/g in the plasma [46]. Taken together, these studies suggest that bilberry anthocyanins can reach the eye of animals by oral administration. Although the effective concentration of anthocyanin from in vitro investigations in the present results may be higher than the plasma concentration of anthocyanin after oral administration, anthocyanin may be able to reach the tissues of the eye and may possess sufficient efficacy to benefit the eye. In future animal studies, it will be necessary to investigate the protective effect of orally administered bilberry extract.
In conclusion, the present study showed that bilberry extract containing anthocyanins protects the murine photoreceptor cell line against blue LED light and DTT-induced retinal photoreceptor cell damage. This was accomplished by modulation of the activation of ATF4 and through the suppression of abnormal aggregation of abnormal proteins. Bilberry may be a useful preventative health food for retinal diseases.
Appendix 1. STR analysis.
To access the data, click or select the words “Appendix 1.”
Appendix 2. Effect of bilberry extract or blue LED light exposure on S-opsin.
(A) The amount of S-opsin mRNA was examined. The levels of mRNA after 6 h exposure to blue LED light and/or treated with bilberry extract of 10 μg/ml determined by real-time RT–PCR. Gapdh mRNA was used as the control. Data are the means ± SEMs (n=14). (B) The amount of S-opsin protein was examined. The levels of S-opsin after 6 h exposure to blue LED light and/or treated with bilberry extract of 10 μg/mL determined by immunoblot with or without cycloheximide. C: Control. B: Bilberry extract. V: Vehicle. To access the data, click or select the words “Appendix 2.”
Appendix 3. Effect of bilberry extract on tunicamycin-induced cell death in murine photoreceptor cells.
(A) Experimental protocol in vitro. Effect of (B) bilberry extract on tunicamycin-induced death of 661W cells. The number of cells exhibiting PI fluorescence was counted and expressed as a percentage of Hoechst 33,342-positive cells. Data are the means ± SEMs (n=6). ##p<0.01 versus control (Student t test). To access the data, click or select the words “Appendix 3.”
References
- 1.Kuse Y, Ogawa K, Tsuruma K, Shimazawa M, Hara H. Damage of photoreceptor-derived cells in culture induced by light emitting diode-derived blue light. Sci Rep. 2014;4:5223. doi: 10.1038/srep05223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Roehlecke C, Schumann U, Ader M, Knels L, Funk RH. Influence of blue light on photoreceptors in a live retinal explant system. Mol Vis. 2011;17:876–84. [PMC free article] [PubMed] [Google Scholar]
- 3.Grimm C, Wenzel A, Williams T, Rol P, Hafezi F, Remé C. Rhodopsin-mediated blue-light damage to the rat retina: effect of photoreversal of bleaching. Invest Ophthalmol Vis Sci. 2001;42:497–505. [PubMed] [Google Scholar]
- 4.Narimatsu T, Negishi K, Miyake S, Hirasawa M, Osada H, Kurihara T, Tsubota K, Ozawa Y. Blue light-induced inflammatory marker expression in the retinal pigment epithelium-choroid of mice and the protective effect of a yellow intraocular lens material in vivo. Exp Eye Res. 2015;132:48–51. doi: 10.1016/j.exer.2015.01.003. [DOI] [PubMed] [Google Scholar]
- 5.Nakamura M, Kuse Y, Tsuruma K, Shimazawa M, Hara H. The involvement of the oxidative stress in murine blue LED light-induced retinal damage model. Biol Pharm Bull. 2017;40:1219–25. doi: 10.1248/bpb.b16-01008. [DOI] [PubMed] [Google Scholar]
- 6.Ooe E, Tsuruma K, Kuse Y, Kobayashi S, Shimazawa M, Hara H. The involvement of ATF4 and S-opsin in retinal photoreceptor cell damage induced by blue LED light. Mol Vis. 2017;23:52–9. [PMC free article] [PubMed] [Google Scholar]
- 7.Baj A, Bombardelli E, Gabetta B, Martinelli EM. Qualitative and quantitative evaluation of vaccinium myrtillus anthocyanins by high-resolution gas chromatography and high-performance liquid chromatography. J Chromatogr A. 1983;279:365–72. [Google Scholar]
- 8.Nakajima J, Tanaka I, Seo S, Yamazaki M, Saito K. LC/PDA/ESI-MS Profiling and Radical Scavenging Activity of Anthocyanins in Various Berries. J Biomed Biotechnol. 2004;2004:241–7. doi: 10.1155/S1110724304404045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ogawa K, Sakakibara H, Iwata R, Ishii T, Sato T, Goda T, Shimoi K, Kumazawa S. Anthocyanin composition and antioxidant activity of the crowberry (Empetrum nigrum) and other berries. J Agric Food Chem. 2008;56:4457–62. doi: 10.1021/jf800406v. [DOI] [PubMed] [Google Scholar]
- 10.Morazzoni P, Bombardelli E. Vaccinium myrtillus I. Fitoterapia. 1996;67:3–29. [Google Scholar]
- 11.Morazoni P, Megistretti MJ. Activity of myrtocyanR, an anthocyanoside complex from Vaccinium myrtillus (VMA), on platelet aggregation and adhesiveness. Fitoterapia. 1990;61:13–21. [Google Scholar]
- 12.Lietti A, Cristoni A, Piccci M. Studies on Vaccinium myrtillus anthocyanosides. Arzneimittelforschung. 1976;26:829–32. [PubMed] [Google Scholar]
- 13.Colantunoni A, Bertuglia S, Magistretti MJ, Donato L. Effects of Vaccinium myrtillus anthocyanosides on arterial vasomotion. Arzneimittelforschung. 1991;41:905–9. [PubMed] [Google Scholar]
- 14.Miyake S, Takahashi N, Sasaki M, Kobayashi S, Tsubota K, Ozawa Y. Vision preservation during retinal inflammation by anthocyanin-rich bilberry extract: cellular and molecular mechanism. Lab Invest. 2012;92:102–9. doi: 10.1038/labinvest.2011.132. [DOI] [PubMed] [Google Scholar]
- 15.Osada H, Okamoto T, Kawashima H, Toda E, Miyake S, Nagai N, Kobayashi S, Tsubota K, Ozawa Y. Neuroprotective effect of bilberry extract in a murine model of photo-stressed retina. PLoS One. 2017;12:e0178627. doi: 10.1371/journal.pone.0178627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ozawa Y, Kawashima M, Inoue S, Inagaki E, Suzuki A, Ooe E, Kobayashi S, Tsubota K. Bilberry extract supplementation for preventing eye fatigue in video display terminal workers. J Nutr Health Aging. 2015;19:548–54. doi: 10.1007/s12603-014-0573-6. [DOI] [PubMed] [Google Scholar]
- 17.Matsunaga N, Imai S, Inokuchi Y, Shimazawa M, Yokota S, Araki Y, Hara H. Bilberry and its main constituents have neuroprotective effects against retinal neuronal damage in vitro and in vivo. Mol Nutr Food Res. 2009;53:869–77. doi: 10.1002/mnfr.200800394. [DOI] [PubMed] [Google Scholar]
- 18.Matsunaga N, Tsuruma K, Shimazawa M, Yokota S, Hara H. Inhibitory actions of bilberry anthocyanidins on angiogenesis. Phytother Res. 2010;24(Suppl 1):S42–7. doi: 10.1002/ptr.2895. [DOI] [PubMed] [Google Scholar]
- 19.Ogawa K, Tsuruma K, Tanaka J, Kakino M, Kobayashi S, Shimazawa M, Hara H. The protective effects of bilberry and lingonberry extracts against UV light-induced retinal photoreceptor cell damage in vitro. J Agric Food Chem. 2013;61:10345–53. doi: 10.1021/jf402772h. [DOI] [PubMed] [Google Scholar]
- 20.Ogawa K, Kuse Y, Tsuruma K, Kobayashi S, Shimazawa M, Hara H. Protective effects of bilberry and lingonberry extracts against blue light-emitting diode light-induced retinal photoreceptor cell damage in vitro. BMC Complement Altern Med. 2014;14:120. doi: 10.1186/1472-6882-14-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tanaka J, Kadekaru T, Ogawa K, Hitoe S, Shimoda H, Hara H. Maqui berry (Aristotelia chilensis) and the constituent delphinidin glycoside inhibit photoreceptor cell death induced by visible light. Food Chem. 2013;139:129–37. doi: 10.1016/j.foodchem.2013.01.036. [DOI] [PubMed] [Google Scholar]
- 22.Tanaka J, Nakamura S, Tsuruma K, Shimazawa M, Shimoda H, Hara H. Purple rice (Oryza sativa L.) extract and its constituents inhibit VEGF-induced angiogenesis. Phytother Res. 2012;26:214–22. doi: 10.1002/ptr.3533. [DOI] [PubMed] [Google Scholar]
- 23.Zhang T, Zhang N, Baehr W, Fu Y. Cone opsin determines the time course of cone photoreceptor degeneration in Leber congenital amaurosis. Proc Natl Acad Sci USA. 2011;108:8879–84. doi: 10.1073/pnas.1017127108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yamakawa MY, Uchino K, Watanabe Y, Adachi T, Nakanishi M, Ichino H, Hongo K, Mizobata T, Kobayashi S, Nakashima K, Kawata Y. Anthocyanin suppresses the toxicity of Aβ deposits through diversion of molecular forms in in vitro and in vivo models of Alzheimer’s disease. Nutr Neurosci. 2016;19:32–42. doi: 10.1179/1476830515Y.0000000042. [DOI] [PubMed] [Google Scholar]
- 25.Al-Ubaidi MR, Font RL, Quiambao AB, Keener MJ, Liou GI, Overbeek PA, Baehr W. Bilateral retinal and brain tumors in transgenic mice expressing simian virus 40 large T antigen under control of the human interphotoreceptor retinoid-binding protein promoter. J Cell Biol. 1992;119:1681–7. doi: 10.1083/jcb.119.6.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tang PH, Buhusi MC, Ma JX, Crouch RK. RPE65 is present in human green/red cones and promotes photopigment regeneration in an in vitro cone cell model. J Neurosci. 2011;31:18618–26. doi: 10.1523/JNEUROSCI.4265-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tsuda T, Horio F, Osawa T. Absorption and metabolism of cyanidin 3‐O‐β‐D‐glucoside in rats. FEBS Lett. 1999;449:179–82. doi: 10.1016/s0014-5793(99)00407-x. [DOI] [PubMed] [Google Scholar]
- 28.Zhang T, Fu Y. A Phe-rich region in short-wavelength sensitive opsins is responsible for their aggregation in the absence of 11-cis-retinal. FEBS Lett. 2013;587:2430–4. doi: 10.1016/j.febslet.2013.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chen WJ, Wu C, Xu Z, Kuse Y, Hara H, Duh EJ. Nrf2 protects photoreceptor cells from photo-oxidative stress induced by blue light. Exp Eye Res. 2017;154:151–8. doi: 10.1016/j.exer.2016.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Miyamoto N, Izumi H, Miyamoto R, Bin H, Kondo H, Tawara A, Sasaguri Y, Kohno K. Transcriptional regulation of activating transcription factor 4 under oxidative stress in retinal pigment epithelial ARPE-19/HPV-16 cells. Invest Ophthalmol Vis Sci. 2011;52:1226–34. doi: 10.1167/iovs.10-5775. [DOI] [PubMed] [Google Scholar]
- 31.Ye P, Mimura J, Okada T, Sato H, Liu T, Maruyama A, Ohyama C, Itoh K. Nrf2- and ATF4-Dependent Upregulation of xCT Modulates the Sensitivity of T24 Bladder Carcinoma Cells to Proteasome Inhibition. Mol Cell Biol. 2014;34:3421–34. doi: 10.1128/MCB.00221-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bai C, Biwersi J, Verkman AS, Matthay MA. A mouse model to test the in vivo efficacy of chemical chaperones. J Pharmacol Toxicol Methods. 1998;40:39–45. doi: 10.1016/s1056-8719(98)00034-3. [DOI] [PubMed] [Google Scholar]
- 33.Nakanishi T, Shimazawa M, Sugitani S, Kudo T, Imai S, Inokuchi Y, Tsuruma K, Hara H. Role of endoplasmic reticulum stress in light-induced photoreceptor degeneration in mice. J Neurochem. 2013;125:111–24. doi: 10.1111/jnc.12116. [DOI] [PubMed] [Google Scholar]
- 34.Uehara T. Accumulation of misfolded protein through nitrosative stress linked to neurodegenerative disorders. Antioxid Redox Signal. 2007;9:597–601. doi: 10.1089/ars.2006.1517. [DOI] [PubMed] [Google Scholar]
- 35.Uehara T, Nakamura T, Yao D, Shi ZQ, Gu Z, Ma Y, Masliah E, Nomura Y, Lipton SA. S-nitrosylated protein-disulphide isomerase links protein misfolding to neurodegeneration. Nature. 2006;441:513–7. doi: 10.1038/nature04782. [DOI] [PubMed] [Google Scholar]
- 36.Ito Y, Shimazawa M, Inokuchi Y, Yamanaka H, Tsuruma K, Imamura K, Onoe H, Watanabe Y, Aihara M, Araie M, Hara H. Involvement of endoplasmic reticulum stress on neuronal cell death in the lateral geniculate nucleus in the monkey glaucoma model. Eur J Neurosci. 2011;33:843–55. doi: 10.1111/j.1460-9568.2010.07578.x. [DOI] [PubMed] [Google Scholar]
- 37.Sato K, Ozaki T, Ishiguro S, Nakazawa M. M-opsin protein degradation is inhibited by MG-132 in Rpe65−/− retinal explant culture. Mol Vis. 2012;18:1516–25. [PMC free article] [PubMed] [Google Scholar]
- 38.Lehrman MA. Oligosaccharide-based information in endoplasmic reticulum quality control and other biological systems. J Biol Chem. 2001;276:8623–6. doi: 10.1074/jbc.R100002200. [DOI] [PubMed] [Google Scholar]
- 39.Rutkowski DT, Arnold SM, Miller CN, Wu J, Li J, Gunnison KM, Mori K, Sadighi Akha AA, Raden D, Kaufman RJ. Adaptation to ER stress is mediated by differential stabilities of pro-survival and pro-apoptotic mRNAs and proteins. PLoS Biol. 2006;4:e374. doi: 10.1371/journal.pbio.0040374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Labunskyy VM, Yoo MH, Hatfield DL, Gladyshev VN. Sep15, a thioredoxin-like selenoprotein, is involved in the unfolded protein response and differentially regulated by adaptive and acute ER stresses. Biochemistry. 2009;48:8458–65. doi: 10.1021/bi900717p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kaufman RJ. Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev. 1999;13:1211–33. doi: 10.1101/gad.13.10.1211. [DOI] [PubMed] [Google Scholar]
- 42.Baraas RC, Hagen LA, Dees EW, Neitz M. Substitution of isoleucine for threonine at position 190 of S-opsin causes S-cone-function abnormalities. Vision Res. 2012;73:1–9. doi: 10.1016/j.visres.2012.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Milbury PE, Vita JA, Blumberg JB. Anthocyanins are bioavailable in humans following an acute dose of cranberry juice. J Nutr. 2010;140:1099–104. doi: 10.3945/jn.109.117168. [DOI] [PubMed] [Google Scholar]
- 44.Czank C, Cassidy A, Zhang Q, Morrison DJ, Preston T, Kroon PA, Botting NP, Kay CD. Human metabolism and elimination of the anthocyanin, cyanidin-3-glucoside: a 13C-tracer study. Am J Clin Nutr. 2013;97:995–1003. doi: 10.3945/ajcn.112.049247. [DOI] [PubMed] [Google Scholar]
- 45.Kalt W, Blumberg JB, McDonald JE, Vinqvist-Tymchuk MR, Fillmore SA, Graf BA, O’Leary JM, Milbury PE. Identification of anthocyanins in the liver, eye, and brain of blueberry-fed pigs. J Agric Food Chem. 2008;56:705–12. doi: 10.1021/jf071998l. [DOI] [PubMed] [Google Scholar]
- 46.Matsumoto H, Nakamura Y, Iida H, Ito K, Ohguro H. Comparative assessment of distribution of blackcurrant anthocyanins in rabbit and rat ocular tissues. Exp Eye Res. 2006;83:348–56. doi: 10.1016/j.exer.2005.12.019. [DOI] [PubMed] [Google Scholar]