Abstract
Camels have cultural value in the Arab society and are considered one of the most important animals in the Arabian Peninsula and arid environments, due to the distinct characteristics of their meat and milk. Moreover, there is a great interest in camel racing and beauty shows. Therefore, treatment of elite animals, increasing the number of camels as well as genetic improvement is an essential demand. Because there are unique camels for milk production, meat, or in racing, the need to propagate genetically superior camels is urgent. Recent biotechnological approaches such as stem cells hold great promise for biomedical research, genetic engineering, and as a model for studying early mammalian developmental biology. Establishment of stem cells lines from camels would tremendously facilitate regenerative medicine for genetically superior camels, permit the gene targeting of the camel genome and the generation of genetically modified animal and be a mean for genome conservation for the elite breeds. In this mini-review, we show the current research, future horizons and potential applications for camel stem cells.
Keywords: Camel, Cloning, Embryo, Regenerative medicine, Stem cells
1. The camel (Camelus dromedarius) agribusiness
The Arabian camel (Camelus dromedarius) is a unique species and can be a better provider of meat and milk in desert areas than other farm animals, which are severely affected by heat and scare feed and water [1]. Camels occupy a special niche in the Arabian agricultural production system. The total population of dromedary is estimated to be about 25 million heads all over the world [updated according [2], [3]].
Camel racing is a highly lucrative and well-organized sport and considered an important traditional and animal agribusiness activity in the Arabian Gulf states [2]. Since the major injuries in racing are fractures and because camels are often nervous, camel orthopedics is poorly understood because of a lack of comprehensive studies on fracture healing [4]. Moreover, there are numerous constraints of camel orthopedics; hence, the principles of bovine and equine orthopedics cannot be applied on camels in absolute terms [5]. Therefore, a basic understanding of bone and cartilage repair is essential to save the lives of thousands of camels used for this agribusiness throughout the world. The bone tissue engineering concept is a relatively new method for repairing damaged bones and involves the regeneration of tissues using stem cells, scaffolds, and growth factors, with stem cells playing a leading role in tissue repair and regeneration [6], [7], [8].
Additionally, industry has attracted investments in camel dairy by-products, as 16.9% of milk consumed by humans comes from species other than cattle [9], [10]. However, there is a significant variability in the milk yield among individuals (i.e. high milk producing camels can produce 12-fold more milk than low-producing ones) [2], [11]. Camels can produce milk and sustain its productivity in harsh and hostile conditions where other animals may not survive [12]. Camel milk is a rich source of proteins with potential antimicrobial and protective activities compared to cow’s milk. In many countries, camel milk is given to babies suffering from malnutrition or milk sensitivity [13]. Compared to cow, buffalo, and ewe milk fat, camel milk fat contains fewer short-chained fatty acids, but the same long-chained fatty acids can be found. Some researchers claim that the value of camel milk is found in the high concentrations of volatile acids especially linoleic and polyunsaturated fatty acids, which are essential for human nutrition [14]. Camel milk has a high vitamin (especially C, B1), minerals and immunoglobulin content. Additionally, camel milk is low in lactose and cholesterol compared to cow's milk [15]. However, the levels of potassium, magnesium, iron, copper, manganese, sodium and zinc are higher than in cow's milk [13], [16]. Therefore, it is necessary to treat injuries and propagate the numbers of high-value milk producers through cloning technology to conserve the genetic merits of these camels, especially when traditional breeding practices are difficult and of low efficiency [17], [18].
2. Camelid nanobodies
Nanobodies are unique compounds secreted by members of the camelids family [19]. According to Muyldermans et al., heavy chain-only antibodies (HCAbs) that circulate in the blood of camels are different from the antibodies produced by other species [20]. These antibodies lack the light chains and are composed of a heavy-chain homodimer. They are termed as variable domain of heavy chain of HCAb (VHH) or nanobody and have various therapeutic advantages. Nanobodies are single domain antibodies derived from heavy chain-only antibodies (HCAbs) produced naturally by camelids [21]. They are considered a new generation of active proteins with unique properties. Nanobodies show excellent tissue distribution, high temperature and pH stability, are easy to produce with recombinant technology and can readily be converted into different formats such as Fc-fusion proteins or heterodimers. Moreover, nanobodies have the unique ability to bind molecular clefts, such as the active site of enzymes, thereby interfering with the function of the target protein [22]. Over the last decade, numerous nanobodies have been developed against proteins involved in inflammation, with the aim to modulate their immune functions [23], [24], [25]. Recently, nanobodies have emerged as potential candidates for targeting cancer. Owing to their very small size, they are able to penetrate the typically inaccessible parts of the solid tumors with low immunogenicity [26]. Nanobodies are also considered as a significant tool in various therapeutic disciplines due to their unique ability to bind or attach to other proteins and nanoparticles by using noncomplex chemical treatments. Furthermore, cell permeable nanobodies have been recently discovered that could permit the co-transport of therapeutically relevant proteins into target cells [27]. This technology constitutes a major step in the labelling, delivery and targeted manipulation of intracellular protein antigens. Ultimately, this approach could open the door towards targeting the intracytoplasmic pathways of living cells and the expansion of immunotherapies to intracellular antigen targets.
3. Embryonic and induced pluripotent stem cells
Embryonic stem cells (ESCs) are pluripotent cells and have the ability to differentiate into all tissues and cells comprising the human and animal tissues, such as muscle, liver, brain, bone and cartilage tissues. ESCs are derived from the inner cell mass (ICM) at the early stage of embryo development, the blastocyst. They can differentiate into three germ layers; the ectoderm, mesoderm and endoderm, which give rise to all the tissues constituting the body [28], [29].
Cells can be transformed into a pluripotent state through cocktail of defined factors such as Oct4 and Sox2 in conjunction with Myc and Klf4 or Nanog and Lin28 as reported recently. The resultant cells were termed induced pluripotent stem cells (iPSCs) and this achievement was awarded Nobel Prize in 2012. The camel is a unique species with high endurance to life in desert conditions; however, little information are known about camels’ early embryonic development and the genes responsible for pluripotency. iPSCs can be generated from differentiated cells by the retrovirus-mediated transfection of four transcription factors, namely Oct3/4, Sox2, c-Myc, and Klf4 [30], [31]. On the other hand, Thomson’s team generated iPSCs using another cocktail of transcription factors which are Oct4, Sox2, Nanog and Lin28 [31]. Interestingly, iPSCs show a great deal of potential for stem cell therapies and clinical applications particularly for elite animals [32], could generate patient- or disease-specific stem cells, which are required for their effective biomedical applications, such as regenerative medicine [33]. Consequently, iPSCs could be used for regenerative medicine in precious genetically superior camels used for racing and beauty shows. The pioneering technology of iPSCs made reprogramming much easier than it had been with early reprogramming technologies, such as somatic cell nuclear transfer (SCNT), as well as avoided the ethical issues such as the embryo or fetus’ destruction that was necessary for harvesting ESCs. Importantly, the generation of patient-specific iPSCs could be used to improve and test new drugs in vitro [34].
Despite their applicative potential, it seems that iPSCs in farm animals have not received the attention they deserve [15], [35], [36]. Few studies have shown the derivation of iPSCs from farm animals such as cattle [37], horses [38], sheep [39], goats [40] and pigs [23], [41]. However, there is no report on the generation of iPSCs from camels.
4. The current situation in camel stem cell research
There are very few studies covered the camel stem cells and this field still in its early infancy. Recently, for the first time, we have isolated ESCs and trophectoderm stem cells from camel embryos on feeder-free conditions and showed the expression of all pluripotency genes (Oct4, Sox2, Klf4 and Myc) in the established cell lines through the conventional and real-time relative quantitative polymerase chain reaction (RT-PCR and RQ-PCR) [42]. The isolated ESCs were successfully differentiated into neuron-like cells. Moreover, we found a differential expression of certain genes such as Klf4, which showed significant increase in trophoblasts when compared with the ESCs which raises the question as to whether Klf4 or other transcripts are essential for pluripotency in camels. These results motivated our team to sequence and identify the whole genes’ sequences responsible for the pluripotency in camels and clone these genes to be easily used for transforming the differentiated somatic cells into pluripotent stem cells following the transfection of the cells with pluripotency transcription factors. Interestingly, BLAST analysis (The Basic Local Alignment Search Tool; https://blast.ncbi.nlm.nih.gov/Blast.cgi) for the predicted camel genes showed ∼90% matching with those known in humans (Table 1).
Table 1.
BLAST sequence alignment for pluripotency genes in comparison with human.
| Gene | Accession No. [Camel (taxid:9838)] | Homology with humans (taxid:9606) |
|---|---|---|
| POU5F1 | XM_010978211.1 | 92% |
| SOX2 | XM_010976367.1 | 93% |
| MYC | XM_010988786.1 | 88% |
| KLF4 | XM_010978705.1 | 90% |
| NANOG | XM_010990807.1 | 81% |
| LIN28A | XM_010993734.1 | 90% |
This preliminary result motivated us to specifically isolate and identify pluripotency genes and used them for generating iPSCs from camels, instead of using commercially available factors designed for human or mouse cells. The best source for isolating the coding DNA sequence (CDS) of these genes are the blastocyst stage of in vivo or in vitro produced embryos. Fortunately, our team has successfully established system for in vitro production of camel embryos either through in vitro techniques [43], [44] or through the flushing of in vivo fertilized ones.
Another team has isolated the mesenchymal stem cells from camel adipose tissue and showed its differentiation capabilities into adipogenic, osteogenic, and chondrogenic cells [45].
Our current work focuses on utilizing novel and easy sources for isolating pluripotent or multipotent stem cells from the camels such as the ovarian follicular cells [35], [46], [47]. These cells could be obtained either from slaughter house or by follicle aspiration from live animals. Our ongoing experiments and results are encouraging and showed that camel follicular cells can be differentiated easily into adipocytes, osteoblasts and neurons.
5. Potential applications of camel stem cells
Pluripotent stem cells are promising not only for medical applications, but could also have numerous uses in biotechnology and agriculture. Advanced reproduction techniques in farm animals could enable the development of genetically modified animals from engineered pluripotent stem cells; SCNT is a method of choice when producing transgenic farm animals [48] and the use of genetically engineered pluripotent stem cells (i.e. ESCs or iPSCs capable of generating offspring through nuclear transfer) as donor cells could efficiently improve the procedure’s success, as already shown in mice [49].
Since the birth of Dolly the sheep [50], the first cloned mammal, an ever-growing number of studies worldwide have helped to substantiate the potential applications of somatic cell nuclear transfer (cloning) to overcome several problems in various biology fields, such as generating copies of particular species including camels [51] and extinct or endangered species and for the propagation of the livestock and elite animals [10], [52], [5], [53]. This technology can be used to propagate camels with the highest potential for milk production, beauty contests or racing champions. However, the use of this technique is limited owing to its low efficiency particularly in camels [52] and several trials have attempted to improve the cloning efficiencies [24]. Moreover, reports have showed the advantageous effects of using iPSCs as donor cells for SCNT [54], [55], [49] to improve the cloning efficiency and to generate genetically modified organisms for therapeutic cloning [56], [53] and transgenesis to generate genetically engineered camels which would be acting as bioreactors to produce specific nanobodies for therapeutic purposes [30], [44], [51], [57], [58]. Production of antibodies from genetically engineered cattle [56], [57], [59], [60], [61] paved the way for the application in the camelids, which are the source of nanobodies.
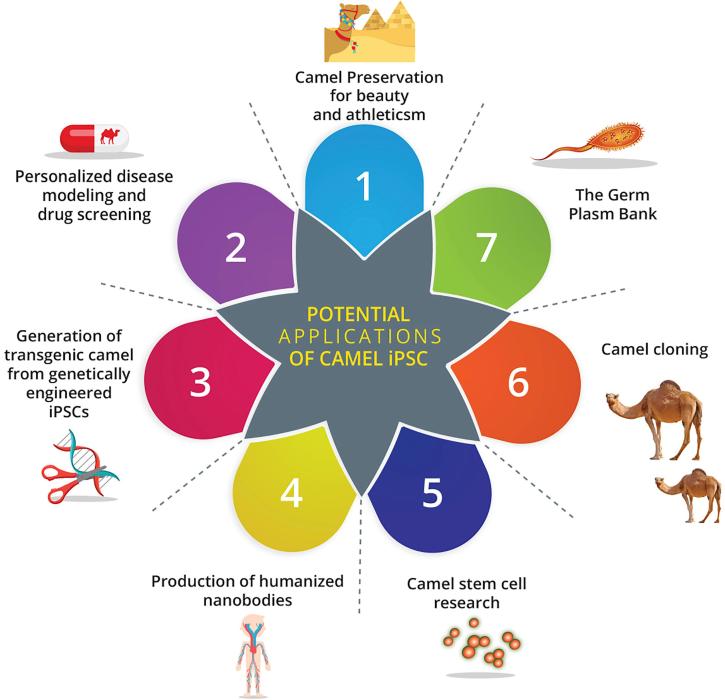
The potential applications of using both embryonic and induced pluripotent stem cells (iPSC) for camel agribusiness can be summarized as following (see also Fig. 1):
-
1.
Regenerative medicine and personalized cell therapy for traumatic, injured, or fractured elite and genetically superior camels used for show, racing or milking.
-
2.
Camel iPSCs will be used for the genome conservation of elite genetically valuable animals, such as high milk producers, racing champions, and males of high genetic merit.
-
3.
Understanding the functions of individual pluripotency gene over-expression on early embryonic development in camels.
-
4.
Improving the cloning efficiency of camels through using pluripotency genes’ over-expressed cell lines, either individual or combined.
-
5.
Generating bioreactors capable of producing therapeutic targeted nanobodies for human diseases.
-
6.
In vitro disease modeling, which could yield new insights into disease mechanisms and drug discovery, especially for orthopedics and neuronal affections pathways.
Fig. 1.
The potential applications of camel induced pluripotent stem cells (iPSC).
Competing interests
The authors declare no competing interests.
Footnotes
Peer review under responsibility of Faculty of Veterinary Medicine, Cairo University.
References
- 1.Schmidt-Nielsen K. The physiology of the camel. Sci Am. 1959;201:140–151. doi: 10.1038/scientificamerican1259-140. [DOI] [PubMed] [Google Scholar]
- 2.Cuadrado, P.S., Young, H., and Vance, A. Camel racing: The multi-million dollar industry mixing modernity and tradition. http://editioncnncom/2017/03/14/sport/camel-racing-robots-uae-thoroughbred-hussain-al-marzooqi/; 2017.
- 3.FAO. FAOSTAT; Live Animals. http://wwwfaoorg/faostat/en/#data/QA; 2013.
- 4.Gahlot T., Chouhan D. Fractures in dromedary (Camelus dromedarius) – a retrospective study. J Camel Pract Res. 1994;1:9–14. [Google Scholar]
- 5.Mohamed A.F. Fractures in single-humped camels: a retrospective Study of 220 Cases (2008–2009) J King Abdulaziz Univ. 2012;23:3–17. [Google Scholar]
- 6.Hayashi K., Ochiai-Shino H., Shiga T., Onodera S., Saito A., Shibahara T. Transplantation of human-induced pluripotent stem cells carried by self-assembling peptide nanofiber hydrogel improves bone regeneration in rat calvarial bone defects. Bdj Open. 2016;2:15007. doi: 10.1038/bdjopen.2015.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hong So G., Winkler T., Wu C., Guo V., Pittaluga S., Nicolae A. Path to the clinic: assessment of iPSC-based cell therapies in vivo in a nonhuman primate model. Cell Rep. 2014;7:1298–1309. doi: 10.1016/j.celrep.2014.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sheyn D., Ben-David S., Shapiro G., De Mel S., Bez M., Ornelas L. Human induced pluripotent stem cells differentiate into functional mesenchymal stem cells and repair bone defects. Stem Cells Transl Med. 2016;5:1447–1460. doi: 10.5966/sctm.2015-0311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Al Kanhal H.A., Al hajand O.A. Compositional, technological and nutritional aspects of dromedary camel milk. Int Dairy J. 2010;20:811–821. [Google Scholar]
- 10.Faye B., Konuspayeva G. The sustainability challenge to the dairy sector – the growing importance of non-cattle milk production worldwide. Int Dairy J. 2012;24:50–56. [Google Scholar]
- 11.Forbes, T. Camel milk industry to receive boost as Australia's largest dairy grows herd. http://wwwabcnetau/news/2017-03-24/camel-milk-beauty-australian-wild-camel-corporation/8380914; 2017.
- 12.Raziq A., Younas M., Kakar M. Camel – a potential dairy animal in difficult environments. Pak J Agri Sci. 2008;45:263–267. [Google Scholar]
- 13.Farah Z. Composition and characteristics of camel milk. J Dairy Res. 2009;60:603–622. doi: 10.1017/s0022029900027953. [DOI] [PubMed] [Google Scholar]
- 14.Khaskheli M., Arain M., Chaudhry S., Soomro A., Qureshi T. Physico-chemical quality of camel milk. J Agric Soc Sci. 2005;2:164–166. [Google Scholar]
- 15.El-Agamy E.I., Nawar M., Shamsia S.M., Awad S., Haenlein G.F.W. Are camel milk proteins convenient to the nutrition of cow milk allergic children? Small Rumin Res. 2009;82:1–6. [Google Scholar]
- 16.Sawaya W.N., Khalil J.K., Al-Shalhat A., Al-Mohammad H. Chemical composition and nutritional quality of camel milk. J Food Sci. 1984;49:744–747. [Google Scholar]
- 17.Long C.R., Walker S.C., Tang R.T., Westhusin M.E. New commercial opportunities for advanced reproductive technologies in horses, wildlife, and companion animals. Theriogenology. 2003;59:139–149. doi: 10.1016/s0093-691x(02)01266-9. [DOI] [PubMed] [Google Scholar]
- 18.Murray J., Anderson G. Genetic engineering and cloning may improve milk, livestock production. Calif Agric. 2000;54:57–65. [Google Scholar]
- 19.Hassanzadeh-Ghassabeh G., Devoogdt N., De Pauw P., Vincke C., Muyldermans S. Nanobodies and their potential applications. Nanomedicine. 2013;8:1013–1026. doi: 10.2217/nnm.13.86. [DOI] [PubMed] [Google Scholar]
- 20.Muyldermans S., Baral T.N., Retamozzo V.C., De Baetselier P., De Genst E., Kinne J. Camelid immunoglobulins and nanobody technology. Vet Immunol Immunopathol. 2009;128:178–183. doi: 10.1016/j.vetimm.2008.10.299. [DOI] [PubMed] [Google Scholar]
- 21.Muyldermans S. Nanobodies: natural single-domain antibodies. Annu Rev Biochem. 2013;82:775–797. doi: 10.1146/annurev-biochem-063011-092449. [DOI] [PubMed] [Google Scholar]
- 22.Kirchhofer A., Helma J., Schmidthals K., Frauer C., Cui S., Karcher A. Modulation of protein properties in living cells using nanobodies. Nat Struct Mol Biol. 2009;17:133–138. doi: 10.1038/nsmb.1727. [DOI] [PubMed] [Google Scholar]
- 23.Esteban M.A., Xu J., Yang J., Peng M., Qin D., Li W. Generation of induced pluripotent stem Cell lines from Tibetan miniature pig. J Biol Chem. 2009;284:17634–17640. doi: 10.1074/jbc.M109.008938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Saadeldin I.M., Kim S.J., Choi Y.B., Lee B.C. Improvement of cloned embryos development by co-culturing with parthenotes: a possible role of exosomes/microvesicles for embryos paracrine communication. Cell Reprogram. 2014;16:223–234. doi: 10.1089/cell.2014.0003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rissiek B.r., Koch-Nolte F., Magnus T. Nanobodies as modulators of inflammation: potential applications for acute brain injury. Front Cell Neurosci. 2014;8:344. doi: 10.3389/fncel.2014.00344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Harmsen M.M., De Haard H.J. Properties, production, and applications of camelid single-domain antibody fragments. Appl Microbiol Biotechnol. 2007;77:13–22. doi: 10.1007/s00253-007-1142-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Herce H.D., Schumacher D., Schneider A.F.L., Ludwig A.K., Mann F.A., Fillies M. Cell-permeable nanobodies for targeted immunolabelling and antigen manipulation in living cells. Nat Chem. 2017;9:762–771. doi: 10.1038/nchem.2811. [DOI] [PubMed] [Google Scholar]
- 28.Evans M.J., Kaufman M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292:154–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- 29.Martin G.R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci USA. 1981;78:7634–7638. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Takahashi K., Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 31.Yu J., Vodyanik M.A., Smuga-Otto K., Antosiewicz-Bourget J., Frane J.L., Tian S. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 32.Schmidt R., Plath K. The roles of the reprogramming factors Oct4, Sox2 and Klf4 in resetting the somatic cell epigenome during induced pluripotent stem cell generation. Genome Biol. 2012;13:251. doi: 10.1186/gb-2012-13-10-251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Takahashi K., Tanabe K., Ohnuki M., Narita M., Ichisaka T., Tomoda K. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 34.Avior Y., Sagi I., Benvenisty N. Pluripotent stem cells in disease modelling and drug discovery. Nat Rev Mol Cell Biol. 2016;17:170–182. doi: 10.1038/nrm.2015.27. [DOI] [PubMed] [Google Scholar]
- 35.Kossowska-Tomaszczuk K., De Geyter C. Cells with stem cell characteristics in somatic compartments of the ovary. Biomed Res Int. 2013;2013:1–8. doi: 10.1155/2013/310859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ogorevc J., Orehek S., Dovč P. Cellular reprogramming in farm animals: an overview of iPSC generation in the mammalian farm animal species. J Anim Sci Biotechnol. 2016;7:10. doi: 10.1186/s40104-016-0070-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Han X., Han J., Ding F., Cao S., Lim S.S., Dai Y. Generation of induced pluripotent stem cells from bovine embryonic fibroblast cells. Cell Res. 2011;21:1509–1512. doi: 10.1038/cr.2011.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nagy K., Sung H.-K., Zhang P., Laflamme S., Vincent P., Agha-Mohammadi S. Induced pluripotent stem cell lines derived from equine fibroblasts. Stem Cell Rev Rep. 2011;7:693–702. doi: 10.1007/s12015-011-9239-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liu J., Balehosur D., Murray B., Kelly J.M., Sumer H., Verma P.J. Generation and characterization of reprogrammed sheep induced pluripotent stem cells. Theriogenology. 2012;77:338–346. doi: 10.1016/j.theriogenology.2011.08.006. e331. [DOI] [PubMed] [Google Scholar]
- 40.Song H., Li H., Huang M., Xu D., Gu C., Wang Z. Induced pluripotent stem cells from goat fibroblasts. Mol Reprod Dev. 2013;80:1009–1017. doi: 10.1002/mrd.22266. [DOI] [PubMed] [Google Scholar]
- 41.Ezashi T., Telugu B.P.V.L., Alexenko A.P., Sachdev S., Sinha S., Roberts R.M. Derivation of induced pluripotent stem cells from pig somatic cells. Proc Natl Acad Sci. 2009;106:10993–10998. doi: 10.1073/pnas.0905284106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Saadeldin I.M., Swelum A.A.-A., Elsafadi M., Moumen A.F., Alzahrani F.A., Mahmood A. Isolation and characterization of the trophectoderm from the Arabian camel (Camelus dromedarius) Placenta. 2017;57:113–122. doi: 10.1016/j.placenta.2017.06.015. [DOI] [PubMed] [Google Scholar]
- 43.Saadeldin I.M., Swelum A.A.-A., Yaqoob S.H., Alowaimer A.N. Morphometric assessment of in vitro matured dromedary camel oocytes determines the developmental competence after parthenogenetic activation. Theriogenology. 2017;95:141–148. doi: 10.1016/j.theriogenology.2017.03.015. [DOI] [PubMed] [Google Scholar]
- 44.Yaqoob S.H., Saadeldin I.M., Swelum A.A.-A., Alowaimer A.N. Optimizing camel (Camelus dromedarius) oocytes in vitro maturation and early embryo culture after parthenogenetic activation. Small Rumin Res. 2017;153:81–86. [Google Scholar]
- 45.Mohammadi-Sangcheshmeh A., Shafiee A., Seyedjafari E., Dinarvand P., Toghdory A., Bagherizadeh I. Isolation, characterization, and mesodermic differentiation of stem cells from adipose tissue of camel (Camelus dromedarius) In Vitro Cell Dev Biol Anim. 2013;49:147–154. doi: 10.1007/s11626-012-9578-9. [DOI] [PubMed] [Google Scholar]
- 46.Kossowska-Tomaszczuk K., De Geyter C., De Geyter M., Martin I., Holzgreve W., Scherberich A. The multipotency of luteinizing granulosa cells collected from mature ovarian follicles. Stem Cells. 2009;27:210–219. doi: 10.1634/stemcells.2008-0233. [DOI] [PubMed] [Google Scholar]
- 47.Riva F., Omes C., Bassani R., Nappi R.E., Mazzini G., Icaro Cornaglia A. In-vitro culture system for mesenchymal progenitor cells derived from waste human ovarian follicular fluid. Reprod BioMed Online. 2014;29:457–469. doi: 10.1016/j.rbmo.2014.06.006. [DOI] [PubMed] [Google Scholar]
- 48.Keefer C.L. Artificial cloning of domestic animals. Proc Natl Acad Sci. 2015;112:8874–8878. doi: 10.1073/pnas.1501718112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhou S., Ding C., Zhao X., Wang E., Dai X., Liu L. Successful generation of cloned mice using nuclear transfer from induced pluripotent stem cells. Cell Res. 2010;20:850–853. doi: 10.1038/cr.2010.78. [DOI] [PubMed] [Google Scholar]
- 50.Wilmut I., Schnieke A.E., McWhir J., Kind A.J., Campbell K.H.S. Viable offspring derived from fetal and adult mammalian cells. Nature. 1997;385:810–813. doi: 10.1038/385810a0. [DOI] [PubMed] [Google Scholar]
- 51.Wani N.A., Wernery U., Hassan F.A.H., Wernery R., Skidmore J.A. Production of the first cloned camel by somatic cell nuclear Transfer1. Biol Reprod. 2010;82:373–379. doi: 10.1095/biolreprod.109.081083. [DOI] [PubMed] [Google Scholar]
- 52.Khatir H., Anouassi A. Preliminary assessment of somatic cell nuclear transfer in the dromedary (Camelus dromedarius) Theriogenology. 2008;70:1471–1477. doi: 10.1016/j.theriogenology.2008.06.094. [DOI] [PubMed] [Google Scholar]
- 53.Saadeldin I.M. Cloning and its Applications. Cloning & Transgenesis. 2015;4:e118. [Google Scholar]
- 54.Fan N., Chen J., Shang Z., Dou H., Ji G., Zou Q. Piglets cloned from induced pluripotent stem cells. Cell Res. 2012;23:162–166. doi: 10.1038/cr.2012.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kou Z., Kang L., Yuan Y., Tao Y., Zhang Y., Wu T. Mice cloned from induced pluripotent stem cells (iPSCs)1. Biol Reprod. 2010;83:238–243. doi: 10.1095/biolreprod.110.084731. [DOI] [PubMed] [Google Scholar]
- 56.Bruggemann M., Osborn M.J., Ma B., Hayre J., Avis S., Lundstrom B. Human antibody production in transgenic animals. Arch Immunol Ther Exp (Warsz) 2015;63:101–108. doi: 10.1007/s00005-014-0322-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.van de Water J.A.J.M., Bagci-Onder T., Agarwal A.S., Wakimoto H., Roovers R.C., Zhu Y. Therapeutic stem cells expressing variants of EGFR-specific nanobodies have antitumor effects. Proc Natl Acad Sci. 2012;109:16642–16647. doi: 10.1073/pnas.1202832109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ishida I., Tomizuka K., Yoshida H., Tahara T., Takahashi N., Ohguma A. Production of human monoclonal and polyclonal antibodies in transchromo animals. Cloning Stem Cells. 2002;4:91–102. doi: 10.1089/153623002753632084. [DOI] [PubMed] [Google Scholar]
- 59.Kuroiwa Y., Kasinathan P., Choi Y.J., Naeem R., Tomizuka K., Sullivan E.J. Cloned transchromosomic calves producing human immunoglobulin. Nat Biotechnol. 2002;20:889–894. doi: 10.1038/nbt727. [DOI] [PubMed] [Google Scholar]
- 60.Luke T., Wu H., Zhao J., Channappanavar R., Coleman C.M., Jiao J.A. Human polyclonal immunoglobulin G from transchromosomic bovines inhibits MERS-CoV in vivo. Sci Transl Med. 2016;8:326ra321. doi: 10.1126/scitranslmed.aaf1061. [DOI] [PubMed] [Google Scholar]
- 61.Kim S.J., Koo O.J., Park H.J., Moon J.H., da Torre B.R., Javaregowda P.K. Oct4 overexpression facilitates proliferation of porcine fibroblasts and development of cloned embryos. Zygote. 2015;23:704–711. doi: 10.1017/S0967199414000355. [DOI] [PubMed] [Google Scholar]