Abstract
Purpose
Bone-modifying agents (BMAs) are recommended for women with bone metastasis from breast cancer to prevent skeletal-related events. We examined the usage patterns and identified the factors associated with the use of BMAs (denosumab and intravenous bisphosphonates) among women in the US.
Patients and methods
Electronic health records from oncology clinics were used to identify women diagnosed with bone metastasis from breast cancer between 2013 and 2014. Patients were excluded if they had recently used a BMA or had concurrent cancer at an additional primary site. The incidence of BMA initiation, interruption, and reinitiation were estimated using competing risk regression models. A generalized linear model was used to estimate risk factors for treatment initiation and interruption.
Results
There were 589 women diagnosed with bone metastasis from breast cancer. By 1 year, 68% of these patients (95% CI: 64%, 71%) had initiated treatment with a BMA. Denosumab and zoledronic acid were the most commonly used agents, whereas pamidronate was used infrequently. Young women were more likely to initiate a BMA than older women (adjusted risk difference: 6.4 [95% CI: 1.5, 10.9]). Of the 412 patients who initiated a BMA, 46% (95% CI: 41%, 51%) experienced an interruption within 1 year. Seventy-four percent (95% CI: 68%, 79%) of patients who interrupted their treatment had reinitiated therapy within 1 year of interruption.
Conclusion
The majority of women diagnosed with bone metastasis from breast cancer initiate a BMA within 1 year of diagnosis, but a large proportion, particularly among the elderly, do not use these therapies.
Keywords: bone-modifying agents, breast cancer, bone metastasis, treatment patterns, electronic health records, denosumab, zoledronic acid, pamidronate
Introduction
Each year, there are approximately 266,000 new cases of female breast cancer in the US.1 The bone is the most common site of distant metastasis for women with breast cancer.2 Depending on the study and the time period evaluated,3 approximately 15% of new breast cancer patients (40,000 cases) are expected to develop bone metastases during the course of their disease.4,5 Not only is this typically a sign of the incurable nature of the underlying disease, but metastatic bone disease is also associated with serious skeletal complications. These are collectively referred to as skeletal-related events (SREs) and include severe bone pain (often requiring radiation), pathological fractures, bone instability requiring surgery, and spinal cord compression.6 SREs are associated with severe pain,7 elevated mortality risk,8 and increased health care costs.9 Nearly 40% of patients with bone metastases from breast cancer experience an SRE within 1 year of developing bone metastasis.10
Three therapies are currently approved in the US for the prevention of SREs in breast cancer patients with bone metastases. Pamidronate, a nitrogen-containing bisphosphonate (approved in 1991), is administered intravenously over a 2-hour period every 3–4 weeks.11 Zoledronic acid, a bisphosphonate (approved in 2001), is administered intravenously over a 15-minute infusion every 3–4 weeks.12 Denosumab, a monoclonal antibody with affinity for the receptor activator of nuclear factor-kappa ligand (approved in 2010), is administered subcutaneously every 4 weeks.13 These agents can reduce the risk of occurrence of SREs, and thereby improve the quality of life and functional independence.14–17 Oral bisphosphonates are not currently recommended to prevent SREs in this population.
Current clinical guidelines recommend bone-modifying agents (BMAs) for breast cancer patients with bone metastases,18,19 but little is known about how often these therapies are used in routine care in the US. We examined real-world data from electronic health records (EHRs) supplemented with important unstructured, clinical data abstracted through medical chart review to measure the initiation, interruption, and reinitiation of three BMAs among women with bone metastasis from breast cancer. We also examined the factors associated with initiation and treatment interruption.
Materials and methods
Data source
We conducted a retrospective cohort study using the Flatiron Health Analytic Database. While these data are not freely available, they may be purchased and used by researchers after all necessary data use agreements are executed. The database incorporates information from 255 cancer clinics (at community-based practices and two academic centers) and 2,330 clinicians across the US, and includes more than 1.3 million active cancer patients. Flatiron provides rich, real-world clinical data generated from the EHR systems routinely used by cancer care providers.20 The EHR captures longitudinal information on clinical diagnoses, medication administrations (including dose), laboratory results, and biomarkers. The data include both structured (ie, drop-down fields in the EHR that capture a patient’s sex or date of birth) and unstructured data (ie, free text from a physician’s note or laboratory report). Flatiron uses validated, technology-enabled chart abstraction21 followed by a manual review of unstructured data elements. In this study, the diagnoses of bone metastasis and SREs were validated using the unstructured data.
Study design
We identified a cohort of adult women with breast cancer (ICD-9 174.x, 175.x or ICD-10 C50.xx) who had a bone metastasis documented within 10 years after the breast cancer diagnosis and within the time period January 1, 2013 through December 31, 2014. A clinic visit was required in the period from 180 days before to 30 days after the date of diagnosis of the bone metastasis to ensure active enrollment in the Flatiron EHR data system. Patients were excluded if they had received denosumab, pamidronate, or zoledronic acid in the 6-month period before the diagnosis of bone metastasis or if they had an additional non-melanoma cancer at a primary site in the 12-month period prior to the diagnosis of bone metastasis.
We created three nested cohorts of patients to examine the cumulative incidence of BMA initiation, interruption, and reinitiation. First, initiation was evaluated among all breast cancer patients with bone metastasis, and was defined as a first administration of denosumab, zoledronic acid, or pamidronate. The index date (ie, when follow-up began) was 30 days after the date of bone metastasis diagnosis to allow for the assessment of the inclusion criterion requiring activity in Flatiron. Second, the interruption of a BMA was evaluated among all patients who initiated a BMA, and was defined as any 45-day interval that included a health care encounter but did not include the intravenous or subcutaneous administration of a BMA. A 45-day interval was chosen to capture the recommended administration time window for BMAs (once per 28 days) plus a short grace period. The index date was the date of BMA initiation. The date of interruption was imputed as the date of the last BMA administration plus 45 days. Third, treatment reinitiation was evaluated among all patients who had experienced a treatment interruption. This was defined as the first BMA administration following an interruption. The index date was the imputed date of interruption.
For all analyses, patients were followed until the earliest of the following events: the outcome of interest (initiation, interruption, reinitiation), end of health care (defined as 90 days without a health care encounter), and death or the end of study (June 30, 2016).
Statistical analysis
We identified covariates using the structured and unstructured data and assessed baseline characteristics prior to the date of bone metastasis. These characteristics were updated at the start of follow-up for each nested cohort. For example, for patients who initiated a BMA and who were followed for a treatment interruption, baseline characteristics were assessed at the date of the BMA initiation. Baseline covariates were defined during the 6-month baseline period or using all available data before the index date to identify chronic comorbidities. Baseline laboratory tests and functional status (measured by the Eastern Cooperative Oncology Group Performance Status [ECOG]) were defined in the 60 days before the diagnosis of bone metastasis. The estimated glomerular filtration rate (eGFR) was calculated using serum creatinine levels and the Chronic Kidney Disease Epidemiology Collaboration equation.22 If there were multiple test results or ECOG measurements during that period, the result closest to the index date was used. Other medications of interest (eg, chemotherapy) were defined during the 30 days prior to the index date. Time-varying covariates were identified: 1) during the 30-day period following the diagnosis of bone metastasis and during each 30-day interval thereafter (for predictors of treatment initiation); and 2) during the 45-day period following treatment initiation and during each 45-day interval thereafter (for predictors of treatment interruption).
We computed descriptive statistics for baseline covariates. The cumulative incidence of initiation, interruption, and reinitiation was calculated using Fine–Gray models that accounted for the competing risk of death.23 These models were stratified by BMA type and calendar year of bone metastasis diagnosis. To assess the risk factors of initiation and interruption, follow-up was discretized into 30-day intervals for initiation and 45-day intervals for interruption. Treatments, comorbidities, and laboratory results were updated within each interval. If laboratory results were missing in a given interval, the last-observation-carried-forward method was used to impute laboratory information for that interval. We estimated the absolute difference in monthly risk of each outcome using multivariable repeated measures generalized models with an identity link function to estimate risk differences. All measured variables were included in the models. Asymptotically correct 95% CIs were obtained using a nonparametric bootstrap. Predictors of each outcome were drawn from the interval prior to the interval in which the outcome occurred, as well as from all prior intervals and the baseline period. Patients only contributed to a specific risk factor analysis during intervals (30-day or 45-day intervals, depending on the analysis) in which they had a health care encounter. This study was approved by the Chesapeake Institutional Review Board. All statistical analyses were performed using R software, version 3.4.0.24
Results
There were 920 women initially identified in the Flatiron database who were diagnosed with bone metastases from breast cancer for whom a medical chart abstraction was completed. After applying study eligibility criteria, there were 589 women remaining, of whom 45% were 65 years and older, 60% were White, 91% had stage II or higher breast cancer when diagnosed, and 12% had a history of pathological fracture (Table 1). The prevalence of biomarkers was as follows: 83% estrogen receptor-positive, 70% progesterone receptor-positive, 21% human epidermal growth factor receptor 2 (HER2)-positive, and 10% triple-negative (negative for all three receptors). During follow-up, 412 women initiated treatment with a BMA, and 258 experienced a subsequent treatment interruption after initiation (Table 1). Compared with the overall cohort measured at the diagnosis of bone metastasis, the demographic and biomarker characteristics of patients who initiated and interrupted BMA treatment were similar, although the prevalence of a history of pathologic fracture was higher (21% and 25% at initiation and interruption, respectively), indicating that patients were experiencing SREs during follow-up (Table 1).
Table 1.
Baseline characteristicsa of women with bone metastasis from breast cancer eligible for initiating, interrupting, or reinitiating BMAs
| Variable | Women with bone metastases from breast cancer
|
Subset who initiated BMA treatment
|
Subset who interrupted BMA treatment
|
|||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| Total | 589 | 100 | 412 | 100 | 258 | 100 |
| Year of index date | ||||||
| 2013 | 290 | 49.2 | 160 | 38.8 | 43 | 16.7 |
| 2014 | 299 | 50.8 | 221 | 53.6 | 101 | 39.1 |
| 2015 | – | – | 29 | 7.0 | 98 | 38.0 |
| 2016 | – | – | 2 | 0.5 | 16 | 6.2 |
| Age (years) | ||||||
| 18–39 | 29 | 4.9 | 22 | 5.3 | 14 | 5.4 |
| 40–49 | 79 | 13.4 | 52 | 12.6 | 34 | 13.2 |
| 50–64 | 215 | 36.5 | 155 | 37.6 | 99 | 38.4 |
| 65+ | 266 | 45.2 | 183 | 44.4 | 111 | 43.0 |
| Race | ||||||
| White | 356 | 60.4 | 261 | 63.3 | 166 | 64.3 |
| Black | 60 | 10.2 | 34 | 8.3 | 24 | 9.3 |
| Asian | 14 | 2.4 | 10 | 2.4 | 9 | 3.5 |
| Other | 88 | 14.9 | 55 | 13.3 | 34 | 13.2 |
| Missing | 71 | 12.1 | 52 | 12.6 | 25 | 9.7 |
| History of SREs | ||||||
| Pathological fracture | 72 | 12.2 | 86 | 20.9 | 66 | 25.6 |
| Spinal cord compression | 9 | 1.5 | 7 | 1.7 | 5 | 1.9 |
| External beam radiation therapy | 9 | 1.5 | 87 | 21.1 | 92 | 35.7 |
| Bone surgery | 19 | 3.2 | 24 | 5.8 | 24 | 9.3 |
| Other sites of metastases | ||||||
| Lung | 69 | 11.7 | 72 | 17.5 | 25 | 9.7 |
| Brain | 15 | 2.5 | 21 | 5.1 | 16 | 6.2 |
| Distant lymph node | 47 | 8 | 67 | 16.3 | 24 | 9.3 |
| Prior cancer treatment | ||||||
| Chemotherapy | 21 | 3.6 | 151 | 36.7 | 101 | 39.1 |
| Colony-stimulating factor | 14 | 2.4 | 47 | 11.4 | 43 | 16.7 |
| Hormonal therapy | 73 | 12.4 | 216 | 52.4 | 97 | 37.6 |
| Renal disease | 86 | 14.6 | 109 | 26.5 | 79 | 30.6 |
| eGFR (mL/min) | ||||||
| <60 | 32 | 5.4 | 34 | 8.3 | 21 | 8.1 |
| ≥60 | 121 | 20.5 | 110 | 26.7 | 73 | 28.3 |
| Missing | 436 | 74 | 268 | 65.0 | 164 | 63.6 |
| ER status | ||||||
| Positive | 490 | 83.2 | 353 | 85.7 | 221 | 85.7 |
| Negative | 93 | 15.8 | 58 | 14.1 | 37 | 14.3 |
| Unknown | 6 | 1 | 1 | 0.2 | 0 | 0.0 |
| PR status | ||||||
| Positive | 410 | 69.6 | 296 | 71.8 | 195 | 75.6 |
| Negative | 167 | 28.4 | 110 | 26.7 | 60 | 23.3 |
| Unknown | 12 | 2 | 6 | 1.5 | 3 | 1.2 |
| HER2 status | ||||||
| Positive | 122 | 20.7 | 78 | 18.9 | 55 | 21.3 |
| Negative | 443 | 75.2 | 323 | 78.4 | 198 | 76.7 |
| Unknown/equivocal | 24 | 4.1 | 11 | 2.7 | 5 | 1.9 |
| Insurance payer | ||||||
| Commercial health insurance plan | 137 | 23.3 | 102 | 24.8 | 69 | 26.7 |
| Medicaid | 18 | 3.1 | 12 | 2.9 | 8 | 3.1 |
| Medicare | 25 | 4.2 | 10 | 2.4 | 5 | 1.9 |
| Missing | 132 | 22.4 | 89 | 21.6 | 40 | 15.5 |
| Multiple | 192 | 32.6 | 140 | 34.0 | 97 | 37.6 |
| Other | 85 | 14.4 | 59 | 14.3 | 38 | 14.7 |
Notes:
Baseline characteristics were measured in the 6-month baseline period or using all available data prior to the index date to identify chronic comorbidities. Baseline characteristics were updated at the start of BMA treatment and on the date of BMA interruption. “−” indicates that there were zero patients in this category.
Abbreviations: BMA, bone-modifying agent; eGFR, estimated glomerular filtration rate; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; PR, progesterone receptor; SRE, skeletal-related event.
Initiation
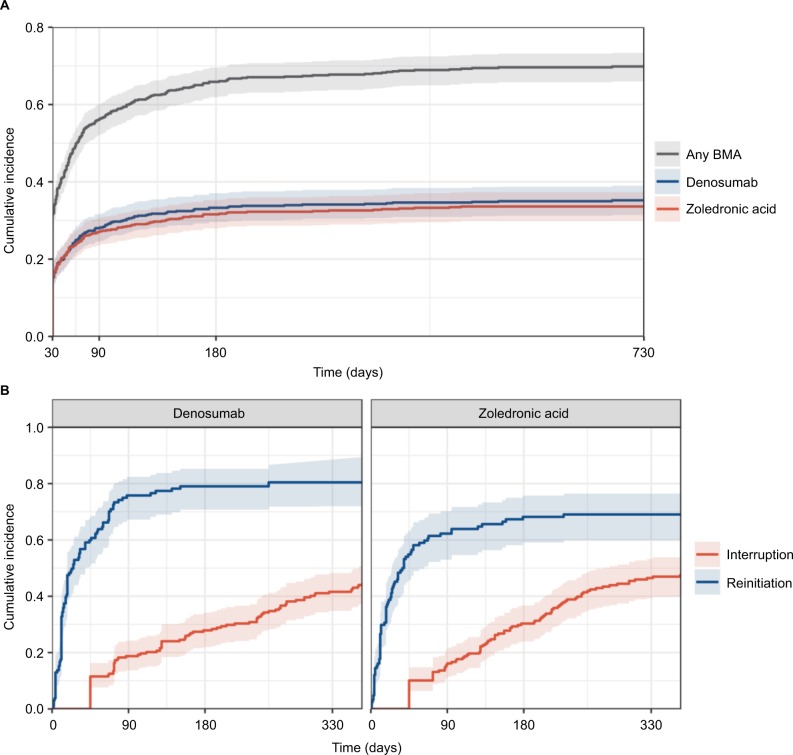
Of the 589 women indicated for BMA therapy, the cumulative incidence of treatment initiation after the diagnosis of bone metastasis was 32% (95% CI: 28%, 35%) at 30 days, 64% (95% CI: 60%, 67%) at 180 days, and 68% (95% CI: 64%, 71%) at 1 year (Figure 1A). The usage of denosumab and zoledronic acid was similar; the cumulative incidence at 90 days was 28% for denosumab and 27% for zoledronic acid. However, there was a small difference between the drugs when stratified by calendar year of bone metastasis diagnosis. Zoledronic acid was used more frequently than denosumab in 2013 (1 year incidence: 33% vs 30%), while denosumab was used more frequently in 2014 (1 year incidence: 38% vs 32%; data not shown), indicating a possible shift in treatment patterns over time.
Figure 1.
Cumulative incidence of bone-modifying agent initiation (A), interruption, and reinitiation (B) among patients with bone metastasis from breast cancer in the US, 2013–2016.
Abbreviation: BMA, bone-modifying agent.
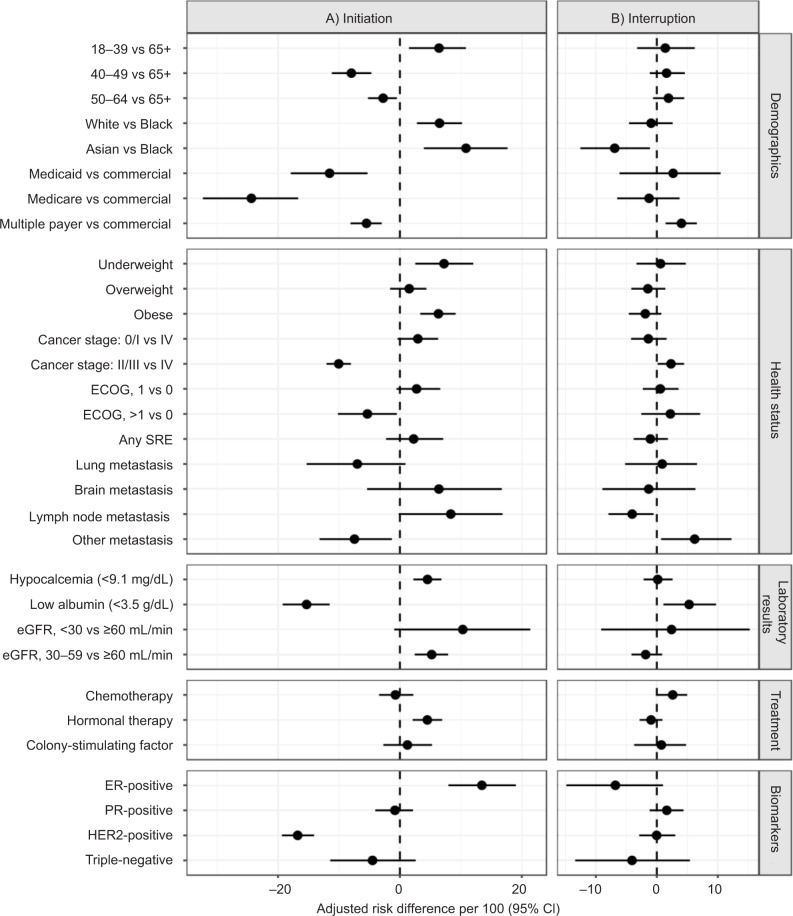
Factors that were positively associated with the use of a BMA included young age and white race. Younger patients were more likely to initiate treatment compared to older women (adjusted risk difference [aRD]: 6.4; 95% CI: 1.5, 10.9), and White patients were more likely to initiate treatment compared to Black patients (aRD: 6.5; 95% CI: 2.8, 10.2). Patients with lower functional status (>1 on ECOG assessment), low albumin, HER2-positive status, and metastases to other sites (not including the lung, brain, and lymph nodes) were less likely to initiate treatment (Figure 2A). Additionally, patients with an eGFR <30 mL/min were less likely to start zoledronic acid (aRD: −12.6; 95% CI: −18.3, −7.1) and more likely to initiate denosumab (aRD: 23.9; 95% CI: 12.5, 35.3) compared to patients with higher eGFR values. Patients with Medicaid and Medicare were less likely to initiate treatment with a BMA, particularly with denosumab (Medicare aRD: −23.3; 95% CI: −28.8, −17.4), compared to those with a commercial health insurance plan; although this finding is based on a relatively small sample of patients with Medicare insurance. Finally, women taking hormonal therapy for their cancer treatment were more likely to initiate denosumab (aRD: 7.3; 95% CI: 5.0, 9.4) and less likely to initiate zoledronic acid (aRD: −2.6; 95% CI: −4.5, −0.5).
Figure 2.
Adjusted risk difference estimates per 100 for variables associated with bone-modifying agent initiation (A) and interruption (B).
Note: Each estimate in the model was adjusted for all other variables listed as well as alkaline phosphatase level and serum calcium level.
Abbreviations: ECOG, Eastern Cooperative Oncology Group Performance Status; eGFR, estimated glomerular filtration rate; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; PR, progesterone receptor; SRE, skeletal-related event.
Interruption and reinitiation
Of the 412 women who initiated a BMA, 46% had a treatment interruption in the first year (95% CI: 41%, 51%) (Figure 1B). The frequencies of interruption of denosumab and zoledronic acid were similar; at 90 days, 19% and 16% of patients taking denosumab and zoledronic acid, respectively, experienced an interruption. Interruption of treatment with either BMA was more likely among patients with low serum albumin and patients with a history of metastasis to other non-bone sites and slightly more likely for patients on chemotherapy (Figure 2B). Only 9.5% of patients experiencing a treatment interruption (N=84) died within 60 days of stopping BMA therapy.
Of the 258 patients who interrupted their BMA therapy, 74% (95% CI: 68%, 79%) reinitiated treatment with the same BMA (Figure 1B), with a median of 18 days (minimum, 1 day; maximum, 402 days) following interruption. The incidence of reinitiation was higher in denosumab patients compared to zoledronic acid patients (80% vs 69% at 1 year, respectively). The number of patients who had a treatment interruption followed by a reinitiation was too small to draw conclusions about the factors associated with reinitiation.
Discussion
Women with breast cancer metastatic to the bone may be candidates for treatment with a BMA for the prevention of SREs. SREs compromise the quality of life, and in some cases, can be fatal. BMAs are used for patients with skeletal metastases alone or with distant metastases at other sites.
In our study of women receiving care at community-based oncology clinics in the US, about one-third of patients with bone metastasis from breast cancer did not receive treatment with a BMA, and there was substantial variation in the timing of initiation for women who did initiate treatment. Among those who started a BMA, about half experienced an interruption by 1 year. However, most patients who experienced a treatment interruption later reinitiated the same type of therapy.
The estimate of BMA initiation in our analysis (68% at 1 year) was similar to a study using administrative health care claims data, which showed that 67% (Medicare) and 58% (commercial health insurance plan) of prevalent breast cancer patients with bone metastasis received a BMA in 2012.25 However, this study focused on prevalent patients rather than incident patients, did not validate bone metastasis, and did not account for censoring. The shift in the type of BMA treatment administered over time was also seen in other solid tumor types, including breast, prostate, and lung. Qian et al26 showed that zoledronic acid was favored in 2012 and 2013, but denosumab was administered more frequently in 2014. The incidence of BMA treatment was lower in our study compared to a large, multicenter cohort study in Germany, where 89% of patients with advanced breast cancer and bone metastasis initiated a BMA. However, a similar distribution between types of drugs was seen; zoledronic acid and denosumab were used almost equally.27 Finally, a study conducted in men with prostate cancer and bone metastasis in routine care in the US showed that BMAs were used at slightly higher rates in this population (77% at 1 year), although incidence of treatment interruption (54% at 1 year) and reinitiation (81% at 1 year) were similar.28
Younger women were more likely to initiate a BMA compared to older women. Older patients likely represent a frailer population with more complex medical regimens and moderate-to-severe comorbidity status. Prior studies have also shown less aggressive treatment patterns for older breast cancer patients.29–31 Similarly, White women were much more likely to initiate BMAs compared to Black women. A similar pattern was observed in a study examining hormonal therapy.32 However, Black women in our study represented a small proportion of the study population (10%).
Patients who were HER2-positive were less likely to initiate treatment compared to those who were HER2-negative. This finding may reflect that HER2-positive breast cancer is more aggressive, more likely to involve lymph nodes, and more likely to recur, which may require more complex treatment.33 Therefore, physicians may be disinclined to initiate non-chemotherapy medications used for supportive care in this population in an effort to reduce the complexity of managing the underlying disease. Several studies have shown that patients who are sicker or show evidence of frailty do not receive preventive therapies.34–36
Patients with Medicaid and Medicare were much less likely to initiate BMAs, particularly denosumab, compared to those with a commercial health insurance plan, although this finding was based on a relatively small sample of patients with Medicare or Medicaid. It is possible that cost could represent a barrier to use, particularly among patients receiving complex and expensive breast cancer treatments.
Renal disease was a common comorbidity in the population at baseline, and its prevalence increased during followup. Patients with an eGFR <30 mL/min were less likely to start zoledronic acid and more likely to initiate denosumab compared to those with higher eGFR values. This finding likely reflects the contraindicated use of zoledronic acid in those with an eGFR <30 mL/min. Although denosumab should also be used with caution in those with renal dysfunction, it is the preferred agent in this population due to the limited evidence of renal toxicity. Renal disease may also be a factor in treatment interruption. While renal disease was not shown to be a predictor of treatment interruption, 31% of patients who had an interruption of any type of BMA treatment had renal disease at some time prior to the interruption.
In this study, we used a 45-day period without treatment to define a treatment interruption; the recommended dosing window for BMAs during the time period of this study was once per 28 days, and we added a short grace period to that dosing schedule. It is possible that dosing patterns could have been influenced by ongoing clinical trials at the time. Specifically, the ZOOM trial37 (conducted in Italy) and the OPTIMIZE-2 trial38 (conducted in the US) both showed that zoledronic acid dosing every 12 weeks was noninferior to dosing every 4 weeks. However, the ZOOM trial was not published until June 2013 and the OPTIMIZE-2 trial was published in 2017. The calendar years of our study (2013–2014) may have been too early to reflect changes in treatment regimens in routine clinical care. Indeed, further examination of dosing regimens among this cohort revealed that very few (<2%) patients initiating denosumab showed evidence of an extended dosing window (ie, Q4 dosing for 6 months followed by Q12 dosing; unpublished data).
Although guidelines recommend the use of denosumab or intravenous bisphosphonates as soon as bone metastases are definitively diagnosed in women with breast cancer, there is limited evidence on the optimal treatment duration.39 This is not something that can be determined from large pivotal trials because the life expectancy of patients with metastatic breast cancer involving the bone may approach or exceed the median length of follow-up in these studies.40–42 For some women with skeletal metastases from breast cancer, life span can be relatively long, exceeding 5 years in 12.5% of the cases;4 thus, these women may be at risk of experiencing a bone complication for several years after their diagnosis of bone metastases. Along with the potential long-term benefits of the drugs, clinicians must also consider the risk of adverse events, including osteonecrosis of the jaw and hypocalcemia. In a recent meta-analysis, denosumab was associated with increased risk of grade 3 or 4 hypocalcemia (relative risk: 1.99; 95% CI: 1.11, 3.54) in comparison to intravenous bisphosphonates, due to denosumab’s powerful antiresorptive effect and reduced risk of renal impairment or toxicity (relative risk: 0.75; 95% CI: 0.61, 0.91) in comparison to intravenous bisphosphonates.40 The rates of osteonecrosis of the jaw can be higher for individuals treated with antiresorptives for longer durations.41 Health care providers should discuss the risks and benefits of BMA therapy with the patients with breast cancer who have metastatic bone disease.
There are several limitations that should be considered when interpreting the results of this study. Our study used EHRs primarily from community-based oncology clinics, and therefore, our results may not be generalizable to other cancer treatment settings because of the limited number of academic centers in the database. Additionally, the EHR systems from oncology centers likely do not fully capture the presence and timing of comorbidities unrelated to cancer care. Indeed, in the present study, the prevalence of chronic comorbidities was substantially lower than what would be expected in this population, and therefore, we were unable to comprehensively assess how these variables relate to BMA treatment initiation or interruption. We were unable to capture detailed clinical information on bone metastasis (eg, number, location) or dental disease or procedures, both of which could be associated with treatment initiation. Furthermore, delays in BMA treatment initiation could be due to ensuring that patients have dental exams, radiographs, and completion of invasive dental procedures prior to starting BMA treatment,18,43,44 or delays could occur while obtaining insurance approval; however, we were unable to differentiate between these in the data. Finally, although laboratory results and functional status (measured via ECOG assessment) were available in the database, they were missing for a large proportion of the population. For example, 79% of the population did not have ECOG measurements during the initiation baseline period. The amount of missing data in these variables decreased with follow-up. Thus, because we treated these variables as time-dependent, we enhanced capture of this information throughout the follow-up.
In summary, based on the data collected in routine clinical practice in the US, approximately one-third of women with bone metastasis from breast cancer do not initiate BMA therapy. Furthermore, among women who do initiate treatment, duration may be suboptimal. Additional studies to further understand the reasons for these observed trends (eg, preferences of the patient or treating clinician), whether altered dosing guidelines changed treatment patterns in later years, as well as the consequences of remaining untreated or experiencing interruptions in treatment, are warranted.
Acknowledgments
This study was funded by Amgen Inc.
Footnotes
Disclosure
RKH, KC, and AL are employees of Amgen Inc and have stock ownership in Amgen Inc. MAB has had research support from Amgen and AstraZeneca, owns equity in NoviSci, LLC, and has served as a scientific advisor/consultant to Amgen, Merck, TargetPharma, Genentech, AbbVie, and RxAnte, LLC. LJM, RAO, DR, and SAN have no potential conflicts of interest to report in this work.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. doi: 10.3322/caac.21442. [DOI] [PubMed] [Google Scholar]
- 2.Li S, Peng Y, Weinhandl ED, et al. Estimated number of prevalent cases of metastatic bone disease in the US adult population. Clin Epidemiol. 2012;4:87–93. doi: 10.2147/CLEP.S28339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang H, Zhu W, Biskup E, et al. Incidence, risk factors and prognostic characteristics of bone metastases and skeletal-related events (SREs) in breast cancer patients: A systematic review of the real world data. J Bone Oncol. 2018;11:38–50. doi: 10.1016/j.jbo.2018.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liede A, Jerzak KJ, Hernandez RK, Wade SW, Sun P, Narod SA. The incidence of bone metastasis after early-stage breast cancer in Canada. Breast Cancer Res Treat. 2016;156(3):587–595. doi: 10.1007/s10549-016-3782-3. [DOI] [PubMed] [Google Scholar]
- 5.Hernandez RK, Wade SW, Reich A, Pirolli M, Liede A, Lyman GH. Incidence of bone metastases in patients with solid tumors: analysis of oncology electronic medical records in the United States. BMC Cancer. 2018;18(1):44. doi: 10.1186/s12885-017-3922-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coleman RE. Clinical features of metastatic bone disease and risk of skeletal morbidity. Clin Cancer Res. 2006;12(20 Pt 2):6243s–6249. doi: 10.1158/1078-0432.CCR-06-0931. [DOI] [PubMed] [Google Scholar]
- 7.Wardley A, Davidson N, Barrett-Lee P, et al. Zoledronic acid significantly improves pain scores and quality of life in breast cancer patients with bone metastases: a randomised, crossover study of community vs hospital bisphosphonate administration. Br J Cancer. 2005;92(10):1869–1876. doi: 10.1038/sj.bjc.6602551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sathiakumar N, Delzell E, Morrisey MA, et al. Mortality following bone metastasis and skeletal-related events among women with breast cancer: a population-based analysis of U.S. Medicare beneficiaries, 1999–2006. Breast Cancer Res Treat. 2012;131(1):231–238. doi: 10.1007/s10549-011-1721-x. [DOI] [PubMed] [Google Scholar]
- 9.Hagiwara M, Delea TE, Chung K. Healthcare costs associated with skeletal-related events in breast cancer patients with bone metastases. J Med Econ. 2014;17(3):223–230. doi: 10.3111/13696998.2014.890937. [DOI] [PubMed] [Google Scholar]
- 10.Jensen AØ, Jacobsen JB, Nørgaard M, Yong M, Fryzek JP, Sørensen HT. Incidence of bone metastases and skeletal-related events in breast cancer patients: a population-based cohort study in Denmark. BMC Cancer. 2011;11:29. doi: 10.1186/1471-2407-11-29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pamidronate Disodium [prescribing information] Bedford, OH. Bedford Laboratories; 2014. [Accessed August 27, 2018]. Available from: https://www.accessdata.fda.gov/drug-satfda_docs/label/2014/021113s017lbl.pdf. [Google Scholar]
- 12.Zometa [prescribing information] East Hanover, NJ: Novartis Pharmaceuticals Corporation; 2016. [Accessed August 27, 2018]. Available from: https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/files/Zometa.pdf. [Google Scholar]
- 13.XGEVA [prescribing information] Thousand Oaks, CA: Amgen Inc; 2017. [Accessed August 27, 2018]. Available from: http://pi.amgen.com/~/media/amgen/repository-sites/pi-amgen-com/xgeva/xgeva_pi.ashx. [Google Scholar]
- 14.Wong MH, Stockler MR, Pavlakis N. Bisphosphonates and other bone agents for breast cancer. Cochrane Database Syst Rev. 2012;2:Cd003474. doi: 10.1002/14651858.CD003474.pub3. [DOI] [PubMed] [Google Scholar]
- 15.Ford JA, Jones R, Elders A, et al. Denosumab for treatment of bone metastases secondary to solid tumours: systematic review and network meta-analysis. Eur J Cancer. 2013;49(2):416–430. doi: 10.1016/j.ejca.2012.07.016. [DOI] [PubMed] [Google Scholar]
- 16.Hatoum HT, Lin SJ, Smith MR, Barghout V, Lipton A. Zoledronic acid and skeletal complications in patients with solid tumors and bone metastases: analysis of a national medical claims database. Cancer. 2008;113(6):1438–1445. doi: 10.1002/cncr.23775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hatoum HT, Lin SJ, Smith MR, Guo A, Lipton A. Treatment persistence with monthly zoledronic acid is associated with lower risk and frequency of skeletal complications in patients with breast cancer and bone metastasis. Clin Breast Cancer. 2011;11(3):177–183. doi: 10.1016/j.clbc.2011.03.015. [DOI] [PubMed] [Google Scholar]
- 18.van Poznak CH, Temin S, Yee GC, et al. American Society of Clinical Oncology executive summary of the clinical practice guideline update on the role of bone-modifying agents in metastatic breast cancer. J Clin Oncol. 2011;29(9):1221–1227. doi: 10.1200/JCO.2010.32.5209. [DOI] [PubMed] [Google Scholar]
- 19.Gralow JR, Biermann JS, Farooki A, et al. NCCN Task Force Report: Bone Health In Cancer Care. J Natl Compr Canc Netw. 2013;11(Suppl 3):S1–50. doi: 10.6004/jnccn.2013.0215. quiz S51. [DOI] [PubMed] [Google Scholar]
- 20.Flatiron Health. [Accessed August 27, 2018]. Available from: https://flatiron.com/real-world-evidence/
- 21.Liede A, Hernandez RK, Roth M, Calkins G, Larrabee K, Nicacio L. Validation of International Classification of Diseases coding for bone metastases in electronic health records using technology-enabled abstraction. Clin Epidemiol. 2015;7:441–448. doi: 10.2147/CLEP.S92209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–612. doi: 10.7326/0003-4819-150-9-200905050-00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fine JP, Gray RJ. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J Am Stat Assoc. 1999;94(446):496–509. [Google Scholar]
- 24.R: A Language and Environment for Statistical Computing [computer program] Vienna: R Foundation for Statistical Computing; 2017. [Google Scholar]
- 25.Hernandez RK, Adhia A, Wade SW, et al. Prevalence of bone metastases and bone-targeting agent use among solid tumor patients in the United States. Clin Epidemiol. 2015;7:335–345. doi: 10.2147/CLEP.S85496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Qian Y, Bhowmik D, Kachru N, Hernandez RK. Longitudinal patterns of bone-targeted agent use among patients with solid tumors and bone metastases in the United States. Support Care Cancer. 2017;25(6):1845–1851. doi: 10.1007/s00520-017-3583-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schroder J, Fietz T, Kohler A, et al. Treatment and pattern of bone metastases in 1094 patients with advanced breast cancer - Results from the prospective German Tumour Registry Breast Cancer cohort study. Eur J Cancer. 2017;79:139–148. doi: 10.1016/j.ejca.2017.03.031. [DOI] [PubMed] [Google Scholar]
- 28.Butler AM, Cetin K, Hernandez RK, et al. Treatment dynamics of bone-targeting agents among men with bone metastases from prostate cancer in the United States. Pharmacoepidemiol Drug Saf. 2018;27(2):229–238. doi: 10.1002/pds.4360. [DOI] [PubMed] [Google Scholar]
- 29.Eaker S, Dickman PW, Bergkvist L, Holmberg L, Uppsala/Orebro Breast Cancer Group Differences in management of older women influence breast cancer survival: results from a population-based database in Sweden. PLoS Med. 2006;3(3):e25. doi: 10.1371/journal.pmed.0030025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Enger SM, Thwin SS, Buist DS, et al. Breast cancer treatment of older women in integrated health care settings. J Clin Oncol. 2006;24(27):4377–4383. doi: 10.1200/JCO.2006.06.3065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bouchardy C, Rapiti E, Blagojevic S, Vlastos AT, Vlastos G. Older female cancer patients: importance, causes, and consequences of undertreatment. J Clin Oncol. 2007;25(14):1858–1869. doi: 10.1200/JCO.2006.10.4208. [DOI] [PubMed] [Google Scholar]
- 32.Sheppard VB, Faul LA, Luta G, et al. Frailty and adherence to adjuvant hormonal therapy in older women with breast cancer: CALGB protocol 369901. J Clin Oncol. 2014;32(22):2318–2327. doi: 10.1200/JCO.2013.51.7367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Burstein HJ. The distinctive nature of HER2-positive breast cancers. N Engl J Med. 2005;353(16):1652–1654. doi: 10.1056/NEJMp058197. [DOI] [PubMed] [Google Scholar]
- 34.Glynn RJ, Knight EL, Levin R, Avorn J. Paradoxical relations of drug treatment with mortality in older persons. Epidemiology. 2001;12(6):682–689. doi: 10.1097/00001648-200111000-00017. [DOI] [PubMed] [Google Scholar]
- 35.Redelmeier DA, Tan SH, Booth GL. The treatment of unrelated disorders in patients with chronic medical diseases. N Engl J Med. 1998;338(21):1516–1520. doi: 10.1056/NEJM199805213382106. [DOI] [PubMed] [Google Scholar]
- 36.Jackson LA, Nelson JC, Benson P, et al. Functional status is a confounder of the association of influenza vaccine and risk of all cause mortality in seniors. Int J Epidemiol. 2006;35(2):345–352. doi: 10.1093/ije/dyi275. [DOI] [PubMed] [Google Scholar]
- 37.Amadori D, Aglietta M, Alessi B, et al. Efficacy and safety of 12-weekly versus 4-weekly zoledronic acid for prolonged treatment of patients with bone metastases from breast cancer (ZOOM): a phase 3, open-label, randomised, non-inferiority trial. Lancet Oncol. 2013;14(7):663–670. doi: 10.1016/S1470-2045(13)70174-8. [DOI] [PubMed] [Google Scholar]
- 38.Himelstein AL, Foster JC, Khatcheressian JL, et al. Effect of Longer-Interval vs Standard Dosing of Zoledronic Acid on Skeletal Events in Patients With Bone Metastases: A Randomized Clinical Trial. JAMA. 2017;317(1):48–58. doi: 10.1001/jama.2016.19425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.van Poznak C, Somerfield MR, Barlow WE, et al. Role of Bone-Modifying Agents in Metastatic Breast Cancer: An American Society of Clinical Oncology-Cancer Care Ontario Focused Guideline Update. J Clin Oncol. 2017;35(35):3978–3986. doi: 10.1200/JCO.2017.75.4614. [DOI] [PubMed] [Google Scholar]
- 40.Menshawy A, Mattar O, Abdulkarim A, et al. Denosumab versus bisphosphonates in patients with advanced cancers-related bone metastasis: systematic review and meta-analysis of randomized controlled trials. Support Care Cancer. 2018;26(4):1029–1038. doi: 10.1007/s00520-018-4060-1. [DOI] [PubMed] [Google Scholar]
- 41.Stopeck AT, Fizazi K, Body JJ, et al. Safety of long-term denosumab therapy: results from the open label extension phase of two phase 3 studies in patients with metastatic breast and prostate cancer. Support Care Cancer. 2016;24(1):447–455. doi: 10.1007/s00520-015-2904-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stopeck AT, Lipton A, Body JJ, et al. Denosumab compared with zoledronic acid for the treatment of bone metastases in patients with advanced breast cancer: a randomized, double-blind study. J Clin Oncol. 2010;28(35):5132–5139. doi: 10.1200/JCO.2010.29.7101. [DOI] [PubMed] [Google Scholar]
- 43.Ruggiero SL, Dodson TB, Fantasia J, et al. American Association of Oral and Maxillofacial Surgeons position paper on medication-related osteonecrosis of the jaw-2014 update. J Oral Maxillofac Surg. 2014;72(10):1938–1956. doi: 10.1016/j.joms.2014.04.031. [DOI] [PubMed] [Google Scholar]
- 44.Khan AA, Morrison A, Hanley DA, et al. Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Miner Res. 2015;30(1):3–23. doi: 10.1002/jbmr.2405. [DOI] [PubMed] [Google Scholar]