Abstract
Patients with acute myeloid leukemia (AML) often relapse after initial therapy because of persistence of leukemic stem cells that frequently express the IL-3 receptor alpha chain CD123. Natural killer (NK) cell-based therapeutic strategies for AML show promise and we explore the NK cell lines, NK-92 and CD16+ NK-92, as a treatment for AML. NK-92 has been tested in phase I clinical trials with minimal toxicity; irradiation prior to infusion prevents risk of engraftment. The CD16 negative NK-92 parental line was genetically modified to express the high affinity Fc gamma receptor, enabling antibody-dependent cell-mediated cytotoxicity, which we utilized in combination with an anti-CD123 antibody to target leukemic stem cells. NK-92 was preferentially cytotoxic against leukemic stem and progenitor cells compared with bulk leukemia in in vitro assays, while CD16+ NK-92 in combination with an anti-CD123 mAb mediated antibody-dependent cell-mediated cytotoxicity against CD123+ leukemic targets. Furthermore, NK-92 infusions (with or without prior irradiation) improved survival in a primary AML xenograft model. Mice xenografted with primary human AML cells had a superior survival when treated with irradiated CD16+NK-92 cells and an anti-CD123 monoclonal antibody (7G3) versus treatment with irradiated CD16+NK-92 cells combined with an isotype control antibody. In this proof-of-principle study, we show for the first time that a CD16+NK-92 cell line combined with an antibody that targets a leukemic stem cell antigen can lead to improved survival in a relevant pre-clinical model of AML.
Introduction
Acute myeloid leukemia (AML) accounts for the majority of acute leukemias in adults and a minority in children.1,2 While up to 70-85% of AML patients treated with current chemotherapy protocols achieve morphological remission,1,3 many relapse because of recurrence from residual leukemic stem cells (LSCs) resulting in an overall 5-year survival of approximately 40%.2 AML was the first malignancy with clear evidence of a stem cell hierarchy, with the LSCs being enriched in the CD34+CD38− fraction.4,5 In addition, they often express the IL-3 receptor alpha chain (CD123), a marker not highly expressed on normal hematopoietic stem cells.6 AML patients with a greater than 1% burden of CD34+CD38−CD123+ LSCs at diagnosis have a reduced disease-free and overall survival rate, directly implicating CD123 as a relevant target antigen.7 Natural killer (NK)-cell-based approaches are under development for the treatment of AML, such as the use of haploidentical NK-cell infusions.8,9 While this shows promise, there is inherent variability in the NK-cell preparations. Another approach is to use a permanent NK cell line, such as NK-92 which was derived from a patient with an NK-cell lymphoma,10 and demonstrates enhanced cytotoxicity over endogenously-derived NK cells against a variety of human leukemia cell lines and primary leukemic blasts.11 However, this cell line lacks the Fc gamma receptor IIIA (CD16), typically expressed by NK cells and, therefore, cannot mediate antibody-dependent cell-mediated cytotoxicity (ADCC). NK-92 has been tested in three published phase I clinical trials, including one clinical trial by our group for relapsed and refractory hematologic cancers (lymphoma and multiple myeloma), which all demonstrated minimal toxicity.12–14 However, to prevent potential engraftment of NK-92 and generate a NK malignancy, the cells are irradiated with 1000 cGy which does not significantly decrease in vitro cytotoxicity.15–17
Natural killer cells typically express CD16 and are able to mediate ADCC against antibody-coated targets, enabling both adaptive and innate immune responses. Since the parental NK-92 cell line lacks CD16, and cannot mediate ADCC, a high-affinity allelic variant (valine at position 176 instead of phenylalanine) of the CD16A Fcγ receptor was transduced into the NK-92 cell line. These gene-modified CD16+NK-92 cells (NK-92.176V and NK-92.176V.GFP) demonstrate ADCC in vitro18 but have not been tested in vivo.
Here, we show that NK-92 preferentially kills leukemic stem cells compared with bulk leukemia cells and can prolong survival with or without prior radiation. Moreover, gene-modified NK-92 expressing the high affinity CD16 receptor (NK-92.176V.GFP) more effectively killed CD123+ targets in vitro and demonstrated an enhanced ability to target LSCs. Finally, irradiated CD16+NK-92 combined with the anti-CD123 antibody, 7G3, enhanced survival in a primary AML xenograft model compared with control arms.
Methods
Cell lines and primary samples
K562 was obtained from the American Type Culture Collection (Manassas, VA, USA) and maintained in IMDM+10% FBS. OCI/AML2, OCI/AML3 and OCI/AML5 were generously provided by Dr. Mark Minden and maintained in MEMalpha+ 10%FBS (OCI/AML2 and OCI/AML3) or MEMalpha+10% FBS and 10% 5637 bladder carcinoma conditioned medium (OCI/AML5). NK-92 was originally kindly provided by Dr. Hans Klingemann, expanded and was maintained in X-VIVO 10 medium (Lonza) supplemented with 450 U/mL of IL-2 and 2.5% human AB serum (GM1). Four primary AML samples were obtained from the Princess Margaret Hospital Leukemia Tissue Bank, Toronto, Canada, according to an approved institutional protocol. NK-92 and NK-92.176V GFP (hereafter referred to as CD16+NK-92) was obtained from Conkwest under a Material Transfer Agreement (MTA) and maintained as described for NK-92. Frozen master cell banks for cell lines were established and new vials utilized to establish new cultures every six weeks. Mycoplasma testing by PCR was conducted periodically with all cultures testing negative.
Chromium release assay
We used a chromium release assay (CRA) as previously described by our group19 and detailed in the Online Supplementary Methods.
Flow cytometry and cell sorting
Immunophenotyping of bone marrow (BM) was performed using an FC500 or Facscalibur flow cytometer. FACS buffer was made with PBS+2mM EGTA+2% FBS. Primary AML and leukemic stem cell fractions were detected using the following antibodies (company; product #; clone): anti-CD34 PE (BD bio-sciences; 348057; 8G12), anti-CD34 FITC (BD Pharmingen; 555821; 581), anti-CD38 APC (ebiosciences; 17-0389-42; HIT2) and anti-CD123 PE (BD Pharmingen; 558714; 7G3), anti-CD45 APC (BD Pharmingen; 557513; TU116) and anti-class I HLA A, B, C (Biolegend; 311404; W6/32). NK-92 cells lines were assessed for CD16 expression using CD16 PE (Biolegend; 302008; 3G8). Leukemia cell lines were evaluated using anti-CD123 PerCy5.5 (BD Biosciences; 560904; 7G3). Cell sorting was performed using a FacsAria cell sorter as described in the Online Supplementary Methods.
Methylcellulose cytotoxicity assay
We used a methylcellulose cytotoxicity assay as previously described19 and included in the Online Supplementary Methods and in Supplementary Figure S1. Briefly, cell line or primary AML cells were incubated with and without NK-92 in a 4-hour assay prior to infusion into methylcellulose. Colonies were assessed at 2-4 weeks and percent colony inhibition calculated. NK-92 did not grow colonies under these conditions.
Animals
NOD/SCID gammanull (NSG) mice from The Jackson Laboratory were bred and maintained in the Ontario Cancer Institute animal facility according to protocols approved by the Animal Care Committee. Mice were fed irradiated food and Baytril containing water ad libitum during experimental periods. Prior to infusion with AML, NSG mice were irradiated with 325 or 225 cGy to facilitate engraftment. We developed a primary AML xenograft model utilizing a patient-derived AML sample (details in the Online Supplementary Methods and Supplementary Figure S2). Mice were sacrificed when humane end points were reached as per our animal use protocol (2791). Primary AML xenografted mice were treated with NK-92 (kindly provided by Hans Klingemann) or CD16+NK-92 (provided by Conkwest), with or without murine antibody therapy as follows: 7G3 (provided to RMR under an MTA with CSL Ltd., Parkville, Australia), BM4 isotype control (under MTA to RMR) and MG2a-53 isotype control (Biolegend; 401502; MG2a-53).
Statistical analysis
Survival analysis was carried out with Kaplan-Meier survival curves using the log rank rest (P<0.05) with Medcalc software. Comparison of cytotoxicity was made with the two-tailed Student t-test (P<0.05) to compare in vitro cytotoxicity and engraftment data using Medcalc software.
Results
NK-92 preferentially kills leukemic stem cells compared with bulk leukemia cells
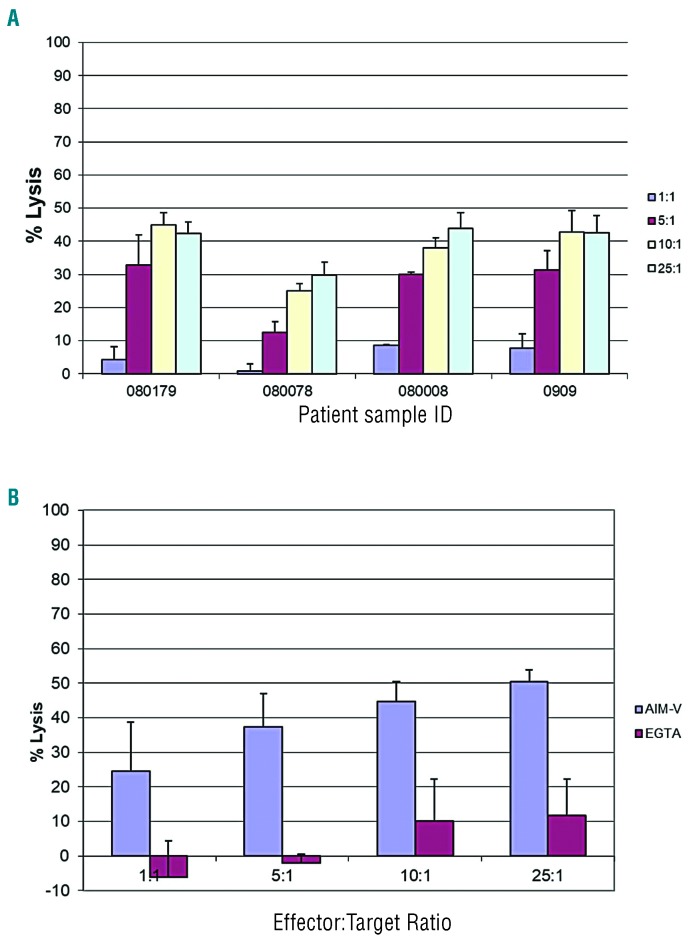
We initially set out to determine the cytotoxicity of NK-92 against primary AML cells using chromium release as a measure of bulk tumor cell kill. A panel of 4 primary AML blast samples treated with NK-92 yielded a dose-dependent response and moderate degrees of cytotoxicity against 4 samples at a 25:1 E:T ratio (% lysis): 080179 (42.3±3.6%), 080078 (29.8±3.6%), 08008 (43.9±1.47%), 0909 (42.6±0.1%) (Figure 1A). Primary AML cells were killed in a dose-dependent manner and killing was abrogated in the presence of the calcium chelator EGTA that blocks granule exocytosis (Figure 1B).
Figure 1.
Chromium release assay of NK-92 against primary acute myeloid leukemia (AML) samples. (A) Four freshly thawed primary AML blast samples were labeled with 100 μCi of Na251CrO4 prior to treatment with NK-92 at four E:T ratios. (B) AML blast sample 080078 was tested in a separate experiment at four E:T ratios with and without calcium chelator EGTA 4 (mM) and MgCl2 (3 mM). Data are presented as the mean percent lysis of triplicate samples (+/−Standard Deviation) from a representative experiment carried out three times.
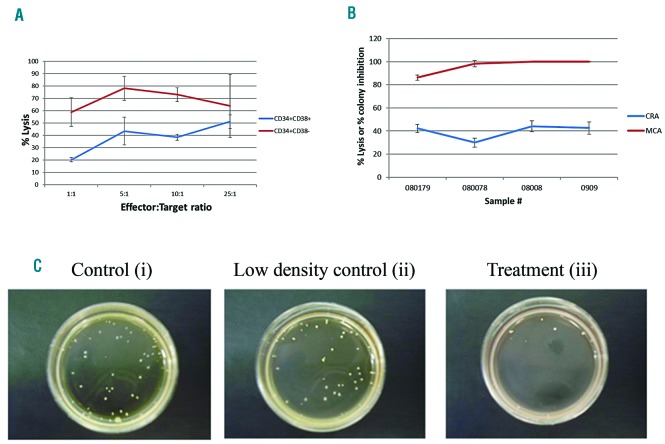
To determine the effect of NK-92 on LSCs, we sorted a primary AML sample into CD34+CD38− and CD34+CD38+ fractions for further testing using the CRA. Primary AML-derived CD34+CD38− cells were more sensitive to killing than CD34+CD38+ blasts by NK-92 in a 4-hour CRA at E:T ratios of 1:1 (58.9±11.5%, 20.3±1.7%), 5:1 (78.3±9.7%, 43.5+11.1%) and 10:1 (72.9±5.6%, 38.5±2.4%); this difference was not significant at a 25:1 E:T ratio (Figure 2A).
Figure 2.
NK-92 cytotoxicity against sorted leukemic stem cells and clonogenic leukemic cells relative to bulk leukemia cells. (A) Primary acute myeloid leukemia (AML) samples were sorted into CD34+CD38− and CD34+CD38+ fractions for subsequent testing in a chromium release assay with NK-92 at a 25:1 E:T ratio. Data are presented as the mean percent (%) lysis of triplicate samples (+/−Standard Deviation) representative of two separate experiments. (B) Four primary AML samples were incubated with or without NK-92 at a 25:1 E:T ratio for 4 hours in 96-well U bottom plates and utilized in either a chromium release assay (CRA) or a methylcellulose cytotoxicity assay (MCA) conducted on the same day. The % lysis values and % colony inhibition values are plotted together (B). An example of the methylcellulose cytotoxicity assay (C) shows a representative assay for one sample (080179) with a control (AML only) (i), low density control (AML + NK-92 infused into methylcellulose only) (ii), and treatment group (AML + NK-92 co-incubated together in a 96-well plate well and then infused in methylcellulose) (iii).
To test the effect of NK-92 against LSCs relative to bulk tumor, we compared the CRA with a methylcellulose cytotoxicity assay (MCA) designed to measure the killing of clonogenic primary AML cells during the 4-hour co-incubation. We also employed a control to correct for the effect of NK-92 against leukemia targets during the 4-week exposure in methylcellulose by enumerating colonies that arose from infusing NK-92 and targets in methylcellulose without prior incubation. The MCA showed that NK-92 at a 25:1 E:T eliminated clonogenic growth of most primary AML blast samples yielding % colony inhibition values of: 86.3±2.3%, 98.4±2.8%, 100±0% and 100±0%, demonstrating much higher cytotoxicity than obtained with the CRA, which was performed on the same day (Figure 2B and C).
To determine if the difference in cytotoxicity measured by the CRA and MCA was not simply due to methodological reasons, we screened additional cell line targets using this methodology. While OCI/AML3 clonogenic cells were more sensitive to NK-92 than bulk tumor, OCI/AML2 bulk and clonogenic cells were equivalently sensitive to NK-92 (Online Supplementary Figure S3). OCI/AML2 was the only target tested which did not have a differential cytotoxicity measured by the CRA and MCA. This demonstrates that the enhanced cytotoxicity measured in the MCA compared with the CRA is, for most targets, not intrinsically related to the method of data comparison.
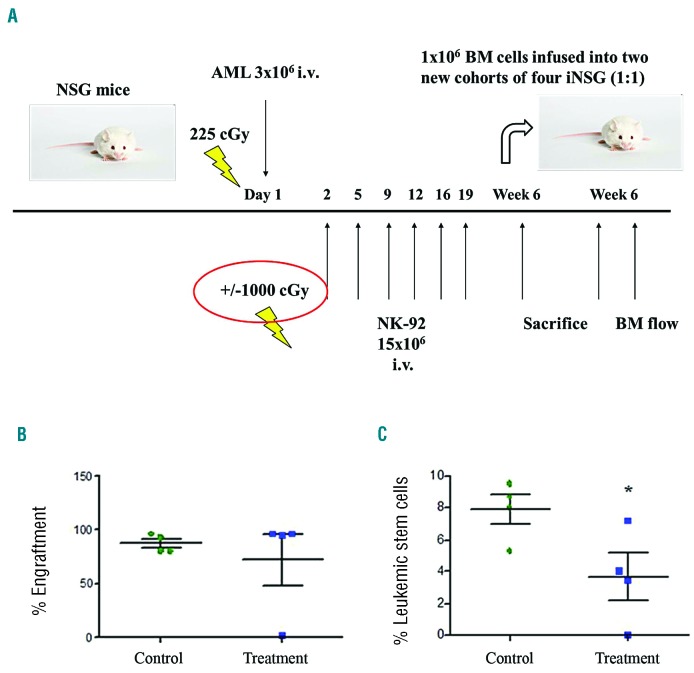
Irradiated NK-92 reduces leukemic stem cell fraction in the secondary transplantation assay
To assess the cytotoxic effect of irradiated NK-92 (iNK-92) on LSCs in the in vivo setting, secondary transplantation experiments were conducted to evaluate total engraftment and fraction of LSCs in secondary recipients. Primary AML cells (3×106) were injected by tail vein into two cohorts of 4 mice and treated with or without iNK-92 from day 2 (15×106 iNK-92 cells) twice weekly to a total dose of 75×106 iNK-92 cells. At six weeks, mice were sacrificed and BM (1×106 cells) from each of the 4 primary recipients (donor mice) in control or treatment groups transplanted into 4 secondary recipient NSG mice (Figure 3A). Evaluation of BM from secondary recipients inoculated with BM from AML-infused mice untreated by iNK-92 revealed a high proportion of human CD45+ cells (80.8, 93.3, 80.4, 96.4 Av=87.7%), while one mouse from AML infused iNK-92 treatment group was leukemia-free with engraftment at background levels of non-injected mice (96.4, 94.7, 1.8, 95.7 Av=72.2%). There was no significant difference between secondary engraftment of AML between groups receiving BM from AML-infused mice in the control and iNK-92 therapy groups (Figure 3B). However, the proportion of CD34+CD38−CD123+ cells in secondary transplanted mice for the AML control group was 7.85% (8.01, 9.48, 8.66, 5.25) and for the AML + iNK-92 group was 3.66% (7.13, 3.46, 0.03, 4.00), which was significantly lower (P=0.05) (Figure 3C).
Figure 3.
Effect of iNK-92 on secondary bone marrow (BM) engraftment of acute myeloid leukemia (AML) cells and leukemic stem cells (LSCs). (A) 3×106 AML cells were also infused intravenously (i.v.) into two cohorts of 4 mice and treated with and without iNK-92 from day 2 and given 15×106 cells twice weekly to a total dose of 75×106 (A). BM (1×106 cells) from each of 4 primary recipients in control and treatment was serially transplanted 1:1 into 4 new NOD/SCID gammanull (NSG) mice. These mice were sacrificed at six weeks and BM assayed for overall leukemic engraftment as determined by presence of % human CD45+ cells (B) and LSC engraftment as determined by % human CD34+CD38−CD123+ cells (C) (*P=0.05).
NK-92 prolongs survival in a primary AML xenograft model
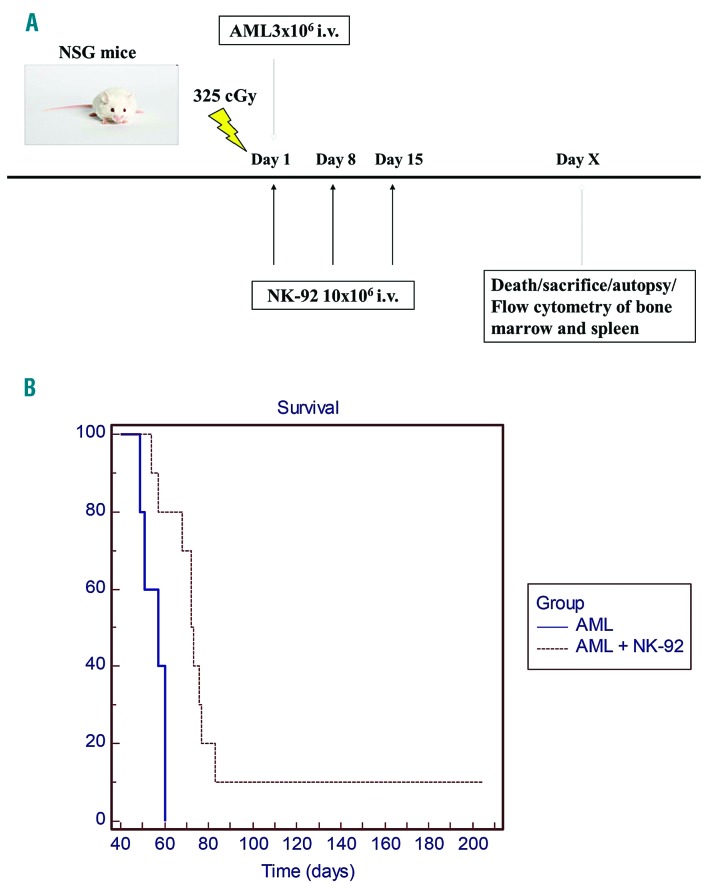
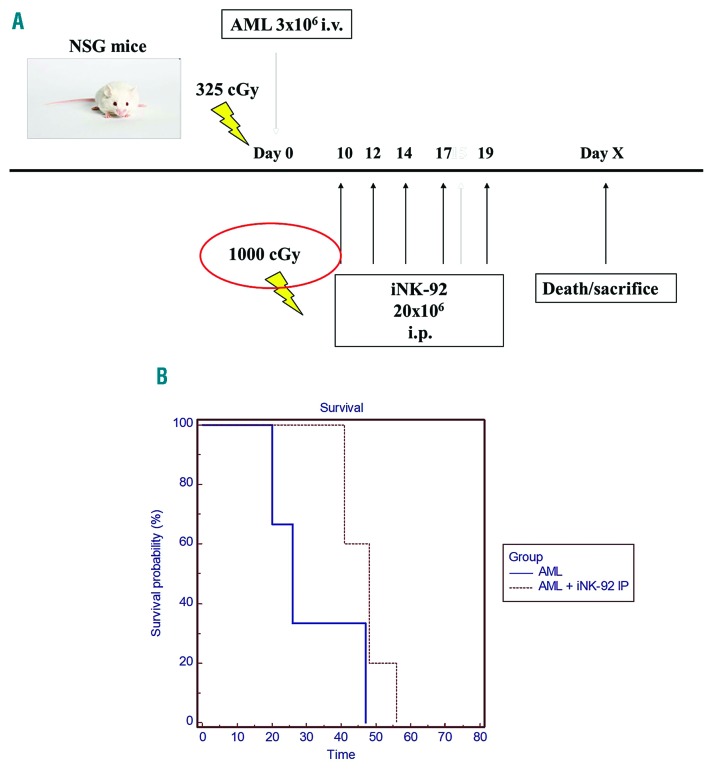
We next sought to assess the impact of NK-92 on survival of mice inoculated with primary human AML. NSG mice inoculated with 3×106 primary AML cells received 10×106 non-irradiated NK-92 weekly for three doses (Figure 4A). This treatment increased median survival from 57 to 72 days (log rank test P<0.01), although most ultimately succumbed to disease (Figure 4B). Autopsy revealed enlarged spleens and pale fragile bones compared with controls. Flow cytometry of BM from NSG mice inoculated with AML only (Online Supplementary Figure S4A), or AML + NK-92 treatments that became symptomatic (Online Supplementary Figure S4B), had 99% engraftment by human leukemia in the bone marrow, while the mouse that survived long term (~9 months) was healthy at sacrifice and did not have evidence of leukemic infiltration in the marrow (Online Supplementary Figure S4C) or splenomegaly. To determine the impact of irradiation on the in vivo activity of NK-92, the cells were irradiated with 1000 cGy prior to infusion into NSG mice inoculated ten days before with 3×106 primary AML cells. NSG mice were administered 20×106 iNK-92 [intraperitoneal injection (i.p.)] weekly × 5 doses and monitored for signs of leukemia (Figure 5A). Survival was improved in the treatment group (26-48 days) to near statistical significance (P=0.0566), but all mice ultimately succumbed to disease (Figure 5B).
Figure 4.
NK-92 therapy of primary acute myeloid leukemia (AML) xenografted NOD/SCID gammanull (NSG) mice. 3×106 primary AML cells were injected intravenously (i.v.) via tail vein into irradiated NOD/SCID gamma null mice to establish disease in control (n=5) and therapy mice (n=10). 10×106 NK-92 were infused via tail vein weekly for three weeks starting on the day of AML inoculation in treatment group (A). Mice were monitored for signs of leukemia and sacrificed at humane end points. Kaplan-Meier survival curves were generated to compare survival in control and treatment groups (P<0.01) (B).
Figure 5.
iNK-92 therapy of primary acute myeloid leukemia (AML) xenografted NOD/SCID gammanull (NSG) mice. 3×106 primary AML cells were injected intravenously (i.v.) via tail vein into irradiated NGS to establish disease in control (n=5) and therapy (n=5) mice. iNK-92 given intraperitoneally (i.p.) 20×206 weekly for six weeks were used to treat AML xenografted mice starting ten days after inoculation (A). Mice were monitored for signs of leukemia and sacrificed at humane end points (B). Kaplan-Meier survival curves were generated to compare survival in control and treatment groups (P=0.0566).
CD16+NK-92 mediates ADCC in vitro and in vivo and prolongs survival in an AML xenograft model by targeting leukemic stem cells
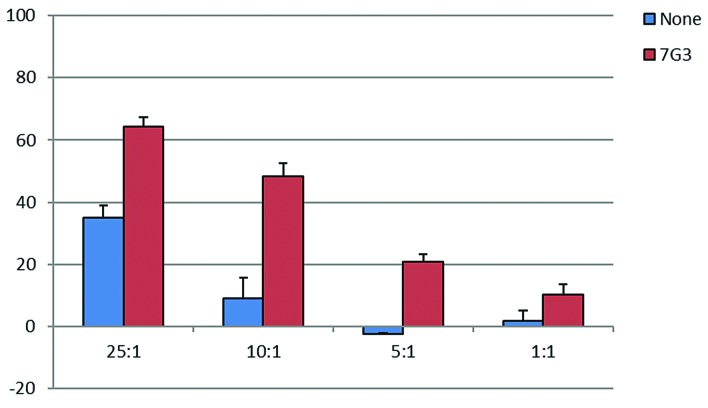
To develop a strategy to enhance killing of LSCs, we utilized a gene-modified CD16+NK-92 transduced with the high affinity CD16 receptor (NK-92.176V GFP), which is capable of mediating ADCC against antibody-coated targets. The proportion of cells expressing CD16 was 2.3% for parental NK-92 and 27.9% for CD16+ NK-92 (Online Supplementary Figure S5). The chromium release assay was modified to measure ADCC by coating target cells with antibodies prior to the 2-hour chromium incubation. The CD123+ leukemia cell line OCI/AML5 was pretreated with anti-CD123 (7G3) antibody at a dose of 10 μg/mL prior to use in a chromium release assay. CD16+NK-92 showed cytotoxicity against OCI/AML5 cells at all E:T ratios, and was significantly enhanced (2-6×) when targets were coated with anti-CD123 mAb (Figure 6), demonstrating effective ADCC.
Figure 6.
CD16+NK-92 in vitro ADCC assay against primary acute myeloid leukemia (AML) and OCI/AML5. OCI/AML5 cells were labeled with 100 μCi of Na251CrO4 for 2 hours +/− 10 μg/mL of 7G3 (anti-CD123 mAb) prior to treatment with CD16+NK-92 in 96-well plates in a standard chromium release assay. Data are presented as the mean percent lysis of triplicate samples (+/−Standard Deviation) from a representative experiment carried out twice.
To enhance the approach of irradiated NK-92 against human AML in the xenograft NSG model, we treated the mice with a CD16+NK-92 cell line, in combination with an anti-CD123 mAb (7G3), given on the same days to facilitate targeting of leukemic stem cells by antibody-dependent cell-mediated cytotoxicity. A blocking dose of isotype control antibody BM4 (200 μg) was given prior to the administration of 7G3 at a low dose (8 μg), due to the known sequestration of 7G3 in the spleen (Online Supplementary Figure S6A). In this pilot experiment, we demonstrated that 7G3 (8 μg) could enhance the therapeutic efficacy of iCD16+ NK-92, as determined by an improvement of median survival by 13 days (P=0.0173) (Online Supplementary Figure S6B).
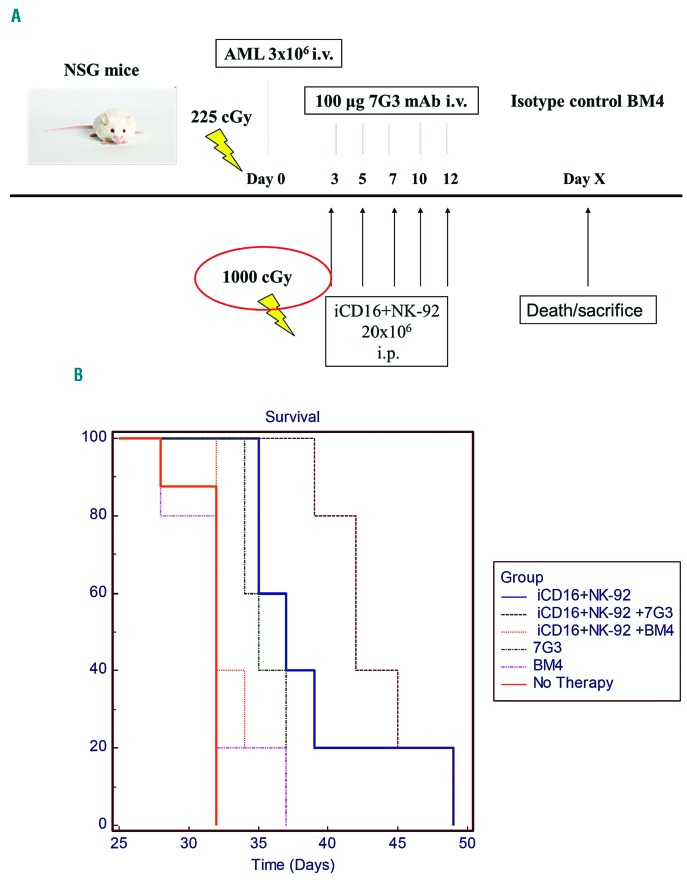
We conducted a more rigorously controlled experiment of CD16+NK-92 and anti-CD123 mAb therapy utilizing an isotype control antibody (BM4) in the control arms (Figure 7A). The cohorts included: no therapy, 7G3, BM4, iCD16+NK-92, iCD16+NK-92 + 7G3, iCD16+NK-92 + BM4. The dosing schedule was iCD16+NK-92, with or without 7G3 or BM4, given on days 3, 5, 7, 10, and 12 after AML inoculation (day 0). NSG mice without therapy had a median survival of 32 days. iCD16+NK-92 alone significantly improved survival to a median of 37 days (P<0.001). Treatment with BM4 did not enhance survival over control with both groups having a median survival of 32 days (P=0.619), but 7G3 significantly improved median survival to 35 days compared with the AML only control (P<0.001), but not the BM4 isotype control (P=0.1509). A combination of 7G3 and iCD16+NK-92 produced the best survival outcome, with a median survival of 42 days, which was significantly enhanced over mice infused with AML only and received no therapy (+10 days; P<0.001), 7G3 (+ 7 days; P<0.0025) and iCD16+NK-92 + BM4 (+ 10 days; P<0.0025) (Figure 7B).
Figure 7.
iCD16+NK-92 +/− 7G3 or isotype control treatment of primary acute myeloid leukemia (AML) xenografted mice. NOD/SCID gammanull (NSG) mice were inoculated intravenously (i.v.) with 3×106 passage human AML spleen-derived cells (day 0) and treated with iCD16+NK-92 +/− 7G3 or BM4 × 5 doses [intraperitoneal injection (i.p.)] (3×/week) starting on day 3 (A). Controls included no therapy and antibodies alone (n=5 for all groups). Survival was determined using Kaplan-Meier survival analysis with a log rank test (B).
Discussion
The overall long-term survival for patients with AML is approximately 40%,2 demonstrating a need for novel treatment strategies, particularly for patients in remission with detectable residual disease. We recently published a phase I clinical trial of NK-92 in relapsed and refractory hematologic malignancies (lymphoma and multiple myeloma) with minimal toxicities despite cumulative doses as high as 150 billion cells, and showed clinical responses.14 Prior studies implicating NK cells as therapeutically relevant in haplotype transplantation for AML20 prompted us to further investigate NK-92 as therapy for AML.
We have studied the mechanism of NK-92 cytotoxicity against primary AML samples and its efficacy in a primary human AML xenograft model. We confirmed initial reports that NK-92 mediates cytotoxicity in vitro against primary AML11 and demonstrated that killing was due primarily to granule exocytosis rather than ligand-mediated cytotoxicity (e.g. via Fas ligand) as shown by inhibiting cytotoxicity with the calcium chelator, EGTA.
We noted that classically defined, sorted CD34+CD38− LSCs4 were more sensitive to NK-92 killing than leukemia blast cells at low E:T ratios in a standard chromium release assay. Given the conflicting reports in the literature of the definitive immunophenotype of the LSC in AML,4,5,21,22 we opted to use a clonogenic assay to assess the effect of immune effector cells against LSCs in a larger set of samples. Primary AML grows well in methylcellulose and leads to the generation of visually detectable single cell-derived colonies that identifies the frequency of individual leukemic stem and progenitor cells. Specifically, we used a methylcellulose cytotoxicity assay (MCA) established previously by our lab19 that enables a comparison of the degree of killing of bulk leukemia blast cells versus colony inhibition in a 4-hour period. This approach provides another means to assay differential cytotoxicity against bulk and LSC populations. The MCA demonstrated a 2-3-fold higher % colony inhibition than the % lysis measured by the CRA. These results support our initial finding using cell sorted LSCs, which showed that NK-92 can preferentially recognize and kill LSCs over bulk leukemia.
Only two studies to date have looked at the in vitro sensitivity of CD34+CD38− LSCs to immune effector cell killing. In the first, lymphokine-activated killer (LAK) cells and allogeneic lymphocytes exerted a modest cytotoxic effect on AML LSCs comparable to the effect on the non-stem cell fraction.23 In a more recent study, endogenous single killer immunoglobulin-like receptor (KIR)-expressing NK cells, mismatched for the HLA of primary AML targets, showed equivalent killing of LSCs and blasts with either the chromium release or methylcellulose-based cytotoxicity assays.24 Here, we demonstrate preferential killing of LSCs versus bulk leukemia by NK-92, not shown by these other studies using a more rigorously controlled methylcellulose cytotoxicity assay than that used by Lankencamp et al. This is consistent with our work on NK-92 treatment of multiple myeloma (MM) cell lines showing that NK-92 preferentially kills clonogenic MM cells over bulk tumor cells.25
To pursue more in-depth studies of the effectiveness of NK-92 in killing LSCs, we developed an animal model of primary human AML by using NSG mice infused with a primary AML sample containing a small fraction of CD34+CD38− cells.
Secondary transplantation is the current gold standard to determine the effect of small molecules on LSCs26 but is rarely used to evaluate cellular therapies for leukemia. We attempted this assay by transplanting bone marrow from control and NK-92 treated mice into new recipients to assess the impact on individual mice rather than on pooled cells. BM engraftment occurred in all AML-only cohort secondary mice, while one mouse from the iNK-92 group was leukemia free, with engraftment at the background levels of non-injected mice. While the average BM engraftment of secondary transplant mice in the therapy groups was less than the control, this was not statistically significant. However, the LSC fraction was significantly decreased in secondary recipients, providing some evidence of NK-92 cytotoxicity against LSCs in the secondary transplant assay.
AML-xenografted NSG mice were effectively treated with NK-92 infusions, leading to improvement in survival versus controls, confirming previous work.11 We accomplished this with lower doses of NK-92 on a less compressed schedule than the original study, and without the use of IL-2 in the regimen. Irradiated NK-92 could prolong survival in mice, but was less effective than the non-irradiated cells. We postulate that the reason for this reduction in therapeutic efficacy is the lack of ability for the cells to proliferate in vivo.
We have demonstrated for the first time that irradiated NK-92 improves survival in an AML xenograft model, which has translational relevance given that only irradiated NK-92 is administered to patients in phase I trials. However, recognizing that the effect of iNK-92 on improving survival in vivo is modest, we wished to add the mechanism of ADCC to cell killing by using the genetically modified CD16+NK-92 in combination with a monoclonal antibody. CD16+NK-92 has been combined with rituximab to enhance killing of CD20+ malignant cells, showing its potential to enhance the killing of cells expressing a tumor-associated antigen.18 CD16+NK-92 cytotoxicity against OCI/AML5 was enhanced via ADCC when target cells were coated with a murine IgG2a anti-human CD123 mAb (7G3), indicating the ability to redirect CD16+NK-92 against a LSC-associated antigen. Cross-species interaction between murine Fc gamma receptors and human immune effectors cells has been reported, with murine IgG2a being the most effective isotype subclass in facilitating ADCC.27,28 However, the enhancement in CD16+NK-92 cytotoxicity seen here with 7G3 is likely an underestimate of the full potential of these cells to mediate ADCC, as the antibodies were murine rather than human in origin.
We then sought to combine iCD16+NK-92 and 7G3 therapy in our AML xenograft model and built upon prior work which demonstrated that systemic treatment with 7G3 alone in an AML NOD/SCID xenograft model reduced BM engraftment of human leukemia.29
We initially used a very small quantity of 7G3 in a single dose, preceded by blocking with a non-specific isotype-matched antibody to block Fc receptors, and improve circulation and binding of 7G3 to CD123, as established by Leyton et al.30 This approach worked in increasing the efficacy of the iCD16+NK-92 cells and improved survival. In a follow-up experiment, we did not use an Fc blocking pre-dose strategy, but gave 100 μg of 7G3 or isotype control antibody BM4 for five doses with or without the iCD16+NK-92 cells. In this experiment, iCD16+NK-92 alone prolonged survival over control. The BM4 antibody had no therapeutic effect and did not enhance iCD16+NK-92, while 7G3 alone had a modest survival benefit above control, which was not statistically significant above BM4 isotype control group. Of note, the best outcome was in the iCD16+NK-92 + 7G3 treated group which had a 10-day improvement in median survival compared with the iCD16+NK-92 + BM4 treatment group. Our data, therefore, demonstrate that the combination of iCD16+NK-92 and 7G3 can improve survival by antibody-dependent cell-mediated cytotoxicity. Furthermore, this represents the first demonstration of in vivo efficacy of the CD16+NK-92 cell line alone and in combination with antibody, which has only previously been tested in vitro.18 The Fc optimized anti-CD123 humanized monoclonal antibody CSL362 (derived from 7G3) can facilitate ADCC from peripheral blood-derived allogeneic NK cells against primary AML and CD123-expressing cell-line targets31 and is currently being tested in several clinical trials for AML. A recent study demonstrated potent CSL362-mediated NK cell ADCC against primary AML blasts and LSCs with comparable results seen with NK cells from AML patients and healthy donors.32 CD16+NK-92 cells which express the high affinity CD16 receptors could be combined with CSL362 as a potentially potent treatment of AML.
Our strategy of targeting LSCs by anti-CD123-facilitated ADCC in vivo had a comparable improvement in median survival to CD123 CAR T-cell therapy in an AML NSG xenograft model.33 The latter, however, used the KG1a line and not primary AML cells, as in our case. Using a different CAR vector, another group tested CD123 CAR T cells in a primary AML model with no improvement in median survival, but had approximately 40% long-term survivors at day 100 (approx. 70 days after control median survival), which was statistically significant.34 One other study of CD123 CAR T cells demonstrated efficacy against LSCs using primary and secondary engraftment models, but did not have a survival end point.35 Another group used CD123 CAR-transduced cytokine-induced killer (CIK) cells, and showed in vitro efficacy against a CD123+ cell line and primary AML targets.36 A limitation of CD123 CAR T-cell therapy is that it can target HSCs with low CD123 expression, making it a potentially myeloablative therapy, suitable only in conjunction with stem cell transplantation.34 In contrast, a clinical study of antibody based targeting of CD123 using CSL360 did not exhibit myeloablation, despite having the potential for NK-mediated ADCC against LSCs with low expression of CD123.37 Therefore, we anticipate that our approach utilizing irradiated CD16+NK-92 in combination with anti-CD123 antibodies may have a reduced risk of causing life-threatening myelosuppression, but this remains to be shown in clinical studies of both these approaches.
In summary, we have shown that NK-92 preferentially targets LSCs over bulk leukemia blasts in vitro and irradiated NK-92 can improve survival in an AML xenograft model, which can be enhanced using CD16+NK-92 combined with an anti-CD123 monoclonal antibody. This provides the first proof-of-principle for the targeting of LSCs by combining an antibody and a standardized cellular therapy. A humanized version of 7G3 (CLS362) is being tested in clinical trials for AML, but is reliant upon a patient’s endogenous NK-cell function for efficacy. Combination therapy with ADCC capable NK cell lines such as CD16+NK-92 or the new haNK platform (CD16+IL-2+NK-92) with CSL362 would enable the therapeutic translation of our approach into a clinical trial in the future. haNK cells are currently in clinical trials for solid tumors in combination with FDA-approved monoclonal antibodies. The approach we have demonstrated can be readily applied to enhance the targeting of any antigen, and its particular novelty here is in demonstrating the targeting of a cancer stem cell marker and a consequent improvement in survival in a murine AML xenograft model.
Supplementary Material
Footnotes
Check the online version for the most updated information on this article, online supplements, and information on authorship & disclosures: www.haematologica.org/content/103/10/1720
Funding
BW was supported by a Terry Fox Foundation Award from the National Cancer Institute of Canada, CIHR clinician scientist training award, as well as grants from the Hospital for Sick Children, Ontario Cancer Institute and University of Toronto. AK was supported by the Gloria and Seymour Epstein Chair in Cell Therapy and Transplantation at the University Health Network and University of Toronto. We would like to acknowledge the generous provision of 7G3 for some experiments by Angel Lopez.
References
- 1.Hurwitz CA, Mounce KG, Grier HE. Treatment of patients with acute myelogenous leukemia: review of clinical trials of the past decade. J Pediatr Hematol Oncol. 1995;17(3):185–197. [DOI] [PubMed] [Google Scholar]
- 2.Lowenberg B, Downing JR, Burnett A. Acute myeloid leukemia. N Engl J Med. 1999;341(14):1051–1062. [DOI] [PubMed] [Google Scholar]
- 3.Ribeiro RC, Razzouk BI, Pounds S, Hijiya N, Pui CH, Rubnitz JE. Successive clinical trials for childhood acute myeloid leukemia at St Jude Children’s Research Hospital, from 1980 to 2000. Leukemia. 2005; 19(12):2125–2129. [DOI] [PubMed] [Google Scholar]
- 4.Lapidot T, Sirard C, Vormoor J, et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367(6464):645–648. [DOI] [PubMed] [Google Scholar]
- 5.Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997;3(7):730–737. [DOI] [PubMed] [Google Scholar]
- 6.Jordan CT, Upchurch D, Szilvassy SJ, et al. The interleukin-3 receptor alpha chain is a unique marker for human acute myelogenous leukemia stem cells. Leukemia. 2000; 14(10):1777–1784. [DOI] [PubMed] [Google Scholar]
- 7.Vergez F, Green AS, Tamburini J, et al. High levels of CD34+CD38low/-CD123+ blasts are predictive of an adverse outcome in acute myeloid leukemia: a Groupe Ouest-Est des Leucemies Aigues et Maladies du Sang (GOELAMS) study. Haematologica. 2011;96(12):1792–1798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Miller JS, Soignier Y, Panoskaltsis-Mortari A, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood. 2005;105(8):3051–3057. [DOI] [PubMed] [Google Scholar]
- 9.Rubnitz JE, Inaba H, Ribeiro RC, et al. NKAML: a pilot study to determine the safety and feasibility of haploidentical natural killer cell transplantation in childhood acute myeloid leukemia. J Clin Oncol. 2010;28(6):955–959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gong JH, Maki G, Klingemann HG. Characterization of a human cell line (NK-92) with phenotypical and functional characteristics of activated natural killer cells. Leukemia. 1994;8(4):652. [PubMed] [Google Scholar]
- 11.Yan Y, Steinherz P, Klingemann HG, et al. Antileukemia activity of a natural killer cell line against human leukemias. Clin Cancer Res. 1998;4(11):2859–2868. [PubMed] [Google Scholar]
- 12.Arai S, Meagher R, Swearingen M, et al. Infusion of the allogeneic cell line NK-92 in patients with advanced renal cell cancer or melanoma: a phase I trial. Cytotherapy. 2008;10(6):625–632. [DOI] [PubMed] [Google Scholar]
- 13.Tonn T, Schwabe D, Klingemann HG, et al. Treatment of patients with advanced cancer with the natural killer cell line NK-92. Cytotherapy. 2013;15(12):1563–1570. [DOI] [PubMed] [Google Scholar]
- 14.Williams BA, Law AD, Routy B, et al. A phase I trial of NK-92 cells for refractory hematological malignancies relapsing after autologous hematopoietic cell transplantation shows safety and evidence of efficacy. Oncotarget. 2017;8(51):89256–89268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Klingemann H, Wong E, Maki G. A cytotoxic NK-cell line (NK-92) for ex vivo purging of leukemia from blood. Bone Marrow Transplant. 1996;2(2):68–75. [PubMed] [Google Scholar]
- 16.Tonn T, Becker S, Esser R, Schwabe D, Seifried E. Cellular immunotherapy of malignancies using the clonal natural killer cell line NK-92. J Hematother Stem Cell Res. 2001;10(4):535–544. [DOI] [PubMed] [Google Scholar]
- 17.Tam YK, Miyagawa B, Ho VC, Klingemann HG. Immunotherapy of malignant melanoma in a SCID mouse model using the highly cytotoxic natural killer cell line NK-92. J Hematother. 1999;8(3):281–290. [DOI] [PubMed] [Google Scholar]
- 18.Binyamin L, Alpaugh RK, Hughes TL, Lutz CT, Campbell KS, Weiner LM. Blocking NK cell inhibitory self-recognition promotes antibody-dependent cellular cytotoxicity in a model of anti-lymphoma therapy. J Immunol. 2008;180(9):6392–6401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Williams BA, Wang XH, Keating A. Clonogenic assays measure leukemia stem cell killing not detectable by chromium release and flow cytometric cytotoxicity assays. Cytotherapy. 2010;12(7):951–960. [DOI] [PubMed] [Google Scholar]
- 20.Ruggeri L, Capanni M, Urbani E, et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science. 2002;295(5562):2097–2100. [DOI] [PubMed] [Google Scholar]
- 21.Taussig DC, Miraki-Moud F, Anjos-Afonso F, et al. Anti-CD38 antibody-mediated clearance of human repopulating cells masks the heterogeneity of leukemia-initiating cells. Blood. 2008;112(3):568–575. [DOI] [PubMed] [Google Scholar]
- 22.Goardon N, Marchi E, Atzberger A, et al. Coexistence of LMPP-like and GMP-like leukemia stem cells in acute myeloid leukemia. Cancer Cell. 2011;19(1):138–152. [DOI] [PubMed] [Google Scholar]
- 23.Costello RT, Mallet F, Gaugler B, et al. Human acute myeloid leukemia CD34+/CD38− progenitor cells have decreased sensitivity to chemotherapy and Fas-induced apoptosis, reduced immunogenicity, and impaired dendritic cell transformation capacities. Cancer Res. 2000; 60(16):4403–4411. [PubMed] [Google Scholar]
- 24.Langenkamp U, Siegler U, Jorger S, et al. Human acute myeloid leukemia CD34+CD38− stem cells are susceptible to allorecognition and lysis by single KIR-expressing natural killer cells. Haematologica. 2009;94(11):1590–1594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Swift BE, Williams BA, Kosaka Y, et al. Natural killer cell lines preferentially kill clonogenic multiple myeloma cells and decrease myeloma engraftment in a bioluminescent xenograft mouse model. Haematologica. 2012;97(7):1020–1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Skrtic M, Sriskanthadevan S, Jhas B, et al. Inhibition of mitochondrial translation as a therapeutic strategy for human acute myeloid leukemia. Cancer Cell. 2011; 20(5):674–688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kipps TJ, Parham P, Punt J, Herzenberg LA. Importance of immunoglobulin isotype in human antibody-dependent, cell-mediated cytotoxicity directed by murine monoclonal antibodies. J Exp Med. 1985;161(1):1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Biddle WC, Pancook J, Goldrosen M, Han T, Foon KA, Vaickus L. Antibody-dependent, cell-mediated cytotoxicity by an anti-class II murine monoclonal antibody: effects of recombinant interleukin 2 on human effector cell lysis of human B-cell tumors. Cancer Res. 1990;50(10):2991–2996. [PubMed] [Google Scholar]
- 29.Jin L, Lee EM, Ramshaw HS, et al. Monoclonal antibody-mediated targeting of CD123, IL-3 receptor alpha chain, eliminates human acute myeloid leukemic stem cells. Cell Stem Cell. 2009;5(1):31–42. [DOI] [PubMed] [Google Scholar]
- 30.Leyton JV, Hu M, Gao C, et al. Auger electron radioimmunotherapeutic agent specific for the CD123+/CD131− phenotype of the leukemia stem cell population. J Nucl Med. 2011;52(9):1465–1473. [DOI] [PubMed] [Google Scholar]
- 31.Busfield SJ, Biondo M, Wong M, et al. Targeting of acute myeloid leukemia in vitro and in vivo with an anti-CD123 mAb engineered for optimal ADCC. Leukemia. 2014;28(11):2213–2221. [DOI] [PubMed] [Google Scholar]
- 32.Xie LH, Biondo M, Busfield SJ, et al. CD123 target validation and preclinical evaluation of ADCC activity of anti-CD123 antibody CSL362 in combination with NKs from AML patients in remission. Blood Cancer J. 2017;7(6):e567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mardiros A, Dos Santos C, McDonald T, et al. T cells expressing CD123-specific chimeric antigen receptors exhibit specific cytolytic effector functions and antitumor effects against human acute myeloid leukemia. Blood. 2013;122(18):3138–3148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gill S, Tasian SK, Ruella M, et al. Preclinical targeting of human acute myeloid leukemia and myeloablation using chimeric antigen receptor-modified T cells. Blood. 2014; 123(15):2343–2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pizzitola I, Anjos-Afonso F, Rouault-Pierre K, et al. Chimeric antigen receptors against CD33/CD123 antigens efficiently target primary acute myeloid leukemia cells in vivo. Leukemia. 2014;28(8):1596–1605. [DOI] [PubMed] [Google Scholar]
- 36.Tettamanti S, Marin V, Pizzitola I, et al. Targeting of acute myeloid leukaemia by cytokine-induced killer cells redirected with a novel CD123-specific chimeric antigen receptor. Br J Haematol. 2013; 161(3):389–401. [DOI] [PubMed] [Google Scholar]
- 37.He SZ, Busfield S, Ritchie DS, et al. A Phase 1 study of the safety, pharmacokinetics and anti-leukemic activity of the anti-CD123 monoclonal antibody CSL360 in relapsed, refractory or high-risk acute myeloid leukemia. Leuk Lymphoma. 2015; 56(5):1406–1415. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.