Abstract
Purpose of review
This review is to summarize the recent progress of vitamin D/VDR and microbiome in intestinal homeostasis, airway function, and other organs.
Recent findings
Microbiome is considered as a newly discovered human organ. It is critical in the synthesis of vitamins and harvest of otherwise inaccessible nutrients, metabolism of xenobiotics, body fat storage, renewal of gut epithelial cells, and mature of immune system. Vitamin D and its receptor vitamin D receptor (VDR) are known to regulate microbiome in health and disease. We will focus on the recent findings published in 12–18 months and discuss the vitamin D supplement and its effects on microbiome, intestinal homeostasis, airway function, and metabolism. We will emphasize the tissue specificity and genetic factor of VDR and microbiome.
Summary
The findings in dietary Vitamin D, VDR, and microbiome with personalized genetic information will be implicated for optimal prevention and treatment of chronic diseases.
Keywords: Vitamin D, Vitamin D Receptor, inflammation, dysbiosis, metabolism, Non-alcoholic fatty liver disease (NAFLD), lung microbiome
Introductions
1,25-Dihydroxyvitamin D (1,25(OH)2D3), the active form of vitamin D, activates the widely expressed vitamin D receptor (VDR). VDR is a nuclear hormone receptor and transcription factor expressed in a variety of tissues, including the intestines, adipose tissue, and liver, as well as most immune and modulates metabolic and immune system processes. 1,25(OH)2D3 is generated in the skin after exposure to UV light or absorbed from a diet of vitamin D-rich foods. Low levels of vitamin D or inactivating polymorphisms in VDR have been associated with inflammatory and metabolic disorders. Classically, vitamin D is known to regulates bone development and calcium homeostasis through its actions in the intestine, kidney, and bone. Vitamin D regulates calcium absorption in the intestine and resorption in the kidney by activating transcellular calcium transport. Vitamin D deficiency causes rickets, characterized by hypoparathyroidism and impaired bone mineralization.
Gut microbiome is considered as a “organ” because it plays critical role in the synthesis of vitamins and harvest of otherwise inaccessible nutrients, metabolism of xenobiotics, renewal of epithelial cells, fat storage, and immunity. The variation of human vdr gene shapes the gut microbiome at the genetic level [1]. Intestinal epithelial VDR conditional knockout (VDRΔIEC) leads to dysbiosis [2]. Gut bacterial abundance is significantly changed in VDRΔIEC mice: decreased genus Lactobacillus and butyrate-producing bacteria. However, the study on biological function of vitamin D and VDR in microbiome is still limited. The purpose of this review is to summarize the recent progress of vitamin D/VDR in microbiome, intestinal function, airway function, and other organs. We will discuss the newly discovered roles of vitamin D/VDR in a tissue specific manner.
Vitamin D supplement regulates gut microbiome and airway microbiome
Diet and nutrition could be a trigger for chronic inflammation, such as inflammatory bowel diseases [3]. Vitamin D deficiency contributes to the pathogenesis of Crohn’s disease (CD), and is supported by the results of intervention studies. A recent study has shown that vitamin D3 supplementationleads to a shift of the intestinal bacterial composition in CD patients, but not in healthy controls[4]. Oral vitamin D3 administration was given with 20,000 IU daily from day 1 until day 3, then every other day for a total of 4 weeks. In this study, a target vitamin D level was set to be between 100 and 150 nmol/L. Serum 25-hydro-xyvitamin D (25[OH]D) levels were measured weekly. Specific species with a high abundancy were found in CD patients during vitamin D administration, but not in healthy controls; the abundancy of Alistipes, Barnesiella, unclassified Porphyromonadaceae (both Actinobacteria), Roseburia, Anaerotruncus, Subdoligranulum and an unclassified Ruminococaceae (all Firmicutes) increased significantly in CD after 1 week vitamin D3 administration.
Vitamin D supplementation changes not only gut microbiome, but also airway microbiome. A double-blind, randomized, placebo-controlled clinical trial has demonstrated that bolus weekly vitamin D3 supplementation has impacts on both intestinal and airway microbiota in patients with cystic fibrosis (CF) [5]. In this study, forty-one CF subjects were classified into two groups: vitamin D insufficient (25-hydroxyvitamin D < 30 ng/mL, n = 23,) and vitamin D sufficient (n = 18). Subjects with vitamin D insufficiency were randomized to receive 50,000 IU of oral vitamin D3 or placebo weekly for 12 weeks. Vitamin D deficiency is a common comorbidity in patients with cystic fibrosis that may influence composition of the gut microbiota. Vitamin D deficiency is associated with dysbiosis that promotes inflammation. Disruption of gut microbiota may exacerbate severity of cystic fibrosis. Thus, vitamin D supplementation has the potential to impact microbiota [5]. As we know, the vitamin D works in a very tissue specific way and has gender difference. It is reported that vitamin D and allergic airway disease shape the murine lung microbiome in a sex-specific manner [6]. However, there is limited research on lung microbiome and vitamin D. We could only speculate how the lung microbiome and intestinal microbiome modulate the functions in the airway.
Nutrition including vitamins plays a role in mental diseases, such as depression [7]. However, antidepressive effect of vitamin D supplementation is currently inconclusive. It is not known whether vitamin D plays a role in the gut-brain axis.
Vdr gene and microbiome
It is always interesting to learn whether intestinal microbiota composition is determined by genetic factors or environment. There is increasing support for a host genetic component shaping and/or structuring between-individual variability in the microbiota. Genome-wide association analysis identifies variation in human Vdr gene and other host factors influencing the gut microbiota [1]. Comprehensively controlling for diet and non-genetic parameters, this study has identified genome-wide significant associations for overall microbial variation and individual taxa at multiple genetic loci, including the Vdr gene. There are significant shifts in the microbiota of Vdr−/− mice relative to control mice and correlations between the microbiota and serum bile and fatty acids in humans. Interestingly, tissue enrichment analysis relates microbial-associated host loci with gastrointestinal and immune-related tissues and cells, thus supporting the functional relevance of the identified loci. Understanding the functional consequences of the genetic variants will require in-depth exploration.
We have reported probiotics in modulating vitamin D/VDR and balancing gut microbiota in health and gastrointestinal diseases [8]. Probiotic LGG treatment did not show a protective role in the VDR deficient mice with Salmonella-colitis, whereas LGG inhibited Salmonella-induced inflammation and injury in the wild-type mice. We thus speculate that the individual response to probiotics may depend on vdr gene and microbiome.
Vitamin D and infant microbiome
The gut microbiome in infancy influences immune system maturation and have an important impact on allergic disease risk and adulthood diseases. In the 913 one month old infants KOALA birth cohort study, researchers found that vitamin D has an influence on key bacterial taxa in infant microbiota [9]. There was a statistically significant negative linear trend between counts of Bifidobacterium spp. and levels of maternal vitamin D supplementation and maternal 25-hydroxyvitamin D quintiles, respectively. A positive linear trend between quintile groups and B. fragilis group counts was observed. Lower counts of C. difficile were associated with vitamin D supplementation of breast fed infants whose mothers were more likely to adhere to an alternative lifestyle in terms of, e.g., dietary habits [9]. There was no influence of vitamin D supplementation of the infant during the first month of life on the prevalence of any of the bacterial species or groups. These data suggest that vitamin D influences the abundance of several key bacterial taxa in the infant microbiota.
In a recent study of infant gut microbiome at age 3–6 months, the researchers find that cord blood vitamin D was linked to increased Lachnobacterium, but decreased Lactococcus [10]. Given microbiotic homeostasis as an important factor in the prevention of immune mediated diseases and that vitamin D status is a modifiable factor, postnatal vitamin D supplementation should be conducted in older infants.
Vitamin D receptor in gut–liver axis
Vitamin D/VDR signaling is critical in intestinal barrier function, innate and adaptive immunity, and homeostasis [11, 12]. A recent study has reported that a Single Nucleotide Polymorphism (SNP) rs731236 in the Vdr gene is associated with decreased levels of VDR protein in patients with Crohn’s Disease[13]. Pro-inflammatory cytokines increased in PBMCs from CD patients homozygous for the C Allele of Vdr gene. In addition, these patients run a higher risk of developing a B3-penetrating phenotype and requiring surgery. This study provides new evidence for the role of Vdr in the disease course. However, the role of microbiome related to the rs731236 in the Vdr gene has not been explored in patients with CD.
Vitamin D deficiency has been associated with non-alcoholic fatty liver disease (NAFLD). In UK children with NAFLD, only 19.2% of children had adequate vitamin D status; most had mean 25OHD levels considered deficient (<25 nmol.L(−1) , 25.5%) or insufficient (<50 nmol.L(−1) , 55.3%). Polymorphisms in the nicotinamide adenine dinucleotide synthase-1/dehydrocholesterol reductase-7 (rs3829251, rs12785878) and vitamin D receptor (rs2228570) genes were independently associated with increased steatosis; while a group-specific component variant (rs4588) was associated with increased inflammation in liver biopsies. Polymorphisms in the vitamin D metabolic pathway are associated with histological severity of pediatric NAFLD[14]. Children with NAFLD had significantly higher amounts of bacteria Proteobacteria, Enterobacteriaceae, and ethanol-producing Escherichia than healthy children [15]. Further studies of VDR at the genetic and functional levels and its regulation of microbiome in gut-liver axis will provide novel mechanistic insights and therapeutic opportunities.
Conclusion:
Environmental factors (e.g. reduced ultraviolet B irradiation and pollution), and changes of lifestyle factors (e.g. decreased outdoor activities and/or poor intake of vitamin D-rich food) are involved in the etiology of vitamin D deficiency and risks of various human diseases. Hundreds of genes are directly or indirectly associated with vitamin D and Vdr. Genetic and genomic studies of vdr gene and microbiome will provide novel insights into the role of vitamin D/VDR in health and disease. The emerging evidence of an important role of vit D and gut microbiota is important. A number of disease conditions seem very “vitamin D responsive”. We still do not know whether a common pathology (e.g., inflammation) or link of theses diseases. Fig. 1 summarizes the current state of understanding of Vitamin D/VDR and microbiome in intestine and other putative organs. Vitamin D supplement has a positive effect in IBD patients and cystic fibrosis patients through regulating microbiome. Further investigation of infant microbiome and vitamin D/VDR will help to reduce the risk of allergy and other diseases in adulthood.
Figure 1.
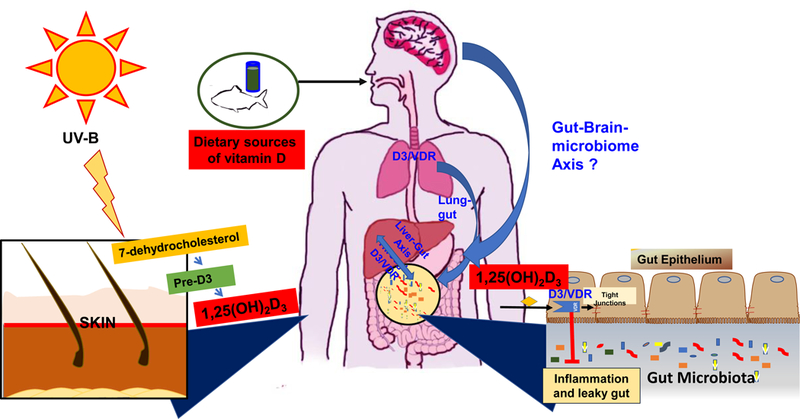
Vitamin D/VDR and microbiome in intestine and other putative organs. There are two main sources to get vitamin D: exposed to sunlight and foods. Vitamin D and its receptor VDR regulate gut microbiome, maintaining barrier functions, and inhibit inflammation in intestine. Bidirecitional host-microbiome interactions seem related to vitamin D/VDR in intestine and also impact lung, liver, and other organs.
Key points:
Vitamin D supplement has a positive effect in IBD patients by modulating gut microbiome and also by increasing the abundance of potential beneficial bacterial strains.
Vitamin D supplement plays novel roles in regulating lung microbiome and airway function.
Progress is made in studying infant microbiome and vitamin D.
The vitamin D/VDR study should be put in the context of tissue specificity.
an interesting insight into Vitamin D regulation of microbiome in IBD patients
of outstanding interest in vitamin D regulation of both gut and airway microbiome.
of special interest in the role of vitamin D in a sex-specific manner.
of special interest in infant microbiome and vitamin D
of special interest in infant microbiome and vitamin D
Acknowledgement
We acknowledge Dr. Ishita Chatterjee for her help with images in Figure 1.
Financial support and sponsorship
This work was supported by the NIDDK 1R01DK105118-01, R01DK114126, and DOD BC160450P1, and UIC Cancer Center (Jun Sun)
Footnotes
Conflicts of interest
None
Recommended reading
Papers of particular interest, published within the annual period of review (18 months/ 2016-2018) have been highlighted as: • of special interest •• of outstanding interest
References
- 1.Wang J, et al. , Genome-wide association analysis identifies variation in vitamin D receptor and other host factors influencing the gut microbiota. Nat Genet, 2016. 48(11): p. 1396–1406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu S, et al. , Intestinal epithelial vitamin D receptor deletion leads to defective autophagy in colitis. Gut, 2015. 64(7): p. 1082–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lewis JD and Abreu MT, Diet as a Trigger or Therapy for Inflammatory Bowel Diseases. Gastroenterology, 2017. 152(2): p. 398–414 e6. [DOI] [PubMed] [Google Scholar]
- 4.Schaffler H, et al. , Vitamin D administration leads to a shift of the intestinal bacterial composition in Crohn’s disease patients, but not in healthy controls. J Dig Dis, 2018. 19(4): p. 225–234. [DOI] [PubMed] [Google Scholar]
- 5.Kanhere M, et al. , Bolus Weekly Vitamin D3 Supplementation Impacts Gut and Airway Microbiota in Adults With Cystic Fibrosis: A Double-Blind, Randomized, Placebo-Controlled Clinical Trial. J Clin Endocrinol Metab, 2018. 103(2): p. 564–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Roggenbuck M, et al. , Vitamin D and allergic airway disease shape the murine lung microbiome in a sex-specific manner. Respir Res, 2016. 17(1): p. 116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Libuda L, et al. , [Nutrition and mental diseases : Focus depressive disorders]. Nervenarzt, 2017. 88(1): p. 87–101. [DOI] [PubMed] [Google Scholar]
- 8.Shang M and Sun J, Vitamin D/VDR, Probiotics, and Gastrointestinal Diseases. Curr Med Chem, 2017. 24(9): p. 876–887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Talsness CE, et al. , Influence of vitamin D on key bacterial taxa in infant microbiota in the KOALA Birth Cohort Study. PLoS One, 2017. 12(11): p. e0188011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sordillo JE, et al. , Factors influencing the infant gut microbiome at age 3–6 months: Findings from the ethnically diverse Vitamin D Antenatal Asthma Reduction Trial (VDAART). J Allergy Clin Immunol, 2017. 139(2): p. 482–491 e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dimitrov V and White JH, Vitamin D signaling in intestinal innate immunity and homeostasis. Mol Cell Endocrinol, 2017. 453: p. 68–78. [DOI] [PubMed] [Google Scholar]
- 12.Bakke D and Sun J, Ancient Nuclear Receptor VDR With New Functions: Microbiome and Inflammation. Inflamm Bowel Dis, 2018. 24(6): p. 1149–1154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gisbert-Ferrandiz L, et al. , A Single Nucleotide Polymorphism in the Vitamin D Receptor Gene Is Associated With Decreased Levels of the Protein and a Penetrating Pattern in Crohn’s Disease. Inflamm Bowel Dis, 2018. [DOI] [PubMed]
- 14.Gibson PS, et al. , Vitamin D status and associated genetic polymorphisms in a cohort of UK children with non-alcoholic fatty liver disease. Pediatr Obes, 2018. [DOI] [PMC free article] [PubMed]
- 15.Zhu L, et al. , Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology, 2013. 57(2): p. 601–9. [DOI] [PubMed] [Google Scholar]


