Abstract
Introduction
A number of plant species, including Cymbopogon schoenanthus, are traditionally used for the treatment of various diseases. C. schoenanthus is currently, traded in the Saudi markets, and thought to have medicinal value. This study aimed at investigating the biological activities of C. schoenanthus against both Gram-positive and Gram-negative bacteria and to identify its chemical ingredients.
Materials and methods
The inhibitory effects of water extracts of C. schoenanthus essential oils were evaluated against ten isolates of both Gram-positive and Gram-negative bacteria using the agar well diffusion and dilution methods. The minimum inhibitory concentration (MIC) was assayed using the Broth microdilution test on five of the ten isolates. The death rates were determined by the time kill assay, done according to the Clinical Laboratory Standards Institute (CLSI) guidelines. The chemical composition of the essential oils of the plant was performed using GC/MS.
Results
The C. schoenanthus essential oil was effective against Escherichia coli, Staphylococcus aureus, methicillin-sensitive (MSSA) S. aureus (MRSA) and Klebsiella pneumoniae. The essential oil was not effective against Staphylococcus saprophyticus at the highest concentration applied of >150 μg/ml. The MIC values were as follows: 9.37 μg/ml for E. coli 4.69 μg/ml for S. aureus (MRSA), 2.34 mg/ml for MSSA and 2.34 μg/ml for K. pneumoniae. The time-kill assay indicated that there was a sharp time dependent decline in K. pneumoniae counts in the presence of the oil. This is in contrast to a gradual decline in the case of S. aureus under the same conditions. The eight major components of the essential oil were: piperitone (14.6%), cyclohexanemethanol (11.6%), β-elemene (11.6%), α-eudesmol (11.5%), elemol (10.8%), β-eudesmol (8.5%), 2-naphthalenemethanol (7.1%) and γ-eudesmol (4.2%).
Conclusion
The results of the present study provide a scientific validation for the traditional use of C. schoenanthus as an antibacterial agent. Future work is needed to investigate and explore its application in the environmental and medical fields. In addition, to evaluating the efficacy of the individual ingredients separately to better understand the underlying mechanism.
Keywords: Cymbopogon schoenanthus, Essential oils, Gram-positive, Bacteria, Gram-negative bacteria
1. Introduction
Medicinal plants have been widely used in traditional medicine for several centuries for the treatment of many health-related ailments. According to the World Health Organization (WHO), the majority of the world’s population depends on traditional medicine for primary healthcare. There has been an increasing interest in medicinal plants and their active ingredients because of their potency and negligible adverse side effects. In Saudi Arabia, medicinal plants account for more than over 50% of all plants spices (1200 out of 2250) (Mossa et al., 1987). Despite the indigenous knowledge of the healing ability of certain plants in Saudi Arabia, few plant extracts and essential oils have been assessed in vitro or in vivo for their therapeutic potentials (Al Yahya et al., 1983). Recent published data on medicinal plants worldwide revealed that some exhibited: antioxidant (Narendran et al., 2016, Noorudheen and Chandrasekharan, 2016, Puthur, 2016, Santhosh et al., 2016), anti-diabetic and attenuation of insulin resistance (Kannan and Agastian, 2015, Balamurugan, 2015), anti-diarrheal activities (Antonisamy et al., 2015), cardio and hepatic protective ability (Nandhini and Bai, 2015, Rathi et al., 2015).
One important medicinal plant, Cymbopogon schoenanthus, locally known as Sakhbar, Izkhir or Athkhar traditionally named as camel grass, is a desert species that grows in dry stony places (Al-Ghamdi et al., 2007, Farooqi, 1998). It was mentioned in Alhadith for its potential applications (Marwat et al., 2009). Its oil has a strong aromatic odor and has great medicinal value. The plant is well known traditionally and is widely used as: antispasmodic, a protection against fever, anti-intestinal ailment problems, anti-malarial, and anti-helminthic (especially against Guinea worms) (Yentéma et al., 2007, Marwat et al., 2009). It is an effective renal antispasmodic and diuretic agent (El-Askary et al., 2003, Elhardallou, 2011, Sabry et al., 2014), and it was shown to possess sedative, digestive and anti-parasitic properties (Sousa et al., 2005). Norbert et al. (2014) demonstrated that it is an antifungal and anti-inflammatory agent used for the prevention and treatment of acute inflammatory skin conditions. The vapor phase is more effective as an antifungal agent as compared to the liquid phase and may be used for the decontamination of air in hospitals. It has also been used as an anti-abortive, anti-convulsive or laxative agent, aroma and anti-rheumatic, asthmatic, and antipyretic agent (Ketoh et al., 2006). Furthermore, C. schoenanthus is used in the treatment of colds, epilepsy, abdominal cramps and pains, as well as in culinary and perfume products (Takaisi-Kikuni et al., 2000). In Saudi traditional medicine, it is mainly used as a diuretic to inhibit kidney stone formation and as an anti-infectious agent in urinary tract infections (Al-Ghamdi et al., 2007).
The aim of this study is to evaluate the antimicrobial activity of the essential oil of C. schoenanthus against susceptible and resistant pathogenic bacteria in order to validate some of its traditionally claimed therapeutic properties.
2. Materials and methods
2.1. Plant collection and extraction
C. schoenanthus was collected from Asfan area, north-east of Jeddah, Saudi Arabia. The plants were washed, dried in the shade, crushed into small pieces, then were subjected to distillation using conventional methods. Water was added to completely cover clean dried crushed plants that had been compressed into a boiling chamber. The mixture was then allowed to simmer and gently brought to boil. Ice cold water was continuously circulated to the condenser to facilitate the condensation process of the generated steam. The process lasted for 48 h. The concentration of the stock solution was determined by dividing the weight of the plant parts used over the volume of the resulting distill. Stock solutions were suspended in Tween 80 to preserve the activity of the oil, divided into small aliquots and stored at −80 °C till the day of the experiment. One aliquot was thawed on ice and used on the day of the experiment and was discarded soon after the completion of the experiment. Tween 80 was added to the control at the same concentration as that in the stock containing the extract to rule out the effect of Tween 80 (Lahlou, 2004).
2.2. Antimicrobial susceptibility testing
The antimicrobial activity of C. schoenanthus essential oil was evaluated using three tests: (i) Agar well-diffusion test, (ii) Broth microdilution test, and (iii) time-kill assay test.
2.2.1. Agar well-diffusion test
2.2.1.1. Preparing the agar plates
Mueller–Hinton agar was used (Oxoid Limited Wade Road Basingstoke Hants, England), and prepared according to the manufacturer’s instructions. Post autoclaving, the agar was allowed to cool down (45–50 °C) in a water bath. Then, the agar was dispensed into Petri dishes, stored in the refrigerator and used within five days.
2.2.1.2. Bacterial cultures
Ten bacterial pathogens were used and purchased from the American Type Culture Collection, ATCC, Virginia, USA. The pathogens included Gram-positive bacteria Staphylococcus aureus, methicillin sensitive (MSSA) (ATCC 6538), methicillin resistant S. aureus (MRSA) (ATCC 33591), Staphylococcus saprophyticus (ATCC 35552), Enterococcus faecalis (VRE) (ATCC 51299) and Enterococcus faecium (ATCC 6569) and Gram-negative bacteria: Escherichia coli (ATCC 11229), Klebsiella pneumoniae (ATCC 4352), Proteus mirabilis (ATCC 7002), Pseudomonas aeruginosa (ATCC 15442) and Serratia marcescens (ATCC 14756),
2.2.1.3. Inoculum preparation
The inoculums were prepared using the direct colony suspension method according to CLSI guidelines. Colonies were fished from a fresh (18–24 h) Tryptic Soy Agar (TSA) (Oxoid Limited, Wade Road Basingstoke Hants, England) plate and inoculated in Tryptic Soy Broth (TSB) (Oxoid Limited, Wade Road Basingstoke Hants, England). The suspensions were mixed by vortexing then the turbidity was adjusted with sterile Tryptic Soy Broth (TSB) to reach a 0.5 McFarland.
2.2.1.4. Inoculation of agar plates
Within 15 min of adjusting turbidity, for each bacterial suspension, a 0.1 ml of bacterial suspension was dispensed and evenly spread over plates containing Mueller–Hinton agar using a glass spreader. The plates were allowed to stand for no longer than 15 min before drilling wells in them using a sterile 8 mm cork borer. Then, 0.1 ml of the essential oil was added into each well. The plates were covered immediately and incubated. A 5 μg/ml oxacillin antibiotic disk (BD biosciences, USA) was also placed on the surface as a reference. The plates were then incubated at 36 ± 1 °C for approximately 18 h in an ambient-air incubator. The zones of inhibition were measured.
2.2.2. Broth microdilution method for MIC test
The MIC test was performed according to the CLSI guidelines (CLSI, 2013), with some modifications. The inoculum was prepared using the direct colony suspension method as indicated earlier. Two milliliters of the prepared bacterial suspension were added to 40 ml of Broth to reach a dilution of 1:20 and a final concentration of approximately 5 × 105 CFU/ml. Within one hour of preparing the bacterial suspension, and after gently mixing by inverting five to six times, the microtiter plate was inoculated. Serial 1:2 dilutions of the oil were performed in the microtiter wells with Mueller–Hinton Broth and subsequently inoculated with the appropriate bacteria. The last well in each row was left blank as negative controls. The test was performed in triplicates. The microtiter plate was covered then incubated at 36 ± 1 °C for 24 h in an ambient-air incubator.
2.2.3. Time-kill assay test
To study the kinetics of inactivation of bacteria by the extract, the time-kill assay was done according to the CLSI guidelines, with some specific modifications. The inoculum was prepared using the direct colony suspension method according to the CLSI guidelines. Several colonies of similar morphology were fished from a fresh (18–24 h) TSA plate and inoculated in TSB. The suspensions were mixed by vortexing, then turbidity was adjusted visually with sterile TSB to reach that of a 0.5 McFarland standard. 0.1 ml of the standardized suspension was transferred to 5 ml of the Broth. In performing the assay, 1 ml of a dilution of the oil, (concentrations determined by MIC test), S. aureus and K. pneumoniae were treated for specified periods of time (0, 2, 4, 6, 8, 10, 12 and 24 h) at room temperature. Phosphate buffered solution was used instead of oil in the case of controls. The activity of the oil was immediately stopped at specified sampling intervals (0, 2, 4, 6, 8, 10, 12 and 24 h) by placing 0.1 ml of the test solution into 0.9 ml of Broth. Colonies of surviving microorganisms were counted using the plate count method and the number of bacteria was estimated.
2.3. Determination of the active ingredients
To determine the major constituents of the essential oil, electron impact mass spectra were determined at 70 eV on a GC 5890 HP instrument. Samples of 1 μL were analyzed by capillary gas chromatography [Hewlett–Packard 5890 Gas chromatograph (GC); Palo Alto, CA, USA] equipped with a mass detector and a 30 m × 0.25 mm HP-5 column with 0.25 μm film thickness. Temperatures were kept at 220 and 300 °C, respectively. Helium was used as the carrier gas; the flow rate through the column was 1 ml/min. Subsequently, the essential oil of C. schoenanthus was analyzed chemically by GC/MS.
3. Results
3.1. Antimicrobial susceptibility testing of C. schoenanthus
3.1.1. Agar well-diffusion test
In vitro antimicrobial activity of the essential oil of C. schoenanthus plant was tested against 10 bacterial pathogens using the agar well-diffusion method. Antimicrobial activity was recorded as the clear zone of inhibition (in millimeters) surrounding the agar well. The means of the zones of inhibition are shown in Table 1. Inhibitory effect was detected on five pathogens, including three Gram-positive (S. aureus, MSSA, S. aureus, MRSA) and two Gram-negative bacteria E. coli and K. pneumonia). The zones of inhibition (in mm) were as follows: the lowest inhibition was noted in the case of S. saprophyticus (10 ± 0.19), for S. aureus (MSSA) (12.5 ± 0.6) and MRSA (11 ± 0.4). In the case of the two Gram negative bacteria, the zone of inhibition was as such: E. coli (15 ± 0.2), K. pneumonia (14 ± 0.16), no antimicrobial activity was observed against P. mirabilis, P. aeruginosa, S. marcescens, E. faecium and E. faecalis.
Table 1.
Antibacterial activity of Cymbopogon schoenanthus essential oil by measuring zones of growth inhibition (mm) using agar well-diffusion assay.a
| Test Microorganism | Zone of Inhibition (mm)b |
|
|---|---|---|
| Essential oil | Oxacillin 5 μg/ml | |
| Staphylococcus aureus ATCC 6538 | 12.5 ± 0.6 | 28 ± 0.14 |
| S. aureus ATCC 33591 (MRSA) | 11 ± 0.14 | 0 |
| S. saprophyticus ATCC 35552 | 10 ± 0.19 | 12 ± 0.17 |
| Escherichia coli ATCC 11229 | 15 ± 0.2 | 0 |
| Klebsiella pneumoniae ATCC 4352 | 14 ± 0.16 | 0 |
| Proteus mirabilis ATCC 7002 | 0 | 0 |
| Pseudomonas aeruginosa ATCC 15442 | 0 | 0 |
| Serratia marcescens ATCC 14756 | 0 | 0 |
| Enterococcus faecium ATCC 6569 | 0 | 0 |
| E. faecalis ATCC 51299 (VRE) | 0 | 0 |
Oxacillin 5 μg/ml was used as a reference.
Each value represents the mean and standard deviation of three separate experiments.
3.2. Broth microdilution MIC test
The MIC of C. schoenanthus was carried out on the five bacterial pathogens that showed positive results using the agar well-diffusion test. Maximum growth of bacteria was achieved at 24 h for the Broth microdilution test. The oil was found effective against E. coli (MIC: 9.37 μg/ml), S. aureus (MSSA) (MIC: 4.69 μg/ml), S. aureus (MRSA) (MIC: 2.34 μg/ml) and K. pneumoniae (MIC: 2.34 μg/ml). The MIC was too high to be detected for S. saprophyticus at 150 μg/ml) (Table 2).
Table 2.
Determination of minimum inhibitory concentration of Cymbopogon schoenanthus extract.
| Escherichia coli | Staphylococcus aureus (MRSA) | S. aureus (MSSA) | Klebsiella pneumoniae | S. saprophyticus |
|---|---|---|---|---|
| 9.37 μg/ml | 4.69 μg/ml | 2.3 μg/ml | 2.3 μg/ml | >150 μg/ml |
Abbreviation: MIC, minimum inhibitory concentration; MSSA, methicillin-sensitive Staphylococcus aureus.
3.3. Time-kill assay
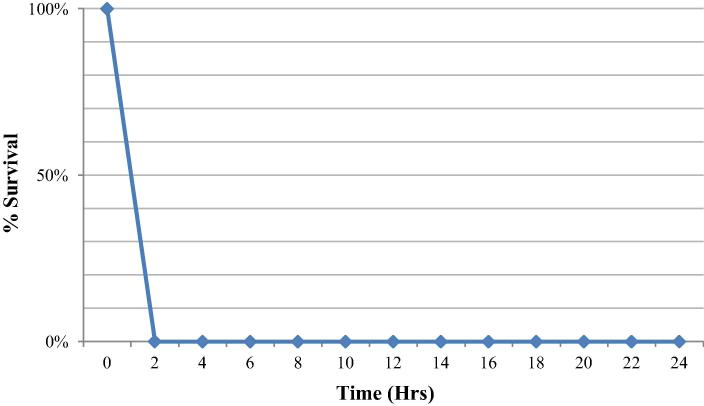
There was a time dependant decline with respect to time in the case of S. aureus, with a 90% reduction reached within 8 h of exposure (Fig. 1). However, K. pneumonia, was a lot more sensitive with an inhibition of 99.95% achieved within the first two hours of contact (Fig. 2).
Figure 1.
Effect of Cymbopogon schoenanthus oil on the survival of Staphylococcus aureus in relation to time.
Figure 2.
Effect of C. schoenanthus oil on the survival of Klebsiella pneumoniae in relation to time.
3.4. Chemical composition of C. schoenanthus essential oil
The solutions of C. schoenanthus were analyzed by GC/MS. The chromatographic profile of the major constituents obtained is shown in Table 3. The eight major components that identified were: piperitone (14.6%), cyclohexanemethanol (11.6%), beta-elemene (11.6%), alpha-eudesmol (11.5%), elemol (10.8%), beta-eudesmol (8.5%), 2-naphthalenemethanol (7.1%) and gamma-eudesmol (4.2%).
Table 3.
Composition of the Cymbopogon schoenanthus oil.
| Compound | Percent |
|---|---|
| Piperitone | 14.6 |
| Cyclohexanemethanol | 11.6 |
| β-Elemene | 11.6 |
| α-Eudesmol | 11.5 |
| Elemol | 10.8 |
| β-Eudesmol | 8.5 |
| 2-Naphthalenemethanol | 7.1 |
| γ-Eudesmol | 4.2 |
4. Discussion
The data obtained demonstrated that the essential oil of C. schoenanthus exhibited antibacterial activity against five of the ten tested bacterial pathogens. Published work by El-Kamali et al. (2005), using the well-diffusion method, indicated that essential oil of C. nervatus had antibacterial activity on all bacteria tested (S. aureus, Bacillus subtilis, E. coli, P. aeruginosa, Salmonella paratyphi A, Salmonella paratyphi B, Shigella dysenteriae, Shigella flexneri, Shigella boydii, P. mirabilis and K. pneumoniae), except for Salmonella typhi. The maximum inhibitory effect was against S. dysenteriae and K. pneumonia. In addition, ethanol and chloroform extracts of C. schoenanthus collected from Salboukh, north of Riyadh, Saudi Arabia were noted to have antibacterial activity against S. aureus. The MICs of both extracts for S. aureus were higher than what has been reported by us (Al Yahya et al., 1983, Lahlou, 2004). This discrepancy might be due to the method of extraction of essential oils. It is not unusual to notice significant differences in data for the same plant species. These variations might be due to many factors, including the method of extraction of essential oils, climatic, seasonal and geographical conditions, and harvest time. For this reason, it is important to standardize the methods of extraction and specify all the conditions that may affect the extraction.
Although it is commonly known that Gram-negative bacteria are slightly more sensitive to essential oils than Gram-positive ones (Chatterjee et al., 2011, Johnson et al., 2011, Ravikumar et al., 2012, Moussa et al., 2012), this is not always true. For instance, in a study by Deans and Ritchie (1987), fifty commercially available essential oils were tested against 25 genera, and no difference in sensitivity was found between Gram-negative and Gram-positive microorganisms. Moreover, E. coli was more susceptible to tea tree oil and other oils than S. aureus (Gustafson et al., 1998). Our study showed that K. pneumoniae, a Gram-negative bacterium, was the most susceptible microorganism. However, other Gram negative bacteria did not follow the same pattern. Takaisi-Kikuni and colleagues (2000) previously studied the effect of various amounts of the essential oil of C. densiflorus on the metabolic activity, growth and morphology of S. aureus. They concluded that relatively high concentrations of the oil impaired staphylococcal growth in a bacteriostatic manner, and in low doses, metabolism became ineffective due to energy losses in the form of heat (Reichling et al., 2009).
McLaughlin et al. (1998) suggested that any study on plant extracts and/or essential oils should include toxicity tests, as bioactive compounds are almost always toxic in high doses. Their results indicated that eight identified components of the oil accounted for 79.9% of the essential oil composition. These compounds belonged to two main classes: monoterpenes and sesquiterpenes. However, the proportion of sesquiterpenes (46.6%) was higher than that of the monoterpenes (14.6%). In a comprehensive review, Heiba and Rizk (1986) studied the essential oils of a number of Cymbopogon species and their components. They reported the presence of citronellol, farnesol, geraniol and sesquiterpene alcohols. Shahi and Tava (1993) studied the chemical composition of several essential oils and found that the main constituent of C. schoenanthus was piperitone (64.71%). Yentéma et al. (2007) tested the chemical composition of essential oil of C. schoenanthus in Burkina Faso and identified sixteen major compounds, which accounted for 65.2% of the whole oil composition. The percentage of monoterpenes (53.2%) was higher than that of sesquiterpenes (12%), and the major compounds were piperitone (42%), δ-2-carene (8.2%) and elemol (6.2%). The results of this study are in agreement with those of previous research in that the major ingredient was piperitone, although in a much lower concentration (14.6%). In 2005, a chemical study performed by Sousa and colleagues, 2005 revealed that the main components of the oil of C. schoenanthus were cis-para-menth-2-en-1-ol, trans-para-menth-2-en-1-ol and elemol when CO2 was used as solvent and cis-piperitol, trans-piperitol and elemol when ethanol was used as solvent (Sousa et al., 2005). It is not always true that the most abundant active ingredient is responsible for the activity of the essential oil. In a study on the anti-fungal activity of a plant extract where piperitone (24.74%) constituted the highest ingredient present, which is similar to our findings, and was tested alone and as a part of the whole extract. The results indicated that activity of the oil of Tagetes patula L. was not a result of the major constituents; rather, it was the result of the synergistic effect of all compounds present in the oil (Romagnoli et al., 2005). However, this synergism was not the case in a different study by El-Saeid et al. (2011), the main inhibitory effect resulted from piperitone against four phytopathogenic fungi.
The antimicrobial action of essential oils against Gram-positive bacteria is likely due to the destruction of the cell walls and cytoplasmic membrane of bacteria, resulting in leakage of the microorganism’s cytoplasmic contents and subsequently its inactivation. In other Gram-positive bacteria sensitive to imidazole and whose cell membranes are rich in unsaturated fatty acids, the rearrangement of the microorganism’s membrane components results in the loss of cell viability and eventually lysis (Kalemba and Kunicka, 2003). The authors concluded that the underlying mechanism of action of essential oils is caused by the inhibition of the synthesis of DNA, RNA, proteins and polysaccharides in both fungal and bacterial cells (Kalemba and Kunicka, 2003).
5. Conclusion
The essential oil of C. schoenanthus has an antibacterial effect against S. aureus, MSSA, S. saprophyticus, E. coli and K. pneumoniae, as indicated by its minimum inhibitory concentration. The results of the present study provide a scientific validation for the traditional use of the medicinal plant C. schoenanthus. Future studies should be conducted to assess the effect of the C. schoenanthus as a possible natural agent to enhance the efficacy of already existing antibiotic agents and to evaluate its application in various fields of medicine. Further, the mechanism of action by which the oil exerts an antibacterial effect has to be elucidated in order to determine more of its potential as an antibacterial agent.
Footnotes
Peer review under responsibility of King Saud University.
References
- Al-Ghamdi S.S., Al-Ghamdi A.A., Shammah A.A. Inhibition of calcium oxalate nephrotoxicity with Cymbopogon schoenanthus (Al-Ethkher) Drug Metab. Lett. 2007;1:241–244. doi: 10.2174/187231207783221420. [DOI] [PubMed] [Google Scholar]
- Al Yahya M.A., Al-Meshal I.A., Mossa J.S., Khatibi A., Hammouda Y. Phytochemical and biological screening of Saudi medicinal plants. Part II. Fitoterapia. 1983;54:21–24. [Google Scholar]
- Antonisamy P., Duraipandiyan V., Ignacimuthu S., Kim J.H. Anti-diarrhoeal activity of friedelin isolated from Azima tetracantha Lam. in Wistar rats. South Indian J. Biol. Sci. 2015;1:34–37. [Google Scholar]
- Balamurugan R. Smilax Chinensis Linn. (Liliaceae) root attenuates insulin resistance and ameliorate obesity in high diet induced obese rat. South Indian J. Biol. Sci. 2015;1:47–51. [Google Scholar]
- Kannan Barathi, Agastian P. In vitro regeneration of a rare antidiabetic plant Epaltes divaricata L. South Indian J. Biol. Sci. 2015;1:52–59. [Google Scholar]
- Chatterjee S.K., Bhattacharjee I., Chandra G. Isolation and identification of bioactive antibacterial components in leaf extracts of Vangueria spinosa (Rubiaceae) Asian Pac. J. Med. 2011;4:35–40. doi: 10.1016/S1995-7645(11)60028-X. [DOI] [PubMed] [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI) Clinical and Laboratory Standards Institute; 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087 USA: 2013. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-Third Informational Supplement. (CLSI document M100-S23 (ISBN 1-56238-865-7 [Print]; ISBN 1-56238-866-5 [Electronic])) [Google Scholar]
- Deans S., Ritchie G. Antibacterial properties of plant essential oils. Int. J. Food Microbiol. 1987;5:165–180. [Google Scholar]
- El-Askary H.I., Meselhy M.R., Galal A.M. Sesquiterpenes from Cymbopogon proximus. Molecules. 2003;8:670–677. [Google Scholar]
- El-Kamali H., Hamza M., El-Amir M. Antibacterial activity of the essential oil from Cymbopogon nervatus inflorescence. Fitoterapia. 2005;76:446–449. doi: 10.1016/j.fitote.2005.03.001. [DOI] [PubMed] [Google Scholar]
- El-Saeid M.H. Antifungal activity of natural piperitone as fungicide on root rot fungi. Am. Eurasian J. Agric. Environ. Sci. 2011;11(2):149–153. [Google Scholar]
- Elhardallou S.B. Cytotoxicity and biological activity of selected Sudanese medicinal plants. Res. J. Med. Plant. 2011;5:201–229. [Google Scholar]
- Farooqi I. Vol. 168. Ilm-o-Irfan Publishers; 9-lower Mall, Aqab Mian Market, Urdu Bazar Lahore: 1998. Ahadith Mein Mazkoor Nabatat, Adwiya Aur Ghizain; pp. 151–152. [Google Scholar]
- Gustafson J., Liew Y., Chew S., Markham J., Bell H., Wyllie S., Warmington J. Effects of tea tree oil on Escherichia coli. Lett. Appl. Microbiol. 1998;26:194–198. doi: 10.1046/j.1472-765x.1998.00317.x. [DOI] [PubMed] [Google Scholar]
- Heiba H.I., Rizk A.M. Constituents of cymbopogon species. Qatar Univ. Sci. J. 1986;6:53–75. [Google Scholar]
- Johnson M., Wesely E.G., Zahir Hussain M.I., Selvan N. In vivo and in vitro phytochemical and antibacterial efficacy of Baliospermum montanum (Wïlld.) Muell. Arg. Asian Pac. J. Med. 2011;3:894–897. [Google Scholar]
- Kalemba D., Kunicka A. Antibacterial and antifungal properties of essential oils. Curr. Med. Chem. 2003;10:813–829. doi: 10.2174/0929867033457719. [DOI] [PubMed] [Google Scholar]
- Ketoh G.K., Koumaglo H.K., Glitho I.A., Huignard J. Comparative effects of Cymbopogon schoenanthus essential oil and piperitone on Callosobruchus maculatus development. Fitoterapia. 2006;77:506–510. doi: 10.1016/j.fitote.2006.05.031. [DOI] [PubMed] [Google Scholar]
- Lahlou M. Methods to study the phytochemistry and bioactivity of essential oils. Phytotherapy Res. 2004;18:435–448. doi: 10.1002/ptr.1465. [DOI] [PubMed] [Google Scholar]
- Marwat S.K., Khan M.A., ur-Rehman F., Bhatti I.U. Aromatic plant species mentioned in the holy Qura’n and Ahadith and their ethnomedicinal importance. Pak. J. Nutr. 2009;8(9):1472–1479. [Google Scholar]
- Mclaughlin J.L., Rogers L.L., Anderson J.E. The use of biological assays to evaluate botanicals. Drug Inf. J. 1998;32:513–524. [Google Scholar]
- Mossa S., Al-Yahya A., Al-Meshal A. King Saud University Press; Riyadh, KSA: 1987. Medicinal Plants of Saudi Arabia. [Google Scholar]
- Moussa A., Noureddine D., Abdelmelek M., Saad A. Antibacterial activity of various honey types of Algeria against pathogenic Gram-Negative Bacilli: Escherichia coli and Pseudomonas aeruginosa. Asian Pac. J. Dis. 2012;2:211–214. [Google Scholar]
- Nandhini V.S., Bai S.G.V. In-vitro phytopharmacological effect and cardio protective activity of Rauvolfia tetraphylla L. South Indian J. Biol. Sci. 2015;1(2):97–102. [Google Scholar]
- Norbert G.K.B.J., Seth N.W., Dodji K.B., Roger N.C.H., Guillaume K.K., Essè A.K., Isabelle G.A. The use of two new formulations of Ocimum Canum Sims and Cymbopogon schoenanthus L. In The Control of Amitermes Evuncifer Silvestri (Termitidae: Termitinae), in Togo. Int. J. Natl. Sci. Res. 2014;2:195–205. [Google Scholar]
- Narendran R.T., Ramavarma S.K.M., Pongathara A.M., Arif R. A study on antimicrobial and antioxidant potential of Biophytum sensitivum in three different solvents. South Indian J. Biol. Sci. 2016;2(1):1–8. [Google Scholar]
- Noorudheen N., Chandrasekharan D.K. Effect of ethanolic extract of Phyllanthus emblica on captan induced oxidative stress in vivo. South Indian J. Biol. Sci. 2016;2016:95–103. [Google Scholar]
- Puthur J.T. Antioxidants and cellular antioxidation mechanism in plants. South Indian J. Biol. Sci. 2016;2(1):9–13. [Google Scholar]
- Ravikumar S., Gokulakrishnan R., Boomi P. In vitro antibacterial activity of the metal oxide nanoparticles against urinary tract infectious bacterial pathogens. Asian Pac. J. Dis. 2012;2:85–89. [Google Scholar]
- Rathi M.A., Meenakshi P., Gopalakrishnan V.K. Hepatoprotective activity of ethanolic extract of Alysicarpus vaginalis against nitrobenzene-induced hepatic damage in rats. South Indian J. Biol. Sci. 2015;1(2):60–65. [Google Scholar]
- Reichling J., Schnitzler P., Suschke U., Saller R. Essential oils of aromatic plants with antibacterial, antifungal, antiviral, and cytotoxic properties-an overview. Forsch. Komplementmed. 2006;16:79. doi: 10.1159/000207196. [DOI] [PubMed] [Google Scholar]
- Romagnoli C., Bruni R., Andreotti E., Rai M., Vicentini C., Mares D. Chemical characterization and antifungal activity of essential oil of capitula from wild Indian Tagetes patula L. Protoplasma. 2005;225:57–65. doi: 10.1007/s00709-005-0084-8. [DOI] [PubMed] [Google Scholar]
- Sabry A., El-Zayat S., El-Said A., Abdel-Motaal F., Magraby T. Mycoflora associated with Halfa-bar leaves and stems (Cymbopogon schoenanthus L. Sprengel), in vitro the antimicrobial activity of the plant leaves and stems secondary metabolites. Int. J. Curr. Microbiol. Appl. Sci. 2014;3:874–882. [Google Scholar]
- Santhosh S.K., Venugopal A., Radhakrishnan M.C. Study on the phytochemical, antibacterial and antioxidant activities of Simarouba glauca. South Indian J. Biol. Sci. 2016;2(1):119–124. [Google Scholar]
- Shahi A.K., Tava A. Essential oil composition of three Cymbopogon species of Indian Thar Desert. J. Essent. Oil Res. 1993;5:639–643. [Google Scholar]
- Sousa E.M.B.D.D., Câmara A.P.C., Costa W.A., Costa A.C.J., Oliveira H.N.M., Galvão E.L., Marques M.M.O. Evaluation of the extraction process of the essential oil from Cymbopogon schoenanthus with pressurized carbon dioxide. Braz. Arch. Biol. Technol. 2005;48:231–236. [Google Scholar]
- Takaisi-Kikuni N.B., Tshilanda D., Babady B. Antibacterial activity of the essential oil of Cymbopogon densiflorus. Fitoterapia. 2000;71:69–71. doi: 10.1016/s0367-326x(99)00097-0. [DOI] [PubMed] [Google Scholar]
- Yentéma O., Alioune O., Dorosso S.A. Chemical composition and physical characteristics of the essential oil of Cymbopogon schoenanthus (L.) Spreng of Burkina Faso. J. Appl. Sci. 2007;7:503–506. [Google Scholar]