Abstract
The effects of poly-γ-glutamic acid (γ-PGA) on the stability of yogurt were studied in terms of texture, rheology, particle size, and microstructure. The effects of γ-PGA on the pH and water holding capacity (WHC) of yogurt during refrigeration were also studied. The tan δ value of the yogurt with 0.04% (w/w) γ-PGA was significantly lower than that of control. The addition of γ-PGA decreased the particle size. The yogurt with 0.02% (w/w) γ-PGA had denser network structure with less porosity, and cross-linking between complexes occurred with greater frequency. The pH of the yogurt supplemented with 0.02% and 0.04% γ-PGA was significantly higher. WHC increased with the amount of γ-PGA. The addition of γ-PGA to yogurt strengthened the antishear ability, inhibited the postacidification phenomenon, and improved stability during refrigeration.
Keywords: Set yogurt, Poly-γ-glutamic acid, Texture, Rheology, Refrigerated storage
Introduction
Set yogurt is a dairy product produced by the fermentation of milk by lactic acid bacteria (Streptococcus thermophilus and Lactobacillus delbrueckii sp. bulgaricus). Its consumption has increased significantly due to its nutritional value and unique flavor. According to Euromonitor data, global yogurt consumption has increased by approximately 23% in recent years.
During the production, transportation, and storage of set yogurt, whey separation and weak body are common problems that directly affect consumers’ acceptance of the product. To maintain the stability of the set yogurt, many stabilizers have been used, including pectin (Kim and Wicker 2011; Zhang et al. 2014), soybean soluble polysaccharides (Nakamura et al. 2003; Nakamura et al. 2006), and κ-carrageenan (Spagnuolo et al. 2005). Pectin has been widely used in yogurt as a thickening agent. Pectin and casein micelles produce a biopolymer mixture through electrostatic interactions (Matia-Merino and Singh 2007). Soybean soluble polysaccharide, which has a pectin-like structure, has been used as a dispersion stabilizer for milk protein under acidic conditions (Nakamura et al. 2003). Increasing the protein content has also been used to strengthen the stability of set yogurt (Sun et al. 2016; Yu et al. 2016). Proteins have been shown to act as fillers or binders within a casein matrix (Lobato-Calleros et al. 2014). Gelatin has been shown to have good gelling capacity in yogurt due to its water holding capacity (WHC) (Pang et al. 2015). Enzymatic cross-linking has been a highly preferred method of improving the functional properties of proteins. For example, transglutaminase, a transferase, has been used in the formation of cross-links in proteins. Şanlı et al. (2011) found that the treatment of milk proteins with transglutaminase was an effective way to increase the techno-functional properties of set yogurt.
Poly-γ-glutamic acid (γ-PGA) is a polymer of the amino acid glutamic acid. It is an edible anionic polypeptide obtained from Bacillus subtilis natto through a fermentation process (Lee and Kuo 2011). Therefore, it may act like protein and become a filler or binder in a casein matrix. At the end of the glutamic acid side chain, peptide bonds occur between the amino group and the carboxyl group (Seenivasagam et al. 2013), giving it a high capacity for absorbing water and metal ions. γ-PGA has been used as a basic material for carrying drugs in the medical industry (Yang et al. 2017), in moisturizers in the cosmetics industry (Ben-Zur and Goldman 2007), and as thickeners, bitterness-relieving agents, cryoprotectants, and mineral absorbents in the food industry (Mitsunaga et al. 2016). Shyu et al. (2008) investigated the effects of γ-PGA on the rheological and thermal properties of wheat dough and found that the use of γ-PGA improved the dough’s rheological and thermal properties and retarded the staling process. Lee and Kuo (2011) discussed the influence of γ-PGA on the rheological properties and microstructure of tofu. Tofu syneresis was reduced by the addition of γ-PGA due to its excellent water absorption property.
This study investigated the effect of γ-PGA on the macroscopic and microscopic properties of yogurt and on its qualitative parameters (pH, WHC) during storage.
Materials and methods
Materials
The γ-PGA powder (molecular weight, 250 kDa; purity, 99.07%) was formed by Bacillus subtils natto fermentation in our laboratory. Whole fat milk (6% fat, 5% protein) was purchased from Shanghai Bright Dairy Co. Ltd (Shanghai, China). Sugar was purchased from China Agri-Industries Holdings Limited, China. Yogurt starter cultures (a mixture of Streptococcus thermophilus, Lactobacillus bulgaricus, Bacillus bifidus, Bacillus acidophilus, and Lactobacillus casei) were purchased from Beijing Chuanxiu Technology Ltd (Beijing, China).
Preparation of the set yogurt
The γ-PGA solution was prepared by dissolving the γ-PGA powder (0.88 g) into 20 mL of sterile water at room temperature. Sugar (19.8 g) and the γ-PGA solution (1.5 mL, 3.0 mL, 4.5 mL, and 6.0 mL; i.e., 0.02%, 0.04%, 0.06%, and 0.08% w/w) were dissolved in whole fat milk (300 mL) with moderate stirring at room temperature. The milk mixture was inoculated with 0.02% (w/v) starter culture and stirred thoroughly. The milk mixture was poured into reusable glass jars to 70% full (150 mL with a lid). The jars were then preheated at 40 °C without agitation for 1 h in an incubator. Next, they were placed in a yogurt maker (Severin JG3516, Germany) and incubated at 43 °C for 6 h. The time was determined by the final pH (about 4.6) of the fermented yogurt (Jørgensen et al. 2016). The pH values of the final product supplemented with different γ-PGA concentrations (control, 0.02%, 0.04%, 0.06%, and 0.08% w/w) were 4.61 ± 0.02, 4.66 ± 0.02, 4.67 ± 0.01, 4.62 ± 0.02, and 4.58 ± 0.02, respectively. The yogurt samples were then transferred to a refrigerator and stored at 4 °C.
Rheological properties
The rheological properties of the set yogurt samples were measured using a rheometer (AR-G2, TA Instruments, New Castle, DE) with a cone and plate geometry (40 mm diameter, 4° angle). Before the experiments, the set yogurt samples were gently stirred 20 times with a spatula. All samples were equilibrated to 25 °C. Measurements were taken in triplicate for each sample.
Measurements in stationary mode were used to characterize the shear viscosity. The set yogurt samples were tested under the controlled rate mode in the shear rate range of 0.01–100 s−1.
Dynamic oscillation tests were performed to determine the viscoelastic properties of the set yogurt samples. Strain sweeps were performed from 0 to 0.5% to identify the linear viscoelastic range. The frequency sweep tests were performed within the linear viscoelastic range to obtain the storage modulus (G′), the loss modulus (G″), and the phase angle (tan δ).
Texture measurements
The texture properties, such as hardness, stringiness, springiness, adhesiveness, cohesiveness, resilience, and chewiness were determined by a texture profile analysis (TPA) test. Texture measurements were performed on a TA-XT texture analyzer (Stable Micro Systems Ltd, Godalming, England). The set yogurt samples were tested in glass jars without stirring. The samples were tested immediately after removal from the refrigerator. The probe was a 25-mm diameter acrylic cylinder. It moved at a pretest speed of 1 mm/s, a test speed of 1 mm/s, and a post-test speed of 1 mm/s. The target value was 40% strain. Three replicates were performed.
Apparent particle size
The apparent particle size of the yogurt samples was determined using a Zetasizer Nano ZSP (Malvern Instruments Ltd., Worcestershire, UK) at room temperature. Based on the principle of dynamic light scattering, the apparent particle size was measured using a 173º scattering angle. Before measurement, all samples were diluted 100-fold with deionized water. Each sample was measured in quintuplicate, and the average value was used.
Microstructure observation
A QUANTA-200 scanning electron microscopy (SEM, FEI Ltd., Eindhoven, The Netherlands) was used to observe the microstructure of the yogurt samples. The yogurt samples were cut into 1 × 1 × 1-mm cubes and fixed with glutaraldehyde solution (3%, v/v) at 4 °C for 3 h. Then, the samples were washed with a phosphate buffer (0.1 M, pH 7.3) three times (10 min each time). The fixed samples were then post-fixed with osmium tetroxide solution (1%, v/v) and washed three times (10 min each time) with phosphate buffer (0.1 M, pH 7.3). The post-fixed samples were frozen immediately in liquid nitrogen and dehydrated using a gradient ethanol series (30%, 50%, 70%, 90%, and 100%) and were replaced using an isoamyl acetate series (30%, 50%, 70%, 90%, and 100%). The samples were then dried with a critical point dryer (CPD, Balzers CPD 030Critical Point Dryer, Leica, DE, Wetzlar, Germany). The dried samples were sliced and mounted on aluminum stubs with double-sided carbon-tapes and sputter-coated with pure gold. Microstructure observations were carried out at an accelerating voltage of 10 kV with a magnification of 8000×.
Stability of the yogurt supplemented with γ-PGA during storage
The changes in pH and WHC during refrigeration were monitored to determine the stability of the yogurt supplemented with different concentrations of γ-PGA (0.02%, 0.04%, 0.06%, and 0.08% w/w). These parameters were determined on the 1st, 3rd, 7th, 14th, 21st, and 28th days of storage.
The pH was determined using a Five Easy Plus pH meter (Mettler Toledo, Inc., Switzerland).
The measurement of WHC was performed using the procedure described by Lin (2011) with some modifications. An empty centrifuge tube was weighed as w0. The centrifuge tube was then one-third filled with a yogurt sample that had just been removed from the refrigerator. The weight was recorded as w1. The tube with the yogurt was centrifuged at 3000 r/min for 20 min at 4 °C. The supernatant was removed, and the tube was weighed as w2. The WHC was calculated using Eq. (1):
| 1 |
The measurements for the pH and WHC were made three times.
Statistical analysis
The results are expressed as mean ± SD from at least three replicates. The effects of γ-PGA on the texture parameters and qualitative parameters (pH, WHC) during storage were analyzed using one-way analysis of variance.
Results and discussion
Rheological properties
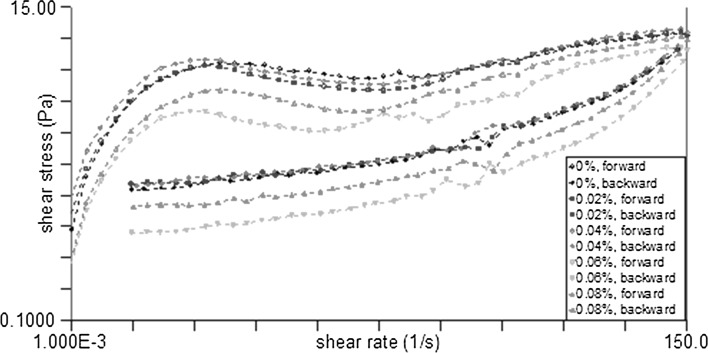
As illustrated in Fig. 1, the flow curves for the yogurt supplemented with different concentrations of γ-PGA (0.02%, 0.04%, 0.06%, and 0.08% w/w) had similar properties as control, indicating presence of similar internal structure. In the ascending curve, as the shear rate increased, the shear stress first increased rapidly and then increased gradually. All samples showed a structural protrusion (corresponding to the maximum shear stress). The emergence of structural protrusions at higher shear rates implied that the system had a stronger gel structure (Milanović et al. 2014). In the downward curve, the shear stress gradually decreased as the shear rate decreased, suggesting that after the up-shear process, the macromolecules and particles were already oriented and could flow quickly in the direction of shear. Thixotropic hysteresis loops were observed in each of the set yogurt samples. These observations were consistent with previous studies of yogurt systems (Cruz et al. 2013; Lee and Kuo 2011). In Fig. 1, the order of the shear rates corresponding to the protruding points of the structure was 0.04%, 0.02%, control, 0.08%, and 0.06% (from high to low). The corresponding shear rates of the latter two samples were obviously smaller than first three samples, indicating that the concentrations lower than 0.04% were favorable to the gel structure of the system.
Fig. 1.
The flow curve of shear stress with shear rate of the set yoghurts supplemented with different concentrations of γ-PGA (0.02%, 0.04%, 0.06% and 0.08%, w/w) at 25 °C
The yogurt was in the linear viscoelastic region when the strain was controlled at 0.1%. The relationships of G′ and G″ with angular frequency were studied. The yogurt system had a solid-attribute at 0.1% strain preference and formed a weak gel net structure, as evidenced by G′ > G″. The tan δ value represents the energy loss of the yogurt system when it was subjected to shearing. A low tan δ value indicates less energy loss in the system during shearing, that is, a high antishear ability. The tan δ values of the yogurt samples supplemented with the 0%, 0.02%, 0.04%, 0.06%, and 0.08% γ-PGA were 0.249 ± 0.002, 0.247 ± 0.001, 0.235 ± 0.003, 0.242 ± 0.004, and 0.250 ± 0.000, respectively. The tan δ value of the yogurt with 0.04% γ-PGA was significantly lower than those of the other yogurt samples, indicating that the 0.04% concentration of γ-PGA strengthened the antishear ability of the set yogurt system.
Texture properties
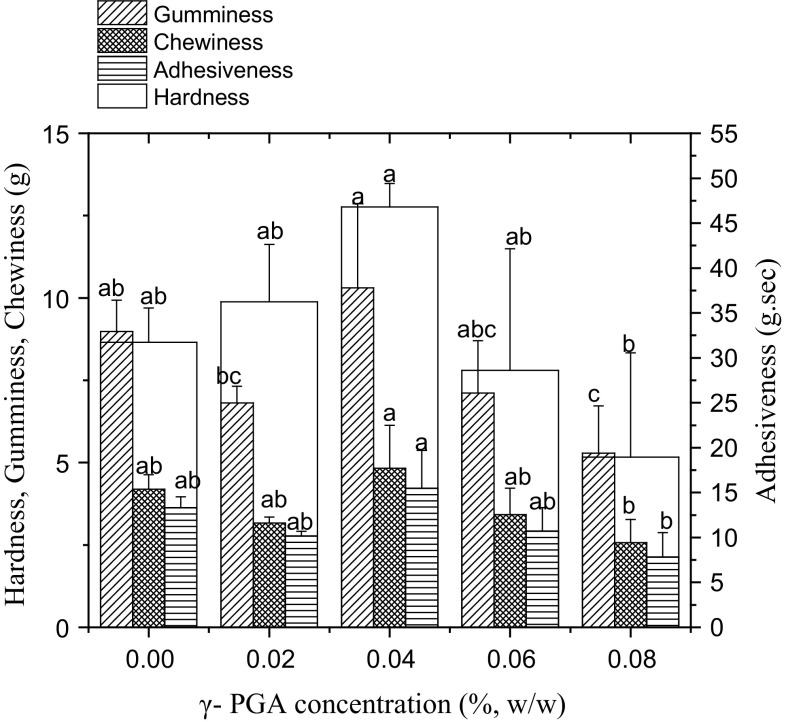
Figure 2 shows the effect of γ-PGA on the set yogurt system’s seven texture attributes: hardness, gumminess, chewiness, adhesiveness, springiness, cohesiveness, and resilience. Hardness, gumminess, chewiness, and adhesiveness initially increased and then decreased as the addition concentration of γ-PGA increased. The hardness was measured as the force required to disrupt the structure (Ramchandran and Shah 2009; Rawson and Marshall 1997), which reflected the strength of the set yogurt. Adhesiveness helped to provide a thicker texture and played a positive role in maintaining yogurt stability. Within a certain range, the yogurts with greater hardness and adhesiveness were deemed delicious (Delikanli and Ozcan 2014). An optimal addition of γ-PGA (0.04% w/w) enhanced the strength and gel elasticity of the coagulated yogurt and strengthened the gelatin state. The interaction between the internal molecules in the yogurt system was improved by the addition of γ-PGA, and a stable network structure was formed (Zhao et al. 1995). For springiness, cohesiveness, and resilience, no significant differences were observed among the five set yogurt samples.
Fig. 2.
The texture parameters of the set yoghurts supplemented with different concentrations of γ-PGA (0.02%, 0.04%, 0.06%, and 0.08%, w/w)
Apparent particle size
The apparent particle sizes were 7.913 ± 0.057, 6.902 ± 0.056, 6.943 ± 0.015, 7.415 ± 0.040, and 7.881 ± 0.012 μm, corresponding to the control, 0.02%, 0.04%, 0.06%, and 0.08% (w/w) γ-PGA concentrations, respectively. Overall, the addition of γ-PGA resulted in smaller particles. When modified starch was added to the set yogurt system, the particle size also decreased (Cui et al. 2014), possibly as a result of the intensive electrostatic force between the positively charged casein micelles and the negatively charged γ-PGA. Under this intensive electrostatic force, the particle size of casein/γ-PGA complexes dramatically decreased. Although the adsorption of γ-PGA on casein micelles increased the particle size of casein/γ-PGA complexes, this positive influence on particle size was masked by the effect of the strong electrostatic force. In a colloidal system, when the particle size is reduced, Brownian motion becomes more intense, which helps to maintain the stability of the colloidal system (Cui et al. 2014). It can be seen from the data that when the γ-PGA concentration was 0.02%, the particle size of the yogurt samples was the smallest, indicating that the electrostatic attraction between the casein micelles and γ-PGA was very strong at this time.
Microstructure
Figure 3 shows the SEM micrographs of the set yogurt system with different concentrations of γ-PGA (control, 0.02%, 0.04%, 0.06%, and 0.08% w/w). From Fig. 3, it can be observed that the set yogurt samples formed a three-dimensional network structure. The whole network structure was formed by stacks of many spherical casein micelles, and a few chain segments were formed, resulting in the low rigidity observed in the yogurt samples (Hu 2013). The structure of the polymer was rich in carboxylic groups that could form salt bridges with the amino groups on the surface of the casein molecules (Hill and Kethireddipalli 2013). Electrostatic repulsion has been shown to play an important role in the formation of the microstructures in acid dairy systems (Peng and Guo 2015; Renard et al. 2006). When the electrostatic repulsion between particles in the system was high, the chain structure formed more easily (Clark et al. 2001). More chain branches helped the formation of a stable and strong network structure (Şanlı et al. 2011). Also, hydrophobic interaction, which has been shown to provide a major impetus for protein folding, leaving hydrophobic residues inside the protein molecule, plays an important role in the complexation by calcium phosphate. In addition, the morphology of casein micelles has been shown to be strongly influenced by pH. When milk was placed in a neutral environment (pH 7.0), the casein micelles had a certain amount of negative charge; the electrostatic repulsion between the micelles was great in this condition, and the casein micelles were relatively stable. When the pH of the milk system began to decrease, the net negative charge of the casein micelles in the system gradually decreased, and the electrostatic repulsion between the micelles gradually weakened. When the pH dropped to the isoelectric point of the protein (about 4.6), a large amount of calcium phosphate in the micelles dissolved, and the charge carried by the micelles changed from negative to positive, resulting in the extreme weakening of the electrostatic repulsion between casein micelles, leading to the aggregation of casein micelles to form a gel. For this reason, the interaction between protein and γ-PGA varies with the pH (Wang et al. 2012). In the control yogurt sample (Fig. 3a), the network structure was formed by disorderly stacks of complex particles, and the structure was very loose and coarse with many large pores. When 0.02% (w/w) γ-PGA was added to the yogurt sample, the network structure was denser with less porosity, and cross-linking between complexes occurred with greater frequency (Fig. 3b). Accordingly, the structure became smoother. However, with the further addition of γ-PGA (exceeding 0.04%, w/w), the structure gradually became looser, and the cross-linking was weaker with larger pores. This may be attributed to the weakened electrostatic repulsion caused by the superfluous γ-PGA or changes in the yogurt’s pH.
Fig. 3.
SEM micrographs of γ-PGA/casein gels system with different γ-PGA concentration. a without γ-PGA, b with 0.02% γ-PGA, c with 0.04% γ-PGA, d with 0.06% γ-PGA and e with 0.08% γ-PGA; ×8000 magnification
Stability of the set yogurt supplemented with γ-PGA during refrigeration
pH
The initial pH values of milk with different γ-PGA concentrations (control, 0.02%, 0.04%, 0.06%, and 0.08% w/w) were 6.83 ± 0.01, 6.82 ± 0.01, 6.79 ± 0.01, 6.78 ± 0.01, and 6.74 ± 0.01, respectively. The pH values of the set yogurt supplemented with different concentrations of γ-PGA during refrigeration are shown in Table 1.
Table 1.
The effect of γ-PGA on the pH of the set yoghurt samples during refrigerated storage (mean ± SD)
| Parameter | γ-PGA (w/w, %) | Storage time (day) | |||||
|---|---|---|---|---|---|---|---|
| 1 | 3 | 7 | 14 | 21 | 28 | ||
| pH | Control | 4.61 ± 0.02E,ab | 4.55 ± 0.01D,a | 4.51 ± 0.03D,b | 4.40 ± 0.02C,a | 4.27 ± 0.04B,a | 4.17 ± 0.01A,a |
| 0.02 | 4.66 ± 0.02D,c | 4.60 ± 0.02C,bc | 4.46 ± 0.03B,a | 4.46 ± 0.01B,b | 4.42 ± 0.04B,c | 4.17 ± 0.04A,a | |
| 0.04 | 4.67 ± 0.01E,c | 4.61 ± 0.01D,c | 4.55 ± 0.02C,b | 4.46 ± 0.01B,b | 4.44 ± 0.03B,c | 4.21 ± 0.03A,a | |
| 0.06 | 4.62 ± 0.02E,b | 4.55 ± 0.03D,a | 4.52 ± 0.01C,b | 4.51 ± 0.01C,c | 4.35 ± 0.01B,b | 4.18 ± 0.01A,a | |
| 0.08 | 4.58 ± 0.02D,a | 4.56 ± 0.04CD,ab | 4.53 ± 0.02CD,b | 4.52 ± 0.02C,c | 4.32 ± 0.03B,b | 4.15 ± 0.04A,a | |
Different ‘abc’ letters on the column values indicated they were significantly different at P < 0.05. Different ‘ABC’ letters on the row values indicated they were significantly different at P < 0.01
As shown in Table 1, the pH values of the set yogurts supplemented with 0.02% and 0.04% γ-PGA were significantly higher than the other three samples, except on the 14th day. According to Sahan et al. (2008), the addition of a β-glucan composite also increased the pH of the yogurt during storage. From the 7th day to the 21st day, the pH values of the yogurts supplemented with 0.02% γ-PGA did not significantly decrease, indicating that the samples were stable during that period. A metamorphosis occurred in all five yogurt samples on the 28th day of storage, indicating that although γ-PGA was beneficial for maintaining the stability of the set yogurt during storage, it could not extend the shelf life of the set yogurt.
WHC
The WHC is directly related to whey separation. During storage, whey separation tends to occur in yogurt with a low WHC. Therefore, WHC is one of the most important characteristics used to evaluate the quality of yogurt. The variation in the WHC among the five yogurt samples supplemented with different concentrations of γ-PGA during refrigeration is shown in Table 2.
Table 2.
The effect of γ-PGA on the WHC of the set yoghurt samples during refrigerated storage (mean ± SD)
| γ-PGA (w/w, %) | Storage time (day) | |||||
|---|---|---|---|---|---|---|
| 1 | 3 | 7 | 14 | 21 | 28 | |
| 0 | 0.55 ± 0.07B,a | 0.48 ± 0.01AB,a | 0.48 ± 0.02AB,a | 0.46 ± 0.06A,a | 0.43 ± 0.03A,a | 0.42 ± 0.01A,a |
| 0.02 | 0.57 ± 0.06B,a | 0.48 ± 0.01B,a | 0.48 ± 0.02AB,a | 0.46 ± 0.06AB,a | 0.43 ± 0.03AB,a | 0.42 ± 0.01A,a |
| 0.04 | 0.59 ± 0.03B,a | 0.53 ± 0.03AB,a | 0.53 ± 0.05AB,a | 0.49 ± 0.04A,a | 0.48 ± 0.07A,a | 0.45 ± 0.03A,a |
| 0.06 | 0.59 ± 0.05A,a | 0.55 ± 0.09A,a | 0.53 ± 0.06A,a | 0.52 ± 0.04A,a | 0.50 ± 0.09A,a | 0.47 ± 0.09A,a |
| 0.08 | 0.60 ± 0.03B,a | 0.55 ± 0.04AB,a | 0.54 ± 0.03AB,a | 0.53 ± 0.00AB,a | 0.50 ± 0.06A,a | 0.47 ± 0.04A,a |
Different ‘abc’ letters on the column values indicated they were significantly different at P < 0.05. Different ‘ABC’ letters on the row values indicated they were significantly different at P < 0.01
As shown in Table 2, the WHC of all five yogurt samples decreased as the storage time increased. As the amount of γ-PGA increased, the WHC also increased. γ-PGA is a large polymer and possesses extremely strong water-absorbing ability (Lee and Kuo 2011); thus, it can be deduced that γ-PGA improved the set yogurt system’s stability during storage by enhancing its WHC.
Conclusion
To investigate the effect of γ-PGA on the stability of set yogurt systems, the rheological properties, texture, and some quality parameters (pH and WHC) during refrigeration were monitored. The use of γ-PGA at a proper concentration improved the antishear ability, inhibited the postacidification phenomenon, and enhanced the WHC during refrigeration. Therefore, γ-PGA is recommended as a texture modifier for set yogurts. The addition of γ-PGA to yogurt helped to solve the problem of whey precipitation and curd failure during the production, transportation, and sale of yogurt. It could improve product quality and reduce company losses.
Acknowledgements
The work was funded by open foundation from Key Laboratory of Biotechnology in Dairy Industry, China (SKLDB2013-09), the Ability Construction Project of the Science and Technology Commission Foundation of Shanghai (No. 15590503500), and “Shu Guang” project (No. 16SG50) supported by Shanghai Municipal Education Commission.
References
- Ben-Zur N, Goldman DM. γ-Poly glutamic acid: a novel peptide for skin care. Cosmet Toilet. 2007;122:65–74. [Google Scholar]
- Clark A, Kavanagh G, Ross-Murphy S. Globular protein gelation—theory and experiment. Food Hydrocoll. 2001;15:383–400. doi: 10.1016/S0268-005X(01)00042-X. [DOI] [Google Scholar]
- Cruz AG, Cavalcanti RN, Guerreiro LMR, Sant’Ana AS, Nogueira LC, Oliveirad CAF, Delizae R, Cunhaa RL, Fariaa JAF, Bolinia HMA. Developing a prebiotic yogurt: rheological, physico-chemical and microbiological aspects and adequacy of survival analysis methodology. J Food Eng. 2013;114:323–330. doi: 10.1016/j.jfoodeng.2012.08.018. [DOI] [Google Scholar]
- Cui B, Tan C, Lu Y, Liu X, Li G. The interaction between casein and hydroxypropyl distarch phosphate (HPDSP) in yoghurt system. Food Hydrocoll. 2014;37:111–115. doi: 10.1016/j.foodhyd.2013.10.032. [DOI] [Google Scholar]
- Delikanli B, Ozcan T. Effects of various whey proteins on the physicochemical and textural properties of set type nonfat yoghurt. Int J Dairy Technol. 2014;67:495–503. doi: 10.1111/1471-0307.12142. [DOI] [Google Scholar]
- Hill AR, Kethireddipalli P. Chapter 8-Dairy products: cheese and yogurt. In: Eskin NM, Shahidi F, editors. Biochemistry of foods. San Diego: Academic Press; 2013. [Google Scholar]
- Hu L. Research on the mechanism of stabilisation of acid casein gels by high methoxyl pectin in yogurt drinks. China: Zhejiang Gongshang University; 2013. [Google Scholar]
- Jørgensen CE, Abrahamsen RK, Rukke EO, Johansen AG, Skeie SB. Fractionation by microfiltration: effect of casein micelle size on composition and rheology of high protein, low fat set yoghurt. Int Dairy J. 2016;74:12–20. doi: 10.1016/j.idairyj.2016.11.018. [DOI] [Google Scholar]
- Kim Y, Wicker L. Charge domain of modified pectins influence interaction with acidified caseins. Food Hydrocoll. 2011;25:419–425. doi: 10.1016/j.foodhyd.2010.07.011. [DOI] [Google Scholar]
- Lee CY, Kuo MI. Effect of γ-polyglutamate on the rheological properties and microstructure of tofu. Food Hydrocoll. 2011;25:1034–1040. doi: 10.1016/j.foodhyd.2010.10.001. [DOI] [Google Scholar]
- Lin H. Studies on the quality improvement and shelf-life stability of set soybean yogurt. China: Zhejing Gongshang University; 2011. [Google Scholar]
- Lobato-Calleros C, Ramírez-Santiago C, Vernon-Carter E, Álvarez-Ramírez J. Impact of native and chemically modified starches addition as fat replacers in the viscoelasticity of reduced-fat stirred yogurt. J Food Eng. 2014;131:110–115. doi: 10.1016/j.jfoodeng.2014.01.019. [DOI] [Google Scholar]
- Matia-Merino L, Singh H. Acid-induced gelation of milk protein concentrates with added pectin: effect of casein micelle dissociation. Food Hydrocoll. 2007;21:765–775. doi: 10.1016/j.foodhyd.2006.12.007. [DOI] [Google Scholar]
- Milanović J, Petrović L, Sovilj V, Katona J. Complex coacervation in gelatin/sodium caseinate mixtures. Food Hydrocoll. 2014;37:196–202. doi: 10.1016/j.foodhyd.2013.10.016. [DOI] [Google Scholar]
- Mitsunaga H, Meissner L, Büchs J, Fukusaki E. Branched chain amino acids maintain the molecular weight of poly (γ-glutamic acid) of Bacillus licheniformis ATCC 9945 during the fermentation. J Biosci Bioeng. 2016;122:400–405. doi: 10.1016/j.jbiosc.2016.03.007. [DOI] [PubMed] [Google Scholar]
- Nakamura A, Furuta H, Kato M, Maeda H, Nagamatsu Y. Effect of soybean soluble polysaccharides on the stability of milk protein under acidic conditions. Food Hydrocoll. 2003;17:333–343. doi: 10.1016/S0268-005X(02)00095-4. [DOI] [Google Scholar]
- Nakamura A, Yoshida R, Maeda H, Corredig M. The stabilizing behaviour of soybean soluble polysaccharide and pectin in acidified milk beverages. Int Dairy J. 2006;16:361–369. doi: 10.1016/j.idairyj.2005.01.014. [DOI] [Google Scholar]
- Pang Z, Deeth H, Sharma R, Bansal N. Effect of addition of gelatin on the rheological and microstructural properties of acid milk protein gels. Food Hydrocoll. 2015;43:340–351. doi: 10.1016/j.foodhyd.2014.06.005. [DOI] [Google Scholar]
- Peng X, Guo S. Texture characteristics of soymilk gels formed by lactic fermentation: a comparison of soymilk prepared by blanching soybeans under different temperatures. Food Hydrocoll. 2015;43:58–65. doi: 10.1016/j.foodhyd.2014.04.034. [DOI] [Google Scholar]
- Ramchandran L, Shah NP. Effect of exopolysaccharides on the proteolytic and angiotensin-I converting enzyme-inhibitory activities and textural and rheological properties of low-fat yogurt during refrigerated storage. J Dairy Sci. 2009;92:895–906. doi: 10.3168/jds.2008-1796. [DOI] [PubMed] [Google Scholar]
- Rawson HL, Marshall VM. Effect of ‘ropy’ strains of Lactobacillus delbrueckii ssp. bulgaricus and Streptococcus thermophilus on rheology of stirred yogurt. Int J Food Sci Technol. 1997;32:213–220. doi: 10.1046/j.1365-2621.1997.00395.x. [DOI] [Google Scholar]
- Renard D, Velde FVD, Visschers RW. The gap between food gel structure, texture and perception. Food Hydrocoll. 2006;20:423–431. doi: 10.1016/j.foodhyd.2005.10.014. [DOI] [Google Scholar]
- Sahan N, Yasar K, Hayaloglu AA. Physical, chemical and flavour quality of non-fat yogurt as affected by a β-glucan hydrocolloidal composite during storage. Food Hydrocoll. 2008;22:1291–1297. doi: 10.1016/j.foodhyd.2007.06.010. [DOI] [Google Scholar]
- Şanlı T, Sezgin E, Deveci O, Şenel E, Benli M. Effect of using transglutaminase on physical, chemical and sensory properties of set-type yoghurt. Food Hydrocoll. 2011;25:1477–1481. doi: 10.1016/j.foodhyd.2010.09.028. [DOI] [Google Scholar]
- Seenivasagam R, Hemavathi K, Sivakumar G, Niranjan V. Discovering novel carriers for oral insulin tablets: a pharmacoinformatics approach. Int J Bioinform Res Appl. 2013;9:184–206. doi: 10.1504/IJBRA.2013.052443. [DOI] [PubMed] [Google Scholar]
- Shyu YS, Hwang JY, Hsu CK. Improving the rheological and thermal properties of wheat dough by the addition of γ-polyglutamic acid. LWT Food Sci Technol. 2008;41:982–987. doi: 10.1016/j.lwt.2007.06.015. [DOI] [Google Scholar]
- Spagnuolo PA, Dalgleish DG, Goff H, Morris ER. Kappa-carrageenan interactions in systems containing casein micelles and polysaccharide stabilizers. Food Hydrocoll. 2005;19:371–377. doi: 10.1016/j.foodhyd.2004.10.003. [DOI] [Google Scholar]
- Sun NX, Liang Y, Yu B, Tan CP, Cui B. Interaction of starch and casein. Food Hydrocoll. 2016;60:572–579. doi: 10.1016/j.foodhyd.2016.04.029. [DOI] [Google Scholar]
- Wang G, Cui B, Tan R, Li T. Research progress of stabilization of casein micelles in yogurt systems by modified starch. J Shandong Inst Light Ind. 2012;26:11–14. doi: 10.3969/j.issn.1004-4280.2012.02.005. [DOI] [Google Scholar]
- Yang N, Wang Y, Zhang Q, Chen L, Zhao Y. γ-Polyglutamic acid mediated crosslinking PNIPAAm-based thermo/pH-responsive hydrogels for controlled drug release. Polym Degrad Stab. 2017 doi: 10.1016/j.polymdegradstab.2017.07.028. [DOI] [Google Scholar]
- Yu HY, Wang L, McCarthy KL. Characterization of yogurts made with milk solids nonfat by rheological behavior and nuclear magnetic resonance spectroscopy. J Food Drug Anal. 2016;24:804–812. doi: 10.1016/j.jfda.2016.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang S, Hsieh FH, Vardhanabhuti B. Acid-induced gelation properties of heated whey protein–pectin soluble complex (part I): effect of initial pH. Food Hydrocoll. 2014;36:76–84. doi: 10.1016/j.foodhyd.2013.07.029. [DOI] [Google Scholar]
- Zhao M, Wang M, Chen Z, Wang R. Study on the coaction of agar, electrolyte and food gum. Food Ferment Ind. 1995;1:1–7. [Google Scholar]