Abstract
Invariant natural killer T (iNKT) cells are activated at sites of local tissue injury, or globally during vaso-occlusive episodes of sickle cell disease (SCD). Tissue damage stimulates production of CD1d-restricted lipid antigens that activate iNKT cells to produce Th1- and Th2-type cytokines. Here, we show that circulating iNKT cells in SCD patients express elevated levels of the ectonucleoside triphosphate diphosphosphohydrolase, CD39, as well the adenosine A2A receptor (A2AR). We also investigated the effects of stimulating cultured human iNKT cells on the expression of genes involved in the regulation of purinergic signaling. iNKT cell stimulation caused induction of ADORA2A, P2RX7, CD38, CD39, ENPP1, CD73, PANX1, and ENT1. Transcription of ADA, which degrades adenosine, was reduced. Induction of CD39 mRNA was associated with increased ecto-ATPase activity on iNKT cells that was blocked by POM1. Exposure of iNKT cells to A2AR agonists during stimulation reduced production of IFN-γ and enhanced production of IL-13 and CD39. Based on these findings, we define “purinergic Th2-type cytokine bias” as an antiinflammatory purinergic response to iNKT cell stimulation resulting from changes in the transcription of several genes involved in purine release, extracellular metabolism, and signaling.
Keywords: Immunology, Inflammation
Keywords: Innate immunity, NK T cells
Stimulation of iNKT cells causes a gradual increase in the expression of purinergic signaling molecules that enhance ATP release, metabolism, and anti-inflammatory adenosine signaling.
Introduction
Invariant natural killer T (iNKT) cells are activated by CD1d-restricted lipid antigens (1, 2) or various cytokines such as IL-12 and IL-18 (3–7), as illustrated in Supplemental Figure 1 (supplemental material available online with this article; https://doi.org/10.1172/jci.insight.91954DS1). Lipid antigens are produced by some bacteria (8, 9), or by injured host tissues (10, 11), and stimulate invariant Vα24Jα18 TCRs that are found on iNKT cells. In individuals with sickle cell disease (SCD), periodic vaso-occlusive episodes are associated with widely disseminated tissue ischemia in response to sickled RBCs. This causes tissue injury and iNKT cell stimulation. Hence, iNKT cells derived from SCD patients are useful for investigating their response to ischemic injury. When acutely stimulated, the majority of human iNKT cells produce a combination of Th1- and Th2-type cytokines (12). The composition of cytokines is sensitive to the structure of the activating lipid antigen (13) and to environmental costimulatory factors that can tip TCR-mediated cytokine responses between antiinflammatory and proinflammatory (14). This plasticity is functionally important. Acute activation of iNKT cells during tissue ischemia-reperfusion injury can enhance tissue injury (10, 11) but stimulation of iNKT cells with αGalCer prior to ischemia modifies subsequent cytokine responses and reduces injury (15, 16). The results of the current study indicate that changes in cytokine production over time following iNKT cell stimulation are associated with changes in purine release, expression of ectoenzymes that metabolize purine nucleotides, and expression of A2ARs.
ATP is released from dead, dying, or in some cases activated cells in inflamed tissues and tumors (17). In the extracellular space, ATP is generally excitatory to most cells by binding to various P2X and P2Y receptors that usually function to elevate intracellular Ca2+ (18). Ecto-ATP is degraded by ectonucleotidases on the surface of cells. iNKT cells have been shown to express CD39 (19) and CD73 (19) as well as P2X7 receptors (18, 20) that respond to ATP by opening nonselective cation channels (21). In mammalian cells, P2X7 receptors can complex with pannexin 1 (PANX1). Prolonged activation by high amounts of ATP stimulates the formation of P2X7R–pannexin 1 pores that are large enough to release nucleotides, including ATP and NAD+ (22, 23). Extracellular ATP is metabolized to ADP and AMP by CD39. Ecto-NAD+ also can be converted to AMP by the combination of CD38 and ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1) (24). AMP is converted to adenosine primarily by ecto-CD73 (25). We have shown in previous studies that adenosine or selective A2AR agonists reduce tissue injury in response to ischemia-reperfusion injury (10) or SCD (26) by binding to A2AR receptors on iNKT cells. In the current study, we describe induction of multiple purinergic genes involved in ATP/NAD+ release, metabolism, and signaling in stimulated iNKT cells. iNKT cell stimulation enhances transcription of PANX1, P2X7R, CD38, CD39, ENPP1, CD73, ENT1, and ADORA2A mRNAs. Adenosine produced in response to the release and metabolism of ATP and NAD+ engages the Gs-coupled A2AR to activate PKA, reduces the expression of IFN-γ, and enhances the expression of IL-13 and CD39. The induction of this purinergic signaling program may be responsible in part for a gradual shift by activated iNKT cells toward PKA-dependent Th2-type cytokine bias.
Results
Human SCD is associated with elevated expression of CD39 on activated iNKT cells.
Inflammatory effects of ischemia reperfusion-injury are mediated in part by iNKT cell stimulation, as first demonstrated in ischemic liver (10) and subsequently in other ischemic tissues (11, 27). In order to study human iNKT cells, Ab 6B11 is useful to detect the Vα24Jα18 CDR3 loop found on the TCR of iNKT cells and not on conventional T cells (28, 29). Invariant NKT cells also can be detected with αGalCer-loaded CD1d tetramers. Supplemental Figure 2 confirms that Ab 6B11 and CD1d tetramers detect a similar population of human iNKT cells. We previously found that a large fraction of 6B11+ circulating iNKT cells, but not conventional T cells, collected from SCD patients during vaso-occlusive episodes express high levels of the NF-κB activation marker phospho-NF-κB p65 (Ser 536) and high levels of A2ARs (30–32).
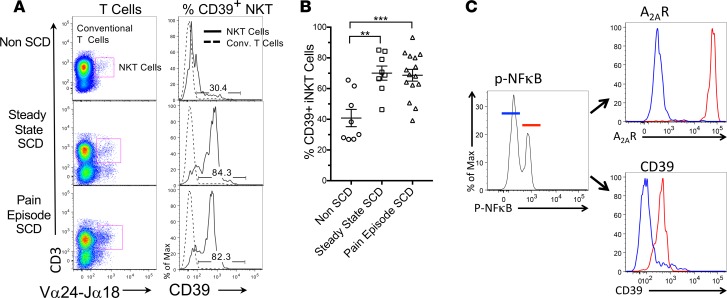
Since most extracellular adenosine is produced from adenine nucleotides by ectoenzymes, we sought to determine if the ecto-ATPase, CD39, which converts ATP and ADP to AMP and is found on iNKT cells (19), is induced as a consequence of SCD. We examined circulating iNKT cells from 8 healthy controls as well as 8 adult SCD patients at steady state and 13 adult SCD patients during acute vaso-occlusive episodes. The mean age of SCD subjects was 30 years. Roughly 50% of SCD subjects were male, 87% had the HbSS genotype, and 87% were prescribed hydroxyurea. Figure 1, A and B show that SCD is associated with a significant increase in CD39 expression on circulating iNKT cells at steady state or during vaso-occlusive episodes. Conventional T cells are not affected (dashed lines in Figure 1A). Figure 1C illustrates that in SCD patients, the same iNKT cells that express phospho-NF-κB p65 (ser 536) also express high levels of the A2AR and CD39. These findings suggest that the concordant induction of ADOR2A and CD39 on activated iNKT cells may function together to increase adenosine production and signaling and limit iNKT cell activation in SCD patients.
Figure 1. CD39 expression on iNKT cells of SCD patients at steady state and during vaso-occlusive crises.
(A) iNKT cells in blood from patients with sickle cell disease (SCD) at steady state (in the absence of vaso-occlusive pain), or during an acute vaso-occlusive pain episode, or cells from normal controls were identified by flow cytometry as 6B11+(Vα14Jα24+) CD3+ cells. Conventional T cells were identified as 6B11–CD3+ (left panels). Histograms of CD39 fluorescence in iNKT cells (solid lines) and conventional T cells (dashed lines) are shown in the right panels. (B) Summary data showing the frequencies of CD39hi iNKT cells from healthy adults (n = 8) and SCD subjects at steady state (n = 8) or during vaso-occlusive crises (n = 15). **P < 0.01, ***P < 0.001 by ANOVA and Bonferroni’s post hoc correction. (C) iNKT cells from a single SCD patient were gated on low (blue) or high (red) expression of phospho-NF-κB p65 (Ser536) and histograms of the expression of the adenosine A2A receptor (A2AR) and CD39 in each NF-κB gate are shown (representative of 4 experiments).
Effects of activating cultured human iNKT cells.
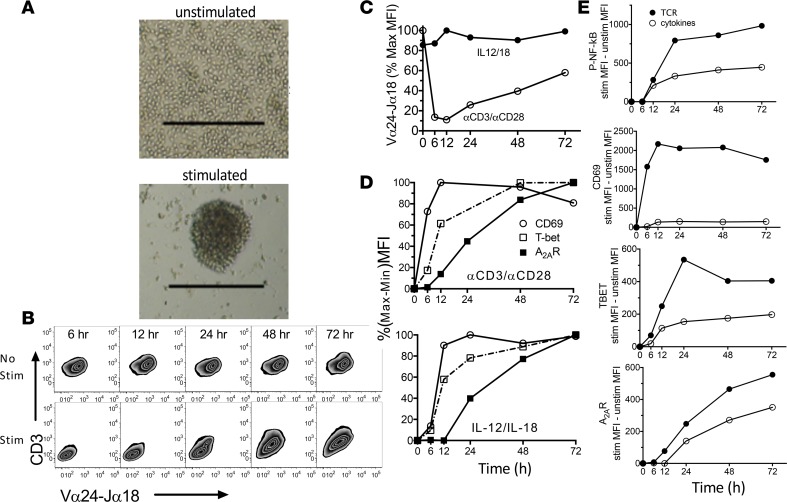
A limitation of studying circulating iNKT cells in vivo is that changes in the characteristics of these cells may be skewed because stimulated cells leave the circulation. In order to investigate the effects of stimulating iNKT cells on purinergic gene expression in a more controlled setting, we next examined the consequences of stimulating purified cultured human cells with anti-CD3 (αCD3) and αCD28 Abs to engage the invariant TCR, or with cytokines (IL-12 and IL-18) in the absence of TCR engagement. We evaluated the magnitudes and kinetics of induction of various activation markers in response to different modes of stimulation. TCR engagement was associated with iNKT cell aggregation (Figure 2A) and rapid downregulation of TCRs and CD3 on the surface of iNKT cells (Figure 2, B and C), whereas activation with cytokines had no effect on cell surface TCR expression (Figure 2C). In order to readily compare the kinetics of increased expression of CD69, T-bet, and A2AR in response to different modes of stimulation, we measured expression kinetics normalized to maximal responses (Figure 2D). These data show that the speed of increased expression is CD69 > T-bet, p-NF-κB >> A2AR. The magnitude of activation of NF-κB as measured by phosphorylation of the p65 subunit (30) was about twice as great in response to TCR as to cytokine stimulation, and a similar difference was noted in the maximal expression of T-bet and the A2AR (Figure 2E). These findings are consistent with the possibility that NF-κB mediates the induction of TBET and ADORA2A, as suggested previously based on the effects of NF-κB inhibitors (30). In contrast to T-bet and the A2AR, maximal expression of CD69 in response to IL-12 and IL-18 was small (8% relative to TCR activation, Figure 2E). Moreover, CD69 expression increased more rapidly than p-NF-κB. These findings suggest that NF-κB activation is not responsible for increasing the expression of CD69 but may mediate increased T-bet and A2AR expression.
Figure 2. Kinetics of changes in the expression of activation markers following stimulation of cultured human iNKT cells.
(A) Images of cultured human iNKT cells after 29 hours of incubation without stimulation or during stimulation with αCD3 and αCD28 Abs as described in the Methods. Scale bars: 250 μm. (B and C) FACS analysis reveals reduced immunofluorescence of CD3 and the invariant TCR (Vα24-Jα18) over time following iNKT cell stimulation by αCD3/αCD28 (Stim) and lack of change in response to IL-12 and IL-18. (D) Kinetics of changes in CD69, T-bet, and adenosine A2A receptor (A2AR) expression over time in response to αCD3/αCD28 or IL-12/IL-18 normalized to the maximal response of each marker. (E) Absolute changes in phospho-NF-κB p65, CD69, T-bet, and A2AR immunofluorescence over time in response to αCD3/αCD28 (TCR) or IL-12/IL-18 (cytokines).
Changes in the transcription of several purinergic genes in response to iNKT cell stimulation.
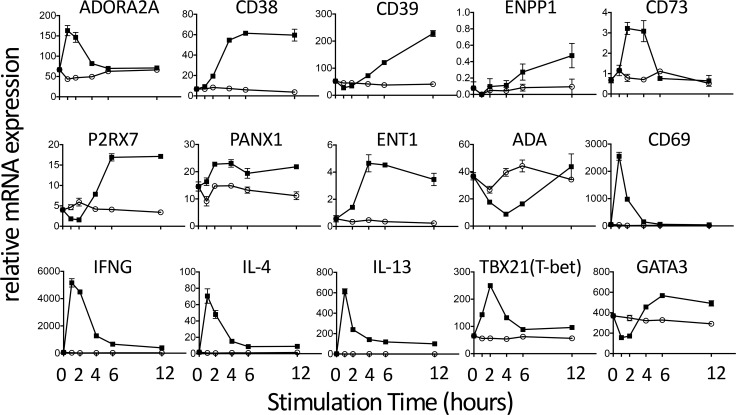
Stimulation of iNKT cells is associated with induction not only of CD39, T-bet, and A2AR, but also of Th1-type (IFN-γ) and Th2-type (IL-4 and IL-13) cytokines (Figure 3). iNKT cell stimulation also evoked a transient increase in TBET and a biphasic effect on GATA3 transcription. The responses of TBET and GATA3 parallel their interactions in Th1 cells where an important function of T-bet is to negatively regulate GATA3 (33). We also examined the effects of iNKT cell activation on the kinetics of expression of transcripts for the 4 adenosine receptor subtypes, A1, A2A, A2B, and A3, as well as various other purinergic genes. Among the adenosine receptor subtypes, the A2AR transcript was more abundant than transcripts for the other adenosine receptors in unstimulated iNKT cells or in cells stimulated with αCD3 (Supplemental Figure 3). These data suggest that the A2AR is the predominant receptor responsible for adenosine signaling in unstimulated or stimulated human iNKT cells. As shown in Figure 3, stimulation of cultured human iNKT cells resulted in a transient increase in ADORA2A and CD73 and more persistent increases in CD38, CD39, PANX1, ENPP1, and ENT1. iNKT cell activation caused a transient reduction of P2X7R transcript for 2 hours, followed by an increase in transcription over the next 4 hours. A rapid reduction in P2X7 receptor expression is expected to reduce the response to extracellular ATP to enhance Ca2+ entry through the P2X7R ligand-gated cation channel, especially if expression of CD39 activity is increased at the same time and lowers extracellular ATP. In addition to mediating Ca2+ entry, P2X7 receptors can form heterodimers with pannexin 1 to produce pores that can conduct ATP and other abundant intracellular small molecules out of cells. Hence, the upregulation of P2X7R under conditions when CD39 and CD73 expression is also elevated may lead to an increase in extracellular ATP-dependent adenosine production. Transcription of ADA, an enzyme that degrades adenosine, is decreased in response to iNKT cell stimulation. This is expected to preserve adenosine and enhance adenosine-mediated signaling. Another purinergic transcript that is strongly induced by iNKT cell stimulation is ENT1, a transporter that equilibrates adenosine across cell membranes. Although the induction of this enzyme is not expected to increase extracellular adenosine derived from ectonucleotides, its induction in cells that are releasing ATP may be needed as a means of facilitating purine salvage via adenosine reuptake. In sum, the effect of iNKT cell stimulation on the transcription of several purinergic genes is predicted to be accelerated ATP release and degradation, increased extracellular adenosine accumulation, and increased antiinflammatory A2AR signaling.
Figure 3. Kinetics of changes in the transcription of purinergic genes, cytokines, and transcription factors following activation of cultured human iNKT cells.
Cells were collected in RLT lysis buffer (Qiagen) at various times after activation by TCR engagement (5 μg/ml plate-bound αCD3 Ab) and transcripts were quantified by RT-PCR. The results are representative of duplicate experiments. Gene names and corresponding proteins are: ADORA2A, adenosine A2A receptor; CD38, cyclic ADP-ribose hydrolase 1/NAD+ nucleosidase; CD39, ectonucleoside triphosphate diphosphohydrolase 1; ENPP1, ectonucleotide pyrophosphatase/phosphodiesterase 1; CD73, ecto-5-nucleotidase; P2RX7, P2X7 receptor; PANX1, pannexin 1; ENT1, equilibrative nucleoside transporter 1; ADA (ADA1), adenosine deaminase; CD69, early lymphocyte activation antigen; IFNG, interferon γ; TBX21 (TBET), T-box 21; GATA3, GATA binding protein 3.
The mode of iNKT cell stimulation and A2AR signaling influence cytokine production.
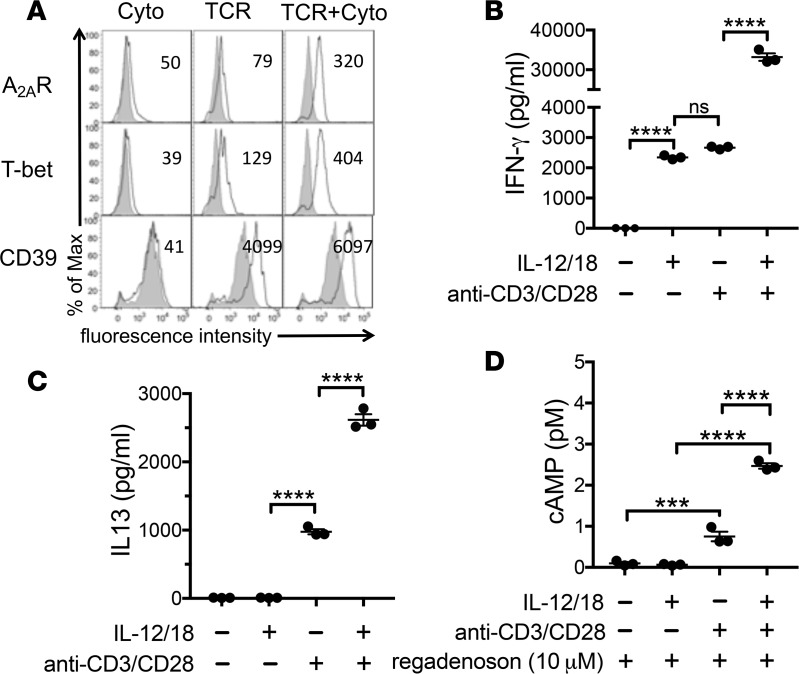
As noted in previous studies, the combination of TCR engagement and stimulation with IL-12 and IL-18 have additive effects on the expression of T-bet, CD39, the Th1 cytokine IFN-γ, and the Th2 cytokine IL-13 (Figure 4, A–C). Stimulation with IL-12/IL-18 alone evoked some production of IFN-γ, but failed to stimulate production of IL-13. Nevertheless, IL-12/IL-18 costimulation with TCR engagement enhanced the production of IFN-γ and IL-13. We next sought to determine how A2AR signaling in iNKT cells is affected by different modes of stimulation. We found that the combination of IL-12/IL-18 and TCR engagement for 48 hours was more efficient than either stimulus alone at increasing A2AR expression (Figure 4A) and at increasing A2AR functional activity assessed by measuring cAMP production in response to the A2AR agonist, regadenoson (Figure 4D). In sum, maximal induction in iNKT cells of IFN-γ, IL-13, or functional A2ARs requires a combination of TCR and IL-12/IL-18 signaling.
Figure 4. Effects of mode of stimulation on activation marker expression and function of iNKT cells.
Cultured iNKT cells were stimulated for 48 hours by 50 ng/ml each of IL-12/IL-18 cytokines (Cyto), TCR engagement (5 μg/ml plate-bound αCD3 and 2 μg/ml soluble αCD28 Abs) or a combination of both. (A) Shaded histograms show immunofluorescence of unstimulated iNKT cells, solid lines show histograms of activation markers on iNKT cells stimulated by Cyto, TCR, or TCR+Cyto. (B and C) Effects of mode of stimulation on iNKT cell production of IFN-γ or IL-13 measured by ELISA during 24 hours of stimulation. (D) Effect of mode of stimulation for 48 hours on subsequent cAMP accumulation in iNKT cells stimulated for 15 minutes with regadenoson, 1 U/ml adenosine deaminase, and 10 μM rolipram. The results are representative of triplicate experiments. ***P < 0.001, ****P < 0.0001. ns, not significant.
Effects of cAMP on IFN-γ, IL-13, and CD39 expression by iNKT cells.
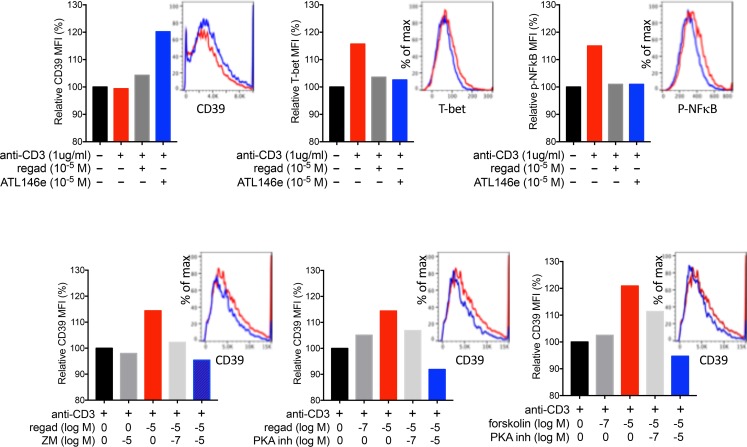
The A2AR is coupled to Gs and cAMP production. Inclusion of the A2AR agonist regadenoson during concurrent TCR engagement produced a dose-dependent decrease in IFN-γ production, but interestingly, a dose-dependent increase in IL-13 production (Figure 5). We also examined the effects of regadenoson on CD39 expression based on a prior report showing that in macrophages, the PKA/CREB pathway enhances CD39 expression (34). As shown in Figure 6A, coactivation of the A2AR during TCR activation enhanced the expression of CD39, while suppressing the expression of phospho-NF-κB p65 (Ser536) and T-bet. In order to confirm that regadenoson acts on iNKT cells via the A2AR to produce cyclic AMP and activate protein kinase A (PKA), we examined another A2AR agonist, ATL-146e (35); an A2AR antagonist, ZM241385; an activator of adenylyl cyclase, forskolin; and a cell-permeant inhibitor of PKA, KT-5720. Figure 6B shows that changes in CD39 expression are mediated by A2AR activation, cAMP production, and PKA activation. Supplemental Figure 4 illustrates the molecular targets of these agents.
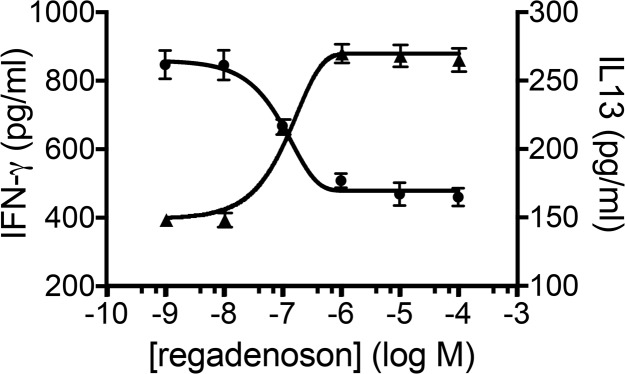
Figure 5. Activation of the A2AR differentially affects IFN-γ and IL-13 production by stimulated iNKT cells.
Adenosine A2A receptor (A2AR) activation with regadenoson during stimulation with αCD3 Ab produces a dose-dependent inhibition of 24-hour IFN-γ production (ED50 = 2.1 μM) and enhancement of IL-13 production (ED50 = 5.2 μM). Each data point is the mean ± SEM of triplicate determinations. The results are representative of triplicate experiments.
Figure 6. iNKT cell CD39 expression is increased by A2AR activation due to activation of protein kinase A (PKA).
(A) Geometric mean fluorescence intensities (MFIs) of CD39, T-bet, and phospho-NF-κB p65 (Ser536) determined by flow cytometry of 4,000–10,000 cultured iNKT cell in response to 48 hours of stimulation with plate-bound αCD3 Ab (1 μg/ml) and the A2AR agonists, regadenoson or ATL146e. Histograms correspond to bars of the same color. (B) The effects of regadenoson, the A2AR antagonist ZM241385 (ZM), the PKA inhibitor KT5720, and the adenylyl cyclase activator forskolin on CD39 expression on iNKT cells stimulated with αCD3. The results are representative of triplicate experiments.
Effects of iNKT cell stimulation on the ecto-ATPase activity of iNKT cells.
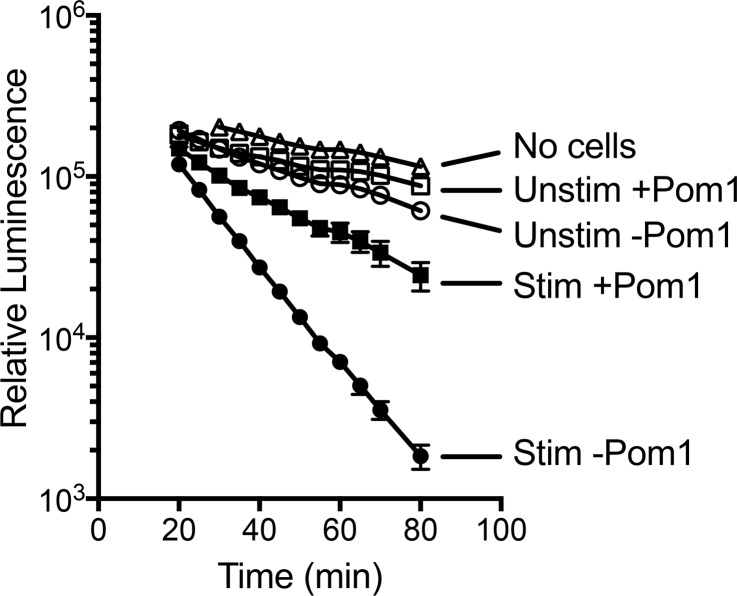
In order to determine if enhanced CD39 transcription and immunoreactivity in iNKT cells is associated with increased CD39 enzymatic activity, we used a firefly luciferase assay to measure ATP outside cultured iNKT cells. As illustrated in Figure 7, following 48 hours of stimulation with αCD3 and αCD28 Abs, iNKT cells exhibited higher ATPase activity than unstimulated cells, and most of the increased activity was inhibited by 50 μM POM1 (Na6O39W12•H2O), an ectonucleotide triphosphate diphosphohydrolase inhibitor with some selectivity for CD39 (36). The increase in ATPase activity was not due to iNKT cell proliferation, since the protein content of cells following incubation with or without stimulation was found to be minimally changed (<10%, data not shown). A low level of ATPase activity noted in the absence of cells is attributable to the ATPase activity of luciferin/luciferase. The data confirm that increased CD39 transcription and immunostaining on stimulated iNKT cells is associated with increased enzymatic activity.
Figure 7. Ecto-ATPase activity on human iNKT cells.
CD4+ NKT cells (50,000) in triplicate wells were cultured with or without αCD3/αCD28 Dynabeads for 48 hours at 37°C and assayed for ecto-ATPase activity as described in the Methods. Vehicle or CD39 inhibitor (50 μM POM1) was added to wells before luciferase plus luciferin followed by 10 μM ATP. Luminescence from each well was measured repetitively at room temperature on a SpectraMax M2 plate reader at various times after addition of ATP. The results are representative of 5 experiments.
Discussion
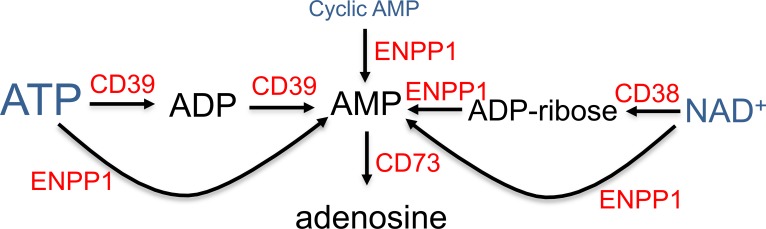
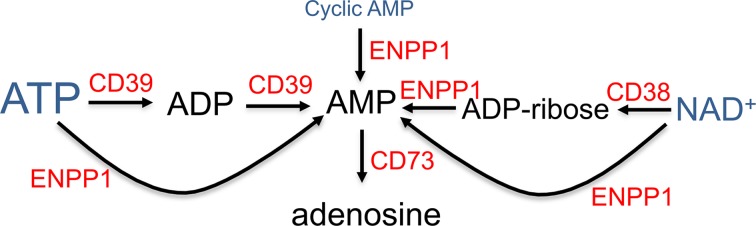
Prior studies have demonstrated that various stimuli increase the transcription and translation of adenosine A2ARs that bind adenosine and function to inhibit proinflammatory cytokine release from iNKT cells or other leukocytes (30, 35, 37, 38). In the current study, we show that iNKT cell stimulation via TCR engagement with αCD3 and αCD28 Abs or cytokine treatment with IL-12 and IL-18 is associated not only with increased expression of A2AR receptors, but also with induction of several other purinergic genes that may function to facilitate adenine nucleotide release and extracellular metabolism to adenosine. These include pannexin 1 and P2X7R, which have been shown to respond to ATP by forming membrane channels large enough to conduct ATP (39), the equilibrative nucleoside transporter ENT1, and several nucleotidases that convert ATP, NAD+, and cyclic AMP to AMP and adenosine (Figure 8).
Figure 8. Diagram of ectoenzymes that produce adenosine.
Adenine nucleotides that are present in all cells (ATP > NAD > cAMP) are released in response to cell necrosis, apoptosis, or various factors that open membrane channels. This diagram shows some of the enzymes that participate in the extracellular conversion of ATP, NAD+, or cyclic AMP to adenosine. CD38, cyclic ADP-ribose hydrolase 1/NAD+ nucleosidase; CD39 (ENTPD1), ectonucleoside triphosphate diphosphohydrolase 1; ENPP1, ectonucleotide pyrophosphatase/phosphodiesterase; CD73 (NT5E), ecto-5-nucleotidase.
Purinergic suppression of iNKT cell activation appears to be important in disease processes inasmuch as iNKT cells have been implicated as mediators of adverse responses to ischemia-reperfusion/transplant injury (10, 31, 40–42) and several autoimmune diseases in which IL-18 concentrations are increased (43). These findings relate to an interesting feature of iNKT cells — their ability to produce diverse cytokines depending in part on environmental cues. Although subsets of NKT1, NKT2, and NKT17 cells that differentiate in the thymus have been defined in mice (44), iNKT cell subsets are not well defined in humans. Rather, the majority of circulating human iNKT cells produce multiple cytokines, and the ratio of Th1-/Th2-type cytokines is known to be influenced by several factors. These include antigen structure, whether the lipid-CD1d complex is found in lipid rafts, and the expression by antigen-presenting cells of costimulatory molecules or cytokines (45). The effect of lipid antigen structure on cytokine responses has important consequences in inflammatory diseases. For example, activation of mouse iNKT cells with αGalCer C20:2 favors Th2-biased cytokine production relative to the closely related compound αGalCer, and treatment with αGalCer C20:2 delays the progression of diabetes in NOD mice (46). Hence, there is great interest in identifying factors that modify cytokine production by the bulk population of mature, differentiated human iNKT cells. The results of the current study suggest that over time after stimulation of human iNKT cells, increased purinergic signaling results in cyclic AMP/PKA–driven Th2-type cytokine bias, e.g., an increase in IL-13 production and a decrease in IFN-γ production.
A2AR agonists may be useful for the treatment of acute inflammation. The data from the current study show that the impact of A2AR signaling on PKA activation and downstream antiinflammatory signaling increases over time. However, when using an A2AR agonist such as regadenoson as a therapy to reduce iNKT cell–mediated type-1 cytokine signaling in response to acute tissue injury, it may be advantageous to begin A2AR treatment as soon as possible in order to optimally inhibit the production of inflammatory cytokines prior to their synthesis and release.
The concentration of extracellular adenosine may be influenced by the expression of P2X7R–pannexin 1 pores that can release ATP and NAD+ (39) as well as the expression of ectoenzymes such as CD38, CD39, and CD73 (47) that generate extracellular adenosine from ATP or NAD+ (48). The concentration of adenosine outside cells, and the cell surface density of A2ARs influence adenosine signaling. In the current study, we show that CD39 expression is increased as a result of iNKT cell activation and also that the expression of the enzyme is increased by activators of PKA. It was shown previously that CD39 transcription and expression are enhanced by the PKA/CREB pathway in macrophages (34) and astrocytes (49), and this pathway is thought to participate in protection from ischemic brain injury (49) and ischemic renal injury (50).
A2AR-mediated PKA activation in mouse iNKT cells was previously found to influence cytokine secretion patterns, causing decreased IFN-γ and increased IL-4, IL-10, and TGF-β production (35, 51). In A2AR-deficient mice, iNKT cells produce only low levels of IL-4, IL-10, and TGF-β (51). Th1-type cytokine production in iNKT cells is also influenced by inhibitors of type 4 and type 7A phosphodiesterases (PDE4 or PDE7A) that elevate cAMP by blocking its degradation (10, 52). Another PDE inhibitor, ibudilast, increases the Th2-/Th1-type cytokine ratio in CD4+ T cells and expands iNKT cells in patients with multiple sclerosis (53).
In addition to A2ARs, T cells express other Gs-coupled receptors that stimulate cAMP production. Calcitonin gene–related peptide (CGRP) was found to suppress IL-2 and IFN-γ production and increase IL-4 production to promote Th2 differentiation by activating the cAMP/PKA pathway during stimulation with αCD3 and αCD28 Abs. IL-4 production and transcriptional activation of Th2-type cytokine mRNAs was also observed in response to the cAMP analogue, dibutyryl-cAMP, in αCD3- and αCD28-stimulated naive T cells. In fact, cAMP/PKA activation in naive T cells stimulated with αCD3 and αCD28 is essential for inducing IL-4 production and promoting Th2 differentiation (54). In Th2 cells, PKA and Jak-STAT signaling have been shown to increase IL-13 secretion and transcription in response to activation by Gs-coupled histamine receptors (55). The mechanism by which PKA produces Th2-type cytokine bias may be due in part to phosphorylation and inactivation of C-terminal Src Kinase (CSK), resulting in inhibition of Lck, and attenuation of the strength of TCR signaling (56).
iNKT cells derived from SCD mice are activated and produce IFN-γ and IFN-γ–inducible chemokines that are important for propagating inflammation and vaso-occlusive injury (31). These responses are attenuated by activation of A2ARs (26, 32). Preconditioning mice with αGalCer was found to protect the liver from ischemia-reperfusion injury by increasing the expression of A2ARs on iNKT cells (16). In the current study, we demonstrate that there is a simultaneous increase in the expression of CD39 and the A2AR on human iNKT cells in response to their activation during vaso-occlusive crises in human SCD. The findings suggest that adenosine signaling may reduce inflammation over time by suppressing IFN-γ production and enhancing the production of Th2-type cytokines by iNKT cells.
The expression on the iNKT cell surface of purinergic molecules is increased in proportion to cell activation: αCD3/αCD28 + IL-12/IL-18 >> αCD3/αCD28 > IL-12/IL-18. On the basis of these results we define “purinergic Th2-type cytokine bias” as an increase in Th2-/Th1-type cytokine ratio due to increased expression on immune cells of P2X7Rs–pannexin 1 channels, ectonucleotidases (CD38, ENPP1, CD39, and CD73), and adenosine A2ARs.
In addition to T cell–intrinsic purinergic regulation, it was previously shown that the phenotypes of DCs also have a strong influence on iNKT cell cytokine production. In mice, the absence of S1P3 receptors on DCs prevents their maturation, evokes a Th2/IL-4 iNKT cell response to antigen presentation, and promotes IL-4–mediated protection from renal ischemia-reperfusion injury (57). Adoptive transfer of DCs treated in vitro with an A2AR agonist protects the kidneys of mice from ischemia-reperfusion injury by regulating DC costimulatory molecules that are important for iNKT cell activation (57). Purinergic signaling is also influenced by chemotaxis of cells that express CD73. Activation of iNKT cells produces chemokines that attract regulatory B cells that express CD73 (58). CD73 is also expressed on mouse Tregs and γδ T cells. Treg cells suppress T effector cells and iNKT cells in part by generating adenosine (59). Activation of γδ T cells significantly lowers their expression of CD73 and this contributes to enhanced proinflammatory activity (60). Hence, purinergic Th2-type cytokine bias may be mediated in part by cell-intrinsic purinergic effects on iNKT cells, and also by purinergic effects on other cells, including DCs, Treg cells, B cells, and γδ T cells. The current study raises the prospect of harnessing purinergic and PKA-dependent signaling to control inflammatory diseases.
Methods
Human iNKT cell culture, purification, and activation.
Ficoll gradient centrifugation was used to prepare peripheral blood mononuclear cells (PBMCs) from normal healthy blood donors. The iNKT cells in PBMCs were expanded by adding 100 ng/ml αGalCer (Funakoshi, KRN7000) and 100 IU/ml IL-2 (NCI) for 12–14 days. Cells were then stained with Live/Dead Aqua (Invitrogen), αCD19 (Invitrogen, clone SJ25-C1), αCD3 (Invitrogen and BD Biosciences), and αVα24Jα18 TCR (eBioscience, 6B11) (28). Live iNKT cells were collected using a BD FACSAria cell sorter and further expanded by coculturing with γ-irradiated (40 Gy) PBMCs pulsed with 100 ng/ml αGalCer, washed and resuspended in medium composed of 47.5% RPMI 1640 with L-glutamine, 25 mM Hepes, and 5.5 μM β-mercaptoethanol, 47.5% AIM V, and 5% heat-inactivated human AB serum supplemented with 100 IU/ml IL-2 for 12–14 days. Prior to their use for experiments, iNKT cells were washed and replated without IL-2 for 18 hours to allow the cells to return toward a resting state. After expansion and washing, 96%–98% of remaining cells were CD3+6B11+ iNKT cells. TCR engagement was achieved by centrifuging cells in medium supplemented with αCD28 (2 μg/ml) (eBioscience, CD28.2) at 200 g for 2 minutes in high-protein-binding plates previously coated overnight at 4°C with αCD3 antibody (5 μg/ml) (eBioscience, OKT3) and washed twice. Activation by cytokines utilized soluble IL-12 (50 ng/ml) and IL-18 (50 ng/ml) (R&D Systems). Cells were further treated with and without the adenosine A2AR agonist regadenoson, or other compounds as indicated.
Flow cytometry.
Invariant NKT cells were identified as live, CD19–, CD3+, Vα24Jα18 TCR+ cells (30). In normal subjects iNKT cells were defined as CD3+6B11+ cells. In some SCD subjects a population of 6B11loCD3lo cells that may include iNKT cells with downregulated TCRs were excluded from analysis because they were difficult to reliably resolve from cells that display nonspecific Ab binding. CD39 was identified with αCD39 (BD Biosciences, A1). NF-κB signaling was assessed by measuring phospho-NF-κB p65 (Ser536) (Cell Signaling Technology, 93H1). Following cell permeabilization, a highly immunoreactive epitope on an intracellular domain of the A2AR was identified with an anti-human monoclonal Ab that we have previously characterized (61) (Santa Cruz Biotechnology, 7F6-G5-A2) conjugated to Alexa Fluor 647. Intracellular cytokines and transcription factors were detected using αIL-4 (BD, 8D4-8), αIFN-γ (eBioscience, 4S.B3), αT-bet (BD, O4-46), and αGATA3 (BD L50-823). Flow cytometry was performed using a BD Bioscience LSRII, and data analyzed using FlowJo software (Tree Star, version 9.6.4).
cAMP assay.
The LANCE Ultra cAMP kit (Perkin-Elmer) was used to quantify cAMP produced by iNKT cells. After 48 hours of activation by TCR engagement, IL-12 and IL-18 treatment, or both, tissue culture plates were centrifuged at 350 g for 4 minutes and the supernatant aspirated from each well. Plated cells were resuspended in 100 μl HBSS/Hepes buffer with BSA (Perkin-Elmer), 1 U/ml adenosine deaminase (Roche), and 10 μM rolipram (Sigma-Aldrich). In some cases, the PKA inhibitor KT5720 was added, or cells were treated with the A2AR agonist regadenoson for 15 minutes at 37°C. We then lysed cells by adding 100 μl 0.6 M perchloric acid and 13 μl 0.36 M potassium carbonate to neutralize the acid. Lysed cells were placed on ice for 1 hour, centrifuged at 10,000 g for 10 minutes, and cAMP in the supernatant was quantified using a SpectraMax M5 plate reader as described by the manufacturer (Molecular Devices).
Enzyme-linked immunosorbent cytokine assays.
After exposing cultured human iNKT cells to different stimuli, supernatants were assayed by enzyme-linked immunosorbent assays (ELISAs) to quantify IL-10, IL-13, or IFN-γ (eBioscience).
Quantitative real-time PCR.
Cultured human iNKT cells were harvested, lysed with RLT Plus lysis buffer (Biolegend), and RNA was extracted using Qiagen Allprep DNA/RNA Micro columns as described by the manufacturer. cDNA was synthesized from RNA samples using Qiagen QuantiTect Reverse Transcription Kits according to the manufacturer’s protocol. Quantitative real-time PCR was performed using TaqMan Gene Expression assays and measured with a Roche LightCycler 480. RNA expression was normalized to RNA polymerase IIA.
Extracellular ATPase activity.
Functional ecto-ATPase activity on iNKT cells was assessed by using an ATP-dependent luciferase assay. Fifty thousand human CD4+ NKT cells in triplicate Corning half-area opaque 96-well plates were cultured in 0.1 ml of RPMI 1640 medium plus 5% human AB serum with or without αCD3/αCD28 Dynabeads for 48 hours at 37°C. Incubation with Dynabeads versus vehicle produced a small reduction in cell protein/well at 48 hours from 12 to 10 μg/well (measured by the Pierce BCA protein assay), so ATPase activity was not normalized to protein. Vehicle or CD39 inhibitor (POM1) (Tocris) was added to wells at a final concentration of 50 μM and incubated for 1 hour at 37°C. Equal volumes (0.1 ml) of assay buffer containing luciferase and luciferin from a Promega CellTiter-Glo Cell Viability Assay kit (Promega) were then added to each well prior to adding 10 μM ATP (Sigma-Aldrich). Luminescence from each well was measured over time at room temperature on the SpectraMax M2 plate reader.
Statistics.
The production of cyclic AMP or cytokines by iNKT cells are plotted as means ± SEM. For the statistical analysis of multiple experimental groups, 1-way ANOVA followed by Bonferonni’s post hoc test was performed using GraphPad Prism for Mac OS X version 6.0e. A P value less than 0.05 was considered significant.
Study approval.
SCD subjects were recruited from the Sickle Cell Clinic or inpatient service at Froedtert Hospital in Milwaukee, Wisconsin, USA. Some were participants in a clinical trial (NCT01788631). iNKT cell samples were shipped overnight at 4°C to the laboratory of Joel Linden. Normal controls were recruited from the La Jolla Institute for Allergy and Immunology. The Human Research Protection Office at the Medical College of Wisconsin and the institutional review board at the La Jolla Institute for Allergy and Immunology approved all study activities and all subjects provided informed consent prior to their participation in the study.
Author contributions
JCY helped with in vitro experiments and helped write the manuscript. GL oversaw flow cytometry, in vitro experiments, data analysis, and FACS. JJF oversaw the collection of blood from SCD patients. JL conceived of experiments and wrote the manuscript.
Supplementary Material
Acknowledgments
The authors thank Cheryl Kim, Kurt Van Gunst, and Lara Nosworthy of the La Jolla Institute Flow Cytometry Core for technical guidance with flow cytometry, and Mitch Kronenberg for his gift of CD1d tetramers. The work was supported by NIH grant R01-HL111969.
Version 1. 09/06/2018
Electronic publication
Footnotes
Conflict of interest: JL owns equity in Adenosine Therapeutics, LLC and Inspyr Therapeutics, Inc. These companies produce drugs targeting adenosine receptors.
Reference information: JCI Insight. 2018;3(17):e91954. https://doi.org/10.1172/jci.insight.91954.
Contributor Information
Gene Lin, Email: glin@lji.org.
Joshua J. Field, Email: joshua.field@bcw.edu.
Joel Linden, Email: jlinden@liai.org.
References
- 1.Dangerfield EM, et al. Species-specific activity of glycolipid ligands for invariant NKT cells. Chembiochem. 2012;13(9):1349–1356. doi: 10.1002/cbic.201200095. [DOI] [PubMed] [Google Scholar]
- 2.Tefit JN, Davies G, Serra V. NKT cell responses to glycolipid activation. Methods Mol Biol. 2010;626:149–167. doi: 10.1007/978-1-60761-585-9_11. [DOI] [PubMed] [Google Scholar]
- 3.Subleski JJ, et al. TCR-dependent and -independent activation underlie liver-specific regulation of NKT cells. J Immunol. 2011;186(2):838–847. doi: 10.4049/jimmunol.1001735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Uchida T, Kinoshita M, Fukasawa M, Habu Y, Shinomiya N, Seki S. IL-18 time-dependently modulates Th1/Th2 cytokine production by ligand-activated NKT cells. Eur J Immunol. 2007;37(4):966–977. doi: 10.1002/eji.200636465. [DOI] [PubMed] [Google Scholar]
- 5.Bourgeois E, et al. The pro-Th2 cytokine IL-33 directly interacts with invariant NKT and NK cells to induce IFN-gamma production. Eur J Immunol. 2009;39(4):1046–1055. doi: 10.1002/eji.200838575. [DOI] [PubMed] [Google Scholar]
- 6.Brennan PJ, et al. Activation of iNKT cells by a distinct constituent of the endogenous glucosylceramide fraction. Proc Natl Acad Sci USA. 2014;111(37):13433–13438. doi: 10.1073/pnas.1415357111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Crowe NY, et al. Glycolipid antigen drives rapid expansion and sustained cytokine production by NK T cells. J Immunol. 2003;171(8):4020–4027. doi: 10.4049/jimmunol.171.8.4020. [DOI] [PubMed] [Google Scholar]
- 8.Taniguchi M, Tashiro T, Dashtsoodol N, Hongo N, Watarai H. The specialized iNKT cell system recognizes glycolipid antigens and bridges the innate and acquired immune systems with potential applications for cancer therapy. Int Immunol. 2010;22(1):1–6. doi: 10.1093/intimm/dxp104. [DOI] [PubMed] [Google Scholar]
- 9.Stanic AK, et al. Another view of T cell antigen recognition: cooperative engagement of glycolipid antigens by Va14Ja18 natural T(iNKT) cell receptor [corrected] J Immunol. 2003;171(9):4539–4551. doi: 10.4049/jimmunol.171.9.4539. [DOI] [PubMed] [Google Scholar]
- 10.Lappas CM, Day YJ, Marshall MA, Engelhard VH, Linden J. Adenosine A2A receptor activation reduces hepatic ischemia reperfusion injury by inhibiting CD1d-dependent NKT cell activation. J Exp Med. 2006;203(12):2639–2648. doi: 10.1084/jem.20061097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li L, et al. NKT cell activation mediates neutrophil IFN-gamma production and renal ischemia-reperfusion injury. J Immunol. 2007;178(9):5899–5911. doi: 10.4049/jimmunol.178.9.5899. [DOI] [PubMed] [Google Scholar]
- 12.Fox L, Hegde S, Gumperz JE. Natural killer T cells: innate lymphocytes positioned as a bridge between acute and chronic inflammation? Microbes Infect. 2010;12(14-15):1125–1133. doi: 10.1016/j.micinf.2010.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Birkholz AM, et al. A novel glycolipid antigen for NKT cells that preferentially induces IFN-γ production. J Immunol. 2015;195(3):924–933. doi: 10.4049/jimmunol.1500070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gumperz JE. CD1d-restricted “NKT” cells and myeloid IL-12 production: an immunological crossroads leading to promotion or suppression of effective anti-tumor immune responses? J Leukoc Biol. 2004;76(2):307–313. doi: 10.1189/jlb.0104038. [DOI] [PubMed] [Google Scholar]
- 15.Homma T, et al. Activation of invariant natural killer T cells by α-galactosylceramide ameliorates myocardial ischemia/reperfusion injury in mice. J Mol Cell Cardiol. 2013;62:179–188. doi: 10.1016/j.yjmcc.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 16.Cao Z, et al. Preactivation of NKT cells with alpha-GalCer protects against hepatic ischemia-reperfusion injury in mouse by a mechanism involving IL-13 and adenosine A2A receptor. Am J Physiol Gastrointest Liver Physiol. 2009;297(2):G249–G258. doi: 10.1152/ajpgi.00041.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pellegatti P, Raffaghello L, Bianchi G, Piccardi F, Pistoia V, Di Virgilio F. Increased level of extracellular ATP at tumor sites: in vivo imaging with plasma membrane luciferase. PLoS One. 2008;3(7):e2599. doi: 10.1371/journal.pone.0002599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cekic C, Linden J. Purinergic regulation of the immune system. Nat Rev Immunol. 2016;16(3):177–192. doi: 10.1038/nri.2016.4. [DOI] [PubMed] [Google Scholar]
- 19.Beldi G, et al. Natural killer T cell dysfunction in CD39-null mice protects against concanavalin A-induced hepatitis. Hepatology. 2008;48(3):841–852. doi: 10.1002/hep.22401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kawamura H, Aswad F, Minagawa M, Govindarajan S, Dennert G. P2X7 receptors regulate NKT cells in autoimmune hepatitis. J Immunol. 2006;176(4):2152–2160. doi: 10.4049/jimmunol.176.4.2152. [DOI] [PubMed] [Google Scholar]
- 21.Khakh BS, North RA. P2X receptors as cell-surface ATP sensors in health and disease. Nature. 2006;442(7102):527–532. doi: 10.1038/nature04886. [DOI] [PubMed] [Google Scholar]
- 22.Scemes E, Suadicani SO, Dahl G, Spray DC. Connexin and pannexin mediated cell-cell communication. Neuron Glia Biol. 2007;3(3):199–208. doi: 10.1017/S1740925X08000069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Locovei S, Bao L, Dahl G. Pannexin 1 in erythrocytes: function without a gap. Proc Natl Acad Sci USA. 2006;103(20):7655–7659. doi: 10.1073/pnas.0601037103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ferretti E, Horenstein AL, Canzonetta C, Costa F, Morandi F. Canonical non-canonical adenosinergic pathways. Immunol Lett. doi: 10.1016/j.imlet.2018.03.007. doi: 10.1016/j.imlet.2018.03.007. [published online ahead of print March 14, 2018]. [DOI] [PubMed] [Google Scholar]
- 25.Antonioli L, Pacher P, Vizi ES, Haskó G. CD39 and CD73 in immunity and inflammation. Trends Mol Med. 2013;19(6):355–367. doi: 10.1016/j.molmed.2013.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wallace KL, Linden J. Adenosine A2A receptors induced on iNKT and NK cells reduce pulmonary inflammation and injury in mice with sickle cell disease. Blood. 2010;116(23):5010–5020. doi: 10.1182/blood-2010-06-290643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sharma AK, et al. Natural killer T cell-derived IL-17 mediates lung ischemia-reperfusion injury. Am J Respir Crit Care Med. 2011;183(11):1539–1549. doi: 10.1164/rccm.201007-1173OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Exley MA, et al. Selective activation, expansion, and monitoring of human iNKT cells with a monoclonal antibody specific for the TCR alpha-chain CDR3 loop. Eur J Immunol. 2008;38(6):1756–1766. doi: 10.1002/eji.200737389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Montoya CJ, et al. Characterization of human invariant natural killer T subsets in health and disease using a novel invariant natural killer T cell-clonotypic monoclonal antibody, 6B11. Immunology. 2007;122(1):1–14. doi: 10.1111/j.1365-2567.2007.02647.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lin G, et al. NF-κB is activated in CD4+ iNKT cells by sickle cell disease and mediates rapid induction of adenosine A2A receptors. PLoS One. 2013;8(10):e74664. doi: 10.1371/journal.pone.0074664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wallace KL, et al. NKT cells mediate pulmonary inflammation and dysfunction in murine sickle cell disease through production of IFN-gamma and CXCR3 chemokines. Blood. 2009;114(3):667–676. doi: 10.1182/blood-2009-02-205492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Field JJ, et al. Sickle cell vaso-occlusion causes activation of iNKT cells that is decreased by the adenosine A2A receptor agonist regadenoson. Blood. 2013;121(17):3329–3334. doi: 10.1182/blood-2012-11-465963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Usui T, et al. T-bet regulates Th1 responses through essential effects on GATA-3 function rather than on IFNG gene acetylation and transcription. J Exp Med. 2006;203(3):755–766. doi: 10.1084/jem.20052165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liao H, Hyman MC, Baek AE, Fukase K, Pinsky DJ. cAMP/CREB-mediated transcriptional regulation of ectonucleoside triphosphate diphosphohydrolase 1 (CD39) expression. J Biol Chem. 2010;285(19):14791–14805. doi: 10.1074/jbc.M110.116905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lappas CM, Rieger JM, Linden J. A2A adenosine receptor induction inhibits IFN-gamma production in murine CD4+ T cells. J Immunol. 2005;174(2):1073–1080. doi: 10.4049/jimmunol.174.2.1073. [DOI] [PubMed] [Google Scholar]
- 36.Lee SY, et al. Polyoxometalates--potent and selective ecto-nucleotidase inhibitors. Biochem Pharmacol. 2015;93(2):171–181. doi: 10.1016/j.bcp.2014.11.002. [DOI] [PubMed] [Google Scholar]
- 37.Murphree LJ, Sullivan GW, Marshall MA, Linden J. Lipopolysaccharide rapidly modifies adenosine receptor transcripts in murine and human macrophages: role of NF-kappaB in A(2A) adenosine receptor induction. Biochem J. 2005;391(Pt 3):575–580. doi: 10.1042/BJ20050888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bruzzese L, et al. NF-κB enhances hypoxia-driven T-cell immunosuppression via upregulation of adenosine A(2A) receptors. Cell Signal. 2014;26(5):1060–1067. doi: 10.1016/j.cellsig.2014.01.024. [DOI] [PubMed] [Google Scholar]
- 39.Iglesias R, et al. P2X7 receptor-Pannexin1 complex: pharmacology and signaling. Am J Physiol, Cell Physiol. 2008;295(3):C752–C760. doi: 10.1152/ajpcell.00228.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fahrner R, Dondorf F, Ardelt M, Settmacher U, Rauchfuss F. Role of NK, NKT cells and macrophages in liver transplantation. World J Gastroenterol. 2016;22(27):6135–6144. doi: 10.3748/wjg.v22.i27.6135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang C, et al. Rapamycin protects kidney against ischemia reperfusion injury through recruitment of NKT cells. J Transl Med. 2014;12:224. doi: 10.1186/s12967-014-0224-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kumar V. NKT-cell subsets: promoters and protectors in inflammatory liver disease. J Hepatol. 2013;59(3):618–620. doi: 10.1016/j.jhep.2013.02.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Boraschi D, Dinarello CA. IL-18 in autoimmunity: review. Eur Cytokine Netw. 2006;17(4):224–252. [PubMed] [Google Scholar]
- 44.Drennan MB, et al. The thymic microenvironment differentially regulates development and trafficking of invariant NKT cell sublineages. J Immunol. 2014;193(12):5960–5972. doi: 10.4049/jimmunol.1401601. [DOI] [PubMed] [Google Scholar]
- 45.Bai L, et al. Distinct APCs explain the cytokine bias of α-galactosylceramide variants in vivo. J Immunol. 2012;188(7):3053–3061. doi: 10.4049/jimmunol.1102414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Forestier C, et al. Improved outcomes in NOD mice treated with a novel Th2 cytokine-biasing NKT cell activator. J Immunol. 2007;178(3):1415–1425. doi: 10.4049/jimmunol.178.3.1415. [DOI] [PubMed] [Google Scholar]
- 47.Bono MR, Fernández D, Flores-Santibáñez F, Rosemblatt M, Sauma D. CD73 and CD39 ectonucleotidases in T cell differentiation: Beyond immunosuppression. FEBS Lett. 2015;589(22):3454–3460. doi: 10.1016/j.febslet.2015.07.027. [DOI] [PubMed] [Google Scholar]
- 48.Morandi F, Horenstein AL, Rizzo R, Malavasi F. The role of extracellular adenosine generation in the development of autoimmune diseases. Mediators Inflamm. 2018;2018:7019398. doi: 10.1155/2018/7019398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Cui M, Ding H, Chen F, Zhao Y, Yang Q, Dong Q. Mdivi-1 protects against ischemic brain injury via elevating extracellular adenosine in a cAMP/CREB-CD39-dependent manner. Mol Neurobiol. 2016;53(1):240–253. doi: 10.1007/s12035-014-9002-4. [DOI] [PubMed] [Google Scholar]
- 50.Grenz A, et al. Contribution of E-NTPDase1 (CD39) to renal protection from ischemia-reperfusion injury. FASEB J. 2007;21(11):2863–2873. doi: 10.1096/fj.06-7947com. [DOI] [PubMed] [Google Scholar]
- 51.Nowak M, et al. The A2aR adenosine receptor controls cytokine production in iNKT cells. Eur J Immunol. 2010;40(3):682–687. doi: 10.1002/eji.200939897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Goto M, et al. Phosphodiesterase 7A inhibitor ASB16165 suppresses proliferation and cytokine production of NKT cells. Cell Immunol. 2009;258(2):147–151. doi: 10.1016/j.cellimm.2009.04.005. [DOI] [PubMed] [Google Scholar]
- 53.Feng J, et al. Ibudilast, a nonselective phosphodiesterase inhibitor, regulates Th1/Th2 balance and NKT cell subset in multiple sclerosis. Mult Scler. 2004;10(5):494–498. doi: 10.1191/1352458504ms1070oa. [DOI] [PubMed] [Google Scholar]
- 54.Tokoyoda K, et al. Up-regulation of IL-4 production by the activated cAMP/cAMP-dependent protein kinase (protein kinase A) pathway in CD3/CD28-stimulated naive T cells. Int Immunol. 2004;16(5):643–653. doi: 10.1093/intimm/dxh072. [DOI] [PubMed] [Google Scholar]
- 55.Elliott KA, Osna NA, Scofield MA, Khan MM. Regulation of IL-13 production by histamine in cloned murine T helper type 2 cells. Int Immunopharmacol. 2001;1(11):1923–1937. doi: 10.1016/S1567-5769(01)00117-5. [DOI] [PubMed] [Google Scholar]
- 56.Vang T, et al. Activation of the COOH-terminal Src kinase (Csk) by cAMP-dependent protein kinase inhibits signaling through the T cell receptor. J Exp Med. 2001;193(4):497–507. doi: 10.1084/jem.193.4.497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bajwa A, et al. Dendritic cell sphingosine 1-phosphate receptor-3 regulates Th1-Th2 polarity in kidney ischemia-reperfusion injury. J Immunol. 2012;189(5):2584–2596. doi: 10.4049/jimmunol.1200999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Almishri W, Deans J, Swain MG. Rapid activation and hepatic recruitment of innate-like regulatory B cells after invariant NKT cell stimulation in mice. J Hepatol. 2015;63(4):943–951. doi: 10.1016/j.jhep.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 59.Deaglio S, et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204(6):1257–1265. doi: 10.1084/jem.20062512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Liang D, Zuo A, Shao H, Chen M, Kaplan HJ, Sun D. Roles of the adenosine receptor and CD73 in the regulatory effect of γδ T cells. PLoS One. 2014;9(9):e108932. doi: 10.1371/journal.pone.0108932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rosin DL, Robeva A, Woodard RL, Guyenet PG, Linden J. Immunohistochemical localization of adenosine A2A receptors in the rat central nervous system. J Comp Neurol. 1998;401(2):163–186. doi: 10.1002/(SICI)1096-9861(19981116)401:2<163::AID-CNE2>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.