Abstract
Purpose
Germline polymorphisms may affect chemotherapy efficacy and toxicity. We examined the effect of polymorphisms in drug metabolism and DNA repair genes on pathological response rates, survival, and toxicity for patients randomised to surgery alone or perioperative ECF chemotherapy in the MRC MAGIC trial.
Experimental design
DNA was extracted from non-tumor resection FFPE blocks. ERCC1, ERCC2, XRCC1, DYPD, and OPRT SNPs were evaluated using Sequenom, GSTP1, GSTT1 deletion and TYMS (TS) 5’ 2R/3R using multiplex PCR. Post PCR amplification TS 2R/3R and GSTT1 samples underwent gel electrophoresis.
Results
Polymorphism data was available for 289/456 (63.4%) operated patients. No polymorphism was statistically significantly associated with pathological response to chemotherapy. Median overall survival (OS) for patients treated with surgery alone with any TS genotype was not different (1.76 years 2R/2R, 1.68 years 2R/3R, 2.09 years 3R/3R). Median OS for patients with a TS 2R/2R genotype treated with chemotherapy was not reached, whereas median OS for 2R/3R and 3R/3R patients were 1.44 and 1.60 years respectively (log rank p value 0.0053). The p value for the interaction between treatment arm and genotype (3R/3R and 3R/2R vs 2R/2R) was 0.029. No polymorphism was statistically significantly associated with chemotherapy toxicity.
Conclusions
In MAGIC, patients with a TS 2R/2R genotype appeared to derive a larger benefit from perioperative ECF chemotherapy than patients with 3R containing genotypes. Further exploration of this potential predictive biomarker in this patient population is warranted.
Keywords: Gastric cancer, gastroesophageal cancer, perioperative chemotherapy, polymorphism, thymidylate synthase
Introduction
Gastric and oesophageal cancers are the third and sixth most common causes of cancer death annually worldwide.(1) Neoadjuvant or perioperative chemotherapy is one standard treatment for patients with operable gastric or gastroesophageal adenocarcinoma prior to surgical resection.(2–5) This approach is associated with a modest (6-13%) absolute overall survival advantage in terms of overall survival compared to surgery alone but also with chemotherapy related toxicities such as nausea and vomiting, and neutropenia. Furthermore, following multimodality therapy half of resected patients develop incurable, metastatic cancer and therefore do not benefit from neoadjuvant chemotherapy.(2,4) Better selection of patients for preoperative chemotherapy might avoid needless toxicity, however currently gastroesophageal cancer patients who are treated with neoadjuvant chemotherapy are selected for treatment based on radiological staging alone as there are no currently validated predictive biomarkers for chemotherapy.
Germline polymorphisms in genes associated with chemotherapy and drug metabolism have been validated as predictors of survival and toxicity outcomes across several tumour types including colorectal and breast cancer.(6–8) Although similar studies have examined the effects of polymorphisms in germline genes relating to chemotherapy metabolism in gastroesophageal cancer, most of these have been retrospective, and all lack an untreated control group.(9–11) The UK MRC MAGIC trial was an open label, multicentre, phase III randomised trial comparing six cycles of perioperative ECF (epirubicin, cisplatin and infused 5-fluorouracil) chemotherapy (3 cycles pre- and 3 cycles post- resection) plus surgery to surgery alone in patients with resectable gastroesophageal cancer.(2) Patients treated with perioperative chemotherapy had improved overall survival (OS) compared to patients treated with surgery alone [5 year OS 36% vs 23%, HR 0.75, (95% CI 0.60-0.93) p=0.009]. As a result, perioperative ECF chemotherapy became one standard treatment regimen for patients with resectable gastroesophageal adenocarcinoma. We hypothesised that selected germline polymorphisms would be associated with pathological response to chemotherapy, overall survival or chemotherapy related toxicity in the MAGIC trial, and herein present the results of this analysis.
Methods
Hematoxylin and eosin (H&E) stained tissue sections from resection specimens were reviewed by a histopathologist (AW). DNA was extracted from non-malignant tissue. Five sections (10 mm thick) were cut and deparaffinised using a standard protocol, and the area of interest was dissected using a sterile scalpel blade. Genomic DNA was extracted using the QIAmp DNA Micro Kit and QIAamp DNA FFPE Tissue Kit (Qiagen, Hilden, Germany), following the manufacturer’s instructions. After dewaxing and rehydrating the slides, tissue was microdissected and placed into a 1.5-mL Eppendorf tube with buffer ATL and proteinase K for digestion (Qiagen). DNA was eluted in buffer ATE (Qiagen) with an elution volume of 60 mL. Quality control of the DNA was performed on the basis of 260:230 and 260:280 ratio values and visual inspection of the wavelength spectral pattern provided by the NanoDrop spectrophotometer (Thermo Scientific, Wilmington, DE). A 260:230 ratio of approximately 2.0, together with a 260:280 ratio of approximately 1.8 and the presence of a peak at 260 nm with a steep decrease toward 280 nm in the wavelength spectrum was considered sufficiently good quality DNA.
Slides from all cases were reviewed and pathological response in tumour graded using the Mandard tumour regression grading (TRG) system.(12)
Genotype Analysis
Ten polymorphisms were selected based on a review of the literature and expected interaction with epirubicin, cisplatin and 5 fluorouracil chemotherapy. These are listed in Table 1. For detailed description of genotype analysis methodology please see supplementary methods.
Table 1. Germline polymorphisms analysed.
| Gene | Polymorphism | rsID |
|---|---|---|
| OPRT | G638C (Gly213Ala) | 1801019 |
| DPYD | IVS14+1G>A | DPYD2A |
| DPYD | A1627G | 1801159 |
| ERCC1 | C118T | 11615 |
| ERCC1 | C8092A | 3212986 |
| ERCC2 | Lys751Gln | 13181 |
| XRCC1 | A399G | 25487 |
| GSTP1 | I105V | 1695 |
| TS 2R/3R 5'UTR | 2R/3R repeat | |
| GSTT1 deletion |
Statistical methods
OS was calculated from surgery to death from any cause or last date of follow up. Date of surgery was selected as the baseline for biomarker analysis to reduce potential bias as only patients with a surgical specimen were available for inclusion. Analyses were performed within treatment arms due to the differences in timing of surgery, to further reduce potential bias in the estimates of effects. Date of surgery could not be confirmed for nine patients in the chemotherapy plus surgery arm and these patients were excluded from the survival analyses. Differences in OS by polymorphism status were assessed using the Kaplan Meier method and compared using Cox regression. To mitigate multiplicity a p-value of <0.01 was considered significant when testing for associations of genotypes with survival and toxicity, and <0.05 when testing interactions Multiple imputation was performed to account for missing polymorphism data. OS results were adjusted for possible confounders of age, subtype, gender, site of primary, WHO, nodal status).
Proportions of patients with good pathological response (TRG 1-2) compared with poor pathological response (TRG 3-5) were compared for each genotype using the Fisher’s exact test. Proportions of patients with toxicities according to genotype were compared using Pearson chi-squared test or Fisher’s exact test where appropriate.
As TS 2R/2R genotype is the polymorphism of interest and is present in approximately 30% of patients, with median OS of 18 months in control (2R/3R + 3R/3R), power of 80%, 5% two sided significance level, to detect a HR of 0.5 would require 85 events. Alternatively as with the same assumptions as above with 70% power 67 events would be required. With respect to pathological response rate in TS genotyped patients, in order to detect a doubling in response rates from 15% to 30%, 206 patients would be required to achieve 70% power. This is based on based on a pathological response rate of 15% in the 3R group, which accounts for 70% of patients, and 30% in the 2R/2R group (which contains 30% of patients). Due to the trial design and retrospective nature of these analyses, all results can only be seem as hypothesis generating and suggestive of future work, with significance levels set to limit the possibility of a type II error.
All analyses were conducted using Stata version 14. The MAGIC protocol was approved by the relevant ethics committees, and patients gave written informed consent for participation in the trial. The translational MAGIC protocol (TransMAGIC) received separate national ethics approval (11/LO/0566).
Results
Polymorphism data was available for 289/456 (63.4%) patients who underwent surgery in the MAGIC trial. There was no difference in distribution of sex, performance status, site of tumour, age or treatment arm between patients with and without polymorphism data, however patients without polymorphism data were more likely to undergo a palliative resection in the view of the operating surgeon (Supplementary Table 1). This resulted in a borderline difference in survival between patients who had polymorphism data available and those who did not, which was more pronounced in the surgery only arm (Supplementary Figure 1).
Discordance in size based polymorphism assessment
We found that on duplicate runs that size based polymorphism assessment that discordance occurred at a rate of 32.7% and 4.2% for GSTT1 and TYMS (TS) respectively.(13) Due to the high rate of discordance in GSTT1 results for this polymorphism was not analysed further.
Genotype frequency
The frequency of each polymorphism genotype is described in Table 2. Genotype frequency was consistent with previously published data and all were in Hardy-Weinberg equilibrium with the exception of DPYD rs1801159.
Table 2. Frequency of each polymorphism.
| Chemo+Surgery | Surgery alone | Total | ||
|---|---|---|---|---|
| TS 2R/3R 5'UTR | 2R/2R | 38 (31%) | 53 (36%) | 91 (34%) |
| 2R/3R | 51 (41%) | 61 (42%) | 112 (41%) | |
| 3R/3R | 35 (28%) | 32 (22%) | 67 (25%) | |
| Total | 124 | 146 | 270 | |
| GSTP1 rs1695 | A | 74 (56%) | 73 (47%) | 147 (51%) |
| AG | 53 (40%) | 67 (43%) | 120 (42%) | |
| G | 6 (5%) | 16 (10%) | 22 (8%) | |
| Total | 133 | 156 | 289 | |
| OPRT rs1801019 | C | 5 (4%) | 3 (2%) | 8 (3%) |
| G | 92 (69%) | 92 (59%) | 184 (64%) | |
| CG | 36 (27%) | 60 (39%) | 96 (33%) | |
| Total | 133 | 155 | 288 | |
| DPYD2A IVS14+1G>A | G | 131 (99%) | 152 (100%) | 283 (>99%) |
| GA | 1 (1%) | 0 (0%) | 1 (<1%) | |
| Total | 132 | 152 | 284 | |
| DPYD rs1801159 | A | 96 (73%) | 100 (65%) | 196 (68%) |
| AG | 27 (20%) | 48 (31%) | 75 (26%) | |
| G | 9 (7%) | 7 (5%) | 16 (6%) | |
| Total | 132 | 155 | 287 | |
| ERCC1 rs11615 | C | 19 (14%) | 29 (19%) | 48 (17%) |
| CT | 70 (53%) | 68 (44%) | 138 (48%) | |
| T | 44 (33%) | 57 (37%) | 101 (35%) | |
| Total | 133 | 154 | 287 | |
| ERCC1 rs3212986 | G | 61 (46%) | 84 (55%) | 145 (51%) |
| GT | 64 (48%) | 60 (39%) | 124 (43%) | |
| T | 7 (5%) | 10 (6%) | 17 (6%) | |
| Total | 132 | 154 | 286 | |
| ERCC2 rs13181 | G | 23 (17%) | 20 (13%) | 43 (15%) |
| GT | 62 (47%) | 63 (41%) | 125 (44%) | |
| T | 48 (36%) | 71 (46%) | 119 (41%) | |
| Total | 133 | 154 | 287 | |
| XRCC1 rs25487 | A | 19 (14%) | 18 (12%) | 37 (13%) |
| AG | 56 (42%) | 67 (44%) | 123 (43%) | |
| G | 57 (43%) | 69 (45%) | 126 (44%) | |
| Total | 132 | 154 | 286 |
Genotype and pathological response to chemotherapy
The association between each polymorphism and pathological response to chemotherapy in chemotherapy treated patients is described in Table 3. No polymorphism was statistically significantly associated with pathological response to chemotherapy.
Table 3. Genotype and pathological response to chemotherapy.
| Genotype | TRG 1-2 | TRG 3-5 | p | OR for TRG 3-5 | 95%CI |
|---|---|---|---|---|---|
| TS | |||||
| 2R/2R | 9 (24.3) | 28 (75.7) | (0.536) | 1.0 | |
| 2R/3R | 8 (16.7) | 40 (83.3) | 0.384 | 1.61 | 0.55-4.68 |
| 3R/3R | 5 (14.7) | 29 (85.3) | 0.313 | 1.86 | 0.56-6.25 |
| 2R/2R | 9 (24.3) | 28 (75.7) | 1.0 | ||
| 2R/3R + 3R/3R | 13 (15.9) | 69 (84.2) | 0.274 | 1.71 | 0.66-4.44 |
| GSTP1 rs1695 | |||||
| A | 11 (15.5) | 60 (84.5) | (0.812) | 1.0 | |
| AG | 10 (20.0) | 40 (80.0) | 0.520 | 0.73 | 0.29-1.89 |
| G | 1 (16.7) | 5 (83.3) | 0.939 | 0.92 | 0.10-8.62 |
| OPRT | |||||
| C | 2 (40.0) | 3 (60.0) | (0.308) | 1.0 | |
| G | 16 (18.2) | 72 (81.8) | 0.249 | 3.0 | 0.46-19.5 |
| GC | 4 (11.8) | 30 (88.2) | 0.128 | 5.0 | 0.63-39.7 |
| DPYD rs1801159 | |||||
| A | 14 (15.4) | 77 (84.6) | (0.413) | 1.0 | |
| AG | 5 (18.5) | 22 (81.5) | 0.698 | 0.80 | 0.26-2.46 |
| G | 3 (33.3) | 6 (66.7) | 0.186 | 0.36 | 0.08-1.62 |
| ERCC1 rs11615 | |||||
| C | 3 (16.7) | 15 (83.3) | (0.211) | 1.0 | |
| CT | 15 (22.7) | 51 (77.3) | 0.580 | 0.68 | 0.17-2.67 |
| T | 4 (9.3) | 39 (90.7) | 0.417 | 1.95 | 0.39-9.77 |
| ERCC1 rs3212986 | |||||
| G | 8 (13.6) | 51 (86.4) | (0.496) | 1.0 | |
| GT | 12 (19.7) | 49 (80.3) | 0.371 | 0.64 | 0.24-1.70 |
| T | 2 (28.6) | 5 (71.4) | 0.308 | 0.39 | 0.06-2.38 |
| ERCC2 rs13181 | |||||
| G | 3 (14.3) | 18 (85.7) | (0.879) | 1.0 | |
| GT | 11 (19.0) | 47 (81.0) | 0.631 | 0.71 | 0.18-2.85 |
| T | 8 (16.7) | 40 (83.3) | 0.804 | 0.83 | 0.20-3.51 |
| XRCC1 rs25487 | |||||
| A | 2 (11.1) | 16 (88.9) | (0.752) | 1.0 | |
| AG | 10 (18.9) | 43 (81.1) | 0.453 | 0.54 | 0.11-2.72 |
| G | 10 (17.9) | 46 (82.1) | 0.503 | 0.58 | 0.11-2.91 |
Genotype and overall survival
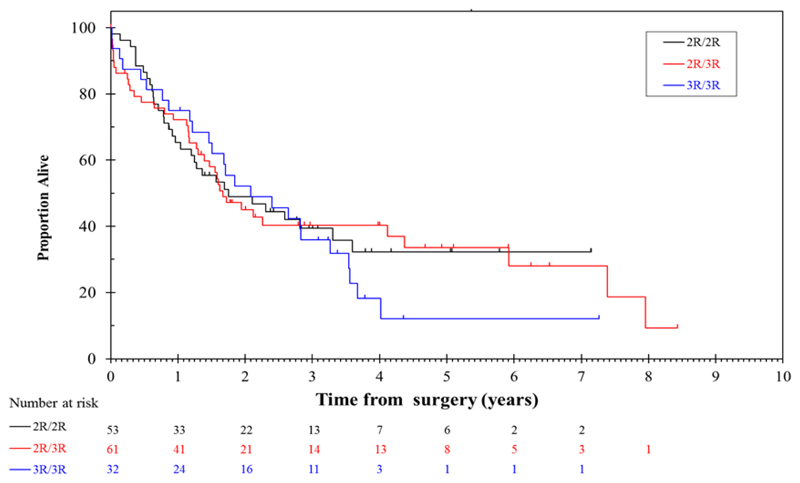
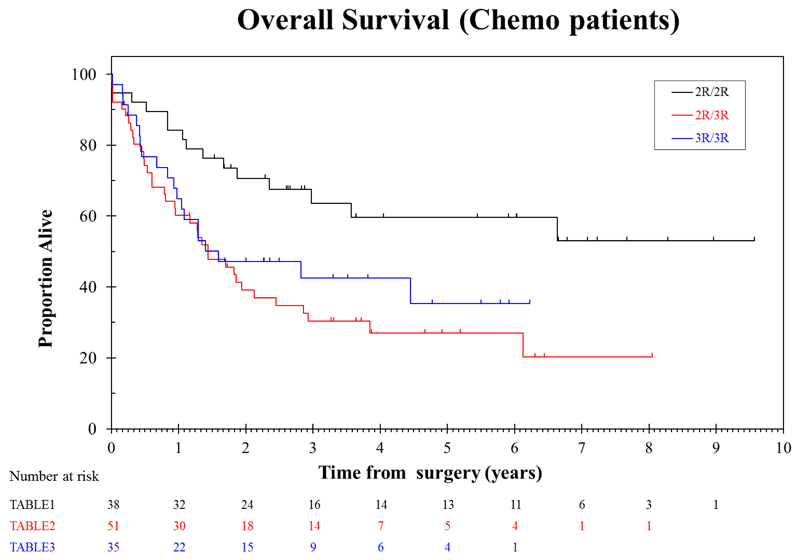
Median overall survival for patients treated with surgery alone who had TS 2R/2R genotype was 1.76 years, compared to 1.68 years for 2R/3R and 2.09 years for 3R/3R (Table 4, Figure 1). These differences were not statistically significant. In contrast, median overall survival for patients with a TS 2R/2R genotype treated with chemotherapy was not reached, whereas survival for 2R/3R and 3R/3R were 1.44 and 1.60 years respectively (log rank p value 0.0053). When all 3R genotypes were combined median overall survival was 1.44 years for chemotherapy treated patients vs not reached for 2R/2R genotype (HR 2.4, p=0.003). The effect of TS genotype on overall survival in chemotherapy treated patients remained statistically significant when adjusted for the potential confounders of age, subtype, gender, site of primary, WHO, nodal status) (Table 4). The p value for the interaction between treatment arm and TS genotype (3R/3R and 3R/2R vs 2R/2R) was 0.029 (with a HR of 0.46).
Table 4. Association between TS genotype and overall survival.
(2nd HR & p-value are adjusted for: age, subtype, gender, site of primary, WHO, nodal status)
| Chemotherapy | Surgery alone | Overall | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 2R/2R | 2R/3R | 3R/3R | 2R/2R | 2R/3R | 3R/3R | 2R/2R | 2R/3R | 3R/3R | |
| Patients | 38 (31%) | 51 (41%) | 35 (28%) | 51 (36%) | 59 (41%) | 32 (23%) | 89 (34%) | 110 (41%) | 67 (25%) |
| Events | 15 | 36 | 20 | 31 | 38 | 25 | 46 | 74 | 45 |
| Median survival | Not reached | 1.44 | 1.60 | 1.76 | 1.68 | 2.09 | 3.31 | 1.62 | 1.84 |
| Log-rank p-value | 0.0053 | 0.7212 | 0.0326 | ||||||
| Hazard ratio | 1 (REF) | 2.66 3.06 |
2.10 2.64 |
1 (REF) | 1.03 1.07 |
1.23 1.45 |
1 (REF) | 1.59 1.73 |
1.53 1.79 |
| HR p-value | 0.002 0.001 |
0.032 0.009 |
0.896 0.778 |
0.448 0.190 |
0.013 0.005 |
0.043 0.008 |
|||
| Combined analysis | |||||||||
| 2R/2R | 2R/3R + 3R/3R | 2R/2R | 2R/3R + 3R/3R | 2R/2R | 2R/3R + 3R/3R | ||||
| Patients | 38 (31%) | 86 | 51 (36%) | 91 | 89 (34%) | 177 | |||
| Events | 15 | 56 | 31 | 63 | 46 | 119 | |||
| Median survival | Not reached | 1.44 | 1.76 | 1.84 | 3.31 | 1/71 | |||
| Log-rank p-value | 0.002 | 0.6519 | 0.009 | ||||||
| Hazard ratio | 1 (REF) | 2.43 2.89 |
1 (REF) | 1.10 1.15 |
1 (REF) | 1.57 1.75 |
|||
| HR p-value | 0.003 0.001 |
0.652 0.531 |
0.010 0.002 |
||||||
Figure 1. Overall survival from surgery by TS genotype (surgery alone arm).
In order to assess the effect of a 4.2% discrepancy in TS genotype status assessment we performed 10000 simulations, randomly changing 4.2% of results. From these 10000 simulations, the 2.5 to 97.5 percentiles of the HR for the interaction between treatment arm and TS genotype (3R/3R and 3R/2R vs 2R/2R) was 0.37 – 0.60, compared to the estimate from the original data of 0.46.
Patients with the (AG) genotype of DPYD rs1801159 Ile543Val had numerically shorter survival compared to the AA genotype in the surgery alone arm of the trial, this difference was statistically significant (HR 1.75, p=0.008). There was no evidence of an interaction between treatment arm and DPYD status. Results were similar when multiple imputation was performed for missing data.
No other genotype was statistically significantly associated with overall survival (see Supplementary Tables 2 A-F).
Genotype and chemotherapy related toxicity
The presence of grade 3 or greater toxicity and association with polymorphism status are detailed in Supplementary Table 1. DPYD2A IVS14+1G>A GA variant was associated with a non-statistically significant trend towards increased rates of ≥ Grade 3 diarrhoea (p=0.039), however only one patient was detected with this variant. No other polymorphism demonstrated a statistically significant relationship with chemotherapy related toxicity.
The mean number of cycles of chemotherapy received for most polymorphisms was five (Supplementary Table 3), with the exception of ERCC1 rs3212986 (GT+TT) variant who had a mean of four cycles (Kruskal-Wallis equality-of-populations rank test p= 0.0425).
Discussion
Our study is the first to evaluate the association between germline polymorphisms and pathological response, overall survival, and chemotherapy related toxicity for patients with operable gastroesophageal cancer in a randomised trial with a control group. We found that patients who have a 2R/2R thymidylate synthase genotype who were treated with perioperative ECF chemotherapy had statistically superior overall survival compared to those who had a 2R/3R or 3R/3R genotype. This difference was not apparent in patients who were treated with surgery alone, and a significant interaction between TS genotype status and treatment arm was noted Additionally, in our study, patients with a TS 2R2R genotype had a non-statistically significant higher rate of good pathological response (TRG 1-2) at 24% compared to 3R allele containing patients. These findings are important as if validated pharmacogenomic genotyping could be used in future to select patients who are more likely to benefit from perioperative chemotherapy.
Thymidylate synthase (TS) acts to produce thymidylate which is an essential precursor for DNA synthesis. The activity of TS is blocked by 5-fluorodeoxyuridylate (5FdUMP), the active metabolite of 5-FU and it is via this mechanism that 5-FU exerts cytotoxicity. The human thymidylate synthase gene is polymorphic through the presence of either double (2R) or triplet (3R) 28 base tandem repeats which are sited upstream of the TS translational start site. [14] These repeats control the transcription and translation of the TS gene; individuals with 3R tandem repeats have higher levels of TS expression in tissue and consequently lower rates of response to fluoropyrimidine chemotherapy.[15] Our findings are in keeping with this biology. Several other series have reported comparable improvements in response rates overall survival similar results in gastric cancer patients with the favourable 2R genotype treated with fluoropyrimidine 5-FU based chemotherapy, however none of these were a randomised trial with an untreated control group.(14–16) However, opposing results have also been demonstrated.(17,18) Potential reasons for this include small, heterogeneous, ethnically diverse populations treated with variable chemotherapy regimens in both advanced and resectable disease settings, and the addition of other related polymorphisms such as the TS 3’UTR 6 base pair polymorphism to analyses.(19) We caveat our discussion with an awareness that length based polymorphism assessment resulted in a discordance rate of 4.2% for TS polymorphism status. However, as our findings for the 2R/2R genotype are quite striking, even when a stringent p.value is applied to correct for multiplicity, and were confirmed with repeated simulation testing to account for any discrepancy in TS genotype assessment we do not think that this is likely to have unduly affected these results.
Dihydropyrimidine dehydrogenase (DPD) is the rate-limiting enzyme in 5-fluorouracil catabolism and variation in DPD levels and activity have profound effects of fluoropyrimidine metabolism and toxicity. The most well described of these is a DPYD 2* splice variant polymorphism which results in a non-functional enzyme and is associated with fluoropyrimidine related toxicity in many studies.(6,20–22) Although low patient deleterious allele frequency and lack of statistical significance due to correction for multiplicity means that we cannot be definitive in our conclusions, our results are consistent with these data. However, we think that these results are of secondary importance to the survival outcomes presented.
We asked two questions from our dataset, firstly, can genotyping be used to differentiate between those who derive a survival benefit from perioperative chemotherapy and those who do not, and secondly, if these genotypes were assessed preoperatively, would it possible to predict excessive toxicity prior to commencing chemotherapy? Regarding survival benefit, our findings relating the favourable effects of the TS 2R/2R genotype are shared with several other large studies. Therefore, is further validation with a clinical trial required? One small genotype directed clinical trial clinical trial evaluated FOLFOX chemotherapy in 25 patients with TS 2R containing genotypes (2R/2R and 2R/3R) and found that radiological response rates did not differ compared to historical control.(33) However, based on our results only the 2R/2R genotype would benefit from this approach; this was also suggested in subgroup analysis of that study. As patients with 3R containing genotypes did not appear to benefit from fluoropyrimidine based chemotherapy in MAGIC, we suggest that alternative treatment options should be evaluated for these patients. Omitting perioperative chemotherapy completely is unlikely to be acceptable as many patients (especially those with proximal tumours) require downstaging prior to surgical resection. Alternatively, patients with 3R containing genotypes could be treated with higher doses of fluoropyrimidines, although this could be result in increased toxicity. This approach in UGT1A1 genotyped patients has demonstrated that patients who are wildtype or heterozygous for the deleterious *28 allele can tolerate increased doses of irinotecan compared to UGT1A1 *28 homozygotes.(34,35) Finally, a non-fluoropyrimidine containing regimen could be considered; for patients with tumours of the gastroesophageal junction or oesophagus chemoradiotherapy with carboplatin and paclitaxel would seem a reasonable alternative.
With respect to avoiding toxicity, the relative rarity of alleles which predict for significant toxicity such as DPYD 2A* is associated with significant screening costs even when toxicity is reduced by the use of pre-emptive dose reductions.(36) As such, neither the European Medicines Agency nor the US Food Drug Administration require testing for DPYD variants prior to treatment with fluoropyrimidines despite the availability of advice from expert groups such Clinical Pharmacogenetics Implementation Consortium and the Dutch Pharmacogenetics Working Group which provide clinical practice guidelines on genotype based drug dosing.(37,38) In the MAGIC trial, the most common grade 3 or greater chemotherapy associated toxicity was neutropenia which is likely to be due to epirubicin and which is not predicted by any of the polymorphisms which we examined. Therefore, routine testing for DPYD 2A* polymorphisms is unlikely to significantly decrease toxicity in patients treated with MAGIC type chemotherapy.
The interaction between chemotherapy and genotype is complex, and coloured by many other clinical variables such as age, ethnicity, gender, hepatorenal function, and the interaction between individual components of each chemotherapy regimen. This has profound implications for the accuracy of toxicity or outcome prediction using genotyping. One potential flaw relating specifically to this work is that not all MAGIC trial participants were included in the current study as not all provided tissue for analysis, therefore we caution that the analysis could be underpowered to detect small effect sizes. On one hand, if a patient did not undergo surgery due to failure to respond to treatment then no tissue was available for analysis. Alternatively, patients with excessive toxicity due to chemotherapy may also have stopped chemotherapy prior to surgery. These potential biases may be reflected in the borderline improved overall survival demonstrated for patients with polymorphism data available. Thus although germline genotype will not be altered by treatment, any true assessment of the predictive power of genotype would preferentially be performed in pre-treatment samples for these reasons. A second issue relates to the technical challenges associated with length based polymorphism assessment; moving forward advances in high throughput next generation sequencing technologies should ensure improved accuracy and speed of results with decreased DNA requirements.
In summary, ours is the first study to examine the effect of germline polymorphisms on pathological response and survival outcomes for patients treated with perioperative chemotherapy for operable oesophagogastric cancer, with a randomised control group. We found that patients with a TS 2R/2R genotype (representing 34% of the population) had excellent survival when treated with perioperative ECF chemotherapy. In contrast, patients with a 3R containing genotype did not appear to derive a similar benefit from standard dose fluoropyrimidine based chemotherapy when compared to patients treated with surgery alone. It is salutary to note that despite recent progress in our understanding of the molecular biology underpinning gastroesophageal cancer that only one targeted drug, trastuzumab, is licenced in this disease, and that almost all patients will receive platinum and fluoropyrimidine based chemotherapy as a component of their treatment.(39,40) Therefore use of available data relating to patient selection for standard of care chemotherapy to design a prospective would appear to be a sound choice.
Supplementary Material
Statement of translational relevance.
Neoadjuvant or perioperative cisplatin and 5-fluorouracil based chemotherapy plus surgery is one standard of care for patients with resectable gastroesophageal cancer. However, chemotherapy benefits only a small proportion of patients, and validated biomarkers predictive of response or toxicity have been elusive. We analysed the effect of multiple germline polymorphisms putatively associated with response or toxicity to chemotherapy in patients treated with chemotherapy plus surgery or surgery alone in the UK Medical Research Council MAGIC Trial. One polymorphism in thymidylate synthase (TS), a 2R/2R tandem repeat, was significantly associated with overall survival in patients treated with chemotherapy, but not in patients treated with surgery alone. These findings suggest that neoadjuvant chemotherapy for patients with operable gastroesophageal cancer could be personalized based on germline polymorphism status.
Statement of significance.
We demonstrate in a randomised trial with a chemotherapy control arm that the presence of the thymidylate synthase 2R/2R genotype is associated with significantly improved overall survival for patients with operable gastroesophageal cancer treated with fluoropyrimidine based chemotherapy plus surgery. Further validation of this potential predictive biomarker for chemotherapy efficacy may be appropriate.
Figure 2.
Author Financial Support
ES, AW, CP, WA, NV, and DC acknowledge funding from the RMH/ICR NIHR BRC. The TransMAGIC project was funded by Cancer Research UK grant C20023/A7217. In Singapore (PT) this work was supported by grant NMRC TCR/009-NUHS/2013.
Footnotes
Acknowledgments/Author contributions
Conception and design: ES, SZ, DC, AW, RS, AO, SS, RL, PT
Development of methodology: ES, SZ, DC, CP, RS, AO, SS, MN, RL, PT
Acquisition of data: ES, SZ, DC, AW, CP, RS, NV, MF, MR, AO, WA, SS, MN, RL, PT
Analysis and interpretation of data: ES, DC, CP, SS, MN, RL, PT
Writing, review, and/or revision of the manuscript: all authors
Administrative, technical, or material support: SZ, DC, AW, CP, NV, MF, MR, WA, SS, MN, RL, PT
Study supervision: DC, RL, PT
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66(1):7–30. doi: 10.3322/caac.21332. [DOI] [PubMed] [Google Scholar]
- 2.Cunningham D, Allum WH, Stenning SP, Thompson JN, Van de Velde CJ, Nicolson M, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med. 2006;355(1):11–20. doi: 10.1056/NEJMoa055531. [DOI] [PubMed] [Google Scholar]
- 3.Ychou M, Boige V, Pignon J-P, Conroy T, Bouché O, Lebreton G, et al. Perioperative Chemotherapy Compared With Surgery Alone for Resectable Gastroesophageal Adenocarcinoma: An FNCLCC and FFCD Multicenter Phase III Trial. Journal of Clinical Oncology. 2011;29(13):1715–21. doi: 10.1200/jco.2010.33.0597. [DOI] [PubMed] [Google Scholar]
- 4.Surgical resection with or without preoperative chemotherapy in oesophageal cancer: a randomised controlled trial. The Lancet. 2002;359(9319):1727–33. doi: 10.1016/s0140-6736(02)08651-8. [DOI] [PubMed] [Google Scholar]
- 5.Alderson D, Langley RE, Nankivell MG, Blazeby JM, Griffin M, Crellin A, et al. Neoadjuvant chemotherapy for resectable oesophageal and junctional adenocarcinoma: Results from the UK Medical Research Council randomised OEO5 trial (ISRCTN 01852072) ASCO Meeting Abstracts. 2015;33(15_suppl):4002. [Google Scholar]
- 6.Lee AM, Shi Q, Pavey E, Alberts SR, Sargent DJ, Sinicrope FA, et al. DPYD variants as predictors of 5-fluorouracil toxicity in adjuvant colon cancer treatment (NCCTG N0147) J Natl Cancer Inst. 2014;106(12) doi: 10.1093/jnci/dju298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Campbell JM, Stephenson MD, Bateman E, Peters MD, Keefe DM, Bowen JM. Irinotecan-induced toxicity pharmacogenetics: an umbrella review of systematic reviews and meta-analyses. Pharmacogenomics J. 2016 doi: 10.1038/tpj.2016.58. [DOI] [PubMed] [Google Scholar]
- 8.Hoskins JM, Carey LA, McLeod HL. CYP2D6 and tamoxifen: DNA matters in breast cancer. Nat Rev Cancer. 2009;9(8):576–86. doi: 10.1038/nrc2683. http://www.nature.com/nrc/journal/v9/n8/suppinfo/nrc2683_S1.html. [DOI] [PubMed] [Google Scholar]
- 9.Goekkurt E, Al-Batran S-E, Hartmann JT, Mogck U, Schuch G, Kramer M, et al. Pharmacogenetic Analyses of a Phase III Trial in Metastatic Gastroesophageal Adenocarcinoma With Fluorouracil and Leucovorin Plus Either Oxaliplatin or Cisplatin: A Study of the Arbeitsgemeinschaft Internistische Onkologie. Journal of Clinical Oncology. 2009;27(17):2863–73. doi: 10.1200/jco.2008.19.1718. [DOI] [PubMed] [Google Scholar]
- 10.Ott K, Rachakonda P, Panzram B, Keller G, Lordick F, Becker K, et al. DNA Repair Gene and MTHFR; Gene Polymorphisms as Prognostic Markers in Locally Advanced Adenocarcinoma of the Esophagus or Stomach Treated with Cisplatin and 5-Fluorouracil-Based Neoadjuvant Chemotherapy. Annals of Surgical Oncology. 2011;18(9):2688–98. doi: 10.1245/s10434-011-1601-y. [DOI] [PubMed] [Google Scholar]
- 11.Kawakami K, Graziano F, Watanabe G, Ruzzo A, Santini D, Catalano V, et al. Prognostic Role of Thymidylate Synthase Polymorphisms in Gastric Cancer Patients Treated with Surgery and Adjuvant Chemotherapy. Clinical Cancer Research. 2005;11(10):3778–83. doi: 10.1158/1078-0432.ccr-04-2428. [DOI] [PubMed] [Google Scholar]
- 12.Smyth EC, Fassan M, Cunningham D, Allum WH, Okines AF, Lampis A, et al. Effect of Pathologic Tumor Response and Nodal Status on Survival in the Medical Research Council Adjuvant Gastric Infusional Chemotherapy Trial. J Clin Oncol. 2016 doi: 10.1200/jco.2015.65.7692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang S, BeeHuat Tan I, Sapari NS, Grabsch H, Okines A, Smyth EC, et al. Technical Reproducibility of Single-Nucleotide and Size-Based DNA Biomarker Assessment Using DNA Extracted from Formalin-Fixed, Paraffin-Embedded Tissues. J Mol Diagn. 2015 doi: 10.1016/j.jmoldx.2014.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goekkurt E, Hoehn S, Wolschke C, Wittmer C, Stueber C, Hossfeld DK, et al. Polymorphisms of glutathione S-transferases (GST) and thymidylate synthase (TS) - novel predictors for response and survival in gastric cancer patients. Br J Cancer. 2005;94(2):281–6. doi: 10.1038/sj.bjc.6602891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ott K, Vogelsang H, Marton N, Becker K, Lordick F, Kobl M, et al. The thymidylate synthase tandem repeat promoter polymorphism: A predictor for tumor-related survival in neoadjuvant treated locally advanced gastric cancer. Int J Cancer. 2006;119(12):2885–94. doi: 10.1002/ijc.22235. [DOI] [PubMed] [Google Scholar]
- 16.Huang K, Shen Y, Zhang F, Wang S, Wei X. Evaluation of effects of thymidylate synthase and excision repair cross-complementing 1 polymorphisms on chemotherapy outcome in patients with gastrointestinal tumors using peripheral venous blood. Oncology letters. 2016;11(5):3477–82. doi: 10.3892/ol.2016.4423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Seo BG, Kwon HC, Oh SY, Lee S, Kim SG, Kim SH, et al. Comprehensive analysis of excision repair complementation group 1, glutathione S-transferase, thymidylate synthase and uridine diphosphate glucuronosyl transferase 1A1 polymorphisms predictive for treatment outcome in patients with advanced gastric cancer treated with FOLFOX or FOLFIRI. Oncol Rep. 2009;22(1):127–36. [PubMed] [Google Scholar]
- 18.Han S-W, Oh D-Y, Im S-A, Park SR, Lee K-W, Song HS, et al. Epidermal growth factor receptor intron 1 CA dinucleotide repeat polymorphism and survival of advanced gastric cancer patients treated with cetuximab plus modified FOLFOX6. Cancer Science. 2010;101(3):793–9. doi: 10.1111/j.1349-7006.2009.01447.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mandola MV, Stoehlmacher J, Muller-Weeks S, Cesarone G, Yu MC, Lenz H-J, et al. A Novel Single Nucleotide Polymorphism within the 5′ Tandem Repeat Polymorphism of the Thymidylate Synthase Gene Abolishes USF-1 Binding and Alters Transcriptional Activity. Cancer Research. 2003;63(11):2898–904. [PubMed] [Google Scholar]
- 20.Wei X, McLeod HL, McMurrough J, Gonzalez FJ, Fernandez-Salguero P. Molecular basis of the human dihydropyrimidine dehydrogenase deficiency and 5-fluorouracil toxicity. The Journal of clinical investigation. 1996;98(3):610–5. doi: 10.1172/jci118830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schwab M, Zanger UM, Marx C, Schaeffeler E, Klein K, Dippon J, et al. Role of genetic and nongenetic factors for fluorouracil treatment-related severe toxicity: a prospective clinical trial by the German 5-FU Toxicity Study Group. J Clin Oncol. 2008;26(13):2131–8. doi: 10.1200/jco.2006.10.4182. [DOI] [PubMed] [Google Scholar]
- 22.Meulendijks D, Henricks LM, Sonke GS, Deenen MJ, Froehlich TK, Amstutz U, et al. Clinical relevance of DPYD variants c.1679T>G, c.1236G>A/HapB3, and c.1601G>A as predictors of severe fluoropyrimidine-associated toxicity: a systematic review and meta-analysis of individual patient data. Lancet Oncol. 2015;16(16):1639–50. doi: 10.1016/s1470-2045(15)00286-7. [DOI] [PubMed] [Google Scholar]
- 23.Ho TV, Scharer OD. Translesion DNA synthesis polymerases in DNA interstrand crosslink repair. Environmental and molecular mutagenesis. 2010;51(6):552–66. doi: 10.1002/em.20573. [DOI] [PubMed] [Google Scholar]
- 24.Hoeijmakers JH, Egly JM, Vermeulen W. TFIIH: a key component in multiple DNA transactions. Current opinion in genetics & development. 1996;6(1):26–33. doi: 10.1016/s0959-437x(96)90006-4. [DOI] [PubMed] [Google Scholar]
- 25.Sung P, Bailly V, Weber C, Thompson LH, Prakash L, Prakash S. Human xeroderma pigmentosum group D gene encodes a DNA helicase. Nature. 1993;365(6449):852–5. doi: 10.1038/365852a0. [DOI] [PubMed] [Google Scholar]
- 26.Macerelli M, Ganzinelli M, Gouedard C, Broggini M, Garassino MC, Linardou H, et al. Can the response to a platinum-based therapy be predicted by the DNA repair status in non-small cell lung cancer? Cancer Treatment Reviews. 2016;48:8–19. doi: 10.1016/j.ctrv.2016.05.004. [DOI] [PubMed] [Google Scholar]
- 27.Krivak TC, Darcy KM, Tian C, Armstrong D, Baysal BE, Gallion H, et al. Relationship Between ERCC1 Polymorphisms, Disease Progression, and Survival in the Gynecologic Oncology Group Phase III Trial of Intraperitoneal Versus Intravenous Cisplatin and Paclitaxel for Stage III Epithelial Ovarian Cancer. Journal of Clinical Oncology. 2008;26(21):3598–606. doi: 10.1200/jco.2008.16.1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bohanes P, LaBonte MJ, Lenz H-J. A Review of Excision Repair Cross-complementation Group 1 in Colorectal Cancer. Clinical Colorectal Cancer. 2011;10(3):157–64. doi: 10.1016/j.clcc.2011.03.024. [DOI] [PubMed] [Google Scholar]
- 29.Lambrechts S, Lambrechts D, Despierre E, Van Nieuwenhuysen E, Smeets D, Debruyne PR, et al. Genetic variability in drug transport, metabolism or DNA repair affecting toxicity of chemotherapy in ovarian cancer. BMC pharmacology & toxicology. 2015;16:2. doi: 10.1186/s40360-015-0001-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Z, Chen JQ, Liu JL, Qin XG, Huang Y. Polymorphisms in ERCC1, GSTs, TS and MTHFR predict clinical outcomes of gastric cancer patients treated with platinum/5-Fu-based chemotherapy: a systematic review. BMC Gastroenterol. 2012;12:137. doi: 10.1186/1471-230x-12-137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sakano S, Hinoda Y, Sasaki M, Wada T, Matsumoto H, Eguchi S, et al. Nucleotide excision repair gene polymorphisms may predict acute toxicity in patients treated with chemoradiotherapy for bladder cancer. Pharmacogenomics. 2010;11(10):1377–87. doi: 10.2217/pgs.10.106. [DOI] [PubMed] [Google Scholar]
- 32.Ruzzo A, Graziano F, Kawakami K, Watanabe G, Santini D, Catalano V, et al. Pharmacogenetic Profiling and Clinical Outcome of Patients With Advanced Gastric Cancer Treated With Palliative Chemotherapy. Journal of Clinical Oncology. 2006;24(12):1883–91. doi: 10.1200/jco.2005.04.8322. [DOI] [PubMed] [Google Scholar]
- 33.Goff LW, Thakkar N, Du L, Chan E, Tan BR, Cardin DB, et al. Thymidylate synthase genotype-directed chemotherapy for patients with gastric and gastroesophageal junction cancers. PLoS One. 2014;9(9):e107424. doi: 10.1371/journal.pone.0107424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Toffoli G, Cecchin E, Gasparini G, D'Andrea M, Azzarello G, Basso U, et al. Genotype-driven phase I study of irinotecan administered in combination with fluorouracil/leucovorin in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28(5):866–71. doi: 10.1200/jco.2009.23.6125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim KP, Hong YS, Lee JL, Bae KS, Kim HS, Shin JG, et al. A phase I study of UGT1A1 *28/*6 genotype-directed dosing of irinotecan (CPT-11) in Korean patients with metastatic colorectal cancer receiving FOLFIRI. Oncology. 2015;88(3):164–72. doi: 10.1159/000368674. [DOI] [PubMed] [Google Scholar]
- 36.Deenen MJ, Meulendijks D, Cats A, Sechterberger MK, Severens JL, Boot H, et al. Upfront Genotyping of DPYD*2A to Individualize Fluoropyrimidine Therapy: A Safety and Cost Analysis. J Clin Oncol. 2016;34(3):227–34. doi: 10.1200/jco.2015.63.1325. [DOI] [PubMed] [Google Scholar]
- 37.Caudle KE, Thorn CF, Klein TE, Swen JJ, McLeod HL, Diasio RB, et al. Clinical Pharmacogenetics Implementation Consortium guidelines for dihydropyrimidine dehydrogenase genotype and fluoropyrimidine dosing. Clin Pharmacol Ther. 2013;94(6):640–5. doi: 10.1038/clpt.2013.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Swen JJ, Nijenhuis M, de Boer A, Grandia L, Maitland-van der Zee AH, Mulder H, et al. Pharmacogenetics: from bench to byte--an update of guidelines. Clin Pharmacol Ther. 2011;89(5):662–73. doi: 10.1038/clpt.2011.34. [DOI] [PubMed] [Google Scholar]
- 39.Comprehensive molecular characterization of gastric adenocarcinoma. Nature. 2014;513(7517):202–9. doi: 10.1038/nature13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bang YJ, Van Cutsem E, Feyereislova A, Chung HC, Shen L, Sawaki A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet. 2010;376(9742):687–97. doi: 10.1016/s0140-6736(10)61121-x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.