Abstract
Dubin-Johnson syndrome (DJS) is a rare, autosomal recessive disorder characterized by predominantly conjugated hyperbilirubinemia, caused by a mutation in the adenosine triphosphate-binding cassette subfamily C member 2 (ABCC2) gene coding the multidrug resistance-associated protein 2 (MRP2) protein. ABCC2 mutations have been identified in patients with DJS worldwide; however, the mutation pattern of ABCC2 in China is not well studied. In the present study, the mutation pattern of the ABCC2 gene in Chinese patients with DJS was investigated. A total of 7 clinically confirmed patients with DJS were enrolled, and mutation analysis of the ABCC2 gene was performed by Sanger sequencing of genomic DNA extracted from whole blood. All 32 exons and the adjacent splice junction areas were sequenced. All cases were identified to harbor at least one non-synonymous variant in the ABCC2 gene, including three known mutations in 3 cases and three novel variants (p.G693R, p.G808V and p.E647X) in the other 4 cases, with the known p.R393W and the novel p.G693R and p.E647X variants identified in 2 of the 7 cases (28.6%), respectively. All the identified mutations were heterozygous, and 1 case presented with a compound heterozygous mutation, namely p.G693R/p.G808V, while the other cases carried only one single mutation. The loss of membrane expression of MRP2 caused by the novel nonsense variant, p.E647X, was confirmed by immunohistochemical analysis of liver biopsy. The present study provided the first report on the mutation patterns of the ABCC2 gene in Chinese patients with DJS, and the clinical association of these mutations with the syndrome.
Keywords: Dubin-Johnson syndrome, mutation, adenosine triphosphate-binding cassette subfamily C member 2, multidrug resistance-associated protein 2
Introduction
Dubin-Johnson syndrome (DJS) is an autosomal recessive disorder, originally described in 1954 (1). The syndrome is characterized by fluctuating predominantly conjugated hyperbilirubinemia without hemolysis, and patients with DJS may suffer from nonspecific symptoms, such as abdominal discomfort (1). Urinary coproporphyrin output is normal, with the majority of the coproporphyrin fraction being coproporphyrin I (2). Cholescintigraphy in DJS patients displays unique delayed visualization of the liver and biliary tract (3). In addition, liver histology in DJS patients demonstrates distinctive deposits of melanin-like pigment in the hepatocyte lysosomes, which gives the organ a characteristic black color. The disorder occurs in all races and nationalities, and in both sexes. It is considered to be a rare disorder, however, in Sephardic Jews a higher incidence is observed of approximately 1:3,000 (4).
The adenosine triphosphate (ATP)-binding cassette subfamily C member 2 (ABCC2) gene is located on chromosome 10q24, which encodes the human canalicular multispecific organic anion transporter, also called the multidrug resistance-associated protein 2 (MRP2). MRP2, comprising 1,545 amino acids, is a specific non-bile acid organic anion transporter, which mediates the primary active export of conjugates of lipophilic compounds with glucuronate or glutathione from cells in an ATP-dependent manner (5). Genetic alterations of the ABCC2 gene have been identified in DJS patients, including deletions, missense mutations, nonsense mutations and splice junction mutations, such as IVS15+2T→C (6). However, no hotspot mutations have been identified in the ABCC2 gene. The majority of the DJS-associated mutations are considered to cause defects in MRP2 protein deletions and maturation, leading to greatly reduced biliary secretion of organic anions (7). Certain mutations may lead to rapid degradation of the mutant mRNA, whereas others may affect protein maturation, protein stability, or the function of correctly localized proteins (8,9).
ABCC2 mutations have been identified in patients with DJS worldwide; however, little is known regarding the mutation pattern of ABCC2 in China. In the present study, the mutation patterns of the ABCC2 gene were investigated in 7 Chinese DJS patients, and genotype and phenotype analysis of the ABCC2 mutations in these patients was conducted.
Patients and methods
Study population
A total of 7 DJS patients enrolled from the China Registry of Genetic/Metabolic Liver Diseases (ClinicalTrials.gov identifier, NCT03131427) between June 2015 and December 2017 were included in the present study. The clinical diagnosis of DJS (7) was based on biochemical evidence of fluctuating predominantly conjugated hyperbilirubinemia with or without family history, and/or histochemistry and immunohistology of the liver. Patients did not present hemolysis, other genetic and metabolic liver diseases, obstruction or dilation of the biliary tree, viral hepatitis, malignant tumors, and drug-induced or autoimmune liver diseases. A total of 4 patients with hepatocellular carcinoma who had undergone hepatectomy in Beijing Friendship Hospital (Beijing, China) from October 2017 to December 2017 were recruited to the current study. Distal normal tissues from the resected hepatic tissues of these patients were collected and used as controls.
The study was conducted in accordance with the Declaration of Helsinki. The Ethics Committee of the Beijing Friendship Hospital at Capital Medical University approved the study protocol. All patients provided written informed consent. Whole blood samples were collected from the 7 patients with DJS, and liver biopsy samples were available and collected from 4 of the 7 patients. The whole blood was stored at −20°C prior to the extraction of genomic DNA for the detection of ABCC2 mutations. For the liver biopsy samples, formalin-fixed and paraffin-embedded tissue was prepared, and 4-µm sections were cut for hematoxylin and eosin (HE) staining, Schmorl's staining and immunohistochemical (IHC) analysis of MRP2 expression.
Sanger sequencing and functional prediction of variants in the ABCC2 gene
Genomic DNA was extracted from whole blood using a Genomic DNA Purification kit (Qiagen, Valencia, CA, USA). All 32 exons and their associated boundary regions (adjacent base sequence in introns) of the ABCC2 gene were amplified by polymerase chain reaction (PCR) using primers designed with Primer3 software (http://bioinfo.ut.ee/primer3-0.4.0/; Table I). PCR amplification (2×Taq PCR MasterMix; cat. no KT201-13; Tiangen Biotech Co., Ltd., Beijing, China) was performed in a PCR cycler (Applied Biosystems Veriti 96-well Thermal Cycler; Thermo Fisher Scientific, Inc., Waltham, MA, USA) under the following conditions: Denaturation for 30 sec at 95°C followed by 38 cycles of 15 sec at 95°C, 30 sec at the annealing temperature of each pair of primers (56°C for exons 2, 3, 10, 14, 17, 18–19, 20–21, 31 and 32; 58°C for exons 1, 7, 9, 12–13, 15, 16, 24, 25, 26, 27–28 and 30; and 60°C for exon 4–6, 8, 11, 22–23, 29) and 70 sec at 72°C, with extension for 10 min at 72°C. PCR products were sequenced in both the forward and reverse orientations using an automated DNA sequencer (Applied Biosystems 3730 DNA Analyzer; Thermo Fisher Scientific, Inc.).
Table I.
Primers used for sequencing of ABCC2 gene.
| No. | Forward | Reverse | Exon |
|---|---|---|---|
| 1 | 5′-GTCTTCGTTCCAGACGCAGT-3′ | 5′-TTCTTGTTGGTGACCACCCTA-3′ | 1 |
| 2 | 5′-ACAATCCTTCCCCTTTGGTC-3′ | 5′-TGCACCAAGGAATTAGAGTTCA-3′ | 2 |
| 3 | 5′-CAGTGGTCTTTTTCCCTTCTCA-3′ | 5′-GGTAAACAGGGCAGAAGTGG-3′ | 3 |
| 4 | 5′-GTCCATGGAGATGAGGCACT-3′ | 5′-GTTGCAGTGAGCCAAGATCA-3′ | 4–6 |
| 5 | 5′-CTGTGGTTCGCTCTTGTTCC-3′ | 5′-TGATGCTGATGTACCCTTGC-3′ | 7 |
| 6 | 5′-GAGCTGCTCAGGCCAGTAAC-3′ | 5′-CCCTGAAAGGACCATCTGAG-3′ | 8 |
| 7 | 5′-TGAGGAGAGAGGCATCCTTG-3′ | 5′-TGAGGGGATTTTCTTTGGTG-3′ | 9 |
| 8 | 5′-GCTTTGTCCATGGGTCCTAA-3′ | 5′-GAAAGCTTATATTCTTCTGGGTGA-3′ | 10 |
| 9 | 5′-CCCTCTCTCATGGAAGCGTA-3′ | 5′-GAGAGCCACTGCTTCTGTCC-3′ | 11 |
| 10 | 5′-GGGCAATCATGTGAGCTGTA-3′ | 5′-GGTCAAACCATTGGTCTCCA-3′ | 12–13 |
| 11 | 5′-TCTCTCTGCTTGTGCTCGTT-3′ | 5′-GCGAATAAGTTTGGGAAGCA-3′ | 14 |
| 12 | 5′-GTCACGTGGGGACCTACATT-3′ | 5′-AATAGGCCAGGCAGTGAGAA-3′ | 15 |
| 13 | 5′-TCAATACCCAACCCCTGCTA-3′ | 5′-ATTCGGGAGTCAGAGGCTTT-3′ | 16 |
| 14 | 5′-TTTGTTTCTTCCCCTCTCCA-3′ | 5′-TCACCCTTGAAGATCCCTTG-3′ | 17 |
| 15 | 5′-TCTTCCTTTTACCCCTCCCTA-3′ | 5′-ACCCATGGCCCAAGTTCTAT-3′ | 18–19 |
| 16 | 5′-TGCTGAAACCAGCAAGATCA-3′ | 5′-TTTGCAAAGGACAGAGGACA-3′ | 20–21 |
| 17 | 5′-CTCCTTGTGGTTGGCATTCT-3′ | 5′-GGGAGCTCACAGCAGGTACT-3′ | 22–23 |
| 18 | 5′-GGAGCCTCTCATCATTCTGC-3′ | 5′-CCTCCCACCGCTAATATCAA-3′ | 24 |
| 19 | 5′-GTTCTGTGAACGCCAAGGTT-3′ | 5′-CCAGGGTTTGAACCTCAGTC-3′ | 25 |
| 20 | 5′-GTTCTGTGAACGCCAAGGTT-3′ | 5′-CCAGGGTTTGAACCTCAGTC-3′ | 26 |
| 21 | 5′-AAAGTCGGCACTGGATTGTC-3′ | 5′-GTGTGATCCCTGGCTGCTAT-3′ | 27–28 |
| 22 | 5′-GCCAGTCACTGCCTCTTACC-3′ | 5′-CCGAGTAGACCGTGGAATTG-3′ | 29 |
| 23 | 5′-CAACCACAAACCAGCTTCCT-3′ | 5′-ACACGAGGAACACGAGGAGT-3′ | 30 |
| 24 | 5′-TGAAAAACGATGCTCACAGC-3′ | 5′-CCTTCTGCCATCAGGTGTTT-3′ | 31 |
| 25 | 5′-GATGTGTGTAGCTGTGGCTCA-3′ | 5′-GACAATCGAGGGGTTTCTCA-3′ | 32 |
Mutations identified in the ABCC2 gene were uploaded to the online Human Gene Mutation Database (HGMD; http://www.hgmd.cf.ac.uk/) to determine whether they were known disease mutations. The online software ExAC (http://exac.broadinstitute.org/) was used to examine the allele frequencies of each mutation worldwide or in specific regions. Results from the online software MutationTaster (http://www.mutationtaster.org/), PolyPhen-2 (http://genetics.bwh.harvard.edu/pph2/) and SIFT (http://sift.bii.a-star.edu.sg/) were used to predict whether the novel mutations would affect the biological function of the ABCC2 gene.
IHC analysis of MRP2 expression
Formalin-fixed and paraffin-embedded liver biopsy tissues were prepared, and 4-µm sections (4 µm) were cut for IHC analysis. Following deparaffinization of the slides, the endogenous peroxidase activity was blocked with 0.3% H2O2 in methanol for 30 min at room temperature. Antigen retrieval was then conducted in antigen unmasking solution (H-3300; Vector Laboratories, Inc., Burlingame, CA, USA) by microwaving at 700 W for 15 min, keeping the solution at boiling and then treating with 5% skimmed milk in phosphate-buffered saline/0.1% bovine serum albumin for at least 1 h at room temperature to block non-specific staining. Immunohistochemical staining was then performed using monoclonal antibodies against MRP2 (cat. no. Ab3373; Abcam, Cambridge, MA, USA) at a dilution of 1:100 at 4°C overnight, followed by addition of secondary antibody (cat. no. MP-7402; dilution, ready-to-use; Vector Laboratories, Inc.) at 37°C for 1 h. Visualization of antigen-antibody reactions was achieved with 3,3′-diaminobenzidine (DAB; SK-4100 kit; Vector Laboratories, Inc.) and examined by an optical microscope (Eclipse E200; Nikon Corporation, Tokyo, Japan).
Results
Patients and clinical data
In total, 7 patients with DJS and a mean age of 30±12 years (range, 13–43 years old) were enrolled into the current study. Three of the patients were female. The median levels of total bilirubin and direct bilirubin of these patients were 91 and 50 µmol/l (normal reference for total bilirubin range was 3.4–17.1 µmol/l and the direct bilirubin range was 0–6.8 µmol/l), respectively. The clinical characteristics of the patients are shown in Table II.
Table II.
Clinical characteristics and non-synonymous variants identified in the patients with Dubin-Johnson syndrome.
| Patient no. | Age (years) | Sex | Total bilirubin (µmol/l) | Direct bilirubin (µmol/l) | Variants |
|---|---|---|---|---|---|
| 1 | 20 | Male | 91 | 56 | p.E647X (c.2052G>T) |
| 2 | 43 | Male | 120 | 79 | p.R393W (c.1290C>T) |
| 3 | 21 | Female | 94 | 54 | p.G693R (c.2190G>A) |
| 4 | 39 | Male | 97 | 49 | p.G693R (c.2190G>A), |
| p.G808V (c.2536G>T) | |||||
| 5 | 39 | Female | 61 | 31 | p.E647X (c.2052G>T) |
| 6 | 13 | Male | 77 | 50 | p.R393W (c.1290C>T), |
| p.V417I (c.1362G>A) | |||||
| 7 | 40 | Female | 50 | 30 | p.Y1275X (c.3938C>G) |
Known mutations reported by previous studies
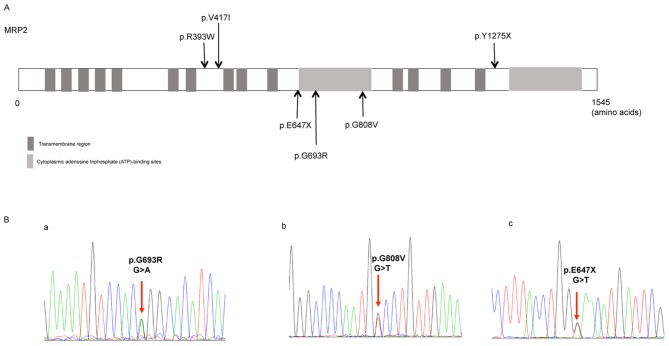
Six different ABCC2 gene variants including three known variants [p.R393W (c.1290C>T) (10), p.Y1275X (c.3938C>G) (11) and p.V417I (c.1362G>A) (12)] were identified across the 7 patients, and the distribution of these variants is shown in Fig. 1A. The missense mutation p.R393W (c.1290C>T) and nonsense mutation p.Y1275X (c.3938C>G) are known disease mutations according to the database HGMD. In addition, the missense variant p.V417I (c.1362G>A) in exon 10 harbored by the case no. 6 has previously been reported as a single-nucleotide polymorphism (SNP) (12). Further details on the variants identified in the ABCC2 gene are shown in Table III.
Figure 1.
Distribution of the identified ABCC2 variants and the representative sequencing of the three novel variants. (A) Distribution of the six ABCC2 gene variants identified in the 7 patients. Three of these variants have already been reported in previous studies (p.R393W, P.V417I and p.Y1275X), whereas the other three are novel, and are located at or close to the cytoplasmic adenosine triphosphate-binding sites. (B) Sequencing of the heterozygous missense mutations (a) c.2190G>A in exon 16 and (b) c.2536G>T in exon 18, and (c) the heterozygous nonsense mutation c.2052G>T in exon 15. ABCC2, adenosine triphosphate-binding cassette subfamily C member 2.
Table III.
Functional analysis of the variants identified in the ABCC2 gene.
| Location | Base change | Amino acid change | Variation types | Patient frequency | Allele frequency (Asian) | Allele frequency (total) | Polyphen-2 prediction | SIFT prediction | HGMD gene result |
|---|---|---|---|---|---|---|---|---|---|
| Exon 9 | c.1290C>T | p.R393W | Missense | 2/7 | 1.817×10−4 | 3.295×10−5 | Probably damaging | Deleterious | Known disease mutation |
| Exon 10 | c.1362G>A | p.V417I | Missense | 1/7 | 2.785×10−1 | 1.953×10−1 | Benign | Tolerated | rs2273697 |
| Exon 15 | c.2052G>T | p.E647X | Nonsense | 2/7 | 6.061×10−5 | 8.248×10−6 | NA | NA | NA |
| Exon 16 | c.2190G>A | p.G693R | Missense | 2/7 | 2.442×10−4 | 4.139×10−5 | Probably damaging | Deleterious | NA |
| Exon 18 | c.2536G>T | p.G808V | Missense | 1/7 | NA | NA | NA | NA | NA |
| Exon 27 | c.3938C>G | p.Y1275X | Nonsense | 1/7 | 4.594×10−4 | 6.59×10−5 | NA | NA | Known disease mutation |
ABCC2, adenosine triphosphate-binding cassette subfamily C member 2.
Novel variants identified in the present study
Of the six variants identified, three were novel variants, including the missense variants p.G693R (c.2190G>A) and p.G808V (c.2536G>T), and the nonsense variant p.E647X (c.2052G>T; Fig. 1B). Software prediction demonstrated that the novel missense mutation p.G693R (c.2190G>A) is probably damaging, while p.E647X is a typical deleterious mutation (Table III). The reports of allele frequency and functional prediction were absent for p.G808V (Table III).
Correlation between the ABCC2 genotypes and histology of liver biopsy
All the identified variants were heterozygous. Among the cases enrolled, 1 patient (case no. 4) presented with compound heterozygous mutations, namely p.G693R/p.G808V, while in another patient (case no. 6), p.R393W/p.V417I was identified, with the p.V417I considered as an SNP (12). Other cases carried only one single mutation.
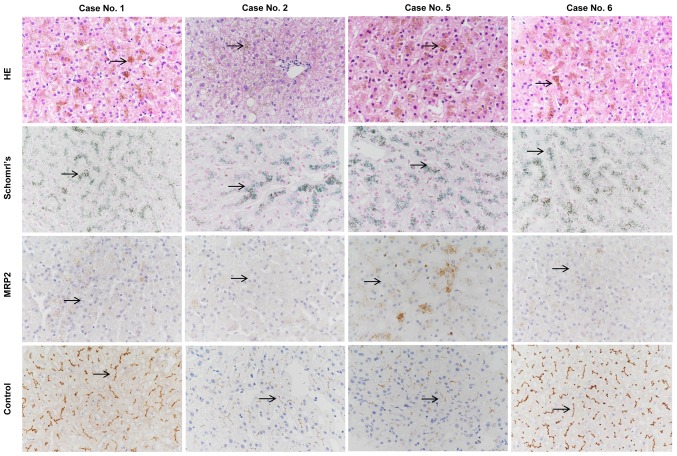
Liver biopsies and immunohistochemical analysis were performed in cases no. 1 and 5 presenting with the novel nonsense variant p.E647X, and in cases no. 2 and 6 presenting with the known missense mutation p.R393W. HE and Schmorl's staining of the liver biopsies demonstrated distinctive melanin-like, dark brown pigment accumulation in the hepatocytes in all 4 cases (Fig. 2).
Figure 2.
Representative HE, Schmorl's and immunohistochemical staining for MRP2 expression in patients with DJS. In cases with the novel p.E647X variant (patients no. 1 and 5) and cases with the known p.R393W variant (patients no. 2 and 6), accumulation of the distinctive dark brown pigment (black arrow) in the hepatocytes was observed with HE (brown) and Schmorl's (blue) staining of liver biopsy tissues. Immunohistochemical analysis revealed that MRP2 expression was absent in the patient with DJS (black arrow), but was positive in control liver (black arrow). Original magnification, ×400. MRP2, multidrug resistance-associated protein 2; HE, hematoxylin and eosin; DJS, Dubin-Johnson syndrome.
Immunohistochemical analysis of the MRP2 protein was negative in all 4 cases compared with the normal liver tissues, which were used as positive controls. Thus, the loss of membrane expression of MRP2 caused by the novel nonsense variant p.E647X and the known missense variant p.R393W was confirmed by the immunohistochemical analysis of liver biopsy (Fig. 2).
Discussion
ABCC2 mutations have been identified in patients with DJS worldwide; however, little is known regarding the mutation pattern of ABCC2 in Chinese patients (13). In the present study, through the mutation analysis of 7 clinically confirmed cases of DJS, it was observed that all cases harbored at least one non-synonymous variant in the ABCC2 gene. All the identified mutations were heterozygous, with only 2 cases presenting with compound heterozygous mutations. Thus, to the best of our knowledge, the present study is the first to report the mutation patterns of the ABCC2 gene in Chinese patients with DJS, as well as their clinical association with DJS.
DJS is an autosomal recessive disorder, and the majority of mutations in this syndrome are truncation mutations, including frame shift mutations, nonsense mutations and splice site mutations. To date, 55 ABCC2 gene mutations have been reported. Most of the reported mutations are predicted to cause defects in MRP2 protein maturation and localization, leading to greatly reduced biliary secretion of organic anions (5). The missense mutation p.R393W (c.1290C>T) (10) and the nonsense mutation p.Y1275X (c.3938C>G) (11) are known disease mutations for DJS previously identified in Asian patients. In the present study, three novel ABCC2 mutations we reported in cases with DJS, including missense mutations p.G693R and p.G808V, and nonsense mutation p.E647X. Software predictions of the biological effects of these mutations revealed that p.G693R is probably damaging, while the p.E647X mutation may lead to truncated MRP2 protein. For p.G808V, the reports of allele frequency and functional prediction were absent.
Notably, DJS is an autosomal recessive disorder; therefore, only homozygous variants in target genes are traditionally considered to cause disease phenotypes. In the present study, although all cases presented phenotypes, the identified genetic variants were all heterozygous, and 2 of the 7 cases harbored compound heterozygous mutations. Recently, studies have demonstrated that autosomal recessive diseases can also be caused by compound heterozygous genotypes. For instance, combinations of missense mutations of MRP2 have been reported in 2 Japanese patients, including p.R393W/p.R768W (10) and p.W709R/p.R1310X (14). In the current study, one novel combination of missense mutations, p.G693R/p.G808V, was reported in the ABCC2 gene. However, in the remaining 6 cases only single heterozygous mutations were identified, suggesting that variants in other associated, yet currently unknown, genes may exist, or that other non-genetic factors may serve a synergistic role. This highlights the importance of future studies involving mutation analysis of the whole genome using next generation sequencing. In our future study with a larger sample size, whole exome sequencing will be conducted for the case with only one heterozygous mutation, and the synergistic role of the non-genetic factors will be explored.
However, there were several limitations in the current study. Firstly, certain novel variants identified in the present study were only predicted by software as deleterious. However, the biological functional analysis of the novel mutations was not conducted with a cell line model, and further functional studies are required to understand the real effect of these mutations. Functional analysis with cell line model will be performed in the future. Furthermore, the number of cases was limited, and whole exon sequencing of cases with single mutations would be helpful to further understand their role in the development of DJS. A greater number of patients with DJS will be enrolled to enlarge the sample size, and whole exon sequencing of cases with single mutations will be conducted in the future.
In conclusion, to the best of our knowledge, the present study is the first to report the mutation patterns of the ABCC2 gene in Chinese patients with DJS and their clinical association with this disorder. The present study may provide information that is essential for understanding of the role of ABCC2 mutations in the development of DJS and may provide a genetic basis for the diagnosis of DJS in China.
Acknowledgements
The authors kindly acknowledge Dr. Yun Zhang (Biobank of Clinical Resources, Beijing Friendship Hospital, Beijing, China) for blood sample storage.
Glossary
Abbreviation
- DJS
Dubin-Johnson syndrome
- ABCC2
adenosine triphosphate-binding cassette subfamily C member 2
- MRP2
multidrug resistance-associated protein 2
Funding
The present study was supported by grants from the National Key Technologies R&D Program of China (grant no. 2015BAI13B09).
Availability of data and materials
All the data were collected from the hospital information system and can be available from the corresponding author upon reasonable request.
Authors' contributions
LW collected and analyzed the clinical data, and drafted the article. JH and XO conceived the paper and revised the manuscript. JJ analyzed the data in the manuscript and supervised the study. SJ performed the polymerase chain reaction amplification analysis of the ABCC2 gene. XZ performed histopathological analysis of the liver tissues of patients. WZ, DZ, AX, WD, ZW, HL, SZ and YN collected the clinical data. All the authors approved the final version of the manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of the Beijing Friendship Hospital at Capital Medical University (Beijing, China).
Patient consent for publication
Written informed consent was obtained from all patients for the publication of this article and any accompanying images.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Dubin IN, Johnson FB. Chronic idiopathic jaundice with unidentified pigment in liver cells; A new clinicopathologic entity with a report of 12 cases. Medicine (Baltimore) 1954;33:155–197. doi: 10.1097/00005792-195409000-00001. [DOI] [PubMed] [Google Scholar]
- 2.Frank M, Doss M, de Carvalho DG. Diagnostic and pathogenetic implications of urinary coproporphyrin excretion in the dubin-johnson syndrome. Hepatogastroenterology. 1990;37:147–151. [PubMed] [Google Scholar]
- 3.Morita M, Kihara T. Intravenous cholecystography and metabolism of meglumine iodipamide (biligrafin) in dubin-johnson syndrome. Radiology. 1971;99:57–60. doi: 10.1148/99.1.57. [DOI] [PubMed] [Google Scholar]
- 4.Shani M, Seligsohn U, Gilon E, Sheba C, Adam A. Dubin-johnson syndrome in israel. I. Clinical, laboratory, and genetic aspects of 101 cases. Q J Med. 1970;39:549–567. [PubMed] [Google Scholar]
- 5.Gazzin S, Masutti F, Vitek L, Tiribelli C. The molecular basis of jaundice: An old symptom revisited. Liver Int. 2017;37:1094–1102. doi: 10.1111/liv.13351. [DOI] [PubMed] [Google Scholar]
- 6.Wakusawa S, Machida I, Suzuki S, Hayashi H, Yano M, Yoshioka K. Identification of a novel 2026G->C mutation of the MRP2 gene in a Japanese patient with Dubin-Johnson syndrome. J Hum Genet. 2003;48:425–429. doi: 10.1007/s10038-003-0052-0. [DOI] [PubMed] [Google Scholar]
- 7.Erlinger S, Arias IM, Dhumeaux D. Inherited disorders of bilirubin transport and conjugation: New insights into molecular mechanisms and consequences. Gastroenterology. 2014;146:1625–1638. doi: 10.1053/j.gastro.2014.03.047. [DOI] [PubMed] [Google Scholar]
- 8.Keitel V, Nies AT, Brom M, Hummel-Eisenbeiss J, Spring H, Keppler D. A common Dubin-Johnson syndrome mutation impairs protein maturation and transport activity of MRP2 (ABCC2) Am J Physiol Gastrointest Liver Physiol. 2003;284:G165–G174. doi: 10.1152/ajpgi.00362.2002. [DOI] [PubMed] [Google Scholar]
- 9.Hashimoto K, Uchiumi T, Konno T, Ebihara T, Nakamura T, Wada M, Sakisaka S, Maniwa F, Amachi T, Ueda K, Kuwano M. Trafficking and functional defects by mutations of the ATP-binding domains in MRP2 in patients with dubin-johnson syndrome. Hepatology. 2002;36:1236–1245. doi: 10.1053/jhep.2002.36368. [DOI] [PubMed] [Google Scholar]
- 10.Machida I, Wakusawa S, Sanae F, Hayashi H, Kusakabe A, Ninomiya H, Yano M, Yoshioka K. Mutational analysis of the MRP2 gene and long-term follow-up of dubin-johnson syndrome in japan. J Gastroenterol. 2005;40:366–370. doi: 10.1007/s00535-004-1555-y. [DOI] [PubMed] [Google Scholar]
- 11.Lee JH, Chen HL, Chen HL, Ni YH, Hsu HY, Chang MH. Neonatal dubin-johnson syndrome: Long-term follow-up and MRP2 mutations study. Pediatr Res. 2006;59:584–589. doi: 10.1203/01.pdr.0000203093.10908.bb. [DOI] [PubMed] [Google Scholar]
- 12.Shen C, Zhang B, Liu Z, Tang Y, Zhang Y, Wang S, Guo Y, Ding Y, Wang S, Ding M. Effects of ABCB1, ABCC2, UGT2B7 and HNF4α genetic polymorphisms on oxcarbazepine concentrations and therapeutic efficacy in patients with epilepsy. Seizure. 2017;51:102–106. doi: 10.1016/j.seizure.2017.07.015. [DOI] [PubMed] [Google Scholar]
- 13.Xiang R, Li JJ, Fan LL, Jin JY, Xia K, Wang F. Identification of a compound heterozygous mutation of ABCC2 in a patient with hyperbilirubinemia. Mol Med Rep. 2017;16:2830–2834. doi: 10.3892/mmr.2017.6926. [DOI] [PubMed] [Google Scholar]
- 14.Uchiumi T, Tanamachi H, Kuchiwaki K, Kajita M, Matsumoto S, Yagi M, Kanki T, Kang D. Mutation and functional analysis of ABCC2/multidrug resistance protein 2 in a japanese patient with dubin-johnson syndrome. Hepatol Res. 2013;43:569–575. doi: 10.1111/j.1872-034X.2012.01103.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data were collected from the hospital information system and can be available from the corresponding author upon reasonable request.