Summary
Mast cells are tissue-resident immune cells. Their overgrowth/overactivation results in a range of common distressing, sometimes life-threatening disorders, including asthma, psoriasis, anaphylaxis, and mastocytosis. Currently, drug discovery is hampered by use of cancer-derived mast cell lines or primary cells. Cell lines provide low numbers of mature mast cells and are not representative of in vivo mast cells. Mast cell generation from blood/bone marrow gives poor reproducibility, requiring 8–12 weeks of culture. Here we report a method for the rapid/robust production of mast cells from pluripotent stem cells (PSCs). An advantageous Gata2Venus reporter enriches mast cells and progenitors as they differentiate from PSCs. Highly proliferative mouse mast cells and progenitors emerge after 2 weeks. This method is applicable for rapid human mast cell generation, and could enable the production of sufficient numbers of physiologically relevant human mast cells from patient induced PSCs for the study of mast cell-associated disorders and drug discovery.
Key words: mast cells, ESC, iPSC, differentiation, Gata2, stem cells, innate immune cells, immune effectors, Venus reporter, rapid protocol
Graphical Abstract
Highlights
-
•
Efficient mast cell production is achieved with novel Gata2-reporter PSCs
-
•
14-day Gata2-reporter PSC culture produces mast cells and self-renewing progenitors
-
•
Gata2-reporter mast cells have high protease content and degranulate
-
•
Gata2-reporter iPSC method advances prospects for human mast cell research
In this article, Dzierzak and colleagues show the rapid and robust production of physiologically relevant mast cells from mouse ESCs via an innovative approach using a Gata2 reporter. This method is applicable to human mast cell production and provides the opportunity for research into patient-specific mast cell disorders.
Introduction
As tissue-resident immune cells, mast cells are the key effectors in common immunological disorders affecting worldwide populations. These include allergies, asthma, eosinophilic esophagitis, celiac disease, mastocytosis, atopic dermatitis, and psoriasis. A characteristic feature of mast cells is the large cytoplasmic granules containing proteases that are released upon activation, for example during an allergic reaction. Activation of mast cells occurs via a number of different agents: immunoglobulin E (IgE), damage-/pathogen-induced molecules, and complement components (reviewed in Rudich et al., 2012). These cells act viscerally in the CNS and also at the blood-brain barrier, causing a wide variety of symptoms in complex diseases that are difficult and often impossible to diagnose. More recently they are suspected to affect pain and neuroinflammation in endometriosis (Theoharides et al., 2015). Strategies to limit the damaging effects of mast calls are needed.
Dependent upon their tissue of residence, mast cells differentially produce a variety of proteases, such as chymase, tryptase and/or histamine (Schwartz and Austen, 1980). Murine connective tissue-type mast cells express heparin, high concentrations of histamine, tryptases (mMCP-6 and -7), chymases (mMCP-4 and -5), and carboxypeptidase A (CPA), whereas mucosal mast cells lack heparin and express low levels of histamine and chymases (mMCP-1 and -2) (Galli et al., 2011). Mast cells in the human also differentially express chymase and tryptase, with classical mast cells being tryptase+chymase+ and tryptase+chymase− (Bischoff, 2007, Weller et al., 2007). Recently a new mast cell type (tryptase−chymase+) has been found in endometrial tissue (De Leo et al., 2017). The fact that these endometrial mast cells express steroid receptors suggests that mast cell function is altered by the local microenvironment.
Treatment options focusing on the modulation of mast cell activity are limited, and the development of new therapies is hampered by challenges in generating sufficient phenotypically mature mast cells for biomedical discovery and drug screening (Holm et al., 2006, Saito et al., 2006, Shimizu et al., 2002, Wang et al., 2006). Direct isolation of mast cells is inefficient (Dahlin et al., 2016) and results in an altered phenotype. Moreover, the ex vivo generation of mast cells from human blood precursors involves extended culture periods, expensive reagents, and low/variable yields (Kirshenbaum and Metcalfe, 2006). Pluripotent stem cells (PSCs) offer an alternative source for obtaining mature mast cells for research. However, the published protocols are time consuming, as mast cells emerge after 4–8 weeks of mouse PSC culture (Moller et al., 2007, Tsai et al., 2002, Westerberg et al., 2012, Yamaguchi et al., 2013) and only after 5–10 weeks of human PSC culture (Kovarova et al., 2010) (Table 1). Further prolonged culture is needed to increase mast cell yield, as the cells are cumulatively harvested and do not enable prompt production of large numbers of mast cells. The lack of an efficient protocol to rapidly obtain large numbers of mature mast cells for research has restricted drug development and progress in understanding and treating mast cell-related disorders; thus, new approaches for mast cell production are needed.
Table 1.
Duration of Cultures Used for Mast Cell Generation from Different Cell/Tissue Sources
| Source | Culture Time | Reference |
|---|---|---|
| hPB | 8–12 weeks | Kirshenbaum and Metcalfe, 2006, Saito et al., 2006, Wang et al., 2006 |
| hCB | 12 weeks | Holm et al., 2006 |
| hBM | 12 weeks | Shimizu et al., 2002 |
| mESC | 4–8 weeks | Moller et al., 2007, Tsai et al., 2002, Westerberg et al., 2012 |
| miPSC | 4 weeks | Yamaguchi et al., 2013 |
| hESC | 10 weeks | Kovarova et al., 2010 |
| hiPSC | 5 weeks | Igarashi et al., 2018 |
| G2V mESC | 14–21 days | this study |
| G2V hESC/iPSC | 12–16 days | this study |
Duration (weeks/days) of cultures for human (h) mast cell generation from progenitors isolated from primary tissue sources such as peripheral blood (hPB), cord blood (hCB), and bone marrow (hBM); from wild-type and Gata2Venus (G2V) reporter mouse embryonic stem cells (mESC); and human ESCs (hESC) and induced pluripotent stem cells (iPSC). References for each of the studies are indicated.
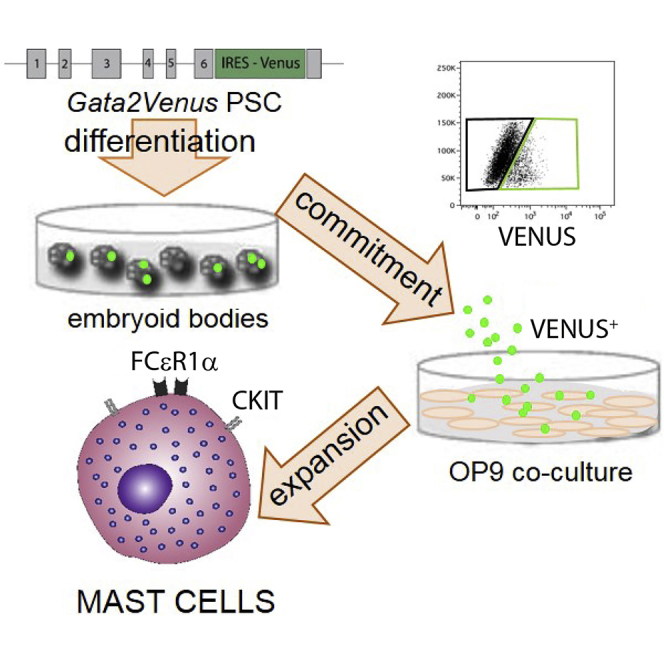
Here we report an innovative method for the rapid and abundant production of mouse mast cells that relies on Gata2Venus (G2V) reporter PSCs (Kaimakis et al., 2016), and show that it is also applicable for the generation of human mast cells. The rationale for this reporter is that the Gata2 transcription factor is highly expressed in mast cells (Jippo et al., 1996). Its expression is essential for mast cell precursor development and expansion (Tsai and Orkin, 1997) and the function of mature mast cells (Masuda et al., 2007). Gata2-deficient mouse and human embryonic stem cells (ESCs) show abrogated hematopoietic differentiation (Huang et al., 2015, Tsai et al., 1994). Previously, we showed in mouse G2V ESCs and G2V embryos that Venus reporter expression mirrors that of Gata2 without affecting Gata2 expression levels (Kaimakis et al., 2016, Kauts et al., 2016, Kauts et al., 2018). Gata2 is expressed in all hematopoietic stem cells (HSCs) and most progenitors (HPCs) in vivo (Kaimakis et al., 2016). With the exception of mast cells and basophils (Sasaki et al., 2016), Gata2 expression is downregulated when immature HPCs differentiate, thus making it a potentially specific reporter for the mast/basophilic cell lineage (Akashi et al., 2000, Guo et al., 2013, Miyamoto et al., 2002, Orlic et al., 1995). We demonstrate here the rapid and efficient production of mast cells from mouse G2V reporter ESCs, and show that this reporter-based system is applicable for the rapid production of mast cells from human G2V ESCs and induced PSCs (iPSCs).
Results
Abundant Production of Phenotypic Mast Cells from Mouse ESCs
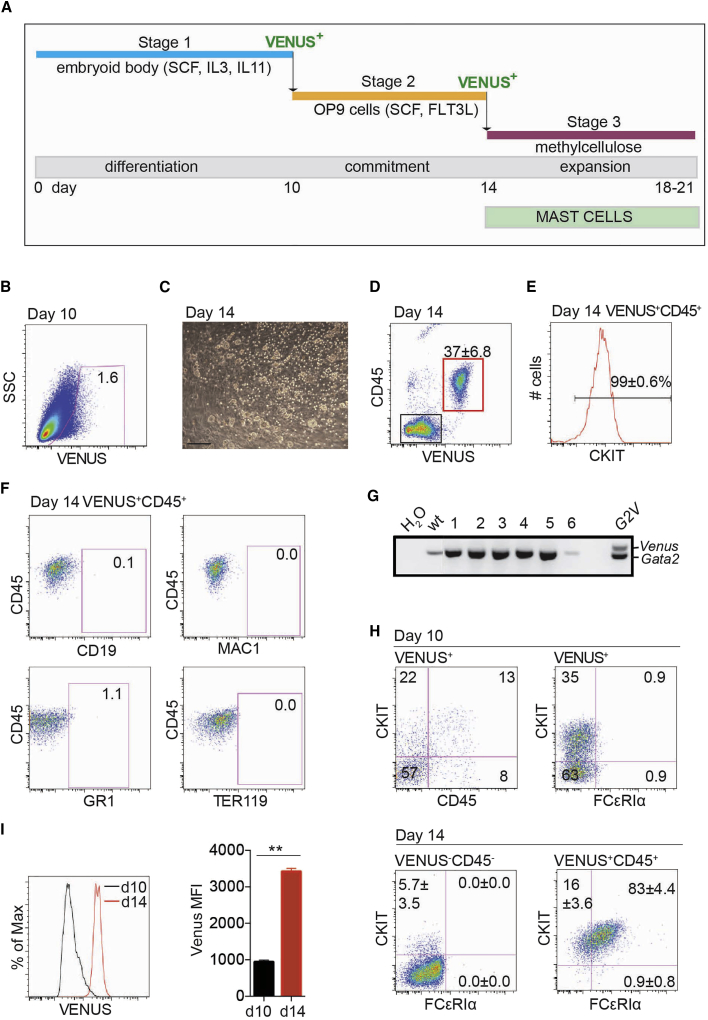
Our recent data show that all functional HPCs generated in mouse ESC (mESC) differentiation cultures are Gata2 expressing, with a peak of HPC activity at day 10 of ESC culture (Kauts et al., 2016, Kauts et al., 2018). With an initial aim to test whether further hematopoietic induction would lead to the development of transplantable HSC/HPC, a three-stage culture was established. In stage 1, G2V mESCs were differentiated to embryoid bodies (EBs) for 10 days (Figure 1A). Venus+ (V+) cells (1.6% of viable EB-derived cells; Figure 1B) were harvested and in stage 2, cultured on a monolayer of OP9 stromal cells for 4 days. The average number of V+ cells obtained from day-10 EBs (3 × 104 starting ESCs) was 1.5 ± 0.3 × 104 (Table 2). Round non-adherent hematopoietic cells appeared in the OP9 co-culture after 2–3 days (Figure 1C) and after 4 days, 37% ± 6.8% of the cells expressed high levels of Venus (Figure 1D). Only V+ cells specifically co-expressed the pan-leukocyte marker CD45, and 99% ± 0.6% of V+CD45+ cells were positive for the CKIT (CD117) HSC/HPC marker (Figure 1E). In the stage 3 culture, day-14 V+ cells were seeded in methylcellulose and progenitor numbers analyzed at days 18–21.
Figure 1.
G2V mESC Differentiation and VENUS Enrichment Facilitates Rapid and Robust Mast Cell Generation
(A) Three-stage differentiation protocol for mast cell production. Stage 1 is hematopoietic commitment in 10-day embryoid body (EB) culture. At day 10, GATA2VENUS+ (V+) cells are sorted and plated onto OP9 cells in stage 2, where mast cell commitment is induced to yield highly proliferating mast cells that appear after 3–4 days. At day 14, V+ mast cells/progenitors are isolated for analysis/further clonal expansion in stage 3.
(B) FACS dot plot of cells from day 10 G2V mouse EBs showing gating used for sorting V+ cells (1.6%) after stage 1.
(C) Visible-light image of a 4-day OP9 co-culture in which round refractive mast cells are observed overlaying the adherent large OP9 cells. Scale bar, 100 μm.
(D) FACS analysis of cells harvested after 4 days of OP9 co-culture. 37% ± 6.8% of cells are V+ and CD45+ (red gated area).
(E) 99% ± 0.6% of V+CD45+ cells shown in (D) are CKIT+ hematopoietic cells. Mean ± SEM, number of independent experiments (n) = 6.
(F) FACS dot plots showing lack of B-lymphoid (CD19), macrophage (MAC1), granulocyte/basophil (GR1), and erythrocyte (TER119) lineage-positive cells in V+CD45+ population after 4 days of OP9 co-culture.
(G) DNA PCR data of six representative spleen colonies from CFU-S assay testing the in vivo repopulation potential of V+CD45+Lin− ESC-derived cells. Sorted cells injected into irradiated mice formed a few small spleen colonies at 9 days post injection. PCR for G2V and Gata2 alleles shows that the CFU-S did not originate from the ESC-derived cells. Negative control is wild-type (wt) ESC DNA. Positive control is G2V ESC DNA.
(H) FACS analysis of (top) day-10 V+ cells for CKIT, CD45, and FCɛR1α expression shows commitment to hematopoietic progenitors but not mature mast cells. FACS analysis of day-14 V−CD45− cells (bottom left) and V+CD45+ cells (bottom right; 83% mast cells) isolated after stage 2. Mean ± SEM, n = 6.
(I) FACS histogram of VENUS expression intensity of day-10 (black) and day-14 (red) differentiated cells. Bar graph quantification of VENUS mean fluorescence intensity (MFI) at days 10 (black) and 14 (red) showing significantly higher Venus protein levels in day-14 mast cells. Mean ± SEM, ∗∗p < 0.01, n = 3.
Table 2.
Mast Cell Generation and Frequency in the G2V mESC Multistep Cultures
| Experiment | Stage 1: V+ Cells from 3 × 104 ESCs | Stage 2: MC/MCp from 3 × 104 ESCs | Stage 3: MC Yield from 3 × 104 ESCs |
|---|---|---|---|
| 1 | 11,504 | 14,923 | 1,242,200 |
| 2 | 30,000 | 2,781 | 937,000 |
| 3 | 7,541 | 2,623 | 3,834,375 |
| 4 | 9,653 | 1,990 | 570,313 |
| 5 | 20,087 | 6,421 | 1,156,250 |
| 6 | 13,764 | 6,875 | 1,906,250 |
| Mean | 15,425 | 5,936 | 1,607,731 |
| ±SEM | ±3,405 | ±1,986 | ±479,952 |
Number of VENUS+ cells after stage 1 at day 10, VENUS+ mast cells/mast cell progenitors (MC/MCp) after OP9 co-culture stage 2 at day 14, and total mast cell yield after stage 3 methylcellulose expansion at days 18–21 from 3 × 104 input Gata2Venus mouse ESCs; six independent experiments, and mean ± SEM.
To assess the V+CD45+ cell population after stage 2, we analyzed mature blood lineage (Lin) marker expression: MAC1 (macrophage), GR1 (granulocyte/monocyte), CD19 (lymphocyte), and TER119 (erythroid) (Figure 1F). None of these markers were expressed on day-14 V+CD45+ cells. As phenotypic HSCs/HPCs are Lin− and express GATA2 (Kaimakis et al., 2016, Kauts et al., 2016, Kauts et al., 2018), CKIT (Sanchez et al., 1996) and CD45 (McKinney-Freeman et al., 2009, North et al., 2002), we examined whether functional HSC/HPC-like cells were generated in the differentiation culture by the in vivo colony forming unit-spleen assay (CFU-S). At day 8 post transplantation of day-14 V+CD45+Lin− cells into lethally irradiated adult mice (two injected with 1,200 and one injected with 20,000 cells), no donor-derived CFU-S activity was found (Figure 1G).
Since mast cells are also known to express GATA2, CKIT, and CD45 (Dahlin et al., 2016, Jippo et al., 1996, Sasaki et al., 2016), we examined differentiated ESCs for expression of the high-affinity IgE receptor 1α (FCɛRIα), a mast cell marker. At day 10, 35%–36% of V+ cells were CKIT+ (Figure 1H, upper panels); 37% of these V+CKIT+ cells were CD45+ and only 2.5% of V+CKIT+ cells were FCɛRIα+. In contrast, 83% ± 4.4% of V+CD45+CKIT+ cells after day 14 were FCɛRIα+ (Figure 1H, lower right panel). 5.7% ± 3.5% of day-14 V−CD45− cells showed some low-level CKIT expression, but none of these cells were FCɛRIα+ (Figure 1H, lower left panel). These data suggest that at day 14, most V+CKIT+CD45+ cells are mast cells/progenitors and that at day 10 most V+CKIT+CD45+/− cells are phenotypic HPCs. As it is thought that mast and basophilic cells express higher levels of GATA2 than HPCs (Kaimakis et al., 2016), the mean fluorescent intensity (MFI) of VENUS expression was examined (Figure 1I). Compared with day-10 differentiated cells, the day-14 cells exhibited a 3.5-fold higher MFI. To further verify the mast cell phenotype of the ESC-derived V+ cells, we examined the expression of a basophilic marker, CD49B, on these cells by fluorescence-activated cell sorting (FACS) analysis (Figure S1). Basophils are known to be CKIT−, FCɛRIα+, and CD49B+. Compared with cultured mouse bone marrow CD45+ cells (Figure S1C), which show both CD49B− mast cells and CD49B+ basophils, only a negligible percentage of ESC-derived cells (Figure S1A) and cultured peritoneal mast cells (Figure S1B) were CD49B+, thus demonstrating that the great majority of ESC-derived cells have acquired a mast cell and/or mast cell progenitor phenotype after 14 days.
Development and Expansion of G2V Mast Cells
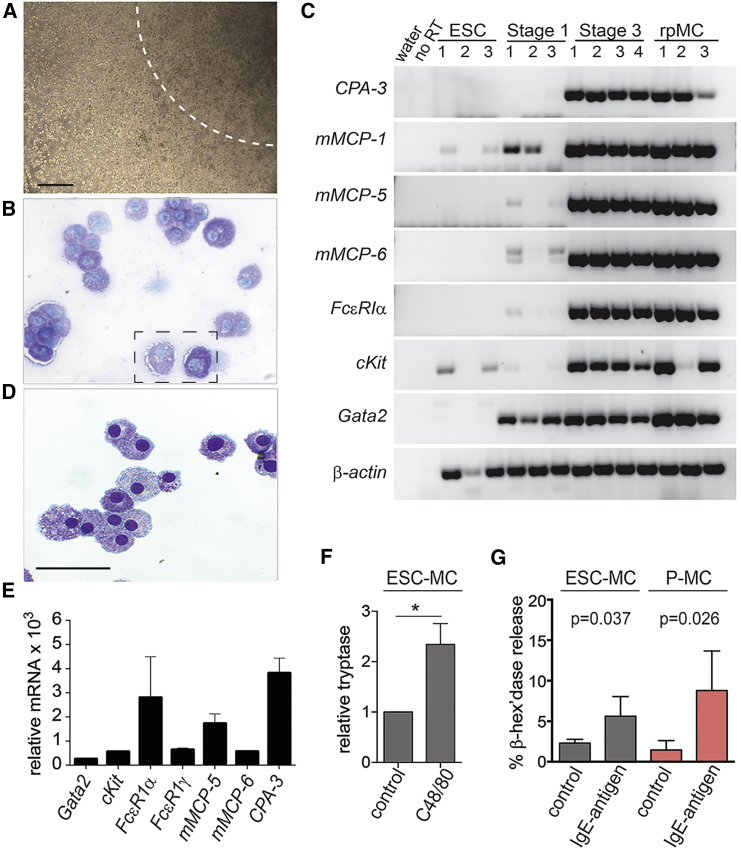
To analyze the growth capacity of the V+ mast cells/progenitors, we plated single V+ cells harvested from day-4 OP9 co-cultures in methylcellulose stage 3 culture. After 3–4 days, macroscopic homogeneous colonies appeared (Figure 2A), indicating a large degree of rapid expansion. In total, a starting culture of 3 × 104 ESCs can yield up to 3.8 × 106 mature mast cells (average 1.6 ± 0.5 × 106) (Table 2). This is up to 8-fold more cells generated in 21 days than previously reported after 5 or more weeks of mESC culture (0.5 × 106 cells) (Westerberg et al., 2012), and 2-fold more mast cells 1 week earlier than from mouse iPSCs (Yamaguchi et al., 2013).
Figure 2.
G2V ESC-Derived V+ Cells Generate Mucosal and Connective Tissue-Type Mast Cells that Express Proteases and Degranulate
(A) Image showing the clonal expansion capacity of a single V+ mast cell harvested from stage 2 after 5 days. Area within the dotted region shows part of such a colony (>1,000 cells). Scale bar, 100 μm.
(B) Image of toluidine blue-stained mast cells generated after stage 3 expansion. Lighter blue indicates mucosal mast cells (mMC) and darker blue connective tissue-like mast cells (ctMC).
(C) Mast cell-specific gene expression in undifferentiated ESCs (number of independent experiments [n] = 3), day-10 V+ hematopoietic cells (stage 1, n = 3), expanded mast cells (stage 3, n = 4) and mast cells after clonal replating (rpMC, n = 3). RT, reverse transcriptase; CPA, carboxypeptidase; mMCP, mouse mast cell protease.
(D) Image of toluidine blue-stained mast cells generated after two serial clonal replatings of a single cell harvested from stage 2 co-culture. Scale bar, 100 μm.
(E) Quantitative gene expression analysis of mast cell-specific genes in expanded G2V mast cells. Expression levels were normalized to 18S expression and compared with the normalized levels in control mouse ear tissue. Mean ± SEM, n = 3.
(F) ELISA assay showing the relative concentration of released tryptase in the medium of c48/80-treated (5 μM) G2V mast cells compared with untreated cells. Tryptase levels were calculated based on a tryptase standard curve. Level was set as 1 in untreated sample. ∗p < 0.05, n = 5.
(G) Immunoglobulin E (IgE) activation of degranulation. Functional assay showing the percentage of β-hexosaminidase release in the supernatant of IgE activation/antigen stimulation-treated ESC-MCs and peritoneal MC (P-MC) compared with controls. β-Hexosaminidase enzymatic activity was measured in supernatants and cell pellets and percent release was calculated as described in Experimental Procedures. Unpaired t test, two-tailed p value; n = 4.
Error bars represent means ± SEM.
Mast cell identity was confirmed by toluidine blue staining (cytoplasmic granule-specific). One hundred percent of the cells after stage 3 culture were toluidine blue positive, but to varying degrees. In contrast to the adult connective tissue-type mast cells in the skin that are dark-staining (due to heparin) and mucosal-type mast cells in the gut and lung that are weak-staining (Bischoff, 2007, Weller et al., 2007), the staining differences in the ESC-derived mast cells are likely to represent different stages of differentiation (Figure 2B).
G2V Mast Cells Express Proteases and Receptors Characteristic of Mature Mast Cells
Mature mast cells produce high levels of inflammatory mediators that are stored in large cytoplasmic secretory granules (Schwartz and Austen, 1980). The expression of genes encoding the inflammatory mediators of connective tissue mast cells (mMCP-5, mMCP-6, and CPA-3) and of mucosal mast cells (mMCP-1) was examined in undifferentiated mESCs, V+ stage 1 cells, and stage 3 expanded mast cells, and after serial clonal replating (rpMC) (Figure 2C). CPA-3, mMCP-1, mMCP-5, and mMCP-6 were highly expressed in all mast cell samples before and after serial replating, with only low/negligible expression in some ESCs or stage 1 samples. Mast cell receptor genes cKit and FcɛR1α were expressed before and after replating in all mast cell samples, with some low cKit expression in ESCs. High levels of Gata2 were detected in stage 1 cells and in stage 3 mast cells before and after replating. The fact that the clonal expansion capacity, cellular morphology (Figure 2D), and gene expression profile (Figure 2C) of the mast cells were retained after serial replating suggests that these cultures maintain self-renewing cells, most likely mast cell progenitors (MCp).
Gene expression levels of the chemical mediators and mast cell receptors in G2V ESC-derived mast cells was compared with expression in control murine ear tissue (the ear is known to contain a high frequency of mast cells). cKit, FcɛR1α, and FcɛR1γ were respectively expressed 560 ± 39, 2,802 ± 1,690 and 644 ± 66 times more than the control, and mMCP-5, mMCP-6, and CPA-3 were expressed at 1,727 ± 402, 570 ± 58, and 3,818 ± 621 times higher than in the control (Figure 2E). Moreover, G2V ESC-derived mast cells (CD45+CKIT+FCɛR1α+) and control peritoneal mast cells (CD45+CKIT+FCɛR1α+) did not express the basophilic protease gene mMcpt8, whereas cultured bone marrow CD45+CKIT−FCɛR1α+ cells expressed high levels of mMcpt8 (Figure S1D). As expected, cKit mRNA was found in both ESC and peritoneal mast cells but not in basophils. These gene expression data confirm the highly enriched and rapid production of mast cells from G2V ESCs and suggest that these mast cells are functional.
G2V Mast Cells Are Activated upon Extracellular Stimulation
Allergic responses in vivo result from the activation and degranulation of mast cells. To test whether G2V mast cells are functionally responsive, we treated cells with compound 48/80 (c48/80) (Lagunoff and Rickard, 1983), a known secretagog of mast cells. After 60 min of c48/80 stimulus (5 μg/mL) a significantly 1.6-fold higher tryptase level was found in the cell media as compared with the control unstimulated sample (Figure 2F). An FCɛR1α-mediated degranulation assay was also performed on G2V-mESC-derived mast cells and control mouse peritoneal mast cells (Figure 2G). In the absence of activation, some leakiness (as measured by percentage of β-hexosaminidase release) was observed. However, when IgE-sensitized G2V-mESC-derived mast cells were stimulated by adding antigen to the ESC-derived mast cells, a significant 2-fold increase in degranulation occurred that was in accordance with the increased degranulation levels (also significant) seen in the peritoneal mast cell controls. Together, these data demonstrate that the G2V ESC-derived mast cells can be activated and respond to common mast cell activating extracellular stimuli, triggering the release of chemical mediators.
Human G2V Reporter PSC Differentiation as an Innovative Approach for Rapid Human Mast Cell Production
In studies related to the involvement of mast cell in endometriosis, we generated mast cells from human CD34+ peripheral blood cells by a widely used method (Kirshenbaum and Metcalfe, 2006). Density gradient-enriched CD34+ progenitor cells were cultured in serum-free medium with growth factors for 8 weeks (Figure S2A). At 2 and 3 weeks, only a few low toluidine blue-stained cells were detected, whereas intensely blue cells indicative of mast cells predominated in 8-week cultures. Further characterization showed upregulated expression of human mast cell protease/receptor genes (Figure S2B). Little to no expression was found at week 1, but statistically significant 20-fold to 70-fold increases of TPSAB1 (tryptase α/β isoforms) and CMA1 (chymase1) expression were found at 4 and 8 weeks. PAR2 (protease activated receptor2) and HR1 (histamine receptor1) expression showed significant increases only at week 8. These results are consistent with the presence of mast cells at week 8 of PBC CD34+ culture. Also, a human mast cell line, HMC-1, showed significantly lower expression of the mast cell genes TPSAB1, CMA1, PAR2, and HR1 compared with 8-week differentiated CD34+ PBCs (Figure S2C), thus demonstrating that HMC1 is not a relevant alternative to overcome the challenge of long duration of mature human mast cell production, and highlighting the need for developing novel culture methods.
Our ability to rapidly produce mast cells from the G2V reporter mESCs, together with the known conservation in hematopoietic development and Gata2 expression/function in mouse and man, suggested that the GATA2 reporter could be applied to human PSCs (hPSCs) for the production of human mast cells. Thus, we established GATA2Venus reporter human ESCs (hESCs) and iPSCs in a strategy similar to that used with G2V mESCs. A 2A-Venus sequence was introduced into the human GATA2 (hGATA2) 3′ UTR by the CRISPR/Cas9 method (Figure S3A). One clone of GATA2Venus reporter human ESCs (G2V-hESC) and two independent clones of iPSCs (G2V-hiPCS1; G2V-hiPCS2) were characterized (Figure S3B) for GATA2 expression and hematopoietic differentiation. V+ sorted cells from day-6 differentiation cultures of all three cell lines expressed high levels of GATA2 compared with V− cells (Figure S3C). At day 12, flow cytometry showed that 38%–70% of cells were V+ and that 6%–18% of V+ cells expressed the CD41 hematopoietic marker (Figure S3D). Moreover, most HPCs (>90%) were enriched in the V+ fraction of differentiated G2V hESCs and hiPSCs (Figure S2E) as shown by in vitro CFU-cell assays. All erythroid and myeloid hematopoietic colony types were present. These results are in line with previous analyses of mESCs (Kauts et al., 2016, Kauts et al., 2018) and hESCs/hiPSCs (Huang et al., 2015), and show that GATA2 is an efficient reporter for human HPCs.
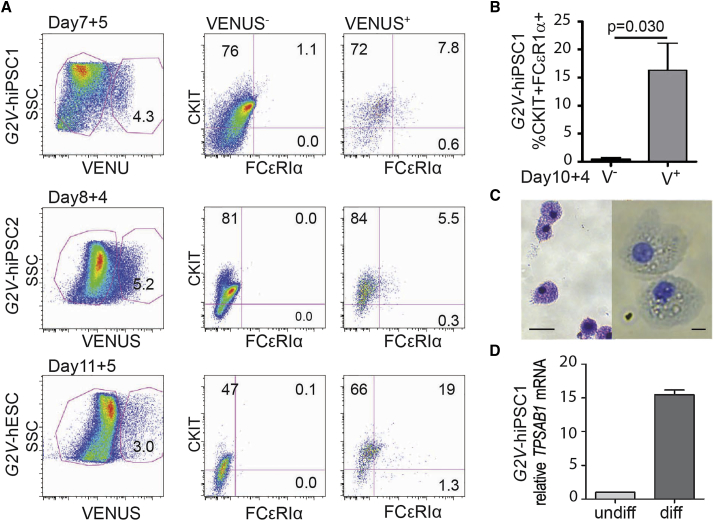
To test whether our rapid Gata2 reporter-based mouse mast cell generation method is also applicable for mast cell generation from hPSCs, we applied the staged differentiation culture protocol to the three independent cell lines, G2V-hiPSC-1, G2V-hiPSC-2, and G2V-hESC. Following 7, 8, and 11 days of differentiation, respectively, cells were sorted based on Venus expression and co-cultured with OP9 cells for 4–5 days. Of the 3.4 × 104, 8.9 × 104, and 3.4 × 104 viable cells harvested from the G2V-hiPSC-1, G2V-hiPSC-2, and G2V-hESC OP9 co-cultures, 4.3%, 5.2%, and 3.0%, respectively were Venus expressing, yielding 278, 241, and 906 CKIT+FCɛRIα+ phenotypic mast cells (7.8%, 5.5%, and 19%, respectively) (Figure 3A). No CKIT+FCɛRIα+ cells were detected in the V− fractions. In further experiments we focused on the G2V hiPSC-1 line (Figure 3B). After 10 days of differentiation and 4 days of OP9 co-culture, 16.30% ± 4.82% CKIT+FCɛRIα+ cells were detected in the cultures of V+ cells, whereas no to few CKIT+FCɛRIα+ cells (0.42% ± 0.33%) were found in the V− cell cultures (n = 3; p = 0.030). Toluidine blue and rapid Romanowsky staining of cells from G2V-hiPSC1 expanded cultures confirmed mast cell morphology (Figure 3C). Moreover, qRT-PCR verified increased expression of the mature mast cell protease, TPSAB1 in differentiated G2V-hiPSC1 as compared with undifferentiated cells (Figure 3D). The consistency of the results from the G2V-hESC and G2V-hiPSC clones indicate that our staged culture and enrichment approach, although not as quantitatively efficient as the G2V-mESC cultures, delivers a promising platform for the generation of human mast cells within 12–16 days of culture initiation.
Figure 3.
Generation of Human GATA2VENUS Mast Cells
(A) The differentiation approach (Figure 1A) was applied to G2V-hESC/hiPSC to generate human mast cells. V+ cells were sorted from three independent cell lines (day-7 G2V-hiPSC1, day-8 G2V-hiPSC2, and day-11 G2V-hESC differentiated EBs) and subjected to OP9 co-culture. After 4 days (G2V-hiPSC1) or 5 days (G2V-hiPSC2, G2V-hESC) in step 2 culture, V+ and V− cells were analyzed for mast cell markers CKIT and FCɛRIα by FACS. Percentages of cells in gated regions are shown (n = 1 independent experiment for each cell line/time frame).
(B) Bar graph showing percentages (mean ± SEM) of CKIT+FCɛRIα+ cells obtained from the VENUS+ and VENUS− fractions of cells harvested from day-10 differentiated and 4-day co-cultured G2V-hiPSC1 cells (n = 3; p = 0.030).
(C) Image of toluidine blue (left) and rapid Romanowsky (right) stained mast cells generated after 5 days of step 3 expansion culture. Scale bars, 50 μm (left) and 10 μm (right).
(D) Gene expression analysis of mast cell-specific protease tryptase (TPSAB1) in undifferentiated (n = 2) and differentiated G2V-hiPSC1 (n = 3) cells. GAPDH controlled for mRNA quantity. Relative expression levels were normalized to OP9 TPSAB1 expression, which was set as 1.
Discussion
The Gata2 reporter PSC approach represents a significant advance in the rapid production of mast cells for research (Table 1). Compared with previously published protocols using limited sources of cells such as human blood (peripheral, umbilical cord) and bone marrow, and unlimited PSC sources, the staged G2V differentiation and enrichment method shortens culture duration considerably (by 1 week for mouse and several weeks for human cultures). This time-saving and resource-efficient method raises prospects for production of mast cells (including patient-specific) for high-throughput drug discovery and mechanistic studies on mast cell-related disorders.
Gata2 Expression as a Reporter for Mast Cells
The Gata2 transcription factor is known to be expressed in HSCs, HPCs, and mast cells (Jippo et al., 1996, Kaimakis et al., 2016). As a nuclear factor it is not amenable to antibody-mediated viable cell enrichment by FACS sorting, and the generation of reporters that accurately mimic Gata2 without disrupting its expression levels or protein function has been difficult (Minegishi et al., 2003). We achieved faithful reporter gene expression by inserting an IRESVenus sequence into the Gata2 3′ UTR, thus precluding disruption of the coding sequences or regulatory elements, or functional modification (as in a fusion protein) of Gata2. Such an approach allows for the parallel expression of the Venus reporter together with the proper qualitative and quantitative expression of Gata2 (Eich et al., 2018). Importantly, our Gata2 reporter approach has been verified in vivo (Kaimakis et al., 2016) and shown to provide the appropriate physiologic levels of Gata2 expression for undisturbed hematopoietic development and differentiation. VENUS protein levels correctly correspond to GATA2 protein levels (Eich et al., 2018), thus guaranteeing appropriate reporting of GATA2 expression in the in vitro G2V ESC/iPSC models.
FACS analysis revealed varying levels of Venus expression in cells from G2V mESC differentiation cultures. We have shown that a significant 3.5-fold increase in Venus MFI between day 10 and day 14 of differentiation (Figure 1I) corresponds to the appearance of mast cells, thus supporting the view that the direct induction of high GATA2 expression is a prerequisite for the rapid commitment to the mast cell lineage (Sasaki et al., 2016) and for the activation of mature mast cells (Masuda et al., 2007).
G2V Mast Cell Expansion Features
In addition to producing mast cells, we found that the staged G2V mESC culture system produces self-renewing mast cell progenitors. Replating experiments revealed large increases in mast cell output over time. While it is known that interleukin-3 (IL-3) and stem cell factor (SCF) lead to increased mast cell output and function (Ito et al., 2012, Lantz et al., 1998, Yamaguchi et al., 2013), our G2V mESC produce these cells in 2–3 weeks, rather than the 4–12 weeks in other culture systems. Most similar to our method, Yamaguchi et al. used co-cultures with OP9 cells to achieve the production of 6.5 × 106 mast cells from 1 × 105 mouse iPSCs in 4 weeks (Yamaguchi et al., 2013). From a starting population of only 3 × 104 G2V mESCs, we have achieved up to 3.8 × 106 mast cells within 3 weeks (or 12.65 × 106 mast cells from 1 × 105 G2V mESC), thus demonstrating not only a more rapid production but a more abundant production of mast cells. Following stage 2, on average every 1:2 to 1:6 Gata2+ cell was an MCp and gave rise to a homogeneous colony containing more than 500 mature mast cells. While the Gata2 reporter-mediated enrichment focuses the co-culture system on hematopoietic progenitors and MCp, we are currently attempting to further shorten the culture period and mast cell output of mESC by other modifications to the culture conditions.
GATA2 Reporter Can Be Exploited for Rapid Human Mast Cell Generation
Our staged ESC culture takes advantage the Gata2 reporter together with OP9 stromal cell co-culture for high-efficiency mouse mast cell induction. Approximately 1%–2% of cells after stage 1 culture are GATA2 expressing and the OP9 co-culture step results in close to 100% differentiation of GATA2-expressing mESC-derived cells to mast cells/mast cell progenitors. We found this method to be applicable to G2V human ESCs/iPSCs. Three independent lines of G2V PSCs were cultured according to the multistep protocol, except under serum-free conditions and with different cytokines in step 1. In contrast to mouse cultures, high percentages (40%–70%) of GATA2-expressing cells were observed after stage 1 (25- to 44-fold more). Following stage 2 co-culture on OP9 cells, almost all mast cell/mast cell progenitors were found in the GATA2+ fraction, as was found for the mouse stage 2 cultures. However, the frequency of human CKIT+FCɛR1α+ cells was many times lower. Nonetheless, human mast cells can be obtained within 12–16 days of differentiation and Gata2 reporter would enable the production of human mast cells that could be enriched to high homogeneity, in contrast to mast cells derived from wild-type non-reporter PSCs.
Our study highlights possible species-specific differences between mPSCs and hPSCs and/or culture requirements for quantitatively efficient mast cell production. For example, mESCs are thought to be naive whereas hESCs/hiPSCs are primed and somewhat differentiated (Weinberger et al., 2016). It will be interesting to test whether the efficiency of hESC/hiPSC hematopoietic differentiation increases in serum-containing cultures. Moreover, OP9 cells are neonatal mouse bone marrow stromal cells that efficiently support mouse hematopoietic cell development and differentiation (Lynch et al., 2011). It would be beneficial to find an equivalent human cell line for the co-culture step in the hESC/hiPSC method And, by extension, examination of which specific molecules produced by OP9 cells are relevant to mast cell differentiation of mESCs would allow for identification and testing of the relevant human homologs. These and other approaches for increasing the efficiency of human mast cell production are under investigation and will open up opportunities for the controlled production of mast cells from patients suffering from highly heterogeneous mast cell disorders such as mastocytosis (Jawhar et al., 2017). CRISPR/Cas9 approaches facilitate the introduction of the Venus reporter gene into the human GATA2 locus, as shown by our ability to generate several G2V hESC and hiPSC lines that correctly report HPCs and mast cells. Isogenic G2V hiPSC lines for mast cell disorders can now be rapidly created by CRISPR/Cas9 engineering of gene mutations (CKIT D816V and/or others) involved in mast cell diseases, such as mastocytosis. The combination of these approaches for staged mast cell differentiation promises to be useful for drug discovery and testing in a disease-specific and patient-specific manner, thus allowing for new treatment strategies and better quality of life for those patients suffering from mast cell-related conditions.
Experimental Procedures
Mouse and Human G2V PSCs
G2V mESCs were derived from the IB10 ESC line that is a subclone of the E14 ESC line (from the 129/Ola mouse strain) and maintained along with IB10 wild-type control ESC as in Kaimakis et al. (2016) and Kauts et al., 2016, Kauts et al., 2018. H1 hESCs (WA01, WiCell) and hiPSCs (Yang et al., 2017) (SFCi55, gift of L. Forrester and CENSO Biotechnologies) were maintained on mouse embryonic fibroblasts, 0.1% gelatin-coated plates, in DMEM/F12+ knockout serum replacement (Thermo Fisher), GlutaMAX (Gibco), minimum essential medium non-essential amino acids (Lonza), 50 μM β-mercaptoethanol (Gibco), and 10 μg/mL basic fibroblast growth factor (bFGF) (PeproTech). Medium was replaced daily.
Mast Cell Generation Culture
The three-stage culture procedure is summarized in Figure 1A.
Stage 1. ESC Differentiation (Days 0–10)
G2V mESCs were trypsinized and feeder depleted by incubation in Iscove’s modified Dulbecco’s medium (IMDM), 20% fetal bovine serum (FBS) (HyClone), and 1% penicillin/streptomycin (P/S) for 30 min at 37°C. For EB formation, G2V mESCs were aggregated by culturing at 25,000 cells/mL in IMDM, 15% FBS, 2 mM GlutaMAX (Gibco), 50 μg/mL ascorbic acid (Sigma), 4 × 10−4 M monothioglycerol (Sigma), and 300 μg/mL transferrin (Roche) in bacterial dishes. After 72 hr the medium was refreshed and supplemented with 5% proteome free hybridoma medium (Gibco). At day 6, medium was supplemented with murine SCF (100 ng/mL), IL-3 (1 ng/mL), and IL-11 (5 ng/mL) (PeproTech) and refreshed every other day. hPSC differentiation was as described by Kennedy et al. (2012). In brief, hPSCs were cut using StemPro EZPassage Tool (Thermo Fisher) and aggregates resuspended in StemPro-34 (Invitrogen), 10 ng/mL P/S, 2 mM L-glutamine (Gibco), 1 mM ascorbic acid (Sigma-Aldrich), 4 × 10−4 M monothioglycerol (Sigma-Aldrich), and 150 mg/mL transferrin (Roche). Human bone morphogenetic protein 4 (10 ng/mL), bFGF (5 ng/mL), activin A, 6 mM SB-431542 (selective inhibitor of the TGF-β type I receptor activin receptor-like kinase receptors), vascular endothelial growth factor (15 ng/mL), Dickkopf-related protein 1 (DKK, 150 ng/mL), IL-6 (10 ng/mL), insulin-like growth factor 1 (25 ng/mL), IL-11 (5 ng/mL), SCF (50 ng/mL), erythropoietin (EPO, 2 U/mL), thrombopoietin (TPO, 30 ng/mL), IL-3 (30 ng/mL), and Fms-related tyrosine kinase 3 ligand (FLT-3L, 10 ng/mL) (PeproTech) were added.
Stage 2. OP9 Co-culture for Hematopoietic/Mast Cell Commitment (Days 10–14)
At day 10, EBs were washed in PBS, incubated with TrypLE Express (Gibco) (37°C, 3–5 min), and deactivated with PBS, 10% fetal calf serum, an 1% P/S. Cells were suspended with a P1000 pipette. Ten thousand V+ cells were sorted and plated on 30,000 OP9 cells pre-seeded1 day earlier in 24-well plates in α-minimal essential medium, 10% heat-inactivated FBS, 1% P/S, 20 ng/mL FLT-3L, 20 ng/mL IL-7 and/or 20 ng/mL IL-6, and 50 ng/mL SCF (PeproTech). For hPSC culture, the same concentration for human cytokines was used (PeproTech). V+ mast cells appeared after 2–4 days and sorted cells used for experiments and further expansion.
Stage 3. Expansion
After 4 days of OP9 co-culture, V+ cells were sorted and plated (500 cells/mL) in methylcellulose (STEMCELL Technologies). After 3–4 days, large dense colonies appeared. Colonies were counted after 3–7 days of culture, then cells were harvested by dissolving methylcellulose in PBS and counted by trypan blue exclusion. For replating, colonies were picked and single cells plated into fresh methylcellulose.
Primary Hematopoietic Cell Cultures
Adult mice were sacrificed by cervical dislocation. Peritoneal cells were obtained by lavage (PBS, 10% FBS, 1% P/S) as previously described (Meurer et al., 2016) and bone marrow cells by PBS-flushing of the long bones. Cell suspensions were centrifuged for 5 min at 1000 rpm at room temperature, resuspended in RPMI 1640 medium, 2 mM L-glutamine (Gibco), 15% heat-inactivated FBS (Gibco), 1% P/S (Gibco), 0.07% β-mercaptoethanol (Gibco), and 10 mM HEPES (Sigma-Aldrich), and cultured (medium changes every 3 days). For mast cell growth (2–3 weeks), 30 ng/mL IL-3 and 20 ng/mL SCF (Peprotech) were added and for basophil growth (10 days), 10 ng/mL IL-3 was added. All mouse experimentation was performed under the UK Animals Scientific Procedures Act 1986 Project License 70/8076 and performed in compliance with Standards for Care and Use of Laboratory Animals.
Flow Cytometry
Venus expression was detected directly. Surface receptor expression was analyzed using antibodies CD45-AF700 (1:400; BioLegend, cat. #103127), CD19-PE (1:200; eBiosciences, cat. #12–0193), GR1-APC-Cy7 (1:400; BD Biosciences, cat. # 557661), CD11B-PerCP-Cy5.5 (1:500; eBioscience, cat. #45-0112-80), TER119-BV421 (1:400; BD Horizon, cat. #563998), CKIT-APCeFluor780 (1:800; eBioscience, cat. #47-1171-80), FCɛR1α-PE (1:200, MAR-1; BioLegend, cat. #134308), CKIT-BV421 (1:200; BD Biosciences, cat. #562609), and CD49B-APC (1:100; BioLegend, cat. #108909) for murine cells and CD41-PE (1:50; BioLegend, cat. #303705), FCɛRIα-PE (1:50; BioLegend, cat. #334610), and CKIT-PECy7 (1:50; BioLegend, cat. #313212) for human cells. Dead cells were excluded with Hoechst 33342 (Life Technologies). Cells were sorted/analyzed on FACSAria IISORP (BD Biosciences) with FlowJo software (Tree Star).
Cytospin Staining
Fifteen thousand mouse mast cells (stage 3) were resuspended in 50 μL of PBS, transferred to sample chambers/glass slides, and centrifuged (5 min, 200 rpm), methanol fixed (30 s), and toluidine blue stained (Scientific Laboratory Supplies) (0.5 N HCl, 1 hr). Human cell cytospins were fixed in Carnoy's fixative (60% absolute alcohol [VWR Prolabo], 30% chloroform, 10% acetic acid [Sigma-Aldrich], 1 hr), incubated in 70% ethanol, and stained in 0.1% toluidine blue (0.5 N HCl, 45 min), then dried, dehydrated (95% alcohol), and mounted with Permafluor (Thermo Fisher Scientific). Some human sample slides were stained with a Rapid Romanowsky Stain Pack kit (TCS Biosciences). An Axioskop2 (Zeiss) microscope with 40× objective was used.
Mast Cell Activation/Degranulation Analyses
Mast cells (stage 3) (1 × 106) were resuspended in 1 mL of PBS and stimulated with c48/80 (Sigma) (5 μg/mL, 60 min, 5% CO2, 37°C). After centrifugation, tryptase concentration in the supernatant was quantified using a Mouse Mast Cell Tryptase (MCT) ELISA Kit (Cusabio). A standard curve was generated using a four-parameter logistic (4-PL) curve fit. Mast cell degranulation (β-hexosaminidase release) was determined according to the protocol of Kuehn et al. (2010). In brief, G2V-mESCS-MCs and control PMCs were sensitized overnight with 1 μg/mL mouse anti-dinitrophenyl (DNP) IgE (Sigma-Aldrich). Following three washes with HEPES buffer (pH 7.4), 50,000 cells/well were activated with DNP-HSA (Sigma-Aldrich) and incubated in 37°C for 60 min. Supernatants were collected and cells lysed using 0.1% Triton X-100 in HEPES buffer. β-Hexosaminidase activity in supernatants and cell lysates was measured using a colorimetric assay with 3.5 mg/mL p-nitrophenyl-N-acetyl-β-D-glucosamide (PNAG; Sigma-Aldrich) as a substrate. After 90-min incubation at 37°C, the reaction was stopped with glycine buffer (400 mM, pH 10.7). The absorbance was measured at 405 nm (Varioskan Flash microplate reader, Thermo Scientific). The percentage of β-hexosaminidase release was calculated according to the formula: %β-hexosaminidase released = 100 × (supernatant content)/(supernatant + lysate content).
Expression Analysis
Total RNA was extracted using an RNA mini or micro kit (Qiagen) and standardized to 100 ng/mL. cDNA was synthesized using Superscript VILO synthesis kit (Invitrogen) at 25°C for 10 min, 42°C for 60 min (primer extension), and 85°C for 5 min (inactivation) in a Thermo Cycler (MJ Research PTC 200 BC-MJPC200). A cDNA standard curve for qPCR primer efficiency validation was made using an RNA dilution series (murine; HMC-1). Together with standard curve R2 value, efficiency (E, %) of all the primer sets was calculated by formula E = (10(−1/slope) − 1) × 100 in 10- and 2-fold dilutions. ΔΔCt analysis was performed for primer sets with an efficiency range of 95%–105% (all primers in this study). TaqMan PCR was used for real-time RNA quantification. Primer sets were designed using the “Universal ProbeLibrary Assay Design Center.” Semi-quantitative RT-PCR was performed using BioMixRed (Bioline). For human Venus-sorted samples, total RNA was isolated and reverse transcribed using oligo-dT (Life Technologies) and SuperScript III (Life Technologies). qRT-PCR used Fast SYBR Green Master Mix (Life Technologies). Primers are listed in Table S1.
Statistics
For two-group analysis, an unpaired two-tailed t test was used. One-way ANOVA was used for multiple-group comparison, followed by Kruskal-Wallis as a secondary test and Dunn's multiple comparisons test. Results are statistically significant at p < 0.05. Data are shown as mean ± SEM, n = number of independent experiments. Data analysis was done using GraphPad Prism (GraphPad Software).
Author Contributions
M.-L.K., and B.D.L. performed the research. P.K., C.R.S., F.G., and A.C.W. generated transgenic cell lines. B.G. and A.C.W. designed the human GATA2 targeting construct. L.F., B.G., and P.S. provided reagents. P.S., H.T., and L.F. provided advice. A.M. and M.B. performed functional and qRT-PCR assays. C.R.S. performed primary hematopoietic cultures and FACS. R.R. performed some cultures and transcription analysis. M.-L.K., B.D.L., and E.D. designed experiments and analyzed and interpreted the data. M.-L.K. and E.D. wrote the manuscript.
Acknowledgments
We thank all the lab members for helpful discussions and support with the experiments, and the QMRI Flow facility for FACS sorting. The authors acknowledge the grant support of the Landsteiner Society for Blood Research (LSBR 1109), ZonMW-Netherlands Scientific Research Council TOP (91211068), the European Research Council advanced grant (341096), and NIHR grant (RP-PG-0310-1002). B.G. and A.C.W. were supported by Bloodwise, NIHR, and core support funding from the Wellcome Trust to the Cambridge Stem Cell Institute.
Published: September 6, 2018
Footnotes
Supplemental Information includes Supplemental Experimental Procedures, three figures, and one table and can be found with this article online at https://doi.org/10.1016/j.stemcr.2018.08.007.
Supplemental Information
References
- Akashi K., Traver D., Miyamoto T., Weissman I.L. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature. 2000;404:193–197. doi: 10.1038/35004599. [DOI] [PubMed] [Google Scholar]
- Bischoff S.C. Role of mast cells in allergic and non-allergic immune responses: comparison of human and murine data. Nat. Rev. Immunol. 2007;7:93–104. doi: 10.1038/nri2018. [DOI] [PubMed] [Google Scholar]
- Dahlin J.S., Malinovschi A., Ohrvik H., Sandelin M., Janson C., Alving K., Hallgren J. Lin- CD34hi CD117int/hi FcepsilonRI+ cells in human blood constitute a rare population of mast cell progenitors. Blood. 2016;127:383–391. doi: 10.1182/blood-2015-06-650648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Leo B., Esnal-Zufiaurre A., Collins F., Critchley H.O.D., Saunders P.T.K. Immunoprofiling of human uterine mast cells identifies three phenotypes and expression of ERbeta and glucocorticoid receptor. F1000Res. 2017;6:667. doi: 10.12688/f1000research.11432.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eich C., Arlt J., Vink C.S., Solaimani Kartalaei P., Kaimakis P., Mariani S.A., van der Linden R., van Cappellen W.A., Dzierzak E. In vivo single cell analysis reveals Gata2 dynamics in cells transitioning to hematopoietic fate. J. Exp. Med. 2018;215:233–248. doi: 10.1084/jem.20170807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galli S.J., Borregaard N., Wynn T.A. Phenotypic and functional plasticity of cells of innate immunity: macrophages, mast cells and neutrophils. Nat. Immunol. 2011;12:1035–1044. doi: 10.1038/ni.2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo G., Luc S., Marco E., Lin T.W., Peng C., Kerenyi M.A., Beyaz S., Kim W., Xu J., Das P.P. Mapping cellular hierarchy by single-cell analysis of the cell surface repertoire. Cell Stem Cell. 2013;13:492–505. doi: 10.1016/j.stem.2013.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holm M., Kvistgaard H., Dahl C., Andersen H.B., Hansen T.K., Schiotz P.O., Junker S. Modulation of chemokine gene expression in CD133+ cord blood-derived human mast cells by cyclosporin A and dexamethasone. Scand. J. Immunol. 2006;64:571–579. doi: 10.1111/j.1365-3083.2006.01835.x. [DOI] [PubMed] [Google Scholar]
- Huang K., Du J., Ma N., Liu J., Wu P., Dong X., Meng M., Wang W., Chen X., Shi X. GATA2(-/-) human ESCs undergo attenuated endothelial to hematopoietic transition and thereafter granulocyte commitment. Cell Regen. (Lond.) 2015;4:4. doi: 10.1186/s13619-015-0018-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Igarashi A., Ebihara Y., Kumagai T., Hirai H., Nagata K., Tsuji K. Mast cells derived from human induced pluripotent stem cells are useful for allergen tests. Allergol. Int. 2018;67:234–242. doi: 10.1016/j.alit.2017.08.008. [DOI] [PubMed] [Google Scholar]
- Ito T., Smrz D., Jung M.Y., Bandara G., Desai A., Smrzova S., Kuehn H.S., Beaven M.A., Metcalfe D.D., Gilfillan A.M. Stem cell factor programs the mast cell activation phenotype. J. Immunol. 2012;188:5428–5437. doi: 10.4049/jimmunol.1103366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jawhar M., Schwaab J., Meggendorfer M., Naumann N., Horny H.P., Sotlar K., Haferlach T., Schmitt K., Fabarius A., Valent P. The clinical and molecular diversity of mast cell leukemia with or without associated hematologic neoplasm. Haematologica. 2017;102:1035–1043. doi: 10.3324/haematol.2017.163964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jippo T., Mizuno H., Xu Z., Nomura S., Yamamoto M., Kitamura Y. Abundant expression of transcription factor GATA-2 in proliferating but not in differentiated mast cells in tissues of mice: demonstration by in situ hybridization. Blood. 1996;87:993–998. [PubMed] [Google Scholar]
- Kaimakis P., de Pater E., Eich C., Solaimani Kartalaei P., Kauts M.L., Vink C.S., van der Linden R., Jaegle M., Yokomizo T., Meijer D. Functional and molecular characterization of mouse Gata2-independent hematopoietic progenitors. Blood. 2016;127:1426–1437. doi: 10.1182/blood-2015-10-673749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kauts M.L., Rodriguez-Seoane C., Kaimakis P., Mendes S.C., Cortes-Lavaud X., Hill U., Dzierzak E. In vitro differentiation of Gata2 and Ly6a reporter embryonic stem cells corresponds to in vivo waves of hematopoietic cell generation. Stem Cell Reports. 2018;10:151–165. doi: 10.1016/j.stemcr.2017.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kauts M.L., Vink C.S., Dzierzak E. Hematopoietic (stem) cell development - how divergent are the roads taken? FEBS Lett. 2016;590:3975–3986. doi: 10.1002/1873-3468.12372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy M., Awong G., Sturgeon C.M., Ditadi A., LaMotte-Mohs R., Zuniga-Pflucker J.C., Keller G. T lymphocyte potential marks the emergence of definitive hematopoietic progenitors in human pluripotent stem cell differentiation cultures. Cell Rep. 2012;2:1722–1735. doi: 10.1016/j.celrep.2012.11.003. [DOI] [PubMed] [Google Scholar]
- Kirshenbaum A.S., Metcalfe D.D. Growth of human mast cells from bone marrow and peripheral blood-derived CD34+ pluripotent progenitor cells. Methods Mol. Biol. 2006;315:105–112. doi: 10.1385/1-59259-967-2:105. [DOI] [PubMed] [Google Scholar]
- Kovarova M., Latour A.M., Chason K.D., Tilley S.L., Koller B.H. Human embryonic stem cells: a source of mast cells for the study of allergic and inflammatory diseases. Blood. 2010;115:3695–3703. doi: 10.1182/blood-2009-08-237206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuehn H.S., Radinger M., Gilfillan A.M. Measuring mast cell mediator release. Curr. Protoc. Immunol. 2010 doi: 10.1002/0471142735.im0738s91. Unit7.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lagunoff D., Rickard A. Evidence for control of mast cell granule protease in situ by low pH. Exp. Cell Res. 1983;144:353–360. doi: 10.1016/0014-4827(83)90414-7. [DOI] [PubMed] [Google Scholar]
- Lantz C.S., Boesiger J., Song C.H., Mach N., Kobayashi T., Mulligan R.C., Nawa Y., Dranoff G., Galli S.J. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature. 1998;392:90–93. doi: 10.1038/32190. [DOI] [PubMed] [Google Scholar]
- Lynch M.R., Gasson J.C., Paz H. Modified ES/OP9 co-culture protocol provides enhanced characterization of hematopoietic progeny. J. Vis. Exp. 2011 doi: 10.3791/2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masuda A., Hashimoto K., Yokoi T., Doi T., Kodama T., Kume H., Ohno K., Matsuguchi T. Essential role of GATA transcriptional factors in the activation of mast cells. J. Immunol. 2007;178:360–368. doi: 10.4049/jimmunol.178.1.360. [DOI] [PubMed] [Google Scholar]
- McKinney-Freeman S.L., Naveiras O., Yates F., Loewer S., Philitas M., Curran M., Park P.J., Daley G.Q. Surface antigen phenotypes of hematopoietic stem cells from embryos and murine embryonic stem cells. Blood. 2009;114:268–278. doi: 10.1182/blood-2008-12-193888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meurer S.K., Ness M., Weiskirchen S., Kim P., Tag C.G., Kauffmann M., Huber M., Weiskirchen R. Isolation of mature (peritoneum-derived) mast cells and immature (bone marrow-derived) mast cell precursors from mice. PLoS One. 2016;11:e0158104. doi: 10.1371/journal.pone.0158104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minegishi N., Suzuki N., Yokomizo T., Pan X., Fujimoto T., Takahashi S., Hara T., Miyajima A., Nishikawa S., Yamamoto M. Expression and domain-specific function of GATA-2 during differentiation of the hematopoietic precursor cells in midgestation mouse embryos. Blood. 2003;102:896–905. doi: 10.1182/blood-2002-12-3809. [DOI] [PubMed] [Google Scholar]
- Miyamoto T., Iwasaki H., Reizis B., Ye M., Graf T., Weissman I.L., Akashi K. Myeloid or lymphoid promiscuity as a critical step in hematopoietic lineage commitment. Dev. Cell. 2002;3:137–147. doi: 10.1016/s1534-5807(02)00201-0. [DOI] [PubMed] [Google Scholar]
- Moller C., Karlberg M., Abrink M., Nakayama K.I., Motoyama N., Nilsson G. Bcl-2 and Bcl-XL are indispensable for the late phase of mast cell development from mouse embryonic stem cells. Exp. Hematol. 2007;35:385–393. doi: 10.1016/j.exphem.2006.11.008. [DOI] [PubMed] [Google Scholar]
- North T.E., de Bruijn M.F., Stacy T., Talebian L., Lind E., Robin C., Binder M., Dzierzak E., Speck N.A. Runx1 expression marks long-term repopulating hematopoietic stem cells in the midgestation mouse embryo. Immunity. 2002;16:661–672. doi: 10.1016/s1074-7613(02)00296-0. [DOI] [PubMed] [Google Scholar]
- Orlic D., Anderson S., Biesecker L.G., Sorrentino B.P., Bodine D.M. Pluripotent hematopoietic stem cells contain high levels of mRNA for c-kit, GATA-2, p45 NF-E2, and c-myb and low levels or no mRNA for c-fms and the receptors for granulocyte colony-stimulating factor and interleukins 5 and 7. Proc. Natl. Acad. Sci. USA. 1995;92:4601–4605. doi: 10.1073/pnas.92.10.4601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudich N., Ravid K., Sagi-Eisenberg R. Mast cell adenosine receptors function: a focus on the a3 adenosine receptor and inflammation. Front. Immunol. 2012;3:134. doi: 10.3389/fimmu.2012.00134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito H., Kato A., Matsumoto K., Okayama Y. Culture of human mast cells from peripheral blood progenitors. Nat. Protoc. 2006;1:2178–2183. doi: 10.1038/nprot.2006.344. [DOI] [PubMed] [Google Scholar]
- Sanchez M.J., Holmes A., Miles C., Dzierzak E. Characterization of the first definitive hematopoietic stem cells in the AGM and liver of the mouse embryo. Immunity. 1996;5:513–525. doi: 10.1016/s1074-7613(00)80267-8. [DOI] [PubMed] [Google Scholar]
- Sasaki H., Kurotaki D., Tamura T. Regulation of basophil and mast cell development by transcription factors. Allergol. Int. 2016;65:127–134. doi: 10.1016/j.alit.2016.01.006. [DOI] [PubMed] [Google Scholar]
- Schwartz L.B., Austen K.F. Enzymes of the mast cell granule. J. Invest. Dermatol. 1980;74:349–353. doi: 10.1111/1523-1747.ep12543620. [DOI] [PubMed] [Google Scholar]
- Shimizu Y., Sakai K., Miura T., Narita T., Tsukagoshi H., Satoh Y., Ishikawa S., Morishita Y., Takai S., Miyazaki M. Characterization of 'adult-type' mast cells derived from human bone marrow CD34(+) cells cultured in the presence of stem cell factor and interleukin-6. Interleukin-4 is not required for constitutive expression of CD54, Fc epsilon RI alpha and chymase, and CD13 expression is reduced during differentiation. Clin. Exp. Allergy. 2002;32:872–880. doi: 10.1046/j.1365-2222.2002.01373.x. [DOI] [PubMed] [Google Scholar]
- Theoharides T.C., Valent P., Akin C. Mast cells, mastocytosis, and related disorders. N. Engl. J. Med. 2015;373:1885–1886. doi: 10.1056/NEJMc1510021. [DOI] [PubMed] [Google Scholar]
- Tsai F.Y., Keller G., Kuo F.C., Weiss M., Chen J., Rosenblatt M., Alt F.W., Orkin S.H. An early haematopoietic defect in mice lacking the transcription factor GATA-2. Nature. 1994;371:221–226. doi: 10.1038/371221a0. [DOI] [PubMed] [Google Scholar]
- Tsai F.Y., Orkin S.H. Transcription factor GATA-2 is required for proliferation/survival of early hematopoietic cells and mast cell formation, but not for erythroid and myeloid terminal differentiation. Blood. 1997;89:3636–3643. [PubMed] [Google Scholar]
- Tsai M., Tam S.Y., Wedemeyer J., Galli S.J. Mast cells derived from embryonic stem cells: a model system for studying the effects of genetic manipulations on mast cell development, phenotype, and function in vitro and in vivo. Int. J. Hematol. 2002;75:345–349. doi: 10.1007/BF02982122. [DOI] [PubMed] [Google Scholar]
- Wang X.S., Sam S.W., Yip K.H., Lau H.Y. Functional characterization of human mast cells cultured from adult peripheral blood. Int. Immunopharmacol. 2006;6:839–847. doi: 10.1016/j.intimp.2005.12.004. [DOI] [PubMed] [Google Scholar]
- Weinberger L., Ayyash M., Novershtern N., Hanna J.H. Dynamic stem cell states: naive to primed pluripotency in rodents and humans. Nat. Rev. Mol. Cell Biol. 2016;17:155–169. doi: 10.1038/nrm.2015.28. [DOI] [PubMed] [Google Scholar]
- Weller C.L., Collington S.J., Hartnell A., Conroy D.M., Kaise T., Barker J.E., Wilson M.S., Taylor G.W., Jose P.J., Williams T.J. Chemotactic action of prostaglandin E2 on mouse mast cells acting via the PGE2 receptor 3. Proc. Natl. Acad. Sci. USA. 2007;104:11712–11717. doi: 10.1073/pnas.0701700104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerberg C.M., Ulleras E., Nilsson G. Differentiation of mast cell subpopulations from mouse embryonic stem cells. J. Immunol. Methods. 2012;382:160–166. doi: 10.1016/j.jim.2012.05.020. [DOI] [PubMed] [Google Scholar]
- Yamaguchi T., Tashiro K., Tanaka S., Katayama S., Ishida W., Fukuda K., Fukushima A., Araki R., Abe M., Mizuguchi H. Two-step differentiation of mast cells from induced pluripotent stem cells. Stem Cells Dev. 2013;22:726–734. doi: 10.1089/scd.2012.0339. [DOI] [PubMed] [Google Scholar]
- Yang C.T., Ma R., Axton R.A., Jackson M., Taylor A.H., Fidanza A., Marenah L., Frayne J., Mountford J.C., Forrester L.M. Activation of KLF1 enhances the differentiation and maturation of red blood cells from human pluripotent stem cells. Stem Cells. 2017;35:886–897. doi: 10.1002/stem.2562. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.