Abstract
Cardiogenic global brain hypoxia-ischemia is a devastating medical problem that is associated with unfavorable neurologic outcomes. Low dose hydrogen gas (up to 2.9%) has been shown to be neuroprotective in a variety of brain diseases. In the present study, we investigated the protective effect of water by electrolysis-derived high concentration hydrogen gas (60%) in a rat model of asphyxia induced-cardiac arrest and global brain hypoxia-ischemia. High concentration hydrogen gas was either administered starting 1 hour prior to cardiac arrest for 1 hour and starting 1 hour post-resuscitation for 1 hour (pre- & post-treatment) or starting 1 hour post-resuscitation for 2 hours (post-treatment). In animals subjected to 9 minutes of asphyxia, both therapeutic regimens tended to reduce the incidence of seizures and neurological deficits within 3 days post-resuscitation. In rats subjected to 11 minutes of asphyxia, significantly worse neurological deficits were observed compared to 9 minutes asphyxia, and pre- & post-treatment had a tendency to improve the success rate of resuscitation and to reduce the seizure incidence within 3 days post-resuscitation. Findings of this preclinical study suggest that water electrolysis-derived 60% hydrogen gas may improve short-term outcomes in cardiogenic global brain hypoxia-ischemia.
Keywords: high concentration hydrogen gas, water electrolysis, cardiac arrest, global brain ischemia, neurological deficit, seizure, rat
INTRODUCTION
Cardiogenic global brain hypoxia-ischemia is a devastating event that is associated with great morbidity and prolonged intensive medical treatment.1,2 According to the 2013 update of Heart Disease and Stroke Statistics by the American Heart Association (AHA), only 9.5% of adult out-of-hospital cardiac arrest (CA) patients and 23.9% of adult in-hospital CA patients survived to hospital discharge.3 For those patients who survive to hospital discharge, neurologic injury remains a source of significant morbidity. Approximately half of surviving patients have moderate to severe cognitive deficits at three months after cardiac arrest. The persistence of unfavorable neurologic outcomes, despite advances in cardiopulmonary resuscitation (CPR), prompted the AHA to emphasize brain injury related to cardiac arrest by proposing “cardiopulmonary-cerebral resuscitation” in its 2000 Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care.4 Therapeutic strategies targeting brain injury after cardiac arrest are thus an important area for basic science and clinical research.5
Molecular hydrogen (H2) has been proposed to be a selective hydroxyl radical and peroxynitrite scavenger in focal brain ischemia.6 It has potential utility as another novel treatment approach in hypoxia-ischemia brain injury in addition to a variety of therapeutic strategies against stroke reported previously.7,8 Emerging evidence has demonstrated the molecular hydrogen's protective role in a variety of diseases including oxidative stress-related disorders, inflammation and allergy using in vivo and in vitro models.9,10,11 These preclinical studies were limited to a maximum dose of 2.9% hydrogen gas due to safety concerns related to explosion.9,10 The recently developed hydrogen-oxygen nebulizer inhalation device (AMS-H-01) is capable of producing up to 67% hydrogen and 33% oxygen gas mixture at rate of 2–3 L/min without a combustion risk.12 Preclinical studies have consistently demonstrated the treatment efficacy of such high concentration H2 gas against a variety of diseases including focal brain ischemia.13,14,15,16,17
In the present study, we investigated the neuroprotective effect of water electrolysis-derived high concentration hydrogen gas (60%) against cardiogenic global brain hypoxia-ischemia in a rat model of asphyxia-induced CA (ACA).
MATERIALS AND METHODS
All protocols were approved by the Animal Health and Safety Committees of Loma Linda University (approved No. OSR 8170006) and were in compliance with Federal regulations. A total of 56 male Sprague-Dawley rats (retired breeder, body weight 450–550 g) were investigated in the following two sets of experiments. Experiment I: Animals were subjected to 9 minutes of asphyxia: 1) ACA (9 minutes, n = 13); 2) ACA (9 minutes) + H2 post-treatment (n = 13); 3) ACA (9 minutes) + H2 pre- & post-treatment (n = 12). Experiment II: To evaluate the effect of H2 treatment in more severe global brain hypoxia-ischemia, two additional groups of rats were studied using 11 minutes of asphyxia. Based on the findings of Experiment I we did not expect H2 post-treatment alone to provide benefit in the setting of more severe global brain hypoxia-ischemia. We did not therefore include the H2 post-treatment group in Experiment II. Two groups of rats (n = 9/group) underwent 11 minutes of asphyxia to induce more severe global brain hypoxia-ischemia, followed by resuscitation: 1) ACA (11 minutes); 2) ACA (11 minutes) + H2 pre- & post-treatment.
Cardiac arrest was induced by asphyxia.18 Animals were anesthetized with pentobarbital (45 mg/kg) and the trachea was orally intubated with a 14-gauge plastic catheter. After tracheal intubation, polyethylene catheters (Becton Dickinson, Franklin Lakes, NJ, USA) were placed through the left femoral artery and vein into the abdominal aorta and the inferior vena cava, respectively. The arterial catheter was used to monitor arterial pressure and the venous catheter was used for administration of epinephrine or sodium bicarbonate during resuscitation. Lead II-ECG was continuously recorded. The animals were mechanically ventilated with room air at a tidal volume of 0.55 mL/100 g and a frequency of 100 breaths per minute for 15 minutes. Asphyxia was induced by ventilator disconnection and tracheal tube obstruction after chemical neuromuscular blockade (vecuronium 2 mg/kg IV). After 9 (Experiment I) or 11 (Experiment II) minutes of asphyxia, cardiopulmonary resuscitation (CPR) was performed using precordial compression with a pneumatically driven mechanical chest compressor and mechanical ventilation with 100% oxygen at a ratio of 2:1. At the start of CPR, a bolus dose of epinephrine (7.5 μg/kg) with sodium bicarbonate (1 mEq/kg) was injected into the left femoral vein. Successful resuscitation was defined as return of spontaneous circulation (ROSC) with the return of supraventricular rhythm and mean arterial pressure over 60 mmHg for 5 minutes.
After resuscitation, mechanical ventilation was continued with 100% oxygen for 30 minutes and reduced to 50% for another 10 minutes and then gradually reduced 10% every 10 minutes to reach 21% oxygen for the last 10 minutes. Ventilation was stopped at 1 hour post-resuscitation and all catheters including the endotracheal tube were removed. Animals were closely observed by the investigator for an additional 2 hours. Body temperature was monitored by a rectal temperature sensor (Model BAT-12, Physitemp Instrument Inc., Clifton, NJ, USA). A heat lamp was used to maintain body temperature at 36.8 ± 0.2°C from anesthesia induction, during surgery preparation and through cardiac arrest and cardiopulmonary resuscitation as well as during the first 3 hours post-resuscitation.
H2 gas was administered to the treatment groups according to protocol: pre- & post-treatment group – starting at 1 hour prior to cardiac arrest for 1 hour and starting again at 1 hour post-resuscitation for 1 hour; post-treatment group – starting at 1 hour post-resuscitation for 2 hours. Sixty percent H2 gas was delivered through a hydrogen treatment chamber with the inlet connected to a hydrogen nebulizer (AMS-H-01, Asclepius Meditec Co. Ltd., Shanghai, China) and the outlet to a ventilating hood. The H2 gas concentration was monitored by a hydrogen detector during treatment.
Electrocardiogram (EKG), end-tidal carbon dioxide (ETCO2) and arterial pressure were continuously recorded from 15 minutes before asphyxia induction until 1 hour after ROSC on a PC-based data-acquisition system supported by WINDAQ software (DATAQ, Akron, OH, USA). Neurologic deficit scores were obtained using the method19 (neurologic deficit score when normal = 0 and coma = 500). The neurologic deficit score was determined and the presence of seizures during a 1 hour observation period was recorded at 24, 48, and 72 hours after ROSC. Survival was observed up to 7 days. At the end of observation, the rats were sacrificed by deep anesthesia using 5% isoflurane followed by transcardial perfusion with 10% formalin.
Quantitative data were presented as the mean ± SD. SigmaPlot 11.0 (Systat Software, Inc., San Jose, CA, USA) was used for statistical analysis. One-way analysis of variance (ANOVA) was applied for multiple comparisons and t-test was applied for the comparison of two groups followed by Student-Newman-Keuls post hoc test or Mann-Whitney Rank Sum test. A P value less than 0.05 was considered statistically significant.
RESULTS
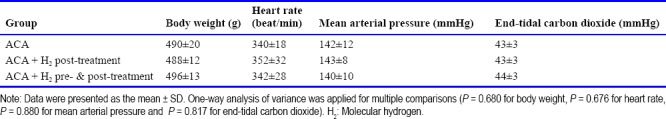
All animals survived the surgical preparation. There were no significant differences in baseline values of body weight, heart rate (HR), mean arterial pressure (MAP) or ETCO2 (Table 1). The average time between the start of asphyxia and MAP decrease to 30 mmHg (80% decrease from the baseline) was 217 ± 28 seconds in ACA control, 225 ± 18 seconds in ACA + H2 post-treatment and 223 ± 19 seconds in ACA + H2 pre- & post-treatment groups, respectively, with no significant difference among the three groups (P = 0.309).
Table 1.
Baseline values of body weight and hemodynamic indicators in a rat model of asphyxia induced-cardiac arrest (ACA)
Experiment I: ACA 9 minutes
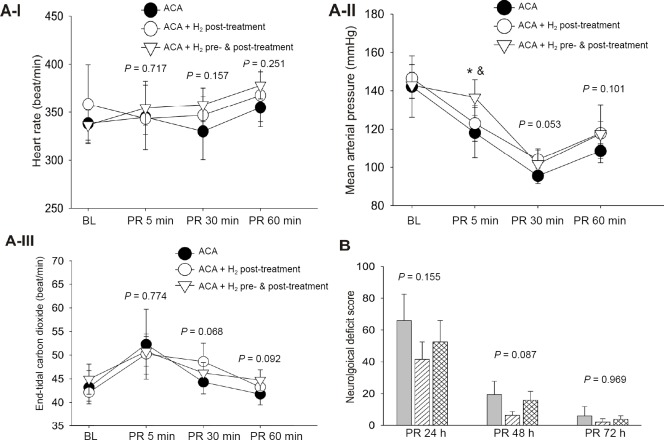
One rat in ACA + H2 post-treatment and one rat in ACA + H2 pre- & post-treatment group were not resuscitated (did not reach ROSC after 5 minutes of CPR). All other animals subjected to 9 minutes of asphyxia achieved ROSC within 1 minute of cardiopulmonary resuscitation (CPR). In the successfully resuscitated rats, we found no between group differences in the time elapsed before ROSC (43 ± 17 seconds in ACA, 36 ± 4 seconds in ACA + H2 post-treatment and 41 ± 7 seconds in ACA + H2 pre- & post-treatment, P = 0.539). After ROSC, the heart rate, MAP and ETCO2 were not significantly different at 5, 30 and 60 minutes post-resuscitation among three groups except for MAP at 5 minutes post-resuscitation (Figure 1A).
Figure 1.
Hemodynamics and neurological outcomes in rats subjected to 9 min of asphyxia.
Note: (A) There were no significant differences in heart rate (A-I), mean arterial pressure (A-II) and endo-tidal carbon dioxide (A-III) among ACA, ACA + H2 post-treatment and ACA + H2 pre- & post-treatment, except mean arterial pressure at 5 min post-resuscitation. *P< 0.05, vs. ACA; &P< 0.05, vs. ACA + H2 post-treatment; (B) There was a tendency toward the improved neurological deficit score in ACA rats received H2 post- and H2 pre- & post-treatment at 24 and 48 h post-resuscitation (PR). Data were presented as the mean ± SD. One-way analysis of variance was applied for multiple comparisons followed by Student-Newman-Keuls post hoc test or Kruskal-Wallis on Rank test. ACA: Asphyxia-induced cardiac arrest; BL: baseline; h: hours; min: minutes; H2: molecular hydrogen.
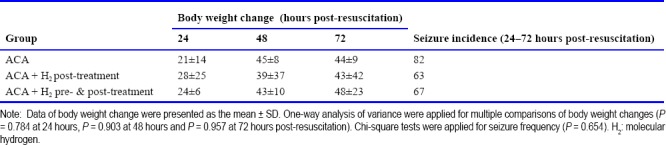
There was no significant change in body weight among the three groups over the 3 days post-resuscitation (Table 2). The incidence of seizures within the 1 hour observation duration at 24 hours post-resuscitation was greater in the ACA group than H2 post-treatment or H2 pre- & post-treatment groups; however the difference was not statistically significant (Table 2). There was a tendency in H2 post-treatment or H2 pre- & post-treatment groups toward improved post-resuscitation neurological deficits at 24, 48 and 72 hours (Figure 1B). All resuscitated rats survived to 7 days except one rat in the ACA group that died at 14 hours post-resuscitation. The 7-day survival was not significantly different among the three groups.
Table 2.
Body weight changes (g) and seizure incidence (%) in rats resuscitated from 9 minutes of asphyxia induced cardiac arrest (ACA)
Experiment II: ACA 11 minutes
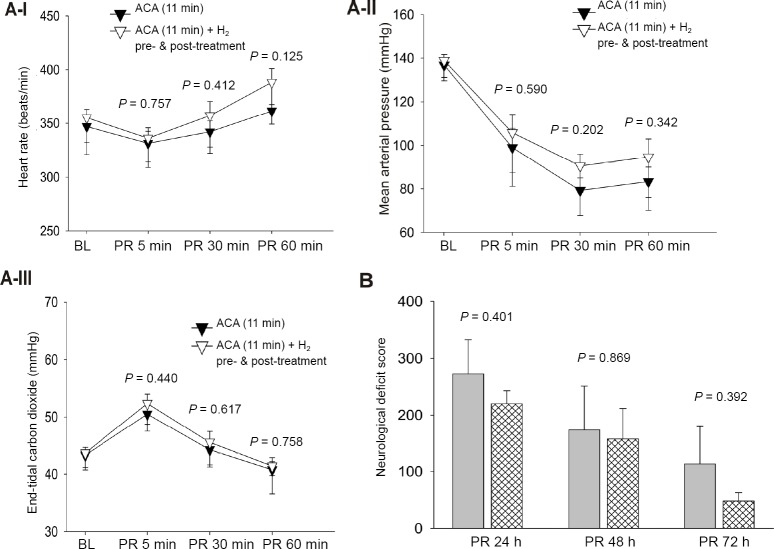
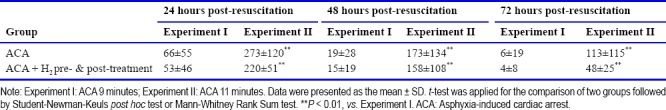
In the animals subjected to 11 minutes of asphyxia, 40% of rats in the ACA group and 62.5% of rats in the ACA + H2 pre- & post-treatment group were successfully resuscitated. However, the difference between groups in successful resuscitation did not reach statistical significance. All the resuscitated animals reached ROSC within 3 minutes of CPR (141 ± 139 seconds in the ACA group and 137 ± 115 seconds in the ACA + H2 pre- & post-treatment group). After ROSC, the heart rate, MAP and ETCO2 were not significantly different at 5, 30 and 60 minutes post-resuscitation (Figure 2A). There was no difference in body weight change between the two groups over 3 days post-resuscitation (Table 3). Compared to animals subjected to 9 minutes of asphyxia, more severe neurological scores were observed in all surviving animals over the three days after resuscitation (Table 4). H2 pre- & post-treatment tended to improve the neurological deficits within the 3 days post-resuscitation (Figure 2B). No seizures were observed at 24 hours post-resuscitation, but the rats had severe neurological deficit and worse overall condition. During the 1 hour observation time at 48 hours post-resuscitation, seizures were observed in 50% of ACA and 20% of ACA + H2 pre- & post-treatment rats (Table 3); however, this difference did not reach statistical significance. Over the 7-day post-resuscitation period, 1 rat died at 24 hours post-resuscitation in the ACA group and 1 rat died at 48 hours post-resuscitation in the H2 pre- & post-treatment group. The overall mortality over 7 days including the non resuscitated rats in H2 pre- & post-treated group (56%) was less than in the ACA group (67%), but this difference was not statistically significant.
Figure 2.
Hemodynamics and neurological outcomes in rats subjected to 11 minutes of asphyxia.
Note: (A) There were no significant differences in heart rate (A-I), mean arterial pressure (A-II) and endo-tidal carbon dioxide (A-III) among ACA and ACA + H2 pre- & post-treatment; (B) There was a tendcy toward the improved neurological difcit score in ACA rats recevied H2 pre- & post-treatment at 24, 48 and 72 h post-resuscitation (PR). Data were presented as the mean ± SD, and t-test was applied for the comparison of two groups followed by Student-Newman-Keuls post hoc test or Mann-Whitney Rank Sum test. ACA: Asphyxia-induced cardiac arrest; BL: baseline; H2: molecular hydrogen; h: hours; min: minutes.
Table 3.
Body weight changes (g) and seizure incidence (%) in rats resuscitated from 11 minutes of asphyxia induced cardiac arrest (ACA)
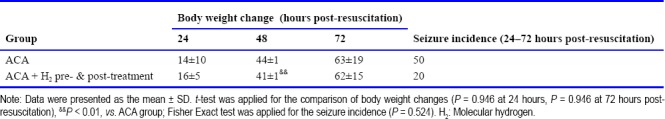
Table 4.
Neurological deficits in resuscitated rats within the 3 days post-resuscitation
DISCUSSION
Using a rat model of asphyxia induced-cardiac arrest, we first investigated the effect of high concentration (60%) H2 gas on global brain hypoxic-ischemia injury. The new findings were: 1) Both high concentration H2 post- and pre- & post-treatment approaches had a similar tendency to reduce the incidence of seizures and improve neurological deficit scores within 3 days following resuscitation; 2) H2 pretreatment tended to increase the tolerance of rats to the subsequent more severe (11 minutes) global hypoxic-ischemia insult, which led to a higher percentage of resuscitation and 7 days survival compared to that of non-treated rats; 3) H2 pretreatment strategy did not significantly affect hemodynamic measurements during the first hour post-resuscitation in rats subjected to either mild or severe global hypoxic-ischemia.
Oxidative stress is a common final mechanism of injury contributing to brain damage following CA and ROSC. Excessive production of free oxygen radicals associated with ischemia and reperfusion injury causes cellular lipid and protein degradation. Treatment with mechanisms that reduce free radicals in the brain may be neuroprotective.20,21 The therapeutic application of H2 as a selective antioxidant was intensively elucidated by Ohsawa et al.6 in a rat model of focal brain ischemia. Use of H2 gas as an antioxidant has the advantages of high biomembrane penetration and selective scavenging of the deleterious hydroxyl radical, potentially making H2 gas superior to traditional antioxidant supplements with strong reductive activity such as vitamin C or vitamin E. Two-hour administration of either 2% H2 in 98% O2 or 1.3% H2 in 25% O2/air at the start of CPR improved neurological outcome in a rat model of ventricular fibrillation cardiac arrest.22,23 Multiple doses of hydrogen-rich saline administered starting at 1 minute before CPR also improved survival and neurological outcomes after CA and CPR in rats.18 In the current study, we used water electrolysis-derived high concentration H2 gas produced by a hydrogen nebulizer. This portable machine that can produce up to 67% H2 at a clinically relevant flow without a combustion risk is ready for patient use and may provide a feasible and safe method for directly administrating H2 gas to patients in clinical practice.
Because most CA patients may not get advanced life support immediately, we applied the 2-hour 60% H2 treatment starting 1 hour after resuscitation. This approach tended to reduce the incidence of seizures and improve neurological deficit scores in the resuscitated rats. However, the time of H2 administration seems to play an important role in that early administration has been shown to be associated with more significant benefits in previous studies.18,22,23 In some patients, global brain ischemia may be induced by certain cardiac surgical procedures, in which pretreatment strategies may be clinically applicable. Exposure of rats to 60% H2 1 hour prior to asphyxia in our study improved tolerance to the subsequent more severe global hypoxic ischemia insult (11 minutes asphyxia), leading to better success rates of return of spontaneous circulation compared to control. This pretreatment benefit appeared less evident if the hypoxic-ischemic injury severity was less (9 minutes asphyxia). In rats that were successfully resuscitated following 11 minutes ACA, the neuroprotective effects of H2 pre- & post-treatment were comparable to that observed in the animals subjected to 9 minutes of asphyxia.
There were some limitations to our study. Only two doses of 60% H2 gas treatment (pre- & post-treatment vs. post-treatment) were applied. Thus, we did not evaluate the best dose effect. Due to the high mortality in the severe model of asphyxia cardiac arrest (Experiment II), the sample size for evaluating post-resuscitation neurological outcomes was relatively small. Future study using larger numbers of animals is warranted to clarify the neuroprotective effect of 60% H2 in the setting of severe global brain ischemia.
In conclusion, water electrolysis derived-high concentration H2 gas provided some degree of short-term neuroprotection in the setting of cardiogenic global brain ischemia. The optimal time for administration and alternative dosage regimens needs to be further investigated to maximize potential neurological benefits.
Additional file
Open peer review report 1 (8.7KB, pdf) .
Acknowledgements
The authors wish to acknowledge the assistance of Suzzanne Marcantonio (Department of Anesthesiology, Loma Linda University) for surgery and neurological evaluation.
Footnotes
Conflicts of interest
None.
Financial support
This study received support from the Floyd S. Brauer Anesthesiology Research Endowment, Loma Linda University.
Institutional review board statement
All protocols were approved by the Animal Health and Safety Committees of Loma Linda University (approved number: OSR 8170006).
Copyright license agreement
The Copyright License Agreement has been signed by all authors before publication.
Data sharing statement
Datasets analyzed during the current study are available from the corresponding author on reasonable request.
Plagiarism check
Checked twice by iThenticate.
Peer review
Externally peer reviewed.
Open peer reviewers
Xue-Jun Sun, Second Military Medical University, China; Sheng Chen, Zhejiang University, China.
Funding: This study received support from the Floyd S. Brauer Anesthesiology Research Endowment, Loma Linda University.
C-Editor: Yang LJ, Zhao M; S-Editor: Yu J; L-Editor: Wang L; T-Editor: Jia Y
REFERENCES
- 1.Madl C, Holzer M. Brain function after resuscitation from cardiac arrest. Curr Opin Crit Care. 2004;10:213–217. doi: 10.1097/01.ccx.0000127542.32890.fa. [DOI] [PubMed] [Google Scholar]
- 2.Moulaert VRM, van Heugten CM, Gorgels TPM, Wade DT, Verbunt JA. Long-term outcome after survival of a cardiac arrest: a prospective longitudinal cohort study. Neurorehabil Neural Repair. 2017;31:530–539. doi: 10.1177/1545968317697032. [DOI] [PubMed] [Google Scholar]
- 3.Go AS, Mozaffarian D, Roger VL, et al. Heart disease and stroke statistics--2014 update: a report from the American Heart Association. Circulation. 2014;129:e28–e292. doi: 10.1161/01.cir.0000441139.02102.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Part 1: Introduction to the International Guidelines 2000 for CPR and ECC: a consensus on science. Circulation. 2000;102:I1–11. doi: 10.1161/01.cir.102.suppl_1.i-1. [DOI] [PubMed] [Google Scholar]
- 5.Yang CL, Wen J, Li YP, Shi YK. Cardiocerebral resuscitation vs cardiopulmonary resuscitation for cardiac arrest: a systematic review. Am J Emerg Med. 2012;30:784–793. doi: 10.1016/j.ajem.2011.02.035. [DOI] [PubMed] [Google Scholar]
- 6.Ohsawa I, Ishikawa M, Takahashi K, et al. Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med. 2007;13:688–694. doi: 10.1038/nm1577. [DOI] [PubMed] [Google Scholar]
- 7.Fan Z, Gao Y, Huang Z, et al. Protective effect of hydrogen-rich saline on pressure overload-induced cardiac hypertrophyin rats: possible role of JAK-STAT signaling. BMC Cardiovasc Disord. 2018;18:32. doi: 10.1186/s12872-018-0773-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang CS, Kawamura T, Toyoda Y, Nakao A. Recent advances in hydrogen research as a therapeutic medical gas. Free Radic Res. 2010;44:971–982. doi: 10.3109/10715762.2010.500328. [DOI] [PubMed] [Google Scholar]
- 9.Chen CH, Manaenko A, Zhan Y, et al. Hydrogen gas reduced acute hyperglycemia-enhanced hemorrhagic transformation in a focal ischemia rat model. Neuroscience. 2010;169:402–414. doi: 10.1016/j.neuroscience.2010.04.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhan Y, Chen C, Suzuki H, Hu Q, Zhi X, Zhang JH. Hydrogen gas ameliorates oxidative stress in early brain injury after subarachnoid hemorrhage in rats. Crit Care Med. 2012;40:1291–1296. doi: 10.1097/CCM.0b013e31823da96d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ohta S. Molecular hydrogen as a preventive and therapeutic medical gas: initiation, development and potential of hydrogen medicine. Pharmacol Ther. 2014;144:1–11. doi: 10.1016/j.pharmthera.2014.04.006. [DOI] [PubMed] [Google Scholar]
- 12.Camara R, Huang L, Zhang JH. The production of high dose hydrogen gas by the AMS-H-01 for treatment of disease. Med Gas Res. 2016;6:164–166. doi: 10.4103/2045-9912.191362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cui J, Chen X, Zhai X, et al. Inhalation of water electrolysis-derived hydrogen ameliorates cerebral ischemia-reperfusion injury in rats - A possible new hydrogen resource for clinical use. Neuroscience. 2016;335:232–241. doi: 10.1016/j.neuroscience.2016.08.021. [DOI] [PubMed] [Google Scholar]
- 14.Li H, Chen O, Ye Z, et al. Inhalation of high concentrations of hydrogen ameliorates liver ischemia/reperfusion injury through A2A receptor mediated PI3K-Akt pathway. Biochem Pharmacol. 2017;130:83–92. doi: 10.1016/j.bcp.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 15.Liu X, Ma C, Wang X, et al. Hydrogen coadministration slows the development of COPD-like lung disease in a cigarette smoke-induced rat model. Int J Chron Obstruct Pulmon Dis. 2017;12:1309–1324. doi: 10.2147/COPD.S124547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Peng Z, Chen W, Wang L, et al. Inhalation of hydrogen gas ameliorates glyoxylate-induced calcium oxalate deposition and renal oxidative stress in mice. Int J Clin Exp Pathol. 2015;8:2680–2689. [PMC free article] [PubMed] [Google Scholar]
- 17.Wang R, Wu J, Chen Z, Xia F, Sun Q, Liu L. Postconditioning with inhaled hydrogen promotes survival of retinal ganglion cells in a rat model of retinal ischemia/reperfusion injury. Brain Res. 2016;1632:82–90. doi: 10.1016/j.brainres.2015.12.015. [DOI] [PubMed] [Google Scholar]
- 18.Huo TT, Zeng Y, Liu XN, et al. Hydrogen-rich saline improves survival and neurological outcome after cardiac arrest and cardiopulmonary resuscitation in rats. Anesth Analg. 2014;119:368–380. doi: 10.1213/ANE.0000000000000303. [DOI] [PubMed] [Google Scholar]
- 19.Hendrickx HH, Rao GR, Safar P, Gisvold SE. Asphyxia, cardiac arrest and resuscitation in rats. I Short term recovery. Resuscitation. 1984;12:97–116. doi: 10.1016/0300-9572(84)90062-5. [DOI] [PubMed] [Google Scholar]
- 20.Chiueh CC, Andoh T, Lai AR, Lai E, Krishna G. Neuroprotective strategies in Parkinson's disease: protection against progressive nigral damage induced by free radicals. Neurotox Res. 2000;2:293–310. doi: 10.1007/BF03033799. [DOI] [PubMed] [Google Scholar]
- 21.Lee YJ, Park KH, Park HH, et al. Cilnidipine mediates a neuroprotective effect by scavenging free radicals and activating the phosphatidylinositol 3-kinase pathway. J Neurochem. 2009;111:90–100. doi: 10.1111/j.1471-4159.2009.06297.x. [DOI] [PubMed] [Google Scholar]
- 22.Hayashida K, Sano M, Kamimura N, et al. H(2) gas improves functional outcome after cardiac arrest to an extent comparable to therapeutic hypothermia in a rat model. J Am Heart Assoc. 2012;1:e003459. doi: 10.1161/JAHA.112.003459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hayashida K, Sano M, Kamimura N, et al. Hydrogen inhalation during normoxic resuscitation improves neurological outcome in a rat model of cardiac arrest independently of targeted temperature management. Circulation. 2014;130:2173–2180. doi: 10.1161/CIRCULATIONAHA.114.011848. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.