Abstract
The diagnosis of Alzheimer disease (AD) dementia is based primarily on the clinical history and examination, but advances in understanding the pathophysiology of AD have led to new diagnostic methods. When used appropriately, the tests can provide strong positive or negative evidence AD dementia. This article described which patients may benefit from additional testing using Cerebrospinal Fluid (CSF) biomarkers, amyloid imaging, quantitative structural magnetic resonance imaging (MRI), and fluoro-deoxyglucose positron emission tomography (FDG-PET).
Introduction
The development of biomarkers and imaging techniques for the diagnosis of Alzheimer disease (AD) has progressed considerably in the last several years. This article will describe the advanced diagnostic methods for AD that are currently available in clinical practice and when they are helpful.
A clear diagnosis of AD dementia can be helpful to patients in many ways. The diagnosis validates and explains the difficulties that patients have been experiencing and that caregivers have been noticing. Patients and caregivers may find comfort and support in educating themselves about the disease. Confirming diagnosis encourages patients and caregivers to join organizations, such as the Alzheimer’s Association, that offer resources to manage the illness. Knowledge of their loved one’s diagnosis may help caregivers to make plans that include choosing an appropriate living situation, completing legal paperwork and making a plan for supportive care. Patients with a clear diagnosis may also be more likely to start medications that help with the symptoms of disease.
Despite advances in diagnostic testing, AD dementia remains a clinical diagnosis. The available tests are not perfect and should be used by a clinician to help choose between diagnoses or, in some cases, to provide greater confidence in the diagnosis of AD dementia. There is no indication for testing in asymptomatic individuals, particularly because we have no effective preventative therapy. A positive result also has not been demonstrated to inevitably result in dementia, and even if it did, it cannot predict the time frame within which the individual may develop symptoms, i.e. whether he or she will develop dementia in 1 year or 20 years. An asymptomatic individual who tests positive is likely to experience anxiety about developing dementia, but cannot significantly change their risk of dementia or predict when they will first manifest symptoms. Therefore, testing is not offered to asymptomatic individuals.
Indications for Referral and/or Advanced Diagnostic Testing
Alzheimer disease is a progressive neurodegenerative disease characterized neuropathologically by amyloid plaques, neurofibrillary tangles, and loss of neurons. The clinical syndrome of cognitive decline that accompanies AD pathology is termed AD dementia.1 Practice parameters developed by the American Academy of Neurology,2 as described by an article in this issue entitled “Alzheimer disease,” allow accurate diagnosis in many cases. The work-up consists of a detailed history from someone who knows the patient well, a screening neurological examination, routine laboratory testing, and brain imaging. The evaluation focuses on ruling out reversible causes of dementia and establishing a clinical history typical of AD dementia.
After a complete initial evaluation, it is still sometimes unclear whether a patient has AD dementia or some other cause of cognitive dysfunction. A referral to a dementia specialist may be indicated for a patient with unusual or complex cognitive problems, particularly if primary care physicians do not have the time or resources to perform a very detailed evaluation. A patient that falls into the following categories may particularly benefit from an evaluation by a dementia specialist and/or advanced diagnostic testing:
1. Early Onset Dementia
A patient younger than 65 with symptoms of dementia and no additional conditions explaining his or her cognitive decline may benefit from either cerebrospinal fluid (CSF) testing or an amyloid-PET scan to help support the diagnosis. Since AD dementia is unusual in patients before age 65,3 and because the diagnosis of AD dementia will likely have a more devastating impact on a younger patient’s life, a supporting laboratory test can reassure both the patient and clinician that the diagnosis is correct. Additionally, in patients with a strong family history of AD dementia and a very early age of onset (before age 55), genetic testing may be considered in cases with an appropriate pedigree demonstrating autosomal dominant inheritance over several generations.
2. Prominent Behavioral or Language Problems
A patient who develops prominent behavioral changes and/or language problems early in the course of disease may have a frontotemporal dementia (FTD). Additional history of the patient’s behavioral changes and a detailed examination of the patient’s language function are important. FDG-PET is sometimes used to evaluate patterns of regional hypometabolism and to help discriminate between dementia caused by FTD or by other diseases, including a rare “frontal variant” of AD4. When indicated, the presence of AD pathology can be established using either CSF testing or an amyloid-PET scan.
3. Prominent Visuospatial Dysfunction
A rare form of AD dementia, posterior cortical dysfunction, presents with prominent visuospatial problems.5 The patient’s eye exam may appear normal, but the patient may have deficits in processing visual information that can render him or her functionally blind. A detailed neurological examination is required to evaluate the extent of the visual deficits. A positive CSF test or an amyloid-PET scan supports the diagnosis of AD. Other causes of posterior cortical dysfunction include Parkinson disease and Creutzfeldt-Jakob disease.
4. Rapidly Progressive Dementia
Alzheimer disease dementia usually causes a very slowly progressive decline in memory and thinking and rarely results in an appreciable change from one week to the next or even one month to the next. When cognition rapidly worsens in a patient with pre-existing AD dementia, it may indicate a medical problem such as an infection. In a patient without pre-existing dementia, a sudden decline in memory and thinking over weeks to months may be caused by a number of different etiologies, some of which may be treatable. The work-up is often complex and may require hospitalization (see article in this series on rapidly progressive dementia).
5. Uncertain Dementia
A patient with very mild memory changes consistent with AD dementia and no complicating factors usually has incipient AD dementia. Further testing is typically not necessary; instead, following the patient clinically over the next year or two usually provides information leading to a diagnosis. In a patient with very mild but consistent cognitive decline who has medical or psychiatric problems that complicate the diagnosis, further testing can help establish whether AD-related brain pathology is present. Positive CSF testing or amyloid-PET supports the diagnosis of AD, but does not rule-out other contributors to cognitive dysfunction. Quantitative MRI allows evaluation for disproportionate cortical or hippocampal atrophy suggestive of AD or another neurodegenerative disease.
6. Individual Factors
Some patients may need a more certain diagnosis. For example, high functioning individuals with very mild AD symptoms may request confirmatory tests because an AD diagnosis would precipitate irreversible career changes (e.g. retiring, selling a company, etc.). The clinician must consider the degree of additional certainty that would be contributed by the test, and whether the results would definitely change an individual’s decisions. To date, insurance companies and Medicare usually do not cover such additional studies.
The new diagnostic tests for AD dementia have resulted from advances in our understanding of AD.6 We have learned that deposition of amyloid-β (Aβ) protein into neuritic plaques begins many years prior to the onset of symptoms and is accompanied by decreases in the 42 amino acid form of Aβ (A42) in CSF. Tau, a protein involved in neuronal structure, increases in the CSF prior to the onset of symptoms, possibly reflecting neuronal injury. Radiolabeled chemicals that bind amyloid in the brain have been developed that allow measurement of the total amount of amyloid in the brain via a PET scan. FDG-PET, which images the metabolism of the brain, can be used to evaluate the pattern of brain hypometabolism. Quantitative structural brain MRI can be used to evaluate for cortical and hippocampal atrophy due to neuronal death, which also begins prior to the onset of dementia.
Cerebrospinal Fluid Biomarkers
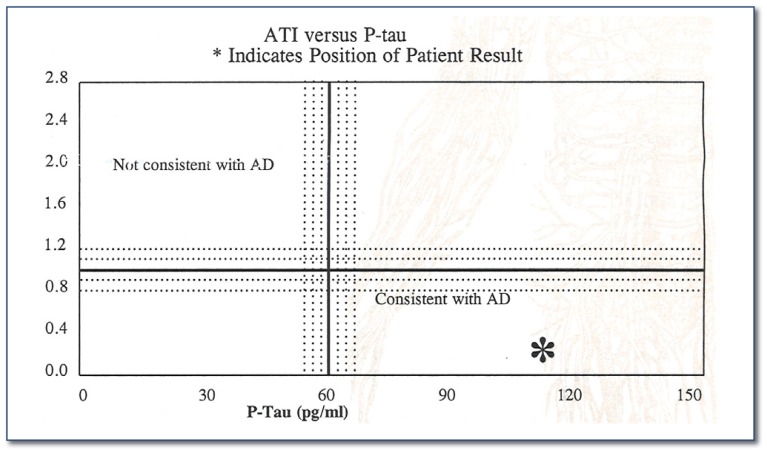
Cerebrospinal Fluid (CSF) biomarkers for AD require collection of the CSF via a lumbar puncture (spinal tap), a procedure which is very safe and reliable when performed by experienced personnel. CSF levels of three AD-related proteins are measured: Aβ42, tau, and tau phosphorylated at position 181. Patients with AD have low levels of CSF Aβ42 and high levels of CSF tau and phospho-tau, leading to elevated ratios of tau:Aβ42 and phosphotau:Aβ42.7 Most patients with even very mild AD dementia have elevated ratios of tau:Aβ42 and phosphotau:Aβ42.8 Athena Diagnostics® is a commercial laboratory that performs the assays (http://www.athenadiagnostics.com/content/test-catalog/find-test/service-detail/q/id/310) and provides a graphical interpretation of the results as consistent with AD (See Figure 1), not consistent with AD, and borderline. Some private insurance companies and Medicare may cover the costs of the lumbar puncture and CSF testing.
Figure 1.
Athena Diagnostics® is a commercial laboratory that performs the assays that provide a graphical interpretation of the results as consistent with AD.
Amyloid Imaging
Several positron emission tomography (PET) tracers have been developed to non-invasively image fibrillary beta-amyloid plaques in the brain.9 One of these PET tracers, florbetapir (AmyvidTM), is currently FDA-approved and commercially available10, 11 and others are likely to be approved soon. The levels of radioactivity in the cerebral cortex measured through florbetapir-PET are correlated with the frequency of neuritic amyloid plaques at autopsy.12 Following intravenous injection of florbetapir, a PET scan is performed that is interpreted as either positive, indicating moderate to frequent neuritic amyloid plaques, or negative, indicating no to sparse neuritic amyloid plaques (See Figure 2).
Figure 2.
Following intravenous injection of florbetapir, a PET scan is performed that is interpreted as either positive, indicating moderate to frequent neuritic amyloid plaques, or negative, indicating no to sparse neuritic amyloid plaques.
A positive scan supports the diagnosis of AD dementia, but is not diagnostic because a substantial number of older individuals have neuritic plaques but are cognitively normal.13 A negative scan makes the diagnosis of AD less likely, but does not completely rule it out. Florbetapir-PET is not currently reimbursed by the Centers for Medicare and Medicaid Services (CMS) or by most private insurance companies. Imaging centers that provide Amyvid-PET are listed at the following website: https://www.amyvidimagingcenterlookup.com/.
Quantitative Structural MRI
Nearly all patients evaluated for AD undergo structural brain imaging, usually MRI. Patients with AD usually have cortical and hippocampal atrophy.14–16 A number of commercially available software packages exist that can perform quantitative volumetric imaging analysis to determine whether an individual’s regional or whole brain volume is within the range of age-and education-matched normal older adults. Some neuroradiologists perform quantification of the degree of atrophy versus established norms using commercial packages or methods developed within the institution. Serial MRI also may be instructive, as AD is associated with progressive cerebral atrophy.
Fluorodeoxyglucose (FDG)-PET
The relative rates of glucose metabolism of different brain regions can be measured non-invasively with PET using the fluorine-18 labeled glucose analogue 2-deoxy-2-fluoro-D-glucose (FDG). FDG is intravenously injected and a PET scan is performed. Brain regions that are metabolically active preferentially take up FDG while brain regions with neuronal loss or synaptic dysfunction do not. The reader evaluates the scan to determine whether the metabolic activity is normal or whether some brain regions show evidence of hypometabolism. AD typically has a pattern of posterior temporal and parietal hypometabolism while FTD usually has frontal and anterior temporal hypometabolism.4, 17 FDG-PET is used routinely for oncologic and neurologic imaging and is available at many PET imaging centers. Brain-FDG/PET is usually reimbursed by Medicare and private insurers when the clinical indication is to help differentiate between AD and FTD.
Biography
Top, from left: Suzanne E. Schindler, MD, PhD, is a Clinical and Research Postdoctoral Fellow. Jonathan McConathy, MD, PhD, Assistant Professor, Radiology. Bottom, from left: Beau M. Ances, MD, PhD, is an Associate Professor, Neurology. Marc I. Diamond, MD, is the David Clayson Professor of Neurology. All are at the Knight Alzheimer’s Disease Research Center at the Washington University School of Medicine in St. Louis.
Contact: schindlers@neuro.wustl.edu




Footnotes
Disclosure
None reported.
References
- 1.McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s & dementia : the journal of the Alzheimer’s Association. 2011;7(3):263–9. doi: 10.1016/j.jalz.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Knopman DS, DeKosky ST, Cummings JL, et al. Practice parameter: diagnosis of dementia (an evidence-based review) Report of the Quality Standards Subcommittee of the American Academy of Neurology Neurology. 2001;56(9):1143–53. doi: 10.1212/wnl.56.9.1143. [DOI] [PubMed] [Google Scholar]
- 3.Ferri CP, Prince M, Brayne C, et al. Global prevalence of dementia: a Delphi consensus study. Lancet. 2005;366(9503):2112–7. doi: 10.1016/S0140-6736(05)67889-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Foster NL, Heidebrink JL, Clark CM, et al. FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer’s disease. Brain. 2007;130(Pt 10):2616–35. doi: 10.1093/brain/awm177. [DOI] [PubMed] [Google Scholar]
- 5.Crutch SJ, Lehmann M, Schott JM, Rabinovici GD, Rossor MN, Fox NC. Posterior cortical atrophy. Lancet Neurol. 2012;11(2):170–8. doi: 10.1016/S1474-4422(11)70289-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jack CR, Jr, Knopman DS, Jagust WJ, et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013;12(2):207–16. doi: 10.1016/S1474-4422(12)70291-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Galasko D, Chang L, Motter R, et al. High cerebrospinal fluid tau and low amyloid beta42 levels in the clinical diagnosis of Alzheimer disease and relation to apolipoprotein E genotype. Archives of neurology. 1998;55(7):937–45. doi: 10.1001/archneur.55.7.937. [DOI] [PubMed] [Google Scholar]
- 8.Fagan AM, Roe CM, Xiong C, Mintun MA, Morris JC, Holtzman DM. Cerebrospinal fluid tau/beta-amyloid(42) ratio as a prediction of cognitive decline in nondemented older adults. Archives of neurology. 2007;64(3):343–9. doi: 10.1001/archneur.64.3.noc60123. [DOI] [PubMed] [Google Scholar]
- 9.Rowe CC, Villemagne VL. Brain amyloid imaging. Journal of nuclear medicine : official publication Society of Nuclear Medicine. 2011;52(11):1733–40. doi: 10.2967/jnumed.110.076315. [DOI] [PubMed] [Google Scholar]
- 10.Doraiswamy PM, Sperling RA, Coleman RE, et al. Amyloid-beta assessed by florbetapir F 18 PET and 18-month cognitive decline: a multicenter study. Neurology. 2012;79(16):1636–44. doi: 10.1212/WNL.0b013e3182661f74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Clark CM, Pontecorvo MJ, Beach TG, et al. Cerebral PET with florbetapir compared with neuropathology at autopsy for detection of neuritic amyloid-beta plaques: a prospective cohort study. Lancet Neurol. 2012;11(8):669–78. doi: 10.1016/S1474-4422(12)70142-4. [DOI] [PubMed] [Google Scholar]
- 12.Clark CM, Schneider JA, Bedell BJ, et al. Use of florbetapir-PET for imaging beta-amyloid pathology. JAMA. 2011;305(3):275–83. doi: 10.1001/jama.2010.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rodrigue KM, Kennedy KM, Devous MD, Sr, et al. beta-Amyloid burden in healthy aging: regional distribution and cognitive consequences. Neurology. 2012;78(6):387–95. doi: 10.1212/WNL.0b013e318245d295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kesslak JP, Nalcioglu O, Cotman CW. Quantification of magnetic resonance scans for hippocampal and parahippocampal atrophy in Alzheimer’s disease. Neurology. 1991;41(1):51–4. doi: 10.1212/wnl.41.1.51. [DOI] [PubMed] [Google Scholar]
- 15.Fotenos AF, Snyder AZ, Girton LE, Morris JC, Buckner RL. Normative estimates of cross-sectional and longitudinal brain volume decline in aging and AD. Neurology. 2005;64(6):1032–9. doi: 10.1212/01.WNL.0000154530.72969.11. [DOI] [PubMed] [Google Scholar]
- 16.van de Pol LA, Hensel A, Barkhof F, Gertz HJ, Scheltens P, van der Flier WM. Hippocampal atrophy in Alzheimer disease: age matters. Neurology. 2006;66(2):236–8. doi: 10.1212/01.wnl.0000194240.47892.4d. [DOI] [PubMed] [Google Scholar]
- 17.Mosconi L. Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. FDG-PET studies in MCI and AD. Eur J Nucl Med Mol Imaging. 2005;32(4):486–510. doi: 10.1007/s00259-005-1762-7. [DOI] [PubMed] [Google Scholar]