Abstract
Previous research has shown that people represent each other’s tasks and actions when acting together. However, less is known about how co-actors represent each other’s action sequences. Here, we asked whether co-actors represent the order of each other’s actions within an action sequence, or whether they merely represent the intended end state of a joint action together with their own contribution. In the present study, two co-actors concurrently performed action sequences composed of two actions. We predicted that if co-actors represent the order of each other’s actions, they should experience interference when the order of their actions differs. Supporting this prediction, the results of six experiments consistently showed that co-actors moved more slowly when performing the same actions in a different order compared to performing the same actions in the same order. In line with findings from bimanual movement tasks, our results indicate that interference can arise due to differences in movement parameters and due to differences in the perceptual characteristics of movement goals. The present findings extend previous research on co-representation, providing evidence that people represent not only the elements of another’s task, but also their temporal structure.
Keywords: Social cognition, Joint action, Coordination, Co-representation, Action sequence, Bimanual control
1. Introduction
Human motor behavior relies on precise action planning and control. We need to decide which button in the elevator to press, when and how far to jump over a puddle, and we need to coordinate our left and right limb during a dance routine. When acting jointly with others, coordination is not only required within an individual’s motor system but also between the independent motor systems of two (or more) individuals (e.g., Knoblich and Jordan, 2003, Wolpert et al., 2003), such as when two dance partners coordinate their steps, or when pianists play a duet together (e.g., Keller, Knoblich, & Repp, 2007).
Previous research has suggested that the coordination of actions within and between individuals may rely on similar processes (e.g., Fine and Amazeen, 2011, Richardson et al., 2007, Schmidt et al., 1990, Schmidt and Turvey, 1994, Schmidt et al., 1998, Schmidt and Richardson, 2008). For instance, when performing repetitive, rhythmic movements, a tendency to entrain to the same movement rhythm was observed between individuals in a group (e.g., Fine and Amazeen, 2011, Richardson et al., 2007, Schmidt et al., 1990) as well as between the limbs of one individual acting bimanually (e.g., Heuer, 1996, Heuer and Klein, 2005, Kelso et al., 1979, Mechsner et al., 2001).
Further similarities between intra- and interpersonal processing have been found at the level of task and action representation. When tasks are distributed between two co-actors, similar response selection conflicts (Atmaca et al., 2011, Sebanz et al., 2003), attention allocation processes (Böckler et al., 2012, Kourtis et al., 2014, Welsh et al., 2005), lexical processes (Hoedemaker et al., 2017, Kuhlen and Abdel Rahman, 2017), and motor priming effects (Griffiths and Tipper, 2009, Welsh et al., 2009) occur as when one individual performs the whole task alone. Further evidence comes from interpersonal movement coordination tasks. When two co-actors concurrently perform movements of different difficulty, they make similar adjustments in action execution (Fine and Amazeen, 2011, Vesper et al., 2013) as one individual performing movements of different difficulty with her two limbs (Fowler et al., 1991, Kelso et al., 1979, Marteniuk et al., 1984). Moreover, van der Wel and Fu (2015; see also Schmitz, Vesper, Sebanz, & Knoblich, 2017) demonstrated that when only one of two co-actors needs to move over an obstacle, the actor without obstacle also increases her movement amplitude. Again, this result pattern resembles findings earlier obtained in a bimanual version of the same task in which the limb without obstacle moved as if it were also clearing an obstacle (Kelso, Putnam, & Goodman, 1983). Finally, della Gatta et al. (2017) showed that when one person draws a line while the other draws a circle, the line trajectories tend to become ovalized. This corresponds to findings from the bimanual literature showing that the same interference occurs when drawing a circle with one hand while drawing a line with the other (Franz, Zelaznik, & McCabe, 1991), indicating that the action representations of line and circle interfere with one another.
Taken together, the research so far indicates that similar mechanisms operate in intrapersonal and interpersonal action planning and action coordination. In particular, people’s tendency to represent a co-actor’s part of a task (e.g., Sebanz et al., 2003) often leads to similar interferences as when one individual performs the whole task alone. This co-representation tendency has been mainly observed in studies where co-actors in a joint action performed discrete, individual actions such as pressing a response button or performing a goal-directed forward jump or an aiming movement. However, in everyday life, people often perform multiple actions in a sequence. Therefore, the present study asked how co-actors represent each other’s actions when they perform sequences of actions to achieve temporal coordination at the end. We examined whether similarities between intra- and interpersonal coordination can be observed.
To illustrate, consider two dancers who perform a dance move that requires them to approach each other so that they arrive synchronously at the center of the dance floor. The male dancer performs a long step followed by a short step whereas the female dancer performs a short step followed by a long step. Our question is whether the two dancers represent the order of actions within each other’s action sequence, or whether they merely represent the end state that the two action sequences produce, together with their own contribution. Does the male dancer represent the female dancer’s sequence of a short step followed by a long one, or does he merely represent her meeting him at the center, while ignoring the specifics of how she is going to get there? Abstracting from the example, we consider a situation where two co-actors perform the same actions in a different order (i.e., B-A vs. A-B), with the joint goal of synchronized arrival at a pre-defined position (see Fig. 1). Reaching a synchronized end state in this type of situation does not necessarily require co-actors to take into account each other’s actions because synchronization can be based on the overall duration of the sequence which is not affected by the order of actions within the sequence (on anticipatory temporal prediction and sensorimotor synchronization, see e.g., Repp and Su, 2013, van der Steen and Keller, 2013).
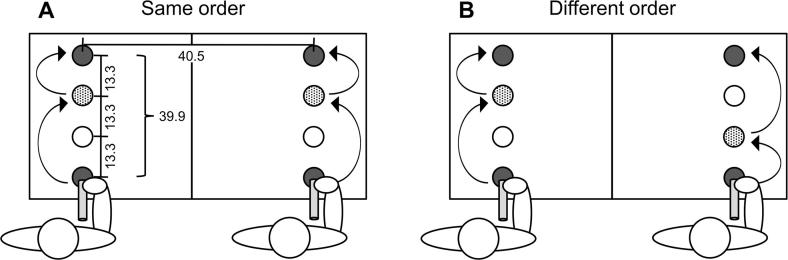
Fig. 1.
Schematic depiction of a joint action situation in which two co-actors perform the same actions in a different order (i.e., B-A vs. A-B), with the joint goal of synchronizing the end state of their action sequences.
Before asking whether co-actors represent the discrete actions that make up each other’s action sequences, we briefly consider the question of how co-actors plan and execute their own action sequences. Prior research has shown that when performing a sequence of two consecutive movements, people do not plan and parametrize each of the movements separately. Rather, both movement segments are planned in advance and stored in a “buffer” such that the second movement can be read from this buffer while the first movement is being executed (“movement integration hypothesis”; Adam et al., 2000). This online preparation of the second movement during the execution of the first movement has the effect of slowing down the first movement: Completing a movement that is followed by a second movement will take longer than completing the same movement on its own – an effect known as the “one-target advantage” (for a recent review, see Bested, de Grosbois, & Tremblay, 2018). In the context of the present research, we assume that even though movement sequences are typically planned in an integrated fashion, the order in which the movement segments are to be executed must surely be part of this plan. Thus, when it comes to co-representing others’ action sequences, we ask whether people represent others’ actions as an ordered sequence or whether they merely take into account the other’s sequence as a whole. An ordered sequence may be represented either in terms of two separate movement segments or as one complex movement with two pre-planned stages.
To test whether co-actors represent the order of actions within each other’s action sequence, we designed a novel joint movement task where two co-actors performed sequences of goal-directed, speeded aiming movements towards targets on a table (Fig. 2). The sequences consisted of two movements of differing distances such that each actor performed a short movement followed by a long one or a long movement followed by a short one. Their joint goal was to synchronize arrival times at the endpoint of the sequence. One way to facilitate synchronization is to make the overall duration of one’s own action sequence as invariant, and thus predictable, as possible (Vesper et al., 2016, Vesper et al., 2011). This strategy does not require representing the order of a co-actor’s actions.
Fig. 2.
Sketch of experimental setup for Experiments 1–3, with co-actors performing long and short movements either in the same order (A) or in a different order (B). Distances are in cm.
However, if co-actors represent the order of actions within each other’s action sequence, they may experience interference when the order of their own actions differs from the order of their co-actor’s actions. This hypothesis follows from the assumption that behavior within and between individuals is organized by similar mechanisms (e.g., Schmidt et al., 1990). In particular, the present interpersonal task relates to studies on bimanual motor control showing that people encounter intermanual interference when trying to simultaneously perform movements of differing spatial characteristics. Interference is reflected in longer initiation times (Diedrichsen et al., 2006, Diedrichsen et al., 2003, Heuer and Klein, 2006, Spijkers et al., 1997) and longer movement times (Albert et al., 2007, Diedrichsen et al., 2001, Heuer and Klein, 2006) for movements of differing distances or directions.
Two distinct sources for this intermanual interference have been identified in the motor control literature. On the one hand, interference can occur at the level of motor representations, where different movement parameters for left and right hand need to be concurrently specified during motor programming (Diedrichsen et al., 2006, Heuer and Klein, 2006, Heuer, 1993, Spijkers et al., 1997). On the other hand, interference can also occur at a higher cognitive level of goal-selection, where different movement goals are selected and assigned to left and right hand (Diedrichsen et al., 2001, Diedrichsen et al., 2003, Diedrichsen et al., 2006, Ivry et al., 2004, Kunde and Weigelt, 2005, Mechsner and Knoblich, 2004, Mechsner et al., 2001, Weigelt, 2007, Weigelt et al., 2007).
For the present interpersonal task, these findings imply that co-actors may show similar interference – at a motor and/or cognitive level – when they represent the actions within each other’s action sequence. At the motor level, actors might be unable to plan and execute their own movements independently of a co-actor’s movements such that interference occurs when a co-actor’s movements differ in crucial movement parameters. In contrast to bimanual aiming movements where this type of movement-related interference is attributed to interhemispheric communication (Diedrichsen et al., 2006, Franz et al., 1996, Kennerley et al., 2002), interpersonal interference would arise from motor simulation processes whereby co-actors use their own motor systems to simulate and predict each other’s actions (e.g., Wilson and Knoblich, 2005, Wolpert et al., 2003). When engaging in such simulation processes, a person not only plans and executes her own movements, but in addition specifies another person’s movement parameters – all within the same motor system. If the two sets of movements differ in crucial parameters, interference may occur.
At the cognitive level, co-actors might represent each other’s actions in terms of action goal states, specifying the perceptual characteristics of movement targets. Thus, when representing not only their own but also a co-actor’s perceptually different movement targets, response selection may get more demanding and actors might have difficulties keeping apart their own and their co-actor’s movement targets. This would be in line with the finding that individuals performing a similar task bimanually have difficulties when needing to select target locations that differ perceptually (cf. Diedrichsen et al., 2003).1 The following six experiments were performed to establish whether co-actors represent the order of each other’s actions and if so, at which level interference arises.
2. Experiment 1
Experiment 1 tested the hypothesis that co-actors represent the order of actions within each other’s action sequences when trying to coordinate the endpoints of these sequences by reaching the final target location at the same time. If this hypothesis is correct, co-actors should show interference when the order of their actions differs, resulting in slower performance than when the order is the same. If co-actors do not represent the actions within each other’s action sequence (but merely the end state of the whole sequence), no interference – and thus no performance slowdown – is expected.
2.1. Method
Participants. In Experiment 1, fifteen women and five men participated in randomly-matched pairs (6 only-female pairs, 1 only-male pair, Mage = 22.5 years, SDage = 3.65 years, range: 18–32). Members of one pair knew each other beforehand. All participants were right-handed and had normal or corrected-to-normal vision. They signed prior informed consent and received monetary compensation (1500 Hungarian Forint ≈ 5 EUR per 45 min). The study was approved by the Hungarian United Ethical Review Committee for Research in Psychology (EPKEB).
Apparatus. The experimental setup consisted of a table (110 cm long, 55 cm wide) with two parallel columns of four circular cardboard markers (diameter 5 cm) attached to it (Fig. 2). The two columns were aligned with the table’s short side at a horizontal distance of 40.5 cm. The center-to-center distance between markers in a column was 13.3 cm. The first marker in each column indicated the start location and the last marker indicated the final target. The two inner markers in each column served as close and far intermediate targets. Start location and final target were colored blue and intermediate targets were colored orange.
The two participants were seated next to each other at the table’s long side (Fig. 2) and held a wooden dowel rod (height: 20 cm, diameter: 2.5 cm, weight: 50 g) in their right hand with a power grip. A Polhemus G4 electro-magnetic motion capture system (www.polhemus.com) was used to record participants’ movement data at a constant sampling rate of 120 Hz via a motion capture sensor that was attached to the top of each dowel rod. Instructions were displayed on a 24″ Asus computer screen (resolution 1920 × 1080 pixels, refresh rate 60 Hz), positioned on a separate table in front of the participants at a distance of 160 cm (see Fig. 4). Data recording was controlled by Matlab 2014a.
Fig. 4.
(A) Displays used in Experiments 1–5 to instruct participants to perform action sequences in the ‘same order’ or in a ‘different order’. ‘Neutral’ refers to the uninformative displays that were presented while co-actors performed the task in Experiments 2–5. (B) The fourth column shows the displays used in the individual baseline.
Procedure and stimuli. The participants were instructed to perform two consecutive aiming movements as fast as possible, moving from the start location to one of the intermediate targets and onwards to the final target while synchronizing their landing times on the final target. Importantly, the synchronization instruction applied to the final target only. Accordingly, the experimenter verbally instructed the two participants to “try to synchronize your movements on the final target and to reach the final target as fast as possible”. At the beginning of each trial, participants rested their dowels on the start location until they heard a short tone (440 Hz, 100 ms) that served as a go signal. Participants then performed the instructed movement sequence. After having landed on the final target, they moved back to the start location. Then the next trial started. There was a 5 s-interval between the start of one trial and the start of the next trial, giving participants sufficient time to complete their movement sequences and to return to the start location for a short rest (for comparable intervals used in a similar task, see Schmitz et al., 2017, van der Wel and Fu, 2015).
There were 12 blocks of twelve trials (144 trials total). Across blocks, it was varied whether participants performed the actions in their respective action sequence in the same order or in a different order, i.e., whether both participants moved to the same intermediate target or not. Accordingly, each block started with an instruction that specified the action sequences to be performed by each participant. The display showed the same layout of markers as seen on the table (Fig. 4A, row 1, panels 1–2). The start and final markers had the same color as the markers on the table. One intermediate marker in each column was colored orange and one was colored white. In blocks of the ‘same order’ condition, the same intermediate targets in the two columns were colored orange (Fig. 4A, row 1, panel 1). This served to instruct the two participants to either both perform a short movement followed by a long movement (‘short-long’) or a long movement followed by a short movement (‘long-short’), see Fig. 3. In blocks of the ‘different order’ condition, different intermediate markers in each column were colored orange (Fig. 4A, row 1, panel 2). This instructed the two participants to perform different action sequences so that one participant performed a short movement followed by a long movement whereas the other participant performed a long movement followed by a short movement, see Fig. 3. Once participants had indicated that they were ready to start, the experimenter manually started the block and a start tone marked the beginning of the first trial. The display remained on the computer screen throughout the entire block.
Fig. 3.
Four types of blocks in Experiments 1–3. Participants performed sequences of two movements of differing distance, in the order ‘short-long’ or ‘long-short’. The two participants in a pair either performed the same sequence or a different sequence. Note that in the ‘different order’ condition (right column) the labels ‘short-long’/‘long-short’ apply to the order for participant P1 only.
The twelve blocks were run in 3 sets of 4 blocks. Each set of blocks consisted of two blocks where participants performed their actions in the same order, one block where Participant 1 performed short-long sequences and Participant 2 performed long-short sequences, and one block where Participant 1 performed long-short sequences and Participant 2 performed short-long sequences (see Fig. 3). The order of blocks was randomized within each set of 4 blocks, with the constraint that blocks of the ‘same order’ condition alternated with blocks of the ‘different order’ condition.
Before performing the joint task, participants completed two blocks of individual trials that served as a baseline. Only one column of markers was displayed either on the left or right side of the computer screen, depending on the participant’s seating location (which remained the same in the joint condition), see Fig. 4B (row 1). Participants performed one block of short-long sequences and one block of long-short sequences. While one participant performed the task individually, the other participant waited outside of the testing room, and then they switched.
At the end of the experiment, participants were asked to report (in writing) whether they had used a specific strategy to achieve the task goal of synchronizing with their partner. The experiment lasted about 45–50 min in total.
Data preparation and analysis. Before extracting movement times (MTs) from participants’ movement data, all movement trajectories were filtered using a 4th-order two-way low-pass Butterworth filter with a cutoff at 20 Hz. Movement onset was defined as the point when movement velocity exceeded 5% of peak velocity for the first time (see, e.g., Zopf, Truong, Finkbeiner, Friedman, & Williams, 2011), with peak velocity defined as the maximal horizontal velocity. Horizontal velocity was calculated by dividing the change in spatial position along the horizontal axis by the change in time. Movement offset was defined as the point of minimal movement height after the intermediate target had been passed. This spatial criterion was used because initial analyses had indicated that it was more reliable for the present data than using a low velocity threshold.2 MT was computed as the time interval between movement onset and offset, thus capturing the time participants needed to move from the start location to the final target. We also computed the time interval between movement onset and arrival at the intermediate target (MT1st) to capture the time participants needed to move from the start location to the intermediate target. The point of arrival at the intermediate target was defined as the first point of minimal movement height. As a measure of coordination performance in each pair, we computed the mean absolute asynchrony between co-actors’ landing times on the final target. Matlab 2013b and 2016a was used for data preparation, and statistical analyses were performed in IBM SPSS 22.
Supplementary material. All data is made publicly available within the Open Science Framework and accessible via the following link: https://osf.io/n3kvt/?view_only=36e58b631c6841dfac2dc1fd4017917d.
2.2. Results
Preliminary analyses indicated that the manipulation of the order of actions within co-actors’ sequences had no effect on movement preparation times. Therefore, we analyzed only MTs and asynchronies. MTs for short-long and long-short movement sequences did not differ in the individual baseline (t(19) = 0.62, p = .542, Cohen’s d = 0.14), making it unlikely that any of the effects reported below can be explained by the order of short and long movements in a sequence alone.3 We excluded the first and last trial in each block to avoid potential starting and stopping effects (see Smits-Engelsman, Van Galen, & Duysens, 2002). Trials in which participants covered less than 75% of the distance between start location and final target were also excluded from further analysis (0.2%). Finally, MTs exceeding two standard deviations around the mean were excluded per participant per block (3% for MT, 2.9% for MT1st) and asynchronies exceeding two standard deviations around the mean were excluded per pair per block (4.5%).
MT. Fig. 5 displays MTs in the ‘same order’ and in the ‘different order’ condition. Participants’ MTs were longer when the co-actor performed her actions in a different order (M = 657 ms, SD = 166 ms) compared to when the co-actor performed her actions in the same order (M = 632 ms, SD = 149 ms). A paired t-test showed that this difference was highly significant, t(19) = −4.94, p < .001, Cohen’s d = −1.11.
Fig. 5.
Movement times (i.e., times between movement onset at start location and movement offset at final target) for the ‘same order’ and the ‘different order’ condition for Experiments 1–3. The target configurations displayed on the bars serve only as exemplary representations of the two conditions. Participants’ movement times were significantly longer when the co-actor performed her actions in a different order. Error bars indicate Standard Errors. (*p < .05; **p < .01; ***p < .001).
MT1st. We tested whether the increase in participants’ overall movement times in the condition where the co-actor performed her movements in a different order depended on the type of movement (i.e., short or long) participants performed themselves. To this end, we analyzed only the time participants needed to move from the start location to the intermediate target (MT1st), to identify whether movement times increased differentially depending on whether participants’ own movement was short or long. We carried out a 2 × 2 ANOVA with the factors Own movement (short, long) and Co-actor order (same, different) for MT1st. This analysis was aimed at determining whether participants assimilated their own movement time to that of the co-actor by slowing down only when they performed a short movement while the co-actor performed a long movement but not when they performed a short movement while the co-actor performed a long movement.
There was a significant main effect of Own movement (F(1, 19) = 127.19, p < .001, ηp2 = .870), indicating that participants needed more time to complete the long compared to the short distance to the intermediate target. Consistent with the findings for overall MT, there was a significant main effect of Co-actor order (F(1, 19) = 9.55, p = .006, ηp2 = .335), showing that participants’ movement times to the intermediate target were longer when the co-actor performed her actions in a different order compared to when the co-actor performed her actions in the same order. Importantly, there was no significant interaction between Own movement and Co-actor order, F(1, 19) = 1.42, p = .248, ηp2 = .070. The absence of an interaction effect suggests that the increase in participants’ movement times did not depend on whether participants performed a short or a long movement themselves (increase for short: 6 ms; increase for long: 15 ms).
Variability of MT. To test the prediction that participants might strategically reduce the temporal variability of their performance to facilitate synchronous arrival at the final target (see Vesper et al., 2011, Vesper et al., 2016), we compared the standard deviations of MT in the ‘same order’ and the ‘different order’ condition. Variability was significantly higher in the ‘different order’ condition (t(19) = −2.44, p = .025, Cohen’s d = −0.54), suggesting that co-actors did not stabilize their movement times more strongly when performing actions in a different order.4 Rather, the longer movement times in the ‘different order’ condition presumably led to higher temporal variability – as variability typically increases the longer a movement lasts (see Bested et al., 2018) while faster movements tend to result in reduced variability (Vesper et al., 2011; Wagenmakers & Brown, 2007).
Asynchrony. Asynchronies in the ‘same order’ condition (M = 35 ms, SD = 7 ms) were numerically smaller than asynchronies in the ‘different order’ condition (M = 42 ms, SD = 14 ms) but the difference was not significant in a paired t-test, t(9) = −2.08, p = .067, Cohen’s d = −0.66.
2.3. Discussion
The results of Experiment 1 showed that co-actors instructed to synchronize the endpoints of two-segment action sequences moved more slowly when they performed their actions in a different order. This increase in movement times occurred not only on the level of the complete sequence but was also present when analyzing the first (and the second5) movement segment separately. The latter analysis indicated that participants’ movement times were longer whenever the co-actor’s action differed in terms of movement distance, regardless of whether participants performed a short or a long movement themselves. This suggests that participants did not assimilate to the co-actor’s movement time by slowing down only when they performed a short movement while the co-actor performed a long movement – a pattern of behavior that would be predicted by accounts of emergent coordination such as temporal entrainment (e.g., Schmidt and Richardson, 2008, Shockley et al., 2003; for a review, see Shockley & Riley, 2015) or temporal assimilation (e.g., Kelso et al., 1979, Kelso et al., 1983).
This finding provides first support for the hypothesis that co-actors represent the order of actions within each other’s action sequence and that this interferes with their own performance. However, it is also possible that observing a co-actor perform different movements might have interfered with actors’ own motor planning and execution (visuomotor interference: Kilner et al., 2003, Sacheli et al., 2012; automatic imitation: Heyes, 2011, Wang and Hamilton, 2012). Moreover, the perceptual content of the instruction screen might also have affected co-actors’ performance because they had continuous visual access to the screen where their own and their co-actor’s target configurations were displayed. Thus, the interference in the ‘different order’ condition might have been caused by the perceptual discrepancy between the two displayed target configurations.
3. Experiment 2
Experiment 2 was conducted to ensure that the slowdown in movement times observed in Experiment 1 was due to co-actors’ conflicting representations of action sequences rather than due to low-level perceptuomotor processes such as visuomotor interference (e.g., Kilner et al., 2003, Sacheli et al., 2012) or automatic imitation (Heyes, 2011, Wang and Hamilton, 2012). To this end, co-actors in Experiment 2 performed the joint task without seeing each other. To exclude the possibility that interference was caused by the different target configurations displayed on the instruction screen, no instructions were displayed during task performance. Rather, the target display was replaced by a neutral display that did not specify the intermediate target positions (Fig. 4A, row 2, panel 3). The same neutral display was used in the individual condition to exclude the possibility that displaying a second set of targets might affect performance.
If co-actors’ representations of each other’s action sequences cause interference, then performing different sequences should again take longer than performing the same sequences. However, if co-actors’ performance slowdown in Experiment 1 was due to visual influences, then no slowdown should occur in Experiment 2.
3.1. Method
The methods in Experiment 2 were the same as in Experiment 1, with the following exceptions:
Participants. Fourteen women and six men participated in randomly-matched pairs (5 only-female pairs, 1 only-male pair, Mage = 24.1 years, SDage = 4.33 years, range: 19–40).
Apparatus. In the joint condition, a partition (styrofoam material on wooden mount; height: 55 cm, width: 65 cm, depth: 0.5 cm) was placed on the table between participants so that they could no longer see each other. The partition was also present in the individual condition.
Procedure and stimuli. The display that informed participants about the current target configurations was only shown before participants started to perform the joint task in each block. Once participants were ready to start, the experimenter started the program and the required sequences for both co-actors were displayed on the screen, with the current intermediate target locations marked in orange (Fig. 4A, row 2, panels 1–2). After 3 s, the display turned ‘neutral’, i.e., the intermediate markers turned white (Fig. 4A, row 2, panel 3). About 2 s later, a start tone was played and the first trial began. In the individual condition, participants’ own target configuration was shown on one side of the screen and in addition, a neutral target configuration was shown on the other side (Fig. 4B, row 2). After 3 s, the display turned neutral and was the same as in the joint condition.
3.2. Results
Prior to analysis, we excluded all trials that did not meet a minimal horizontal distance criterion (0.2%). Values exceeding two standard deviations around the mean were excluded (2.9% for MT, 3.1% for MT1st, 3.5% for asynchrony).
Preliminary manipulation check. There was no indication that the presence of an additional target display in the individual baseline affected participants’ performance, as a comparison between individuals’ MTs from Experiment 1 and 2 was not significant (t(38) = 0.33, p = .744, Cohen’s d = 0.10).
MT. Fig. 5 displays MTs in the ‘same order’ and in the ‘different order’ condition. As in Experiment 1, participants’ MTs were significantly longer when the co-actor performed her actions in a different order (M = 632 ms, SD = 125 ms) compared to the same order (M = 617 ms, SD = 118 ms), t(19) = −3.15, p = .005, Cohen’s d = −0.70.
MT1st. As in Experiment 1, we performed a 2 × 2 ANOVA with the factors Own movement (short, long) and Co-actor order (same, different) for MT1st. The results were similar to Experiment 1, showing significant main effects of Own movement (F(1, 19) = 117.75, p < .001, ηp2 = .861) and of Co-actor order (F(1, 19) = 4.48, p = .048, ηp2 = .191), yet no significant interaction effect between the two factors (F(1, 19) = 0.27, p = .610, ηp2 = .014). This indicates that participants needed more time to complete the long compared to the short distance to the intermediate target and that participants’ movement times to the intermediate target were longer when the co-actor performed her actions in a different order compared to when the co-actor performed her actions in the same order. Importantly, the absence of an interaction effect suggests that the increase in movement times occurred irrespective of whether participants performed a short or a long movement themselves (increase for short: 8 ms; increase for long: 4 ms).
Asynchrony. Asynchronies in the ‘same order’ condition (M = 44 ms, SD = 12 ms) were significantly smaller than in the ‘different order’ condition (M = 53 ms, SD = 13 ms), t(9) = −3.85, p = .004, Cohen’s d = −1.23, indicating that co-actors were less well coordinated when they performed their actions in a different order. Based on previous literature showing that less variable temporal performance correlates with smaller asynchronies, we conducted a post-hoc t-test comparing the variability (in form of standard deviations) of MTs in the two conditions. The results showed that standard deviations were significantly higher in the ‘different order’ condition (Mdifferent = 38 ms) compared to the ‘same order’ condition (Msame = 33 ms), t(19) = −3.30, p = .004, Cohen’s d = −0.74.
3.3. Discussion
Experiment 2 provided evidence that the slowdown in co-actors’ performance was likely due to co-representing the order of each other’s actions within an action sequence. The results showed that co-actors moved more slowly when they performed actions in a different order even when they could not see each other moving. Hence, the slowdown cannot be explained in terms of visuomotor interference or automatic imitation. Moreover, the slowdown cannot be attributed to the display on the instruction screen because a neutral display was displayed while the task was performed. It is thus unlikely that visual access to a co-actor’s movements and/or to the target display contributed to the slowdown in Experiment 1. Moreover, the analysis of the first movement segment suggests that the overall increase in movement times did not depend on whether participants performed short or long movements themselves.
The fact that similar effects as in Experiment 1 were observed when co-actors could not see each other’s actions is crucial in that it distinguishes the present findings from earlier findings on automatic imitation of action sequences (e.g., Forbes and Hamilton, 2017, Pan and Hamilton, 2015; for a recent meta-analysis on automatic imitation, see Cracco et al., 2018). In the latter studies, it was tested whether participants’ own performance of an action sequence is affected by prior observation of a (virtual) actor performing the same or a different action sequence. For example, participants first watched an actor in a video perform three consecutive taps on three different drums; then participants tapped their own three drums either in the same or in a different order as the actor (Pan & Hamilton, 2015). In another study (Forbes & Hamilton, 2017), participants first observed an actor perform a series of three target-directed pointing movements in the presence or absence of obstacles between the targets. Afterwards, participants pointed to the same targets as the actor but there were no obstacles between the targets. Results showed that participants performed their actions faster when tapping the drums in the same order as the actor (Pan & Hamilton, 2015) and that they performed their own movements higher after having observed the actor move over obstacles compared to when there were no obstacles in the actor’s movement path (Forbes & Hamilton, 2017), respectively. These findings have been interpreted in the theoretical framework of automatic imitation which relies on the assumption that observing the movements of a human-like actor will prime the observer’s own motor system, resulting in, for example, faster performance for movements that are anatomically congruent to the observed movements, or in an assimilation of the observer’s movement trajectory to the observed trajectory. The mechanism underlying automatic imitation is typically explained in terms of the mirror neuron system in its function of providing a common underlying format for perception and action (e.g., Jeannerod, 2001, Kilner et al., 2003, Prinz, 1997; for a comprehensive review, see Rizzolatti & Sinigaglia, 2010). Importantly, for automatic imitation to occur, an action must be visually observed. In contrast, the theoretical framework of co-representation is built on the assumption that visual information is not necessary but solely the knowledge of another person’s task or action is sufficient to mentally represent that task or action (e.g., Sebanz, Knoblich, & Prinz, 2005; Sebanz, Bekkering, & Knoblich, 2006; van der Wel & Fu, 2015). In the present study, we aimed to investigate how simultaneously executed, yet unobservable, action sequences are co-represented, rather than how observed action sequences prime participants’ subsequent performance via the mechanism of automatic imitation (Pan & Hamilton, 2015).
In contrast to Experiment 1, co-actors coordinated their actions more successfully when the order of their actions was the same. This finding may reflect a regularity demonstrated in previous research: In the absence of perceptual feedback, less variable temporal performance facilitates coordination (Glover and Dixon, 2017, Vesper et al., 2011, Vesper et al., 2016, Vesper et al., 2013). Indeed, a post-hoc analysis showed that the variability of MTs was significantly higher when co-actors performed their actions in a different order, suggesting that the lower variability in the ‘same order’ condition may have facilitated coordination.
4. Experiment 3
In Experiments 1 and 2, the two co-actors were given the joint instruction to synchronously arrive at the final target location. Experiment 3 investigated whether having such a joint goal is a necessary precondition for co-actors to take each other’s action sequences into account. Previous research has indicated that people represent others’ actions even when coordination is not required (e.g., Böckler et al., 2012, Eskenazi et al., 2013, Sebanz et al., 2003, van der Wel and Fu, 2015). Thus, performing an action sequence alongside another person might be sufficient to trigger a representation of this person’s action sequence. To test this, participants in Experiment 3 performed the same task without being instructed to coordinate. If having a joint goal is not a necessary pre-condition for taking each other’s action sequences into account, co-actors should again move more slowly when performing their actions in a different order than when performing their actions in the same order.
4.1. Method
The methods in Experiment 3 were the same as in Experiment 2, with the following exceptions:
Participants. Fourteen women and six men participated in randomly-matched pairs (4 only-female pairs, Mage = 22.7 years, SDage = 3.89 years, range: 19–38). Members of one pair knew each other beforehand.
Procedure. Participants were instructed to perform their movements as fast as possible. The same instruction was given in the joint condition and in the individual baseline. After the experiment, participants were asked whether they thought they had acted differently in the joint condition compared to the individual baseline. This question served to assess whether participants had tried to synchronize with their partner even though not explicitly instructed to do so.
4.2. Results
Prior to analysis, we excluded all trials that failed to meet a minimal horizontal distance criterion (1%). Values exceeding two standard deviations around the mean were excluded (3% for MT, 2.8% for MT1st, 3.6% for asynchrony).
MT. Fig. 5 displays MTs in the ‘same order’ and in the ‘different order’ condition. As in the previous two experiments, participants’ MTs were significantly longer when the co-actor performed her actions in a different order (M = 580 ms, SD = 106 ms) compared to the same order (M = 573 ms, SD = 103 ms), t(19) = −2.34, p = .031, Cohen’s d = −0.52.
MT1st. As in the first two experiments, the analysis of MT1st showed that participants needed more time to complete the long compared to the short distance to the intermediate target, indicated by a significant main effect of Own movement, F(1, 19) = 520.97, p < .001, ηp2 = .965. In contrast to the previous experiments, there was no significant main effect of Co-actor order (F(1, 19) = 2.48, p = .132, ηp2 = .115), showing that participants’ movement times to the intermediate target were not markedly affected by the order of the co-actor’s movements. However, MT1st was numerically higher in the different order condition (increase of 3 ms for short and long). There was no significant interaction effect, F(1, 19) = 0.003, p = .957, ηp2 < .001.
Asynchrony. Asynchronies in the ‘same order’ condition (M = 83 ms, SD = 36 ms) did not differ from asynchronies in the ‘different order’ condition (M = 79 ms, SD = 28 ms), t(9) = −0.92, p = .382, Cohen’s d = 0.29.
4.3. Discussion
The results of Experiment 3 indicated that having a joint goal was not a necessary precondition for co-actors to represent each other’s action sequences. Acting alongside a co-actor was sufficient to trigger co-representation. Whereas this result is in line with earlier research on task co-representation (e.g., Sebanz et al., 2003, Sebanz et al., 2005), it is less consistent with recent evidence demonstrating that co-actors need to conceive of their actions as joint to take each other’s actions into account (della Gatta et al., 2017).
Co-actors in Experiment 3 tended to move faster compared to Experiment 2 (ME3 = 577 ms; ME2 = 625 ms). Most likely, the faster movement times can be attributed to the absence of a coordination constraint. Not being required to coordinate, co-actors shifted – intentionally or unintentionally – towards a form of competitive behavior where they tried to beat each other to the target. Indeed, 12 out of 20 participants reported that they moved faster in the joint condition because they were affected by the sound of their task partner’s target hits, which motivated them to reach their own target at least as fast or faster than the other. Five of these participants mentioned explicitly that they experienced the task as an “undeliberate competition”. Thus, interference from a co-represented action sequence seems to even occur under conditions that are perceived to involve competition. This is in line with evidence for co-representation of a task partner’s actions in a competitive context (Ruys & Aarts, 2010).
In Experiment 3, asynchronies were distinctly higher (MAsyncE3 = 81 ms) than in the previous two experiments (MAsyncE1 = 39 ms; MAsyncE2 = 49 ms). This is expected given that co-actors were not instructed to synchronize their landing times in Experiment 3.
5. Experiment 4
Experiment 4 tested whether interference from co-represented action sequences is specific to differences in movement distance or whether it is a more general phenomenon. If the interference reflects a more general tendency to plan and execute own action sequences while additionally specifying parameters of a co-actor’s action sequence, interference should also occur when another’s movements differ in other parameters that affect movement difficulty. We tested this by varying target sizes, a parameter that is known to systematically affect movement difficulty (Fitts, 1954).
5.1. Method
The methods in Experiment 4 were the same as in Experiment 3, with the following exceptions:
Participants. Thirteen women and seven men participated in randomly-matched pairs (4 only-female pairs, 1 only-male pair, Mage = 21.5 years, SDage = 1.72 years, range: 19–26). Members of one pair knew each other beforehand.
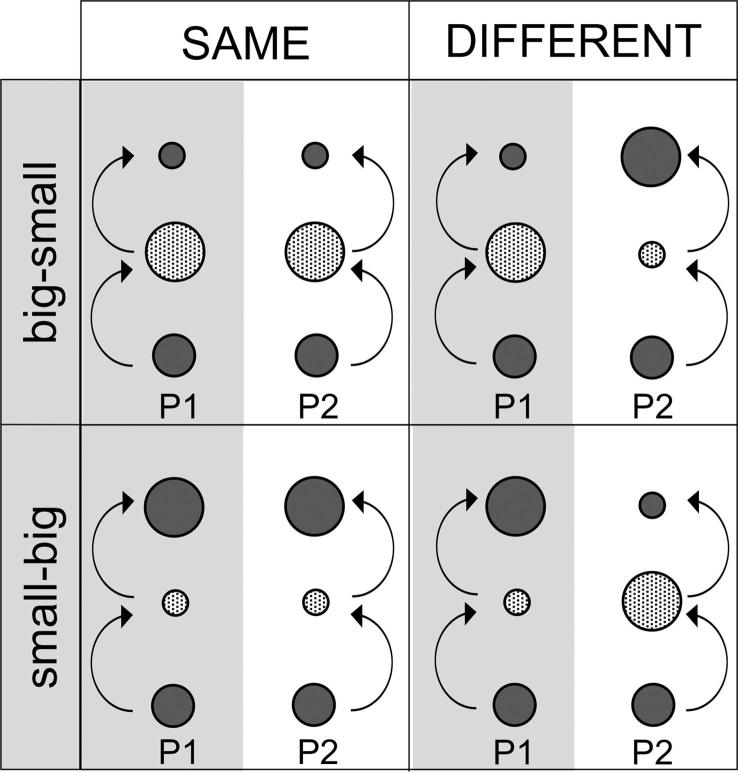
Apparatus. The intermediate target was located exactly half-way between start location and final target, dividing the total distance into two equal distances of ∼20 cm, see Fig. 6. Whereas the start marker had a diameter of 5 cm as in previous experiments, the intermediate and the final markers differed in size, with diameters of 3.8 cm and 7.6 cm, respectively. By selecting these sizes, we matched movement difficulty in Experiment 1–4 in terms of their index of difficulty (ID = log2(2 × distance/width)) as defined by Fitts’ law (1954). In Experiment 4, big targets (corresponding to short movements in Experiment 1–3) had an ID of 2.4 and small targets (corresponding to long movements in Experiment 1–3) had an ID of 3.4.
Fig. 6.
Four types of blocks in Experiment 4. Participants performed sequences of two movements to targets of different sizes, in the order ‘big-small’ or ‘small-big’. The two participants in a pair either performed the same sequence or a different sequence. Note that in the ‘different order’ condition (right column) the labels ‘big-small’/‘small-big’ apply to the order for participant P1 only.
Procedure and stimuli. Participants were instructed to synchronize their landing times on the final target and to perform their movements as fast as possible.
Across blocks, it was varied whether the sizes of the intermediate and final target were the same or different for the two participants. Before each block, the experimenter arranged the small and big target markers on the table and the corresponding target configuration was displayed on the instruction screen. The display showed the intermediate and final markers pictured as a small circle within a bigger circle, outlined in black. In blocks of the ‘same order’ condition (Fig. 4A, row 3, panel 1), the intermediate markers were colored such that the entire big circles were orange whereas for the final markers only the small inner circles were orange, or vice versa. This served to instruct the two participants to either both perform a movement to a big target followed by a movement to a small target (‘big-small’) or a movement to a small target followed by a movement to a big target (‘small-big’), see Fig. 6. In blocks of the ‘different order’ condition, the circles in the two columns were colored differently such that for the intermediate marker in one column the entire big circle was orange whereas for the intermediate marker in the other column only the small inner circle was orange, and vice versa for the final markers (Fig. 4A, row 3, panel 2). This instructed the two participants to perform different action sequences so that one participant performed a movement to a big target followed by a movement to a small target whereas the other participant performed a movement to a small target followed by a movement to a big target (Fig. 6). After 3 s, both intermediate and final target circles on the instruction screen turned white such that only the black outlines of the small circles within the big circles were displayed while co-actors performed the task (Fig. 4A, row 3, panel 3).
Before performing the joint task, participants completed two blocks of individual baseline trials. Participants performed one block of big-small sequences and one block of small-big sequences.
5.2. Results
Prior to analysis, we checked whether any trials failed to meet a minimal horizontal distance criterion but no trials had to be excluded. Values exceeding two standard deviations around the mean were excluded (3.2% for MT, 2.9% for MT1st, 4.1% for asynchrony).
MT. Fig. 7 displays MTs in the ‘same order’ and in the ‘different order’ condition. As in the previous experiments, participants’ MTs were significantly longer when the co-actor performed her actions in a different order (M = 683 ms, SD = 151 ms) compared to the same order (M = 669 ms, SD = 151 ms), t(19) = −3.72, p = .001, Cohen’s d = −0.83.
Fig. 7.
Movement times for the ‘same order’ and the ‘different order’ condition for Experiments 4–6 (where ‘same’/‘different’ corresponds to ‘4 colors’/‘2 colors’ in Exp. 6). In all experiments, participants' movement times were significantly longer when the co-actor performed her actions in a different order. Error bars indicate Standard Errors. (**p < .01; ***p < .001).
MT1st. We performed a 2 × 2 ANOVA with the factors Own movement (big, small) and Co-actor order (same, different) for MT1st. There was a significant main effect of Co-actor order (F(1, 19) = 4.68, p = .043, ηp2 = .198), indicating that participants’ movement times to the intermediate target were longer when the co-actor performed her actions in a different order compared to when the co-actor performed her actions in the same order. Unexpectedly, the main effect of Own movement was not significant (F(1, 19) = 1.87, p = .187, ηp2 = .090), showing that participants’ movement times did not differ as a function of target size.6 A significant interaction effect between Co-actor order and Own movement (F(1, 19) = 17.27, p < .001, ηp2 = .476) showed that the increase in MT1st occurred only when participants moved to a small target (increase for big target: −5 ms; increase for small target: 18 ms).
Asynchrony. Asynchronies in the ‘same order’ condition (M = 40 ms, SD = 14 ms) and the ‘different order’ condition (M = 41 ms, SD = 13 ms) did not differ significantly, t(9) = −0.26, p = .803, Cohen’s d = −0.08, indicating that co-actors were equally well coordinated regardless of whether the order of their actions differed.
5.3. Discussion
Experiment 4 showed that participants moved more slowly when a co-actor performed movements to targets that differed in size from their own targets. This finding generalizes the results from Experiments 1–3 by demonstrating that the previously observed interference is not specific to movement distance but more generally related to movement difficulty. Thus, the results from Experiments 1–4 suggest that the interference occurred at a motor level where concurrent specification of a co-actor’s movement parameters led to a slowdown in specifying own movement parameters.
The target size manipulation in Experiment 4 improved the current task design in one important aspect. In Experiments 1–3, the target layout on the table consisted of two intermediate targets, yet only one of these targets was task-relevant in a given block of trials. Thus, actors had to actively remember and select the target that was currently task-relevant and inhibit a response to the other one, which may have implied an additional cognitive cost. In Experiment 4, however, only one intermediate target was present so that the possibility of such additional costs was excluded.
Participants’ explicit reports indicate that most of them were not aware that the target size manipulation affected their behavior. Only a single participant reported that she had moved more slowly when the order of her partner’s target sizes had differed from her own order.
6. Experiment 5
Experiment 5 aimed to test whether interference from co-representing another’s action sequence can also arise at the cognitive level of goal selection. Research on bimanual control has established that bimanual interference during reaching movements is not exclusively related to the specification of movement parameters but may also emerge due to processes related to selecting and specifying target locations (e.g., see Diedrichsen et al., 2006). It has been shown that it is easier to coordinate two hands when the final goal states of the required movements are the same, e.g., when target locations are specified by the same cues (Diedrichsen et al., 2001, Diedrichsen et al., 2006, Hazeltine et al., 2003, Weigelt et al., 2007), when targets share perceptual characteristics such as color (Diedrichsen et al., 2003), or when the instructed final orientation of two manipulated objects is the same (Kunde & Weigelt, 2005). Bimanual costs related to goal-selection can be substantially larger than those related to motor programming (Diedrichsen et al., 2006), and benefits during goal-selection may even cancel out movement-related costs (Diedrichsen et al., 2003, Weigelt et al., 2007).
Based on these findings from the bimanual domain, we predicted that interference might occur when the action sequences performed by two co-actors imply selecting targets with different perceptual characteristics. To test this prediction, we removed any differences in movement parameters so that co-actors performed two sequential movements of equal movement difficulty. The difficulty of the goal selection process was manipulated by varying the color of the movement targets in the action sequences of the two co-actors. Co-actors either moved to targets of the same color or to targets of different colors, implying same or different goal states. If interference arises due to the difficulty of selecting between different goal states, a slowdown in performance should occur when co-actors move to targets of different colors.
6.1. Method
The methods in Experiment 5 were the same as in Experiment 4, with the following exceptions:
Participants. Fourteen women and six men participated in randomly-matched pairs (5 only-female pairs, 1 only-male pair, Mage = 22.6 years, SDage = 2.50 years, range: 19–29).
Apparatus. Intermediate and final markers both had a diameter of 5.4 cm (see Fig. 8). This size was chosen to create an ID of 2.9 for both movements, representing the average of the two IDs (2.4 and 3.4) used in the previous experiments. The intermediate and the final markers were yellow and brown, respectively.
Fig. 8.
Four types of blocks in Experiment 5. Participants performed sequences of two movements to targets of different colors, in the order ‘yellow-brown’ or ‘brown-yellow’. The two participants in a pair either performed the same sequence or a different sequence. Note that in the ‘different order’ condition (right column) the labels ‘yellow-brown’/‘brown-yellow’ apply to the order for participant P1 only.
Procedure and stimuli. Across blocks, it was varied whether the colors of the intermediate and final target were the same or different for the two participants. Before each block, the experimenter arranged the yellow and brown target markers on the table and the corresponding target configuration was displayed on the instruction screen.
In blocks of the ‘same order’ condition, the intermediate and final markers in the two columns on the instruction screen were of the same color (Fig. 4A, row 4, panel 1). Thus, the two participants both performed a movement to a yellow target followed by a movement to a brown target (‘yellow-brown’) or a movement to a brown target followed by a movement to a yellow target (‘brown-yellow’), see Fig. 8. In blocks of the ‘different order’ condition, the intermediate and final markers in the two columns differed in color (Fig. 4A, row 4, panel 2). Thus, the two participants performed different action sequences so that one participant performed a movement to a yellow target followed by a movement to a brown target whereas the other participant performed a movement to a brown target followed by a movement to a yellow target (Fig. 8).
Before performing the joint task, participants completed two blocks of individual baseline trials. Participants performed one block of yellow-brown sequences and one block of brown-yellow sequences. At the end of the experiment, participants were asked whether they had noticed any difference in their performance with respect to the order of their own and their partner’s target colors.
6.2. Results
Prior to analysis, we excluded all trials that failed to meet a minimal horizontal distance criterion (0.2%). Values exceeding two standard deviations around the mean were excluded (2.3% for MT, 2.9% for MT1st, 4.4% for asynchrony).
MT. Fig. 7 displays MTs in the ‘same order’ and in the ‘different order’ condition. Participants’ MTs were significantly longer when the order of the co-actor’s target colors was different (M = 689 ms, SD = 106 ms) than when it was the same (M = 674 ms, SD = 107 ms), t(19) = −4.13, p < .001, Cohen’s d = −0.92.
MT1st. We performed a 2 × 2 ANOVA with the factors Own target color (yellow, brown) and Co-actor order (same, different) for MT1st. Consistent with the results for overall MT, participants’ movement times to the intermediate target were significantly longer when the order of the co-actor’s target colors was different than when it was the same, F(1, 19) = 12.16, p = .002, ηp2 = .390 (Mdiff = 8 ms). There was no significant main effect of Own target color (F(1, 19) = 0.01, p = .920, ηp2 = .001) and no significant interaction effect (F(1, 19) = 0.30, p = .590, ηp2 = .016).
Asynchrony. A comparison between asynchronies from the ‘same order’ and the ‘different order’ condition revealed no significant differences, t(9) = −1.34, p = .215, Cohen’s d = −0.42, indicating that co-actors coordinated their movements equally well regardless of whether the order of target colors was the same (M = 37 ms, SD = 9 ms) or different (M = 39 ms, SD = 6 ms).
6.3. Discussion
Experiment 5 showed that participants moved more slowly when a co-actor performed movements to targets that differed in color from their own targets. This finding demonstrates that interference from co-representing another’s action sequence can also arise at the cognitive level of goal selection. As the action sequences performed by two co-actors did not differ in terms of movement parameters, interference cannot be attributed to difficulties with motor programming but must have emerged due to the difficulty of selecting goal states that differ from a co-actor’s goal states. Similar costs during goal selection have been observed for bimanual performance (Diedrichsen et al., 2003). It is noteworthy that according to their explicit reports, none of the participants in the present experiment was aware that their performance was affected by their partner’s target colors.
7. Experiment 6
The aim of our final experiment was to disentangle two hypotheses about how co-representing another’s action sequence causes interference at the level of goal selection. One possibility is that unspecific interference occurs whenever the goal states of two co-actors’ individual actions (in an action sequence) differ in their perceptual characteristics. Another possibility is that specific interference occurs if task-relevant characteristics overlap between the goal states of two co-actors’ individual actions (cf. Diedrichsen et al., 2003, Experiment 3) such as when they produce actions with the same goal states in a different order. Here, interference would arise because a co-represented action sequence specifies the same goal states as one’s own action sequence while the order of these states differs. In this case, actors might experience confusion because the characteristics of the co-actor’s goal states are highly self-relevant (e.g., targets of the same color as their own targets) yet reverse-ordered and thus in conflict with their own goal state representation. That is, the specific relation between co-actors’ goal states, e.g., the fact that one actor’s intermediate target has the same characteristics as the co-actor’s final target (and vice versa), would lead to interference.
To test whether interference is specific, we compared a condition where co-actors had two different sets of target colors and moved to targets of different color (no overlap in task-relevant perceptual characteristics) to a condition where co-actors had the same set of target colors and moved to targets of different color (overlap in task-relevant perceptual characteristics, same as the ‘different’ condition in Experiment 5). If interference is specific, a slowdown in movement times is only expected in the latter but not in the former condition.
7.1. Method
The methods in Experiment 6 were the same as in Experiment 5, with the following exceptions:
Participants. Fifteen women and five men participated in randomly-matched pairs (6 only-female pairs, 1 only-male pair, Mage = 21.3 years, SDage = 1.73 years, range: 18–24). Participants were asked whether they could clearly distinguish all target colors and all participants answered this question affirmatively. However, after the experiment one participant reported a strong preference for a specific color combination. This participant and her task partner were replaced by a new pair of participants.
Apparatus. The movement distances and target sizes were identical to Experiment 5, yet the colors of the targets differed. The color of the start marker was grey. The colors of the intermediate and final markers varied between conditions; they could be either yellow, brown, blue, pink, green, or purple (see Fig. 9 and below for details).
Fig. 9.
Four types of blocks in Experiment 6. Participants performed sequences of two movements to targets of different colors. In blocks of the ‘4 colors’ condition (left column), the target colors were different for the two participants in a pair such that P1’s colors were pink and blue while P2’s colors were green and purple. In blocks of the ‘2 colors’ condition (right column), the two participants shared the same set of brown and yellow targets but the order in which the colors occurred was different (e.g., P1: brown-yellow, P2: yellow-brown).
Procedure and stimuli. The design of Experiment 6 differed from the previous experiments as we did not compare a ‘same order’ to a ‘different order’ condition. Rather, we reran the ‘different order’ condition from Experiment 5 where the order of the two participants’ target colors differed (‘yellow-brown’ vs. ‘brown-yellow’) and compared it to a condition where the two participants’ targets had four entirely different colors. We refer to the former as the ‘2 colors’ condition and to the latter as the ‘4 colors’ condition. Before each block, the experimenter arranged the differently colored markers on the table and the corresponding target configuration was displayed on the instruction screen.
In blocks of the ‘4 colors’ condition, Participant 1 performed a movement to a pink target followed by a movement to a blue target whereas Participant 2 performed a movement to a green target followed by a movement to a purple target (P1: pink-blue, P2: green-purple), or Participant 1 performed a movement to a blue target followed by a movement to a pink target while Participant 2 performed a movement to a purple target followed by a movement to a green target (P1: blue-pink, P2: purple-green), see Fig. 9. In blocks of the ‘2 colors’ condition, one participant performed a movement to a yellow target followed by a movement to a brown target while the other performed a movement to a brown target followed by a movement to a yellow target (see Fig. 9).
Before performing the joint task, participants completed four blocks of individual baseline trials. Participants performed one block of yellow-brown sequences, one block of brown-yellow sequences, one block of pink-blue/green-purple sequences, and one block of blue-pink/purple-green sequences.
7.2. Results
Prior to analysis, we excluded all trials that failed to meet a minimal horizontal distance criterion (0.1%). Values exceeding two standard deviations around the mean were excluded (3.1% for MT, 3.1% for MT1st, 3.8% for asynchrony).
MT. Fig. 7 displays MTs in the ‘4 colors’ and in the ‘2 colors’ condition. Participants’ MTs were significantly longer when the co-actor’s movement targets had the same two colors arranged in different order (M = 614 ms, SD = 112 ms) than when the co-actor’s movement targets had entirely different colors (M = 605 ms, SD = 114 ms), t(19) = −3.24, p = .004, Cohen’s d = −0.72.
MT1st. We performed a 2 × 2 ANOVA with the factors Own target color (yellow/pink/green, brown/blue/purple) and Co-actor order (same, different) for MT1st, where ‘same order’ refers, for instance, to blocks where participant’s own order was pink-blue and the co-actor’s order was green-purple while ‘different order’ refers to blocks where participant’s own order was yellow-brown and the co-actor’s order was brown-yellow (see details above). Participants’ movement times to the intermediate target were not significantly longer when the order of the co-actor’s target colors was different than when it was the same, F(1, 19) = 3.53, p = .076, ηp2 = .157, yet there was a numerical difference of ∼4 ms. There was no significant main effect of Own target color (F(1, 19) = 0.10, p = .759, ηp2 = .005) and no significant interaction effect (F(1, 19) = 0.29, p = .599, ηp2 = .015).
Asynchrony. A comparison between asynchronies from the ‘2 colors’ and the ‘4 colors’ condition revealed no significant effects, t(9) = −1.13, p = .289, Cohen’s d = −0.36, indicating that co-actors coordinated their movements equally well irrespective of whether they shared the same set of target colors (M = 46 ms, SD = 17 ms) or not (M = 44 ms, SD = 17 ms).
7.3. Discussion
The findings of Experiment 6 supported the hypothesis that interference from representing a co-actor’s action sequence is specific and occurs only if one’s own action sequence and another’s action sequence overlap in the perceptual characteristics of action goal states. Movement times were slowed down when co-actors moved to targets that shared the same color set compared to when they moved to targets whose colors were from entirely different color sets.
These results do not fully rule out the theoretical possibility that additional unspecific interference may occur when two co-actors perform action sequences that specify non-overlapping goal states, as in the ‘4 colors’ condition in Experiment 6.
8. General discussion
The aim of the present study was to investigate whether co-actors have a tendency to represent each other’s action sequences, even if doing so is not necessary for joint task performance. To this end, we designed a novel joint movement task where two co-actors performed sequences of goal-directed actions. Each sequence consisted of two individual actions that differed in terms of the difficulty of the movements to be performed (Experiments 1–4) or in terms of the perceptual characteristics of the action goal states (Experiments 5–6). Supporting our prediction that co-actors represent each other’s action sequences, the results of Experiments 1–5 consistently showed that co-actors moved more slowly when performing the same actions in a different order compared to performing the same actions in the same order.
Importantly, this slowdown cannot be attributed to visuomotor processes (e.g., Heyes, 2011, Kilner et al., 2003, Sacheli et al., 2012, Wang and Hamilton, 2012) but must be a result of internal representations because it also occurred when co-actors could not observe each other’s movements (Experiments 2–6). The present finding adds to previous findings on automatic imitation of action sequences (e.g., Forbes and Hamilton, 2017, Pan and Hamilton, 2015) in at least two important ways: Pan & Hamilton (2015) have shown that people respond faster when they perform an action sequence that is congruent with a previously observed action sequence than when it is incongruent with the observed sequence, providing evidence for automatic imitation of action sequences. In contrast, the present study went beyond visually induced motor interference effects, showing that co-actors co-represent each other’s action sequences (1) when direct visual input is absent and (2) when they perform their actions concurrently and with a joint goal rather than acting sequentially in response to a virtual actor on a video screen. Thus, whereas Pan and Hamilton (2015) have already shown that people have a tendency to automatically imitate another’s observed action sequence, we show that simultaneously executed, yet unobservable, movement sequences are co-represented and how this, in turn, affects co-actors’ own performance.
In line with previous research on co-representation (e.g., Sebanz et al., 2003, van der Wel and Fu, 2015), we found that a joint coordination goal was not a prerequisite for co-actors to represent each other’s action sequences as a slowdown was also observed when co-actors did not have the joint goal of synchronizing arrival times at the final location (Experiment 3). In line with findings from research on the coordination of bimanual movements (e.g., Diedrichsen et al., 2006), we found that interference can arise due to differences in the difficulty of the individual movements that two co-actors perform as part of an action sequence (Experiments 1–4) as well as due to differences in the perceptual characteristics of the goal states two co-actors’ actions are directed at (Experiments 5–6).
The fact that participants’ own actions were affected by the order of their co-actor’s actions suggests that participants co-represented the order of their co-actor’s actions in some form – whether in form of two separate movement segments or as one complex movement with two pre-planned stages (see Adam et al., 2000) remains an open question that cannot be answered based on the present set of experiments. However, what seems clear is that participants did take into account that their co-actor’s action sequence consisted of two ordered segments, and that they did not merely represent the sequence as a whole without distinguishing between the first and the second segment.
The results of Experiment 6 indicate that interference at the level of goal selection occurs if one’s own action sequence and another’s action sequence overlap in the perceptual characteristics of goal states. More generally, this suggests that interference due to co-representing another’s action sequence is restricted to situations where co-actors perform the same actions in a different order and does not occur (or at least to a smaller extent) in situations where co-actors perform entirely different actions.
At present, it remains an open question whether interference in all of our six experiments was caused at the level of representing action goal states. This assumption could explain the differences in movement times observed in Experiments 1–4 because participants may have represented the action sequences as “far target – close target” or “small target – big target” at the level of the individual actions’ goal states. Alternatively, interference in Experiments 1–4 may have occurred at the level of specifying movement parameters, as suggested by previous findings on bimanual control (e.g., Heuer, 1993, Heuer et al., 1998, Spijkers and Heuer, 1995, Spijkers et al., 1997). Thus, based on the present data, we can say with confidence that in two experiments (Exp. 5–6) interference arose at the goal level. The open question to be addressed in future research is whether in the other experiments there was interference at the motor level, or possibly at both levels.
Manipulating movement difficulty and perceptual characteristics of action goal states within the same experiment may help to answer this question. For example, building on the present experimental setup, one could design an experiment with (1) a baseline condition where movement difficulty and action goal states are the same (e.g., same target distances; same color for all targets), (2) a condition where movement difficulty is manipulated while action goal states are the same (e.g., different target distances as in Experiments 1–3; same color for all targets), (3) a condition where action goal states are manipulated while movement difficulty is the same (same target distances; different target colors as in Experiment 5), and (4) a condition where movement difficulty and action goal states are manipulated. Comparing the effects of movement times across these conditions should allow one to determine whether the two factors (movement difficulty and action goal states) separately lead to effects of similar size, and whether these effects are additive (in condition 4). Still, the possibility remains that a movement parameter (e.g., a long distance) could be represented in terms of the respective goal state (e.g., a far target). A crucial step for future research would be to experimentally disentangle these alternatives, if at all possible.
An important issue of interpretation concerns the directionality of the observed effect. The question is whether the difference in movement times should be interpreted as interference (i.e., increased movement times in the ‘different order’ condition) or as facilitation (i.e., decreased movement times in the ‘same order’ condition)? Strictly speaking, one would need to compare movement times in the ‘same order’ and the ‘different order’ condition to a neutral baseline to determine the directionality of the effect. In this neutral baseline, co-actors should neither perform the same nor different actions. Such a baseline was not included in the present design, and it is generally difficult to conceive of. Given this lack of a proper joint baseline comparison, we instead compared performance in the joint conditions to performance in the individual baseline condition. If movement times in the joint ‘same order’ condition were faster than in the individual baseline, this would support a facilitation account. If movement times in the joint ‘different order’ condition were slower than in the individual baseline, this would suggest interference. However, the results7 of the baseline-comparison were not consistent across experiments: In Exp. 1, 2, and 6, MTs in the joint ‘same order’ condition were significantly shorter than in the individual baseline while there was no difference between the joint ‘different order’ condition and the individual baseline. In Exp. 3, MTs in both joint conditions were significantly shorter than in the individual baseline. In Exp. 4 and 5, there were no significant differences between MTs in the individual baseline and the joint conditions. Thus, no clear conclusions about the directionality of the effect can be drawn from these data. Moreover, shorter movement times in the joint condition are generally hard to interpret because people have been shown to often speed up their actions to support coordination (e.g., Vesper et al., 2011, Vesper et al., 2016).
From a theoretical point of view and based on previous literature, we suggest that the present data are more consistent with an interference account than with a facilitation account. This is because first of all, the rationale of the present study was based on the fact that there are similarities between inter- and intrapersonal coordination (e.g., Fine and Amazeen, 2011, Richardson et al., 2007, Schmidt et al., 1998) and that therefore the effects observed in bimanual motor control, often referred to as intermanual “interference” or “constraints”, (e.g., Diedrichsen et al., 2001, Diedrichsen et al., 2003, Kelso et al., 1979) might translate to interpersonal coordination. Secondly, the study relied on previous literature on co-representation which consistently interpreted similar effects (i.e., participants’ temporal or spatial deviations whose occurrence was ascribed to a co-actor’s incongruent task or action) in an interference framework (e.g., Kuhlen and Abdel Rahman, 2017, Schmitz et al., 2017, Sebanz et al., 2003, van der Wel and Fu, 2015).
Furthermore, there is some indication of interference in the present data. Specifically, we found longer movement times in the ‘different order’ condition in Experiment 6 (2 colors; different colors for spatially aligned targets) compared to what one may call a relatively ‘neutral’ condition (4 colors), suggesting that performing the same actions in a different order than a co-actor interferes with performance compared to performing non-related actions. However, Experiment 6 is crucially lacking a ‘same order’ condition (2 colors; same colors for spatially aligned targets). Only a comparison between ‘neutral’ and ‘same order’ conditions could show whether movement times in ‘same’ might be even shorter than in ‘neutral’, indicating a facilitatory effect. Thus, to sum up, we suggest that an interpretation of the observed effect in terms of interference is more in line with theoretical assumptions and prior literature, yet we cannot fully rule out the theoretical possibility that some or all of the effect is due to facilitation. A combination of interference and facilitation is also possible.
It is important to consider potential alternative explanations for the effects observed in the present study. Given that co-actors received visual feedback (Experiment 1) and auditory feedback about each other’s actions, they may have unintentionally entrained with one another. Entrainment is a form of emergent coordination based on low-level perception-action links that can be described as a coupling of rhythmic oscillators and is observed in mechanical as well as in biological systems (e.g., Schmidt and Richardson, 2008, Shockley et al., 2003, Shockley and Riley, 2015). Co-actors in the present experiments may have become coupled simply because they could see each other’s movements (Experiment 1) or hear each other landing on the targets. A temporal coupling would imply that co-actors assimilate their movement times to each other such that the actor with the longer distance speeds up her movement or the actor with the shorter distance slows down her movement such that they arrive synchronously at the intermediate target. However, our analyses of the first movement segment indicate that participants did not assimilate their movement times to each other. Rather, we found a general increase in movement times (for the first movement segment, the second movement segment, and the complete sequence) whenever co-actors performed their actions in a different order. For example, when P1 performed a short movement while P2 performed a long movement (or vice versa), movement times of both participants were longer compared to when they performed the same movements.
Still, one might object that a more general form of audiomotor interference might have occurred. In fact, there is evidence indicating that humans possess not only a visual but also an auditory mirror neuron system which responds to the sounds of actions (Gazzola, Aziz-Zadeh, & Keysers, 2006). Thus, hearing the co-actor land on her targets might have interfered, via auditory mirror neurons, with participants’ own action execution. However, we believe that this explanation cannot fully account for participants’ pattern of behavior. To see this, consider the behavior of P1 in the following case: P1’s first movement is short and P2’s first movement is long (Experiments 1–3). We found longer movement times for P1 (and also for P2) in this case compared to the case where P2’s first movement is also short. This increase in P1’s movement time cannot be directly caused by P1 hearing P2 land on the first target because P1 will land on her own target before P2 will land on her target which is further away. Thus, the slowdown in P1’s movement time must be of a predictive (rather than reactive) nature: Simply knowing that P2 will perform a longer movement affects P1’s own movement time. This suggests that P1 must hold a representation of P2’s action order – and that it is this representation, rather than the direct auditory feedback from P2’s actions, that interferes with P1’s performance.
Importantly, both alternative explanations advanced above, i.e., temporal entrainment and more general audiomotor interference, cannot account for the results from Experiments 5 and 6. As co-actors’ movement difficulties (target IDs) did not differ in these experiments, co-actors naturally tended to land on the two targets roughly at the same time. Hence, there was neither a need for co-actors to assimilate to each other’s movement times (temporal entrainment) nor an obvious way of how hearing each other land on the targets should have affected own movement times (audiomotor interference). Thus, the observed increase in movement times in the last two experiments cannot be attributed to temporal entrainment or to audiomotor interference.
Previous studies have demonstrated that the tendency to perform in a coordinated manner emerges not only between biomechanically coupled actors such as a person’s limbs but also between actors that lack a centralized controller such as the limbs of two different individuals (e.g., Richardson et al., 2007, Schmidt et al., 1990, Schmidt and Turvey, 1994). For example, in a task where two actors (two hands belonging to either one or to two individuals) moved to targets of unequal target ID, the movement time of the actor with the easier target ID increased to assimilate to the movement time of the actor with the more difficult target ID (Fine and Amazeen, 2011, Fowler et al., 1991, Kelso et al., 1979, Marteniuk et al., 1984). In contrast, in the present study, we did not observe an assimilation of co-actors’ movement times (e.g., an increase in MT for the short distance assimilating to the MT for the long distance) but we observed generally longer movement times whenever co-actors performed actions in different order. These findings do not speak so much to the processes of emergent coordination between individuals but rather provide insights into how the compatibility of simultaneously executed movement sequences is co-represented and how this affects co-actors’ individual performances.
Regarding the effects of representing a co-actor’s action order, it is interesting to consider a recent finding by Gambi, Van de Cavey, and Pickering (2015). Using a joint picture naming task, these authors found that participants took longer to name pictures when they believed that their task partner was also naming pictures (Gambi et al., 2015). Specifically, when participants named two pictures, their naming latencies increased when they believed that their task partner was naming the same pictures in the reverse order. Whereas this result seems to perfectly correspond to the findings of the present study, Gambi et al. (2015) found a similar increase in naming latencies when participants believed that their task partner was naming the pictures in the same order. The authors suggest that speakers use their own language production system to represent their task partner’s linguistic intention (but not the content of their utterance), such that concurrent language production is slowed down (Gambi et al., 2015). The difference in the results pattern between Gambi and colleagues’ study (2015) and the present study indicates that there may be a systematic difference in how task partners represent linguistic sequences and action sequences. Thoroughly investigating such differences in future studies seems worthwhile.
The present findings raise several further questions for future research. One question is whether interference from co-representing another’s action sequence occurs exclusively when actions are performed concurrently or whether similar interference occurs when co-actors take turns in performing their actions. A recent study on picture naming (Gambi et al., 2017) found an increase in participants’ speech length when a co-actor concurrently described a picture in a different voice (active vs. passive) compared to the same voice – yet there was no difference when participant and co-actor spoke consecutively. However, Diedrichsen et al. observed interference in a bimanual task in which participants used their left and right hand in alternation (Diedrichsen et al., 2003, Experiment 4) suggesting that interference from co-representing another’s action sequence may also occur in tasks where co-actors take turns. A similar prediction could also be derived from earlier studies on co-representation where participants took turns in performing actions defined by different task rules (Sebanz et al., 2003).
Another interesting question is whether the presence of a co-actor is necessary or whether the belief that another person is acting in another room is sufficient to trigger co-representation of action sequences. Previous research (e.g., Atmaca et al., 2011, Tsai et al., 2008, Vlainic et al., 2010; but see Welsh, Higgins, Ray, & Weeks, 2007) suggests that the mere belief might be sufficient. Follow-ups on the present study could address the question of whether such beliefs have differential effects on the co-representation of action sequences at the level of specifying action goal states and at the level of specifying movement parameters.
A further important question concerns the social nature of the observed effects. Would participants be affected in similar ways when performing their actions next to a non-human (e.g., robotic) co-actor instead of a human co-actor? The present experiments cannot rule out this possibility. However, the possibility that similar effects might occur in a non-social context does not challenge the present finding that two human co-actors co-represent each other’s action sequences. Even if non-social interference effects were observed in a future study, it would not necessarily follow that the effects in the present study are also of a non-social nature, because the possibility remains that different mechanisms might be at play in social and non-social contexts.
The findings from the present study can be applied to a number of everyday life situations. For instance, consider members of a choir singing a round: They sing the same voice but begin and end at different times, such as in the famous French nursery rhyme ‘Frère Jacques’. Interference might occur because singers concurrently perform the same actions in a different order, similar to participants in the present study. Based on the results from Experiment 6, one should predict that no (or less) interference will occur if people sing different, independent voices at the same time. Similar interference might occur in activities such as ballroom dance or in other games or sports activities that involve sequences of actions. It would be interesting to investigate how experts at joint action such as professional musicians or dancers (learn to) avoid or overcome such interference. First insights into this question have been provided by recent studies showing that practicing a task while receiving visual feedback of one’s own actions leads to a better distinction between self- and other-generated actions. This enhanced ability in self-other distinction makes people less susceptible to movement interference from others’ actions (Roberts et al., 2016, Roberts et al., 2018).
Finally, further studies are needed to address the generality of the observed interference from co-representing another’s action sequence. Would introducing more extreme differences between co-actors’ movement difficulty lead to a proportional increase in interference? Would increasing the number of individual actions in a sequence increase or reduce interference? What are the conditions that determine whether co-representing others’ action sequences facilitates or interferes with own performance? Answering these questions could make an important contribution towards advancing our understanding of when and how individuals mind others when jointly performing actions.
Conflicts of interest
The authors declare no conflict of interest.
Acknowledgements
This research was supported by the European Research Council under the European Union's Seventh Framework Program (FP7/2007-2013)/ERC grant agreement n° 609819, SOMICS, and ERC grant agreement n° 616072, JAXPERTISE. We thank Dávid Csűrös, Fruzsina Elekes, and Anna Mária Lisincki for their help with data collection, as well as Tim Welsh and two anonymous reviewers for their helpful comments on an earlier version of this manuscript.
Footnotes
In a task where participants performed bimanual reaching movements, participants initiated and executed their movements more slowly when they had to move their two hands to targets of different colors compared to the same colors. This suggests that costs may arise during target selection when targets with different (rather than the same) perceptual characteristics need to be selected (Diedrichsen et al., 2003).
We initially tried to identify movement offset as the first moment when velocity goes below 5% of maximum velocity (after it has already passed 50% of the maximum velocity). This method turned out to be unreliable, presumably because some participants did not come to a complete standstill at the final target but immediately started moving back to the start location. Hence, they did not always reach the expected low velocity threshold at the final target (participants were not explicitly instructed to pause on the final target but could freely return to the start).
There were also no MT differences between short-long and long-short sequences in the individual baselines of Experiment 2 (t(19) = 1.40, p = .179, Cohen’s d = 0.31) and Experiment 3 (t(19) = 1.85, p = .080, Cohen’s d = 0.41), respectively.
Variability was also significantly higher in the ‘different order’ condition in Experiments 2 and 4 (E2: p = .004; E4: p = .042). The same numerical yet non-significant trend was observed in Experiments 3, 5, and 6.
The results of the analysis for the second movement segment are provided in the supplementary material.
As the analysis of MT1st suggests that our manipulation of target size did not affect the difficulty of the first movement, we checked additionally whether target size had affected the time participants needed to move from the intermediate to the final target (MT2nd). A significant main effect of Own movement (F(1, 19) = 4.89, p = .039, ηp2 = .205) demonstrated that participants needed more time to complete their movements to a small final target compared to a big final target, showing that movement difficulty had been affected by the manipulation of target size, as intended, at least for the second movement phase.
The full report of this analysis is provided in the supplementary material.
Supplementary data associated with this article can be found, in the online version, at https://doi.org/10.1016/j.cognition.2018.08.008.
Appendix A. Supplementary material
The following are the Supplementary data to this article:
References
- Adam J.J., Nieuwenstein J.H., Huys R., Paas F.G., Kingma H., Willems P., Werry M. Control of rapid aimed hand movements: The one-target advantage. Journal of Experimental Psychology: Human Perception and Performance. 2000;26(1):295–312. doi: 10.1037//0096-1523.26.1.295. [DOI] [PubMed] [Google Scholar]
- Albert N.B., Weigelt M., Hazeltine E., Ivry R.B. Target selection during bimanual reaching to direct cues is unaffected by the perceptual similarity of the targets. Journal of Experimental Psychology: Human Perception and Performance. 2007;33(5):1107–1116. doi: 10.1037/0096-1523.33.5.1107. [DOI] [PubMed] [Google Scholar]
- Atmaca S., Sebanz N., Knoblich G. The joint flanker effect: Sharing tasks with real and imagined co-actors. Experimental Brain Research. 2011;211(3–4):371–385. doi: 10.1007/s00221-011-2709-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bested S.R., de Grosbois J., Tremblay L. Better together: Contrasting the hypotheses explaining the one-target advantage. Human Movement Science. 2018;58:347–356. doi: 10.1016/j.humov.2017.11.001. [DOI] [PubMed] [Google Scholar]
- Böckler A., Knoblich G., Sebanz N. Effects of a coactor's focus of attention on task performance. Journal of Experimental Psychology: Human Perception and Performance. 2012;38(6):1404–1415. doi: 10.1037/a0027523. [DOI] [PubMed] [Google Scholar]
- Cracco E., Bardi L., Desmet C., Genschow O., Rigoni D., De Coster L.…Brass M. Automatic imitation: A meta-analysis. Psychological Bulletin. 2018;144(5):453–500. doi: 10.1037/bul0000143. [DOI] [PubMed] [Google Scholar]
- Della Gatta F., Garbarini F., Rabuffetti M., Viganò L., Butterfill S.A., Sinigaglia C. Drawn together: When motor representations ground joint actions. Cognition. 2017;165:53–60. doi: 10.1016/j.cognition.2017.04.008. [DOI] [PubMed] [Google Scholar]
- Diedrichsen J., Grafton S., Albert N., Hazeltine E., Ivry R.B. Goal-selection and movement-related conflict during bimanual reaching movements. Cerebral Cortex. 2006;16(12):1729–1738. doi: 10.1093/cercor/bhj108. [DOI] [PubMed] [Google Scholar]
- Diedrichsen J., Hazeltine E., Kennerley S., Ivry R.B. Moving to directly cued locations abolishes spatial interference during bimanual actions. Psychological Science. 2001;12(6):493–498. doi: 10.1111/1467-9280.00391. [DOI] [PubMed] [Google Scholar]
- Diedrichsen J., Ivry R.B., Hazeltine E., Kennerley S., Cohen A. Bimanual interference associated with the selection of target locations. Journal of Experimental Psychology: Human Perception and Performance. 2003;29(1):64–77. doi: 10.1037//0096-1523.29.1.64. [DOI] [PubMed] [Google Scholar]
- Eskenazi T., Doerrfeld A., Logan G.D., Knoblich G., Sebanz N. Your words are my words: Effects of acting together on encoding. The Quarterly Journal of Experimental Psychology. 2013;66(5):1026–1034. doi: 10.1080/17470218.2012.725058. [DOI] [PubMed] [Google Scholar]
- Fitts P. The information capacity of the human motor system in controlling the amplitude of movement. Journal of Experimental Psychology. 1954;47(6):381–391. [PubMed] [Google Scholar]
- Fine J.M., Amazeen E.L. Interpersonal Fitts’ law: When two perform as one. Experimental Brain Research. 2011;211(3–4):459–469. doi: 10.1007/s00221-011-2707-y. [DOI] [PubMed] [Google Scholar]
- Forbes P.A., Hamilton A.F.D.C. Moving higher and higher: Imitators’ movements are sensitive to observed trajectories regardless of action rationality. Experimental Brain Research. 2017;235(9):2741–2753. doi: 10.1007/s00221-017-5006-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fowler B., Duck T., Mosher M., Mathieson B. The coordination of bimanual aiming movements: Evidence for progressive desynchronization. The Quarterly Journal of Experimental Psychology. 1991;43(2):205–221. doi: 10.1080/14640749108400967. [DOI] [PubMed] [Google Scholar]
- Franz E.A., Eliassen J.C., Ivry R.B., Gazzaniga M.S. Dissociation of spatial and temporal coupling in the bimanual movements of callosotomy patients. Psychological Science. 1996;7(5):306–310. [Google Scholar]
- Franz E.A., Zelaznik H.N., McCabe G. Spatial topological constraints in a bimanual task. Acta psychologica. 1991;77(2):137–151. doi: 10.1016/0001-6918(91)90028-x. [DOI] [PubMed] [Google Scholar]
- Gambi, C., Van de Cavey, J., & Pickering, M. J. (2017, July). Joint interference in picture description: Evidence for linguistically-detailed simulation of others’ utterances, but only when speaking concurrently. Paper presented at the 7th joint action meeting (JAM7), London, UK.
- Gambi C., Van de Cavey J., Pickering M.J. Interference in joint picture naming. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2015;41(1):1–21. doi: 10.1037/a0037438. [DOI] [PubMed] [Google Scholar]
- Gazzola V., Aziz-Zadeh L., Keysers C. Empathy and the somatotopic auditory mirror system in humans. Current Biology. 2006;16(18):1824–1829. doi: 10.1016/j.cub.2006.07.072. [DOI] [PubMed] [Google Scholar]
- Glover S., Dixon P. The role of predictability in cooperative and competitive joint action. Journal of Experimental Psychology: Human Perception and Performance. 2017;43(4):644. doi: 10.1037/xhp0000362. [DOI] [PubMed] [Google Scholar]
- Griffiths D., Tipper S.P. Priming of reach trajectory when observing actions: Hand-centred effects. The Quarterly Journal of Experimental Psychology. 2009;62(12):2450–2470. doi: 10.1080/17470210903103059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hazeltine E., Diedrichsen J., Kennerley S.W., Ivry R.B. Bimanual cross-talk during reaching movements is primarily related to response selection, not the specification of motor parameters. Psychological Research. 2003;67(1):56–70. doi: 10.1007/s00426-002-0119-0. [DOI] [PubMed] [Google Scholar]
- Heuer H. Structural constraints on bimanual movements. Psychological Research. 1993;55(2):83–98. doi: 10.1007/BF00419639. [DOI] [PubMed] [Google Scholar]
- Heuer H., Klein W. The influence of movement cues on intermanual interactions. Psychological Research. 2006;70(4):229–244. doi: 10.1007/s00426-005-0218-9. [DOI] [PubMed] [Google Scholar]
- Heuer H., Klein W. Intermanual interactions in discrete and periodic bimanual movements with same and different amplitudes. Experimental Brain Research. 2005;167(2):220–237. doi: 10.1007/s00221-005-0015-0. [DOI] [PubMed] [Google Scholar]
- Heuer H., Spijkers W., Kleinsorge T., van der Loo H., Steglich C. The time course of cross-talk during the simultaneous specification of bimanual movement amplitudes. Experimental Brain Research. 1998;118(3):381–392. doi: 10.1007/s002210050292. [DOI] [PubMed] [Google Scholar]
- Heuer, H. (1996). Coordination. In: H. Heuer, & S. W. Keele (Eds.), Handbook of perception and action. Vol. 2: Motor skills (pp. 121–180). London: Academic Press.
- Heyes C. Automatic imitation. Psychological Bulletin. 2011;137(3):463–483. doi: 10.1037/a0022288. [DOI] [PubMed] [Google Scholar]
- Hoedemaker R.S., Ernst J., Meyer A.S., Belke E. Language production in a shared task: Cumulative Semantic Interference from self-and other-produced context words. Acta Psychologica. 2017;172:55–63. doi: 10.1016/j.actpsy.2016.11.007. [DOI] [PubMed] [Google Scholar]
- Ivry R., Diedrichsen J., Spencer R., Hazeltine E., Semjen A. A cognitive neuroscience perspective on bimanual coordination and interference. In: Swinnen S.P., Duysens J., editors. Neuro-behavioral determinants of interlimb coordination. Springer; Boston, MA: 2004. pp. 259–295. [Google Scholar]
- Jeannerod M. Neural simulation of action: A unifying mechanism for motor cognition. Neuroimage. 2001;14(1):S103–S109. doi: 10.1006/nimg.2001.0832. [DOI] [PubMed] [Google Scholar]
- Keller P.E., Knoblich G., Repp B.H. Pianists duet better when they play with themselves: On the possible role of action simulation in synchronization. Consciousness and Cognition. 2007;16(1):102–111. doi: 10.1016/j.concog.2005.12.004. [DOI] [PubMed] [Google Scholar]
- Kelso J.A.S., Putnam C.A., Goodman D. On the space-time structure of human interlimb co-ordination. The Quarterly Journal of Experimental Psychology. 1983;35(2):347–375. doi: 10.1080/14640748308402139. [DOI] [PubMed] [Google Scholar]
- Kelso J.A.S., Southard D.L., Goodman D. On the nature of human interlimb coordination. Science. 1979;203(4384):1029–1031. doi: 10.1126/science.424729. [DOI] [PubMed] [Google Scholar]
- Kennerley S.W., Diedrichsen J., Hazeltine E., Semjen A., Ivry R.B. Callosotomy patients exhibit temporal uncoupling during continuous bimanual movements. Nature Neuroscience. 2002;5(4):376–381. doi: 10.1038/nn822. [DOI] [PubMed] [Google Scholar]
- Kilner J.M., Paulignan Y., Blakemore S.J. An interference effect of observed biological movement on action. Current Biology. 2003;13(6):522–525. doi: 10.1016/s0960-9822(03)00165-9. [DOI] [PubMed] [Google Scholar]
- Knoblich G., Jordan J.S. Action coordination in groups and individuals: Learning anticipatory control. Journal of Experimental Psychology: Learning, Memory, and Cognition. 2003;29(5):1006–1016. doi: 10.1037/0278-7393.29.5.1006. [DOI] [PubMed] [Google Scholar]
- Kourtis D., Knoblich G., Wozniak M., Sebanz N. Attention allocation and task representation during joint action planning. Journal of Cognitive Neuroscience. 2014;26(10):2275–2286. doi: 10.1162/jocn_a_00634. [DOI] [PubMed] [Google Scholar]
- Kuhlen A.K., Abdel Rahman R. Having a task partner affects lexical retrieval: Spoken word production in shared task settings. Cognition. 2017;166:94–106. doi: 10.1016/j.cognition.2017.05.024. [DOI] [PubMed] [Google Scholar]
- Kunde W., Weigelt M. Goal congruency in bimanual object manipulation. Journal of Experimental Psychology: Human Perception and Performance. 2005;31(1):145–156. doi: 10.1037/0096-1523.31.1.145. [DOI] [PubMed] [Google Scholar]
- Marteniuk R.G., MacKenzie C.L., Baba D.M. Bimanual movement control: Information processing and interaction effects. The Quarterly Journal of Experimental Psychology. 1984;36(2):335–365. [Google Scholar]
- Mechsner F., Kerzel D., Knoblich G., Prinz W. Perceptual basis of bimanual coordination. Nature. 2001;414(6859):69–73. doi: 10.1038/35102060. [DOI] [PubMed] [Google Scholar]
- Mechsner F., Knoblich G. Do muscles matter for coordinated action? Journal of Experimental Psychology: Human Perception and Performance. 2004;30(3):490–503. doi: 10.1037/0096-1523.30.3.490. [DOI] [PubMed] [Google Scholar]
- Pan X., Hamilton A.F.D.C. Automatic imitation in a rich social context with virtual characters. Frontiers in Psychology. 2015;6:790. doi: 10.3389/fpsyg.2015.00790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prinz W. Perception and action planning. European Journal of Cognitive Psychology. 1997;9(2):129–154. [Google Scholar]
- Repp B.H., Su Y.H. Sensorimotor synchronization: A review of recent research (2006–2012) Psychonomic Bulletin & Review. 2013;20(3):403–452. doi: 10.3758/s13423-012-0371-2. [DOI] [PubMed] [Google Scholar]
- Richardson M.J., Marsh K.L., Baron R.M. Judging and actualizing intrapersonal and interpersonal affordances. Journal of Experimental Psychology: Human Perception and Performance. 2007;33(4):845–859. doi: 10.1037/0096-1523.33.4.845. [DOI] [PubMed] [Google Scholar]
- Richardson M.J., Marsh K.L., Isenhower R.W., Goodman J.R., Schmidt R.C. Rocking together: Dynamics of intentional and unintentional interpersonal coordination. Human Movement Science. 2007;26(6):867–891. doi: 10.1016/j.humov.2007.07.002. [DOI] [PubMed] [Google Scholar]
- Rizzolatti G., Sinigaglia C. The functional role of the parieto-frontal mirror circuit: Interpretations and misinterpretations. Nature Reviews. Neuroscience. 2010;11(4):264–274. doi: 10.1038/nrn2805. [DOI] [PubMed] [Google Scholar]
- Roberts J.W., Constable M.D., Burgess R., Lyons J.L., Welsh T.N. The influence of intrapersonal sensorimotor experiences on the corticospinal responses during action–observation. Social Neuroscience. 2018;13(2):246–256. doi: 10.1080/17470919.2017.1289979. [DOI] [PubMed] [Google Scholar]
- Roberts J.W., Katayama O., Lung T., Constable M.D., Elliott D., Lyons J.L., Welsh T.N. The modulation of motor contagion by intrapersonal sensorimotor experience. Neuroscience Letters. 2016;624:42–46. doi: 10.1016/j.neulet.2016.04.063. [DOI] [PubMed] [Google Scholar]
- Ruys K.I., Aarts H. When competition merges people's behavior: Interdependency activates shared action representations. Journal of Experimental Social Psychology. 2010;46(6):1130–1133. [Google Scholar]
- Sacheli L.M., Candidi M., Pavone E.F., Tidoni E., Aglioti S.M. And yet they act together: Interpersonal perception modulates visuo-motor interference and mutual adjustments during a joint-grasping task. PLoS ONE. 2012;7 doi: 10.1371/journal.pone.0050223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt R.C., Bienvenu M., Fitzpatrick P.A., Amazeen P.G. A comparison of intra-and interpersonal interlimb coordination: Coordination breakdowns and coupling strength. Journal of Experimental Psychology: Human Perception and Performance. 1998;24(3):884–900. doi: 10.1037//0096-1523.24.3.884. [DOI] [PubMed] [Google Scholar]
- Schmidt R.C., Carello C., Turvey M.T. Phase transitions and critical fluctuations in the visual coordination of rhythmic movements between people. Journal of Experimental Psychology: Human Perception and Performance. 1990;16(2):227–247. doi: 10.1037//0096-1523.16.2.227. [DOI] [PubMed] [Google Scholar]
- Schmidt R.C., Richardson M.J. Dynamics of interpersonal coordination. In: Fuchs A., Jirsa V., editors. Coordination: Neural, behavioral and social dynamics. Springer; Berlin: 2008. pp. 281–308. [Google Scholar]
- Schmidt R.C., Turvey M.T. Phase-entrainment dynamics of visually coupled rhythmic movements. Biological Cybernetics. 1994;70(4):369–376. doi: 10.1007/BF00200334. [DOI] [PubMed] [Google Scholar]
- Schmitz L., Vesper C., Sebanz N., Knoblich G. Co-representation of others’ task constraints in joint action. Journal of Experimental Psychology: Human Perception and Performance. 2017;43(8):1480–1493. doi: 10.1037/xhp0000403. [DOI] [PubMed] [Google Scholar]
- Sebanz N., Bekkering H., Knoblich G. Joint action: Bodies and minds moving together. Trends in Cognitive Sciences. 2006;10(2):70–76. doi: 10.1016/j.tics.2005.12.009. [DOI] [PubMed] [Google Scholar]
- Sebanz N., Knoblich G., Prinz W. Representing others' actions: Just like one's own? Cognition. 2003;88(3):B11–B21. doi: 10.1016/s0010-0277(03)00043-x. [DOI] [PubMed] [Google Scholar]
- Sebanz N., Knoblich G., Prinz W. How two share a task: Corepresenting stimulus-response mappings. Journal of Experimental Psychology: Human Perception and Performance. 2005;31(6):1234–1246. doi: 10.1037/0096-1523.31.6.1234. [DOI] [PubMed] [Google Scholar]
- Shockley K., Riley M.A. Interpersonal couplings in human interactions. In: Webber C., Marwan N., editors. Recurrence quantification analysis. Springer; Cham: 2015. pp. 399–421. [Google Scholar]
- Shockley K., Santana M.V., Fowler C.A. Mutual interpersonal postural constraints are involved in cooperative conversation. Journal of Experimental Psychology: Human Perception and Performance. 2003;29(2):326–332. doi: 10.1037/0096-1523.29.2.326. [DOI] [PubMed] [Google Scholar]
- Smits-Engelsman B.C.M., Van Galen G.P., Duysens J. The breakdown of Fitts’ law in rapid, reciprocal aiming movements. Experimental Brain Research. 2002;145(2):222–230. doi: 10.1007/s00221-002-1115-8. [DOI] [PubMed] [Google Scholar]
- Spijkers W., Heuer H. Structural constraints on the performance of symmetrical bimanual movements with different amplitudes. The Quarterly Journal of Experimental Psychology. 1995;48(3):716–740. [Google Scholar]
- Spijkers W., Heuer H., Kleinsorge T., van der Loo H. Preparation of bimanual movements with same and different amplitudes: Specification interference as revealed by reaction time. Acta Psychologica. 1997;96(3):207–227. [Google Scholar]
- Tsai C.C., Kuo W.J., Hung D.L., Tzeng O.J. Action co-representation is tuned to other humans. Journal of Cognitive Neuroscience. 2008;20(11):2015–2024. doi: 10.1162/jocn.2008.20144. [DOI] [PubMed] [Google Scholar]
- van der Steen M.M., Keller P.E. The ADaptation and Anticipation Model (ADAM) of sensorimotor synchronization. Frontiers in Human Neuroscience. 2013;7:253. doi: 10.3389/fnhum.2013.00253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Wel R.P., Fu E. Entrainment and task co-representation effects for discrete and continuous action sequences. Psychonomic Bulletin & Review. 2015;22(6):1685–1691. doi: 10.3758/s13423-015-0831-6. [DOI] [PubMed] [Google Scholar]
- Vesper, C., Schmitz, L., Sebanz, N., & Knoblich, G. (2013). Joint action coordination through strategic reduction of variability. In M. Knauff, M. Pauen, N. Sebanz, & I. Wachsmuth (Eds.), Proceedings of the 35th annual conference of the Cognitive Science Society (pp. 1522–1527). Austin, TX: Cognitive Science Society.
- Vesper C., Schmitz L., Safra L., Sebanz N., Knoblich G. The role of shared visual information for joint action coordination. Cognition. 2016;153:118–123. doi: 10.1016/j.cognition.2016.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesper C., van der Wel R.P., Knoblich G., Sebanz N. Making oneself predictable: Reduced temporal variability facilitates joint action coordination. Experimental Brain Research. 2011;211(3–4):517–530. doi: 10.1007/s00221-011-2706-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesper C., van der Wel R.P., Knoblich G., Sebanz N. Are you ready to jump? Predictive mechanisms in interpersonal coordination. Journal of Experimental Psychology: Human Perception and Performance. 2013;39(1):48–61. doi: 10.1037/a0028066. [DOI] [PubMed] [Google Scholar]
- Vlainic E., Liepelt R., Colzato L.S., Prinz W., Hommel B. The virtual co-actor: The social Simon effect does not rely on online feedback from the other. Frontiers in Psychology. 2010;1:208. doi: 10.3389/fpsyg.2010.00208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wagenmakers E.J., Brown S. On the linear relation between the mean and the standard deviation of a response time distribution. Psychological Review. 2007;114:830–841. doi: 10.1037/0033-295X.114.3.830. [DOI] [PubMed] [Google Scholar]
- Wang Y., Hamilton A.F.D.C. Social top-down response modulation (STORM): A model of the control of mimicry in social interaction. Frontiers in Human Neuroscience. 2012;6:153. doi: 10.3389/fnhum.2012.00153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weigelt M. Re-examining structural constraints on the initiation of bimanual movements: The role of starting locations, movement amplitudes, and target locations. Human Movement Science. 2007;26(2):212–225. doi: 10.1016/j.humov.2007.01.010. [DOI] [PubMed] [Google Scholar]
- Weigelt M., Rieger M., Mechsner F., Prinz W. Target-related coupling in bimanual reaching movements. Psychological Research. 2007;71(4):438–447. doi: 10.1007/s00426-005-0043-1. [DOI] [PubMed] [Google Scholar]
- Welsh T.N., Elliott D., Anson J.G., Dhillon V., Weeks D.J., Lyons J.L., Chua R. Does Joe influence Fred's action? Inhibition of return across different nervous systems. Neuroscience Letters. 2005;385(2):99–104. doi: 10.1016/j.neulet.2005.05.013. [DOI] [PubMed] [Google Scholar]
- Welsh T.N., Higgins L., Ray M., Weeks D.J. Seeing vs. believing: Is believing sufficient to activate the processes of response co-representation? Human Movement Science. 2007;26(6):853–866. doi: 10.1016/j.humov.2007.06.003. [DOI] [PubMed] [Google Scholar]
- Welsh T.N., McDougall L.M., Weeks D.J. The performance and observation of action shape future behaviour. Brain and Cognition. 2009;71(2):64–71. doi: 10.1016/j.bandc.2009.04.001. [DOI] [PubMed] [Google Scholar]
- Wilson M., Knoblich G. The case for motor involvement in perceiving conspecifics. Psychological Bulletin. 2005;131(3):460–473. doi: 10.1037/0033-2909.131.3.460. [DOI] [PubMed] [Google Scholar]
- Wolpert D.M., Doya K., Kawato M. A unifying computational framework for motor control and interaction. Philosophical Transactions of the Royal Society of London B: Biological Sciences. 2003;358(1431):593–602. doi: 10.1098/rstb.2002.1238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zopf R., Truong S., Finkbeiner M., Friedman J., Williams M.A. Viewing and feeling touch modulates hand position for reaching. Neuropsychologia. 2011;49(5):1287–1293. doi: 10.1016/j.neuropsychologia.2011.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.