Abstract
Purpose
The objective of this study was to compare tumor characteristics, biomarkers, and surrogate subtypes of breast cancer between Sudanese and German women.
Methods
Tumor characteristics and immunohistochemistry markers (estrogen receptor [ER], progesterone receptor [PR], and human epidermal growth factor receptor 2 [HER2]) were collected from the routine assessment of consecutive patients with invasive breast cancer diagnosed from 2010 to 2015 (Gezira University Pathology Laboratory, Gezira, Sudan) and from 1999 to 2013 (Breast Centre, Martin-Luther-University, Halle, Germany).
Results
A total of 2,492 patients (German [n = 1,932] and Sudanese [n = 560]) were included. Age at diagnosis ranged from 20 to 94 years. Sudanese women were, on average, 10 years younger than German women, with a mean (± standard deviation) age of 48.8 (13.5) and 58.6 (12.4) years, respectively. The Sudanese women had a higher grade, larger tumor, and more lymph node positivity compared with German women. ER-, PR-, and HER2-negative proportions were 55%, 61.8%, and 71.3%, respectively, for Sudanese women versus 22.7%, 32.3%, and 82.5%, respectively, for German women. The triple-negative subtype was more prevalent in Sudanese women (34.5%) than in German women (14.2%). The strongest factor associated with ER-negative disease was grade III (odds ratio, 19.6; 95% CI 11.6 to 33.4; P < .001). Sudanese patients were at higher risk for ER-negative breast cancer, with an odds ratio of 2.01 (P = .001; adjusted for age, size, nodal status, histologic type, and grade). Stratified by grade, the influence of origin was observed in grade I and grade II tumors, but not in grade III tumors.
Conclusion
Sudanese women had more aggressive tumor characteristics and unfavorable prognostic biomarkers. After adjustment, Sudanese origin was still associated with hormone receptor–negative disease, especially in grade I and II tumors. These findings suggest differences in tumor biology among these ethnic groups.
INTRODUCTION
Breast cancer (BC) is the most common malignancy in women and is the primary cause of cancer mortality in developed countries and most developing countries. In developing countries, the incidence is relatively low compared with developed countries; however, mortality rates are alarmingly high.1,2 BC is a highly diversified cancer in terms of its clinical presentation, morphology, molecular markers, prognosis, and treatment outcome.
BC in African and African American women is commonly an aggressive disease. Many studies have described that native African, African in diaspora, and African American women present at an earlier age, with higher grade, advanced stage, and with higher hormone receptor (HR) –negative rates than their white counterparts.3-6
African-based studies have reported high proportions of estrogen receptor (ER) – and progesterone receptor (PR) –negative BCs—for instance, 76% of BC cases in Nigeria,6 Ghana,7 and Kenya8; 65% of cases in Uganda9; and 61% of cases in Mali10 were found to be ER negative. Recent studies, however, have shown relatively low proportions of ER- and PR-negative BC: Ethiopia (34.7%),11 South Africa (37%),12 Sudan (36%),13 Nigeria (35%),14 and Eritrea (32%).15 These recent findings are comparable with that from African American premenopausal women in the Carolina Breast Cancer Study (39%).16 A recent systematic review and meta-analysis in Africa showed that the proportion of ER- and PR-negative and human epidermal growth factor receptor 2 (HER2)–positive BC in Africa is highly variable. The review proposed that young age, high grade, retrospective assessment, tissue collection, early year of diagnosis, and place of origin (West Africa) were contributing factors to HR-negative BC.17
The taxonomy of the molecular subtypes of BC—luminal A and B, HER2 overexpressing, and basal like—that was described first by Perou et al18 in 2000 has revolutionized the management of BC, and these subtypes are independent prognostic markers. Luminal A and B are more common in older white women and have a good outcome and prognosis.19,20 Conversely, HER2-enriched (ER- and PR-negative and HER2-positive) and triple-negative BCs (TNBCs; negative for ER, PR, and HER2) are 85% concordant with the basal-like subtype and have poor outcome.21 TNBCs are often poorly differentiated, insensitive to hormone therapy, and have a poor prognosis and outcome. TNBCs are usually treated with radiochemotherapy, whereas HER2-enriched BCs are candidates for HER2-targeted (trastuzumab) therapy. TNBCs have been found to be more dominant in younger women and in African women or those of African descent. Studies have shown that these intrinsic subtypes show remarkable variation in terms of race and ethnicity, geographic distribution, survival, and therapeutic response.16,19 The racial and geographic variation could be a result of demographic structure, reproductive patterns, access to health care, and intrinsic biologic factors. These regional and ethnic variations warrant a more thorough determination of biomarkers as part of routine assessment22 and personalized medicine in BC treatment.
A few studies have shown tumor characteristics and biomarker distribution between native African women and white women. Amadori et al23 found that Tanzanian women had a higher histologic grade and proliferative index, advanced stage, and higher ER- and/or PR-negative BC than white Italian women. Another two studies that compared Sudanese women with Italian women13 and Ghanaian women with Norwegian women24 found a similar proportion of ER and PR positivity in both African and European women, although African women presented at a younger age, with higher grade, and more advanced stage. These few studies were conducted in small populations with sample sizes of 114 and 51.
The purpose of this study was to compare tumor characteristics and the distribution of ER, PR, and HER2 biomarkers and their derived intrinsic subtypes of BC in a large case series among Sudanese and German women.
METHODS
Data Collection
Ethical approval was obtained from the research committees of both institutions. Data were retrieved by using a standard protocol from the Histopathology Department of the University of Gezira and electronically from the Breast Clinic at Martin-Luther-University (MLU). This study was a retrospective, facility-based consecutive case series from 2010 to 2015 for Sudanese women and 1999 to 2013 for German women—all women with histologically confirmed invasive breast carcinoma and ER, PR, and HER2 results available were included in the study. Demographic data and tumor characteristics were obtained from medical records, and HR status (ER and PR) and HER2 expression were determined by using a standard method. Tumor grade was assessed according to the modified Nottingham Bloom-Richardson grading system.25 Tumor size and nodal status was described according to the TNM classification.26
Study Setting
Sudan, the largest country in Africa, is located in the northeast of Africa with diverse ethnic groups, including African, Arab, and African-Arab tribes. The state of Gezira has approximately 4 million inhabitants; the University of Gezira histopathology laboratory is the only public institution that serves this state. The department of gynecology at MLU is a certified breast center that serves the population of Halle and its surrounding areas.
Immunohistochemistry
For Sudanese women, ER, PR, and HER2 immunostaining was performed manually at the Gezira University pathology laboratory using standard immunohistochemistry (IHC). In brief, formalin-fixed, paraffin-embedded breast tumor blocks were obtained and 4-µm tissues were sectioned, deparaffinized, and rehydrated. Antigen retrieval was performed by using a water bath in 10 mM citrate buffer (pH 6.0) at 95°C for 45 minutes and blocked with 3% hydrogen peroxide in phosphate buffered saline. Tissue section was permeabilized and blocked with blocking agent (BioGenex, Kent, United Kingdom), then incubated for 1 hour with primary antibodies at room temperature: anti-ER (clone EPR703; BioGenex), anti-PR (clone PR88; BioGenex), and anti-HER2neu (clone CB11), followed by biotinylated horse anti-mouse or goat anti-rabbit secondary antibodies. Staining was visualized by using diaminobenzidine and counterstained with hematoxylin.
For Halle patients, IHC was performed at MLU as follows: 4-μm paraffin-embedded sections were prepared and tissue sections were boiled in 10 mM citrate buffer (pH 6.0) for 10 minutes followed by cooling at 25°C. Sections were covered with monoclonal mouse anti-human ER (clone 1D5; Zytomed Systems, Berlin, Germany), monoclonal mouse anti-human PR (clone 636; Dako, Carpinteria, CA), and HercepTest (Dako) for HER2/neu by using a semiautomated system (IntelliPath; Biocare Medical, Pacheco, CA).
ER and PR were considered positive if ≥1% nuclei of tumor cells stained according to the ASCO/College American Pathology guidelines22 for both the Sudanese and German patients. HER2 was scored as 0, 1+, 2+, or 3+. Fluorescent in situ hybridization was not performed for intermediate 2+ HER2 in both groups; only a score of 3+ was considered HER2 enriched, whereas scores ≤ 2+ were assumed to be HER2 negative. Furthermore, Ki-67 was not assessed to evaluate mitotic index
Subtypes were defined as luminal A–like (ER- and/or PR positive and HER2-negative), luminal B–like (ER- and/or PR-positive and HER2-positive), HER2 type (ER- and PR-negative and HER2-positive), and triple negative (ER-, PR-, and HER2-negative).28,29
Data analysis was performed by using SPSS for Windows version 19 (SPSS, Chicago, IL). Tumor characteristics and biomarkers of Sudanese and German women were compared across BC subtypes by using the χ2 test for categorical variables. Logistic regression analysis was used to determine the odds ratio (OR) to evaluate the effect of age, tumor size, grade, and histology on the probability of ER-positive tumors or tumor subtypes. All P values were based on two-tailed tests of significance, where P < .05 was considered statistically significant.
RESULTS
A total of 2,492 women with invasive BC—1,932 from Halle, Germany, and 560 from Gezira, Sudan—were included in this cohort study.
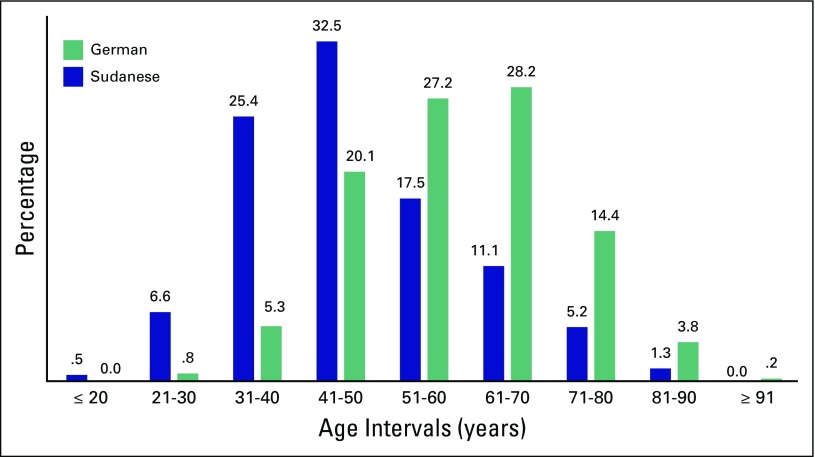
As noted in Table 1, the age range for Sudanese and German patients was similar, but Sudanese women, on average, were diagnosed 10 years earlier than German women. The majority of Sudanese women who were diagnosed with BC were younger than 50 years of age, whereas the majority of German patients were age older than 50 years of age (Fig 1).
Table 1.
Comparison of Tumor Characteristics and Biomarkers Among Sudanese and German Women With Breast Cancer
Fig 1.
Bar graph showing the distribution of age at diagnosis among Sudanese and German women.
The most frequent histologic type of cancer in both the Sudanese and German women was invasive carcinoma of no special type. Invasive lobular carcinoma was three times more common in German patients. Aggressive histologic subtypes, such as primary invasive squamous carcinoma were only reported in Sudanese women (n = 2). More than one half (56.3%) of German patients had a tumor size of ≤ 2 cm; fewer Sudanese patients (15.5%) had tumors of this size. Sudanese women presented with higher stages of BC (four times higher for T3 and three times higher for T4) than German women. Similarly, Sudanese women presented with poorly differentiated tumors (two times as many cases of grade III BC, but six times fewer cases of grade I) compared with German women. German women had a higher proportion of node-negative disease (65.7%) compared with Sudanese women (47.3%).
A significant difference was observed in ER-negative and/or PR-negative BC (54% v 21%; P = .001) between Sudanese and German patients. ER- and PR-negative rates of Sudanese patients were 55% and 62%, respectively, compared with ER- and PR-negative rates of 22.2% and 32%, respectively, in German patients. HER2 expression was higher in Sudanese women than in German women at 28.7% and 17.5%, respectively.
In logistic regression multivariate analysis adjusted for age, origin, grade, and histologic type, it was noted that Sudanese women had a two-fold higher risk for ER-negative BC (P < .001; OR = 2.01) compared with German women. Women age ≤ 50 years were more likely to develop ER-negative BC (OR = 1.28). Of note, women who were diagnosed with grade II BC had a 4.5-fold higher risk of ER-negative BC compared with those diagnosed with grade I BC. Surprisingly, the risk was extremely high (OR, approximately 20) for those who were diagnosed with grade III BC. Similarly, there was a notable increase in the risk of ER negativity as tumor size increased. Women who were diagnosed with T2 BC had a 1.5-fold higher risk of ER-negative tumors compared with those who were diagnosed with T1 BC, and risk was slightly higher for those diagnosed with T3 and T4 BC (OR, 2.03 and OR, 1.70, respectively). There was no significant association between ER negativity and lymph node metastasis (Table 2).
Table 2.
Proportions and Risk Ratios for ER-Negative Disease Adjusted for Age, Origin, Histologic Type, Tumor Size, Lymph Node Status, and Grade in German and Sudanese Patients
For sensitivity analysis multivariate logistic regression, we stratified by tumor grade and adjusted for origin, age, tumor size, and nodal status (Table 3). In grade I and II tumors (n = 1,827), the strongest predictive factor for ER-negative disease was Sudanese origin (OR, 3.51), followed by larger tumor size (OR, 2.91 for T3 tumors). In grade III tumors (n = 658), neither origin, nor tumor size predicted ER status; however, invasive lobular carcinomas were more likely to be ER positive, even in poorly differentiated cancers (OR, 0.12).
Table 3.
Multivariate Logistic Regression Sensitivity Analysis Stratified by Grade and Adjusted for Origin, Histologic Type, Tumor Size, Lymph Node Status, and Age Among German and Sudanese Women With Breast Cancer
Our study revealed significant differences in the percentage of molecular subtypes of BC between the two ethnicities. German women had a higher rate of luminal A and a lower rate of more aggressive types of BC (HER2 and TNBCs).
Regarding tumor characteristics in relation to molecular subtype (Table 4), approximately three quarters (74.1%) of Sudanese women with TNBC were ≤ 50 years of age, whereas German women were ≥ 50 years of age.
Table 4.
Intrinsic Subtypes and Clinicopathologic Characteristics of Sudanese and German Women (immunohistochemistry based)
Approximately 73% of Sudanese women and 90% of German women had Luminal A subtype with grade I and II BC; however, German women had fewer luminal B type BCs (25%) with grade III BC than Sudanese women (65%). There were no differences in tumor grade with HER2-enriched and TNBC subtypes in both groups. There were notable differences in tumor size and lymph node metastasis among BC subtypes between the two ethnicities (Table 4).
DISCUSSION
This study was a large hospital-based case series that compared BC between African (Gezira, Sudan) and white (Halle, Germany) patients. The study revealed that Sudanese women were younger and had worse prognostic pathologic markers, such as larger tumor size, higher grade, frequent positive lymph node status, and HR-negative status, compared with German women, which is consistent with previous findings.6-9 We also found that women with larger tumor size and younger age (age ≤ 50 years) were more likely to develop ER- and PR-negative BC. When the case series was stratified for grade, Sudanese origin and larger tumor size demonstrated a significant risk for ER-negative BC in patients with grade I and II tumors, but not in grade III tumors, which suggests that poorly differentiated breast tumors are an independent risk factor for ER-negative status.
We noted a higher proportion of ER- and PR-negative BCs compared with recently published studies in Africa.11-15,29,30 Publications that directly compared African and white patients show differences in ER status of various magnitudes. Similar to our findings, the result of a higher rate of receptor-negative BC was reported in Tanzania compared with Italian patients23; however, the investigation that compared Sudanese women (from a single institution in Khartoum) with Italian women (from Milan, Italy)13 and, similarly, Ghanaian women with Norwegian women reported minimal difference.24 The latter could, in part, be a result of the inherent bias of small sample size case series. Our Sudanese patients had almost two times more HER2 overexpression than did German women; a similar figure was reported between Swiss women and Saudi Arabian women.31
The main predictive factor of a high proportion of ER-negative disease was the proportion of grade III tumors. Eng et al17 suggested that African ethnic origin could increase the prevalence of ER- and PR-negative disease to some extent. Nonbiologic factors, such as young age at diagnosis, premenopausal status, reproductive pattern (high parity and breast feeding), socioeconomic status, and feeding habits, have been documented as risk factors for ER-negative BC and contribute to geographic variation.6,11,17,21,30 In our case series, we adjusted for tumor grade and other factors, but still we found differences in receptor status between Sudanese and German patients that implies color difference contributes to disparity of BC.
We also found a significant association between histologic grade and ER negativity, regardless of origin, age, tumor size, and nodal status. Sudanese women with BC had a lower tendency to acquire well-differentiated (grade I) cancer (3.6% v 18%) and had a higher predisposition for poorly differentiated (grade III) cancer (42% v 22%; P < .001) compared with German women. This could explain the absolute excess of ER-negative BC in Sudanese women, as poorly differentiated BCs are more likely to be HR negative. A study has shown that grade I and III invasive ductal carcinomas have distinct genetic pathways32 and different gene expression profiles with minimal overlap.33 This suggests that Sudanese women may have inherent genetic factors that contribute to the development of more aggressive, poorly differentiated HR-negative BC. In contrast, it has to be considered that the German population may have an additional large proportion of well-differentiated (GI) cancer that is ER positive, which is different from the Sudanese population. This is consistent with previous studies that revealed that white women develop more well-differentiated tumors than women of other ethnicities.13,33
Sudanese women had a lower percentage of luminal A and a higher rate of the more aggressive BC phenotypes, HER2-enriched BC and TNBC, compared with German women. This finding is similar to that noted for Nigerian women,6,14 other East African reports (Uganda and Tanzania),23,34 and North Africa (Egypt).35 Many studies have reported that TNBCs are the dominant phenotype in native African women6,15,34 and African Americans compared with white women.16,36 Our findings support the existing reports,6,23,34 although a previous single study from Sudan13 has reported much lower rates of ER- and PR-negative BC than our current study. The basal-like and TNBC subtypes are considered more common in younger women; associated with multiple live births, aggressive clinicopathologic and biologic prognostic markers, as well as hereditary BCs with BRCA1/2 gene mutations.37 These risk factors and adverse prognostic markers were documented in Sudanese women, but not in German women. BRCA1/2 was not investigated in this study; however, previous case series studies from the same study area in Sudan have reported significant novel BRCA1/2 mutations.38
Tissue fixation (prolonged warm and/or cold ischemia) and technical variations in IHC (staining and scoring) are claimed to contribute to variations in biomarkers.17,22 If this is the case, many patients may have been missed and treated unnecessarily with more aggressive regimens when simple endocrine therapy could have sufficed; however, this suggestion is counter-argued by some investigators. A study that controlled for fixation and analytical variables appropriately revealed high ER negativity.6 Other supportive findings are the disparity between African American and white American women.16,35 We found a similar proportion of ER-negative disease in grade III tumors among Sudanese and German women. In cases of severe technical variation, we would have expected to find differences in ER status in patients with grade III BCs; therefore, this may point toward some reliability in the technical procedures in our study, but detail gene analysis is needed to confirm real biologic differences.
These adverse prognostic factors in African women could be explained by the lack of early screening programs, poor health care facilities, and poor health-seeking behavior. As a result of late presentation and being a long-standing tumor without intervention, BCs in African women could possibly undergo tumor progression and may eventually develop aggressive behavior, thereby allowing the cancer to become enlarged, poorly differentiated, and likely to metastasize to the lymph nodes. However, in our study, differences persist even after adjusting for tumor stage, which suggests that this is a result of inherent tumor behavior rather than tumor dedifferentiation over time.
The disparity in the proportion of molecular subtypes between Sudanese and German women could be a result of both nonbiologic and inherent biologic factors, although a systematic study to establish the exact etiology is needed. Environmental factors, such as exposure to insecticides and agricultural pesticides,39 hydrocarbons40 and viral and parasitic infections, could possibly induce Sudanese women to develop more aggressive disease. These factors could modulate the immune and tumor microenvironment. For instance, the relationship between malaria and Burkitt’s lymphoma is well documented.41,42 It is also time to investigate the molecular pathways and genetics in African women with BC in collaboration with other international research networks to reveal underlying biologic differences.
The strength of this study is the large sample size and availability of a range of clinical and pathologic information in individual patients. The limitations of the study include retrospective data collection in the hospital setting, but as both centers serve the surrounding population, we do not assume a selection bias. Fluorescent in situ hybridization was not performed for HER2 equivocal results and was solely determined by IHC. This could underestimate HER2-positive BC. Moreover, laboratory procedures were performed in two different laboratories, possibly leading to technical differences. Because the proportions of ER-negative disease were similar in grade III tumors, we do not assume a high fraction of false-negative results.
In conclusion, there was a remarkable difference in tumor characteristics and biomarkers between Sudanese and German cohorts. Sudanese women with BC had a higher proportion of aggressive tumor types (TNBC and HER2) than did German women. This may point toward a difference in the underlying tumor biology.
ACKNOWLEDGMENT
We thank the staff of the pathology departments of Gezira University and Martin-Luther-University for their dedication in obtaining clinical data. A.T.S. thanks Tigsti Fkadu for helping with data entry and Timothy Rebbeck, MD, for valuable comment.
AUTHOR CONTRIBUTIONS
Conception and design: Asmerom Tesfamariam Sengal, Eva Johanna Kantelhardt
Provision of study materials or patients: Asmerom Tesfamariam Sengal, Nada Suliman Haj Mukhtar, Ahmed Mohammed Elhaj, Ahmed Abdalla Mohamedani, Claudia Wickenhauser, Eva Johanna Kantelhardt
Collection and assembly of data: Asmerom Tesfamariam Sengal, Nada Suliman Haj Mukhtar, Eva Johanna Kantelhardt
Data analysis and interpretation: Asmerom Tesfamariam Sengal, Martina Vetter, Christoph Thomssen, Claudia Wickenhauser, Eva Johanna Kantelhardt
Manuscript writing: Asmerom Tesfamariam Sengal, Eva Johanna Kantelhardt
Final approval of manuscript: All authors
Accountable for all aspects of the work: All authors
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to www.asco.org/rwc or ascopubs.org/jco/site/ifc.
Asmerom Tesfamariam Sengal
No relationship to disclose
Nada Suliman
No relationship to disclose
Haj Mukhtar
No relationship to disclose
Martina Vetter
No relationship to disclose
Ahmed Mohammed Elhaj
No relationship to disclose
Shahinaz Bedri
No relationship to disclose
Steffen Hauptmann
No relationship to disclose
Christoph Thomssen
No relationship to disclose
Ahmed Abdalla Mohamedani
No relationship to disclose
Claudia Wickenhauser
No relationship to disclose
Eva Johanna Kantelhardt
Consulting or Advisory Role: Daiichi Sankyo
Research Funding: Roche (Inst)
REFERENCES
- 1.Allemani C, Weir HK, Carreira H, et al. : Global surveillance of cancer survival 1995-2009: Analysis of individual data for 25,676,887 patients from 279 population-based registries in 67 countries (CONCORD-2). Lancet 385:977-1010, 2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.International Agency for Research on Cancer : GLOBOCAN 2012 v1.0: Cancer incidence and mortality worldwide: IARC CancerBase No. 11. http://globocan.iarc.fr/Default.aspx [Google Scholar]
- 3.Bowen RL, Duffy SW, Ryan DA, et al. : Early onset of breast cancer in a group of British black women. Br J Cancer 98:277-281, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eley JW, Hill HA, Chen VW, et al. : Racial differences in survival from breast cancer. Results of the National Cancer Institute black/white cancer survival study. JAMA 272:947-954, 1994 [DOI] [PubMed] [Google Scholar]
- 5.Chu KC, Anderson WF: Rates for breast cancer characteristics by estrogen and progesterone receptor status in the major racial/ethnic groups. Breast Cancer Res Treat 74:199-211, 2002 [DOI] [PubMed] [Google Scholar]
- 6.Huo D, Ikpatt F, Khramtsov A, et al. : Population differences in breast cancer: Survey in indigenous African women reveals over-representation of triple-negative breast cancer. J Clin Oncol 27:4515-4521, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kantelhardt EJ, Zerche P, Mathewos A, et al. : Breast cancer survival in Ethiopia: A cohort study of 1,070 women. Int J Cancer 135:702-709, 2014 [DOI] [PubMed] [Google Scholar]
- 8.Ohene-Yeboah M, Adjei E: Breast cancer in Kumasi, Ghana. Ghana Med J 46:8-13, 2012 [PMC free article] [PubMed] [Google Scholar]
- 9.Bird PA, Hill AG, Houssami N: Poor hormone receptor expression in East African breast cancer: Evidence of a biologically different disease? Ann Surg Oncol 15:1983-1988, 2008 [DOI] [PubMed] [Google Scholar]
- 10.Nalwoga H, Arnes JB, Wabinga H, et al. : Frequency of the basal-like phenotype in African breast cancer. APMIS 115:1391-1399, 2007 [DOI] [PubMed] [Google Scholar]
- 11.Kantelhardt EJ, Mathewos A, Aynalem A, et al. : The prevalence of estrogen receptor-negative breast cancer in Ethiopia. BMC Cancer 14:895, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McCormack VA, Joffe M, van den Berg E, et al. : Breast cancer receptor status and stage at diagnosis in over 1,200 consecutive public hospital patients in Soweto, South Africa: A case series. Breast Cancer Res 15:R84, 2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Awadelkarim KD, Arizzi C, Elamin E O M, et al. : Pathological, clinical and prognostic characteristics of breast cancer in Central Sudan versus Northern Italy: Implications for breast cancer in Africa. Histopathology 52:445-456, 2008 [DOI] [PubMed] [Google Scholar]
- 14.Adebamowo CA, Famooto A, Ogundiran TO, et al. : Immunohistochemical and molecular subtypes of breast cancer in Nigeria. Breast Cancer Res Treat 110:183-188, 3008, 2008 [DOI] [PubMed] [Google Scholar]
- 15.Tesfamariam A, Roy I: Molecular biology of breast cancer in the Horn of Africa: Case series—A pilot study of breast cancer from Eritrea. ISRN Pathol 2013:787495, 2013 [Google Scholar]
- 16.Carey LA, Perou CM, Livasy CA, et al. : Race, breast cancer subtypes, and survival in the Carolina Breast Cancer Study. JAMA 295:2492-2502, 2006 [DOI] [PubMed] [Google Scholar]
- 17.Eng A, McCormack V, dos-Santos-Silva I: Receptor-defined subtypes of breast cancer in indigenous populations in Africa: A systematic review and meta-analysis. PLoS Med 11:e1001720, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Perou CM, Sørlie T, Eisen MB, et al. : Molecular portraits of human breast tumours. Nature 406:747-752, 2000 [DOI] [PubMed] [Google Scholar]
- 19.Sørlie T, Perou CM, Tibshirani R, et al. : Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA 98:10869-10874, 2001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rezai M, Kellersmann S, Knispel S, et al. : Translating the concept of intrinsic subtypes into an oncoplastic cohort of more than 1000 patients: Predictors of recurrence and survival. Breast 24:384-390, 2015 [DOI] [PubMed] [Google Scholar]
- 21.Goldhirsch A, Wood WC, Coates AS, et al. : Strategies for subtypes: Dealing with the diversity of breast cancer: Highlights of the St. Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2011. Ann Oncol 22:1736-1747, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hammond ME, Hayes DF, Wolff AC, et al. : American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Oncol Pract 6:195-197, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Amadori D, Serra P, Bravaccini S, et al. : Differences in biological features of breast cancer between Caucasian (Italian) and African (Tanzanian) populations. Breast Cancer Res Treat 145:177-183, 2014 [DOI] [PubMed] [Google Scholar]
- 24.Adjei EK, Owusu-Afriyie O, Awuah B, et al. : Hormone receptors and Her2 expression in breast cancer in sub-Saharan Africa. A comparative study of biopsies from Ghana and Norway. Breast J 20:308-311, 2014 [DOI] [PubMed] [Google Scholar]
- 25.Elston CW, Ellis IO: Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: Experience from a large study with long-term follow-up. Histopathology 41:154-161, 2002. (suppl 3A) [PubMed] [Google Scholar]
- 26.Sobin LH, Gospodarowicz MK, Wittekind C: TNM Classification of Malignant Tumours. New York, NY, John Wiley & Sons; 2011 [Google Scholar]
- 27. Reference deleted. [Google Scholar]
- 28.Singh M, Ding Y, Zhang LY, et al. : Distinct breast cancer subtypes in women with early-onset disease across races. Am J Cancer Res 4:337-352, 2014 [PMC free article] [PubMed] [Google Scholar]
- 29.Dickens C, Duarte R, Zietsman A, et al. : Racial comparison of receptor-defined breast cancer in Southern African women: Subtype prevalence and age-incidence analysis of nationwide cancer registry data. Cancer Epidemiol Biomarkers Prev 23:2311-2321, 2014 [DOI] [PubMed] [Google Scholar]
- 30.Ly M, Antoine M, Dembélé AK, et al. : High incidence of triple-negative tumors in sub-saharan Africa: A prospective study of breast cancer characteristics and risk factors in Malian women seen in a Bamako university hospital. Oncology 83:257-263, 2012 [DOI] [PubMed] [Google Scholar]
- 31.Al-Kuraya K, Schraml P, Sheikh S, et al. : Predominance of high-grade pathway in breast cancer development of Middle East women. Mod Pathol 18:891-897, 2005 [DOI] [PubMed] [Google Scholar]
- 32.Roylance R, Gorman P, Hanby A, et al. : Allelic imbalance analysis of chromosome 16q shows that grade I and grade III invasive ductal breast cancers follow different genetic pathways. J Pathol 196:32-36, 2002 [DOI] [PubMed] [Google Scholar]
- 33.Sotiriou C, Wirapati P, Loi S, et al. : Gene expression profiling in breast cancer: Understanding the molecular basis of histologic grade to improve prognosis. J Natl Cancer Inst 98:262-272, 2006 [DOI] [PubMed] [Google Scholar]
- 34.Galukande M, Wabinga H, Mirembe F, et al. : Molecular breast cancer subtypes prevalence in an indigenous Sub-Saharan African population. Pan Afr Med J 17:249, 2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Elesawy BH, Abd El hafez A, Shawky Ael-A, et al. : Immunohistochemistry-based subtyping of breast carcinoma in Egyptian women: A clinicopathologic study on 125 patients. Ann Diagn Pathol 18:21-26, 2014 [DOI] [PubMed] [Google Scholar]
- 36.O'Brien KM, Cole SR, Tse C, et al. : Intrinsic breast tumour subtypes, race, and long-term survival in the Carolina Breast Cancer Study. Clin Cancer Res 16:6100-6110, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Millikan RC, Newman B, Tse CK, et al. : Epidemiology of basal-like breast cancer. Breast Cancer Res Treat 109:123-139, 2008. [Erratum: Breast Cancer Res Treat 109: 141, 2008] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Awadelkarim KD, Aceto G, Veschi S, et al. : BRCA1 and BRCA2 status in a Central Sudanese series of breast cancer patients: Interactions with genetic, ethnic and reproductive factors. Breast Cancer Res Treat 102:189-199, 2007 [DOI] [PubMed] [Google Scholar]
- 39.Dich J, Zahm SH, Hanberg A, et al. : Pesticides and cancer. Cancer Causes Control 8:420-443, 1997 [DOI] [PubMed] [Google Scholar]
- 40.Kaplan C: Indoor air pollution from unprocessed solid fuels in developing countries. Rev Environ Health 25:221-242, 2010 [DOI] [PubMed] [Google Scholar]
- 41.Thorley-Lawson D, Deitsch KW, Duca KA, et al. : The link between Plasmodium falciparum malaria and endemic Burkitt’s lymphoma: New insight into a 50-year-old enigma. PLoS Pathog 12:e1005331, 2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Maizels RM: Parasite immunomodulation and polymorphisms of the immune system. J Biol 8:62, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]