Abstract
Background
Pulmonary hypertension (PH) due to left heart failure (HF) is the most common form of PH. However, treatment is unclear because there are conflicting results about safety and efficacy of PH-targeted therapies.
Objectives
To assess the effects of PH-targeted therapy on exercise capacity in HF patients.
Methods
MEDLINE, EMBASE and the Cochrane Library were searched from January 1990 to July 2017 for randomized controlled trials comparing PH-targeted therapies to conventional therapy in HF. The primary outcome was to assess the effects on exercise capacity. Secondary outcomes included mortality, hospitalisation, NT-proBNP levels, echocardiographic and hemodynamics parameters and discontinuation rate.
Results
22 studies were included (n = 5448), including 3, 8 and 11 studies with low, high and unknown risk of bias, respectively. PH-targeted therapies were associated with an improvement of exercise capacity (standardized mean difference 0.29;95%CI:0.08–0.50, p = 0.006). Pre-specified subgroup analyses found that this improvement was predominantly observed in studies evaluating phosphodiesterase-5 inhibitors and prostanoids and in patients with reduced ejection fraction. Moreover, systolic pulmonary artery pressure measured by echocardiography was improved (mean difference: -7.5mmHg; [95%CI]: -14.9,-0.1, p = 0.05), which was also entirely driven by studies evaluating phosphodiesterase-5 inhibitors. However, PH-targeted therapies were associated with an increased treatment discontinuation rates and a potential increase in mortality compared to standard treatment.
Conclusions
In conclusion, PH-targeted therapies and especially phosphodiesterase-5 inhibitors may improve exercise capacity in patients with HF. However, an increase in adverse outcomes was likely. Moreover, most studies were at high or unknown risk of bias, precluding confident conclusions about the effects of PH-targeted therapies.
Introduction
Pulmonary hypertension due to left heart disease (PH-LHD) appears to be the most common form of PH[1]. Epidemiological studies suggest that PH develops in up to 80% of patients with heart failure with preserved (HFpEF) and reduced (HFrEF) ejection fraction [2]. When present, PH-LHD is associated with more severe symptoms and worse exercise tolerance, and exerts a negative impact on outcomes, doubling the risk of mortality as compared to patients with HFpEF/HFrEF without PH[3].
Chronically elevated pressures within the left ventricle (LV) and atrium lead to pathological changes characterized by enlarged and thickened pulmonary veins, pulmonary capillary dilatation, interstitial oedema, alveolar haemorrhage, and lymphatic vessel and lymph node enlargement. In addition to this phenotype of isolated post-capillary PH, the passive elevation in pressures frequently triggers a superimposed precapillary component in some patients, combining pulmonary vasoconstriction, endothelial dysfunction and vascular remodelling[4]. At this stage, pulmonary artery pressure increases further, and this seems to be in excess of the elevation of pulmonary artery occlusion pressure[5]. Although the definition of combined post-capillary and pre-capillary PH is debated[6, 7], recent evidences suggest these patients are at particularly high risk of morbidity and mortality, long-term prognosis being similar to patients with PAH in many series[8].
Importantly, while established treatments may be effective for improving LV function and reducing LV filling pressures, they provide limited improvements in pulmonary vascular remodelling[9]. Some studies suggested that PH-targeted therapies that were shown to improve outcomes in pulmonary arterial hypertension (PAH)[10] could also have a positive effect on endothelial function and the course of PH-LHD[11, 12]. However, most studies were characterized by a small sample size and many had conflicting results. It is thus a matter of concern that drugs approved for the treatment of PAH are commonly used in patients with PH-LHD despite insufficient data supporting their safety and efficacy[13].
We therefore systematically assessed the efficacy and safety of PH-targeted therapies for patients with HFpEF/HFrEF. The aim of the present systematic review and meta-analysis was to assess the effect of PH-targeted therapies for patient with HFpEF/HFrEF on clinically relevant outcomes including exercise capacity, hospitalisation and death compared to conventional therapies.
Materials and methods
The methods for this systematic review are in accordance with the methodological guidelines for systematic reviews of randomized control trials from «Cochrane Handbook for Systematic Reviews of Interventions»[14]. The complete study protocol is available on PROSPERO (CRD42017083114).
Study objectives
The primary objective of the study was to determine whether PH-targeted therapies improve exercise capacity compared to conventional therapies alone in HFpEF/HFrEF. Given the heterogeneity in mechanism of action, the primary outcome was assessed according to drug classes. Whenever possible, we also assessed if these outcomes were homogeneous amongst subgroups, including with or without PH (as defined in each study), baseline functional class, HFrEF (<45%) or HFpEF (≥45%), and study duration (<6 months and ≥ 6 months). Secondary objectives were to assess the impact of PH-targeted therapies on mortality, hospitalisation, NT-proBNP, echocardiographic and hemodynamics parameters, as well as treatment discontinuation.
Search strategy and selection criteria
We searched MEDLINE, EMBASE and the Cochrane Library from January 1990 through July 2017 for randomised controlled trials evaluating PH-targeted therapies for patients with HFpEF/HFrEF. We used free terms and appropriate indexation terms referring to the population and the intervention of interest (online supplement). No filter for randomised controlled trials was used to ensure maximum sensitivity. We also searched for additional articles using the bibliographies of each included studies and any review articles that we retrieved. In addition, we explored grey literature by hand searching the relevant conference abstracts (online supplement). Non-English papers were translated to English. Studies were included in the systematic review if they met inclusion criteria defined a priori. Studies had to (1) be prospective randomised controlled trials evaluating the effect of PH-targeted therapies, including prostanoids, endothelin receptor antagonists, type-5 phosphodiesterase inhibitors (PDE5-inhibitors) and soluble guanylate cyclase stimulators compared to conventional therapies in adult with HFpEF/HFrEF of all-causes; (2) have a clear identification of a comparator (placebo or conventional therapies alone); (3) report one of the outcomes of interest of the present systematic review and; (4) have a duration of at least 12 weeks. Titles and abstracts were independently assessed by two reviewers (CAG/LVMT). Relevant papers were then reviewed for a final decision about their inclusion in the review. Reviewers were blinded to authors’ names, journal and year of publication throughout this process. Discrepancies were resolved by consensus or by consulting a third reviewer (SP). Reasons for rejection of citations were kept and the agreement between the two reviewers was measured using the quadratic weighted kappa. Two significant studies were published after the research period and were included in the analysis [15, 16].
Assessment of methodological quality
The risk of bias of the selected studies was evaluated independently by two reviewers (CAG/LVMT) using the Cochrane’s Risk of Bias Assessment Tool[17]. The reviewers assigned a low, high or unknown risk of bias for each category. A study was considered to have a high risk of bias or an unknown risk of bias if there was at least one category with a high risk or an unknown risk of bias, respectively. Primary analyses were made on all retrieved studies while sensitivity analyses were made including only articles with a low or an unknown risk of bias.
Data extraction and analysis
The whole process of data extraction was independently made by two reviewers (CAG/LVMT) with tested and validated data collection forms. Retained information included study design, patient characteristics and mean treatment effect on exercise capacity, mortality (all-cause and cardiac-related), hospitalisation (all-cause and cardiac-related), NT-proBNP levels, echocardiographic and hemodynamic parameters and discontinuation rate. 2x2 tables were constructed based on treatment received and available data for the primary and secondary dichotomous outcomes.
Forest plots were created for each outcome. Data were analysed using the Mantel-Haenszel method based on a random-effects model, which accounts for within-study and between-study variability[18]. For continuous outcomes, when multiple scales were used (e.g. exercise capacity), effect sizes were computed using standardized mean difference (SMD) between measures obtained at the end of the study for each comparison group. When the same scale was used, weighted mean difference (MD) were calculated. When mean values or standard deviations were not available, these were estimated as previously described (online supplement)[14, 19]. For dichotomous outcomes, risk ratios (RR) were calculated with their 95% confidence intervals (CIs). If one of the cells contained a value of zero, 0.5 was added to each cell, whereas the studies were excluded when there was no event in both groups. When BNP levels where reported, they were transformed in NT-proBNP using a previously published formula[20]. Cochrane’s Q test and I2 test were used to assess between study heterogeneity and were considered statistically significant at P<0.10 and I2>50%. A sensitivity analysis was also performed using the fixed-effect model.
Subgroup analyses were planned a priori to investigate sources of heterogeneity in the main analyses. Sensitivity analyses were made according to the method used to assess exercise capacity. Sensitivity analysis was also made excluding one trial due to its important weight and its suspiciously delayed publication 15 years after its completion[21]. Publication bias was assessed visually using funnel plots made according to the method used to assess exercise capacity because standardized mean differences are naturally correlated with their standard error and can therefore produce false asymmetry in a funnel plot[14]. All analyses were performed with Review Manager (The Cochrane Collaboration, Oxford, England). The report was written according to the PRISMA statement[22].
Results
Characteristics of the selected studies
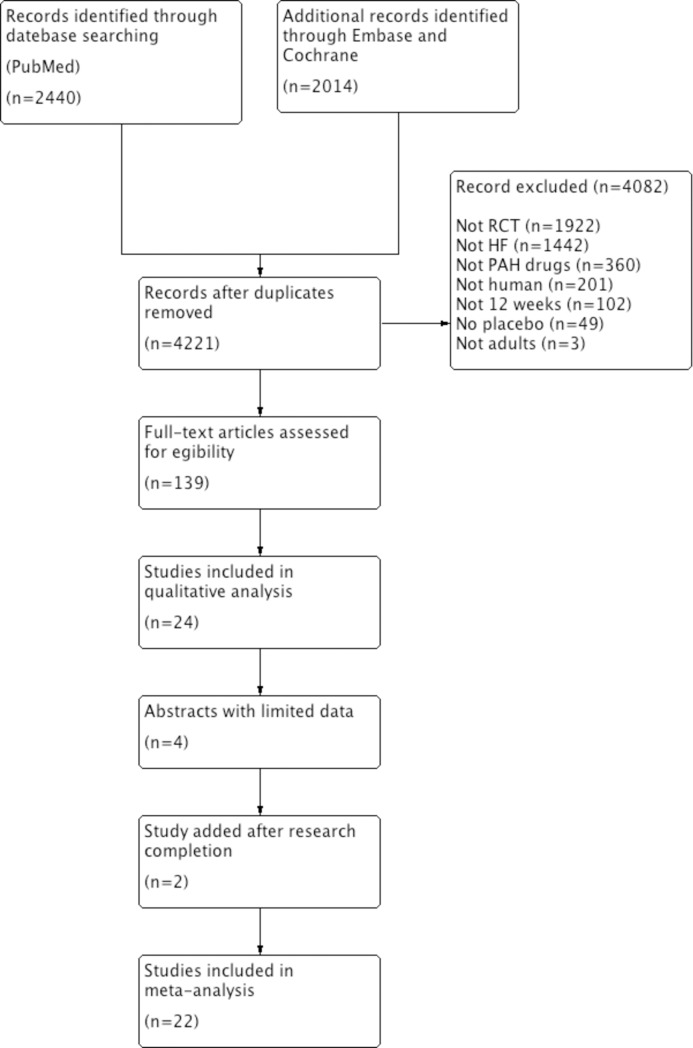
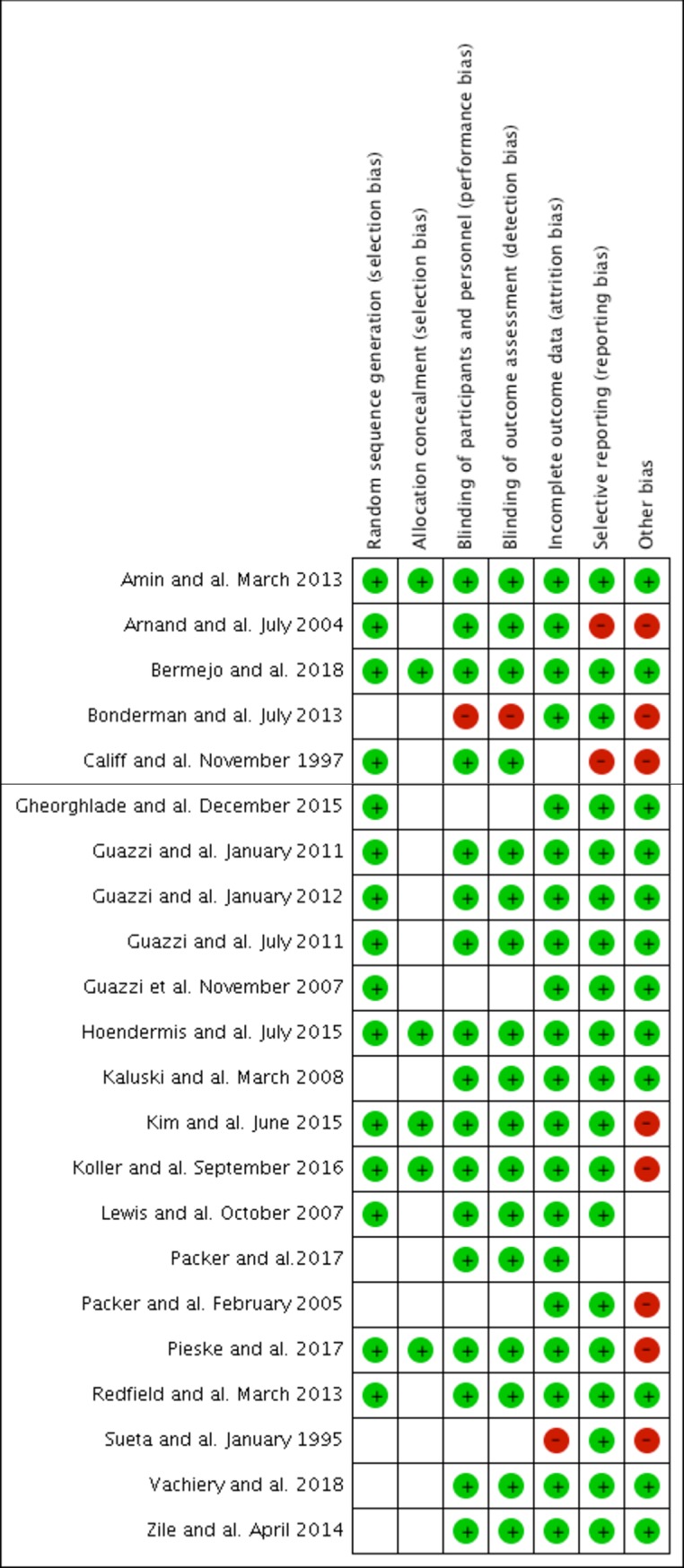
Four thousand one hundred and seventy-five studies were retrieved from our literature search. The primary reviewers included 22 [11, 12, 15, 16, 21, 23–39] separate randomized trials representing 5448 patients (quadratic weighted Kappa:0.82;95%CI:0.71–0.94). The reasons for excluding studies appear in Fig 1. The included publications are described in Table 1. Fifteen studies reported the effects of endothelin receptor antagonists (n = 4)[16, 27, 30, 31], PDE5-inhibitors (n = 8)[11, 12, 23, 25, 26, 32–34], prostanoids (n = 2)[35, 36] and soluble guanylate cyclase stimulators (n = 1)[37] on exercise capacity, whereas 7 other studies [15, 21, 24, 28, 29, 38, 39] were included for the evaluation of secondary endpoints. The duration of the trials ranged from 12 to 52 weeks (median: 22 weeks) and mostly included Caucasian patients with NYHA functional class II or III. Exercise capacity was assessed by 6MWT, VO2 max and treadmill in 9, 5 and 1 studies, respectively. Three studies [15, 26, 34] had a low risk of bias, 8 [27, 28, 31, 33, 35–37, 39] had a high risk and 11 [11, 12, 16, 21, 23–25, 29, 30, 32, 38] had an unknown risk (Fig 2). Lack of information on allocation concealment was the most frequent reason for unknown risk (S1 Table).
Fig 1. Flow chart.
Table 1. Characteristics of included studies.
| Study | n | Length (weeks) |
Baseline therapy (%) | Intervention | Female (%) | NYHA (%) | PH |
HF type (mean EF±SD) | Outcomes | |
|---|---|---|---|---|---|---|---|---|---|---|
| Phosphodiesterase-5 inhibitors | ||||||||||
| Lewis, 2007 [12] | 34 | 12 | ACE-1/ARB (83), Diuretics (100), BB (97), MRA (53), Digitalis (68), ICD (86), CRT (29) | Sildenafil 25 to 75 mg TID | 15 | II (53) III (38) IV (9) |
100% P: 33±3 T: 30±2 (mPAP) |
HFrEF (<40%) P: 20±2 T: 19±2 |
Primary: VO2 max Secondary: 6MWD, hemodynamics, QOL, biomarkers | |
| Guazzi, 2007 [11] | 46 | 26 | IECA (80), ARB (17), Furosemide (67), BB (65), MRA (52), Digitalis (26) | Sildenafil 50 mg BID | 0 | II III |
N/A P: 32±3 T: 34±3 (sPAP) |
HFrEF (<45%) P: 32±3 T: 31±3 |
VO2max, brachial artery FMD, ergoreflex, QOL | |
| Guazzi, Jan 2011 [25] | 45 | 52 | ACE-I (87), ARB (24), BB (84), MRA (42), Digitalis (11), CRT (38) | Sildenafil 50 mg TID | 0 | II (42) III (58) |
N/A P: 38±3 T: 38±6 (sPAP) |
HFrEF (<40%) P: 30±4 T: 30±3 |
Acute sildenafil response, cardiac dimension, echocardiographic parameters, NT-proBNP, CPET, QOL | |
| Guazzi, July 2011 [24] | 44 | 52 | ACE-1/ARB (95), Diuretics (77), BB (82), Digitalis (11) | Sildenafil 50 mg TID | 20 | N/A | 100% P: 36±5 T: 39±5 (mPAP) |
HFpEF (>50%) P: 60±6 T: 60±4 |
Hemodynamics, pulmonary function evaluations, echocardiographic parameters, QOL | |
| Guazzi,2012 [23] | 32 | 52 | ACE-I(75), ARB (25), BB (78), MRA (50), Digitalis (9), CRT (53) | Sildenafil 50 mg TID | 0 | III (91) IV (9) |
100% P: 34±3 T: 35±4 (mPAP) |
HFrEF (<45%) P: 28±7 T: 29±8 |
EOB, hemodynamics, VO2 max, QOL | |
| Amin, 2013 [26] | 106 | 12 | ACE-I/ARB (94), Diuretics (99), BB (95), MRA (64), Digitalis (27), CRT (7), ICD (9) | Sildenafil 50 mg 3 times a week | 26 | II (53) III (47) |
N/A | HFrEF (<35%) |
Primary: mean BP and 6MWD Secondary: hospitalization, survival |
|
| Redfield, 2013 [32] | 216 | 24 | ACE-I\ARB (70), Diuretics (86), BB (76), MRA (11) | Sildenafil 60 mg TID | 48 | II (47) III (53) |
N/A P: 43±15 T: 41±14 (sPAP) |
HFpEF (>50%) P: 60±7 T: 60±7 |
Primary: changes in VO2max at 24 weeks Secondary: composite clinical status score (death, hospitalization, MLHFQ), 6MWD at 12 and 24 weeks, peak sildenafil levels and PCGM at 12 and 24 weeks, left ventricular structure, vascular function, PASP, biomarkers, safety |
|
| Kim, 2015 [33] | 41 | 12 | ACE-I/ARB (100), Diuretics (100), BB (85), MRA (37), CRT/ICD (7) | Udenafil 100 mg BID | 32 | II (76) III (24) |
N/A P: 43±12 T: 41±9 (sPAP) |
HFrEF (<40%) P: 29±7 T: 30±6 |
Primary: change in VO2max at 12 weeks Secondary: changes in ventilatory efficiency, LVEF, E/e', LAVI, PASP, NYHA FC at 12 weeks, changes in BNP at 4 and 12 weeks |
|
| Hoendermis, 2015 [34] | 52 | 12 | ACE-I/ARB (75), Diuretics (90), BB (87), MRA (35) | Sildenafil 60 mg TID | 71 | II (21) III (79) |
100% P: 35±7 T: 35±10 (mPAP) |
HFpEF (>45%) P: 58±4 T: 58±4 |
Primary: mPAP Secondary: PAWP, CO, VO2max |
|
| Bermejo, 2017 [15] | 200 | 24 | ACE-I (39), ARB (21), Diuretics (87), BB (48), MRA (42), Digitalis (42), CCB (17) | Sildenafil 40 mg TID | 77 | I (8) II (48) III (42) (WHO) |
100% P: 40±9 T: 38±7 (mPAP) |
N/A |
Primary: composite clinical score (death, HF hospitalisation with diuretics IV, WHO FC, patient’s global assessment) Secondary: Adjusted composite score, all-cause mortality, cardiac mortality, HF hospitalisations, changes in 6MWD, WHO FC, BNP, PASP, stroke volume at 6 months |
|
| Endothelin receptor antagonist | ||||||||||
| Arnand, 2004 [27] | 642 | 24 | ACE-I/ARB (93), Diuretics (91), BB (75), MRA (40) Digitalis (46) |
Darusentan 10, 25, 50, 100 or 300 mg DIE | 18 | II (20) III (79) IV (1) |
N/A | HFrEF (<35%) P: 27±12 T: 26±11 |
Primary: LVESV Secondary: Changes in LVEF, neurohumoral measures, 6MWD, QOL, NYHA class, global assessment, composite clinical status |
|
| Packer, 2005 [28] | 370 | 26 | ACE-I (89), ARB (12), BB (24), Diuretics (100), MRA (14), Digitalis (74) | Bosentan 500 mg BID | 23 | III (69) IV (31) |
N\A | HFrEF (<35%) P: 23± 7 T: 24±6 |
Primary: clinical status Secondary: combined risk of all-cause mortality and worsening HF |
|
| Kaluski, 2008 [29] | 94 | 20 | ACE-I/ARB (99) Furosemid (99) Thiazide diuretic (23), BB (95) MRA (31) |
Bosentan 125 mg BID | 29 | III (83) IV (17) |
100% P: 49±9 T: 52±10 (sPAP) |
HFrEF (<35%) |
Primary: PASP Secondary: CI Exploratory: others echocardiographic parameters |
|
| Zile, 2014 [30] | 192 | 24 | ACE-I/ARB (80), Diuretics (77), BB (64) | Sitaxsentan 100 mg DIE | 63 | II (56) III (44) |
N/A | HFpEF (>50%) P: 60±13 T: 61±12 |
Primary: Changes in TET Secondary: E/e', LVMI, proportion of subjects achieving improvement, no change or worsening in TET, QOL, NYHA, safety |
|
| Koller, 2016 [31] | 20 | 12 | ACE-I (45), ARB (40), BB (65) Furosemid (80), Thiazide diuretics (45) |
Bosentan 125 mg BID | 45 | II III |
100% P: 66±17 T: 61±17 (sPAP) |
HFpEF (>50%) P: 65±7 T: 56±5 |
Primary: 6MWD at 12 weeks Secondary: 6MWD at 24 weeks and NT-proBNP, NYHA, echocardiographic parameters of RV, QOL at 12 and 24 weeks |
|
| Packer, 2017 [21] | 1613 | 36 | ACE-I /ARB (96), BB (51), Loop diuretics (95), MRA (26), Digitalis (58), Nitrates (44), Hydralazine (2), ICD (7) | Bosentan 125 mg BID | 26 | IIIb (91) IV (9) |
N/A | HFrEF (<35%) P: 25±6 T: 25±7 |
Primary: hierarchical clinical composite, all-cause mortality, hospitalization for HF Secondary: all cause mortality |
|
| Vachiery, 2018 [16] | 63 | 12 | ACE-I /ARB (64), BB (68), Loop diuretics (94), MRA (41), Thiazide diuretics (25), CCB (29) | Macitentan 10 mg DIE | 65 | II (24) III (72) |
100% P: 47±11 T: 46±10 (mPAP) |
Both N/A |
Primary: composite of significant fluid retention or worsening in NYHA Exploratory: haemodynamic and echocardiographic parameters, NT-proBNP, 6MWD, HF related hospitalisations |
|
| Soluble guanylate cyclase stimulators | ||||||||||
| Bonderman, 2013 [37] | 201 | 16 | ACE-I (71), ARB (28), Furosemide (94), Thiazide diuretics (16), BB (93), MRA (76), CD (60) | Riociguat 0.5,1 or 2 mg TID | 14 | II (60) III (38) IV (2) |
100% P: 40±1 T: 37±2 (mPAP) |
HFrEF (<40%) P: 27±5 T: 28±9 |
Primary: Changes mPAP Secondary: changes in hemodynamics, echocardiographic parameters Exploratory: composite of incidence of clinical worsening, composite of cardiovascular death and hospitalization, QOL, WHO/NYHA class, 6MWD, NT-proBNP |
|
| Gheorghlade, 2015 [38] | 456 | 12 | ACE-I (61), ARB (23), Diuretics (94), BB (90), MRA (62), ICD (21), CRT-D (7) | Vericiguat 1.25, 2.5, 5 or 10 mg DIE | 20 | I/II (53) III/IV (47) |
N/A | HFrEF (<45%) P: 29±9 T: 30±8 |
Primary: Change in log transformed NT-proBNP Exploratory: changes in LVEF, LVEDV, LVESV), clinical events, BP, HR, biomarker levels |
|
| Pieske, 2016 [39] | 477 | 12 | ACE-I (40), ARB (34), Diuretics (92), BB (80), MRA (37), CCB (36) |
Vericiguat 1.25, 2.5, 5 or 10 mg DIE | 48 | II (55) III/IV (45) |
N/A | HFpEF (>45%) P: 57± 7 T: 58±6 |
Primary: Change in log transformed NT-proBNP, change in left atrial volume Exploratory: KCCQ, EQ-5D, mortality, morbidity, echocardiography at rest, |
|
| Prostanoids | ||||||||||
| Sueta, 1995 [36] | 33 | 12 | ACE-I (85), Diuretics (100), Digoxin (91) | Max tolerated epoprostenol infusion | 12 | III (36) IV (64) |
N/A | HFrEF (<30%) P: 17±7 T: 17±7 |
6MWD, LVEF, NYHA, hemodynamics | |
| Califf, 1997 [35] | 471 | 36 | ACE-I (84), Diuretics (98), BB (0), Digitalis (91) | Max tolerated epoprostenol infusion | 24 | III (41) IV (59) |
N/A P: 40±9 T: 38±10 (mPAP) |
HFrEF (<25%) P: 18±6 T: 17±6 |
Primary: time until death Secondary: clinical events, exercise capacity, QOL, resource use |
|
n; numbers of study, NYHA; New York Heart Association, PH; pulmonary hypertension, LVEF; left ventricular ejection fraction, ACE-I; angiotensin convertor enzyme inhibitor, ARB; angiotensin receptor blocker, BB; beta-blocker MRA; mineralocorticoid receptor antagonist; REF; reduced ejection fraction, LVESV; left ventricular end systolic volume, 6MWD; 6 minute walking distance, QOL; quality of life, 6MWT; 6 meters walking test, HF; heart failure, PASP; pulmonary arterial systolic pressure, CI; cardiac index, PEF; preserved ejection fraction, TET; treadmill exercise time, E/e'; early diastolic mitral inflow velocity to early diastolic mitral annular velocity, LVMI; left ventricular mass index, NT-proBNP; N-terminal prohormone of brain natriuretic peptide, RV; right ventricule, ICD; implantable cardioverter defibrillator, CRT; cardiac resynchronisation therapy, P; placebo, T; treatment, VO2max; peak oxygen uptake, FMD; flow mediated dilatation, CPET; cardiopulmonary exercise testing, EOB; exercise oscillatory breathing, BP; blood pressure, MLHFQ; Minnesota Living With Heart Failure Questionnaire, PCGM; plasma cyclic guanosine phosphate, LAVI; left atrial volume, BNP; brain natriuretic peptide, mPAP; mean pulmonary arterial pressure, PAWP; pulmonary arterial wedge pressure, CO; cardiac output, WHO; World Health Organization, LVEDV; left ventricular end-diastolic volume, LVESV; left ventricular end-systolic volume, HR; hearth rate, CCB; calcium channel blocker, KCCQ; Kansas City Cardiomyopathy Questionnaire, EQ-5D; EuroQol-5 dimension.
Fig 2. Summary of risk of bias analysis.
Primary outcome
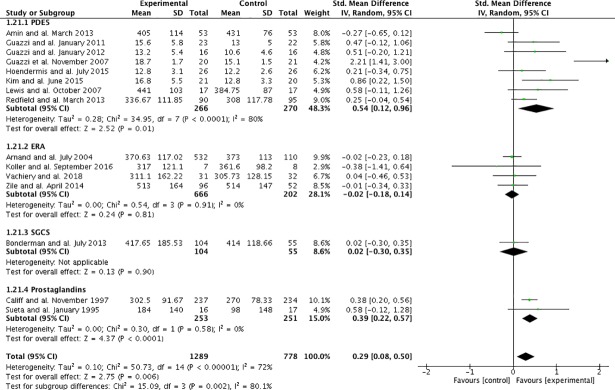
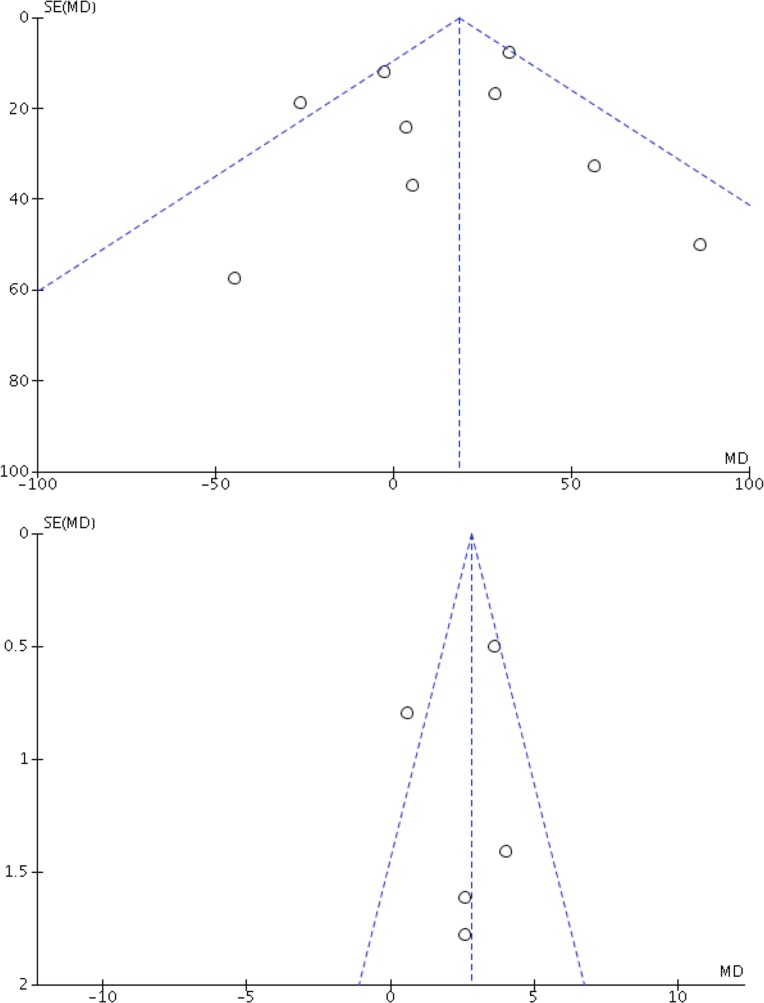
PH-targeted therapies were associated with a significant improvement of exercise capacity (SMD:0.29;95%CI:0.08–0.50, p = 0.006) (Fig 3). The visual analysis of the funnel plots suggested no publication bias (Fig 4). Statistically significant heterogeneity was noted (I2 = 72%;phomogeneity<0.001). Pre-specified subgroup analyses found that this improvement was observed in studies evaluating PDE5-inhibitors (pinteraction = 0.01) and prostanoids (pinteraction<0.001), as well as studies using VO2 peak as the exercise capacity endpoint (pinteraction<0.001), studies of longer duration (pinteraction<0.001) and studies evaluating PH-targeted therapies for HFrEF (pinteraction = 0.005) (Table 2). Overall, the mean increase in VO2 peak was +2.62ml/kg/min (95%CI; 1.08–4.16, p<0.001), whereas no changes in 6MWD were observed (MD +14; 95%CI:-6-34 meters, p = 0.16). Statistical results from fixed-effect suggested similar confidence intervals results except for changes in 6MWD (MD +18; 95%CI: 8–29 meters, p<0.001). These results were similar when studies with a high risk of bias were excluded (S1 Fig, S2 Table), although all studies evaluating prostanoids were at high risk of bias.
Fig 3. Forest plot of primary outcome.
Fig 4.
Study of publication bias: funnel plot for the primary outcome of the meta-analysis, including (A) the 6MWT and; (B) VO2peak.
Table 2. Primary outcome and prespecified subgroups analysis.
| Outcomes | All studies | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| n | References |
Random effect model | Fixed effects model | Homogeneity | |||||
| SMD | 95% CI (p value) | SMD | 95% CI (p value) | P value | I2 (%) | ||||
| Primary outcome | |||||||||
| Exercise capacity | 15 |
[11, 12, 16, 23, 25–27, 30–37] | 0.29 | 0.08–0.50 (p = 0.006) | 0.21 | 0.11–0.30 (p<0.001) | <0.001 | 72 | |
| Type of exercise test | |||||||||
| 6MWT | 9 | [12, 16, 26, 27, 31, 32, 35–37] | MD: 14.1 | -5.6–33.7 (p = 0.16) | MD: 18.4 | 7.8–29.1 (p<0.001) | 0.03 | 53 | |
| VO2 max | 5 | [11, 23, 25, 33, 34] | MD: 2.62 | 1.08–4.16 (p<0.001) | MD: 2.82 |
2.1–3.6 (p<0.001) | 0.03 | 64 | |
| Treadmill | 1 | [30] | MD: -1.0 | -52.7–50.7 (p = 0.97) | MD: -1.0 | -52.7–50.7 (p = 0.97) | N/A | N/A | |
| Classes of PH-targeted therapies | |||||||||
| PDE-5i | 8 | [11, 12, 23, 25, 26, 32–34] | 0.54 | 0.12–0.96 (p = 0.01) | 0.33 | 0.16–0.51 (p<0.001) | <0.001 | 80 | |
| ERA | 4 | [16, 27, 30, 31] | -0.02 |
-0.18–0.14 (p = 0.81) | -0.02 |
-0.18–0.14 (p = 0.81) | 0.91 | 0 | |
| sGC stimulators | 1 | [37] | 0.02 | -0.30–0.35 (p = 0.90) | 0.02 | -0.30–0.35 (p = 0.90) | N/A | N/A | |
| Prostanoids | 2 | [35, 36] | 0.39 | 0.22–0.57 (p<0.001) | 0.39 | 0.22–0.57 (p<0.001) | 0.58 | 0 | |
| Pulmonary hypertension | |||||||||
| With PH | 7 | [12, 16, 23, 31, 34, 35, 37] | 0.25 | 0.07–0.42 (p = 0.006) | 0.28 | 0.14–0.41 (p<0.001) | 0.30 | 17 | |
| Without PH/Unknown | 8 | [11, 25–27, 30, 32, 33, 36] | 0.40 | 0.05–0.75 (p = 0.03) | 0.15 | 0.02–0.27 (p = 0.03) | <0.001 | 83 | |
| Left ventricular ejection fraction | |||||||||
| Preserved | 4 | [30–32, 34] | 0.13 | -0.07–0.33 (p = 0.20) | 0.13 | -0.07–0.33 (p = 0.20) | 0.51 | 0 | |
| Reduced | 10 | [11, 12, 23, 25–27, 33, 35–37] | 0.43 | 0.13–0.72 (p = 0.005) |
0.24 | 0.23–0.35 (p<0.001) | <0.001 | 81 | |
| Both | 1 | [16] | 0.04 | -0.46–0.53 (p = 0.89) |
0.04 | -0.46–0.53 (p = 0.89) | N/A | N/A | |
| Study duration | |||||||||
| ≤ 6 months | 8 | [12, 16, 26, 31, 33, 34, 36, 37] | 0.17 | -0.10–0.44 (p = 0.21) |
0.11 | -0.07–0.29 (p = 0.24) | 0.05 | 49 | |
| > 6 months | 7 | [11, 23, 25, 27, 30, 32, 35] | 0.40 | 0.09–0.72 (p = 0.01) |
0.24 | 0.13–0.35 (p<0.001) | <0.001 | 83 | |
| NYHA functional class | |||||||||
| Up to II | 0 | N/A | N/A | N/A | N/A | N/A | N/A | N/A | |
| Up to III | 8 | [11, 16, 25, 26, 30, 31, 33, 34] | 0.36 | -0.09–0.81 (p = 0.11) |
0.18 | 0.00–0.36 (p = 0.05) | <0.001 | 82 | |
| Up to IV | 7 | [12, 23, 27, 32, 35–37] | 0.24 | 0.05–0.42 (p = 0.01) |
0.22 | 0.11–0.33 (p<0.001) | 0.05 | 51 | |
Secondary outcomes
PH-targeted therapies had no effects on other patients-centered outcomes, except for a significant increase in treatment discontinuation compared to standard treatment (Table 3). No differences were noted on safety outcome when comparing studies including only PH patients and studies including patients without PH. However, sensitivity analyses suggested a possible increase in all-cause mortality (RR 1.26; 95%CI:1.04–1.53, p = 0.02, I2 = 0% and phomogeneity = 0.70) when the ENABLE study [21] was excluded. Systolic pulmonary artery pressure (sPAP) measured by echocardiography was decreased by PH-targeted drugs and NT-proBNP were not influenced by PH-targeted therapies. However, statistically significant heterogeneity was observed for both measures. Subgroup analyses suggested that decreases in sPAP were driven by studies evaluating PDE5-inhibitors (pinteraction = 0.004), whereas decreases in NT-proBNP levels were observed in studies that included patients with HFrEF only (pinteraction = 0.02) (S4 Table). These results were similar when studies with a high risk of bias were excluded (S3 and S4 Tables). Given that PDE5-inhibitors were associated with significant improvements in exercise capacity and sPAP, exploratory analyses were performed and suggested that PDE5-inhibitors were not associated with improvements in patients-centered outcomes. (S5 Table).
Table 3. Secondary outcomes.
| Outcomes | All studies | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| n | References | Proportion of events (%) | Random effect model | Fixed effects model | Homogeneity | ||||
| RR | 95% CI (p value) | RR | 95% CI (p value) | P value | I2 (%) | ||||
| Patient centered secondary outcomes | |||||||||
| All-cause mortality | 22 | [11, 12, 15, 16, 21, 23–39] | (T) 357/3356 (10.6) (P) 289/2080(13.9) Total: 646/5436(11.9) |
1.09 | 0.92–1.29 (p = 0.32) |
1.07 |
0.93–1.23 (p = 0.34) |
0.39 | 6 |
| Cardiac mortality | 20 | [11, 12, 15, 16, 23–38] | (T) 20/1022 (2.0) (P) 12/587 (2.0) Total: 32/1609 (2.0) |
0.86 | 0.41–1.80 (p = 0.69) | 0.89 | 0.45–1.75 (p = 0.73) |
0.65 | 0 |
| All-cause hospitalization | 10 | [11, 12, 15, 23, 25, 27, 28, 31–33, 38] | (T) 180/1362 (13.2) (P) 86/538 (16.0) Total: 266/1900(14.0) |
0.85 | 0.64–1.12 (p = 0.25) | 0.85 | 0.66–1.10 (p = 18) |
0.38 | 7 |
| Cardiac Hospitalization | 13 | [11, 12, 15, 16, 21, 23, 25, 27, 28, 31, 33, 37, 38] | (T) 366/2289 (16) (P) 242/1407 (17.2) Total: 608/3696(16.5) |
0.97 | 0.74–1.27 (p = 0.81) | 1.01 | 0.87–1.18 (= 0.88) |
0.08 | 41 |
| Treatment discontinuation | 22 | [11, 12, 15, 16, 21, 23–39] | (T) 544/3120 (17.4) (P) 270/1848 (14.6) Total: 814/4968 (16.4) |
1.31 | 1.15–1.50 (p<0.001) | 1.34 | 1.17–1.54 (p<0.001) |
0.84 | 0 |
| Hemodynamic outcomes | |||||||||
| Outcomes | n | References | Random effect model | Fixed effects model | Homogeneity | ||||
| MD | 95% CI (p value) | MD | 95% CI (p value) | P value | I2 (%) | ||||
| sPAP | 7 | [11, 24, 25, 29, 31–33] | N/A | -7.5 | -14.9, -0.1 (p = 0.05) |
-12.4 |
-13.5, -11.3 (p<0.001) | <0.001 | 97 |
| NT-proBNP | 10 | [12, 25, 27, 31–34, 37–39] | N/A | -240 | -578, -98 (p = 0.16) |
-214 |
-324, -104 (p<0.001) | <0.001 | 78 |
Discussion
The present systematic review with meta-analysis documented that PH-targeted therapies may modestly improve exercise capacity in patients with HFpEF/HFrEF. These findings were similar when only studies with a low or an unknown risk of bias were taken into consideration and when the fixed-effects model was used, substantiating the robustness of these results. However, significant heterogeneity was noted and predefined subgroup analyses suggested that this observation was driven by studies 1) of longer duration; 2) evaluating the effects of PDE5-inhibitors or prostanoids; 3) using VO2 peak as the evaluative modality and; 4) recruiting patients with HFrEF. Intriguingly, the presence of PH did not influence the primary outcome. PDE5-inhibitors were also associated with a significant decrease in sPAP and exploratory analyses suggested they might be associated with decreased cardiac-specific hospitalizations in heart failure patient but not in patients with corrected valvulopathy and persistent PH. Importantly, however, these results should be cautiously tempered by the fact that PH-targeted therapies were associated with an increase in treatment discontinuation, that most studies had high or unknown risk of bias and that sensitivity analyses suggested that an increased mortality with PH-targeted therapies cannot be ruled out.
In addition to the passive elevation of blood pressure within the pulmonary circulation, HFpEF/HFrEF has long been recognized to promote venular remodeling, and in some instances, arteriolar remodeling with various combinations of medial hypertrophy, intimal proliferation, adventitial thickening, microthrombi and more rarely fibrinoid necrosis[40]. These histological abnormalities were compatible with clinical and hemodynamic demonstration of a “precapillary component”, either reactive or fixed, of PH-LHD[9]. A hemodynamic definition of these phenotypes has been tentatively proposed, including “isolated post-capillary PH” and “combined post-capillary and pre-capillary PH”, according to a diastolic pressure difference (defined as diastolic PAP minus mean PAWP) <7 mmHg or ≥7 mmHg, respectively[9]. Although the precise characteristics defining this “out-of-proportion PH” and their pathobiological consequences remain controversial [6, 7], the presence of PH-LHD identifies a subgroup of HFpEF [2] and HFrEF [41, 42] at high risk of morbidity and mortality[8]. Interestingly, PH-LHD with a significant precapillary component shares many pathobiological features with PAH. The utilization of pulmonary vasoactive therapies in these patients was thus appealing. As such, numerous randomized controlled trials assessed the effects of these therapies in HFpEF/HFrEF. However, most studies were characterized by a small sample size and many had conflicting results. Some studies even resulted in increased mortality [35] and morbidity events[28].
The heterogeneity in the scales used to assess exercise capacity in included studies led to the use of a SMD for pooled data. SMD can be hard to interpret because the overall intervention effect is not reported with traditional scales. Rules of thumbs have been suggested to interpret effect sizes: 0.2 being a small effect, 0.5 a moderate one and 0.8 a large one[43]. Using this scale, the observed effect size represents a small-to-moderate effect of intervention on exercise capacity. Subgroup analyses suggested that this effect was driven by studies evaluating PDE5-inhibitors and prostanoids. Interestingly, these studies predominantly used the VO2 peak to assess the effects of PH-targeted therapies on exercise capacity, compared to other trials that used the 6MWT. The increase in VO2 peak was similar to the one observed in a previous meta-analysis including shorter trials[44]. The 6MWT and CPET have excellent discriminative properties and have been reported to closely correlate with functional class, disease severity and survival in HFrEF/HFpEF[45]. However, the responsiveness of the 6MWT to detect effective interventions in heart failure studies has been questioned[46]. Therefore, the possibility that other drug classes did not result in improved exercise as a result of the limited evaluative properties of the 6MWT in HFpEF/HFrEF patients cannot be excluded. Our meta-analysis also demonstrated that PH-targeted therapy is associated with a significant decrease in sPAP. This is of clinical interest knowing that elevated sPAP strongly predicts mortality [3], yet a reduction in sPAP is not a surrogate marker for improved survival.
Importantly, PH-targeted therapies were associated with a higher risk of treatment discontinuation. Moreover, sensitivity analyses suggested that an increase in all-cause mortality (RR 1.26; 95%CI:1.04–1.53, p = 0.02, I2 = 0% and phomogeneity = 0.70) could not be ruled out with PH-targeted therapies when the ENABLE trial was excluded, despite the fact that this trial was characterized by an increased risk of early worsening in HF necessitating hospitalisation[21]. Consistently, sildenafil increased the risk major clinical events (including hospitalizations) compared to placebo in patients with persistent PH following valvular heart surgery, whereas macitentan almost doubled the incidence of significant fluid retention in PH-LHD in recently published trials[15, 16]. Past observational studies also suggested an increase in morbidity/mortality with the use of other endothelin receptor antagonists [28] or prostanoids [35, 36]. These observations were speculatively explained by cardiac decompensation due to an increasing pulmonary blood flow in the presence of an already elevated left-sided filling pressure and fluid retention[28]. This finding thus supports previous concerns about PH-targeted therapies in HFpEF/HFrEF patients, including PDE5-inhibitors[47]. Conversely, a recent meta-analysis on PDE5-inhibitors in HFrEF/HFpEF observed a reduction of the composite of death or hospitalisation [48, 49], whereas another meta-analysis could not find significant changes in mortality with endothelin receptor antagonists[50]. However, these meta-analyses mainly included trials of short duration. Moreover, when taking into account our results, the significant reduction of this composite outcome observed in PDE5-inhibitors were likely driven by the reduction of hospitalization rather than mortality. Most importantly, recent trials confirmed the frequent disconnection between exercise capacity and the incidence of hard events (e.g. hospitalisations) in PH-LHD [15], underscoring the need of using clinical outcomes (e.g. death or hospitalisation) rather than surrogate outcomes (e.g. exercise capacity or sPAP) in future trials in the field. Taken together, these observation support current recommendations, stipulating that there is currently insufficient evidence supporting the use of PH-targeted therapies in PH-LHD[51].
The present study has several limitations, including the variability in the scale used to assess exercise capacity and in trial duration (ranging from 12 to 52 weeks), the protracted period between the publication of the first and the last trials (around 20 years) and the impossibility to include 4 studies due to unpublished data. The fact that we had to transform median to average and BNP to NT-proBNP using an already published formula is another limitation of our meta-analysis. In addition, most of the studies included in our meta-analysis had methodological flaws with many studies lacking information about key aspects of the methodology, resulting in an unknown or a high risk of bias. Yet, sensitivity analyses excluding studies with high risk of bias resulted in similar improvements suggesting that our results are not explained by the risk of bias. While our subgroup analyses of aggregate data could not find significant differences in exercise capacity, mortality or other clinically important outcomes between these categories of patients, it remains unknown whether meta-analysis of patient-level data would have delineated patterns or specific clinical syndromes predicting treatment responders. Most importantly, the lack of homogeneity in the study population is a major issue. Indeed, the type of HF and the severity of PH were highly variable from one study to the other, and many studies did not even describe whether patients had concomitant PH[11, 21, 25–28, 30, 32, 33, 35, 36, 38, 39]. When assessed, pulmonary hemodynamics were most commonly estimated non-invasively, precluding precise characterization of the PH. These inclusion criteria are somewhat surprising since pulmonary vasoactive therapies could be expected to be mostly effective in patients with a precapillary component of PH. In addition, the prevalence of additional left-sided valvular disease, particularly functional mitral regurgitation, was generally not reported. Finally, whether HF therapy was fully optimized before randomization was generally poorly described. Thus, robust evidence on the safety and efficacy of PH-targeted therapies will require long-term multicentre randomized, controlled trials of hemodynamically phenotyped patients that are clinically stable on optimized background therapy to allow delineating subgroups of patients whom benefit most from PH-targeted therapies[52].
In conclusion, the use of PH-targeted therapies may modestly improve exercise capacity in patients with HFpEF/HFrEF. Subgroup analyses suggested this effect was mostly driven by the use of PDE5-inhibitors. However, PH-targeted therapies were associated with a significant increase in treatment discontinuation and an excess of mortality could not be entirely ruled out. Moreover, most studies were at high or unknown risk of bias and patients’ phenotypic description precluded the delineation a subgroup of patients that could benefit from PH-targeted therapies. Although the potential use of PH-targeted therapies in PH associated with HFpEF/HFrEF is based on a sound pathobiological rationale, these observations do not provide evidence to support the use of these drugs in the clinical management of patients until future trials provide stronger evidence of safety and efficacy.
Supporting information
(DOCX)
(DOC)
(TIF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
We’d like to thank Caroline Lambert for her continuous help.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
The authors received no specific funding for this work.
References
- 1.Simonneau G, Gatzoulis MA, Adatia I, Celermajer D, Denton C, Ghofrani A, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2013;62(25 Suppl):D34–41. 10.1016/j.jacc.2013.10.029 . [DOI] [PubMed] [Google Scholar]
- 2.Lam CS, Roger VL, Rodeheffer RJ, Borlaug BA, Enders FT, Redfield MM. Pulmonary hypertension in heart failure with preserved ejection fraction: a community-based study. J Am Coll Cardiol. 2009;53(13):1119–26. Epub 2009/03/28. 10.1016/j.jacc.2008.11.051 ; PubMed Central PMCID: PMCPMC2736110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bursi F, McNallan SM, Redfield MM, Nkomo VT, Lam CS, Weston SA, et al. Pulmonary pressures and death in heart failure: a community study. J Am Coll Cardiol. 2012;59(3):222–31. Epub 2012/01/14. 10.1016/j.jacc.2011.06.076 ; PubMed Central PMCID: PMCPMC3258551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guazzi M, Borlaug BA. Pulmonary hypertension due to left heart disease. Circulation. 2012;126(8):975–90. 10.1161/CIRCULATIONAHA.111.085761 . [DOI] [PubMed] [Google Scholar]
- 5.Naeije R, Vachiery JL, Yerly P, Vanderpool R. The transpulmonary pressure gradient for the diagnosis of pulmonary vascular disease. Eur Respir J. 2013;41(1):217–23. 10.1183/09031936.00074312 . [DOI] [PubMed] [Google Scholar]
- 6.Palazzini M, Dardi F, Manes A, Bacchi Reggiani ML, Gotti E, Rinaldi A, et al. Pulmonary hypertension due to left heart disease: analysis of survival according to the haemodynamic classification of the 2015 ESC/ERS guidelines and insights for future changes. Eur J Heart Fail. 2017. 10.1002/ejhf.860 . [DOI] [PubMed] [Google Scholar]
- 7.Guazzi M, Naeije R. Pulmonary Hypertension in Heart Failure: Pathophysiology, Pathobiology, and Emerging Clinical Perspectives. J Am Coll Cardiol. 2017;69(13):1718–34. Epub 2017/04/01. 10.1016/j.jacc.2017.01.051 . [DOI] [PubMed] [Google Scholar]
- 8.Gerges C, Gerges M, Lang MB, Zhang Y, Jakowitsch J, Probst P, et al. Diastolic pulmonary vascular pressure gradient: a predictor of prognosis in "out-of-proportion" pulmonary hypertension. Chest. 2013;143(3):758–66. Epub 2013/04/13. 10.1378/chest.12-1653 . [DOI] [PubMed] [Google Scholar]
- 9.Vachiery JL, Adir Y, Barbera JA, Champion H, Coghlan JG, Cottin V, et al. Pulmonary hypertension due to left heart diseases. J Am Coll Cardiol. 2013;62(25 Suppl):D100–8. 10.1016/j.jacc.2013.10.033 . [DOI] [PubMed] [Google Scholar]
- 10.Lajoie AC, Lauziere G, Lega JC, Lacasse Y, Martin S, Simard S, et al. Combination therapy versus monotherapy for pulmonary arterial hypertension: a meta-analysis. Lancet Respir Med. 2016;4(4):291–305. Epub 2016/03/05. 10.1016/S2213-2600(16)00027-8 . [DOI] [PubMed] [Google Scholar]
- 11.Guazzi M, Samaja M, Arena R, Vicenzi M, Guazzi MD. Long-term use of sildenafil in the therapeutic management of heart failure. J Am Coll Cardiol. 2007;50(22):2136–44. 10.1016/j.jacc.2007.07.078 . [DOI] [PubMed] [Google Scholar]
- 12.Lewis GD, Shah R, Shahzad K, Camuso JM, Pappagianopoulos PP, Hung J, et al. Sildenafil improves exercise capacity and quality of life in patients with systolic heart failure and secondary pulmonary hypertension. Circulation. 2007;116(14):1555–62. 10.1161/CIRCULATIONAHA.107.716373 . [DOI] [PubMed] [Google Scholar]
- 13.Trammell AW, Pugh ME, Newman JH, Hemnes AR, Robbins IM. Use of pulmonary arterial hypertension-approved therapy in the treatment of non-group 1 pulmonary hypertension at US referral centers. Pulm Circ. 2015;5(2):356–63. Epub 2015/06/13. 10.1086/681264 ; PubMed Central PMCID: PMCPMC4449248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Higgins JPT, Green S, Cochrane Collaboration. Cochrane handbook for systematic reviews of interventions. Chichester, England; Hoboken, NJ: Wiley-Blackwell; 2008. xxi, 649 p. p. [Google Scholar]
- 15.Bermejo J, Yotti R, Garcia-Orta R, Sanchez-Fernandez PL, Castano M, Segovia-Cubero J, et al. Sildenafil for improving outcomes in patients with corrected valvular heart disease and persistent pulmonary hypertension: a multicenter, double-blind, randomized clinical trial. Eur Heart J. 2018;39(15):1255–64. Epub 2017/12/28. 10.1093/eurheartj/ehx700 ; PubMed Central PMCID: PMCPMC5905634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vachiery JL, Delcroix M, Al-Hiti H, Efficace M, Hutyra M, Lack G, et al. Macitentan in pulmonary hypertension due to left ventricular dysfunction. Eur Respir J. 2018;51(2). Epub 2018/02/14. . [DOI] [PubMed] [Google Scholar]
- 17.Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928 10.1136/bmj.d5928 ; PubMed Central PMCID: PMCPMC3196245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Poole C, Greenland S. Random-effects meta-analyses are not always conservative. Am J Epidemiol. 1999;150(5):469–75. Epub 1999/09/03. . [DOI] [PubMed] [Google Scholar]
- 19.Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135 10.1186/1471-2288-14-135 ; PubMed Central PMCID: PMCPMC4383202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Alibay Y, Schmitt C, Beauchet A, Dubourg O, Alexandre JA, Boileau C, et al. [Non-radioimmunometric NT-ProBNP and BNP assays: impact of diluent, age, gender, BMI]. Ann Biol Clin (Paris). 2005;63(1):43–9. . [PubMed] [Google Scholar]
- 21.Packer M, McMurray JJV, Krum H, Kiowski W, Massie BM, Caspi A, et al. Long-Term Effect of Endothelin Receptor Antagonism With Bosentan on the Morbidity and Mortality of Patients With Severe Chronic Heart Failure: Primary Results of the ENABLE Trials. JACC Heart Fail. 2017;5(5):317–26. Epub 2017/04/30. 10.1016/j.jchf.2017.02.021 . [DOI] [PubMed] [Google Scholar]
- 22.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med. 2009;151(4):264–9, W64. Epub 2009/07/23. . [DOI] [PubMed] [Google Scholar]
- 23.Guazzi M, Vicenzi M, Arena R. Phosphodiesterase 5 inhibition with sildenafil reverses exercise oscillatory breathing in chronic heart failure: a long-term cardiopulmonary exercise testing placebo-controlled study. Eur J Heart Fail. 2012;14(1):82–90. 10.1093/eurjhf/hfr147 . [DOI] [PubMed] [Google Scholar]
- 24.Guazzi M, Vicenzi M, Arena R, Guazzi MD. Pulmonary hypertension in heart failure with preserved ejection fraction: a target of phosphodiesterase-5 inhibition in a 1-year study. Circulation. 2011;124(2):164–74. 10.1161/CIRCULATIONAHA.110.983866 . [DOI] [PubMed] [Google Scholar]
- 25.Guazzi M, Vicenzi M, Arena R, Guazzi MD. PDE5 inhibition with sildenafil improves left ventricular diastolic function, cardiac geometry, and clinical status in patients with stable systolic heart failure: results of a 1-year, prospective, randomized, placebo-controlled study. Circ Heart Fail. 2011;4(1):8–17. 10.1161/CIRCHEARTFAILURE.110.944694 . [DOI] [PubMed] [Google Scholar]
- 26.Amin A, Mahmoudi E, Navid H, Chitsazan M. Is chronic sildenafil therapy safe and clinically beneficial in patients with systolic heart failure? Congest Heart Fail. 2013;19(2):99–103. 10.1111/chf.12008 . [DOI] [PubMed] [Google Scholar]
- 27.Anand I, McMurray J, Cohn JN, Konstam MA, Notter T, Quitzau K, et al. Long-term effects of darusentan on left-ventricular remodelling and clinical outcomes in the EndothelinA Receptor Antagonist Trial in Heart Failure (EARTH): randomised, double-blind, placebo-controlled trial. Lancet. 2004;364(9431):347–54. 10.1016/S0140-6736(04)16723-8 . [DOI] [PubMed] [Google Scholar]
- 28.Packer M, McMurray J, Massie BM, Caspi A, Charlon V, Cohen-Solal A, et al. Clinical effects of endothelin receptor antagonism with bosentan in patients with severe chronic heart failure: results of a pilot study. J Card Fail. 2005;11(1):12–20. . [DOI] [PubMed] [Google Scholar]
- 29.Kaluski E, Cotter G, Leitman M, Milo-Cotter O, Krakover R, Kobrin I, et al. Clinical and hemodynamic effects of bosentan dose optimization in symptomatic heart failure patients with severe systolic dysfunction, associated with secondary pulmonary hypertension—a multi-center randomized study. Cardiology. 2008;109(4):273–80. 10.1159/000107791 . [DOI] [PubMed] [Google Scholar]
- 30.Zile MR, Bourge RC, Redfield MM, Zhou D, Baicu CF, Little WC. Randomized, double-blind, placebo-controlled study of sitaxsentan to improve impaired exercise tolerance in patients with heart failure and a preserved ejection fraction. JACC Heart Fail. 2014;2(2):123–30. 10.1016/j.jchf.2013.12.002 . [DOI] [PubMed] [Google Scholar]
- 31.Koller B, Steringer-Mascherbauer R, Ebner CH, Weber T, Ammer M, Eichinger J, et al. Pilot Study of Endothelin Receptor Blockade in Heart Failure with Diastolic Dysfunction and Pulmonary Hypertension (BADDHY-Trial). Heart Lung Circ. 2016. 10.1016/j.hlc.2016.09.004 . [DOI] [PubMed] [Google Scholar]
- 32.Redfield MM, Chen HH, Borlaug BA, Semigran MJ, Lee KL, Lewis G, et al. Effect of phosphodiesterase-5 inhibition on exercise capacity and clinical status in heart failure with preserved ejection fraction: a randomized clinical trial. JAMA. 2013;309(12):1268–77. 10.1001/jama.2013.2024 ; PubMed Central PMCID: PMCPMC3835156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim KH, Kim HK, Hwang IC, Cho HJ, Je N, Kwon OM, et al. PDE 5 inhibition with udenafil improves left ventricular systolic/diastolic functions and exercise capacity in patients with chronic heart failure with reduced ejection fraction; A 12-week, randomized, double-blind, placebo-controlled trial. Am Heart J. 2015;169(6):813–22 e3. 10.1016/j.ahj.2015.03.018 . [DOI] [PubMed] [Google Scholar]
- 34.Hoendermis ES, Liu LC, Hummel YM, van der Meer P, de Boer RA, Berger RM, et al. Effects of sildenafil on invasive haemodynamics and exercise capacity in heart failure patients with preserved ejection fraction and pulmonary hypertension: a randomized controlled trial. Eur Heart J. 2015;36(38):2565–73. 10.1093/eurheartj/ehv336 . [DOI] [PubMed] [Google Scholar]
- 35.Califf RM, Adams KF, McKenna WJ, Gheorghiade M, Uretsky BF, McNulty SE, et al. A randomized controlled trial of epoprostenol therapy for severe congestive heart failure: The Flolan International Randomized Survival Trial (FIRST). Am Heart J. 1997;134(1):44–54. . [DOI] [PubMed] [Google Scholar]
- 36.Sueta CA, Gheorghiade M, Adams KF Jr., Bourge RC, Murali S, Uretsky BF, et al. Safety and efficacy of epoprostenol in patients with severe congestive heart failure. Epoprostenol Multicenter Research Group. Am J Cardiol. 1995;75(3):34A–43A. . [DOI] [PubMed] [Google Scholar]
- 37.Bonderman D, Ghio S, Felix SB, Ghofrani HA, Michelakis E, Mitrovic V, et al. Riociguat for patients with pulmonary hypertension caused by systolic left ventricular dysfunction: a phase IIb double-blind, randomized, placebo-controlled, dose-ranging hemodynamic study. Circulation. 2013;128(5):502–11. 10.1161/CIRCULATIONAHA.113.001458 . [DOI] [PubMed] [Google Scholar]
- 38.Gheorghiade M, Greene SJ, Butler J, Filippatos G, Lam CS, Maggioni AP, et al. Effect of Vericiguat, a Soluble Guanylate Cyclase Stimulator, on Natriuretic Peptide Levels in Patients With Worsening Chronic Heart Failure and Reduced Ejection Fraction: The SOCRATES-REDUCED Randomized Trial. JAMA. 2015;314(21):2251–62. 10.1001/jama.2015.15734 . [DOI] [PubMed] [Google Scholar]
- 39.Pieske B, Maggioni AP, Lam CSP, Pieske-Kraigher E, Filippatos G, Butler J, et al. Vericiguat in patients with worsening chronic heart failure and preserved ejection fraction: results of the SOluble guanylate Cyclase stimulatoR in heArT failurE patientS with PRESERVED EF (SOCRATES-PRESERVED) study. Eur Heart J. 2017;38(15):1119–27. Epub 2017/04/04. 10.1093/eurheartj/ehw593 ; PubMed Central PMCID: PMCPMC5400074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tandon HD, Kasturi J. Pulmonary vascular changes associated with isolated mitral stenosis in India. Br Heart J. 1975;37(1):26–36. Epub 1975/01/01. ; PubMed Central PMCID: PMCPMC484151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Meyer P, Filippatos GS, Ahmed MI, Iskandrian AE, Bittner V, Perry GJ, et al. Effects of right ventricular ejection fraction on outcomes in chronic systolic heart failure. Circulation. 2010;121(2):252–8. Epub 2010/01/06. 10.1161/CIRCULATIONAHA.109.887570 ; PubMed Central PMCID: PMCPMC2877272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Damy T, Goode KM, Kallvikbacka-Bennett A, Lewinter C, Hobkirk J, Nikitin NP, et al. Determinants and prognostic value of pulmonary arterial pressure in patients with chronic heart failure. Eur Heart J. 2010;31(18):2280–90. Epub 2010/08/10. 10.1093/eurheartj/ehq245 . [DOI] [PubMed] [Google Scholar]
- 43.Cohen J. Statistical power analysis for the behavioral sciences. 2nd ed. Hillsdale, N.J.: L. Erlbaum Associates; 1988. xxi, 567 pages p. [Google Scholar]
- 44.Wu X, Yang T, Zhou Q, Li S, Huang L. Additional use of a phosphodiesterase 5 inhibitor in patients with pulmonary hypertension secondary to chronic systolic heart failure: a meta-analysis. Eur J Heart Fail. 2014;16(4):444–53. 10.1002/ejhf.47 . [DOI] [PubMed] [Google Scholar]
- 45.Guazzi M, Bandera F, Ozemek C, Systrom D, Arena R. Cardiopulmonary Exercise Testing: What Is its Value? J Am Coll Cardiol. 2017;70(13):1618–36. Epub 2017/09/25. 10.1016/j.jacc.2017.08.012 . [DOI] [PubMed] [Google Scholar]
- 46.Olsson LG, Swedberg K, Clark AL, Witte KK, Cleland JG. Six minute corridor walk test as an outcome measure for the assessment of treatment in randomized, blinded intervention trials of chronic heart failure: a systematic review. Eur Heart J. 2005;26(8):778–93. Epub 2005/03/19. 10.1093/eurheartj/ehi162 . [DOI] [PubMed] [Google Scholar]
- 47.Varma A, Shah KB, Hess ML. Phosphodiesterase inhibitors, congestive heart failure, and sudden death: time for re-evaluation. Congest Heart Fail. 2012;18(4):229–33. Epub 2012/04/18. 10.1111/j.1751-7133.2012.00293.x . [DOI] [PubMed] [Google Scholar]
- 48.De Vecchis R, Cesaro A, Ariano C, Giasi A, Cioppa C. Phosphodiesterase-5 Inhibitors Improve Clinical Outcomes, Exercise Capacity and Pulmonary Hemodynamics in Patients With Heart Failure With Reduced Left Ventricular Ejection Fraction: A Meta-Analysis. J Clin Med Res. 2017;9(6):488–98. Epub 2017/05/13. doi: 10.14740/jocmr3008w ; PubMed Central PMCID: PMCPMC5412522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hwang IC, Kim YJ, Park JB, Yoon YE, Lee SP, Kim HK, et al. Pulmonary hemodynamics and effects of phosphodiesterase type 5 inhibition in heart failure: a meta-analysis of randomized trials. BMC Cardiovasc Disord. 2017;17(1):150 Epub 2017/06/14. 10.1186/s12872-017-0576-4 ; PubMed Central PMCID: PMCPMC5468951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Xiong B, Nie D, Cao Y, Zou Y, Yao Y, Tan J, et al. Clinical and Hemodynamic Effects of Endothelin Receptor Antagonists in Patients With Heart Failure. Int Heart J. 2017;58(3):400–8. Epub 2017/05/26. 10.1536/ihj.16-307 . [DOI] [PubMed] [Google Scholar]
- 51.Galie N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, et al. 2015 ESC/ERS Guidelines for the Diagnosis and Treatment of Pulmonary Hypertension. Rev Esp Cardiol (Engl Ed). 2016;69(2):177 10.1016/j.rec.2016.01.002 . [DOI] [PubMed] [Google Scholar]
- 52.Hoeper MM, Lam CSP, Vachiery JL, Bauersachs J, Gerges C, Lang IM, et al. Pulmonary hypertension in heart failure with preserved ejection fraction: a plea for proper phenotyping and further research. Eur Heart J. 2017;38(38):2869–73. Epub 2016/12/25. 10.1093/eurheartj/ehw597 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOC)
(TIF)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.