Abstract
Context:
Contrast baths (CB) is a thermal treatment modality used in sports medicine, athletic training, and rehabilitation settings. Proposed physiological effects of CB include increasing tissue blood flow and oxygenation and decreasing tissue swelling and edema to promote better healing, improved limb function, and quicker recovery.
Objective:
To investigate the physiological effects of CB on the intramuscular hemodynamics and oxygenation of the lower leg muscles using near-infrared spectroscopy (NIRS), an optical method for monitoring changes in tissue oxygenated (O2Hb), deoxygenated (HHb), and total hemoglobin (tHb) as well as tissue oxygen saturation index (TSI%).
Design:
Descriptive laboratory study.
Patients or Other Participants:
Ten healthy men and women with a mean age of 29 (range = 17 ± 42) years, mean body mass index of 24.6 ± 3.2, and mean adipose tissue thickness of 6.4 ± 2.2 mm.
Intervention(s):
Conventional CB (10-minute baseline, 4 : 1-minute hot : cold ratio) was applied to the left lower leg.
Main Outcome Measure(s):
Changes in chromophore concentrations of O2Hb, HHb, tHb, and TSI% of the gastrocnemius muscle were monitored during 10 minutes of baseline measurement, a 30-minute CB protocol, and 10 minutes of recovery using a spatially resolved NIRS.
Results:
After a 30-minute CB protocol, increases (P < .05) in tissue O2Hb (7.4 ± 4 μM), tHb (7.6 ± 6.1 μM), and TSI% (3.1% ± 2.3%) were observed as compared with baseline measures.
Conclusions:
Application of CB induced a transient change in the hemodynamics and oxygenation of the gastrocnemius muscle in healthy individuals. The effect of CB application in improving tissue hemodynamics and oxygenation may, therefore, support the therapeutic benefits of CB in the treatment of muscle injuries.
Key Words: thermal modalities, muscle injuries, NIRS, blood flow, cryotherapy
Key Points
Contrast baths (CB) is a thermal intervention commonly used by clinicians, athletic trainers, physiotherapists, and athletes to reduce upper and lower limb inflammation, swelling, and pain and improve posttraining recovery.
By increasing the level of intramuscular oxygenated blood volume, a 30-minute protocol of CB improved intramuscular hemodynamics and oxygenation in healthy people.
Further research is required to support potential therapeutic applications of CB in people with acute and chronic muscle damage and soreness.
Near-infrared spectroscopy is a noninvasive optical method that can be used to study intramuscular hemodynamics and oxygenation.
Contrast baths (CB) is a thermal treatment modality used to reduce upper and lower limb pain, soft tissue inflammation and edema, and muscle spasm and joint stiffness at subacute phases and to facilitate recovery from training.1,2 It is an accessible, safe, noninvasive, simple, and inexpensive modality with a long history in musculoskeletal therapy.1 Besides its application in rehabilitation and musculoskeletal medicine, CB is commonly used by athletes to promote recovery and reduce muscle damage and the pain associated with delayed-onset muscle soreness (DOMS).3–7
Contrast baths describes the repeated immersion of a limb in hot and then cold water for a specified timing, duration, and temperature. It is thought that alternating the application of hot and cold water causes intermittent vasoconstriction and vasodilation that induce a vascular pumping effect, which results in increased tissue blood flow and oxygenation that improve healing, enhances tissue waste-product transportation that reduces edema, improves limb function, and promotes a quicker recovery.4 Hot whirlpool has also been reported to result in reduced joint stiffness and increased extensibility of soft tissue, which causes hemoglobin to release oxygen more readily into the tissues.5 However, CB is believed to be more efficient in increasing circulation than simply placing the limb in a hot whirlpool.5,6 The hand, wrist, elbow, forearm, foot, ankle, knee, and lower leg are frequent sites for CB treatment, and rheumatoid arthritis, diabetes, carpal tunnel syndrome, foot and ankle sprains, and soft tissue trauma are among the common conditions that have been reported to benefit from CB treatment.7–13
Protocols for CB vary by temperature range, timing of each immersion, and overall duration.7 Opinions also differ regarding the appropriate order of immersion—hot first versus cold first—and whether the limb needs to exercise or rest while immersed in the baths.7,8 However, the most typical protocol reported in the literature is an initial immersion of the resting limb for 10 minutes in hot water (38°C–40°C), alternated with a 1-minute immersion in cold water (8°C–10°C), followed by alternate immersions of 4 minutes in hot and 1 minute in cold (4 : 1) water for another 3 repetitions, for a total duration of 30 minutes.7,10,14,15 Some protocols do not include the initial 10 minutes of hot-water immersion or use a ratio of 3 : 1 minutes of hot and cold water.7,16 The CB modality is frequently used by therapists in private practices, outpatient hospitals, physician offices, and sports clubs and at home by individuals.1,2,6 Up to 70% of physical therapists providing hand therapy included CB among their treatment options.1
Although clinicians are familiar with CB and it is frequently described in the literature, its use is controversial, with insufficient evidence to support its application.6,17,18 Currently no standard explanation or rationale is accepted for the best ranges of temperature and immersion duration. Also, although the physiological effects of CB on superficial tissues have been reported by a number of researchers,1,5,7,12,14,19 its effects on intramuscular oxygenation and perfusion remain unclear.
We conducted the present study to investigate the physiological effects of CB on intramuscular hemodynamics and oxygenation using near-infrared spectroscopy (NIRS). Near-infrared spectroscopy is a noninvasive optical technique that uses near-infrared light to study tissue hemodynamics and oxygenation in real time by monitoring changes in tissue chromophore concentrations transcutaneously.20 When light in the near-infrared spectrum penetrates living tissue, a portion of photons is lost to scatter, others are transmitted directly through the tissue, and still others are absorbed by tissue chromophores. Variations in photon absorption at specific wavelengths can be monitored to generate raw optical data that can then be converted by software-based mathematical algorithms into real-time concentration changes for each chromophore using a modification of the Beer-Lambert law.21,22 The principal findings derived from NIRS are changes in the concentration of oxygenated (O2Hb) and deoxygenated (HHB) hemoglobin chromophores, from which changes in oxygen supply and demand can be inferred, and total hemoglobin (tHb) [O2Hb + HHb], which reflects variations in local blood volume.23 The absolute ratio of O2Hb to tHb can also be determined using a spatially resolved configuration; hence, absolute tissue oxygen saturation can be calculated.24 The unique capabilities of NIRS may thus be relevant in monitoring the effect of physical modalities on tissue hemodynamics and oxygenation.25–28
The purpose of our study was to investigate the effects of CB on intramuscular perfusion and oxygenation of the lower leg in healthy individuals using noninvasive NIRS. We hypothesized that by altering local hemodynamics, CB would increase the level of intramuscular oxygenated blood volume.
METHODS
Participants
Ten adult volunteers (5 men and 5 women; average age = 29 [range = 17 ± 42] years) were recruited. Their body mass index was 24.6 ± 3.2, and mean adipose tissue thickness beneath the NIRS probes was 6.4 ± 2.2 mm. Volunteers were included if they were adults, were healthy, had no history of lower limb abnormality or surgery, had no history of skin hypersensitivity, were nonsmokers, could provide informed consent, and had subcutaneous adipose tissue thickness of less than 15 mm at the site used for NIRS monitoring over the gastrocnemius muscle. This maximum subcutaneous adipose tissue thickness was necessary to ensure effective NIRS light penetration to the underlying muscle tissue. All participants provided written informed consent, and the study was approved by the Clinical Research Ethics Board of the University of British Columbia.
Preparation
Participants refrained from consuming caffeine and alcohol and from exercising for 48 hours before testing to minimize potential effects on muscle blood flow; measurements were performed during the afternoon. The midportion of the medial gastrocnemius muscle belly of both legs was located and marked for NIRS sensor placement. Using a skinfold caliper (Jamar 2058; Sammons Preston Rolyan, Bolingbrook, IL), we measured and recorded subcutaneous fat-layer thickness at the marked points.
Instrumentation
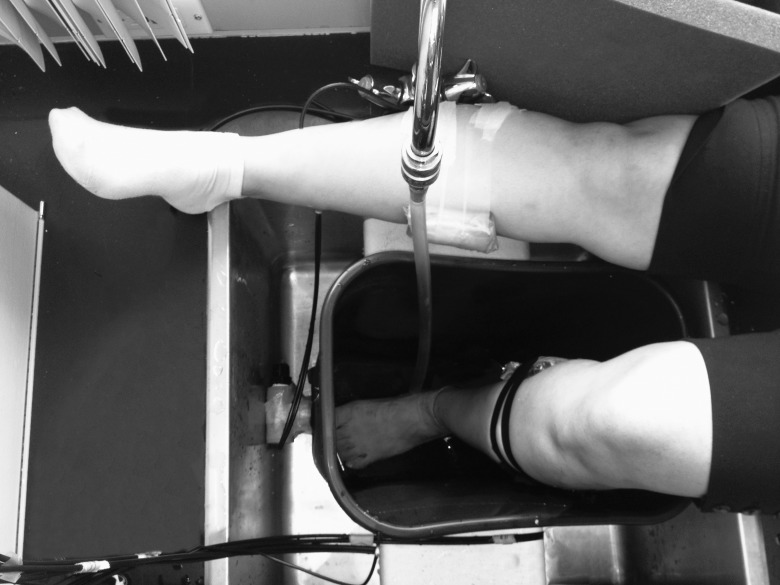
Two sets of wireless SR-NIRS devices (PortaMon; Artinis Medical Systems BV, Elst, The Netherlands) were placed and fixed with surgical taping over the measurement points. To make the device waterproof, each device was covered with an optically clear, transparent, thin silicone sheath before the placement (Figure 1).
Figure 1.
The placement of 2 sets of wireless near-infrared spectroscopy devices, covered by optically clear and transparent silicone sheaths, over the right and left medial gastrocnemius muscles.
The NIRS system used in this study was a self-contained wireless device with light-emitting diodes as the NIR light source and optical geometry for the spatially resolved measurement of oxygen saturation. The device weighed 84 g, and its full specifications have been described previously.29 Three paired light-emitting diodes with wavelengths of 760 and 850 nm were mounted at distances of 30, 35, and 40 mm from the silicon photodiode sensor. Power was supplied by a rechargeable lithium polymer battery, and the unit incorporated Bluetooth broadcast technology (Kirkland, WA) for data transfer to a laptop computer up to 20 m away. Data were collected at 10 Hz, and data analysis and graphic display were managed by proprietary NIRS software (Oxysoft; Artinis Medical Systems BV). Raw optical data were analyzed from each source-detector distance, and changes derived and displayed for changes in concentration from the baselines of O2Hb, HHb, tHb, and an absolute measure of tissue oxygenation was expressed as the tissue saturation index (TSI%). Maximum depth of penetration of the device for monitoring O2Hb, HHb, and tHb and calculating TSI% was about 20 mm, which was sufficient for monitoring gastrocnemius muscle hemodynamics and oxygenation. The NIRS data were collected before, during, and after application of a common protocol of CB (explained in the next paragraph) to monitor changes in intramuscular tissue perfusion and oxygenation. We started monitoring bilateral (left side under study versus right side control) NIRS measures 10 minutes before CB application, to record a baseline measure, and continued during the CB procedure and for another 10 minutes after completion of the procedure.
Contrast Baths
One 30-L bath with an adjustable valve was used as the reservoir for the hot and cold water. Water was transferred to the bath from separate hot and cold containers and temperatures were controlled subsequently, according to the protocol. With the participant wearing shorts and sitting upright, after 10 minutes of baseline NIRS measurement, the participant's left limb, with the knee flexed to 90° and the ankle in resting position, was immersed in hot (38°C–40°C) water (depth of ∼50 cm) for 10 minutes. The water was then alternated to cold water (8°C–10°C) for 1 minute, followed by 4 minutes in hot water and 1 minute in cold water for another 3 repetitions, for a total duration of 30 minutes. Each water exchange took 10 to 15 seconds. During the procedure, the right (control) limb rested on the floor similarly, with the knee flexed to 90° and the ankle in resting position. Participants were instructed to not activate their lower limb muscles during the experiment. The room temperature was constant at approximately 22°C during all experiments.
Statistical Analysis
Participant characteristics were collected and summarized using descriptive statistics. Mean changes in NIRS-derived O2Hb, HHb, and tHb at the channel with the highest interoptode distance (4 cm) and TSI% measures were calculated for each stage of CB. The measures at each stage of the CB were compared with baseline measures by performing a paired-samples Student t test. Data are presented as mean ± standard deviation. Statistical significance was set at P < .05 for all comparisons. All statistical analyses were performed using SPSS (version 17.0; SPSS Inc, Chicago, IL).
RESULTS
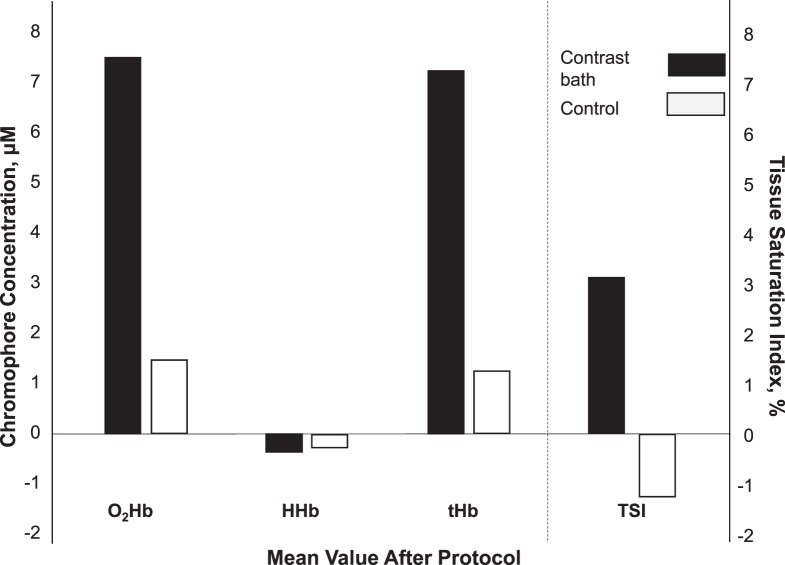
The mean values of O2Hb, HHb, and tHb in both the CB and control legs are provided in Figure 2. After a 30-minute protocol of CB, we observed significant overall increases in tissue O2Hb (7.4 ± 4 μM), tHb (7.6 ± 6.1 μM), and TSI% (3.1% ± 2.3%) but no change on the control side. The mean values of tissue O2Hb, HHb, and tHb during each stage of CB (10-minute hot, 1-minute cold, 4-minute hot, 1-minute cold, 4-minute hot, 1-minute cold, 4-minute hot, 1-minute cold, 4-minute hot) are provided in the Table. In the experimental leg, tissue O2Hb and tHb increased in all 5 stages of the hot bath and decreased in all 4 stages of the cold bath. Significant changes in HHb were evident after 14 minutes, toward the end of CB. In general, tHb showed a consistent pattern of increases in the hot bath and decreases in the cold bath. Values of tissue O2Hb, HHb, and tHb were not different during any stage of CB on the control side.
Figure 2.
Changes in the mean values of tissue oxygenated (O2Hb), deoxygenated (HHb), and total (tHb) hemoglobin and tissue oxygen-saturation level (TSI) after application of the 30-minute contrast-baths protocol in the experimental and control legs.
Table.
Absolute Tissue Oxygenated, Deoxygenated, and Total Hemoglobin Concentration Changes During Each Stage of the Contrast Baths and 10 Minutes of Recovery Timea (Mean ± SD)
| Hemoglobin Concentration, μM |
Hot (10 min) |
1st Cold (1 min) |
1st Hot (4 min) |
2nd Cold (1 min) |
2nd Hot (4 min) |
3rd Cold (1 min) |
3rd Hot (4 min) |
4th Cold (1 min) |
4th Hot (4 min) |
Recovery (10 min) |
| Oxygenated | 3.5 ± 0.9b | −1.2 ± 2.4 | 2.4 ± 2.3b | −0.6 ± 0.9b | 1.8 ± 0.8b | −1.1 ± 0.9b | 1.8 ± 0.9b | −1.5 ± 1.4b | 2.4 ± 1.3b | 1.1 ± 0.7 |
| Deoxygenated | −0.1 ± 0.9 | −0.9 ± 1.4b | 0.5 ± 1.4 | −0.4 ± 0.7b | 0.3 ± 0.5b | −0.4 ± 0.5b | 0.4 ± 0.4b | 0 ± 0.7b | 0.2 ± 0.7b | 0.1 ± 0.6 |
| Total | 3.4 ± 1.6b | −2.2 ± 3.7 | 2.9 ± 3.6b | −1 ± 1.4b | 2.1 ± 1.1b | −1.5 ± 1.2b | 2.2 ± 1.2b | −1.5 ± 1.7b | 2.6 ± 1.8a | 1.2 ± 1 |
A positive value indicates an increase and a negative value indicates a decrease in the concentration during each stage of the protocol.
Significant difference in measure during each stage (P < .05).
DISCUSSION
A 30-minute protocol of CB application altered lower leg intramuscular hemodynamics and oxygenation in healthy people. Hot-water immersion for 4 minutes increased the gastrocnemius intramuscular perfusion and oxygenation level, whereas a 1-minute cold bath decreased the intramuscular oxygenated blood volume.
The key contribution of this study to the field is that the effects of CB on intramuscular perfusion and oxygenation had not been established previously. Significant changes in the NIRS-derived measures of tissue oxygenation and perfusion in subsequent hot and cold stages provided evidence for the efficacy of CB in altering tissue hemodynamics. Increases in intramuscular O2Hb, tHb, and TSI% after completion of CB, compared with the baseline measure, indicated the efficacy of the intervention in increasing intramuscular perfusion and oxygenation. This effect on intramuscular hemodynamics may explain the therapeutic use of CB in rehabilitation.
In exercise and sports medicine, CB interventions have been widely used over the years to improve postexercise recovery and reduce DOMS after high-intensity activities more effectively than either cold- or warm-water immersion. These positive effects of CB are based on both subjective assessments and more objective measures such as isometric force production and various field performance tests.30–32 Nonetheless, the physiological basis of such effects is not yet fully established, just as the optimal treatment protocol is not yet clear. Authors33 have looked into outcome measures such as variations in heart rate, blood pressure, and respiratory minute volume as well as peripheral catecholamine concentration. The meaningful changes demonstrated in this study further support the use of CB in performance recovery and functional rehabilitation.
One limitation of our study was the small sample size. We did not perform a power analysis due to the lack of previous data. Another limitation was the lack of long-term tissue-hemodynamic monitoring after the CB. In addition, all participants were healthy individuals without injury at the measurement site, and consequently, it is inappropriate to extend these findings to a population with acute or chronic injuries. Future researchers should pursue larger sample sizes to assess the extent and durability of CB effects in the rehabilitation of acute and chronic musculoskeletal conditions and for recovery after intense sport activity. It is also important to establish optimal protocols for various applications of CB by correlating subjective data such as reported symptoms, objective performance or clinical assessment outcomes, and NIRS hemodynamic findings.
CONCLUSIONS
We demonstrated that CB application induced transient changes in the hemodynamics and oxygenation of the gastrocnemius muscle of healthy individuals. The physiological effects of CB on lower limb muscles, as shown in this study, included increasing the level of intramuscular oxygenated blood volume after a 30-minute protocol of CB. The effect of CB application in improving the tissue hemodynamics and oxygenation that are considered essential mechanisms of tissue repair may, therefore, support the therapeutic use of CB in rehabilitation and sports medicine. Further studies of people with acute and chronic muscle damage and DOMS are warranted.
REFERENCES
- 1.Stanton DB, Bear-Lehman J, Graziano M, Ryan C. Contrast baths: what do we know about their use? J Hand Ther. 2003;16(4):343–346. doi: 10.1197/s0894-1130(03)00161-3. [DOI] [PubMed] [Google Scholar]
- 2.Versey NG, Halson SL, Dawson BT. Water immersion recovery for athletes: effect on exercise performance and practical recommendations. Sports Med. 2013;43(11):1101–1130. doi: 10.1007/s40279-013-0063-8. [DOI] [PubMed] [Google Scholar]
- 3.French DN, Thompson KG, Garland SW, et al. The effects of contrast bathing and compression therapy on muscular performance. Med Sci Sports Exerc. 2008;40(7):1297–1306. doi: 10.1249/MSS.0b013e31816b10d5. [DOI] [PubMed] [Google Scholar]
- 4.Cochrane DJ. Alternating hot and cold water immersion for athlete recovery: a review. Phys Ther Sport. 2004;5(1):26–32. [Google Scholar]
- 5.Campion MR. Hydrotherapy: Principles and Practice. Oxford, England: Butterworth-Heinemann;; 1998. [Google Scholar]
- 6.Cameron MH. Physical Agents in Rehabilitation: From Research to Practice 2nd ed. Philadelphia, PA: WB Saunders Company;; 2003. pp. 418–419. [Google Scholar]
- 7.Breger Stanton D, Lazaro R, MacDermid JA. Systemic review of the effectiveness of contrast baths. J Hand Ther. 2009;22(1):57–70. doi: 10.1016/j.jht.2008.08.001. [DOI] [PubMed] [Google Scholar]
- 8.Janssen RG, Schwartz DA, Velleman PF. A randomized controlled study of contrast baths on patients with carpal tunnel syndrome. J Hand Ther. 2009;22(3):200–207. doi: 10.1016/j.jht.2009.02.001. [DOI] [PubMed] [Google Scholar]
- 9.Struijs PA, Kerkhoffs GM. Ankle sprain. BMJ Clin Evid. 2010;2010:1115. [PMC free article] [PubMed] [Google Scholar]
- 10.Jackins S, Jamieson A. Use of heat and cold in physical therapy. In: Lehmann JF, editor. Therapeutic Heat and Cold 4th ed. Baltimore, MD: Williams & Wilkins;; 1990. p. 656. [Google Scholar]
- 11.Knight KL, Londeree BR. Comparison of blood flow in the ankle of uninjured subjects during therapeutic applications of heat, cold and exercise. Med Sci Sports Exerc. 1980;12(1):76–80. doi: 10.1249/00005768-198021000-00015. [DOI] [PubMed] [Google Scholar]
- 12.Moseley B, Korson R, Petrofsky J, et al. The effects of contrast baths on skin blood flow in people with type 2 diabetes and age-matched controls. J Investig Med. 2006;54(suppl 1):S118–S119. doi: 10.1080/09593980701209295. [DOI] [PubMed] [Google Scholar]
- 13.Myrer JW, Draper DO, Durrant E. Contrast therapy and intramuscular temperature in the human leg. J Athl Train. 1994;29(4):318–322. [PMC free article] [PubMed] [Google Scholar]
- 14.Shih CY, Lee WL, Lee CW, Huang CH, Wu YZ. Effect of time ratio of heat to cold on brachial artery blood velocity during contrast baths. Phys Ther. 2012;92(3):448–453. doi: 10.2522/ptj.20100394. [DOI] [PubMed] [Google Scholar]
- 15.Myrer JW, Measom G, Durrant E, Fellingham GW. Cold- and hot-pack contrast therapy: subcutaneous and intramuscular temperature change. J Athl Train. 1997;32(3):238–241. [PMC free article] [PubMed] [Google Scholar]
- 16.Hayes KW. Manual for Physical Agents 5th ed. Upper Saddle River, NJ: Prentice Hall Health;; 2000. pp. 64–45. [Google Scholar]
- 17.Artzberger S. Edema reduction techniques: a biologic rationale for selection. In: Cooper C, editor. Fundamentals of Hand Therapy: Clinical Reasoning and Treatment Guidelines for Common Diagnoses of the Upper Extremity. St Louis, MO: Mosby;; 2007. p. 43. [Google Scholar]
- 18.Hecox B, Leinanger PM. Hydrotherapy. In: Hecox B, Andemicael-Mehreteab T, Weisberg J, Sanko J, editors. Integrating Physical Agents in Rehabilitation 2nd ed. Upper Saddle River, NJ: Prentice Hall;; 2006. pp. 421–422. [Google Scholar]
- 19.Fiscus KA, Kaminski TW, Powers ME. Changes in lower-leg blood flow during warm-, cold-, and contrast-water therapy. Arch Phys Med Rehabil. 2005;86(7):1404–1410. doi: 10.1016/j.apmr.2004.11.046. [DOI] [PubMed] [Google Scholar]
- 20.Ferrari M, Mottola L, Quaresima V. Principles, techniques and imitations of near infrared spectroscopy. Can J Appl Physiol. 2004;29(4):463–487. doi: 10.1139/h04-031. [DOI] [PubMed] [Google Scholar]
- 21.Delpy DT, Cope M. Quantification in tissue near-infrared spectroscopy. Philos Trans R Soc Lond Biol Sci. 1997;352(1354):649–659. [Google Scholar]
- 22.van Beekvelt MCP, Borghuis MS, van Engelen BGM, Wevers RA, Colier WNJM. Adipose tissue thickness affects in vivo quantitative near-IR spectroscopy in human skeletal muscle. Clin Sci (Lond) 2001;101(1):21–28. doi: 10.1042/cs20000247. [DOI] [PubMed] [Google Scholar]
- 23.Boushel R, Langberg H, Olesen J, Gonzales-Alonzo J, Bulow J, Kjaer M. Monitoring tissue oxygen availability with near infrared spectroscopy (NIRS) in health and disease. Scand J Med Sci Sports. 2001;11(4):213–222. doi: 10.1034/j.1600-0838.2001.110404.x. [DOI] [PubMed] [Google Scholar]
- 24.Suzuki S, Takasaki S, Ozaki T, Kobayashi Y. A tissue oxygenation monitor using NIR spatially resolved spectroscopy. Proc Soc Photo Optic Instrum Eng. 1999;3597:582–592. [Google Scholar]
- 25.Banzer W, Hubscher M, Seib M, Vogt L. Short-time effects of laser needle stimulation on the peripheral microcirculation assessed by laser Doppler spectroscopy and near-infrared spectroscopy. Photomed Laser Surg. 2006;24(5):575–580. doi: 10.1089/pho.2006.24.575. [DOI] [PubMed] [Google Scholar]
- 26.Lo MY, Ong MW, Chen WY, Sun WZ, Lin JG. The effects of acupuncture on cerebral and muscular microcirculation: a systematic review of near-infrared spectroscopy studies. Evid Based Complement Alternat Med. 2015;2015:839470. doi: 10.1155/2015/839470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Muthalib M, Jubeau M, Millet GY, Maffiuletti NA, Ferrari M, Nosaka K. Biceps brachii muscle oxygenation in electrical muscle stimulation. Clin Physiol Funct Imaging. 2010;30(5):360–368. doi: 10.1111/j.1475-097X.2010.00953.x. [DOI] [PubMed] [Google Scholar]
- 28.Shadgan B, Med S, Pakravan AH, Hoens A, Reid WD. Subcutaneous and intramuscular hemodynamics and oxygenation after cold-spray application as monitored by near-infrared spectroscopy. J Athl Train. 2015;50(8):800–805. doi: 10.4085/1062-6050-50.6.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shadgan B, Reid WD, Gharakhanlou R, Stothers L, Macnab A. Wireless near-infrared spectroscopy of skeletal muscle oxygenation and hemodynamics during exercise and ischemia. Spectroscopy. 2009;23(5):233–241. [Google Scholar]
- 30.Vaile JM, Gill ND, Blazevich AJ. The effect of contrast water therapy on symptoms of delayed onset muscle soreness. J Strength Cond Res. 2007;21(3):697–702. doi: 10.1519/R-19355.1. [DOI] [PubMed] [Google Scholar]
- 31.Higgins TR, Heazlewood IT, Climstein M. A random control trial of contrast baths and ice baths for recovery during competition in U/20 rugby union. J Strength Cond Res. 2011;25(4):1046–1051. doi: 10.1519/JSC.0b013e3181cc269f. [DOI] [PubMed] [Google Scholar]
- 32.Versey NG, Halson SL, Dawson BT. Effect of contrast water therapy duration on recovery of running performance. Int J Sports Physiol Perform. 2012;7(2):130–140. doi: 10.1123/ijspp.7.2.130. [DOI] [PubMed] [Google Scholar]
- 33.Bleakley CM, Davison GW. What is the biochemical and physiological rationale for using cold-water immersion in sports recovery? A systematic review. Br J Sports Med. 2010;44(3):179–187. doi: 10.1136/bjsm.2009.065565. [DOI] [PubMed] [Google Scholar]