Abstract
Chronic inflammatory enteropathies (CIE) in dogs are a group of disorders that are characterized by chronic persistent or recurrent signs of gastrointestinal disease and histologic evidence of mucosal inflammation. These CIEs are classified as either food‐responsive, antibiotic‐responsive, or immunosuppressant‐responsive enteropathy. Patients not clinically responding to immunomodulatory treatment are grouped as nonresponsive enteropathy and dogs with intestinal protein loss as protein‐losing enteropathy. Disease‐independent clinical scoring systems were established in dogs for assessment of clinical disease severity and patient monitoring during treatment. Histopathologic and routine clinicopathologic findings are usually not able to distinguish the subgroups of CIE. Treatment trials are often lengthy and further diagnostic tests are usually at least minimally invasive. Biomarkers that can aid in defining the presence of disease, site of origin, severity of the disease process, response to treatment, or a combination of these would be clinically useful in dogs with CIE. This article summarizes the following biomarkers that have been evaluated in dogs with CIE during the last decade, and critically evaluates their potential clinical utility in dogs with CIE: functional biomarkers (cobalamin, methylmalonic acid, folate, α1‐proteinase inhibitor, immunoglobulin A), biochemical biomarkers (C‐reactive protein, perinuclear anti‐neutrophilic cytoplasmic antibodies, 3‐bromotyrosine, N‐methylhistamine, calprotectin, S100A12, soluble receptor of advanced glycation end products, cytokines and chemokines, alkaline phosphatase), microbiomic biomarkers (microbiome changes, dysbiosis index), metabolomic biomarkers (serum metabolome), genetic biomarkers (genomic markers, gene expression changes), and cellular biomarkers (regulatory T cells). In addition, important performance criteria of diagnostic tests are briefly reviewed.
Keywords: antibiotic‐responsive enteropathy, dog, food‐responsive enteropathy, inflammation, inflammatory bowel disease
Abbreviations
- α1PI
alpha1‐proteinase inhibitor
- AP
alkaline phosphatase
- ARE
antibiotic‐responsive enteropathy
- 3‐BrY
serum 3‐bromotyrosine
- CCL
CC chemokine ligand
- CIE
chronic inflammatory enteropathy
- cPLI
canine specific pancreatic lipase
- CR
complete remission
- CRP
C‐reactive protein
- CXCL
CXC chemokine ligand
- DAMP
damage‐associated molecular pattern
- FRE
food‐responsive enteropathy
- IgA
immunoglobulin A
- IL
interleukin
- IRE
immunosuppressant‐responsive enteropathy
- MMA
methylmalonic acid
- NMH
N‐methylhistamine
- NPV
negative predictive value
- NR
no response
- NRE
nonresponsive (immunosuppressant‐refractory) enteropathy
- pANCA
perinuclear anti‐neutrophilic cytoplasmic antibodies
- PLE
protein‐losing enteropathy
- PPV
positive predictive value
- PR
partial response
- RAGE
receptor for advanced glycation end products
- sRAGE
soluble RAGE
- SNP
single‐nucleotide polymorphism
- TLR
Toll‐like receptor
- UPC
urine protein‐to‐creatinine ratio.
1. INTRODUCTION
Chronic inflammatory enteropathies (CIE) in dogs comprise a group of disorders that are characterized by chronic persistent or recurrent gastrointestinal signs, histologic evidence of mucosal inflammation, and the exclusion of other underlying gastrointestinal or extra‐gastrointestinal diseases.1, 2, 3, 4 Clinicopathologic variables, fecal parasite examination, and diagnostic imaging are important means to rule out other etiologies with a similar clinical presentation. The CIEs are retrospectively classified based on the response to empirical treatment as food‐responsive (FRE), antibiotic‐responsive (ARE), or immunosuppressant‐responsive enteropathy (IRE). Patients that do not respond to immunomodulatory treatment are categorized as having nonresponsive enteropathy (NRE).3, 4, 5, 6 The term protein‐losing enteropathy (PLE) is used for a subgroup of affected CIE dogs with intestinal protein loss, overall carrying a worse prognosis.3, 6, 7 Disease‐independent clinical scoring systems have been established in dogs8, 9 and can be used to semi‐objectively assess the severity of clinical signs at the time of diagnosis and to monitor patient improvement during treatment. Histopathologic and routine clinicopathologic findings usually are not able to distinguish different subgroups of CIE,5, 10 but younger dogs with less severe clinical signs are more likely to be diagnosed with FRE.5, 9 Treatment trials often are lengthy and further diagnostic tests, after appropriately designed dietary and antibiotic trials have failed, are usually invasive.3, 4, 5, 6 Biomarkers that can aid in diagnostic evaluation or patient monitoring or that can assess the response to various forms of treatment thus would be clinically useful in dogs with CIE. Several functional, biochemical, microbiomic, metabolomic, genetic, and cellular biomarkers have been evaluated in dogs with CIE during the last decade. This article will summarize selected biomarkers that have been evaluated in dogs with CIE and will critically evaluate their potential clinical utility. In addition, important performance criteria of diagnostic tests are discussed.
2. BIOMARKER CHARACTERISTICS
Understanding the clinical utility andas the limitations of biomarkers is very important when using biomarker data in clinical cases and to prevent pitfalls in the interpretation of biomarker results in clinical practice.11 Thus, the goals of using biomarkers and the parameters of importance for the interpretation of biomarker assays in the clinical setting are briefly reviewed.
2.1. Clinical utility of biomarkers
For a biomarker to be clinically useful, it must aid in evaluating organ function, assess the risk for disease development, diagnose a specific disease process, assess disease activity or severity, predict the patient's response to treatment or predict disease outcome, monitor disease progress, or some combination of these.11, 14 Because a single biomarker is very unlikely to satisfy all of these goals, it is very important to consider what clinical information is expected to be gained from using a specific biomarker, and in what clinical situation it will serve as a good surrogate marker for existing algorithms (eg, definition of baseline disease activity, response to treatment, or maintenance of clinical remission) to improve diagnostic decision making, treatment recommendations, patient monitoring, or even prophylactic measures (Table 1).11, 12, 13
Table 1.
Biomarker goals
| • Assessment of the risk for disease development |
| • Diagnosis of a disease process |
| • Evaluation of organ function |
| • Determination of organ origin |
| • Assessment of disease severity |
| • Assessment of response to treatment |
| • Prediction of individual outcome |
| • Monitoring of a disease process |
| • Detection of disease flares |
2.2. Classification of biomarkers
Disease biomarkers are either static or dynamic, and based on the desired characteristics described above, they typically are subclassified as functional, biochemical, cellular, genomic, proteomic, metabolomic, microbiomic, behavioral, or outcome biomarkers.14, 15, 16 Of these, functional, biochemical, and genomic biomarkers currently are the most practical to use and most widely available and are currently of particular clinical interest for CIE management in dogs (Figure 1).
Figure 1.
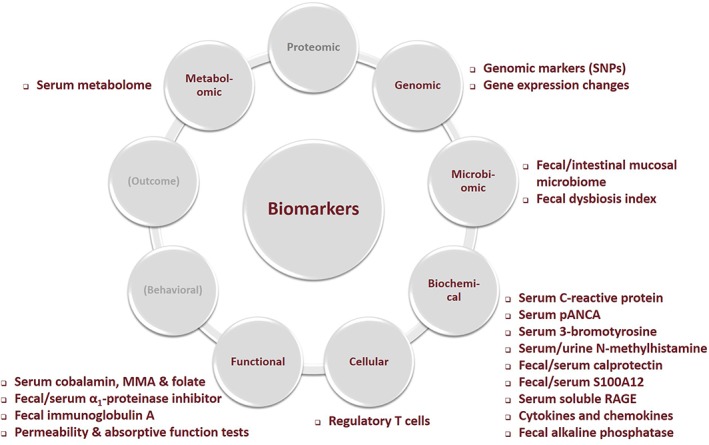
Groups of biomarkers in dogs with chronic inflammatory enteropathies (CIE). The figure shows the functional, biochemical, metabolomic, microbiomic, genomic, and cellular biomarkers that have been evaluated in dogs with CIE. MMA, methylmalonic acid; pANCA, perinuclear anti‐neutrophilic cytoplasmic antibodies; RAGE, receptor for advanced glycation end products; SNP, single‐nucleotide polymorphism
An important consideration for the interpretation of biomarker data is that based on the characteristics of the biomarker assay, either a qualitative or quantitative test result is obtained. Qualitative assays yield a dichotomized (eg, positive or negative) test result, whereas quantitative assays offer a numerical test result (ie, continuous or ordinal data). Interpretation of quantitative assays usually requires the use of either population‐ or patient‐based cut‐off values,18, 19, 20 and the results of these assays might also fall within a gray range (ie, ambiguous test results).
2.3. Biomarker performance criteria
Understanding the performance characteristics of diagnostic tests is important for correct interpretation. Any diagnostic test result must be interpreted in light of patient history, clinical signs, physical examination findings, and also in combination with the results of other diagnostic tests (eg, clinicopathologic variables, diagnostic imaging findings) performed.11, 14, 17
Although often assumed (and usually beyond the clinician's responsibility), it is important to recognize that the diagnostic performance of an assay relies heavily on its analytical performance.21 This includes preanalytic effects associated with the collection and handling (including storage) of samples and analytical variability arising from the methodology, instrumentation, and also technical skills.18, 21, 22 This aspect becomes especially important for the clinician when utilizing patient‐side (ie, in‐house) diagnostic tests.23 Important variables that are determined for the preanalytical validation of a biomarker assay include the lower detection limit of the assay, assay sensitivity, dilutional parallelism and linearity, assay accuracy, precision, and reproducibility.14, 21 In addition, quality assurance of a biomarker test requires that a quality control program be in place.22, 23
2.3.1. Biological variability
Biological variability, an inherent characteristic of all biochemical analytes, is an important aspect in the interpretation of the diagnostic performance of a biomarker.18 Normal biological variability comprises within‐subject (intraindividual) and among‐subject (interindividual) variation, both of which might be similar or different between health and various disease states.18, 24
Sources of biological variation include rhythmic expression or secretion, variation in gastrointestinal passage or patchy distribution of cells expressing and releasing a biomarker measured in feces, and the metabolism and half‐life of a biomarker (determined by its size, charge, hydrophobicity, and route of excretion).18, 24 Such sources of biological variation must be recognized and strategies developed to counterbalance such variation (eg, by collecting specimens on multiple days or at a certain time of the day).
Components of biological variability (ie, intra‐ and interindividual variability) usually are estimated from a relatively small set of specimens (depending upon the biological variability) obtained from a small group of healthy individuals during a relatively short time period.18 For most biomarkers, this approach allows for determining the critical change value (based on the indices of individuality and heterogeneity) and thus assessing the utility of a conventional population‐based reference interval.18
The critical change value is the percentage (or concentration) that a biomarker must change between sequential measurements to be considered a clinically relevant alteration and not merely a reflection of biological or analytical variability.18, 24 This variable also is an important determinant of the ability of a biomarker to distinguish diseased individuals from healthy individuals and from disease controls.
Biomarkers for which the use of a conventional population‐based reference interval is appropriate generally are useful for diagnosing a disease (ie, as an “event marker”).18 In contrast, biomarkers for which using a traditional population‐based reference interval is not feasible generally are better for monitoring a condition (ie, as a “chronic disease marker”).18
Establishing the lower and upper limits of the reference interval for a quantitative biomarker requires a large reference sample group (ideally n > 120),19, 20 and can be done by several different methods, depending on the number of individuals included in the reference sample group and the distribution of the data.19, 20 Because some biomarkers can vary with age, sex, neuter status, body condition, muscle condition, or other environmental factors (eg, diet, medications), stratification of the reference population based on such variables may be necessary.
2.3.2. Diagnostic accuracy
Diagnostic accuracy of a test typically is described by means of its estimated sensitivity and specificity.14, 25, 26
Sensitivity of a diagnostic test is the likelihood of a positive test result in a patient affected with the disease (true positive rate) that was diagnosed based on an independent gold standard diagnostic method or technique. Sensitivity is calculated as the number of all true positive individuals divided by the number of all diseased individuals. The higher the sensitivity, the lower the false negative rate (ie, great confidence exists that a negative test result is true). Consequently, diagnostic tests with a high sensitivity are useful to rule out a disease (ie, as screening tests).11, 14, 25
Specificity of a diagnostic test is the likelihood of a negative test result in a patient not having the disease (true negative rate) as determined by an independent gold standard diagnostic test. Specificity is calculated as the number of all true negative individuals divided by the number of all nondiseased individuals. The higher the specificity, the lower the false positive rate (ie, great confidence exists that a positive test result is true). Thus, diagnostic tests with a high specificity are useful for confirming a diagnosis (confirmatory tests).11, 14, 25
2.3.3. Diagnostic performance
Predictive values of a diagnostic test can be determined if (in addition to the diagnostic accuracy of the test) the prevalence of the disease in the population tested is known.14, 25 Predictive values are important variables in clinical practice when using a biomarker to determine the patient's disease status, because they can help select the patient population in which a specific test is likely to yield meaningful results.14, 20, 26
The positive predictive value (PPV) is the chance that a patient with a positive test result has the disease.26 It is calculated as the number of all true positive test results divided by the number of all individuals testing positive. Diagnostic tests with a low specificity have a low PPV when the disease prevalence is low. These tests are good confirmatory tests in patients with a strong suspicion for the disease and absent any factors (eg, medication) that could produce false‐positive results, but they should not be used to screen for an uncommon disease. The negative predictive value (NPV) is the chance that a patient with a negative test result does not have the disease.26 It is calculated as the number of all true negative test results divided by the number of all individuals testing negative. Diagnostic tests with a low sensitivity have a low NPV when the disease prevalence is high. These tests are good screening tests for large patient populations, but should not be used to rule out the diagnosis when there is a strong suspicion for the disease.11, 14
Because PPV and NPV depend on the disease prevalence, studies reporting PPV and NPV should be critically evaluated and interpreted with caution if predictive values are estimated based on the study prevalence as it might not reflect the prevalence of the disease in the population for which the test will be used. The study prevalence might be different from the disease prevalence in a specific clinical practice setting and, unfortunately, the true prevalence of few diseases is known in veterinary medicine.
3. BIOMARKERS IN CHRONIC INFLAMMATORY ENTEROPATHIES OF DOGS
3.1. Routine clinicopathologic variables in CIE
Routine clinicopathologic variables usually are not specific for a diagnosis of CIE but are important to rule out other diseases causing similar clinical signs and to assess the overall status of the patient.1, 3, 6
Serum albumin concentration can be decreased in some dogs with CIE because of gastrointestinal protein loss (PLE) and is of prognostic value.7, 9, 27, 28 Serum calcium and cholesterol concentrations also may be decreased in dogs with PLE.6, 7, 29, 30 Serum bile acid concentrations can help further evaluate the patient for hepatopathy, and urinalysis with a urine protein‐to‐creatinine (UPC) ratio can help exclude renal loss of protein if the UPC is <0.5.
A baseline serum cortisol concentration or an ACTH stimulation test can exclude or confirm a diagnosis of (atypical) hypoadrenocorticism. A fecal parasite examination also should be performed. Subnormal serum canine trypsin‐like immunoreactivity concentrations can identify dogs with exocrine pancreatic insufficiency.31 Serum canine specific pancreatic lipase (cPLI) concentration also should be measured to evaluate the patient for concurrent pancreatitis. Increased serum cPLI concentrations appeared to be a negative prognostic factor in dogs with CIE in 1 study.32 Serum gastrin concentrations may indicate a gastrinoma if increased >10 times above the normal reference interval.33
Despite the importance of the minimum database comprised of routine clinicopathologic tests, these diagnostic tests usually are not specific for CIE in dogs, and additional biomarkers that are more disease‐specific, organ‐specific, or both appear to be a very useful additional tool for the management of dogs with CIE.
3.2. Clinical utility of biomarkers for CIE
Biomarkers that could be clinically useful in dogs with CIE are markers that reflect the individual risk to develop CIE, evaluate gastrointestinal function (ie, digestion, absorption, secretion, or a combination of these), aid in diagnosing the inflammatory disease process, indicate disease activity or severity, predict individual response to specific forms of treatment or disease outcome, monitor the severity of gastrointestinal inflammation, or even a combination of these.12, 13, 14 The utility of a marker in routine clinical practice also requires that it be easy to evaluate, inexpensive, and minimally invasive, in addition to being stable in routine biological specimens under clinical conditions.11
3.3. Classes of biomarkers for CIE
Biomarkers with potential clinical utility that have been evaluated in dogs with CIE are functional, biochemical, and genomic markers (Figure 1). Selected biomarkers are discussed below.
3.4. Functional biomarkers
The functional biomarkers include fecal and serum alpha1‐proteinase inhibitor (α1PI) concentrations, serum folate (vitamin B9), and cobalamin (vitamin B12) concentrations, markers to evaluate gastrointestinal permeability and absorptive function (eg, 51Cr‐EDTA or iohexol absorption, serum or urine lactulose/rhamnose ratio, or the xylose/methylglucose ratio), fecal immunoglobulin A (IgA) concentrations, serum metabolite profiles, and the fecal dysbiosis index. Because markers to assess gastrointestinal permeability and absorptive function are fairly invasive and impractical, these tests are not useful clinically and will not be discussed in detail.
3.4.1. Fecal and serum alpha1‐proteinase inhibitor (α1PI)
Alpha1‐proteinase inhibitor (α1PI, also known as alpha1‐antitrypsin in human medicine) is a major proteinase inhibitor that is primarily synthesized in the liver.34 Despite being protective against the effects of trypsin, chymotrypsin, and neutrophil proteases, the role of canine α1PI as an acute‐phase reactant still is controversial.35, 36, 37, 38 Canine α1PI has a molecular weight similar to that of albumin,39 and with diseases causing gastrointestinal protein loss both should be lost at approximately the same rate. Unlike albumin, α1PI is resistant against proteolysis, allowing its extraction and quantification in fecal samples.40
Increased fecal canine α1PI concentrations are clinically useful as a marker for gastrointestinal protein loss and histologic lesions seen with PLE in dogs (ie, lacteal dilatation, crypt abscesses, or both).41, 42, 43 Relatively large day‐to‐day variation in fecal α1PI concentrations necessitates that fecal samples be collected on 3 consecutive days,40, 44 and a 3‐day mean fecal α1PI concentration ≥13.9 μg/g or a 3‐day maximum fecal α1PI concentration ≥21.0 μg/g is interpreted as abnormal.40 A 3‐day mean fecal α1PI concentration ≥19.0 μg/g43 is a good confirmatory test (PPV > 80%) for histologic lesions typically seen with PLE in dogs with CIE that have failed elimination diet and antibiotic trials (where the prevalence of hypoalbuminemia is 50–56%27, 45; Table 2).
Table 2.
Performance characteristics of selected biomarkers evaluated in dogs with chronic inflammatory enteropathies
| Biomarker | Group comparison | Cut‐off | Sensitivity | Specificity | Critical change value | Reference |
|---|---|---|---|---|---|---|
| Fecal α1PI (3‐day mean) | PLE versus non‐PLE‐CIEa | ≥19.0 μg/g | 44% | 85% | nd | [43] |
| Serum α1PI | nd ≤1,087 mg/L |
nd 64% |
nd 93% |
510 mg/L nd |
[48] [43] |
|
| Serum‐to‐fecal α1PI ratio | ≤53.6 g/mL | 49% | 89% | nd | [43] | |
| Serum CRP | IRE/NRE versus FRE/ARE | ≥9.1 mg/L nd |
72% nd |
100% nd |
nd 270% |
[50] [92] |
| Serum pANCA | IRE/NRE versus other causes of diarrhea or healthy | pANCA positivity | 51% | 83% | nd | [98] |
| FRD versus IRE/NRE | 62% | 77% | nd | [100] | ||
| IRE/NRE versus lymphoma | 37% | 83% | nd | [101] | ||
| FRD versus IRE/NRE/healthy | 61% | 100% | nd | [99] | ||
| Fecal calprotectin | IRE/NRE: PR/NR versus CR | ≥15.2 μg/g | 80% | 75% | nd | [50] |
| Serum calprotectin | IRE/NRE versus healthy | ≥296 μg/L nd |
82% nd |
68% nd |
nd 6.4 mg/L |
[94] [173] |
| Fecal S100A12 | Endoscopic score: ≤1 versus ≥2 | ≥273 ng/g | 71% | 89% | nd | [133] |
| IRE/NRE versus FRE/ARE | ≥490 ng/g | 64% | 77% | nd | [45] | |
| IRE/NRE: NR versus CR/PR | ≥2,700 ng/g | 100% | 73% | nd | [45] | |
| Serum S100A12 | nd nd |
nd nd |
nd nd |
nd nd |
85% 115% |
[132] [131] |
| Serum sRAGE | IRE/NRE versus. healthy | ≤340 ng/L | 90% | 73% | nd | [27] |
| Fecal dysbiosis index | CIE versus healthy | ≥0 | 74% | 95% | nd | [68] |
Abbreviations: α1PI, alpha1‐proteinase inhibitor; ARE, antibiotic‐responsive enteropathy; CIE, chronic inflammatory enteropathy; CRP, C‐reactive protein; CR, complete remission; FRE, food‐responsive enteropathy; IRE, immunosuppressive‐responsive enteropathy; nd, not determined; NR, no response; pANCA, perinuclear anti‐neutrophilic cytoplasmic antibodies; PLE, protein‐losing enteropathy; PR, partial response; sRAGE, soluble receptor for advanced glycation end products.
All dogs with CIE independent of the disease classification.
Fecal canine α1PI appears to be particularly useful for the early detection of gastrointestinal protein loss because it can be increased before the onset of clinical signs, hypoalbuminemia (or panhypoproteinemia), or both.41 The α1PI test also might be useful to differentiate gastrointestinal protein loss from hepatic causes of hypoalbuminemia. Using a 3‐day mean fecal α1PI concentration of ≥4.0 μg/g appears to be a good test to screen (NPV > 90%) for PLE in dogs with CIE (the prevalence of histologic lesions typically seen with PLE is 20% of dogs with hypoalbuminemia and 20%‐30% of all dogs with CIE5, 9, 43, 45; Table 2).43 However, although a value of ≥4.0 μg/g has high sensitivity for PLE, it is not very specific and many dogs with values ≥4.0 μg/g but <19.0 μg/g do not have PLE. Also, increased fecal canine α1PI concentrations in dogs <6–12 months of age should be interpreted with caution and should be verified at ≥1 year of age.40
Chronic gastrointestinal loss of α1PI caused by PLE will deplete systemic α1PI concentrations, resulting in a decreased serum α1PI concentration43, 46, 47 and presumably an altered plasma proteinase‐proteinase inhibitor balance. Consequently, the serum‐to‐fecal α1PI ratio appears to further improve the diagnostic accuracy for PLE in hypoalbuminemic dogs,43 but caution must be exercised with the interpretation of serum α1PI concentrations in corticosteroid‐treated dogs.48 Also, the serum concentration of α1PI must decrease by at least 510 mg/L (minimum critical difference) for that decrease to be considered clinically relevant.48
3.4.2. Serum cobalamin, methylmalonic acid (MMA), and folate concentrations
Cobalamin (vitamin B12) and folate (vitamin B9) are water‐soluble vitamins that can be measured in serum and might serve as indicators of CIE.
Cobalamin is exclusively absorbed in the distal small intestine (ileum) where specific receptors internalize the intrinsic factor–cobalamin complex. Hypocobalaminemia frequently is detected in dogs with chronic enteropathies (19%‐54%)5, 9, 43, 45, 49, 50 and is presumed to reflect distal small intestinal malabsorption, secondary small intestinal dysbiosis (with increased utilization of cobalamin by the intestinal microbiota), or both. Hypocobalaminemia is a negative prognostic factor in dogs with CIE and is associated with hypoalbuminemia.5, 9 However, hypocobalaminemia is not specific for CIE and also can be observed in dogs with exocrine pancreatic insufficiency.31 Also, a normal serum cobalamin concentration does not rule out a diagnosis of CIE. Subnormal or low normal serum cobalamin concentrations (<400 ng/L) indicate a need for parenteral (50 μg/kg SQ once weekly for 6 weeks, then every 2‐4 weeks) or PO supplementation (50 μg/kg PO q24h for at least 12 weeks) with cyanocobalamin.51, 52
Methylmalonic acid (MMA) is a metabolite that accumulates when the activity of methylmalonyl‐CoA mutase is decreased because of a lack of intracellular cobalamin.53 Thus, increased MMA production is a useful marker for cobalamin deficiency at the cellular level as a result of cobalamin malabsorption, decreased cobalamin transport, or both.53, 54 Concentrations of MMA can be determined in serum and urine samples,49, 53, 54 and measurement of both serum cobalamin and serum or urine MMA appears to be superior to serum cobalamin concentration alone for proper assessment of cobalamin status in dogs. However, measurement of MMA currently is not routinely performed in companion animals51 because of the cost and technical difficulty of the assay, but it might be a reasonable future strategy. Serum MMA concentrations can be increased in patients with renal insufficiency49 and must be interpreted in light of the serum creatinine or symmetric dimethylarginine (SDMA) concentration.
Folate is primarily absorbed in the proximal small intestine (ie, duodenum and proximal jejunum) as folate monoglutamate via folate carriers. Hypofolatemia can result from chronic malabsorption in the proximal small intestine and was detected in 14% of dogs with CIE in 1 study,50 but a decreased serum folate concentration is not specific for CIE and normofolatemia does not exclude a diagnosis of CIE. Serum folate concentration can be falsely normal or increased because of secondary small intestinal dysbiosis (with increased folate production by some members of the intestinal microbiota) or hypocobalaminemia and should be monitored after cobalamin supplementation.55 Oral folic acid supplementation (10 μg/kg or 200‐400 μg/dog PO q24h for 30 days) is recommended in patients with moderate or marked hypofolatemia.
3.4.3. Fecal immunoglobulin A
Concentrations of IgA can be measured in canine feces presumably reflecting IgA synthesis and secretion by the intestinal mucosa.56, 57, 58 Impaired function of IgA‐producing plasma cells and decreased fecal IgA concentrations (relative or absolute IgA deficiency) have been detected in German Shepherd dogs with chronic gastrointestinal disease, which could be a cause or consequence of the disease process.56, 59, 60, 61, 62 Although decreased fecal IgA concentrations might be detected in dogs with CIE, currently published data do not lend enough support for the clinical utility of measuring fecal IgA concentrations in dogs suspected to have CIE.
3.5. Microbiomic biomarkers
3.5.1. Fecal and mucosal intestinal microbiome
Alterations in the fecal bacterial microbiome have been found in dogs with CIE.63, 64, 65, 66 The most significant decreases were detected in Faecalibacterium spp. and Fusobacteria, which are short‐chain fatty acid producers of importance for intestinal health.65 Some differences in the abundance of individual bacterial taxa (ie, an unclassified genus of Neisseriaceae and Bilophila in the duodenum, Burkholderia in the colon) also were identified between the intestinal mucosal microbiome in IRE dogs and dogs with FRE.66
3.5.2. Fecal dysbiosis index
The fecal dysbiosis index indicates changes in the gut bacterial microbiome (ie, presence of normobiosis versus dysbiosis) by evaluating 8 bacterial groups that commonly are altered in dogs with CIE, particularly dogs diagnosed with IRE (ie, Blautia, Clostridium hiranonis, Escherichia coli, Faecalibacterium, Fusobacterium, Streptococcus, Turicibacter, and total bacteria) and distinguishes IRE from health with 74% sensitivity and 95% specificity.67, 68
3.6. Metabolomic biomarkers
3.6.1. Serum metabolite profile
Serum metabolome changes in dogs with CIE were shown to have a distinct signature (ie, a higher abundance of 3‐hydroxybutyrate, hexuronic acid, ribose, and gluconic acid lactone) indicating CIE in dogs to be associated with oxidative stress and also functional changes in the gut microbiome.63
Serum metabolite profiles together with the fecal dysbiosis index might be clinically useful tools and offer an approach to individualized treatment and monitoring, but high expense and lack of wide availability currently limit their application in clinical practice.
3.7. Inflammatory biomarkers
For an inflammatory biomarker to be clinically useful, it ideally should be quantifiable without any temporal delay in expression, secretion, or both to reflect disease severity.11, 14 Specificity for the gastrointestinal tract (ie, organ‐specific expression or by type of specimen) would be another desirable characteristic for which fecal biomarkers hold great promise.12, 13, 17
Biochemical markers that have been evaluated in dogs with CIE are serum C‐reactive protein (CRP), perinuclear anti‐neutrophilic cytoplasmic antibodies (pANCA), serum 3‐bromotyrosine (3‐BrY), urine and fecal N‐methylhistamine (NMH) concentrations, serum and fecal S100A8/A9 complex (calprotectin) concentrations, serum and fecal S100A12 concentrations, serum soluble receptor of advanced glycation end products (sRAGE), several cytokines and chemokines, and intestinal alkaline phosphatase (AP) activity.
3.7.1. Serum C‐reactive protein
C‐reactive protein is a positive type II acute phase protein expressed in the liver69 in response to infection, inflammation, or cancer,70 and the serum CRP concentration is a nonspecific marker of inflammation.71, 72, 73, 74, 75, 76, 77, 78, 79, 80 Quantification of canine CRP in serum can be done using several assay formats,81, 82, 83, 84, 85, 86, 87, 88 all of which have a reference interval of approximately 0–8 mg/L. A high‐sensitivity CRP (hs‐CRP) assay with improved sensitivity for lower serum CRP concentrations89, 90 currently is not routinely available in veterinary medicine.91
High biological variability of serum CRP concentrations in dogs92 limits its utility as a diagnostic biomarker in dogs with CIE. Serum CRP appears to be clinically more useful as a surrogate marker to assess disease progression and response to treatment in dogs with CIE,8, 50, 93, 94, 95 although some studies did not find a relationship with the severity of clinical signs or histologic lesions.9, 96, 97 The serum concentration of CRP must increase or decrease at least 2.7‐fold for this change to be considered clinically relevant.92
A recent study found that a serum CRP concentration ≥9.1 mg/L distinguished dogs with CIE requiring anti‐inflammatory or immunosuppressive treatment from those dogs responding to an elimination diet or antibiotic trial with a sensitivity of 72% and a specificity of 100%.50 With a pretest probability of ~30%, serum CRP has high PPV (~100%) for the diagnosis of IRE or NRE.
3.7.2. Perinuclear anti‐neutrophilic cytoplasmic antibodies
Perinuclear anti‐neutrophilic cytoplasmic antibodies (pANCA) are serum autoantibodies against neutrophil granule components (eg, nuclear histone, proteinase 3, myeloperoxidase) and suspected cross‐reactivity with a gastrointestinal bacterial antigen98 that can be detected by indirect immunofluorescence assays.98, 99
Seropositivity for pANCA (and also pANCA titers) is higher in dogs with FRE (61%‐62%) than in those with IRE or NRE (0%‐37%).95, 99, 100, 101 However, pANCA seropositivity is not specific for CIE and also can occur with other immune‐mediated, infectious, or neoplastic diseases.101, 102 Positivity for pANCA is associated with PLE, protein‐losing nephropathy, or both in Soft Coated Wheaten Terriers, with pANCA positivity being detected >2 years before the onset of hypoalbuminemia.103 Although pANCA is a difficult assay to acquire, it might be useful for diagnosing dogs with FRE and for early detection of protein‐losing disease in Soft Coated Wheaten Terriers (where fecal α1PI also might predict gastrointestinal protein loss before the development of hypoalbuminemia).
3.7.3. Serum 3‐bromotyrosine
3‐Bromotyrosine (3‐BrY) is the stable metabolite of eosinophil peroxidase,104 which is released from eosinophils after their activation and degranulation.105 Thus, serum 3‐BrY is a biomarker of eosinophilic inflammation.104
Histopathologic findings in dogs with CIE can vary and, although the lamina propria cellular infiltrate often is primarily lymphoplasmacytic, an eosinophilic component or mixed inflammation also can be present.2 Dogs with CIE have increased serum 3‐BrY concentrations,106 with dogs requiring anti‐inflammatory or immunosuppressive treatment having higher serum 3‐BrY concentrations compared to dogs responding to an elimination diet.107 However, sensitivity and specificity of the serum 3‐BrY concentration to differentiate dogs with IRE or NRE from those with FRE or ARE have not been determined. Therefore, further research is needed before serum 3‐BrY can be recommended as a biomarker in routine clinical practice.
3.7.4. N‐methylhistamine
N‐methylhistamine (NMH), a stable product of histamine metabolism, is a proinflammatory biomarker of mast cell activation and degranulation.108 It can be measured in serum, urine, and fecal specimens.109, 110
Mast cell‐mediated inflammation appears to be a component in some dogs with CIE.110, 111, 112 Mast cell degranulation and fecal NMH concentrations were increased in Norwegian Lundehunds and Soft Coated Wheaten Terriers with CIE.111, 112 Some dogs of other breeds with CIE (36–43%) also have increased urine NMH concentrations.109, 110 The NMH concentrations correlated with the severity of histologic lesions in 1 study,110 but not with the number of mast cells in the duodenal mucosa or the severity of clinical signs.109, 110 N‐methylhistamine might be a clinically useful biomarker, but sensitivity and specificity remain to be determined.
3.7.5. Fecal and serum S100A8/A9 protein complex
Calprotectin, the S100A8/A9 protein complex, belongs to the S100/calgranulin family of damage‐associated molecular pattern (DAMP) molecules that accumulate at sites of inflammation.113 Calprotectin is expressed and released by activated macrophages and neutrophils, but expression also can be induced in epithelial cells. Calprotectin is a ligand for Toll‐like receptor (TLR) 4, which plays a role in acute and chronic inflammation.114 Mucosal S100‐mRNA concentrations also are upregulated in dogs with CIE.115
Serum calprotectin concentrations are increased in dogs with CIE,47, 94, 116 but serum calprotectin is not specific for the gastrointestinal tract and must be interpreted with caution in corticosteroid‐treated dogs.94, 95, 117, 118
Calprotectin is a stable protein complex that can be extracted and measured in fecal samples.50, 119, 120 Fecal calprotectin concentrations have been evaluated in dogs with chronic gastrointestinal inflammation50, 95, 121 and appear to serve as a useful biomarker of intestinal inflammation in dogs. Fecal calprotectin concentrations are a good surrogate marker of disease severity in dogs with CIE,50, 95, 121 with a cut‐off value of ~50 μg/g feces to identify dogs with severe clinical disease.121 Fecal calprotectin concentration also appears to have utility in predicting the response to treatment in dogs with CIE,50 and concentrations ≥15.2 μg/g distinguish partial responders or nonresponders (PR/NR) from dogs with IRE that achieve complete clinical remission (CR) with a sensitivity of 80% and a specificity of 75%. Given the 45% prevalence of PR/NR in that study, PPV (77%) and NPV (82%) for fecal calprotectin to identify PR/NR are fairly high.
Because fecal calprotectin has been shown to be an indicator of overall gastrointestinal health58 and also can be increased in acute gastrointestinal inflammatory conditions,122 the patient population for fecal calprotectin testing must be carefully selected to gain relevant information from this marker. The possibility of an effect of gastrointestinal neoplasia on fecal calprotectin concentrations remains to be determined.
3.7.6. Fecal and serum S100A12
Calgranulin C (S100A12) is another DAMP molecule that also belongs to the S100/calgranulin protein family123 and has a cellular distribution similar to that of calprotectin.124 The S100A12 protein has a number of different target proteins, such as the pattern recognition receptor RAGE (receptor for advanced glycation end products)125, 126 and plays a central role in the innate and acquired immune response.113, 127
It is a very sensitive and specific marker of localized inflammatory processes, such as gastrointestinal inflammation,128 and can be increased in serum with various inflammatory disorders.47, 116, 118, 129, 130 In contrast to calprotectin, corticosteroid treatment does not affect serum S100A12 concentrations.131
The S100A12 protein also is stable in fecal samples from which it can be extracted and measured using a species‐specific assay.131, 132 Fecal S100A12 concentration also appears to be a useful biomarker of gastrointestinal inflammation in dogs (Table 2).14, 27, 45, 50, 133 Fecal canine S100A12 concentrations are correlated with the severity of clinical signs and endoscopic lesions, but not with the severity of histopathologic changes.50, 133 A fecal S100A12 concentration ≥490 ng/g can distinguish dogs requiring anti‐inflammatory or immunosuppressive treatment (IRE/NRE) from those with FRE or ARE with a sensitivity of 64% and a specificity of 77%.45 Thus, there is a high chance for a dog to respond to elimination diet and antibiotic trial (NPV > 80%) if the fecal S100A12 concentration is <490 ng/g (20%‐40% prevalence of IRE/NRE in CIE dogs5, 45). Fecal S100A12 concentrations ≥2,700 ng/g distinguished dogs with IRE/NRE that were refractory to treatment (NR) from those with at least a PR with a sensitivity of 100% and a specificity of 76%.45 Thus, dogs with IRE are very likely to have at least PR (NPV ~100%) if fecal S100A12 concentration is <2,700 ng/g (11%‐16% pretest probability45, 50). A significant association between increased fecal S100A12 concentrations and disease outcome also was reported in dogs with CIE.27
Fecal S100A12 concentrations also can be increased with acute gastrointestinal inflammatory conditions,122 thus patient selection for use of this marker is important. The possibility of an effect of gastrointestinal neoplasia on fecal S100A12 concentrations remains to be determined.
3.7.7. Soluble receptor for advanced glycation end products
Soluble RAGE (sRAGE) is a truncated variant of transmembrane RAGE, a pattern‐recognition receptor involved in chronic inflammatory disease processes.134 Soluble RAGE functions as an anti‐inflammatory decoy receptor sequestering ligands of RAGE (eg, DAMP molecules such as S100A12) and thus abrogating cellular proinflammatory RAGE signaling,134 rendering sRAGE also a potential therapeutic target in patients with chronic inflammatory diseases.135 The lack of a correlation between serum sRAGE and S100A12 concentrations suggests that sRAGE is a nonspecific decoy receptor in dogs.27
Similar to inflammatory bowel disease in humans,136 serum sRAGE concentrations are significantly decreased in dogs with CIE but do not correlate with the severity of clinical signs, overall histologic lesions, or outcome.27 However, a weak correlation exists between serum sRAGE concentrations and histologic duodenal lesions (Cabrera Garcia, personal communication 2018). Serum sRAGE concentration also may be a good surrogate marker to assess response to treatment in dogs with CIE because serum sRAGE concentrations increased (normalized) only in dogs achieving complete clinical remission.27 However, further studies are needed to evaluate the potential clinical utility of serum sRAGE as a biomarker in dogs with CIE.
3.7.8. Cytokines and chemokines
Several reports describe various components of the cytokine signature (interleukin [IL]‐1β, IL‐2, IL‐4, IL‐5, IL‐6, IL‐8, IL‐10, IL‐12p40, IL‐17, IL‐18, IL‐23, IL‐25, IL‐33, IFN‐γ, TNF‐α, and TGF‐β1) in dogs with CIE,137, 138, 139, 140, 141, 142, 143 all of which do not support a Th1 nor Th2 polarization as described for inflammatory bowel diseases in humans.144, 145 There also is no evidence of a Th17 signature in dogs with CIE.146, 147 One problem with using cytokines as biomarkers in dogs is that there are many cytokine assays available. However, most of them either have not been analytically validated or have failed basic analytical validation. Recently, a multiplex assay that concurrently measures concentrations of IL‐2, IL‐6, IL‐8, and TNF‐α has been analytically validated, and initial studies have shown that dogs with CIE commonly have increases of serum concentrations of IL‐2, IL‐6, and TNF‐α, but not IL‐8 (Buono, personal communication 2018).
The chemokine signature identified in dogs with CIE (ie, CC chemokine ligand [CCL]2, CCL20, CCL25, CCL28, and CXC chemokine ligand [CXCL]8) is characterized by an increase in chemotaxins for T and B lymphocytes.148
Other signaling molecules of the innate immune response that are dysregulated in dogs with CIE include intracellular signaling (eg, nuclear factor‐κB) and intercellular adhesion molecules (eg, vascular cell adhesion molecule [VCAM]‐1, mucosal vascular addressin cell adhesion molecule [MAdCAM]‐1, and intercellular adhesion molecule [ICAM]‐1),141, 149 as well as other gene products involved in innate immunity, inflammation, cell replication, cellular detoxification, iron and calcium transport, intestinal barrier function, and extracellular matrix degradation.115, 150 Evidence of alterations in the serotonergic neuroendocrine system also have been identified in dogs with CIE.151
3.7.9. Intestinal alkaline phosphatase (AP)
Mucosal expression and activity as well as fecal concentrations of intestinal AP were shown to be decreased in dogs with CIE, especially those with moderate or severe disease.152 More research is needed to determine the potential role of fecal AP concentration as a biomarker in dogs with CIE.
3.8. Genomic biomarkers
Genomic biomarkers include single nucleotide polymorphisms (SNPs) in TLR4, TLR5, nucleotide oligomerization domain (NOD)2, and the neutrophil cytosolic factor (NCF2) gene,153, 154, 155, 156, 157, 158, 159, 160, 161 a reduction of genetic diversity (eg, T cell receptor repertoire),162 and also alterations in the expression of several other genes (eg, genes encoding TLR2, TLR4, TLR5, TLR9, caspase‐3, and B‐cell lymphoma [Bcl]‐2).115, 163, 164, 165, 166 Overexpression of some marker genes also was associated with clinical disease severity.164, 165, 166 To date, these genomic markers have been utilized as research tools to investigate the pathogenesis of CIE in dogs, but none of these markers currently have any relevance in routine clinical practice.
3.8.1. Genetic polymorphisms
Polymorphisms in the genes encoding TLR4 (risk‐associated SNPs: A1571T, G1807A), TLR5 (risk‐associated SNP: G22A; protective SNPs: C100T, T1844C; leucine rich repeat domain‐SNP: T443C), and NOD2 (a cytosolic pattern recognition receptor that recognizes peptidoglycan motifs of Gram− and Gram+ bacteria) have been linked to CIE in dogs.153, 154, 155, 156, 157, 158 In addition to breed‐unspecific SNPs, individual breeds may harbor unique SNPs. The functional implication of the TLR5 SNPs associated with CIE is a hyper‐responsiveness of TLR5 to stimulation with flagellin.159, 160 Genetic polymorphisms in the NCF2 gene (which encodes for a subunit of neutrophil nicotinamide adenine dinucleotide phosphate hydrogen [NADPH] oxidase involved in intracellular autophagy) were detected in Boxer dogs with granulomatous colitis.161
3.8.2. Alterations in gene expression
Mucosal expression of TLR9 (intracellular) as well as TLR2 and TLR4 (apical epithelial surface) is increased in dogs with CIE, but correlates only weakly or not at all with disease severity and response to treatment.163, 164, 165 Decreased TLR5 expression can be observed in dogs with CIE, which however also is not correlated with the severity of the disease.164 However, these results remain to be confirmed at the protein level.167
3.9. Cellular biomarkers
3.9.1. Regulatory T cells
Regulatory T cells (Treg) play an important role in cell‐mediated mucosal immunotolerance and were decreased in duodenal biopsy specimens from dogs with CIE.168, 169 Numbers of circulating Treg were also decreased in peripheral blood samples from dogs with CIE (IRE and PLE), and this cellular biomarker might be useful to monitor disease progression (ie, disease activity) and assess response to treatment.170 However, further studies are needed to determine the clinical utility of assessing the disturbance in Treg homeostasis as a cellular biomarker in dogs with CIE.
4. SUMMARY AND OUTLOOK
Several biomarkers have been evaluated and are potentially clinically useful in dogs with CIE. Although the lack of wide availability, expense, and low stability in biological specimens limits the routine analysis of some of these clinically useful small molecules (eg, cytokines and chemokines), a number of biomarkers can easily be quantified in serum, urine, and even feces, or a combination of these biological samples.
Some functional biomarkers, such as serum cobalamin and folate concentrations as well as fecal α1PI are already widely used in dogs with suspected CIE in the clinical setting. Genomic biomarkers currently are not readily available, but may be able to provide the clinician more information about the individual risk of a dog to develop CIE or even a specific form of CIE in the future. Genomic biomarkers also may represent an avenue to individualized treatment and monitoring. Inflammatory biomarkers beyond serum CRP, such as serum 3‐BrY, fecal calprotectin, or fecal S100A12, currently are not widely available, but a human fecal calprotectin assay holds promise for fecal calprotectin measurement in dogs.120 Inflammatory markers appear to be useful surrogate markers for guiding the management of dogs with CIE, especially to predict response to treatment because therapeutic trials often are tedious, invasive diagnostic tests are impractical for evaluating response to treatment, and routine clinicopathologic and histopathologic findings usually are not able to distinguish different forms of CIE in dogs.171, 172 Combining the results of several inflammatory biomarkers (eg, serum CRP and fecal calprotectin50) also may increase the diagnostic accuracy of each individual biomarker.
Incorporating the information gained from several inflammatory biomarkers (ie, cellular components and severity of the inflammatory disease process) and functional biomarkers (ie, digestive, absorptive, and secretory capacity of the gastrointestinal tract as well as the degree of dysbiosis) into an algorithm or several algorithms may provide an improved strategy to manage dogs with CIE in the future. Such a biomarker panel also may be a useful tool to further characterize CIE in dogs, and further research is warranted to identify such algorithms for different subgroups of dogs with CIE.
5. CONCLUSION
Several functional, biochemical, and genomic biomarkers have been evaluated in dogs with CIE over the last 2 decades and great strides have been made increasing the diagnostic and monitoring armamentarium in canine gastroenterology. However, further prospective and longitudinal studies are now needed to critically evaluate the utility of currently established biomarker assays in dogs with CIE in the routine clinical setting.
CONFLICT OF INTEREST DECLARATION
Dr Jörg Steiner is affiliated with the Gastrointestinal Laboratory at the Texas A&M University College of Veterinary Medicine and Biomedical Sciences where some of the biomarker assays reviewed in this article are offered on a fee‐for‐service basis.
INSTITUTIONAL ANIMAL CARE AND USE COMMITTEE (IACUC) OR OTHER APPROVAL DECLARATION
Authors declare no IACUC or other approval was needed.
ACKNOWLEDGMENTS
The authors thank Dr Niels Grützner for critically reviewing this article. They also acknowledge support from the German Research Foundation (DFG) and Leipzig University within the program of Open Access Publishing.
OFF‐LABEL ANTIMICROBIAL DECLARATION
Authors declare no off‐label use of antimicrobials.
Heilmann RM, Steiner JM. Clinical utility of currently available biomarkers in inflammatory enteropathies of dogs. J Vet Intern Med. 2018;32:1495–1508. 10.1111/jvim.15247
Funding information Leipzig University; German Research Foundation
REFERENCES
- 1. Washabau RJ, Day MJ, Willard MD. WSAVA International Gastrointestinal Standardization Group. Endoscopic, biopsy, and histopathologic guidelines for the evaluation of gastrointestinal inflammation in companion animals. J Vet Intern Med. 2010;24:10‐26. [DOI] [PubMed] [Google Scholar]
- 2. Day MJ, Bilzer T, Mansell J, et al. Histopathological standards for the diagnosis of gastrointestinal inflammation in endoscopic biopsy samples from the dog and cat: a report from the World Small Animal Veterinary Association Gastrointestinal Standardization Group. J Comp Pathol. 2008;138:S1‐S43. [DOI] [PubMed] [Google Scholar]
- 3. Dandrieux JR. Inflammatory bowel disease versus chronic enteropathy in dogs: are they one and the same? J Small Anim Pract. 2016;57:589‐599. [DOI] [PubMed] [Google Scholar]
- 4. Allenspach K, Culverwell C, Chan D. Long‐term outcome in dogs with chronic enteropathies: 203 cases. Vet Rec. 2016;178:368. [DOI] [PubMed] [Google Scholar]
- 5. Volkmann M, Steiner JM, Fosgate GT, et al. Chronic diarrhea in dogs – retrospective study in 136 cases. J Vet Intern Med. 2017;31:1043‐1055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Erdmann C, Heilmann RM. Diagnostic and therapeutic approach to chronic inflammatory enteropathies in dogs [Article in German]. Tierärztl Prax (K). 2017;45:317‐327. [DOI] [PubMed] [Google Scholar]
- 7. Dossin O, Lavoué R. Protein‐losing enteropathies in dogs. Vet Clin North Am Small Anim Pract. 2011;41:399‐418. [DOI] [PubMed] [Google Scholar]
- 8. Jergens AE, Schreiner CA, Frank DE, et al. A scoring index for disease activity in canine inflammatory bowel disease. J Vet Intern Med. 2003;17:291‐297. [DOI] [PubMed] [Google Scholar]
- 9. Allenspach K, Wieland B, Gröne A, Gaschen F. Chronic enteropathies in dogs: evaluation of risk factors for negative outcome. J Vet Intern Med. 2007;21:700‐708. [DOI] [PubMed] [Google Scholar]
- 10. Kawano K, Shimakura H, Nagata N, et al. Prevalence of food‐responsive enteropathy among dogs with chronic enteropathy in Japan. J Vet Med Sci. 2016;78:1377‐1380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Parikh NI, Vasan RS. Assessing the clinical utility of biomarkers in medicine. Biomark Med. 2007;1:419‐436. [DOI] [PubMed] [Google Scholar]
- 12. Duvoisin G, Lopez RN, Day AS, et al. Novel biomarkers and the future potential of biomarkers in inflammatory bowel disease. Mediators Inflamm. 2017;2017:1936315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Vermeire S, Van Assche G, Rutgeerts P. Laboratory markers in IBD: useful, magic, or unnecessary toys? Gut. 2006;55:426‐431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Heilmann RM. Evaluation of canine S100A12 and sRAGE as novel disease markers in dogs with inflammatory bowel disease [PhD Thesis]. College Station, TX: Texas A&M University; 2015.
- 15. Strimbu K, Tavel JA. What are biomarkers? Curr Opin HIV AIDS. 2010;5:463‐466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Huss R. Biomarkers In: Atala A, Allickson J, eds. Translational Regenerative Medicine. Oxford: Elsevier; 2015:235‐241. [Google Scholar]
- 17. Viennois E, Zhao Y, Merlin D. Biomarkers of inflammatory bowel disease: from classical laboratory tools to personalized medicine. Inflamm Bowel Dis. 2015;21:2467‐2464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Fraser CG, Harris EK. Generation and application of data on biological variation in clinical chemistry. Crit Rev Clin Lab Sci. 1989;27:409‐437. [DOI] [PubMed] [Google Scholar]
- 19. Geffré A, Friedrichs K, Harr K, et al. Reference values: a review. Vet Clin Pathol. 2009;38:288‐298. [DOI] [PubMed] [Google Scholar]
- 20. Friedrichs KR, Harr KE, Freeman KP, et al. ASCVP reference interval guidelines: determination of de novo reference intervals in veterinary species and other related topics. Vet Clin Pathol. 2012;41:441‐453. [DOI] [PubMed] [Google Scholar]
- 21. Valentin MA, Ma S, Zhao A, et al. Validation of immunoassay for protein biomarkers: bioanalytical study plan implementation to support pre‐clinical and clinical studies. J Pharm Biomed Anal. 2011;55:869‐877. [DOI] [PubMed] [Google Scholar]
- 22. Gunn‐Christie RG, Flatland B, Friedrichs KR, et al. ASVCP quality assurance guidelines: control of preanalytical, analytical, and postanalytical factors for urinalysis, cytology, and clinical chemistry in veterinary laboratories. Vet Clin Pathol. 2012;41:18‐26. [DOI] [PubMed] [Google Scholar]
- 23. Flatland B, Freeman KP, Vap LM, Harr KE. ASVCP. ASVCP guidelines: quality assurance for point‐of‐care testing in veterinary medicine. Vet Clin Pathol. 2013;42:405‐423. [DOI] [PubMed] [Google Scholar]
- 24. Ricós C, Iglesias N, García‐Lario JV, et al. Within‐subject biological variation in disease: collated data and clinical consequences. Ann Clin Biochem. 2007;44:343‐352. [DOI] [PubMed] [Google Scholar]
- 25. Parikh R, Mathai A, Parikh S, et al. Understanding and using sensitivity, specificity and predictive values. Indian J Ophthalmol. 2008;56:45‐50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Simon R. Sensitivity, specificity, PPV, and NPV for predictive biomarkers. JNCI J. 2015;107:djv153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Heilmann RM, Otoni CC, Jergens AE, et al. Systemic levels of the anti‐inflammatory decoy receptor sRAGE (soluble receptor for advanced glycation end products) are decreased in dogs with inflammatory bowel disease. Vet Immunol Immunopathol. 2014;161:184‐192. [DOI] [PubMed] [Google Scholar]
- 28. Wennogle SA, Priestnall SL, Webb CB. Histopathologic characteristics of intestinal biopsy samples from dogs with chronic inflammatory enteropathy with and without hypoalbuminemia. J Vet Intern Med. 2017;31:371‐376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Titmarsh HF, Gow AG, Kilpatrick S, et al. Low vitamin D status is associated with systemic and gastrointestinal inflammation in dogs with a chronic enteropathy. PLoS One. 2015;10:e0137377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Titmarsh H, Gow AG, Kilpatrick S, et al. Association of vitamin D status and clinical outcome in dogs with a chronic enteropathy. J Vet Intern Med. 2015;29:1473‐1478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Heilmann RM, Steiner JM. Laboratory testing for the exocrine pancreas In: Bonagura JD, Twedt DC, eds. Kirk's Current Veterinary Therapy XV. St. Louis, Missouri: Saunders Elsevier; 2014:554‐557. [Google Scholar]
- 32. Kathrani A, Steiner JM, Suchodolski JS, et al. Elevated canine pancreatic lipase immunoreactivity concentration in dogs with inflammatory bowel disease is associated with a negative outcome. J Small Anim Pract. 2009;50:126‐132. [DOI] [PubMed] [Google Scholar]
- 33. Heilmann RM, Berghoff N, Grützner N, et al. Effect of antisecretory therapy and biological variation of serum gastrin concentration in dogs with chronic enteropathies. BMC Vet Res. 2017;13:321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Travis J. Structure, function, and control of neutrophil proteinases. Am J Med. 1988;84:37‐42. [DOI] [PubMed] [Google Scholar]
- 35. Conner JG, Eckersall PD, Ferguson J, Douglas TA. Acute phase response in the dog following surgical trauma. Res Vet Sci. 1988;45:107‐110. [PubMed] [Google Scholar]
- 36. Ganrot K. Plasma protein response in experimental inflammation in the dog. Res Exp Med (Berl). 1973;161:251‐261. [DOI] [PubMed] [Google Scholar]
- 37. Grützner N, Heilmann RM, Hokamp JA, et al. Serum alpha1‐proteinase inhibitor concentrations in dogs with exocrine pancreatic disease, chronic hepatitis, or chronic kidney disease. J Vet Intern Med. 2014;28:1048 (abstract). [DOI] [PubMed] [Google Scholar]
- 38. Heilmann RM, Grützner N, Thames BE, et al. Serum alpha1‐proteinase inhibitor concentrations in dogs with systemic inflammatory response syndrome (SIRS) or sepsis. J Vet Emerg Crit Care (San Antonio). 2017;27:674‐683. [DOI] [PubMed] [Google Scholar]
- 39. Melgarejo T, Williams DA, Griffith G. Isolation and characterization of α1‐protease inhibitor from canine plasma. Am J Vet Res. 1996;57:258‐263. [PubMed] [Google Scholar]
- 40. Heilmann RM, Paddock CG, Ruhnke I, et al. Development and analytical validation of a radioimmunoassay for the measurement of α1‐proteinase inhibitor concentrations in feces from healthy puppies and adult dogs. J Vet Diagn Invest. 2011;23:476‐485. [DOI] [PubMed] [Google Scholar]
- 41. Vaden SL, Vidaurri A, Levine JF, et al. Fecal α1‐proteinase inhibitor activity in Soft Coated Wheaten Terriers. J Vet Intern Med. 2002;16:382 (abstract). [Google Scholar]
- 42. Murphy KF, German AJ, Ruaux CG, et al. Fecal alpha1‐proteinase inhibitor concentration in dogs with chronic gastrointestinal disease. Vet Clin Pathol. 2003;32:67‐72. [DOI] [PubMed] [Google Scholar]
- 43. Heilmann RM, Parnell NK, Grützner N, et al. Serum and fecal canine alpha1‐proteinase inhibitor concentrations reflect the severity of intestinal crypt abscesses and/or lacteal dilation in dogs. Vet J. 2016;207:131‐139. [DOI] [PubMed] [Google Scholar]
- 44. Steiner JM, Ruaux CG, Miller MD, et al. Intra‐individual variability of fecal α1‐proteinase inhibitor concentration in clinically healthy dogs. J Vet Intern Med. 2003;17:445 (abstract). [Google Scholar]
- 45. Heilmann RM, Volkmann M, Otoni CC, et al. Fecal S100A12 concentration predicts a lack of response to treatment in dogs affected with chronic enteropathy. Vet J. 2016;215:96‐100. [DOI] [PubMed] [Google Scholar]
- 46. Grützner N, Heilmann RM, Bridges CS, et al. Serum concentrations of canine alpha1‐proteinase inhibitor in cobalamin‐deficient Yorkshire Terrier dogs. J Vet Diagn Invest. 2013;25:376‐385. [DOI] [PubMed] [Google Scholar]
- 47. Equilino M, Théodoloz V, Gorgas D, et al. Evaluation of serum biochemical marker concentrations and survival time in dogs with protein‐losing enteropathy. J Am Vet Med Assoc. 2015;246:91‐99. [DOI] [PubMed] [Google Scholar]
- 48. Heilmann RM, Ruaux CG, Burgener IA, et al. Serum alpha1‐proteinase inhibitor concentrations in healthy dogs – method validation and determination of reference interval and intra‐individual variation. Vet Clin Pathol. 2013;42:190‐195. [DOI] [PubMed] [Google Scholar]
- 49. Berghoff N, Parnell NK, Hill SL, et al. Serum cobalamin and methylmalonic acid concentrations in dogs with chronic gastrointestinal disease. Am J Vet Res. 2013;74:84‐89. [DOI] [PubMed] [Google Scholar]
- 50. Heilmann RM, Berghoff N, Mansell J, et al. Association of fecal calprotectin concentrations with disease severity, response to treatment, and other biomarkers in dogs with chronic inflammatory enteropathies. J Vet Intern Med. 2018;32:679‐692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Toresson L, Steiner JM, Suchodolski JS, Spillmann T. Oral cobalamin supplementation in dogs with chronic enteropathies and hypocobalaminemia. J Vet Intern Med. 2016;30:101‐107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Toresson L, Steiner JM, Razdan P, et al. Comparison of efficacy of oral and parenteral cobalamin supplementation in normalising low cobalamin concentrations in dogs: a randomised controlled study. Vet J. 2018;232:27‐32. [DOI] [PubMed] [Google Scholar]
- 53. Grützner N, Heilmann RM, Stupka KC, et al. Serum homocysteine and methylmalonic acid concentrations in Chinese Shar‐Pei dogs with cobalamin deficiency. Vet J. 2013;197:420‐426. [DOI] [PubMed] [Google Scholar]
- 54. Berghoff N, Suchodolski JS, Steiner JM. Association between serum cobalamin and methylmalonic acid concentrations in dogs. Vet J. 2012;191:306‐311. [DOI] [PubMed] [Google Scholar]
- 55. Ruaux CG. Laboratory testing for the diagnosis of intestinal disorders In: Steiner JM, ed. Small Animal Gastroenterology. Hannover. Germany: Schlütersche; 2008:50‐55. [Google Scholar]
- 56. Tress U, Suchodolski JS, Williams DA, Steiner JM. Development of a fecal sample collection strategy for extraction and quantification of fecal immunoglobulin A in dogs. Am J Vet Res. 2006;67:1756‐1759. [DOI] [PubMed] [Google Scholar]
- 57. Grellet A, Mila H, Heilmann RM, et al. Effect of age, gestation, and lactation on fecal immunoglobulin A and calprotectin concentrations in dogs. J Nutr Sci. 2014;3:e41‐e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Grellet A, Heilmann RM, Polack B, et al. Influence of breed size, age, fecal quality, and enteropathogen shedding on fecal calprotectin and immunoglobulin A concentrations in puppies during the weaning period. J Vet Intern Med. 2016;30:1056‐1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Batt RM, Barnes A, Rutgers HC, Carter SD. Relative IgA deficiency and small intestinal bacterial overgrowth in German Shepard dogs. Res Vet Sci. 1991;50:106‐111. [DOI] [PubMed] [Google Scholar]
- 60. German AJ, Hall EJ, Day MJ. Relative deficiency in IgA production by duodenal explants from German Shepherd dogs with small intestinal disease. Vet Immunol Immunopathol. 2000;76:25‐43. [DOI] [PubMed] [Google Scholar]
- 61. Littler RM, Batt RM, Lloyd DH. Total and relative deficiency of gut mucosal IgA in German Shepherd dogs demonstrated by faecal analysis. Vet Rec. 2006;158:334‐341. [DOI] [PubMed] [Google Scholar]
- 62. Lee A, Kathrani A, Priestnall SL, et al. Lack of correlation between mucosal immunoglobulin A‐positive plasma cell numbers and TLR5 genotypes in German shepherd dogs with idiopathic chronic enteropathy. J Comp Pathol. 2015;152:201‐205. [DOI] [PubMed] [Google Scholar]
- 63. Minamoto Y, Otoni CC, Steelman SM, et al. Alteration of the fecal microbiota and serum metabolit profiles in dogs with idiopathic inflammatory bowel disease. Gut Microbes. 2015;6:33‐47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Suchodolski JS, Xenoulis PG, Paddock CG, et al. Molecular analysis of the bacterial microbiota in duodenal biopsies from dogs with idiopathic inflammatory bowel disease. Vet Microbiol. 2010;142:394‐400. [DOI] [PubMed] [Google Scholar]
- 65. Suchodolski JS, Markel ME, Garcia‐Mazcorro JF, et al. The fecal microbiome in dogs with acute diarrhea and idiopathic inflammatory bowel disease. PLoS One. 2012;7:e51907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Kalenyak K, Isaiah A, Heilmann RM, et al. Comparison of the intestinal mucosal microbiota in dogs diagnosed with idiopathic inflammatory bowel disease and dogs with food‐responsive diarrhea before and after treatment. FEMS Microbiol Ecol. 2018;94:173. [DOI] [PubMed] [Google Scholar]
- 67. Suchodolski JS. Diagnosis and interpretation of intestinal dysbiosis in dogs and cats. Vet J. 2016;215:30‐37. [DOI] [PubMed] [Google Scholar]
- 68. AlShawaqfeh MK, Wajid B, Minamoto Y, et al. A dysbiosis index to assess microbial changes in fecal samples of dogs with chronic inflammatory enteropathy. FEMS Microbiol Ecol. 2017;93:136. [DOI] [PubMed] [Google Scholar]
- 69. Lewis DH, Chan DL, Pinheiro D, et al. The immunopathology of sepsis: pathogen recognition, systemic inflammation, the compensatory anti‐inflammatory response, and regulatory T cells. J Vet Intern Med. 2012;26:457‐482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Rhodes B, Fürnrohr BG, Vyse TJ. C‐reactive protein in rheumatology: biology and genetics. Nat Rev Rheumatol. 2011;7:282‐289. [DOI] [PubMed] [Google Scholar]
- 71. Dabrowski R, Wawron W, Kostro K. Changes in CRP, SAA and haptoglobin produced in response to ovariohysterectomy in healthy bitches and those with pyometra. Theriogenology. 2007;67:321‐327. [DOI] [PubMed] [Google Scholar]
- 72. Nakamura M, Takahashi M, Ohno K, et al. C‐reactive protein concentration in dogs with various diseases. J Vet Med Sci. 2008;70:127‐131. [DOI] [PubMed] [Google Scholar]
- 73. Griebsch C, Arndt G, Raila J, et al. C‐reactive protein concentration in dogs with primary immune‐mediated hemolytic anemia. Vet Clin Pathol. 2009;38:421‐425. [DOI] [PubMed] [Google Scholar]
- 74. Lowrie M, Penderis J, Eckersall PD, et al. The role of acute phase proteins in diagnosis and management of steroid‐responsive meningitis arteritis in dogs. Vet J. 2009;182:125‐130. [DOI] [PubMed] [Google Scholar]
- 75. Chan DL, Rozanski EA, Freeman LM. Relationship among plasma amino acids, C‐reactive protein, illness severity, and outcome in critically ill dogs. J Vet Intern Med. 2009;23:559‐563. [DOI] [PubMed] [Google Scholar]
- 76. Gebhardt C, Hirschberger J, Rau S, et al. Use of C‐reactive protein to predict outcome in dogs with systemic inflammatory response syndrome or sepsis. J Vet Emerg Crit Care (San Antonio). 2009;19:450‐458. [DOI] [PubMed] [Google Scholar]
- 77. Reimann MJ, Ljungvall I, Hillström A, et al. Increased serum C‐reactive protein concentration in dogs with congestive heart failure due to myxomatous mitral valve disease. Vet J. 2016;209:113‐118. [DOI] [PubMed] [Google Scholar]
- 78. Hillström A, Bylin J, Hagman R, et al. Measurement of serum C‐reactive protein concentration for discriminating between suppurative arthritis and osteoarthritis in dogs. BMC Vet Res. 2016;12:240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Grobman M, Outi H, Rindt H, Reinero C. Serum thymidine kinase 1, canine C‐reactive protein, haptoglobin, and vitamin D concentrations in dogs with immune‐mediated hemolytic anemia, thrombocytopenia, and polyarthropathy. J Vet Intern Med. 2017;31:1430‐1440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Gommeren K, Desmas I, Garcia A, et al. Inflammatory cytokine and C‐reactive protein concentrations in dogs with systemic inflammatory response syndrome. J Vet Emerg Crit Care (San Antonio). 2018;28:9‐19. [DOI] [PubMed] [Google Scholar]
- 81. Eckersall PD, Conner JG, Harvie J. An immunoturbidimetric assay for canine C‐reactive protein. Vet Res Commun. 1991;15:17‐24. [DOI] [PubMed] [Google Scholar]
- 82. Berghoff N, Suchodolski JS, Steiner JM. Assessment of stability and determination of a reference range for canine C‐reactive protein in serum. J Vet Intern Med. 2006;20:791 (abstract). [Google Scholar]
- 83. Parra MD, Tuomola M, Cabezas‐Herrera J, Cerón JJ. Analytical and clinical validation of a time‐resolved immunofluorometric assay (TR‐IFMA) for canine C‐reactive protein in serum. Vet Res Commun. 2006;30:113‐126. [DOI] [PubMed] [Google Scholar]
- 84. Hillström A, Hagman R, Tvedten H, Kjelgaard‐Hansen M. Validation of a commercially available automated canine‐specific immunoturbidimetric method for measuring canine C‐reactive protein. Vet Clin Pathol. 2014;43:235‐243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Plickert HD, Einspanier R, Arndt G, et al. Evaluation of a point‐of‐care test for canine C‐reactive protein. Vet Clin Pathol. 2011;40:384‐388. [DOI] [PubMed] [Google Scholar]
- 86. Klenner S, Bauer N, Moritz A. Evaluation of three automated human immunoturbidimetric assays for the detection of C‐reactive protein in dogs. J Vet Diagn Invest. 2010;22:544‐552. [DOI] [PubMed] [Google Scholar]
- 87. Hindenberg S, Klenner‐Gastreich S, Kneier N, et al. Evaluation of a species‐specific C‐reactive protein assay for the dog on the ABX Pentra 400 clinical chemistry analyzer. BMC Vet Res. 2017;13:146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Piñero M, Pato R, Soler L, et al. A new automated turbidimetric immunoassay for the measurement of canine C‐reactive protein. Vet Clin Pathol. 2018;47:130‐137. [DOI] [PubMed] [Google Scholar]
- 89. Roberts WL, Sedrick R, Moulton L, et al. Evaluation of four automated high‐sensitivity C‐reactive protein methods: implications for clinical and epidemiological applications. Clin Chem. 2000;46:461‐468. [PubMed] [Google Scholar]
- 90. Poullis AP, Zar S, Sundaram KK, et al. A new, highly sensitive assay for C‐reactive protein can aid the differentiation of inflammatory bowel disorders from constipation‐ and diarrhoea‐predominant functional bowel disorders. Eur J Gastroenterol Hepatol. 2002;14:409‐412. [DOI] [PubMed] [Google Scholar]
- 91. Hillström A, Hagman R, Söder J, et al. Validation and application of a canine‐specific automated high‐sensitivity C‐reactive protein assay. J Vet Diagn Invest. 2015;27:182‐190. [DOI] [PubMed] [Google Scholar]
- 92. Carney PC, Ruaux CG, Suchodolski JS, Steiner JM. Biological variability of C‐reactive protein and specific pancreatic lipase immunoreactivity (Spec cPL) in apparently healthy dogs. J Vet Intern Med. 2011;25:825‐830. [DOI] [PubMed] [Google Scholar]
- 93. Jergens AE, Crandell J, Morrison JA, et al. Comparison of oral prednisone and prednisone combined with metronidazole for induction therapy of canine inflammatory bowel disease: a randomized‐controlled trial. J Vet Intern Med. 2010;24:269‐277. [DOI] [PubMed] [Google Scholar]
- 94. Heilmann RM, Jergens AE, Ackermann MR, et al. Serum calprotectin concentrations in dogs with idiopathic inflammatory bowel disease. Am J Vet Res. 2012;73:1900‐1907. [DOI] [PubMed] [Google Scholar]
- 95. Otoni CC, Heilmann RM, García‐Sancho M, et al. Serologic and fecal markers to predict response to induction therapy in dogs with idiopathic inflammatory bowel disease. J Vet Intern Med. 2018;32:999‐1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. McCann TM, Ridyard AE, Else RW, Simpson JW. Evaluation of disease activity markers in dogs with idiopathic inflammatory bowel disease. J Small Anim Pract. 2007;48:620‐625. [DOI] [PubMed] [Google Scholar]
- 97. Münster M, Suchodolski JS, Bilzer T, et al. Influence of physiological disturbances on treatment success of dietary therapy in dogs with chronic enteropathies. Berl Munch Tierarztl Wochenschr. 2010;123:74‐82. [PubMed] [Google Scholar]
- 98. Allenspach K, Luckschander N, Styner M, et al. Evaluation of assays for perinuclear antineutrophilic cytoplasmic antibodies and antibodies to Saccharomyces cerevisiae in dogs with inflammatory bowel disease. Am J Vet Res. 2004;65:1279‐1283. [DOI] [PubMed] [Google Scholar]
- 99. Florey J, Viall A, Streu S, et al. Use of a granulocyte immunofluorescence assay designed for humans for detection of antineutrophil cytoplasmic antibodies in dogs with chronic enteropathies. J Vet Intern Med. 2017;31:1062‐1066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Luckschander N, Allenspach K, Hall J, et al. Perinuclear antineutrophilic cytoplasmic antibody and response to treatment in diarrheic dogs with food responsive disease or inflammatory bowel disease. J Vet Intern Med. 2006;20:221‐227. [DOI] [PubMed] [Google Scholar]
- 101. Mancho C, Sainz Á, García‐Sancho M, et al. Evaluation of perinuclear antineutrophilic cytoplasmic antibodies in sera from dogs with inflammatory bowel disease or intestinal lymphoma. Am J Vet Res. 2011;72:1333‐1337. [DOI] [PubMed] [Google Scholar]
- 102. Karagianni AE, Solano‐Gallego L, Breitschwerdt EB, et al. Perinuclear antineutrophil cytoplasmic autoantibodies in dogs infected with various vector‐borne pathogens and in dogs with immune‐mediated hemolytic anemia. Am J Vet Res. 2012;73:1403‐1409. [DOI] [PubMed] [Google Scholar]
- 103. Allenspach K, Lomas B, Wieland B, et al. Evaluation of perinuclear anti‐neutrophilic cytoplasmic autoantibodies as an early marker of protein‐losing enteropathy and protein‐losing nephropathy in Soft Coated Wheaten Terriers. Am J Vet Res. 2008;69:1301‐1304. [DOI] [PubMed] [Google Scholar]
- 104. Wu W, Chen Y, d'Avignon A, Hazen SL. 3‐Bromotyrosine and 3,5‐dibromotyrosine are major products of protein oxidation by eosinophil peroxidase: potential markers for eosinophil‐dependent tissue injury in vivo. Biochemistry. 1999;38:3538‐3548. [DOI] [PubMed] [Google Scholar]
- 105. Bastan I, Robinson NA, Ge XN, et al. Assessment of eosinophil peroxidase as a potential diagnostic and prognostic marker in dogs with inflammatory bowel disease. Am J Vet Res. 2017;78:36‐41. [DOI] [PubMed] [Google Scholar]
- 106. Sattasathuchana P, Grützner N, Lopes R, et al. Stability of 3‐bromotyrosine in serum and serum 3‐bromotyrosine concentrations in dogs with gastrointestinal diseases. BMC Vet Res. 2015;11:5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Sattasathuchana P, Allenspach K, Lopes R, et al. Evaluation of serum 3‐bromotyrosine concentrations in dogs with steroid‐responsive diarrhea and food‐responsive diarrhea. J Vet Intern Med. 2017;31:1056‐1061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. He SH. Key role of mast cells and their major secretory products in inflammatory bowel disease. World J Gastroenterol. 2004;10:309‐318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Anfinsen KP, Berghoff N, Priestnall SL, et al. Urinary and faecal N‐methylhistamine concentrations do not serve as markers for mast cell activation or clinical disease activity in dogs with chronic enteropathies. Acta Vet Scand. 2014;56:90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Berghoff N, Hill S, Parnell NK, et al. Fecal and urinary N‐methylhistamine concentrations in dogs with chronic gastrointestinal disease. Vet J. 2014;201:289‐294. [DOI] [PubMed] [Google Scholar]
- 111. Berghoff N, Suchodolski JS, Steiner JM. Fecal N‐methylhistamine concentrations in Norwegian Lundehunds with gastrointestinal disease. J Vet Intern Med. 2008;22:748 (abstract). [Google Scholar]
- 112. Vaden SL, Hammerberg B, Orton SM, et al. Mast cell degranulation responses in Soft‐Coated Wheaten Terriers with protein‐losing enteropathy and/or nephropathy. J Vet Intern Med. 2000;14:348 (abstract). [DOI] [PubMed] [Google Scholar]
- 113. Foell D, Wittkowski H, Vogl T, Roth J. S100 proteins expressed in phagocytes: a novel group of damage‐associated molecular pattern molecules. J Leukoc Biol. 2007;81:28‐37. [DOI] [PubMed] [Google Scholar]
- 114. Vogl T, Tenbrock K, Ludwig S, et al. Mrp8 and Mrp14 are endogenous activators of Toll‐like receptor 4, promoting lethal, endotoxin‐induced shock. Nat Med. 2007;13:1042‐1049. [DOI] [PubMed] [Google Scholar]
- 115. Wilke VL, Nettleton D, Wymore MJ, et al. Gene expression in intestinal mucosal biopsy specimens obtained from dogs with chronic enteropathy. Am J Vet Res. 2012;73:1219‐1229. [DOI] [PubMed] [Google Scholar]
- 116. Heilmann RM, Allenspach K, Procoli F, et al. Serum calgranulin concentrations in dogs with inflammatory bowel disease. J Vet Intern Med. 2011;25:1486 (abstract). [Google Scholar]
- 117. Barr JW, Black DM, Perrett JJ, et al. Serum cS100A12 and cS100A8/A9 concentrations in critically ill dogs. J Vet Emerg Crit Care. 2012;22:S23 (abstract). [Google Scholar]
- 118. Heilmann RM, Xenoulis PG, Müller K, et al. Serum S100/calgranulin concentrations in Miniature Schnauzers with idiopathic hyperlipidemia. J Vet Intern Med. 2018;32:535 (abstract). [Google Scholar]
- 119. Heilmann RM, Suchodolski JS, Steiner JM. Development and analytic validation of a radioimmunoassay for the quantification of canine calprotectin in serum and feces from dogs. Am J Vet Res. 2008;69:845‐853. [DOI] [PubMed] [Google Scholar]
- 120. Truar K, Nestler J, Schwarz J, et al. Feasibility of measuring fecal calprotectin concentrations in dogs and cats by the fCAL® turbo immunoassay. J Vet Intern Med. 2018;32:580 (abstract). [Google Scholar]
- 121. Grellet A, Heilmann RM, Lecoindre P, et al. Fecal calprotectin concentrations in adult dogs with chronic diarrhea. Am J Vet Res. 2013;74:706‐711. [DOI] [PubMed] [Google Scholar]
- 122. Heilmann RM, Guard MM, Steiner JM, et al. Fecal inflammatory biomarkers and microbial changes in dogs with acute hemorrhagic diarrhea syndrome (AHDS). J Vet Emerg Crit Care. 2017;27:586‐589. [DOI] [PubMed] [Google Scholar]
- 123. Heizmann CW. The multifunctional S100 protein family In: Vogel HJ, ed. Methods in Molecular Biology. Calcium‐Binding Protein Protocols, Reviews and Case Studies. 1st ed. Totowa: Humana Press; 2002:69‐80. [DOI] [PubMed] [Google Scholar]
- 124. Vogl T, Pröpper C, Hartmann M, et al. S100A12 is expressed exclusively by granulocytes and acts independently from MRP8 and MRP14. J Biol Chem. 1999;274:25291‐25296. [DOI] [PubMed] [Google Scholar]
- 125. Hofmann MA, Drury S, Fu C, et al. RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell. 1999;97:889‐901. [DOI] [PubMed] [Google Scholar]
- 126. Pietzsch J, Hoppmann S. Human S100A12: a novel key player in inflammation? Amino Acids. 2008;36:381‐389. [DOI] [PubMed] [Google Scholar]
- 127. Gebhardt C, Riehl A, Durchdewald M, et al. RAGE signaling sustains inflammation and promotes tumor development. J Exp Med. 2008;205:275‐285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Foell D, Wittkowski H, Roth J. Monitoring disease activity by stool analyses: from occult blood to molecular markers of intestinal inflammation and damage. Gut. 2009;58:859‐868. [DOI] [PubMed] [Google Scholar]
- 129. Grützner N, Heilmann RM, Cranford SM, et al. Inflammatory, immunological, and intestinal disease biomarkers in Chinese Shar Peis with marked hypocobalaminemia. J Vet Diagn Invest. 2015;27:31‐40. [DOI] [PubMed] [Google Scholar]
- 130. Craig SM, Lidbury JA, Fry JK, et al. Serum C‐reactive protein and S100A12 concentrations in dogs with hepatic disease. J Small Anim Pract. 2016;57:459‐464. [DOI] [PubMed] [Google Scholar]
- 131. Heilmann RM, Cranford SM, Ambrus A, et al. Methodological and preanalytical validation of an enzyme‐linked immunosorbent assay for the measurement of canine S100A12. Vet Clin Pathol. 2016;45:135‐147. [DOI] [PubMed] [Google Scholar]
- 132. Heilmann RM, Lanerie DJ, Ruaux CG, et al. Development and analytic validation of an immunoassay for the quantification of canine S100A12 in serum and fecal samples and its biological variation in serum from healthy dogs. Vet Immunol Immunopathol. 2011;144:200‐209. [DOI] [PubMed] [Google Scholar]
- 133. Heilmann RM, Grellet A, Allenspach K, et al. Association between fecal S100A12 concentration and histologic, endoscopic, and clinical disease severity in dogs with idiopathic inflammatory bowel disease. Vet Immunol Immunopathol. 2014;158:156‐166. [DOI] [PubMed] [Google Scholar]
- 134. Bierhaus A, Humpert PM, Morcos M, et al. Understanding RAGE, the receptor for advanced glycation end products. J Mol Med. 2005;83:876‐886. [DOI] [PubMed] [Google Scholar]
- 135. Hudson BI, Bucciarelli LG, Wendt T, et al. Blockade of the receptor for advanced glycation end products: a new target for therapeutic intervention in diabetic complications and inflammatory disorders. Arch Biochem Biophys. 2003;419:80‐88. [DOI] [PubMed] [Google Scholar]
- 136. Meijer B, Hoskin T, Ashcroft A, et al. Total soluble and endogenous secretory receptor for advanced glycation end products (RAGE) in IBD. J Crohns Colitis. 2014;8:513‐520. [DOI] [PubMed] [Google Scholar]
- 137. German AJ, Helps CR, Hall EJ, Day MJ. Cytokine mRNA expression in mucosal biopsies from German shepherd dogs with small intestinal enteropathies. Dig Dis Sci. 2000;45:7‐17. [DOI] [PubMed] [Google Scholar]
- 138. Peters IR, Helps CR, Calvert EL, et al. Cytokine mRNA quantification in duodenal mucosa from dogs with chronic enteropathies by real‐time reverse transcriptase polymerase chain reaction. J Vet Intern Med. 2005;19:644‐653. [DOI] [PubMed] [Google Scholar]
- 139. Jergens AE, Sonea IM, O'Connor AM, et al. Intestinal cytokine mRNA expression in canine inflammatory bowel disease: a meta‐analysis with critical appraisal. Comp Med. 2009;59:153‐162. [PMC free article] [PubMed] [Google Scholar]
- 140. Kołodziejska‐Sawerska A, Rychlik A, Depta A, et al. Cytokines in canine inflammatory bowel disease. Pol J Vet Sci. 2013;16:165‐171. [DOI] [PubMed] [Google Scholar]
- 141. Okanishi H, Kabeya H, Maruyama S, et al. Activation of nuclear factor‐kappa B and cell adhesion molecule mRNA expression in duodenal mucosa of dogs with lymphocytic‐plasmacytic enteritis. Vet Immunol Immunopathol. 2013;154:145‐152. [DOI] [PubMed] [Google Scholar]
- 142. Tamura Y, Ohta H, Yokoyama N, et al. Evaluation of selected cytokine gene expression in colonic mucosa from dogs with idiopathic lymphocytic‐plasmacytic colitis. J Vet Med Sci. 2014;76:1407‐1410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Osada H, Ogawa M, Hasegawa A, et al. Expression of epithelial cell‐derived cytokine genes in the duodenal and colonic mucosae of dogs with chronic enteropathy. J Vet Med Sci. 2017;79:393‐397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Neurath MF, Finotto S, Glimcher LH. The role of Th1/Th2 polarization in mucosal immunity. Nat Med. 2002;8:567‐573. [DOI] [PubMed] [Google Scholar]
- 145. Heilmann RM, Suchodolski JS. Is inflammatory bowel disease in dogs and cats associated with a Th1 or Th2 polarization? Vet Immunol Immunopathol. 2015;168:131‐134. [DOI] [PubMed] [Google Scholar]
- 146. Schmitz S, Garden OA, Werling D, Allenspach K. Gene expression of selected signature cytokines of T cell subsets in duodenal tissues of dogs with and without inflammatory bowel disease. Vet Immunol Immunopathol. 2012;146:87‐91. [DOI] [PubMed] [Google Scholar]
- 147. Ohta H, Takada K, Sunden Y, et al. CD4+ T cell cytokine gene and protein expression in duodenal mucosa of dogs with inflammatory bowel disease. J Vet Med Sci. 2014;76:409‐414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Maeda S, Ohno K, Nakamura K, et al. Quantification of chemokine and chemokine receptor gene expression in duodenal mucosa of dogs with inflammatory bowel disease. Vet Immunol Immunopathol. 2011;144:290‐298. [DOI] [PubMed] [Google Scholar]
- 149. Luckschander N, Hall JA, Gaschen F, et al. Activation of nuclear factor‐kappa B in dogs with chronic enteropathies. Vet Immunol Immunopathol. 2010;133:228‐236. [DOI] [PubMed] [Google Scholar]
- 150. Jergens AE, Nettleton D, Suchodolski JS, et al. Interplay of commensal bacteria, host gene expression, and clinical disease activity in the pathogenesis of canine inflammatory bowel disease. J Vet Intern Med. 2010;24:1570 (abstract). [Google Scholar]
- 151. Bailey C, Ruaux C, Stang BV, Valentine BA. Expression of serotonin, chromogranin‐A, serotonin receptor‐2B, tryptophan hydroxylase‐1, and serotonin reuptake transporter in the intestine of dogs with chronic enteropathy. J Vet Diagn Invest. 2016;28:271‐278. [DOI] [PubMed] [Google Scholar]
- 152. Ide K, Kato K, Sawa Y, et al. Comparison of the expression, activity, and fecal concentration of intestinal alkaline phosphatase between healthy dogs and dogs with chronic enteropathy. Am J Vet Res. 2016;77:721‐729. [DOI] [PubMed] [Google Scholar]
- 153. Kathrani A, House A, Catchpole B, et al. Polymorphisms in the TLR4 and TLR5 gene are significantly associated with inflammatory bowel disease in German shepherd dogs. PLoS One. 2010;5:e15740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Kathrani A, House A, Werling D, et al. Overdominant single nucleotide polymorphisms in the nucleotide oligomerization domain two (NOD2) gene are significantly associated with canine inflammatory bowel disease. J Vet Intern Med. 2010;24:725 (abstract). [Google Scholar]
- 155. Allenspach K. Clinical immunology and immunopathology of the canine and feline intestine. Vet Clin North Am Small Anim Pract. 2011;41:345‐360. [DOI] [PubMed] [Google Scholar]
- 156. Kathrani A, House A, Catchpole B, et al. Breed‐independent toll‐like receptor 5 polymorphisms show association with canine inflammatory bowel disease. Tissue Antigens. 2011;78:94‐101. [DOI] [PubMed] [Google Scholar]
- 157. Kathrani A, Werling D, Allenspach K. Mutational analysis of the TLR5 gene in Boxer dogs with inflammatory bowel disease reveals novel polymorphisms in the leucine rich repeat domain. J Vet Intern Med. 2011;25:696 (abstract). [Google Scholar]
- 158. Kathrani A, Lee H, White C, et al. Association between nucleotide oligomerization domain two (Nod2) gene polymorphisms and canine inflammatory bowel disease. Vet Immunol Immunopathol. 2014;161:32‐41. [DOI] [PubMed] [Google Scholar]
- 159. Allenspach K, Kathrani A, House A , et al. Toll‐like receptor 5 mutations in German Shepherd Dogs are hyper‐responsive to stimulation with flagellin. J Vet Intern Med. 2011;25:688 (abstract). [Google Scholar]
- 160. Kathrani A, Werling D, Holder A, et al. Polymorphisms in Toll‐like receptor 5 results in hyper‐responsiveness to flagellin. J Vet Intern Med. 2011;25:687‐688 (abstract). [Google Scholar]
- 161. Craven M, Acland GM, Mezey J, et al. Genome‐wide association scan reveals polymorphisms in the P67phox subunit (Ncf2) of the NADPH oxidase complex in Boxer dogs with adherent and invasive E.coli‐associated granulomatous colitis: a potential model of chronic granulomatous disease. Gastroenterology. 2010;138:S683 (abstract). [Google Scholar]
- 162. Olivero D, Turba ME, Gentilini F. Reduced diversity of immunoglobulin and T‐cell receptor gene rearrangements in chronic inflammatory gastrointestinal diseases in dogs. Vet Immunol Immunopathol. 2011;144:337‐345. [DOI] [PubMed] [Google Scholar]
- 163. Burgener IA, König A, Allenspach K, et al. Upregulation of toll‐like receptors in chronic enteropathies in dogs. J Vet Intern Med. 2008;22:553‐560. [DOI] [PubMed] [Google Scholar]
- 164. Allenspach K, House A, Smith K, et al. Evaluation of mucosal bacteria and histopathology, clinical disease activity and expression of Toll‐like receptors in German Shepherd Dogs with chronic enteropathies. Vet Microbiol. 2010;146:326‐335. [DOI] [PubMed] [Google Scholar]
- 165. McMahon LA, House AK, Catchpole B, et al. Expression of Toll‐like receptor 2 in duodenal biopsies from dogs with inflammatory bowel disease is associated with severity of disease. Vet Immunol Immunopathol. 2010;135:158‐163. [DOI] [PubMed] [Google Scholar]
- 166. Dandrieux JR, Bornand VF, Doherr MG, et al. Evaluation of lymphocyte apoptosis in dogs with inflammatory bowel disease. Am J Vet Res. 2008;69:1279‐1285. [DOI] [PubMed] [Google Scholar]
- 167. Heilmann RM, Allenspach K. Pattern recognition receptors: signaling pathways and dysregulation in canine chronic enteropathies – brief review. J Vet Diagn Invest. 2017;29:781‐787. [DOI] [PubMed] [Google Scholar]
- 168. Junginger J, Schwittlick U, Lemensieck F, et al. Immunohistochemical investigation of Foxp3 expression in the intestine in healthy and diseased dogs. Vet Res. 2012;43:23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Maeda S, Ohno K, Fujiwara‐Igarashi A, et al. Changes in Foxp3‐positive regulatory T cell number in the intestine of dogs with idiopathic inflammatory bowel disease and intestinal lymphoma. Vet Pathol. 2016;53:102‐112. [DOI] [PubMed] [Google Scholar]
- 170. Volkmann M, Hepworth MR, Ebner F, et al. Frequencies of regulatory T cells in the peripheral blood of dogs with primary immune‐mediated thrombocytopenia and chronic enteropathy: a pilot study. Vet J. 2014;202:630‐633. [DOI] [PubMed] [Google Scholar]
- 171. García‐Sancho M, Rodríguez‐Franco F, Sainz A, et al. Evaluation of clinical, macroscopic, and histopathologic response to treatment in nonhypoproteinemic dogs with lymphocytic‐plasmacytic enteritis. J Vet Intern Med. 2007;21:11‐17. [DOI] [PubMed] [Google Scholar]
- 172. Schreiner NM, Gaschen F, Gröne A, et al. Clinical signs, histology, and CD3‐positive cells before and after treatment of dogs with chronic enteropathies. J Vet Intern Med. 2008;22:1079‐1083. [DOI] [PubMed] [Google Scholar]
- 173. Heilmann RM, Ruaux CG, Grützner N, Suchodolski JS. Steiner, JM . Biological variation of canine calprotectin concentrations in serum. J Vet Intern Med. 2015;29:446 (abstract). [Google Scholar]


