Abstract
Objective
Aging which is accompanied by loss of skeletal muscle and increase of body fat in some adults older than 60 years does not only result in remarkable influences on daily life function but also increases the risk of cardiovascular events. This study used electrical acupuncture together with essential amino acid supplementation to treat sarcopenic obesity (SO) in male older adults.
Methods
A total of 48 male participants with SO (>60 years old) were randomized to electrical acupuncture with oral essential amino acids (EA + AA) or oral essential amino acids alone (AA). Acupuncture points on the limbs were punctured and stimulated electrically once every 3 days for 12 weeks. All participants received essential amino acids orally, twice per day for 28 weeks. Body fat percentage (BFP) and appendicular skeletal muscle index (ASM/H2) was determined by bioelectrical impedance analysis.
Results
Both groups exhibited significant changes in BFP after 12, 20, and 28 weeks compared with baseline values; for ASM/H2, there were significant differences to baseline values after 12, 20, and 28 weeks in the EA + AA group, but only after 28 weeks in the AA group. Between the two groups, there were significant differences in BFP after 12, 20, and 28 weeks, and in ASM/H2 after 20 and 28 weeks.
Conclusion
Both methods decrease BFP and increase ASM/H2. In male older adults, electrical acupuncture with oral essential amino acids is more effective and can increase muscle mass in a shorter time than oral essential amino acids alone.
Keywords: Sarcopenic obesity, Male older adults, Electrical acupuncture, Essential amino acid
Introduction
Sarcopenic obesity (SO) is defined as loss of skeletal muscle and increase of body fat in some adults aged 60 years or older; it carries remarkable impacts on both daily functioning and cardiovascular risk [1, 2]. Fat mass reaches its peak between 60 and 75 years of age; the decrease of muscle mass is beginning near the age of 30 years and accelerates after the age of 60, with an increase of visceral and muscular fat [3, 4]. High levels of inflammation in fatty infiltrates of muscle tissue have been confirmed as an important mechanism of SO; IL-6 and tumor necrosis factor (TNF), secreted from fat cells and macrophages, exacerbate the inflammation, which thereby reduces the quality and strength of muscle tissue [5, 6, 7]. Hormonal changes contribute to the onset of SO, as determined by epidemiological investigations of Asian people: high levels of serum insulin are positively correlated with SO, while low levels of 25-OH-vitamin D are negatively correlated with SO [8, 9]. Some prospective studies have shown that low levels of testicular hormone and growth hormone result in decreased muscle mass in SO patients [10, 11]. Other investigations have revealed that low amounts of exercise and protein intake may induce SO [12, 13].
SO in older adults should receive attention in the clinic because of its heavy burden on the family and society [14]. Resistance training and a high-protein diet seem to be the most effective methods to increase muscle mass and decrease fat [15, 16], but a large proportion of older adults experience joint problems, which limit their ability to engage in training. Moreover, decreased food intake is another problem that faces the high-protein diet approach.
In 2008, it was shown that daily supplementation with 16 g essential amino acids (EAAs) for 8 months can increase lean mass [17]. In recent years, electrical acupuncture has been proven to promote protein synthesis in an aging amyotrophic rat, affecting muscle protein associated with the IGF (insulin-like growth factor)/PKB (protein kinase B) pathway [18]. In our clinical practice, we confirmed the improvement of muscle power in a large number of older obese patients after several months of electrical acupuncture treatment in arms or legs via simple muscle strength tests. We therefore hypothesized that electrical acupuncture may have increased the muscle mass in those patients, especially in those with sarcopenia. When combined with EAA intake, this effect may occur in a shorter period of time. In the present study, we investigated the therapeutic effects of acupuncture combined with oral EAA therapy in SO patients.
Material and Methods
Study Design and Subjects
The research involved 60 male participants aged between 60 and 80 years who had a diagnosis of SO according to the criteria from the Asian Working Group for Sarcopenia [19] and the World Health Organization (WHO). Participants were recruited from the Yuexiu District Community Pension Center in Guangzhou, from January 2016 to October 2017. The study was approved by the Hospital Ethics committee (thereby meeting the standards of the Declaration of Helsinki in its revised version of 1975 and its amendments of 1983, 1989, and 1996); all literate participants gave their signed informed consent, while all illiterate participants gave their verbal consent after detailed verbal explanation of the study by our staff.
Body composition was determined by bioelectrical impedance analysis (BIA). SO participants were evaluated for their appendicular skeletal muscle index (ASM/Height2) as established by the Asian Working Group for Sarcopenia (male ≤ 7.0 kg/m2). Obesity was diagnosed with WHO criteria of body fat percentage (BFP ≥ 25% in men).
Participants were excluded from the study if they suffered from diseases that affect fat metabolism and muscle maintenance: diabetes, hyperthyroidism or hypothyroidism, adrenalism, severe gastrointestinal dysfunction, renal insufficiency, malignant tumor, neuromuscular disease, fracture in the preceding 3 months, hemiplegia, or addiction to cigarettes. Participants with mental defects were also excluded from the study. Further, we observed the preliminary food intake of the participants and excluded those exhibiting abnormal appetites according to Basal Energy Expenditure (BEE) analysis [20]. For this, the participants were asked for weighing every kind of food before cooking and eating, recording every meal and snack, and transmitting the immediate data of food (species and weight) to the researchers over 1 week. We then calculated the total calories intake on account of the data.
The participants were randomized into two groups: the electrical acupuncture and oral EAA group (EA + AA), and the oral EAA group (AA). Randomization was performed by minimization [21], using prognostic factors of age (weight = 2) and BFP (weight = 1); the age was stratified as 60–70 years and 70–80 years; the BFP was stratified as 25–30%, 31–36%, and ≥37%; and the BMI was stratified as 27–33 and 34–40 kg/m2. The predefined participant characteristics were all continuous data, expressed as means and standard deviations (SD). P values of <0.05 were considered to be significant.
Experimental Design and Training Program
A total of 60 male participants were included in the study. Five were excluded before the training, 5 for the intolerance to electrical acupuncture during the experiment in the EA + AA group, and 2 for health problems unrelated to the study in the AA group. Ultimately, 48 participants completed the experiment. The participants in the EA + AA group had been treated with electrical acupuncture using the LI14 and LI11 pair, and the ST31 and ST34 pair on both sides of the body. The four acupuncture points belong to the Yang meridian, and they were chosen because the Yang meridian is associated with activation in traditional Chinese medicine theory [22]. The acupuncture points were punctured 5 mm subcutaneously, using sterile disposable acupuncture needles (CE-0197; Suzhou Hualun Medical Appliance Co., Ltd., Suzhou, China), and were stimulated with an electric stimulator (KWD-808I; Changzhou YinDi Electronic Medical Device Co., Ltd., Changzhou, China) for 20 min with a frequency of 5 Hz, wave duration of 1 ms, and strength of 1.5 mA, once every 3 days for 12 weeks. The acupuncturists employed were two qualified acupuncture physicians who were blinded to the grouping situation; the researchers greeted the participants and informed the physicians of each participant's treatment needs. All participants in each group took EAAs orally twice per day (20 g in total) for 28 weeks. All participants received a reminder phone call for drug intake once every 7 days. Simple food recipes, three meals per day made by the nutritional department of the hospital, were supplied for all participants in the center. The total calories of the food were 1.58 × (13.5 × weight (kg) + 487) [23], with the nutrition distribution of protein 10–15%, fat 20–30% and carbohydrate 55–65%. We weighed the participants every week to adjust the calories of the food. We scheduled the breakfast, lunch, and dinner at 7:00 AM, 12:00 noon, and 7:00 PM. The extra food, if there is any, was also recorded; the food materials were purchased in close contact with the researchers, two cooks were responsible for the meal supply, materials were cooked and mixed solely according to the recipe. Participants were required to take all food we supplied every meal. Those who take less than 80% total calories 5 times would be excluded from the study. Thus, we could control the caloric intake to a great extent. However, we did not intervene regarding the amount of daily exercise; all participants were instructed to engage in daily moderate exercise without fatigue.
The oral EAA mixture (supplied by the nutritional department of the hospital) was administered as a nutritional support snack at 10:00 AM and 5:00 PM. The EAA mixture comprised 10 g/day, divided as follows (based on recommendations of the Food and Agriculture Organization of the United Nations): L-leucine 1.7 g; L-lysine 1.3 g; L-isoleucine 1.3 g; L-valine 1.4 g; L-threonine 1.0 g; L-phenylalanine 2.0 g; L-methionine 1.0 g; and L-tryptophan 0.3 g.
The primary evaluation criteria were ASM/H2 and BFP; the temporal points for the evaluation were before initiation, and 4, 12, and 20 weeks after initiation as well as at the end of the experiment.
We used BIA to detect BFP and ASM. The participants were required to comply with a series of rules before the detection: fast and refrain from alcohol and exercise for at least 12 h, and from food and water for 3 h; relieve the bladder at least 30 min before detection. Wear single layer cloths without any mental ornament. The time for the detection was 8:00–10:00 AM.
In spite of the definite food intake we supplied, we also observed and recorded the extra food intake of the participants. We therefore asked the participants for weighting every kind of extra food before cooking and eating to calculate the extra calories.
Statistical Analysis
SPSS software 18.0 (IBM, Armonk, NY, USA) was applied for analyses, the data were distributed and presented as mean and SD. Statistical differences were analyzed using independent Student's t-test and paired t-test. Differences were considered significant if p < 0.05.
Results
As shown in table 1, no significant differences were observed between the two groups at baseline regarding age, BMI, BFP, and ASM/H2.
Table 1.
Age, BMI, body fat percentage (BFP), appendicular skeletal muscle mass-height index (ASM/H2) and extra calories of two groups at baseline
| Group | Age, years | BMI | BFP, % | ASM/H2, kg/m2 | Extra calories, kcal |
|---|---|---|---|---|---|
| EA+AA (n = 23) | 70.35 ± 5.36 | 33.41 ± 3.33 | 33.17 ± 2.66 | 6.04 ± 0.50 | 1.43 ± 0.19 × 104 |
| AA (n = 25) | 68.80 ± 5.08* | 32.94 ± 3.69** | 31.80 ± 2.83※ | 5.94 ± 0.49※※ | 1.48 ± 0.20 × 104† |
No significant difference from EA (electrical acupuncture) + AA (amino acids) group on baseline
p = 0.310
p = 0.643
p = 0.090
p = 0.510
p = 0.327
As shown in table 2, for BFP in the EA + AA group, there was no significant difference between baseline and 4 weeks, but there were significant differences at 12 weeks, 20 weeks, and 28 weeks; for BFP in the AA group, there was no significant difference between baseline and 4 weeks, but there were significant differences at 12 weeks, 20 weeks, and 28 weeks. For ASM/H2 in the EA + AA group, there was no significant difference between baseline and 4 weeks, but there were significant differences at 12 weeks, 20 weeks, and 28 weeks. For ASM/H2 in the AA group, there was no significant difference between baseline and 4 weeks, 12 weeks, or 20 weeks, but there was a significant difference at 28 weeks.
Table 2.
Mean of the body fat percentage (BFP) and the appendicular skeletal muscle mass-height index (ASM/H2) throughout the experiment
| EA+AA (n = 23) |
AA (n = 25) |
|||
|---|---|---|---|---|
| BFP, % | ASM/H2, kg/m2 | BFP, % | ASM/H2, kg/m2 | |
| Pre | 33.17 ± 2.66 | 6.04 ± 0.50 | 31.80 ± 2.83 | 5.94 ± 0.49 |
| 4 weeks | 32.96 ± 2.88* | 6.07 ± 0.51* | 31.64 ± 2.71* | 5.96 ± 0.48* |
| 12 weeks | 27.30 ± 1.96** | 6.19 ± 0.52** | 30.16 ± 2.84** | 5.96 ± 0.48** |
| 20 weeks | 26.61 ± 1.92 *** | 6.94 ± 0.24*** | 30.16 ± 2.84*** | 5.98 ± 0.45*** |
| 28 weeks | 25.91 ± 1.00**** | 6.97 ± 0.24**** | 29.60 ± 2.75**** | 6.41 ± 0.53**** |
For BFP, compared with the baseline in EA+AA,
p = 0.096
p = 0.000
p = 0.000
p = 0.000
compared with the baseline in AA group,
p = 0.212
p = 0.000
p = 0.000
p = 0.000
compared with the baseline in EA+AA,
p = 0.069
p = 0.000
p = 0.000
p = 0.000
compared with the baseline in AA group,
p = 0.083
p = 0.083
p = 0.125
p = 0.000
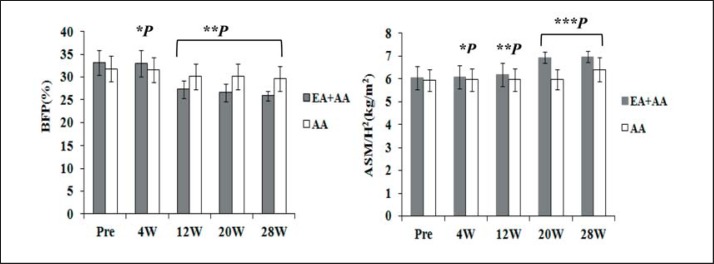
As shown in figure 1, for BFP, there was no significant difference between the two groups at 4 weeks, but there were significant differences between the groups at 12 weeks, 20 weeks, and 28 weeks. For ASM/H2, there was no significant difference between the groups at 4 weeks or 12 weeks, but there were significant differences at 20 weeks and 28 weeks.
Fig. 1.
Body fat percentage (BFP) and appendicular skeletal muscle mass-height index (ASM/H2) after treatment between electrical acupuncture (EA) + amino acids (AA) and AA group. Data are mean ± SEM. For BFP, compared with AA group, *p = 0.110 at 4 weeks, but **p = 0.00 at 12, 20, and 28 weeks. For ASM/H2, compared with AA group, *p = 0.480 at 4 weeks and **p = 0.128 at 12 weeks, but ***p = 0.000 at 20 and 28 weeks.
Discussion
SO has attract geriatricians' interest in recent years for its detrimental effect on physical function and functional ability in older age, as it is associated with changes in skeletal muscle mass and total body fat. In Beijing, a large-scale survey of elderly people in the community showed that many elderly people suffered from SO and that this clinical picture did not receive sufficient attention and treatment [24]. Thus great expenditures of money, resources, and materials are necessary in China to treat cardiovascular diseases and fractures caused by SO. Extended clinical management regarding nutrition as well as maintenance and increase of muscle mass is important for participants suffering from SO.
The data from the present study indicate that both AA and EA + AA therapy can decrease fat mass and increase muscle mass compared to baseline values, but that EA + AA increases muscle mass in a shorter time and continues to produce therapeutic effects for the remaining 16 weeks after completion of EA. Overall, EA + AA is more effective than AA. These observations suggest that EAA supplementation and EA stimulation may be important in clinical management of SO.
A possible mechanism by which acupuncture increases muscle mass is increased muscle blood flow (MBF). In fact, early studies have shown that acupuncture increases MBF in humans [25, 26, 27] and animals [28, 29, 30]. Some researchers suggested that it the elevated MBF is caused by increases in nitric oxide and prostaglandins after acupuncture [31]. Notably, circulation improvement is beneficial for muscle growth and the mitigation of atrophy through enhancement of oxygen content in muscle tissue. Another study revealed that the stimulation of local and distant acupuncture points from the trapezius muscle was able to increase ipsilateral and contralateral muscle strength [32]. Indeed, stimulation in the points of the Yang meridian can increase the pattern of muscle response according to the Traditional Chinese Medicine theory.
In general, muscle stretches with exercise and changes in body position, thereby causing muscle spindle excitation and increased receptor sensitivity due to increased signal impulses from efferent nerves. Acupuncture is correlated with the induction of signal impulses from the muscle spindle [33]. The continuous and stable output of EA stimulation, is suggested to be positively correlated with muscle growth. Compared to manually administered traditional acupuncture, it is more convenient and manageable. Takaoka et al. [34] demonstrated that EA treatment induces a satellite cell-related proliferative reaction in non-atrophic skeletal muscle via the suppression of myostatin expression. Using a senile mouse model, this study uncovered the effectiveness of EA therapy to prevent age-related muscle atrophy. In another investigation, the muscle mass in the EA group was greater than in the control group after 8 months, with a significant increase in related protein synthesis of rat muscle protein [18]. Despite the limitations of human experimentation regarding muscle dry weight, changes in soft tissue can be detected intuitively with BIA; thus, its reliability has been examined in different a variety of patients [35, 36].
Compared with young people, the elderly have lower protein synthesis and require more protein intake to accelerate skeletal protein synthesis; an unreasonable dietary structure for elderly people would be the induction of a change in body composition, accelerating fat growth and loss of muscle [13]. High TNF levels are caused by increased fat mass, which affects protein metabolism directly [6]. According to the present research, correcting dietary composition and supplementation with EAAs could be important for the maintenance and increase of muscle mass. In China, it is proposed that high-quality protein of biological value should comprise half of the daily diet [37]. In fact, raising the ratio of leucine in the diet (>2.8 g) can effectively improve skeletal protein synthesis in older people [38]. Supplement of branched-chain amino acids (BCCA; containing leucine, valine, and isoleucine) increases nitrogen retention and protein synthesis, and raises the flow rate of nitrogen in skeletal muscle [39]. In muscle proteins, BCCA comprises 35% of EAAs, which is absorbed by the thigh muscle directly during exercise.
IL-6 is considered to be a key inflammatory factor, which is highly expressed in obese subjects and is produced by macrophages that have infiltrated fat tissue [40]. Inhibition of lipoprotein enzyme activity is caused by IL-6, which leads to lipid deposition in fat tissue. The possible mechanism for the decreased body fat after EA may be a decline of IL-6 expression as shown in animal experiments [41].
In our study, EA + AA combination therapy was superior to AA monotherapy as EA and EAA supplementation trigger different mechanisms with regard to reversal of muscle atrophy. EA stimulates muscle cells via electrical signals, while EAAs produce chemical signals to reverse aging-related atrophy. The effect can be maintained for several months after EA with the administration of EAAs. During the experiment, some participants retreated because of the unbearable discomfort of EA (sore, swelling, pain), which provides a research prospect for noninvasive acupuncture point stimulation in SO. In consideration of the changes in hormone levels of elderly women, which may differentially affect the metabolism of muscle and fat, the study did not include female participants, which may affect its clinical application. We suggest conducting a study in a female patient population to determine whether or not the observed results can be sustained.
Conclusion
EA with oral EAA supplementation significantly decreased fat mass and increased muscle mass in elderly SO participants. EA has an important practical application in combination clinical management of SO because combined therapy increased muscle mass in a shorter time than oral AA only.
Disclosure Statement
The authors declare that they have no conflicts of interest.
Acknowledgments
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- 1.Pereira FB, Leite AF, Paula AP. Relationship between pre-sarcopenia, sarcopenia and bone mineral density in elderly men. Arch Endocrinol Metab. 2015;59:59–65. doi: 10.1590/2359-3997000000011. [DOI] [PubMed] [Google Scholar]
- 2.Chung JY, Kang HT, Lee DC, Lee HR, Lee YJ. Body composition and its association with cardio metabolic risk factors in the elderly: a focus on sarcopenic obesity. Arch Gerontol Geriatr. 2013;56:270–278. doi: 10.1016/j.archger.2012.09.007. [DOI] [PubMed] [Google Scholar]
- 3.Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. Sarcopenic obesity: definition, cause and consequences. Curr Opin Clin Nutr Metab Care. 2008;11:693–700. doi: 10.1097/MCO.0b013e328312c37d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zoico E, Di Francesco V, Guralnik JM, Mazzali G, Bortolani A, Guariento S, Sergi G, Bosello O, Zamboni M. Physical disability and muscular strength in relation to obesity and different body composition indexes in a sample of healthy elderly women. Int J Obes Relat Metab Disord. 2004;28:234–241. doi: 10.1038/sj.ijo.0802552. [DOI] [PubMed] [Google Scholar]
- 5.Zamboni M, Mazzali G, Fantin F, Rossi A, Di Francesco V. Sarcopenic obesity: a new category of obesity in the elderly. Nutr Metab Cardiovasc Dis. 2008;18:388–395. doi: 10.1016/j.numecd.2007.10.002. [DOI] [PubMed] [Google Scholar]
- 6.Schaap LA, Pluijm SM, Deeg DJ, Visser M. Inflammatory markers and loss of muscle mass (sarcopenia) and strength. Am J Med. 2006;119:526. doi: 10.1016/j.amjmed.2005.10.049. [DOI] [PubMed] [Google Scholar]
- 7.Hung J, McQuillan BM, Thompson PL, Beilby JP. Circulating adiponectin levels associate with inflammatory markers, insulin resistance and metabolic syndrome independent of obesity. Int J Obes (Lond) 2008;32:772–779. doi: 10.1038/sj.ijo.0803793. [DOI] [PubMed] [Google Scholar]
- 8.Kim TN, Park MS, Lim KI, Choi HY, Yang SJ, Yoo HJ, Kang HJ, Song W, Choi H, Baik SH, Choi DS, Choi KM. Relationships between sarcopenic obesity and insulin resistance, inflammation, and vitamin D status: the Korean Sarcopenic Obesity Study. Clin Endocrinol (Oxf) 2013;78:525–532. doi: 10.1111/j.1365-2265.2012.04433.x. [DOI] [PubMed] [Google Scholar]
- 9.Hwang B, Lim JY, Lee J, Choi NK, Ahn YO, Park BJ. Prevalence rate and associated factors of sarcopenic obesity in Korean elderly population. J Korean Med Sci. 2012;27:748–755. doi: 10.3346/jkms.2012.27.7.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schaap LA, Pluijm SM, Smit JH, van Schoor NM, Visser M, Gooren LJ, Lips P. The association of sex hormone levels with poor mobility, low muscle strength and incidence of falls among older men and women. Clin Endocrinol (Oxf) 2005;63:152–160. doi: 10.1111/j.1365-2265.2005.02315.x. [DOI] [PubMed] [Google Scholar]
- 11.Cappola AR, Bandeen RK, Wand GS, Volpato S, Fried LP. Association of IGF-I levels with muscle strength and mobility in older women. J Clin Endocrinol Metab. 2001;86:4139–4146. doi: 10.1210/jcem.86.9.7868. [DOI] [PubMed] [Google Scholar]
- 12.Duvigneaud N, Matton L, Wijndaele K, Deriemaeker P, Lefevre J, Philippaerts R, Thomis M, Delecluse C, Duquet W. Relationship of obesity with physical activity, aerobic fitness and muscle strength in Flemish adults. J Sports Med Phys Fitness. 2008;48:201–210. [PubMed] [Google Scholar]
- 13.Houston DK, Nicklas BJ, Ding J, Harris TB, Tylavsky FA, Newman AB, Lee JS, Sahyoun NR, Visser M, Kritchevsky SB, Health ABC Study Dietary protein intake is associated with lean mass change in older, community-dwelling adults: the Health, Aging, and Body Composition (Health ABC) Study. Am J Clin Nutr. 2008;87:150–155. doi: 10.1093/ajcn/87.1.150. [DOI] [PubMed] [Google Scholar]
- 14.Lim S, Kwon SY, Yoon JW, Kim SY, Choi SH, Park YJ, Yoon HI, Chang YS, Lee JH, Lee CT, Kim KW, Park KS, Jang HC. Association between body composition and pulmonary function in elderly people: the Korean Longitudinal Study on Health and Aging. Obesity (Silver Spring) 2011;19:631–668. doi: 10.1038/oby.2010.167. [DOI] [PubMed] [Google Scholar]
- 15.Benton MJ, Whyte MD, Dyal BW. Sarcopenic obesity: strategies for management. Am J Nurs. 2011;111:38–44. doi: 10.1097/01.NAJ.0000408184.21770.98. quiz 36–45. [DOI] [PubMed] [Google Scholar]
- 16.Gordon MM, Bopp MJ, Easter L, Miller GD, Lyles MF, Houston DK, Nicklas BJ, Kritchevsky SB. Effects of dietary protein on the composition of weight loss in post-menopausal women. J Nutr Health Aging. 2008;12:505–509. doi: 10.1007/BF02983202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Solerte SB, Gazzaruso C, Bonacasa R, Rondanelli M, Zamboni M, Basso C, Locatelli E, Schifino N, Giustina A, Fioravanti M. Nutritional supplements with oral amino acid mixtures increases whole body lean mass and insulin sensitivity in elderly subjects with sarcopenia. Am J Cardiol. 2008;101:69E–77E. doi: 10.1016/j.amjcard.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 18.Ma CC, Xu P, Wei Y. The effect of preventive electroacupuncture on the morphology of musculoskeletal system of different month age rats and the expressions of gap associated protein under the pathway IGF/AKT. Chin J Gerontol. 2016;36:4683–4686. [Google Scholar]
- 19.Chen LK, Liu LK, Woo J, Assantachai P, Auyeung TW, Bahyah KS, Chou MY, Chen LY, Hsu PS, Krairit O, Lee JS, Lee WJ, Lee Y, Liang CK, Limpawattana P, Lin CS, Peng LN, Satake S, Suzuki T, Won CW, Wu CH, Wu SN, Zhang T, Zeng P, Akishita M, Arai H. Sarcopenia in Asia: consensus report of the Asian Working Group for Sarcopenia. J Am Med Dir Assoc. 2014;15:95–101. doi: 10.1016/j.jamda.2013.11.025. [DOI] [PubMed] [Google Scholar]
- 20.Roza AM, Shizgal HM. The Harris Benedict equation reevaluated: resting energy requirements and the body cell mass. Am J Clin Nutr. 1984;40:168–182. doi: 10.1093/ajcn/40.1.168. [DOI] [PubMed] [Google Scholar]
- 21.Scott NW, McPherson GC, Ramsay CR, Campbell MK. The method of minimization for allocation to clinical trials: a review. Control Clin Trials. 2002;23:662–674. doi: 10.1016/s0197-2456(02)00242-8. [DOI] [PubMed] [Google Scholar]
- 22.Guang JW, Hossein AM, Wei BZ. Meridian studies in China: a systematic review. J Acupunct Meridian Stud. 2010;3:1–9. doi: 10.1016/S2005-2901(10)60001-5. [DOI] [PubMed] [Google Scholar]
- 23.Yu K. Vol. 5. Beijing: Peking Union Medical College publishing house; 2004. Clinical Nutrition Iatreusiology; pp. pp 45–48. [Google Scholar]
- 24.Ping M, Yi X, Hu L, Fan L, Zhang Y, Zhang MX, Sun J, Liu Y, Li M, Yang Y, Wang LH, Zhang Y, Gao W, Han XQ, Chen LK. Sarcopenia and sarcopenic obesity among men aged 80 years and older in Beijing: prevalence and its association with functional performance. Geriatr Gerontol Int. 2014;23:29–35. doi: 10.1111/ggi.12211. [DOI] [PubMed] [Google Scholar]
- 25.Sandberg M, Lundeberg T, Lindberg LG, Gerdle B. Effects of acupuncture on skin and muscle blood flow in healthy subjects. Eur J Appl Physiol. 2003;90:114–119. doi: 10.1007/s00421-003-0825-3. [DOI] [PubMed] [Google Scholar]
- 26.Sandberg M, Lindberg LG, Gerdle B. Peripheral effects of needle stimulation (acupuncture) on skin and muscle blood flow in fibromyalgia. Eur J Pain. 2004;8:163–171. doi: 10.1016/S1090-3801(03)00090-9. [DOI] [PubMed] [Google Scholar]
- 27.Sandberg M, Larsson B, Lindberg LG, Gerdle B. Different patterns of blood flow response in the trapezius muscle following needle stimulation (acupuncture) between healthy subjects and patients with fibromyalgia and work-related trapezius myalgia. Eur J Pain. 2005;9:497–510. doi: 10.1016/j.ejpain.2004.11.002. doi:10.1016/j.ejpain.2004.11.002. [DOI] [PubMed] [Google Scholar]
- 28.Shinbara H, Okubo M, Sumiya E, Fukuda F, Yano T, Kitade T. Effects of manual acupuncture with sparrow pecking on muscle blood flow of normal and denervated hindlimb in rats. Acupunct Med. 2008;26:149–159. doi: 10.1136/aim.26.3.149. [DOI] [PubMed] [Google Scholar]
- 29.Shinbara H, Okubo M, Kimura K, Kunio M, Eiji S. Acetylcholine does not participate in the increase in local muscle blood flow following manual acupuncture in rats (in Japanese) JAM. 2011;1:33–38. [Google Scholar]
- 30.Shinbara H, Okubo M, Kimura K, Mizunuma K, Sumiya E. Participation of calcitonin gene related peptide released via axon reflex in the local increase in muscle blood flow following manual acupuncture. Acupunct Med. 2013;31:81–87. doi: 10.1136/acupmed-2012-010253. [DOI] [PubMed] [Google Scholar]
- 31.Shinbara H, Okubo M, Kimura K, Mizunuma K, Sumiya E. Contributions of nitric oxide and prostaglandins to the local increase in muscle blood flow following manual acupuncture in rats. Acupunct Med. 2015;33:65–71. doi: 10.1136/acupmed-2014-010634. [DOI] [PubMed] [Google Scholar]
- 32.Leandro L, de Souza, Fernanda LB, Mucciaroni TS, de Araujo JE. Unilateral and immediate stimulation of acupuncture points Xiaohai (SI8) and Jianwaishu (SI14) of the small intestine meridian increases electromyographic activity and strength in the ipsilateral and contralateral upper trapezius muscle. J Acupunct Meridian Stud. 2016;9:250–256. doi: 10.1016/j.jams.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 33.Ding GH, Shen XY, Dai JH, Liu H, Yao W, Li X. Research and development on the dynamic system for detecting the force of acupuncture process in the clinical practice of Traditional Chinese Medicine. J Biomed Eng. 2003;20:121–124. [PubMed] [Google Scholar]
- 34.Takaoka Y, Ohta M, Ito A, Takamatsu K, Sugano A, Funakoshi K, Takaoka N, Sato N, Yokozaki H, Arizono N, Goto S, Maeda E. Electroacupuncture suppresses myostatin gene expression: cell proliferative reaction in mouse skeletal muscle. Physiol Genomics. 2007;30:102–110. doi: 10.1152/physiolgenomics.00057.2006. [DOI] [PubMed] [Google Scholar]
- 35.Kyle UG, Bosaeus I, De Lorenzo AD, Deurenberg P, Elia M, Gómez JM, Heitmann BL, Kent-Smith L, Melchior JC, Pirlich M, Scharfetter H, Schols AM, Pichard C, Composition of the ESPEN Working Group Bioelectrical impedance analysis - part I: review of principles and methods. Clin Nutr. 2004;23:1226–1243. doi: 10.1016/j.clnu.2004.06.004. [DOI] [PubMed] [Google Scholar]
- 36.Sun G, French CR, Martin GR, Younghusband B, Green RC, Xie YG, Mathews M, Barron JR, Fitzpatrick DG, Gulliver W, Zhang H. Comparison of multifrequency bioelectrical impedance analysis with dual-energy X-ray absorptiometry for assessment of percentage body fat in a large, healthy population. Am J Clin Nutr. 2005;81:74–78. doi: 10.1093/ajcn/81.1.74. [DOI] [PubMed] [Google Scholar]
- 37.Wei JM, Cao WX, Sun JQ, et al. The experts consensus on nutrition therapy in elderly patients the nutritional support for sarcopenia (in Chinese) Chin J Geriatr. 2013;32:913–928. [Google Scholar]
- 38.Verhoeven S, Vanschoonbeek K, Verdijk k LB, Koopman R, Wodzig WK, Dendale P, van Loon LJ. Long term leucine supplementation does not increase muscle mass or strength in healthy elderly men. Am J Clin Nutr. 2009;89:1468–1475. doi: 10.3945/ajcn.2008.26668. [DOI] [PubMed] [Google Scholar]
- 39.Xu ZQ, Sun YM, He H, et al. The effect of branched chain nitrogen acid on protein metabolism of rats in sports (in Chinese) Acta Nutrimenta Sinica. 2001;23:102–105. [Google Scholar]
- 40.Weisberg SP, Mccann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–1800. doi: 10.1172/JCI19246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu ZL, Tang CL, Yu M, et al. Effect of intensities of electro acupuncture on C-reactive protein and interleukin-6 in adipose tissues from high fat diet-induced obese rats. J Chongqing Med Univ. 2011;36:429–432. [Google Scholar]