Abstract
Background
Until recently, lung transplantation was not considered in patients with human immunodeficiency virus (HIV). HIV seropositive patients with suppressed viral loads can now expect long-term survival with the advent of highly active antiretroviral therapies (HAART); however, HIV remains a relative contraindication to lung transplantation. We describe, to our knowledge, the first HIV seropositive lung transplant recipient in Canada. We also review the literature of previously reported cases of solid-organ transplantation in patients with HIV with a focus on immunosuppression considerations.
Case presentation
A 48-year old man received a bilateral lung transplant for a diagnosis of desquamative interstitial pneumonia (DIP) attributed to cigarette and cannabis smoking. His control of HIV infection pre-transplant was excellent on HAART, and he had no other contraindications to lung transplantation. The patient underwent bilateral lung transplantation using basiliximab, methylprednisolone, and mycophenolate mofetil (MMF) as induction immunosuppression. He was maintained on MMF, prednisone, and tacrolimus thereafter, and restarted his HAART regimen immediately post-operatively. His post-transplant course was complicated by Grade A1 minimal acute cellular rejection, as well as an enterovirus/rhinovirus graft infection. Despite these complications, his functional status and control of HIV infection remain excellent 24 months post-transplant.
Conclusions
Our patient is one of only several HIV seropositive lung transplant recipients reported globally. With growing acceptance of transplantation in this population, there is a need for clarification of prognosis post-transplantation, as well as optimal immunosuppression regimens for these patients. This case report adds to the recent literature that suggests HIV seropositivity should not be considered a contraindication to lung transplantation, and that post-transplant patients with HIV can be managed safely with basiliximab, tacrolimus, MMF and prednisone.
Keywords: HIV, Lung transplant, Desquamative interstitial pneumonia, Immunosuppression
Background
Until recently, lung transplantation was not considered in patients with human immunodeficiency virus (HIV) given the poor prognosis associated with being HIV seropositive and the need for additional post-transplant immunosuppression. With the advent of highly active antiretroviral therapies (HAART) for HIV, seropositive patients with suppressed viral loads can now expect long-term survival. Despite these advances, HIV seropositivity remains a relative contraindication to lung transplantation per International Society for Heart and Lung Transplantation (ISHLT) guidelines [1], and there are few reports of HIV seropositive patients who have undergone lung transplantation [2–4].
Here, we describe, to our knowledge, the first HIV seropositive lung transplant recipient in Canada. We also provide a literature review of previously reported cases of solid-organ transplantation in patients with HIV, with a focus on immunosuppression considerations and prognosis.
Case presentation
A 48-year-old man received a bilateral lung transplant for a diagnosis of desquamative interstitial pneumonia (DIP) that was attributed to smoking cigarettes and cannabis, both of which he ceased 20 months prior to transplantation. The diagnosis of DIP was based on compatible imaging and a prior surgical lung biopsy that was performed at the age of 44 years (Figs. 1 and 2).
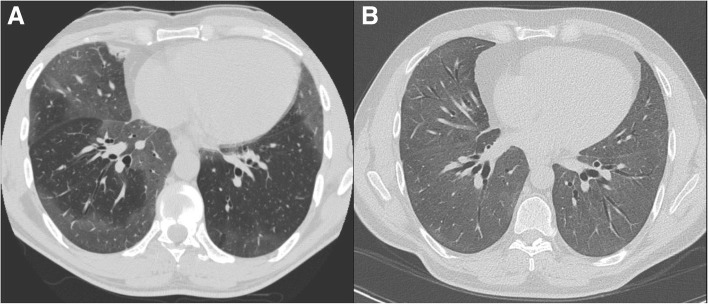
Fig. 1.
High-resolution computed tomography (HRCT) of the chest in axial cut from time of initial presentation (a), and 2 years post-diagnosis (b). Both HRCTs demonstrate multifocal ground glass consolidation, primarily in the lower lung zones, progressive over an 8-year period
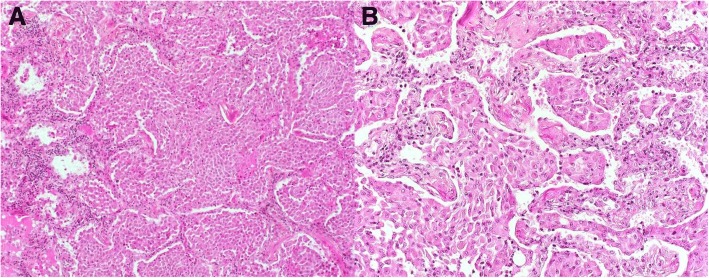
Fig. 2.
Haematoxylin phyloxin saffron stain at 100× (a), and 200× (b) of surgical lung biopsy specimen showing filling of alveolar spaces by macrophages. There is mild interstitial inflammatory infiltrate
The patient was diagnosed with HIV at the age of 38 years and had started HAART 20 months prior to transplantation at age 47. He achieved excellent control of his HIV infection, with a pre-transplant CD4+ cell count of 950 cells/μL, CD4+:CD8+ ratio of 0.69, no detectable viral load, and no history of AIDS-defining illnesses. His HIV was initially managed with cobicistat, elvitegravir, emtricitabine, and tenofovir, which were subsequently changed to abacavir, dolutegravir, and lamivudine when lung transplantation became a consideration due to pharmacokinetic interactions between cobicistat and tacrolimus [5–8].
The patient had progressively worsening dyspnea and lung function that prompted initiation of long-term prednisone and eventually mycophenolate mofetil (MMF), which were both prescribed in consultation with his HIV specialist. His interstitial lung disease continued to progress both clinically and radiographically (Fig. 1), with pre-transplant lung function showing a forced expiratory volume in 1 s (FEV1) 55% predicted, forced vital capacity (FVC) 50% predicted, and diffusing capacity for carbon monoxide (DLCO) 36% predicted. Prior to transplant, he required 2 l oxygen via nasal prongs at rest.
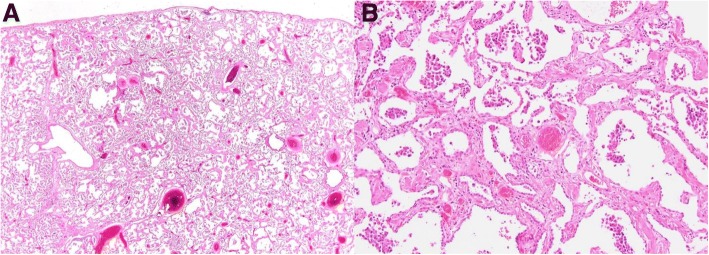
His induction immunosuppression included basiliximab (20 mg on date of transplantation and post-operative day 4), methylprednisolone (500 mg at induction, 500 mg at reperfusion, and three doses of 125 mg on the day of transplant), and MMF (1 g pre-transplant). His explanted lungs still showed a pattern of DIP, but with a decreased number of airspace macrophages and a picture that in areas was more consistent with fibrotic nonspecific interstitial pneumonia (NSIP) (Fig. 3). A similar progression of DIP to an NSIP-like picture has been reported previously [9].
Fig. 3.
10× (a) and 100× (b) views of the explanted lungs. The number of airspace macrophages has considerably decreased compared to the surgical lung biopsy, and the process more closely resembles fibrotic non-specific interstitial pneumonia than desquamative interstitial pneumonia
Post-transplant, his maintenance immunosuppression included MMF 1 g twice daily, prednisone 15 mg daily, and tacrolimus, dosed to a target trough level of 10-12 ng/mL. His previous HAART regimen was also continued. The patient had an uncomplicated course in hospital and was discharged from the intensive care unit on post-operative day 3 and discharged from hospital on day 18. He was maintained on daily valganciclovir 900 mg, trimethoprim-sulfamethoxazole 160-800 mg, and azithromycin 250 mg for prophylaxis against opportunistic infections. Valganciclovir was discontinued 6 months post-transplant as cytomegalovirus DNA remained undetectable in his serum. His tacrolimus trough levels were reduced to 8-10 ng/mL 10 months post-transplant. His prednisone dose was reduced to 10 mg and then subsequently 5 mg daily at 6- and 14-months post-transplant respectively.
He had an episode of Grade A1 minimal acute cellular rejection detected on surveillance transbronchial biopsy 3 months post-transplant. This was not treated, and there was no evidence of graft rejection on repeat biopsies 1 month later. He had an enterovirus/rhinovirus graft infection with significant allograft dysfunction 11 months post-transplant and received 3 daily doses of methylprednisolone 500 mg with a following prednisone dose of 60 mg daily, tapered by 5 mg every 5 days to a baseline dose of 10 mg thereafter. At the time of this infection, he also received a single dose of intravenous immunoglobulin 0.5 g/kg.
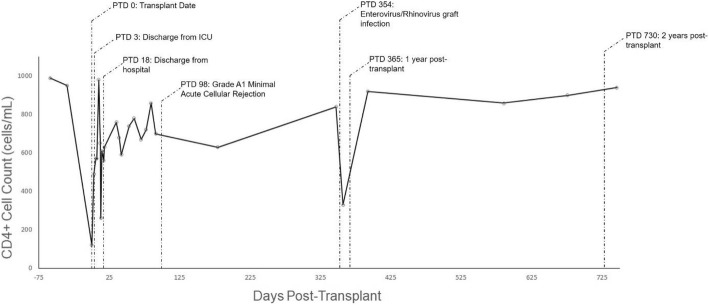
Post-transplant, the patient’s markers of HIV infection continue to be controlled, with CD4+ cell count nadir of 120 cells/μL on post-operative day 3. CD4+ count increased to 630 cells/μL at time of discharge from hospital, and was 940 cells/μL at 24 months post-transplant, with a CD4+:CD8+ ratio of 1.03, and undetectable viral load throughout (Fig. 4). He continues to have normal functional status, no dyspnea, normal lung function, and normal ambulatory oxygenation 24 months post-transplantation.
Fig. 4.
CD4+ cell count in relation to date of bilateral lung transplant. Significant events during the patient’s course are denoted by annotations with post-transplant day (PTD) specified
Discussion and conclusions
There have been eight previously reported cases of lung transplantation in HIV seropositive patients globally [2–4]. Of the 5 patients who received transplantation more than 3 years ago, 4 have achieved 3-year survival, which is a comparable outcome to non-HIV seropositive patients [4, 10]. There are also numerous reports of HIV seropositive patients who have received heart transplants, the majority of which have had good outcomes since the introduction of HAART [11]. Observational data from renal and liver transplant populations have generally suggested that HIV seropositive patients have similar survival compared to seronegative patients if maintained on HAART [12–15]. Conversely, the largest cohort study of HIV seropositive liver and kidney recipients to date reported an increased risk of graft failure in recipients from both subpopulations [16], and a higher risk of mortality in liver recipients. Multiple other studies have also suggested a higher than expected risk of acute rejection in HIV seropositive kidney recipients [13, 15, 17, 18].
Until recently, induction immunosuppression was thought to increase the risks of adverse outcomes in HIV seropositive patients [19]. Modern immunosuppression regimens for induction and acute rejection in solid-organ transplant recipients rely on the inhibition of lymphocyte proliferation and activity, and occasionally the more widespread reduction of lymphocyte populations. This latter approach presents unique challenges in HIV seropositive transplant recipients given CD4+ cell populations and CD4+:CD8+ ratios are important prognostic factors in the management of HIV progression and treatment response [20–22]. This applies to both medications frequently used in the induction and maintenance phases of post-lung transplantation immunosuppression.
Induction therapies for lung transplant typically include anti-thymocyte globulin (ATG) and basiliximab. ATG is a polyclonal antibody that actively depletes T-lymphocytes. ATG reduces CD4+ cell counts immediately after treatment, and also causes prolonged suppression of CD4+:CD8+ ratios [23–25]. When used as induction immunosuppression in lung transplantation, ATG may increase overall survival by reducing graft loss despite also increasing the risk of infection [26]. In the largest cohort of HIV seropositive liver and kidney transplant recipients to date, ATG use in the first week post-kidney transplantation was associated with increased mortality and graft loss [13, 16]. Other studies in HIV seropositive renal transplant recipients have instead suggested that induction immunosuppression with ATG does not increase the risk of infection compared to HIV seronegative controls, and furthermore decreases the risk of graft loss and increases survival [19, 27]. Basiliximab is an IL-2 receptor antagonist that inhibits proliferation and differentiation of T-cells. IL-2 receptor antagonists continue to be the most frequently used induction agents in HIV seronegative lung transplant recipients [28]. Basiliximab use in induction for HIV seropositive renal transplant recipients has been associated with a lower incidence of delayed graft function, reduced graft loss, and a trend towards longer survival compared to no induction [19]. These findings therefore support the use of basiliximab in the induction phase for HIV seropositive lung and renal transplant recipients. Basiliximab may be a preferable induction agent for HIV seropositive patients given ATG’s adverse effects on CD4+ cell populations and its association with increased mortality and graft loss in HIV seropositive renal transplant populations. Our patient tolerated the use of basiliximab as induction therapy well, and had no major adverse events and long-term sequelae attributed to its use.
Maintenance immunosuppressive therapies post-lung transplantation in most centers typically include tacrolimus, MMF, and prednisone, which all exert cytostatic effects on T-cell populations via several mechanisms [29–35]. MMF also exerts apoptotic effects on CD4+ cells in vitro [36]. Despite these findings, neither HIV seropositive or seronegative patients treated with these medications exhibit sustained decreases in their absolute CD4+ cell counts or declines in their CD4+:CD8+ ratios [13, 18, 25, 36–40]. Cobicistat and the protease inhibitor medications increase plasma calcineurin inhibitor levels via cytochrome P450 3A and P-glycoprotein inhibition [5–8], necessitating close monitoring of drug levels when these medications are co-administered with tacrolimus. These interactions were avoided in our patient with alterations to his HAART. Based on this information, HIV seropositive lung transplant recipients can safely be managed with MMF and tacrolimus. Our patient received these medications, as well as prednisone, and had no long-term complications with respect to graft rejection or control of HIV over a 2-year period, which further suggests that HIV seropositive patients can tolerate this immunosuppressant regimen if their HAART is carefully tailored.
In summary, this is the first HIV seropositive lung transplant recipient in Canada to our knowledge, and one of only several patients reported globally. Our immunosuppression selection included basiliximab, corticosteroids, tacrolimus and MMF; a regimen that is supported by evidence drawn from kidney and liver transplant populations. This case report adds to the growing body of literature that these medications are safe in HIV seropositive patients undergoing lung transplantation, and that these patients can achieve long-term outcomes that are comparable to HIV seronegative patients. There is, however, a need for large-scale studies of this specific population given the lack of evidence in optimal immunosuppression for these patients, as well as their long-term prognosis. This is especially the case with future trends towards increasing acceptance of solid-organ transplantation in selected HIV seropositive patients.
Abbreviations
- ATG
Anti-thymocyte globulin
- DIP
Desquamative interstitial pneumonia
- DLCO
Diffusing capacity for carbon monoxide
- FEV1
Forced expiratory volume in 1 s
- FVC
Forced vital capacity
- HAART
Highly active antiretroviral therapy
- HIV
Human immunodeficiency virus
- ISHLT
International Society for Heart and Lung Transplantation
- MMF
Mycophenolate mofetil
- NSIP
Nonspecific interstitial pneumonia
Author’s contributions
The first draft of the manuscript was produced by SO, RDL, and CJR. All authors reviewed, edited, and approved the final version of the submitted manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
This case report details information regarding a human patient, and was reported with the express written consent of this patient.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Shaun Ong, Email: shaunong91@alumni.ubc.ca.
Robert D Levy, Email: robert.levy@vch.ca.
John Yee, Email: john.yee@vch.ca.
Nilu Partovi, Email: nilu.partovi@vch.ca.
Andrew Churg, Email: achurg@mail.ubc.ca.
Philippe Roméo, Email: philippe.romeo.chum@ssss.gouv.qc.ca.
Jean Chalaoui, Email: jchalaoui@hotmail.com.
Roland Nador, Email: roland.nador@ubc.ca.
Alissa Wright, Email: alissa.wright@ubc.ca.
Hélène Manganas, Email: hmanganas@yahoo.ca.
Christopher J Ryerson, Phone: 604-806-8818, Email: chris.ryerson@hli.ubc.ca.
References
- 1.Weill D, Benden C, Corris PA, Dark JH, Davis RD, Keshavjee S, et al. A consensus document for the selection of lung transplant candidates: 2014—an update from the pulmonary transplantation Council of the International Society for heart and lung transplantation. J Heart Lung Transplant. 2015;34(1):1–15. doi: 10.1016/j.healun.2014.06.014. [DOI] [PubMed] [Google Scholar]
- 2.Bertani A, Grossi P, Vitulo P, DAncona G, Arcadipane A, Costa AN, et al. Successful lung transplantation in an HIV- and HBV-positive patient with cystic fibrosis. Am J Transplant. 2009;9(9):2190–2196. doi: 10.1111/j.1600-6143.2009.02779.x. [DOI] [PubMed] [Google Scholar]
- 3.Kern RM, Seethamraju H, Blanc PD, Sinha N, Loebe M, Golden J, et al. The feasibility of lung transplantation in HIV-seropositive patients. Ann Am Thoracic Soc. 2014;11(6):882. doi: 10.1513/AnnalsATS.201402-083OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Koval C, Malinis M, Mueller N, Krisl J, Hannan M, Grossi P, et al. Heart and lung transplantation outcomes in HIV-positive recipients. HOBOKEN: WILEY; 2018. [DOI] [PubMed] [Google Scholar]
- 5.Sheikh AM, Wolf DC, Lebovics E, Goldberg R, Horowitz HW. Concomitant human immunodeficiency virus protease inhibitor therapy markedly reduces tacrolimus metabolism and increases blood levels. Transplantation. 1999;68(2):307–309. doi: 10.1097/00007890-199907270-00027. [DOI] [PubMed] [Google Scholar]
- 6.Barau C, Blouin P, Creput C, Taburet AM, Durrbach A, Furlan V. Effect of coadministered HIV-protease inhibitors on tacrolimus and sirolimus blood concentrations in a kidney transplant recipients. Fundam Clin Pharmacol. 2009;23(4):423–425. doi: 10.1111/j.1472-8206.2009.00706.x. [DOI] [PubMed] [Google Scholar]
- 7.Jain AKB, Venkataramanan R, Shapiro R, Scantlebury VP, Potdar S, Bonham CA, et al. The interaction between antiretroviral agents and tacrolimus in liver and kidney transplant patients. Liver Transpl. 2002;8(9):841–845. doi: 10.1053/jlts.2002.34880. [DOI] [PubMed] [Google Scholar]
- 8.Han Z, Kane BM, Petty LA, Josephson MA, Sutor J, Pursell KJ. Cobicistat significantly increases tacrolimus serum concentrations in a renal transplant recipient with human immunodeficiency virus infection. Pharmacother J Hum Pharmacol Drug Ther. 2016;36(6):e53. doi: 10.1002/phar.1752. [DOI] [PubMed] [Google Scholar]
- 9.Tazelaar HD, Wright JL, Churg A. Desquamative interstitial pneumonia. Histopathology. 2011;58(4):509–516. doi: 10.1111/j.1365-2559.2010.03649.x. [DOI] [PubMed] [Google Scholar]
- 10.Lund LH, Khush KK, Cherikh WS, Goldfarb S, Kucheryavaya AY, Levvey BJ, et al. The registry of the International Society for Heart and Lung Transplantation: thirty-fourth adult heart transplantation report—2017; focus theme: allograft ischemic time. J Heart Lung Transplant. 2017;36(10):1037–1046. doi: 10.1016/j.healun.2017.07.019. [DOI] [PubMed] [Google Scholar]
- 11.Agüero F, Castel MA, Cocchi S, Moreno A, Mestres CA, Cervera C, et al. An update on heart transplantation in human immunodeficiency virus-infected patients: heart transplantation in HIV-infected patients. Am J Transplant. 2016;16(1):21–28. doi: 10.1111/ajt.13496. [DOI] [PubMed] [Google Scholar]
- 12.Qiu JX, Terasaki PI, Waki K, Cai JC, Gjertson DW. HIV-positive renal recipients can achieve survival rates similar to those of HIV-negative patients. Transplantation. 2006;81(12):1658–1661. doi: 10.1097/01.tp.0000226074.97314.e0. [DOI] [PubMed] [Google Scholar]
- 13.Stock PG, Barin B, Murphy B, Hanto D, Diego JM, Light J, et al. Outcomes of kidney transplantation in HIV-infected recipients. N Engl J Med. 2010;363(21):2004–2014. doi: 10.1056/NEJMoa1001197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cooper C, Kanters S, Klein M, Chaudhury P, Marotta P, Wong P, et al. Liver transplant outcomes in HIV-infected patients: a systematic review and meta-analysis with synthetic cohort. AIDS. 2011;25(6):777–786. doi: 10.1097/QAD.0b013e328344febb. [DOI] [PubMed] [Google Scholar]
- 15.Roland ME, Barin B, Carlson L, Frassetto LA, Terrault NA, Hirose R, et al. HIV-infected liver and kidney transplant recipients: 1- and 3-year outcomes. Am J Transplant. 2008;8(2):355–365. doi: 10.1111/j.1600-6143.2007.02061.x. [DOI] [PubMed] [Google Scholar]
- 16.Roland ME, Barin B, Huprikar S, Murphy B, Hanto DW, Blumberg E, et al. Survival in HIV-positive transplant recipients compared with transplant candidates and with HIV-negative controls. AIDS. 2016;30(3):435–444. doi: 10.1097/QAD.0000000000000934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tan HP, Kaczorowski DJ, Basu A, Khan A, McCauley J, Marcos A, et al. Living-related donor renal transplantation in HIV plus recipients using alemtuzumab preconditioning and steroid-free tacrolimus monotherapy: a single center preliminary experience. Transplantation. 2004;78(11):1683–1688. doi: 10.1097/01.TP.0000145880.38548.0D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mazuecos A, Fernandez A, Zarraga S, Andres A, Rodriguez-Benot A, Jimenez C, et al. High incidence of delayed graft function in HIV-infected kidney transplant recipients. Transplant Int. 2013;26(9):893–902. doi: 10.1111/tri.12147. [DOI] [PubMed] [Google Scholar]
- 19.Kucirka LM, Durand CM, Bae S, Avery RK, Locke JE, Orandi BJ, et al. Induction immunosuppression and clinical outcomes in kidney transplant recipients infected with human immunodeficiency virus. Am J Transplant. 2016;16(8):2368–2376. doi: 10.1111/ajt.13840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Choi S, et al. CD4 lymphocytes are an incomplete surrogate marker for clinical progression in persons with asymptomatic HIV infection taking zidovudine. Ann Intern Med. 1993;118(9):674–680. doi: 10.7326/0003-4819-118-9-199305010-00003. [DOI] [PubMed] [Google Scholar]
- 21.Serrano-Villar S, Moreno S, Fuentes-Ferrer M, Sánchez-Marcos C, Ávila M, Sainz T, et al. The CD4:CD8 ratio is associated with markers of age-associated disease in virally suppressed HIV-infected patients with immunological recovery. HIV Medicine. 2014;15(1):40–49. doi: 10.1111/hiv.12081. [DOI] [PubMed] [Google Scholar]
- 22.Serrano-Villar S, Gutiérrez C, Vallejo A, Hernández-Novoa B, Díaz L, Abad Fernández M, et al. The CD4/CD8 ratio in HIV-infected subjects is independently associated with T-cell activation despite long-term viral suppression. J Infect 2013. 2012;66(1):57–66. doi: 10.1016/j.jinf.2012.09.013. [DOI] [PubMed] [Google Scholar]
- 23.Müller TF, Grebe SO, Neumann MC, Heymanns J, Radsak K, Sprenger H, et al. Persistent long-term changes in lymphocyte subsets induced by polyclonal antibodies. Transplantation. 1997;64(10):1432. doi: 10.1097/00007890-199711270-00010. [DOI] [PubMed] [Google Scholar]
- 24.Carter JT, Melcher ML, Carlson LL, Roland ME, Stock PG. Thymoglobulin-associated Cd4 T-cell depletion and infection risk in HIV-infected renal transplant recipients. Am J Transplant. 2006;6(4):753–760. doi: 10.1111/j.1600-6143.2006.01238.x. [DOI] [PubMed] [Google Scholar]
- 25.Stock PG, Roland ME, Carlson L, Freise CE, Roberts JP, Hirose R, et al. Kidney and liver transplantation in human immunodeficiency virus-infected patients: a pilot safety and efficacy study. Transplantation. 2003;76(2):370–375. doi: 10.1097/01.TP.0000075973.73064.A6. [DOI] [PubMed] [Google Scholar]
- 26.Hachem RR, Edwards LB, Yusen RD, Chakinala MM, Alexander Patterson G, Trulock EP. The impact of induction on survival after lung transplantation: an analysis of the International Society for Heart and Lung Transplantation registry. Clin Transpl. 2008;22(5):603–608. doi: 10.1111/j.1399-0012.2008.00831.x. [DOI] [PubMed] [Google Scholar]
- 27.Locke JE, James NT, Mannon RB, Mehta SG, Pappas PG, Baddley JW, et al. Immunosuppression regimen and the risk of acute rejection in HIV-infected kidney transplant recipients. Transplant J. 2014;97(4):446–450. doi: 10.1097/01.TP.0000436905.54640.8c. [DOI] [PubMed] [Google Scholar]
- 28.Van Raemdonck D, Hartwig MG, Hertz MI, Davis RD, Cypel M, Hayes D, et al. Report of the ISHLT working group on primary lung graft dysfunction part IV: prevention and treatment: a 2016 consensus group statement of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant. 2017;36(10):1121–1136. doi: 10.1016/j.healun.2017.07.013. [DOI] [PubMed] [Google Scholar]
- 29.Allison AC, Eugui EM. Mycophenolate mofetil and its mechanisms of action. Immunopharmacology. 2000;47(2–3):85–118. doi: 10.1016/S0162-3109(00)00188-0. [DOI] [PubMed] [Google Scholar]
- 30.Allison AC, Eugui EM. Immunosuppressive and other effects of mycophenolic-acid and an ester prodrug, mycophenolate mofetil. Immunol Rev. 1993;136(1):5–28. doi: 10.1111/j.1600-065X.1993.tb00652.x. [DOI] [PubMed] [Google Scholar]
- 31.Eugui EM, Allison AC. Immunosuppressive activity of mycophenolate mofetil. Ann N Y Acad Sci. 1993;685(1 Immunomodulat):309–329. doi: 10.1111/j.1749-6632.1993.tb35881.x. [DOI] [PubMed] [Google Scholar]
- 32.Ho S, Clipstone N, Timmermann L, Northrop J, Graef I, Fiorentino D, et al. The mechanism of action of Cyclosporin a and FK506. Clin Immunol Immunopathol. 1996;80(3):S45. doi: 10.1006/clin.1996.0140. [DOI] [PubMed] [Google Scholar]
- 33.Kiani A, Rao A, Aramburu J. Manipulating immune responses with immunosuppressive agents that target NFAT. Immunity. 2000;12(4):359–372. doi: 10.1016/S1074-7613(00)80188-0. [DOI] [PubMed] [Google Scholar]
- 34.Granellipiperno A, Nolan P, Inaba K, Steinman RM. The effect of immunosuppressive agents on the induction of nuclear factors that bind to sites on the interleukin-2 promoter. J Exp Med. 1990;172(6):1869–1872. doi: 10.1084/jem.172.6.1869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tocci MJ, Matkovich DA, Collier KA, Kwok P, Dumont F, Lin S, et al. The immunosuppressant FK506 selectively inhibits expression of early T cell activation genes. J Immunol. 1989;143(2):718–726. [PubMed] [Google Scholar]
- 36.Pantaleo G, Paolo Rizzardi G, Chapuis AG, Acha-Orbea H, Attinger A, Fleury S, et al. Effects of mycophenolic acid on human immunodeficiency virus infection in vitro and in vivo. Nat Med. 2000;6(7):762–768. doi: 10.1038/77489. [DOI] [PubMed] [Google Scholar]
- 37.Press N, Kimel G, Harris M, Yip B, Craib KJP, Montaner JSG. Case series assessing the safety of mycophenolate as part of multidrug rescue treatment regimens. HIV clinical trials. 2002;3(1):17–20. doi: 10.1310/B6T0-N98J-1J3M-EQPK. [DOI] [PubMed] [Google Scholar]
- 38.Gómez V, Fernández A, Galeano C, Oliva J, Diez V, Bueno C, et al. Renal transplantation in HIV-infected patients: experience at a tertiary Hospital in Spain and Review of the literature. Transplant Proc. 2013;45(3):1255–1259. doi: 10.1016/j.transproceed.2013.02.032. [DOI] [PubMed] [Google Scholar]
- 39.Bravo Soto JA, de la Rosa RE, del Castillo JL, Morales SC, Olivares EG, Ortega AO, et al. Effect of mycophenolate mofetil regimen on peripheral blood lymphocyte subsets in kidney transplant recipients. Transplant Proc. 2003;35(4):1355–1359. doi: 10.1016/S0041-1345(03)00364-6. [DOI] [PubMed] [Google Scholar]
- 40.Sankatsing SUC, Jurriaans S, van Swieten P, van Leth F, Cornelissen M, Miedema F, et al. Highly active antiretroviral therapy with or without mycophenolate mofetil in treatment-naive HIV-1 patients. AIDS. 2004;18(14):1925–1931. doi: 10.1097/00002030-200409240-00008. [DOI] [PubMed] [Google Scholar]