Abstract
Background
The association of sodium intake with the risk of cardiovascular morbidity and mortality is inconsistent. Thus, the present meta-analysis was conducted to summarize the strength of association between sodium intake and cardiovascular morbidity and mortality.
Methods
PubMed, Embase, and the Cochrane Library were searched systematically to identify the relevant studies up to October 2017. The effect estimates for 100 mmol/day increase in sodium intake were calculated using 95% confidence intervals (CIs) of cardiac death, total mortality, stroke, or stroke mortality for low (< 3 g/d), moderate (3–5 g/d), or heavy (> 5 g/d) sodium intake, and minimal sodium intake comparison.
Results
A total of 16 prospective cohort studies reported data on 205,575 individuals. The results suggested that an increase in sodium intake by 100 mmol/d demonstrated little or no effect on the risk of cardiac death (P = 0.718) and total mortality (P = 0.720). However, the risk of stroke incidence (P = 0.029) and stroke mortality (P = 0.007) was increased significantly by 100 mmol/day increment of sodium intake. Furthermore, low sodium intake was associated with an increased risk of cardiac death (P = 0.003), while moderate (P < 0.001) or heavy (P = 0.001) sodium intake was associated with an increased risk of stroke mortality.
Conclusions
These findings suggested that sodium intake by 100 mmol/d increment was associated with an increased risk of stroke incidence and stroke mortality. Furthermore, low sodium intake was related to an increased cardiac death risk, while moderate or heavy sodium intake was related to an increased risk of stroke mortality.
Keywords: Sodium intake, Cardiovascular outcomes, Dose-response, Meta-analysis, Prospective cohort studies
Background
Cardiovascular diseases (CVD) are the major causes of mortality and morbidity in the general population, accounting for approximately 17.5 million deaths worldwide. The World Health Organization (WHO) estimated over 30% of all the deaths worldwide annually due to CVDs [1]. Several studies have recommended several lifestyle factors such as intake of yogurt [2], dietary magnesium [3], nuts [4], whole grains [5], dietary fibers [6], milk [7], and saturated and trans unsaturated fatty acids [8] that prevent the progression of CVD. However, the relatively high residual risk for CVD should be addressed, and it is necessary to understand the association of individual dietary components with CVD at the population level to alter the dietary habits and improve the health conditions.
Dietary sodium intake has been documented as a modifiable risk factor for blood pressure, which in turn, is associated with the progression of CVD [9–12]. Currently, WHO recommends a sodium intake of < 2 g/d, which is largely based on the small and short-term clinical trials that evaluated the effect of modest salt reduction on blood pressure in general population [13]. However, the effect of dietary sodium intake on subsequent cardiovascular morbidity and mortality is limited and inconclusive.
Several prospective studies have indicated that long-term interventions aiming at sodium reduction may reduce the risk of CVD [14, 15]. Moreover, the results of another prospective study did not show any correlation between sodium intake and CVD [16]. Furthermore, several studies suggested that high sodium intake may decrease the risk of cardiac death [17, 18]. Hence, clarifying the optimal daily intake of sodium is essential in the general population as it has not yet been determined. Herein, we attempted to investigate the available prospective cohort studies on a large-scale to determine the association of sodium intake and cardiovascular morbidity and mortality.
Methods
Data sources, search strategy, and selection criteria
This study was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines [19]. Studies with a prospective cohort design evaluating the impact of sodium intake and the risk of major cardiovascular outcomes, without any language bias (English or another language), were included in this meta-analysis. Electronic databases, such as PubMed, Embase, and Cochrane Library were searched for literature published up to October 2017. The core search terms used were “dietary salt” OR “sodium” AND (“cardiovascular disease” OR “stroke” OR “cardiac death” OR “mortality” OR “death” OR “CVD” OR “myocardial infarction” OR “coronary events”) AND “clinical trials” AND “human”. The reference lists from potentially relevant studies were searched to select the additional eligible studies. Parameters such as the study topic, design, participants’ status, exposure, and reported outcomes were employed to identify the relevant studies.
The literature search and study selection was conducted by two authors independently, and any inconsistencies were settled by group discussion until a consensus was reached. The inclusion criteria for the studies were as follows: (1) prospective cohort design; (2) evaluation of the impact of sodium intake and the risk of major cardiovascular outcomes; (3) reported at least 1 of the following outcomes: cardiac death, total mortality, stroke, or stroke mortality; (4) the data should provide the effect estimates, such as relative risk (RR), hazard ratio (HR), or odds ratio (OR,) and 95% confidence intervals (CIs) or crude data that compared the different categories of sodium intake vs. the minimal sodium intake with respect to 100 mmol/day increments and the risk of major cardiovascular outcomes. All the retrospective observational studies were excluded as various confounding factors could bias the results.
Data collection and quality assessment
The data collected from the eligible studies included the first author’s name, publication year, country, sample size, age at baseline, percentage male, assessment of exposure, reference category of sodium intake, reported outcomes, follow-up duration, and covariates in the fully adjusted model. Also, the crude data on the number of cases/persons or person-years, effect of different exposure categories, and the 95% CIs were collected. In addition, the effect estimates that were maximally adjusted for potential confounders, if the study provided several adjusted effect estimates, were selected.
The comprehensive Newcastle–Ottawa Scale (NOS) has been partially validated for evaluating the quality of the observational studies in the meta-analysis, and hence, was used to evaluate the quality of the study method [20]. The NOS evaluated the quality of the observational studies based on selection (4 items), comparability (1 item), and outcome (3 items). The maximum score was 9, and the minimum score was 0 (Additional file : Table S1). The data were extracted and quality assessed by 2 authors independently, and any inconsistencies were referred to the original studies by an additional author.
Statistical analysis
To evaluate the impact of sodium intake and the risk of major cardiovascular outcomes, we collected the effect estimates (RR, HR, or OR) and the 95% CIs or the relevant crude data from each study. The summary RRs and 95% CIs for the low (< 3 g/d), moderate (3–5 g/d), or heavy (> 5 g/d) sodium intake vs. and the minimized intake of sodium and the risk of major cardiovascular outcomes were calculated using the random-effects model [21, 22]. Next, we evaluated the estimates of the RR associated with every 100 mmol/day increase in sodium by the generalized least-squares method for trend estimation [23], assuming the presence of a linear relationship between the natural logarithm of the RR and increasing sodium intake. The mid-point for closed categories and median for open categories putatively determined each sodium intake category, presuming a normal distribution for sodium intake. The summary RRs for 100 mmol/day increase in sodium intake was calculated using random-effects meta-analysis [22, 24].
The heterogeneity among the included studies was assessed using the I2 and Q statistic, and a P-value < 0.10 was considered as significant heterogeneity [25, 26]. Subgroup analyses were conducted for cardiac death, total mortality, and stroke according to publication year, sample size, percentage male, assessment of exposure, following-up duration, and with or without adjusted body mass index (BMI), smoking, alcohol, previous CVD, diabetes mellitus (DM), physical activity (PA), and level of potassium. The P-value between subgroups was evaluated by chi-square test and meta-regression [27]. A sensitivity analysis was evaluated the impact of individual studies by removing individual study from the meta-analysis [28]. Funnel plot and Egger [29] and Begg [30] tests investigated the outcomes that were also used to evaluate any potential publication bias. All the reported P-values are 2-sided, and P < 0.05 was considered statistically significant. The statistical analyses were conducted using STATA software (version 10.0; Stata Corporation, College Station, TX, USA).
Results
Literature search
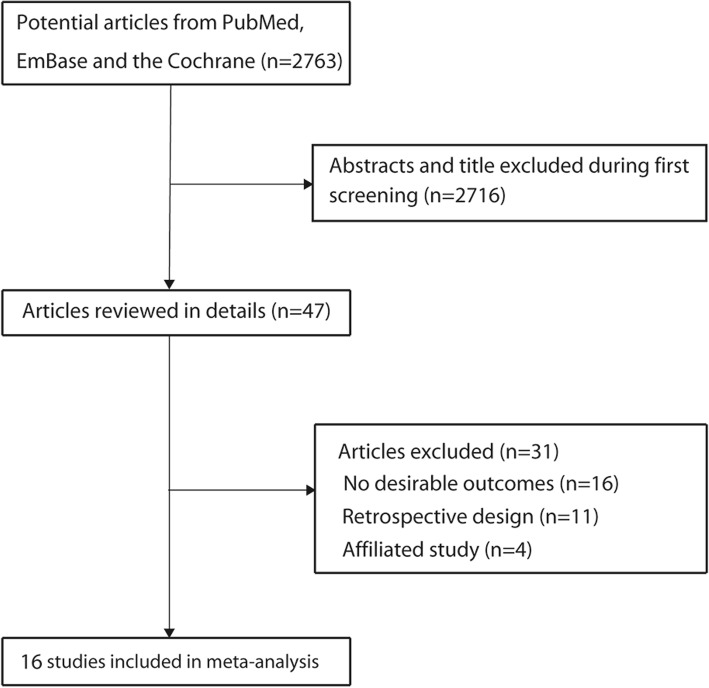
The results of the study selection process were presented in Fig. 1. A total of 2763 articles were identified in the initial electronic search. Of these, 2716 were excluded as they were duplicates and irrelevant studies. Thus, a total of 47 potentially eligible studies were selected. After a detailed evaluation, 16 prospective cohort studies were selected for the final meta-analysis [14, 16, 18, 31–43]. A manual search of the reference lists of these studies did not yield any new eligible studies. The general characteristics of the included studies were presented in Table 1.
Fig. 1.
Schematic representation of the study selection process
Table 1.
Baseline characteristic of studies included in this meta-analysis
| Study | Publication year | Country | Sample size | Age at baseline | Percentage male (%) | Assessment of exposure | Reference category of sodium intake | Reported outcomes | Follow-up (year) | Adjusted factors | NOS score |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Alderman [31] | 1995 | US | 2937 | 53.0 | 64.7 | 24 h urine collection | Quartile I | Stroke | 3.5 | Age, and race | 7 |
| Tunstall-Pedoe [32] | 1997 | Scotland | 11,629 | 40.0–59.0 | 49.5 | 24 h urine collection | 129.6 mmol/day | Cardiac death, total mortality | 7.6 | Age | 8 |
| He [33] | 1999 | US | 9485 | 25.0–74.0 | 38.9 | 24 h urine collection | Quartile I | Cardiac death, stroke, stroke mortality, total mortality | 19.0 | Age, sex, race, SBP, SC, BMI, DM, diuretic use, PA, education, alcohol, smoking, EI | 9 |
| Tuomilehto [34] | 2001 | Finland | 2436 | 25.0–64.0 | 48.2 | 24 h urine collection | < 159 mmol/day | Cardiac death, stroke, total mortality | 8.0 | Age, study year, smoking, HDL, SBP, and BMI | 9 |
| Nagata [35] | 2004 | Japan | 29,079 | > 35.0 | 49.6 | FFQ | Stroke mortality | 7.0 | Age, EI, marital status, education, BMI, smoking, alcohol, PA, hypertension, DM, and intake of protein, potassium, and vitamin E | 9 | |
| Cohen [36] | 2008 | US | 8699 | > 30.0 | 44.9 | FFQ | Cardiac death, total mortality | 8.7 | Age, sex, race, education, added table salt, PA, alcohol, smoking, DM, history of cancer, SBP, TC, dietary potassium, weight, antihypertensive drug | 9 | |
| Larsson [37] | 2008 | Finland | 26,556 | 50.0–69.0 | 100 | FFQ | Stroke | 13.6 | Age, supplementation group, smoking, BMI, SBP and DBP, SC, serum HDL, DM and CVD, PA, alcohol and EI | 8 | |
| Umesawa [14] | 2008 | Japan | 58,730 | 40.0–79.0 | 39.4 | FFQ | Cardiac death, stroke mortality | 12.7 | BMI, smoking, alcohol, history of hypertension, DM, menopause, HRT, PA, educational, perceived mental stress, calcium, and potassium intake | 9 | |
| Ekinci [38] | 2011 | Australia | 638 | 64.0 | 56.0 | 24 h urine collection | Cardiac death, total mortality | 9.9 | Age, sex, previous CVD, eGFR, atrial fibrillation, SBP, DM duration | 6 | |
| Stolarz-Skrzypek [18] | 2011 | Belgium | 3595 | 40.9 | 47.3 | 24 h urine collection | Cardiac death, stroke, total mortality | 7.9 | Study population, sex, age, BMI, SBP, potassium excretion, antihypertensive drug, smoking, alcohol, DM, TC, and educational | 9 | |
| Yang [16] | 2011 | US | 12,267 | > 20.0 | 48.1 | FFQ | Cardiac death, total mortality | 14.8 | Sex, race/ethnicity, educational, BMI, smoking, alcohol, TC, HDL, PA, family history of CVD, and EI | 9 | |
| O’Donnell [39] | 2011 | Canada | 28,880 | > 55.0 | 70.6 | 24 h urine collection | Cardiac death, stroke, total mortality | 4.8 | Age, sex, race/ethnicity, history of stroke or MI, creatinine, BMI, comorbid vascular risk factors, treatment allocation, fruit and vegetable, PA, SBP, and urinary potassium | 7 | |
| Gardener [40] | 2012 | US | 2657 | 69.0 | 36.0 | FFQ | Stroke | 10.0 | Age, sex, race/ethnicity, education, alcohol, smoking, PA, EI, total fat, saturated fat, carbohydrates, protein, DM, hypercholesterolemia, hypertension, previous CVD, BMI | 9 | |
| Mills [41] | 2016 | US | 3757 | 57.8 | 55.6 | FFQ | Stroke | 6.8 | Age, sex, race, clinic site, education; waist circumference, BMI, smoking; alcohol, PA, LDL, glucose; history of CVD; use of antidiabetic, lipid-lowering, and BP- lowering medications, urinary creatinine excretion, baseline estimated GFR | 9 | |
| Kalogeropoulos [42] | 2015 | US | 2642 | 74.6 | 48.8 | FFQ | Total mortality | 10.0 | Age, sex, race, BMI, smoking, PA, previous CVD, pulmonary disease, DM, depression, BP, heart rate, electrocardiogram abnormalities, and serum glucose, albumin, creatinine, and SC | 9 | |
| Horikawa [43] | 2014 | Japan | 1588 | 58.7 | 52.5 | FFQ | Total mortality | 7.0 | Age, sex, BMI, HbA1c, DM duration, LDL, HDL, log-transformed triglycerides, insulin, lipid-lowering agents, smoking, alcohol, EI, and PA | 7 |
*BMI body mass index, BP blood pressure, CHD coronary heart disease, CVD cardiovascular disease, DBP diastolic blood pressure, DM diabetes mellitus, EI energy intake, FFQ food frequency questionnaire, eGFR estimated glomerular filtration rate, GFR glomerular filtration rate, HbA1c glycated hemoglobin, HDL high density lipoprotein, HRT hormone replacement therapy, LDL low density lipoprotein, MACEs major cardiovascular events, MI myocardial infarction, PA physical activity, SBP systolic blood pressure, SC serum cholesterol, TC total cholesterol
Study characteristics
A total of 16 prospective cohort studies with 205,575 individuals were eligible for this study. The follow-up period of the participants ranged from 3.5–19.0 years and 638–58,730 individuals were included in each study. A total of 7 studies were conducted in the USA [16, 31, 33, 36, 40–42], 4 in Europe [18, 32, 34, 37], 3 in Japan [14, 35, 43], 1 in Australia [38], and 1 in Canada [39]. Seven studies used 24-h urine collection [18, 31–34, 38, 39], and the remaining 9 studies used food frequency questionnaires (FFQ) to assess the dietary sodium exposure [14, 16, 35–37, 40–43]. The study quality was assessed using the NOS (Table 1). A score of ≥7 was considered as high quality for the study. Overall, 10 studies had a score of 9 [14, 16, 18, 33–36, 40–42], 2 had a score of 8 [32, 37], 3 had a score of 7 [32, 39, 43], and the remaining 1 study had a score of 6 [38].
Cardiac death
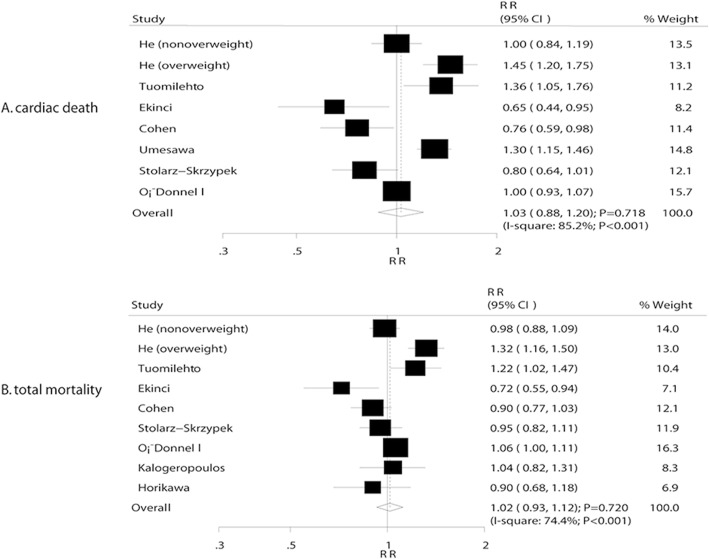
A total of 7 studies reported an association between sodium intake and cardiac death. The summary RR showed that a 100 mmol increment per day in sodium intake was not associated with cardiac death (RR, 1.03; 95% CI, 0.88–1.20; P = 0.718; Fig. 2a); however, accumulating evidence suggested significant heterogeneity (I2 = 85.2%, P < 0.001). Sensitivity analysis indicated that the conclusion was unaffected after sequential exclusion of each study from the pooled analysis. Furthermore, the low sodium intake was found to be associated with an increased risk of cardiac death (RR: 1.19; 95% CI: 1.06–1.33; P = 0.003), while moderate (RR: 0.91; 95% CI: 0.71–1.15; P = 0.421) and heavy (RR: 1.02; 95%CI: 0.92–1.13; P = 0.762) sodium intake did not demonstrate a significant effect (Table 2). Subgroup analysis indicated that an increment of 100 mmol/day in sodium intake exerted detrimental effects on cardiac death if the duration of follow-up was ≥10 years (RR: 1.24; 95% CI: 1.02–1.50; P = 0.034; Table 3). Conversely, increased sodium intake was associated with the reduced risk of cardiac death if the study was not adjusted for BMI (RR: 0.72; 95% CI: 0.59–0.90; P = 0.003; Table 3).
Fig. 2.
a Association between sodium intake and cardiac death. b Association between sodium intake and total mortality
Table 2.
Summary results for different categories of sodium and subsequent major cardiovascular outcomes
| Outcomes | Low sodium | P value | Moderate sodium | P value | Heavy sodium | P value |
|---|---|---|---|---|---|---|
| Cardiac death | 1.19 (1.06–1.33) | 0.003 | 0.91 (0.71–1.15) | 0.421 | 1.02 (0.92–1.13) | 0.762 |
| Total mortality | 1.02 (0.89–1.18) | 0.779 | 0.98 (0.85–1.14) | 0.806 | 1.09 (0.94–1.27) | 0.257 |
| Stroke | 1.25 (0.85–1.85) | 0.260 | 1.11 (1.00–1.24) | 0.058 | 1.02 (0.93–1.11) | 0.720 |
| Stroke mortality | 1.20 (0.96–1.50) | 0.117 | 1.50 (1.20–1.88) | < 0.001 | 1.81 (1.29–2.55) | 0.001 |
Table 3.
Subgroup analyses for cardiac death
| Factor | Subgroup | RR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity | P value between subgroups |
|---|---|---|---|---|---|---|
| Publication year | Before 2010 | 1.15 (0.94–1.41) | 0.170 | 82.7 | < 0.001 | < 0.001 |
| 2010 or after | 0.85 (0.67–1.08) | 0.174 | 73.8 | 0.022 | ||
| Sample size | ≥ 10,000 | 1.13 (0.88–1.47) | 0.334 | 92.8 | < 0.001 | 0.513 |
| < 10,000 | 0.98 (0.76–1.25) | 0.846 | 84.8 | < 0.001 | ||
| Percentage male (%) | ≥ 60.0 | 1.00 (0.93–1.07) | 1.000 | – | – | 0.023 |
| < 60.0 | 1.03 (0.83–1.26) | 0.812 | 85.7 | < 0.001 | ||
| Assessment of exposure | FFQ | 1.01 (0.59–1.70) | 0.982 | 92.9 | < 0.001 | 0.025 |
| 24 h urine collection | 1.03 (0.86–1.23) | 0.746 | 82.2 | < 0.001 | ||
| Follow-up duration (years) | ≥ 10.0 | 1.24 (1.02–1.50) | 0.034 | 78.0 | 0.011 | < 0.001 |
| < 10.0 | 0.91 (0.74–1.11) | 0.329 | 77.4 | 0.001 | ||
| Adjusted BMI | Yes | 1.13 (0.96–1.32) | 0.146 | 85.2 | < 0.001 | < 0.001 |
| No | 0.72 (0.59–0.90) | 0.003 | 0.0 | 0.506 | ||
| Adjusted smoking | Yes | 1.09 (0.88–1.33) | 0.433 | 85.3 | < 0.001 | 0.003 |
| No | 0.84 (0.56–1.27) | 0.415 | 78.5 | 0.031 | ||
| Adjusted alcohol | Yes | 1.04 (0.83–1.31) | 0.735 | 87.5 | < 0.001 | 0.028 |
| No | 0.99 (0.74–1.34) | 0.959 | 80.4 | 0.006 | ||
| Adjusted Previous CVD | Yes | 0.84 (0.56–1.27) | 0.415 | 78.5 | 0.031 | 0.003 |
| No | 1.09 (0.88–1.33) | 0.433 | 85.3 | < 0.001 | ||
| Adjusted DM | Yes | 0.98 (0.78–1.23) | 0.855 | 87.4 | < 0.001 | 0.125 |
| No | 1.14 (0.84–1.53) | 0.400 | 80.3 | 0.024 | ||
| Adjusted PA | Yes | 1.09 (0.91–1.30) | 0.355 | 87.2 | < 0.001 | 0.113 |
| No | 0.90 (0.59–1.38) | 0.636 | 85.0 | 0.001 | ||
| Adjusted potassium | Yes | 0.97 (0.78–1.19) | 0.744 | 88.2 | < 0.001 | 0.070 |
| No | 1.10 (0.82–1.46) | 0.533 | 83.6 | < 0.001 |
*BMI body mass index, CI confidence interval, CVD cardiovascular disease, DM diabetes mellitus, FFQ food frequency questionnaire, PA physical activity, RR relative risk
Total mortality
A total of 8 studies reported a correlation between sodium intake and total mortality. However, the results did not reveal any significant association of 100 mmol increments per day in sodium intake with the total mortality risk (RR: 1.02; 95% CI: 0.93–1.12; P = 0.720; Fig. 2b). Although substantial heterogeneity was observed in the magnitude of the effect across the studies (I2 = 74.4%, P < 0.001), after sequential exclusion of each study from pooled analyses, the conclusion was not affected by the exclusion of any specific study. Furthermore, the low (RR: 1.02; 95% CI: 0.89–1.18; P = 0.779), moderate (RR: 0.98; 95% CI: 0.85–1.14; P = 0.806), and heavy (RR: 1.09; 95% CI: 0.94–1.27; P = 0.257) sodium intake was not associated with the risk of total mortality (Table 2). Subgroup analysis indicated that an increment of 100 mmol/day in the sodium intake was associated with an increased risk of total mortality if the sample size was ≥10,000 (RR: 1.06; 95% CI: 1.01–1.12; P = 0.029; Table 4), and the percentage male was ≥60.0%.
Table 4.
Subgroup analyses for total mortality
| Factor | Subgroup | RR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity | P value between subgroups |
|---|---|---|---|---|---|---|
| Publication year | Before 2010 | 1.09 (0.91–1.30) | 0.351 | 85.2 | < 0.001 | 0.328 |
| 2010 or after | 0.96 (0.85–1.08) | 0.493 | 60.2 | 0.040 | ||
| Sample size | ≥ 10,000 | 1.06 (1.01–1.12) | 0.029 | – | – | 0.428 |
| < 10,000 | 1.00 (0.89–1.14) | 0.958 | 77.1 | < 0.001 | ||
| Percentage male (%) | ≥ 60.0 | 1.06 (1.01–1.12) | 0.029 | – | – | 0.428 |
| < 60.0 | 1.00 (0.89–1.14) | 0.958 | 77.1 | < 0.001 | ||
| Assessment of exposure | FFQ | 0.93 (0.83–1.04) | 0.212 | 0.0 | 0.570 | 0.032 |
| 24 h urine collection | 1.05 (0.93–1.17) | 0.441 | 80.4 | < 0.001 | ||
| Follow-up duration (years) | ≥ 10.0 | 1.11 (0.90–1.37) | 0.343 | 83.9 | 0.002 | 0.139 |
| < 10.0 | 0.97 (0.87–1.09) | 0.616 | 69.6 | 0.005 | ||
| Adjusted BMI | Yes | 1.07 (0.98–1.17) | 0.146 | 68.2 | 0.004 | 0.001 |
| No | 0.83 (0.67–1.02) | 0.082 | 51.4 | 0.151 | ||
| Adjusted smoking | Yes | 1.04 (0.92–1.17) | 0.522 | 74.5 | 0.001 | 0.991 |
| No | 0.89 (0.61–1.30) | 0.560 | 87.0 | 0.005 | ||
| Adjusted alcohol | Yes | 1.01 (0.87–1.17) | 0.908 | 80.4 | < 0.001 | 0.471 |
| No | 1.02 (0.87–1.19) | 0.804 | 70.8 | 0.016 | ||
| Adjusted Previous CVD | Yes | 0.95 (0.77–1.17) | 0.649 | 74.1 | 0.021 | 0.999 |
| No | 1.04 (0.91–1.19) | 0.573 | 78.8 | < 0.001 | ||
| Adjusted DM | Yes | 0.98 (0.85–1.12) | 0.716 | 77.7 | < 0.001 | 0.132 |
| No | 1.11 (0.97–1.25) | 0.119 | 52.4 | 0.147 | ||
| Adjusted PA | Yes | 1.04 (0.94–1.15) | 0.475 | 74.2 | 0.002 | 0.291 |
| No | 0.96 (0.74–1.24) | 0.735 | 81.4 | 0.005 | ||
| Adjusted potassium | Yes | 0.99 (0.88–1.10) | 0.790 | 64.0 | 0.062 | 0.341 |
| No | 1.03 (0.88–1.21) | 0.712 | 79.8 | < 0.001 |
*BMI body mass index, CI confidence interval, CVD cardiovascular disease, DM diabetes mellitus, FFQ food frequency questionnaire, PA physical activity, RR relative risk
Stroke and stroke mortality
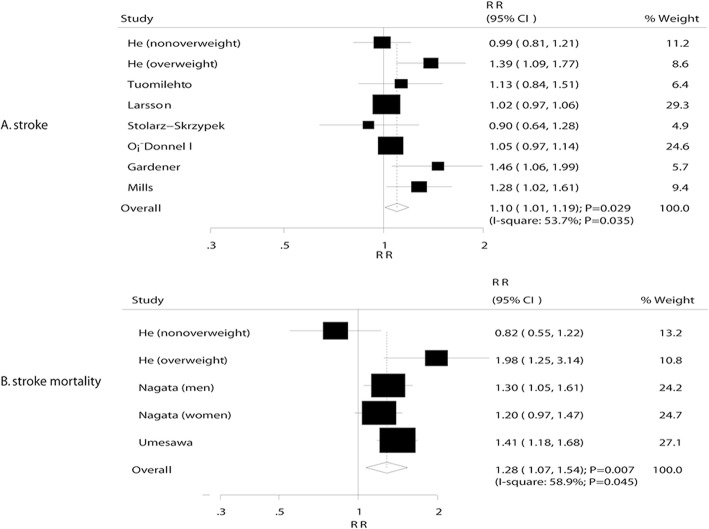
A total of 7 studies reported an association between sodium intake and stroke, and 3 studies reported the association of sodium intake and stroke mortality. Pooled analysis of stroke and stroke mortality indicated that a 100 mmol increment per day in sodium intake exerted a harmful effect (stroke: RR, 1.10; 95% CI, 1.01–1.19; P = 0.029, Fig. 3a; stroke mortality: RR, 1.28; 95% CI, 1.07–1.54; P = 0.007, Fig. 3b). Heterogeneity was observed in the magnitude of the effect across the studies (I2 = 53.7%, P = 0.035 for stroke; I2 = 58.9%, P = 0.045 for stroke mortality). However, the conclusion was not affected by excluding any specific study after sequential exclusion of each study from all the pooled analyses. Furthermore, low (RR: 1.25; 95% CI: 0.85–1.85; P = 0.260), moderate (RR: 1.11; 95% CI: 1.00–1.24; P = 0.058), and heavy (RR: 1.02; 95% CI: 0.93–1.11; P = 0.720) sodium intake did demonstrate any effect on the subsequent stroke risk (Table 2). In addition, low sodium intake did not affect the stroke mortality (RR: 1.20; 95% CI: 0.96–1.50; P = 0.117), while moderate (RR: 1.50; 95% CI: 1.20–1.88; P < 0.001) and heavy (RR: 1.81; 95% CI: 1.29–2.55; P = 0.001) sodium intake was associated with a high risk of stroke mortality (Table 2). In addition, subgroup analysis suggested that a 100 mmol per day increment in sodium intake was associated with an increased risk of stroke if the sample size was < 10,000 (RR: 1.18; 95% CI: 1.02–1.36; P = 0.029), the proportion of males was < 60.0% (RR: 1.18; 95% CI: 1.02–1.36; P = 0.029), the study adjusted for BMI (RR: 1.10; 95% CI: 1.01–1.19; P = 0.029), smoking status (RR: 1.13; 95% CI: 1.00–1.28; P = 0.048), and PA(RR: 1.11; 95% CI: 1.01–1.22; P = 0.026), and the study not adjusted for the level of potassium (RR: 1.16; 95% CI: 1.02–1.33; P = 0.029) (Table 5). The subgroup analysis for stroke mortality was not conducted due to the small number of studies included in this investigation on the association of sodium intake and stroke mortality.
Fig. 3.
a Association between sodium intake and stroke. b Association between sodium intake and stroke mortality
Table 5.
Subgroup analyses for stroke
| Factor | Subgroup | RR and 95% CI | P value | Heterogeneity (%) | P value for heterogeneity | P value between subgroups |
|---|---|---|---|---|---|---|
| Publication year | Before 2010 | 1.09 (0.95–1.24) | 0.211 | 54.5 | 0.086 | 0.248 |
| 2010 or after | 1.14 (0.96–1.35) | 0.127 | 58.2 | 0.067 | ||
| Sample size | ≥ 10,000 | 1.03 (0.99–1.07) | 0.181 | 0.0 | 0.537 | 0.020 |
| < 10,000 | 1.18 (1.02–1.36) | 0.029 | 46.5 | 0.096 | ||
| Percentage male (%) | ≥ 60.0 | 1.03 (0.99–1.07) | 0.181 | 0.0 | 0.537 | 0.020 |
| < 60.0 | 1.18 (1.02–1.36) | 0.029 | 46.5 | 0.096 | ||
| Assessment of exposure | FFQ | 1.19 (0.95–1.50) | 0.125 | 76.0 | 0.016 | 0.503 |
| 24 h urine collection | 1.08 (0.96–1.21) | 0.190 | 36.8 | 0.176 | ||
| Follow-up duration (years) | ≥ 10.0 | 1.15 (0.97–1.37) | 0.116 | 72.4 | 0.012 | 0.453 |
| < 10.0 | 1.08 (0.98–1.20) | 0.136 | 18.2 | 0.300 | ||
| Adjusted BMI | Yes | 1.10 (1.01–1.19) | 0.029 | 53.7 | 0.035 | – |
| No | – | – | – | – | ||
| Adjusted smoking | Yes | 1.13 (1.00–1.28) | 0.048 | 60.2 | 0.020 | 0.865 |
| No | 1.05 (0.97–1.14) | 0.236 | – | – | ||
| Adjusted alcohol | Yes | 1.14 (0.99–1.31) | 0.072 | 66.2 | 0.011 | 0.744 |
| No | 1.06 (0.98–1.14) | 0.174 | 0.0 | 0.636 | ||
| Adjusted Previous CVD | Yes | 1.09 (0.99–1.20) | 0.074 | 64.4 | 0.038 | 0.395 |
| No | 1.10 (0.91–1.32) | 0.317 | 49.7 | 0.114 | ||
| Adjusted DM | Yes | 1.14 (0.99–1.31) | 0.072 | 66.2 | 0.011 | 0.744 |
| No | 1.06 (0.98–1.14) | 0.174 | 0.0 | 0.636 | ||
| Adjusted PA | Yes | 1.11 (1.01–1.22) | 0.026 | 64.6 | 0.015 | 0.892 |
| No | 1.03 (0.82–1.29) | 0.811 | 0.0 | 0.326 | ||
| Adjusted potassium | Yes | 1.04 (0.96–1.13) | 0.309 | 0.0 | 0.396 | 0.963 |
| No | 1.16 (1.02–1.33) | 0.029 | 65.2 | 0.013 |
*BMI body mass index, CI confidence interval, CVD cardiovascular disease, DM diabetes mellitus, FFQ food frequency questionnaire, PA physical activity, RR relative risk
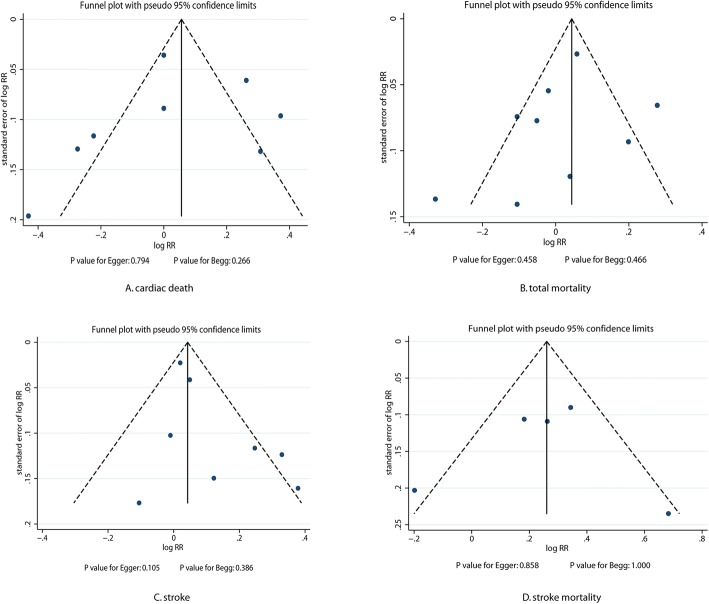
Publication bias
The review of the funnel plots did not exclude the potential for publication bias for cardiac death, total mortality, stroke, and stroke mortality (Fig. 4). The Egger and Begg test’s results did not show any evidence of publication bias for cardiac death (P-value for Egger: 0.794; P-value for Begg: 0.266), total mortality (P-value for Egger: 458; P-value for Begg: 0.466), stroke (P-value for Egger: 0.105; P-value for Begg: 0.386), and stroke mortality (P-value for Egger: 0.858; P-value for Begg: 1.000).
Fig. 4.
Publication bias tests for cardiac death, total mortality, stroke, and stroke mortality. Each point represents an independent study of the indicated association
Discussion
The current study included the prospective cohort studies and explored the possible correlations between sodium intake and the outcomes of cardiac death, total mortality, stroke, and stroke mortality. This quantitative meta-analysis included a total of 205,575 individuals from 16 prospective cohort studies with a broad range of populations. The meta-analysis findings suggested that an increment of 100 mmol/day in sodium intake did not affect the incidence of cardiac death and total mortality. However, a 100 mmol per day increment in sodium intake significantly increased the risk of stroke and stroke mortality. Furthermore, parameters such as sample size, the proportion of males, assessment of exposure, follow-up duration, and several other adjusted factors were found to be associated with the correlation between sodium intake and major cardiovascular outcomes.
A previous meta-analysis suggested that high sodium intake was associated with a significantly increased risk of stroke and total cardiovascular diseases [44]. However, other 2 meta-analysis studies based on randomized controlled trials suggested that the reduced dietary salt did not affect the cardiovascular morbidity or mortality [45, 46]. The inherent limitation of this study included shorter duration of follow-up period than that required to show a clinical benefit, especially when the rate of events was lower than expected, which without any statistically significant difference. Furthermore, reduced dietary sodium intake seems to be associated with the degree of control achieved. Finally, the range of sodium intake and the cut-off values for the three categories differed among various studies. Therefore, we conducted a dose-response meta-analysis of these prospective studies for evaluating the optimal dose of sodium intake.
The current findings were in agreement with a recently published large cohort study conducted in Manhattan [40]. Our meta-analysis study included 2657 individuals and found that the participants who consumed > 4000 mg/d sodium demonstrated a 159% increased risk of stroke. Also, the risk percentage of stroke was increased by 17% for each increase in 500 mg/d. He et al. suggested that high sodium intake was strongly and independently associated with an increased risk of stroke mortality in overweight individuals, thereby significantly increasing the risk of total mortality [33]. Also, the current study indicated that increased sodium intake significantly elevated the risk of stroke and stroke mortality, while no effect on cardiac death and total mortality was demonstrated, which might be attributed to the increased blood pressure and hypertension due to high sodium levels by stiffening the endothelial cells, thickening and narrowing of resistance arteries, and blocking of nitric oxide synthesis [47].
The current study did not demonstrate a significant difference between 100 mmol increments of sodium intake per day and the risk of cardiac death. However, inconsistent results were reported by individual studies. O’Donnell et al. indicated that sodium excretion > 7 g/d was associated with an increased risk of cardiac death and coronary heart disease (CHD) as compared to sodium excretion of 4–5.99 g/d [39]. Furthermore, TunstallPedoe et al. suggested that high sodium intake significantly increased the cardiac death and CHD by 36% and 34%, respectively [32]. This phenomenon might be attributed to the inclusion of other prospective studies encompassing general individuals; however, these 2 studies specifically included individuals with high risk of cardiovascular disease, rendering them susceptible to extreme sodium intake.
Subgroup analysis suggested that a 100 mmol increment of sodium intake per day was associated with cardiac death reduction if the study was not adjusted for BMI; also, the risk of cardiac death was increased significantly if the follow-up duration was ≥10 years. In addition, the risk of total mortality was increased if the sample size was ≥10,000 and percentage male was ≥60.0%. Finally, and increased sodium intake by 100 mmol per day was associated with an elevated risk of stroke if the sample size was < 10,000, the percentage of males was < 60.0%, the study adjusted for BMI, smoking status, PA, and the study not adjusted for potassium level. However, these conclusions might be unreliable due to the inclusion of small cohorts in each subset. Therefore, this study provided a relative result as well as a synthetic and comprehensive review.
The three strengths of our study should be highlighted. Firstly, only prospective cohort studies were included, which eliminated the selection as well as recall bias and could be a concern for retrospective case-control studies. Secondly, a large sample size allowed us to quantitatively assess the association of sodium intake with the risk of cardiovascular morbidity and mortality, which in turn, demonstrated that our findings are potentially more robust than any individual studies. Thirdly, the pooled analysis included a wide range of sodium intake levels, which subsequently allowed an accurate assessment of the relation of sodium intake and major cardiovascular risk outcomes.
Nevertheless, the present study had some limitations as follows: (1) the adjusted models used in the included studies are different, and these factors might play a critical role in the development of CVDs; (2) the minimal intake of sodium in individual study varied, which might introduce uncontrolled biases and potential heterogeneity; (3) heterogeneity across included studies was high, and hence, the results of publication bias test were not reliable; (4) high heterogeneity was not investigated by subgroup analysis due to the minimal intake of sodium and cutoff value, and the adjusted factors were not consistent among included studies; (5) the meta-analysis used pooled data due to the unavailability of individual data, which restricted a detailed and relevant analysis in order to obtain comprehensive results.
Conclusions
In conclusion, the results of this study suggested that increased sodium intake might play a major role in the risk of stroke morbidity and mortality. However, the increased sodium intake did not have a significant effect on cardiac death and total mortality. Nevertheless, future studies focusing on specific populations for analyzing the secondary prevention of major cardiovascular outcomes are warranted.
Acknowledgments
Funding
This study was supported by the Beijing Natural Science Foundation (No. 7184204, No. 7182042), Beijing Municipal Health Bureau High-Level Talent Cultivation (Nos. 2014–3-043, 2015–3-048, and 2015–3-051), and Beijing Municipal Administration of Hospital Incubating Program (PX20166046).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BMI
Body mass index
- CHD
Coronary heart disease
- CIs
Confidence intervals
- CVD
Cardiovascular diseases
- HR
Hazard ratio
- NOS
Newcastle–Ottawa Scale
- OR
Odds ratio
- RR
Relative risk
Authors’ contributions
YBZ1 (YZ) and JZ conducted the studies, participated in data collection, and drafted the manuscript. ZQL and YL performed the statistical analysis and participated in the experimental design. XF, YPZ, and YBZ2 (YZ) helped in drafting the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yaobin Zhu, Email: zhuyaobin@126.com.
Jing Zhang, Email: 2802581220@qq.com.
Zhiqiang Li, Phone: +86-18600372914, Email: cardiacsurgeon@yeah.net.
Yang Liu, Email: liuyang2010strive@163.com.
Xing Fan, Email: fanxingshasha_163@163.com.
Yaping Zhang, Email: ping.zhang@hotmail.com.
Yanbo Zhang, Phone: +86-18600372914, Email: yanbozhang@126.com.
References
- 1.Collaborators GMaCoD Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the global burden of disease study 2013. Lancet. 2015;385:117–171. doi: 10.1016/S0140-6736(14)61682-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu L, Sun D. Consumption of yogurt and the incident risk of cardiovascular disease: a meta-analysis of nine cohort studies. Nutrients. 2017;9:3. doi: 10.3390/nu9030315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fang X, Wang K, Han D, He X, Wei J, Zhao L, et al. Dietary magnesium intake and the risk of cardiovascular disease, type 2 diabetes, and all-cause mortality: a dose-response meta-analysis of prospective cohort studies. BMC Med. 2016;14:210. doi: 10.1186/s12916-016-0742-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aune D, Keum N, Giovannucci E, Fadnes LT, Boffetta P, Greenwood DC, et al. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: a systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016;14:207. doi: 10.1186/s12916-016-0730-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li B, Zhang G, Tan M, Zhao L, Jin L, Tang X, et al. Consumption of whole grains in relation to mortality from all causes, cardiovascular disease, and diabetes: dose-response meta-analysis of prospective cohort studies. Medicine (Baltimore) 2016;95:e4229. doi: 10.1097/MD.0000000000004229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim Y, Je Y. Dietary fibre intake and mortality from cardiovascular disease and all cancers: a meta-analysis of prospective cohort studies. Arch Cardiovasc Dis. 2016;109:39–54. doi: 10.1016/j.acvd.2015.09.005. [DOI] [PubMed] [Google Scholar]
- 7.Larsson SC, Crippa A, Orsini N, Wolk A, Michaelsson K. Milk consumption and mortality from all causes, cardiovascular disease, and Cancer: a systematic review and meta-analysis. Nutrients. 2015;7:7749–7763. doi: 10.3390/nu7095363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.de Souza RJ, Mente A, Maroleanu A, Cozma AI, Ha V, Kishibe T, et al. Intake of saturated and trans unsaturated fatty acids and risk of all cause mortality, cardiovascular disease, and type 2 diabetes: systematic review and meta-analysis of observational studies. BMJ. 2015;351:h3978. doi: 10.1136/bmj.h3978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Group ICR Intersalt: an international study of electrolyte excretion and blood pressure. Results for 24 hour urinary sodium and potassium excretion. Intersalt cooperative research group. BMJ. 1988;297:319–328. doi: 10.1136/bmj.297.6644.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Iso H, Stampfer MJ, Manson JE, Rexrode K, Hennekens CH, Colditz GA, et al. Prospective study of calcium, potassium, and magnesium intake and risk of stroke in women. Stroke. 1999;30:1772–1779. doi: 10.1161/01.STR.30.9.1772. [DOI] [PubMed] [Google Scholar]
- 11.Whelton PK, Appel LJ, Espeland MA, Applegate WB, Ettinger WH, Jr, Kostis JB, et al. Sodium reduction and weight loss in the treatment of hypertension in older persons: a randomized controlled trial of nonpharmacologic interventions in the elderly (TONE). TONE collaborative research group. JAMA. 1998;279:839–846. doi: 10.1001/jama.279.11.839. [DOI] [PubMed] [Google Scholar]
- 12.Group ToHPCR Effects of weight loss and sodium reduction intervention on blood pressure and hypertension incidence in overweight people with high-normal blood pressure. The trials of hypertension prevention, phase II. The trials of hypertension prevention collaborative research group. Arch Intern Med. 1997;157:657–667. doi: 10.1001/archinte.1997.00440270105009. [DOI] [PubMed] [Google Scholar]
- 13.He FJ, MacGregor GA. Effect of modest salt reduction on blood pressure: a meta-analysis of randomized trials. Implications for public health. J Hum Hypertens. 2002;16:761–770. doi: 10.1038/sj.jhh.1001459. [DOI] [PubMed] [Google Scholar]
- 14.Umesawa M, Iso H, Date C, Yamamoto A, Toyoshima H, Watanabe Y, et al. Relations between dietary sodium and potassium intakes and mortality from cardiovascular disease: the Japan collaborative cohort study for evaluation of Cancer risks. Am J Clin Nutr. 2008;88:195–202. doi: 10.1093/ajcn/88.1.195. [DOI] [PubMed] [Google Scholar]
- 15.Cook NR, Cutler JA, Obarzanek E, Buring JE, Rexrode KM, Kumanyika SK, et al. Long term effects of dietary sodium reduction on cardiovascular disease outcomes: observational follow-up of the trials of hypertension prevention (TOHP) BMJ. 2007;334:885–888. doi: 10.1136/bmj.39147.604896.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang Q, Liu T, Kuklina EV, Flanders WD, Hong Y, Gillespie C, et al. Sodium and potassium intake and mortality among US adults: prospective data from the third National Health and nutrition examination survey. Arch Intern Med. 2011;171:1183–1191. doi: 10.1001/archinternmed.2011.257. [DOI] [PubMed] [Google Scholar]
- 17.Cohen HW, Hailpern SM, Fang J, Alderman MH. Sodium intake and mortality in the NHANES II follow-up study. Am J Med. 2006;119:275.e7–275.14. doi: 10.1016/j.amjmed.2005.10.042. [DOI] [PubMed] [Google Scholar]
- 18.Stolarz-Skrzypek K, Kuznetsova T, Thijs L, Tikhonoff V, Seidlerova J, Richart T, et al. Fatal and nonfatal outcomes, incidence of hypertension, and blood pressure changes in relation to urinary sodium excretion. JAMA. 2011;305:1777–1785. doi: 10.1001/jama.2011.574. [DOI] [PubMed] [Google Scholar]
- 19.Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA group Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Plos Med. 2009;6:e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wells GA, Shea B, O’Connell D. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa (ON): Ottawa hospital research institute; 2009. [Google Scholar]
- 21.DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–188. doi: 10.1016/0197-2456(86)90046-2. [DOI] [PubMed] [Google Scholar]
- 22.Ades AE, Lu G, Higgins JP. The interpretation of random-effects meta-analysis in decision models. Med Decis Mak. 2005;25:646–654. doi: 10.1177/0272989X05282643. [DOI] [PubMed] [Google Scholar]
- 23.Orsini N, Bellocco R. Generalized least squares for trend estimation of summarized dose-response data. Stata J. 2006;6:40–57. [Google Scholar]
- 24.Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301–1309. doi: 10.1093/oxfordjournals.aje.a116237. [DOI] [PubMed] [Google Scholar]
- 25.Deeks JJ, Higgins JPT, Altman DG. Analyzing data and undertaking meta-analyses. In: Higgins J, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions 5.0.1., edn. Oxford: The Cochrane Collaboration; 2008. [Google Scholar]
- 26.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Deeks JJ, Altman DG, Bradburn MJ. Statistical methods for examining heterogeneity and combining results from several studies in meta-analysis. In: Egger M, Davey Smith G, Altman DG, editors. Systematic reviews in health care: Metaanalysis in context. 2. London: BMJ Books; 2001. pp. 285–312. [Google Scholar]
- 28.Tobias A. Assessing the influence of a single study in meta-analysis. Stata Tech Bull. 1999;47:15–17. [Google Scholar]
- 29.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–1101. doi: 10.2307/2533446. [DOI] [PubMed] [Google Scholar]
- 31.Alderman MH, Madhavan S, Cohen H, Sealey JE, Laragh JH. Low urinary sodium is associated with greater risk of myocardial infarction among treated hypertensive men. Hypertension. 1995;25:1144–1152. doi: 10.1161/01.HYP.25.6.1144. [DOI] [PubMed] [Google Scholar]
- 32.Tunstall-Pedoe H, Woodward M, Tavendale R, A’Brook R, McCluskey MK. Comparison of the prediction by 27 different factors of coronary heart disease and death in men and women of the Scottish heart health study: cohort study. BMJ. 1997;315:722–729. doi: 10.1136/bmj.315.7110.722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.He J, Ogden LG, Vupputuri S, Bazzano LA, Loria C, Whelton PK. Dietary sodium intake and subsequent risk of cardiovascular disease in overweight adults. JAMA. 1999;282:2027–2034. doi: 10.1001/jama.282.21.2027. [DOI] [PubMed] [Google Scholar]
- 34.Tuomilehto J, Jousilahti P, Rastenyte D, Moltchanov V, Tanskanen A, Pietinen P, et al. Urinary sodium excretion and cardiovascular mortality in Finland: a prospective study. Lancet. 2001;357:848–851. doi: 10.1016/S0140-6736(00)04199-4. [DOI] [PubMed] [Google Scholar]
- 35.Nagata C, Takatsuka N, Shimizu N, Shimizu H. Sodium intake and risk of death from stroke in Japanese men and women. Stroke. 2004;35:1543–1547. doi: 10.1161/01.STR.0000130425.50441.b0. [DOI] [PubMed] [Google Scholar]
- 36.Cohen HW, Hailpern SM, Alderman MH. Sodium intake and mortality follow-up in the third National Health and nutrition examination survey (NHANES III) J Gen Intern Med. 2008;23:1297–1302. doi: 10.1007/s11606-008-0645-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Larsson SC, Virtanen MJ, Mars M, Mannisto S, Pietinen P, Albanes D, et al. Magnesium, calcium, potassium, and sodium intakes and risk of stroke in male smokers. Arch Intern Med. 2008;168:459–465. doi: 10.1001/archinte.168.5.459. [DOI] [PubMed] [Google Scholar]
- 38.Ekinci EI, Clarke S, Thomas MC, Moran JL, Cheong K, MacIsaac RJ, et al. Dietary salt intake and mortality in patients with type 2 diabetes. Diabetes Care. 2011;34:703–709. doi: 10.2337/dc10-1723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.O’Donnell MJ, Yusuf S, Mente A, Gao P, Mann JF, Teo K, et al. Urinary sodium and potassium excretion and risk of cardiovascular events. JAMA. 2011;306:2229–2238. doi: 10.1001/jama.2011.1729. [DOI] [PubMed] [Google Scholar]
- 40.Gardener H, Rundek T, Wright CB, Elkind MS, Sacco RL. Dietary sodium and risk of stroke in the northern Manhattan study. Stroke. 2012;43:1200–1205. doi: 10.1161/STROKEAHA.111.641043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mills KT, Chen J, Yang W, Appel LJ, Kusek JW, Alper A, et al. Sodium excretion and the risk of cardiovascular disease in patients with chronic kidney disease. JAMA. 2016;315:2200–2210. doi: 10.1001/jama.2016.4447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kalogeropoulos AP, Georgiopoulou VV, Murphy RA, Newman AB, Bauer DC, Harris TB, et al. Dietary sodium content, mortality, and risk for cardiovascular events in older adults: the health, aging, and body composition (health ABC) study. JAMA Intern Med. 2015;175:410–419. doi: 10.1001/jamainternmed.2014.6278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Horikawa C, Yoshimura Y, Kamada C, Tanaka S, Tanaka S, Hanyu O, et al. Dietary sodium intake and incidence of diabetes complications in Japanese patients with type 2 diabetes: analysis of the Japan diabetes complications study (JDCS) J Clin Endocrinol Metab. 2014;99:3635–3643. doi: 10.1210/jc.2013-4315. [DOI] [PubMed] [Google Scholar]
- 44.Strazzullo P, D’ Elia L, Kandala N. Salt intake, stroke, and cardiovascular disease: metaanalysis of prospective studies. BMJ. 2009;339:b4567. doi: 10.1136/bmj.b4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Taylor RS, Ashton KE, Moxham T, Hooper L, Ebrahim S. Reduced dietary salt for the prevention of cardiovascular disease: a meta-analysis of randomized controlled trials (Cochrane review) Am J Hypertens. 2011;24:843–853. doi: 10.1038/ajh.2011.115. [DOI] [PubMed] [Google Scholar]
- 46.Taylor RS, Ashton KE, Moxham T, Hooper L, Ebrahim S. Reduced dietary salt for the prevention of cardiovascular disease. Cochrane Database Syst Rev. 2011;12:Cd009217. doi: 10.1002/14651858.CD009217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bussemaker E, Hillebrand U, Hausberg M, Pavenstadt H, Oberleithner H. Pathogenesis of hypertension: interactions among sodium, potassium, and aldosterone. Am J Kidney Dis. 2010;55:1111–1120. doi: 10.1053/j.ajkd.2009.12.022. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.