Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis is characterised by pauci-immune necrotising vasculitis of the small blood vessels [1]. The association between ANCA-associated vasculitis (AAV) and usual interstitial pneumonia (UIP) pattern of interstitial lung disease (ILD) is poorly understood.
Short abstract
Initial evaluation for ANCA should be performed at diagnosis in all patients with pulmonary fibrosis; ANCA positivity will increase risk of development of vasculitis and promote further evaluation and specific monitoring http://ow.ly/hf4730kVZF8
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis is characterised by pauci-immune necrotising vasculitis of the small blood vessels [1]. The association between ANCA-associated vasculitis (AAV) and usual interstitial pneumonia (UIP) pattern of interstitial lung disease (ILD) is poorly understood.
The occurrence of AAV and ILD in the same patient, though rare, is being increasingly recognised. Patients with an initial diagnosis of pulmonary fibrosis occasionally acquire seropositivity for myeloperoxidase (MPO)-ANCA, which may develop into microscopic polyangitis (MPA) [2]. This association between ILD and ANCA/AAV has received increasing attention both clinically and pathophysiologically. Herein, the association between ILD with a UIP pattern and ANCA/AAV is reviewed.
Case report
A 66-year-old Indian female presented with complaints of cough, dypnoea on exertion (grade 2 Medical Research Council ) and weakness. There was no past medical or relevant family history. She was a nonsmoker, housewife with no history of any exposure and was not taking any medication. There was no history of joint pains, rashes or any other history of symptoms suggestive of connective tissue disease or vasculitis.
Clinical examination was normal (body mass index 25 kg·m-2) other than hypoxia (arterial oxygen saturation measured by pulse oximetry 94% on room air) and fine bibasal inspiratory crackles on auscultation of the chest.
Blood work-up revealed a raised erythrocyte sedimentation rate (ESR) of 43 (normal 0–30) mm·h−1 and a C-reactive protein of 2.95 (normal <0.748) mg·dL−1. Angiotensin converting enzyme was raised at 86.3 (normal 20–70) units·L−1. Anti-nuclear antibody (ELISA) was raised at 2.21. Rheumatoid factor was raised at 30 (normal <14) units·mL−1 and MPO antibodies were elevated at 99.4 (normal <10). The rest of her serology and laboratory work-up was normal as detailed in table 1.
Table 1.
Initial laboratory data from 2013
| Variable | Patient's result | Normal value |
| WBC count cells·mm−3 | 11 100 | 4000–11 000 |
| Neutrophil count % of WBC | 73 | 40–75 |
| Lymphocyte count % of WBC | 20 | 20–40 |
| Monocyte count % of WBC | 4 | 2–10 |
| Eosinophil count % of WBC | 3 | 1–5 |
| Basophil count % of WBC | 0 | 0–1 |
| Haemoglobin g·dL−1 | 13 | 12–14 |
| Haematocrit % | 38.3 | 36–42 |
| Platelet count cells·mm−3 | 210 000 | 140 000–440 000 |
| International normalised ratio | 1.1 | <1.0 |
| Urea nitrogen mg·dL−1 | 17 | 6–30 |
| Creatinine mg·dL−1 | 0.80 | 0.5–1.0 |
| Creatine kinase units·L−1 | 83 | 30–135 |
| Alanine transaminase units·L−1 | 26 | 9–52 |
| Aspartate transaminase units·L−1 | 28 | 5–40 |
| Urine analysis | Normal | |
| ESR mm·h−1 | 43 | 0–30 |
| C-reactive protein mg·dL−1 | 2.95 | <0.748 |
| Anti-nuclear antibody | 2.21 | <1:40 |
| Rheumatoid factor units·mL−1 | 30 | <14 |
| Anti-cyclic citrullinated protein antibody units·mL−1 | 0.50 | <5 |
| Angiotensin converting enzyme units·L−1 | 86.5 | 20–70 |
| Anti-MPO antibody | 99.4 | <10 |
| Anti–SSa antibody | 7.2 | <20 |
| Anti-Scl-70 antibody | 2.79 | <20 |
| Anti-dsDNA units·mL−1 | 29.59 | <30 |
| Anti-proteinase 3 antibody | 4.04 | <10 |
| Anti–Jo-1 antibody | 0.48 | <3 |
| Anti-Smith antibody | 6.83 | <20 |
| C3 complement mg·dL−1 | 156.38 | 90–180 |
| C4 complement mg·dL−1 | 38.25 | 10–40 |
WBC: white blood cell.
Pulmonary function tests were performed and revealed a restrictive pattern with forced expiratory volume in 1 s of 1.27 L (67% predicted) and a reduced diffusion capacity of the lung for carbon monoxide of 47%. Transthoracic echocardiogram demonstrated normal cardiac structure, function and ejection fraction except for a pulmonary artery pressure of 40 mmHg.
Bronchoscopy with bronchoalveolar lavage and transbronchial lung biopsy (TBLB) was performed. All bronchoalveolar lavage stains and cultures were negative. Histopathology on TBLB reported a mild lympho-histiocytic infiltrate with possible incipient non-necrotic micro-granuloma in the bronchial wall. There was no evidence of vasculitis, eosinophilic infiltrate or granuloma in the alveolar tissue. Ziehl–Neelsen and Periodic Acid–Schiff stains were negative. Open lung biopsy could not be performed due to lack of consent from the patient.
Task 1
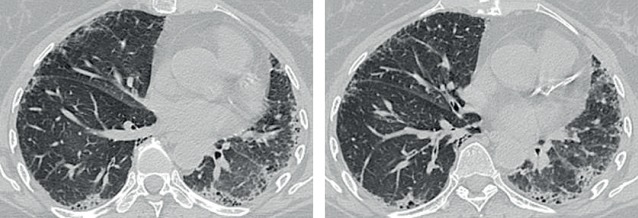
Interpret the high-resolution computed tomography (HRCT) chest findings (figure 1).
Figure 1.
Chest HRCT scan from 2013.
Answer 1
The HRCT scan showed widespread interstitial fibrotic changes with mild subpleural honeycombing consistent with UIP. The distribution of UIP on HRCT is characteristically basal and peripheral, though often patchy.
Task 2
What is your initial diagnosis based on history, radiological evidence and laboratory reports at this point of the case summary?
UIP-ILD
Sarcoidosis
AAV
Answer 2
Initial diagnosis of UIP-ILD and possible sarcoidosis
A differential diagnosis of UIP-ILD and possible sarcoidosis was entertained at this stage with raised anti-nuclear antibody and MPO antibodies (p-ANCA). The patient was started on prednisolone (1 mg·kg−1) and remained clinically stable with subjective improvement of symptoms. Steroids were continued for 1 year and then stopped.
After 3 months of starting steroids the patient was started on pirfenidone 1200 mg daily (could not tolerate >1200 mg daily) and N-acetyl cysteine (600 mg twice a day) which were continued for 2 years. During this time she remained clinically stable with no acute exacerbations or hospitalisation. She was regularly monitored with annual HRCT and 6-monthly pulmonary function testing (table 2).
Table 2.
Sequential lung function test: 2013–2017
| Parameter | Predicted value | Absolute value (% predicted value) | ||||
| 12/07/2013 | 23/05/2014 | 23/03/2015 | 01/04/2016 | 24/05/2017 | ||
| FVC L | 2.3 | 1.58 (69) | 2.06 (90) | 1.78 (78) | 2.02 (88) | 1.7 (73) |
| TLC L | 4.5 | 2.62 (58) | 3.06 (68) | 3.2 (71) | 3.94 (86) | |
| KCO DLCO·L−1 | 4.47 | 2.09 (47) | 2.12 (47) | 2.52 (57) | 3.23 (74) | 1.61 (42) |
| TLCO L | 20.15 | 3.85 (19) | 10.95 (55) | 7.47 (37) | 7.68 (38) | 6.1 (32) |
FVC: forced vital capacity; TLC: total lung capacity; KCO: transfer coefficient of the lung for carbon monoxide; DLCO: diffusing capacity of the lung for carbon monoxide; TLCO: transfer factor of the lung for carbon monoxide.
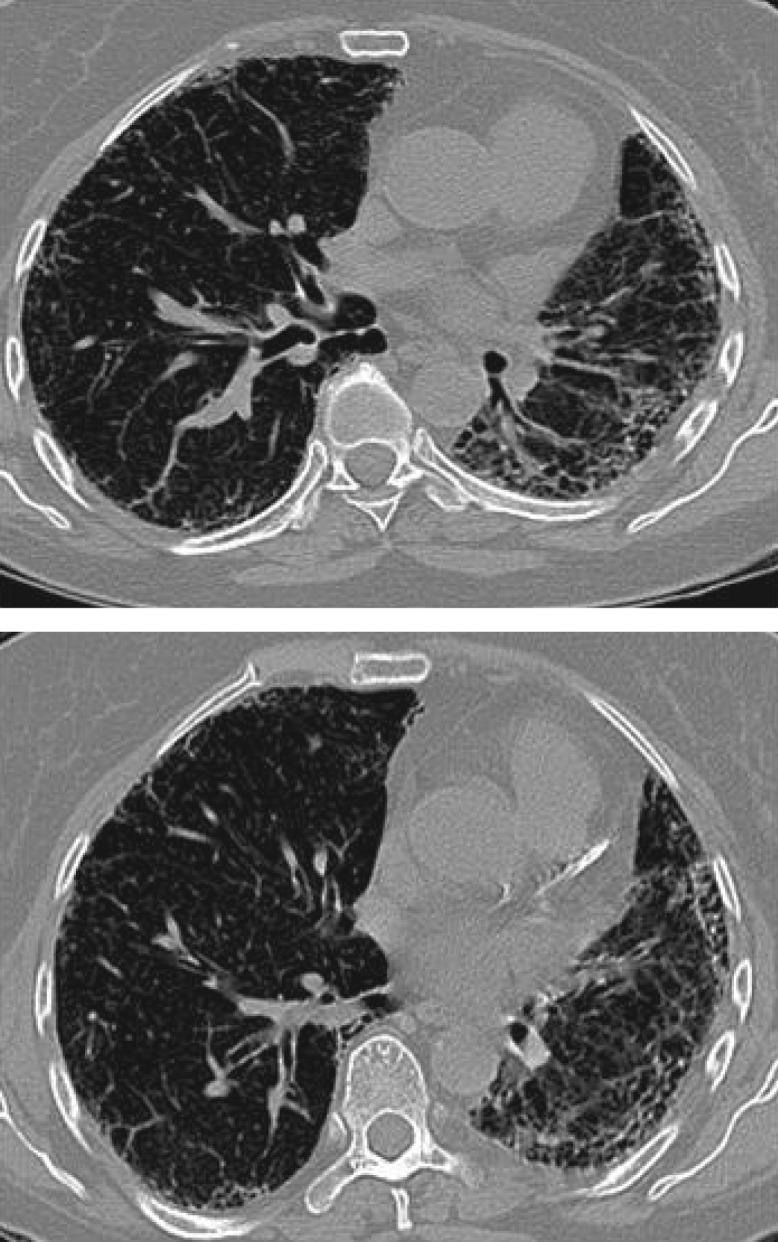
A HRCT scan performed in 2017 showed widespread interstitial fibrotic changes with an increase in subpleural honeycombing when compared to the HRCT from 2013, which is consistent with a UIP pattern (figure 2).
Figure 2.
Chest HRCT scan from 2017 showing progression of disease with increase in subpleural honeycombing.
In February 2017 (3.5 years after initial presentation), the patient developed diplopia due to third nerve palsy which resolved spontaneously in May 2017. The patient also developed abnormal lower limb sensation with tingling, pins and needles and cold intolerance with diminished peripheral pulses on examination.
Task 3
What is your final diagnosis based on history, radiological evidence and laboratory reports at this point of the case summary?
UIP
Sarcoidosis
Rheumatoid arthritis
AAV with ILD
Answer 3
AAV
ANCA has been associated with different lung diseases, mostly vasculitis with pulmonary involvement: Wegener's granulomatosis, Churg–Strauss syndrome and MPA. The coexistence of pulmonary fibrosis and ANCA-positive vasculitis, essentially MPA, has been consistently reported in recent years.
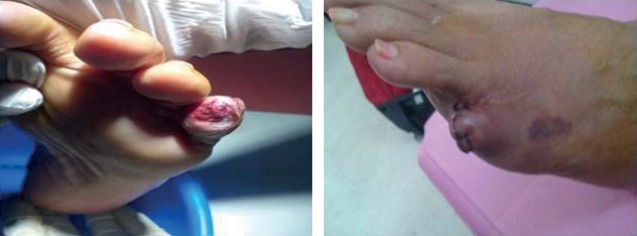
In May 2017 the patient's left little toe showed necrosis on the tip and dry gangrene set in. The toe was amputated after 2 months (figure 3). In August 2017 she developed a macular rash on the anterior aspect of her lower left leg. She also developed discolouration of the second right toe at this time.
Figure 3.
Gangrene in left toe.
During these episodes she was reviewed by a rheumatologist who diagnosed her with AAV-MPA and started her on cyclophosphamide (pulsed intravenous) and steroids.
She is currently under active follow-up by the rheumatology and pulmonology teams.
Discussion
ANCA are auto-antibodies directed against antigens found in cytoplasmic granules of neutrophils and monocytes. AAV is characterised by pauci-immune necrotising vasculitis of the small blood vessels [1], which comprise three different clinical syndromes: eosinophilic granulomatosis with polyangiitis (Churg–Strauss syndrome), granulomatosis with polyangiitis (Wegener's granulomatosis) and MPA.
The possible link between pulmonary fibrosis, ANCA positivity and vasculitis is poorly understood.
Many studies have assessed the relationship between ILD and ANCA. Arulkumaran et al. [3] suggested that out of 510 patients treated for ANCA vasculitis, 38% percent carried a diagnosis of MPA and 62% had granulomatosis with polyangiitis or eosinophilic granulomatosis with polyangiitis. 2.7% had ILD, which occurred in patients with MPA (7.2% of MPA patients) only.
In 1990, Nada et al. [4] reported three patients with an initial diagnosis of idiopathic pulmonary fibrosis (IPF) but were later correctly diagnosed as having pulmonary renal vasculitis. MPO antibodies (p-ANCA) were detected in two out of the three patients. Various cases reported emphasise the importance of a high index of suspicion for systemic vasculitis in elderly patients and the need to consider vasculitis in the differential diagnosis of IPF.
It has also been reported in a few studies that the diagnosis of ILD with UIP features often precedes the onset of features of vasculitis, particularly MPA [5]. In our patient, ILD with UIP features on HRCT preceded the clinical onset of AAV (MPA) by 4 years.
The other confounding element in our case is the presence of the rudimentary granuloma on the bronchial biopsy without any features of vasculitis/capillaritis, necrosis, eosinophilic infiltration or granulomatosis in the alveolar tissue. On directed re-evaluation of the histology, following the diagnosis of vasculitis, the pathologist was of the opinion that the probable incipient granuloma in the bronchial wall was, in all likelihood, a red-herring and non-contributory to the final diagnosis as there were no other features of vasculitis in the TBLB material, and an open lung biopsy was not available.
Kagiyama et al. [6] concluded that among 504 patients with IPF, 7.2% tested positive for ANCA (4% MPO and 3.2% proteinase-3 ANCA) at the time of first evaluation in the pulmonology clinic, in the absence of signs of vasculitis. Half of the patients had serial ANCA measurements, 11% of whom later developed a positive ANCA (5.7% MPO, 5.3% proteinase-3 ANCA). A clinical diagnosis of MPA developed in 25% of those patients with a positive MPO-ANCA.
Our patient had a UIP-ILD pattern on HRCT. This finding correlates with a French retrospective multicentre study including 49 patients with pulmonary fibrosis associated with AAV in which a typical UIP was the main HRCT pattern (43%) [7]. In another French retrospective study of 17 patients presenting with pulmonary fibrosis and a positive ANCA, HRCT analysis showed honeycombing, reticular intralobular opacities and traction bronchiectasis in all the patients with some degree of ground-glass attenuation (usually limited), whereas air-space consolidation was rare [8].
There are multiple theories regarding the pathomechanisms of ILD and AAV [6]. The first theory considers that repeated episodes of alveolar haemorrhage due to pulmonary capillaritis could be the pathogenesis of pulmonary fibrosis. Schanbel et al. [9] reported that subclinical alveolar bleeding was indeed a common finding in AAV. The second theory states that MPO-ANCA may play a direct role in the pathogenesis of pulmonary fibrosis. Foucher et al. [10] reported patchy inflammatory cell infiltrates throughout the parenchyma of the lung in their MPO-induced rat model of AAV and suggested that the presence of an anti-MPO directed autoimmune response contributes to generalised pulmonary tissue injury. The third theory is that IPF may induce ANCA and AAV and that pulmonary fibrosis is clinically manifested at the time of diagnosis in the majority of patients at the time of diagnosis of AAV. Namely, ANCA might be produced as a result of neutrophil destruction during the chronic inflammatory process [11].
In most studies, AAV patients with ILD have a worse prognosis than those without it, and patients with pulmonary fibrosis and ANCA had as low a prognosis as patients with IPF without ANCA [5]. Hence it is important that physicians are aware of this clinical association and the presence of ILD should be considered in all patients with AAV, especially those with MPO-ANCA positivity. The possibility that patients with ILD may subsequently develop features of systemic vasculitis should also be remembered. Currently, there is no specific treatment for patients with ILD and AAV/ANCA positivity [3] and treatment is on the same lines as patients with AAV.
Conclusion
It is important for physicians to be aware of the fact that AAV can present as an ILD with a UIP pattern. At presentation, all patients with ILD should be assessed for AAV and tested for ANCA positivity. Such patients should be monitored closely for signs and symptoms of vasculitis and should be treated early and aggressively for vasculitis.
Currently no guidelines exist for treating patients with ILD and AAV and these patients are managed along the lines of AAV patients [3]. However, in view of their worse prognosis [5], more evidence and research is required to evaluate whether it is beneficial to add an anti-fibrotic agent to the regimen of these patients on the same lines as management of patients with UIP-ILD.
Footnotes
Conflict of interest: None declared.
References
- 1.Jennette JC, Falk RJ, Bacon PA, et al. . 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum 2013; 65: 1–11. [DOI] [PubMed] [Google Scholar]
- 2.Ando M, Miyazaki E, Ishii T, et al. . Incidence of myeloperoxidase anti-neutrophil cytoplasmic antibody positivity and microscopic polyangitis in the course of idiopathic pulmonary fibrosis. Respir Med 2013; 107: 608–615. [DOI] [PubMed] [Google Scholar]
- 3.Arulkumaran N, Periselneris N, Gaskin G, et al. . Interstitial lung disease and ANCA associated vasculitis: a retrospective observational cohort study. Rheumatology 2011; 50: 2035–2043. [DOI] [PubMed] [Google Scholar]
- 4.Nada AK, Torres VE, Ryu JH, et al. . Pulmonary fibrosis as an unusual clinical manifestation of a pulmonary renal vasculitis in elderly patients. Mayo Clin Proc 1990; 65: 847–856. [DOI] [PubMed] [Google Scholar]
- 5.Katsumata Y, Kawaguchi Y, Yamanaka H, et al. . Interstitial lung disease with ANCA-associated vasculitis. Clin Med Insights Circ Respir Pulm Med 2015; 9: 51–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kagiyama N, Takayanagi N, Kanauchi T, et al. . Antineutrophil cytoplasmic antibody-positive conversion and microscopic polyangiitis development in patients with idiopathic pulmonary fibrosis. BMJ Open Respir Res 2015; 2: e000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Comarmond C, Crestani B, Tazi A, et al. . Pulmonary fibrosis in antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis: a series of 49 patients and review of the literature. Medicine 2014; 93: 340–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foulon G, Delaval P, Valeyre D, et al. . ANCA-associated lung fibrosis: analysis of 17 patients. Respir Med 2008; 102: 1392–1398. [DOI] [PubMed] [Google Scholar]
- 9.Schnabel A, Reuter M, Csernok E, et al. . Subclinical alveolar bleeding in pulmonary vasculitides: correlation with indices of disease activity. Eur Respir J 1999; 14: 118–124. [DOI] [PubMed] [Google Scholar]
- 10.Foucher P, Heeringa P, Petersen AH, et al. . Antimyeloperoxidase-associated lung disease. An experimental model. Am J Respir Crit Care Med 1999; 160: 987–994. [DOI] [PubMed] [Google Scholar]
- 11.Tzelepis GE, Kokosi M, Tzioufas A, et al. . Prevalence and outcome of pulmonary fibrosis in microscopic polyangiitis. Eur Respir J 2010; 36: 116–121. [DOI] [PubMed] [Google Scholar]