Abstract
Background
Antibodies targeting PD-1 and its ligand PDL1 are used in cancer immunotherapy but may lead to autoimmune diseases, including type 1 diabetes (T1D). It remains unclear whether PDL1 is expressed in pancreatic islets of people with T1D and how is it regulated.
Methods
The expression of PDL1, IRF1, insulin and glucagon was evaluated in samples of T1D donors by immunofluorescence. Cytokine-induced PDL1 expression in the human beta cell line, EndoC-βH1, and in primary human pancreatic islets was determined by real-time RT-PCR, flow cytometry and Western blot. Specific and previously validated small interference RNAs were used to inhibit STAT1, STAT2, IRF1 and JAK1 signaling. Key results were validated using the JAK inhibitor Ruxolitinib.
Findings
PDL1 was present in insulin-positive cells from twelve T1D individuals (6 living and 6 deceased donors) but absent from insulin-deficient islets or from the islets of six non-diabetic controls. Interferons-α and -γ, but not interleukin-1β, induced PDL1 expression in vitro in human islet cells and EndoC-βH1 cells. Silencing of STAT1 or STAT2 individually did not prevent interferon-α-induced PDL1, while blocking of JAKs – a proposed therapeutic strategy for T1D – or IRF1 prevented PDL1 induction.
Interpretation
These findings indicate that PDL1 is expressed in beta cells from people with T1D, possibly to attenuate the autoimmune assault, and that it is induced by both type I and II interferons via IRF1.
Keywords: Type 1 diabetes, PDL1, PDL-1, Pancreatic beta cells, Pancreatic islets, IRF1, Immune checkpoint inhibitor, CD274
Research in context.
Evidence before this study
Antibodies targeting PD-1 and its ligand PDL1 are used in cancer immunotherapy but may lead to autoimmune diseases, including type 1 diabetes (T1D). PDL1 expression is induced by several proinflammatory stimuli in cancer cells, particularly by interferons (IFNs), IL-1β, IL6, IL10, IL12, IL17, TGF-β and TNFα, and can also be induced in human islets by IFNs. The PD-1-PDL1 system is crucial to the preservation of tolerance to pancreatic beta cell antigens and, if disrupted, immune-mediated beta cell loss might proceed more quickly in genetically predisposed individuals.
Sources: Pubmed.
Search terms: PD-1; PDL1; cancer immunotherapy; endocrine autoimmune diseases; type 1 diabetes; pancreatic islets.
Added value of this study
We observed that PDL1 is present in insulin-positive cells from twelve T1D individuals but absent from insulin-deficient islets or from the islets of non-diabetic controls. Interferons-α and -γ, but not interleukin-1β, induce PDL1 expression in vitro in human beta cells. Silencing of STAT1 or STAT2 individually does not prevent interferon-α-induced PDL1, while blocking of JAKs – a proposed therapeutic strategy for T1D – or IRF1 prevents PDL1 induction. These findings indicate that PDL1 is expressed in beta cells from people with T1D, possibly to attenuate the autoimmune assault, and that it is induced by both type I and II interferons via IRF1.
Implications of all the available evidence
The present findings suggest the presence of an active dialog between beta cells and immune cells during insulitis, mediated by the release of pro- and anti-inflammatory cytokines by both immune cells and beta cells and by “danger signals” released from stressed or dying beta cells. It is usually assumed that this dialog has a largely negative outcome for the beta cells, but the present data suggest that two of the cytokines that are locally released during insulitis, namely IFNα and IFNγ, up-regulate PDL1 expression in human beta cells. Up-regulation of this immune checkpoint inhibitor may delay progression of human T1D, and may explain why beta cell destruction is heterogeneous in the pancreas if, for example, some beta cells express PDL1 to a greater extent than others. New drugs should be developed to prevent IFNα-induced pro-inflammatory effects, i.e. HLA class I up-regulation, chemokine production and ER stress, while preserving up-regulation of the protective PDL1. Our previous and present observations that inhibition of STAT2 prevents IFNα-induced HLA class I but not PDL1 up-regulation suggest that this may be feasible.
Alt-text: Unlabelled Box
1. Introduction
The introduction of immune checkpoint inhibitors into clinical practice represents a major improvement for the treatment of advanced cancers [1]. Antibodies targeting the programmed death receptor-1 (PD-1) and its ligand, programmed death-ligand 1 (PDL1) [2] are particularly efficacious. These reagents counteract the normally inhibitory effects of PDL1 (often up-regulated on tumor cells) on PD-1-expressing cytotoxic T-cells, thereby facilitating the targeting of the tumor cells by infiltrating lymphocytes.
PDL1 expression is induced by several proinflammatory stimuli in cancer cells, particularly by interferons (IFNs), IL-1β, IL6, IL10, IL12, IL17, TGF-β and TNFα [3]. The JAK/STAT-IRF1 pathway is the key regulator of IFN-mediated PDL1 expression in melanoma cells [4], while NF-κB activation is crucial for lipopolysaccharide (LPS)-induced PDL1 in macrophages [5]. A type I interferon signature precedes the development of autoimmunity in children genetically at risk for T1D [6] and IFNα, a member of the type I IFN family, is expressed in human islets from type 1 diabetic patients [7].
Immune checkpoints have physiological function, namely the maintenance of peripheral tolerance to self-antigens [8]. In accord with this, nearly 15% of patients treated with immune checkpoint inhibitors develop endocrine autoimmune diseases [9]. These individuals are prone to autoimmune diseases affecting the hypophysis, thyroid, adrenals and pancreatic beta cells [10], in the latter case, leading to type 1 diabetes [11]. In line with this, inhibition of PD-1-PDL1 signaling accelerates diabetes in NOD mice [12], while overexpression of PDL1 in beta cells prevents diabetes in these animals [13]. When coupled with induction of islet neogenesis in the liver, this can revert hyperglycemia [14].
Such findings indicate that the PD-1-PDL1 system is crucial to the preservation of tolerance to pancreatic beta cell antigens and that, if disrupted, immune-mediated beta cell loss might proceed more quickly in genetically predisposed individuals. It remains to be defined, however, whether PDL1 is expressed in the pancreatic islets of people with type 1 diabetes and, if that is the case, which factors mediate its up-regulation and the signaling pathways involved.
2. Materials and methods
2.1. Tissue
Formalin-fixed paraffin embedded pancreatic sections from the Exeter Archival Diabetes Biobank (EADB; http://foulis.vub.ac.be/) or from the DiViD biopsy study of living donors with recent-onset type 1 diabetes [15] were studied. These comprised samples from 6 non-diabetic patients (age range: 4–58 years) and 12 individuals with type 1 diabetes, in a total of 18 independent subjects studied (Table S1). All samples were studied with full ethical approval and following the adequate reporting standards for human studies.
2.2. Immunofluorescence
After standard dewaxing and rehydration, samples were subjected to heat–induced epitope retrieval (HIER) in Universal Buffer (Abcam, UK) then probed in a sequential manner with three different antibodies (Table S2). The relevant antigen-antibody complexes were detected using secondary antibodies conjugated with fluorescent dyes (Alexa Fluor™ anti-mouse 555, anti-rabbit TSA 488, anti-guinea pig 647) (Invitrogen, Paisley, U.K). Cell nuclei were stained with DAPI. After mounting, images were captured with a Leica AF6000 microscope (Leica, Milton Keynes, UK) and processed using the standard LASX Leica software platform. For the analysis of the immunostaining profile, two independent observers assessed the presence or absence and intensity of staining for each antigen (insulin and PDL1) starting from a position in which the immunostaining is negative (for instance, most control islets are scored as zero because they do not stain positively for PDL1; see Results) until cases in which most islets were positive and with intense staining (scored as 3). We have minimised bias by ensuring that each observer was blind to the status of the sample (group assignment and outcome assessment) and then comparing the scoring among the observers in a post hoc analysis. In cases where doubt persisted, two additional observers were invited to score the samples in a similarly blinded manner to verify the initial observations. The values shown describe the mean ± SEM of the two observers and the “n” represents independent biological data.
2.3. Culture of human EndoC-βH1 cells and human islets, and cell treatments
The human beta cell line EndoC-βH1 (kindly provided by Dr. R. Scharfmann, University of Paris, France [16], RRID:CVCL_L909) was cultured in Matrigel-fibronectin-coated plates as described [17]. Human islet isolation [18] from 10 non-diabetic organ donors (Table S3) was performed in accordance with the local Ethical Committee in Pisa, Italy. After arrival in Brussels, the islets were dispersed and cultured as described in [17]. All experiments shown with EndoC-βH1 cells or human islet cells (indicated as “n” in the figures) refer to independent biological samples.
Where indicated, cells were pre-treated with human IFNα (PeproTech Inc., Rocky Hill, NJ, RRID:SCR_006802) 2000 U/ml, human IFNγ (PeproTech Inc., RRID:SCR_006802) 1000 U/ml or human IL-1β (R&D Systems, Abingdon, UK, RRID:SCR_006140) 50 U/ml [17]. In some experiments cells were pre-treated for 1 h with ruxolitinib 1 or 4 μM (Selleckchem, Germany, RRID:SCR_003823) [19] and the drug maintained in the medium during the subsequent exposure to cytokines, or transfected with an unspecific control siRNA (siCTL) or siRNAs targeting STAT1 or STAT2 (these siRNAs have been previously validated by us, and were used at similar concentrations (30 nM) and transfection conditions as previously described [17], JAK1 and IRF1 before exposure to cytokines. The sequences of the siRNAs used are listed in Table S4.
2.4. Real-time PCR, Western blot and flow cytometry analysis
Poly(A) + mRNA was isolated using the Dynabeads mRNA DIRECT kit (Invitrogen) following the manufacturer's instructions, and reverse transcribed as described [17]. Quantification by real-time PCR was carried out using SYBR Green. Gene expression values were corrected by the housekeeping gene β-actin, as its expression is not affected by the experimental conditions used in this study [17]. The primers used are listed in Table S4.
For Western blot cells were lysed with Laemmli buffer. Total protein was resolved by 10% SDS-PAGE gel and then transferred to a nitrocellulose membrane. Immunoblot analysis was performed with the specific antibodies as indicated. Peroxidase-conjugated antibodies were used as secondary antibodies (list of antibodies in the Key Resources Table). Chemiluminescent substrate (SuperSignal West Femto, Thermo Scientific, Chicago, USA) was used to detect immunoreactive bands using a Bio-Rad chemi DocTM XRS+ system (Bio-Rad laboratories). Densitometric analysis of the bands was performed using the ImageLab software (Bio-Rad laboratories), and the values were normalized by the housekeeping protein α-tubulin and then by the highest value of each experiment considered as 1.
Flow cytometry analysis was performed as previously described [17], in brief, EndoC-βH1 cells were plated in 24-well plates and exposed to IFNα for 24 h. After that, cells were incubated with rabbit monoclonal anti-PDL1 antibody [28–8] (1:250) for 2 h at 4 °C, without permeabilisation, and subsequently with secondary antibody conjugated with fluorescent dyes for 1 h (1:500) (Alexa Fluor™ anti-rabbit 647) before performing flow cytometry analysis (FACSCanto; BD Biosciences, San Jose, CA, USA). Analysis was performed using FACSDiva software version 1.0 (BD Biosciences, San Jose, CA, USA). For the experiments with co-stain for PDL1 plus insulin, human islets were incubated with rabbit monoclonal anti-PDL1 antibody [28–8] (Abcam) (1:250) for 2 h at 4 °C, without permeabilisation, and subsequently with secondary antibody conjugated with fluorescent dyes for 1 h (1:500) (Alexa Fluor™ anti-rabbit 647). After that cells were fixed with paraformaldehyde 4%, permeabilized with saponin 1% and incubated with guinea pig anti-insulin (1:200) (DAKO, #A0564) and subsequently with secondary antibody conjugated with fluorescent dyes for 1 h (1:500) (Alexa Fluor™ anti-guinea pig 488) before performing flow cytometry analysis (FACSCanto; BD Biosciences, San Jose, CA, USA).
2.5. Promoter studies
EndoC-βH1 cells or dispersed human islets were transfected with plasmid constructs containing the firefly luciferase gene under the control of the human PDL1 promoter presenting or not a site-directed mutagenesis deletion in the IRF1 binding site (a kind gift from Prof. A. Ribas (University of California, UCLA, USA) [4]) using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer instructions. Twelve hours later, cells were exposed to IFNα (2000 U/ml) or IFNγ (1000 U/ml) for 24 h. Luciferase activities were assayed using the Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA). Values were corrected for the luciferase activity of the internal control plasmid, pRL-CMV, and shown as fold induction compared with untreated cells. In some experiments these constructs were co-transfected with siControl, siIRF1 #1 or #2, in the same conditions as described above.
2.6. Statistical analysis
Data are expressed as means ± SEM. A significant difference between experimental conditions was assessed by one-way or two-ways ANOVA followed by Bonferroni correction for multiple comparisons using the GraphPad Prism program version 6.0 (www.graphpad.com). Results with p < .05 were considered statistically significant.
3. Results
3.1. PDL1 expression is elevated in the insulin-containing islets (ICI) of individuals with type 1 diabetes
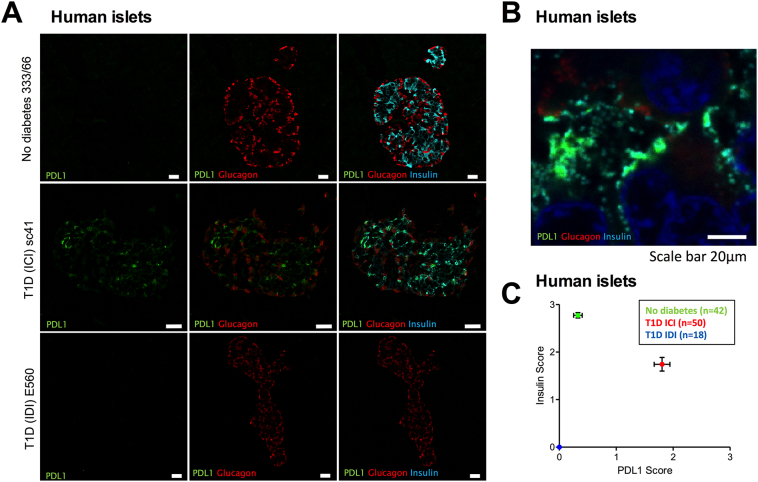
Initially we assessed the expression of PDL1 in the islets of both people with type 1 diabetes (3 children and 3 adults) along with age-matched controls. PDL1 immunostaining was absent (and consequently scored as zero) or, at best, observed as very weak in a few islets of non-diabetic individuals (Fig. 1A, upper panels; mean score 0.25) but was seen clearly in those with type 1 diabetes (Fig. 1A, middle panels; mean score 1.8, i.e. >7-fold higher than in non-diabetic individuals). The up-regulation of PDL1 was detected most readily in beta cells (Figs. 1A and B) although occasional alpha cells also appeared immunopositive for PDL1 (Fig. 1A). Positive immunostaining was not seen in insulin-deficient islets (Fig. 1A, lower panels). Scoring of islet PDL1 and insulin expression by two independent observers (unaware of sample identity) based on the intensity of immunostaining (0 to 3) in a large number of islets from each of the patient groups confirmed these observations (Fig. 1C). In non-diabetic individuals, most islets (70%) were negative for PDL1 with only 15 of 50 (30%) showing evidence of very weak expression. By contrast, among the Exeter Archival Diabetes Biobank (EADB) type 1 diabetes cases, 40 of 42 (95%) of the insulin-containing islets were clearly immunopositive for PDL1. All T1D insulin-deficient islets (n = 18) scored 0.
Fig. 1.
PDL1 expression is elevated in the insulin-containing islets (ICI) of individuals with type 1 diabetes, but not in insulin-deficient islets (IDI) or in islets from individuals without diabetes.
(A) Immunostaining of PDL1 alone (green; left panel), PDL1 (green) and glucagon (red; middle panel) or PDL1 (green) glucagon (red) and insulin (light blue; right panel) is shown in representative islets from individuals without diabetes (upper panels, A) and those with type 1 diabetes (middle and lower panels). Scale bar 20 μm.
(B) Higher magnification image demonstrating that PDL1 (green) is localized in beta cells (insulin; light blue).
(C) Scoring of PDL1 and insulin expression (scale of 0–3; as judged by independent observers) in islets from individuals without diabetes (green circle; n = 42 across 6 cases); insulin-containing islets (red circle; n = 50 across 6 cases) and insulin deficient islets (blue circle; n = 18, across 6 cases) from type 1 diabetes. The mean values ± SEM of the scores are shown.
PDL1 mRNA expression was also 5–8 higher in islets of a relatively recent-onset T1D individual as compared to the islets of a long-lasting T1D individual and three healthy donors (Fig. S1A) [20]. On the other hand, there was no increase in PDL1 expression in islets from individuals with impaired glucose tolerance (IGT) or type 2 diabetes (T2D) [21] (Fig. S1B) or in human islets exposed in vitro to the metabolic stressor palmitate [22] (Fig. S1C). Thus, PDL1 expression is increased during insulitis but is not modulated by dysglycemia and metabolic stressors related to T2D.
3.2. Interferons are the main regulators of PDL1 expression in human pancreatic beta cells
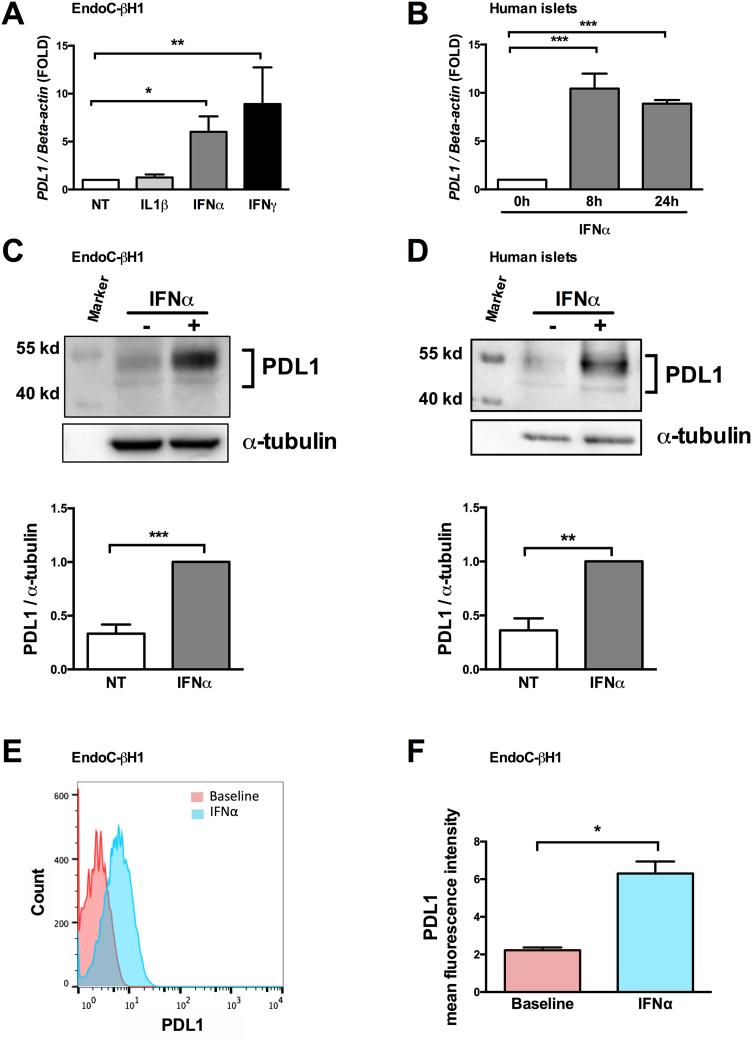
IFNα and IFNγ, but not IL-1β, induced PDL1 mRNA expression in EndoC-βH1 cells (Fig. 2A). PDL1 induction by both IFNs was of similar magnitude, which is in line with the observed similar expression of the receptors for IFNα and IFNγ in both EndoC-βH1 cells (Fig. S1D) and human islets (Fig. S1E). We focused the subsequent experiments on IFNα, which plays a key role in early insulitis [17]. IFNα induced both PDL1 mRNA and protein expression in EndoC-βH1 cells (Figs. 2A and C) and human islet cells (Figs. 2B and D), and led to a 2.8-fold increase in cell surface expression of PDL1 in EndoC-βH1 cells, as shown by flow cytometry (Figs. 2E and F).
Fig. 2.
Interferons type I and II, but not interleukin-1β, up-regulate PDL1 mRNA and protein expression in human pancreatic beta cells.
EndoC-βH1 cells (A, C, E and F) and primary human islets (B and D) were exposed to interferon-α (IFNα, 2000 U/ml), interferon-γ (IFNγ, 1000 U/ml) or interleukin-1β (IL1β, 50 U/ml) for 24 h (A, C, D, E, F) for the indicated time points (B).
(A and B) PDL1 mRNA expression was evaluated by real time RT-PCR. The values were normalized by the housekeeping gene beta-actin and are represented as fold induction compared to non-treated control cells (NT) (n = 3–5, * p < .05, ** p < .01, *** p < .001, ANOVA with Bonferroni correction).
(C and D) PDL1 protein expression was determined by Western blot (upper part of the figure), quantified by densitometry and normalized by the housekeeping protein α-tubulin and then by the highest value of each experiment considered as 1 (n = 6–7, ** p < .01, *** p < .001, paired t-test).
(E and F) PDL1 cell surface expression was quantified by flow cytometry analysis at baseline or after exposure to IFNα.
(E) Histograms represent changes in mean fluorescence intensity.
(F) The geometric mean of the mean fluorescence intensity was quantified at baseline and after exposure to IFNα (n = 3, * p < .05, paired t-test).
The mean values ± SEM are shown.
3.3. Inhibition of JAK1 decreases interferon-α-induced PDL1 expression
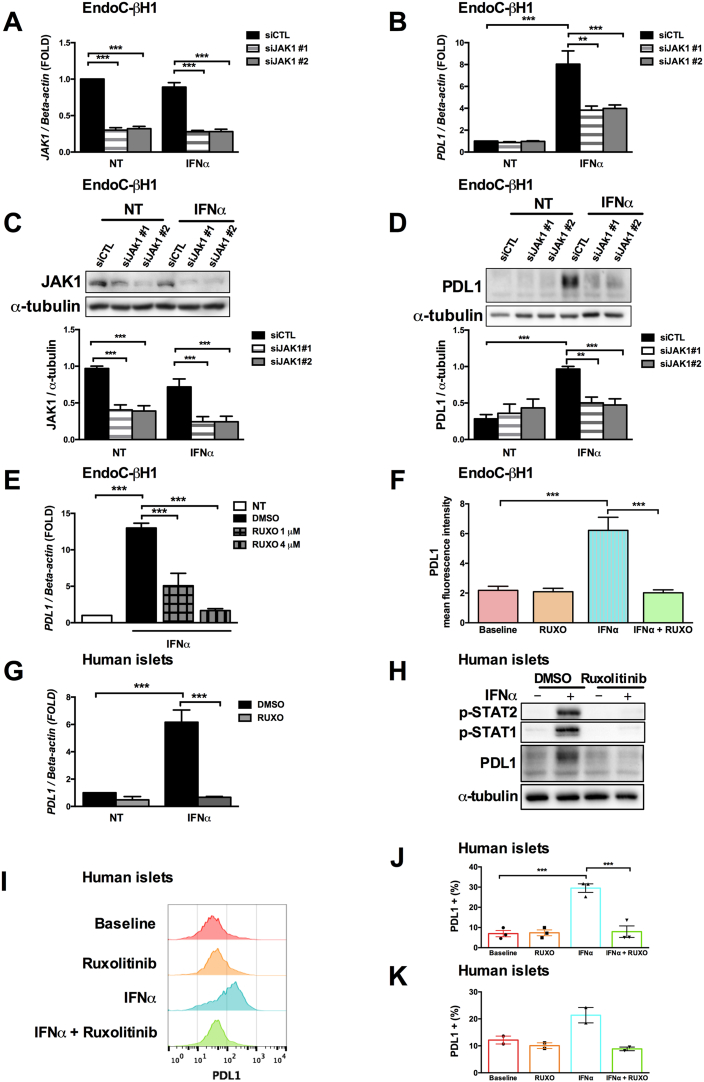
Inhibition of JAK1 (a key kinase for the signal transduction downstream of the IFNα receptor) by two specific siRNAs (Figs. 3A to D) reduced by 70% JAK1 expression at the mRNA (Fig. 3A) and protein levels (Fig. 3C) and led to a 55% reduction in interferon-induced PDL1 mRNA (Fig. 3B) and protein PDL1 expression (Fig. 3D). This partial inhibition in IFNα-mediated PDL1 induction after JAK1 silencing could be due to a second kinase, Tyk2, also involved in the downstream signaling of the type I interferon receptor [23] and/or to the fact that JAK1 silencing was only partial. To evaluate this, we used ruxolitinib, an FDA-approved JAK kinase inhibitor [24], which promoted a dose-dependent decrease in IFNα-induced PDL1 mRNA expression (Fig. 3E), and completely abolished PDL1 mRNA expression (Fig. 3E) and its cell surface expression in EndoC-βH1 cells (Fig. 3F) at the dose of 4 μM. Similar to EndoC-βH1 cells, the pretreatment of human islets with ruxolitinib prevented the IFNα-induced signaling pathways (Fig. 3H) leading to severe inhibition in PDL1 mRNA (Fig. 3G) and protein expression (Figs. 3H and I), which was confirmed by a decrease in global PDL1 positive (+) cells or PDL1+ and insulin+ cells as evaluated by flow cytometry (Figs. 3J and 3K). These results confirm the importance of JAK1 for IFNα-mediated PDL1 regulation.
Fig. 3.
JAK1 blocking by siRNAs or a chemical inhibitor decreases interferon-α-induced PDL1 expression.
(A to C) EndoC-βH1 cells were transfected with an unspecific control siRNA (siCTL) or two independent siRNAs targeting JAK1 (siJAK1 #1 and #2). After 48 h of recovery, the cells were exposed or not to interferon-α (IFNα, 2000 U/ml) for 24 h. Then, JAK1 and PDL1 mRNA and protein levels were determined by real time RT-PCR or Western blot, respectively. (n = 4–7, ** p < .01, *** p < .001, ANOVA with Bonferroni correction).
EndoC-βH1 cells (E and F) or human islets (G to K) were pretreated for 1 h with the JAK1 inhibitor ruxolitinib at the indicated doses or the vehicle DMSO (control) and then exposed to IFNα for 24 h.
(E and G) PDL1 mRNA expression was evaluated by real time RT-PCR. The values were normalized by the housekeeping gene beta-actin and are represented as fold induction compared to non-treated cells (NT) (n = 3–5, *** p < .001, ANOVA with Bonferroni correction).
(H) Phosphorylated STAT1, phosphorylated STAT2, PDL1 and α-tubulin protein expression were determined by Western blot (image representative of three independent experiments).
(F, I, J and K). IFNα-induced PDL1 cell surface expression was measured by flow cytometry in the presence or not of chemical JAK inhibition.
(I) Histograms represent changes in mean fluorescence intensity.
(J and K) The frequency of PDL1 positive cells was quantified in the entire islet population (J, n = 3) or among the insulin positive cells only (K, n = 2) (*** p < .001, ANOVA with Bonferroni correction).
The mean values ± SEM are shown.
3.4. Interferon-α-induced PDL1 expression depends on STAT1 and STAT2 activity
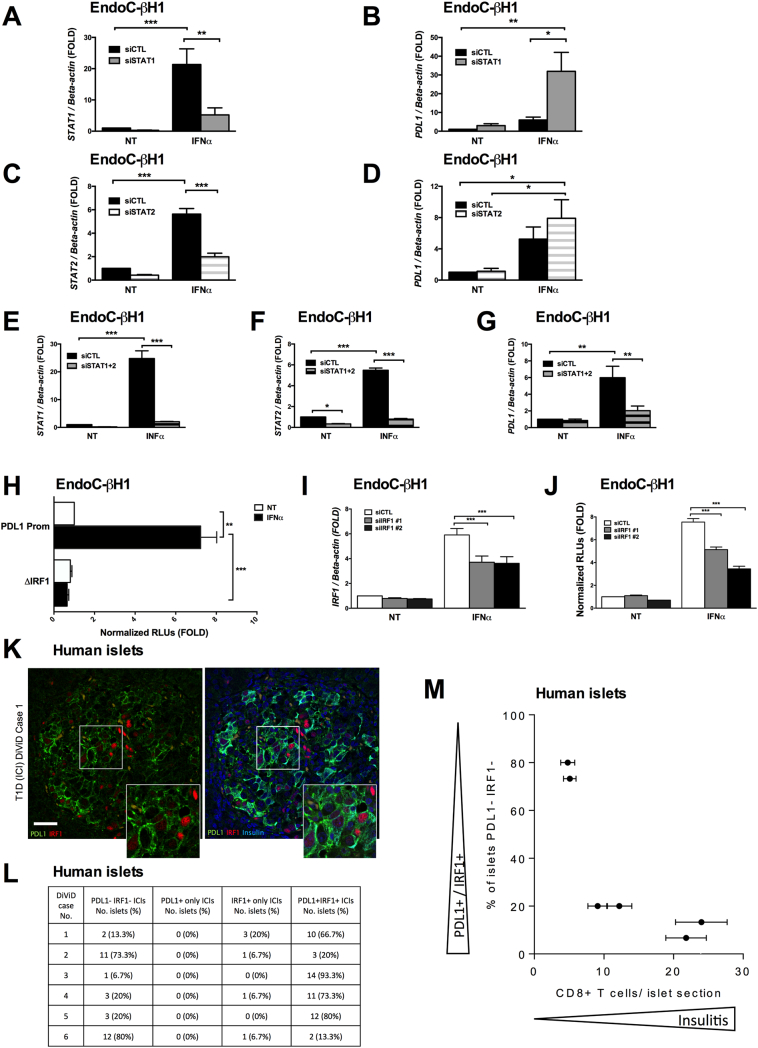
A time-course analysis of the key transcription factors induced by IFN showed a very early and marked activation of STAT1 and STAT2 (with peak at 0.5 h), followed by IRF1 activation (peak by 2 h) and then PDL1 up-regulation, starting at 8 h (Figs. S2A to F). STAT2 is a crucial transcription factor mediating IFNα-induced HLA class I expression and ER stress in human beta cells [17]. Surprisingly, STAT2 knockdown (KD) (Fig. 4C and S3B) induced a paradoxical effect, namely a clear trend for increased PDL1 expression (Fig. 4D), present in each individual experiment (not shown). Inhibition of STAT1 (Fig. 4A and S3A) led to an even more marked (>5-fold) paradoxical up-regulation of IFNα-induced PDL1 up-regulation at mRNA (Fig. 4B) and protein level (Fig. S3C). On the other hand, STAT1 inhibition (Fig. S3E) significantly decreased IFNγ-induced PDL1 expression (Fig. S3F), which is in line with the key role for this transcription factor in IFNγ signaling in beta cells [25]. STAT1 leads to STAT2 up-regulation (and vice versa) following IFNα exposure [17], a finding confirmed in the present study (Fig. S3D). To investigate whether this compensatory mechanism may explain the presently observed paradoxical effect of STAT1 and STAT2 KD on IFNα-induced PDL1 expression (Fig. 4B and 4D), we knocked down STAT1 and STAT2 in parallel (Figs. 4E and F) and observed that this double KD of STAT1/2 reduced IFNα-induced PDL1 mRNA (Fig. 4G) and protein (Fig. S3C) expression by 80%. These findings were confirmed in primary human islets (Fig. S4A to C).
Fig. 4.
Combined STAT1 plus 2 or IRF1 silencing decrease interferon-α-induced PDL1 expression.
(A to G) EndoC-βH1 cells were transfected with control siRNA (siCTL) or siRNAs targeting STAT1 (siSTAT1) and/or STAT2 (siSTAT2). After 48 h of recovery, the cells were exposed or not to interferon-α (IFNα, 2000 U/ml) for 24 h. The knockdown efficiency (A, C, E, F) and the PDL1 expression (B, D, G) were determined by real time RT-PCR. The values were normalized by the housekeeping gene beta-actin and are represented as fold induction compared to non-treated cells (NT) (n = 3–6, * p < .05, ** p < .01, *** p < .001, ANOVA with Bonferroni correction).
(H) EndoC-βH1 cells were transfected with a luciferase reporter construct containing the wild type human PDL1 promoter or the same construct presenting a deletion in the IRF1 binding site plus a pRL-CMV plasmid (used as internal control); cells were then exposed to IFNα for 24 h and luciferase activity was assayed. The values were corrected for the activity of the internal control (pRL-CMV) and are presented as normalized relative luciferase units (RLUs) in relation to non-treated cells (NT) considered as 1 (n = 4, ** p < .01, *** p < .001, ANOVA with Bonferroni correction).
(I and J) EndoC-βH1 cells were transfected with control siRNA (siCTL) or two independent siRNAs targeting IRF1 (siIRF1 #1 and #2) plus the wild type PDL1 promoter reporter and the pRL-CMV plasmid. The cells were then exposed to IFNα for 24 h and luciferase activity was assayed (J). The knockdown efficiency (I) was determined by real time RT-PCR. (n = 4, *** p < .001, ANOVA with Bonferroni correction).
(K) Immunostaining of PDL1 (green), IRF1 (red), insulin (light blue) and nuclei (dapi; dark blue) in the islets of an individual with T1D. The insets show the regions highlighted in each islet with a white box at higher magnification.
(L) 15 islets were selected at random from each of 6 individuals with type 1 diabetes (from the DiViD) cohort) and analysed for the proportion that were immunonegative for both PDL1 and IRF1, or were immunopositive for PDL1 alone, IRF1 alone or both PDL1 and IRF1.
(M) The proportion of islets that were immunonegative for PDL1 and IRF1 was scored in relation to islet inflammation measured as the mean number of CD8+ T-cells present in each individual patient studied.
The mean values ± SEM are shown.
IRF1, a key downstream transcription factor regulated by STATs in beta cells [25], was up-regulated several fold by IFNα (Fig. S2E). Transfection of EndoC-βH1 cells and dispersed human islets with a luciferase reporter construct of the human PDL1 promoter presenting or not a deletion of the IRF1 binding site [4] showed that IFNα induced a > 8-fold increase in PDL1 promoter activity, while IRF1 deletion abolished this effect (Figs. 4H and S4D); similar findings were obtained with IFNγ (Fig. S3G). Co-transfection of EndoC-βH1 cells with two independent siRNAs silencing IRF1 (Fig. 4I) plus the wild type PDL1 promoter partially (40–50%) decreased PDL1 promoter activity (Fig. 4J). The IRF1 knockdown (Fig. S4E) in human islets also decreased PDL1 protein expression induced by IFNα (Fig. S4F).
We next evaluated the correlation between PDL1 and IRF1 expression in human pancreas from people with T1D. IRF1 was not present in the islets of any control sample studied (not shown) but was markedly up-regulated in certain islet cells in T1D (Fig. 4K). PDL1 was frequently increased in concert with IRF1 immunopositivity (Fig. 4K) although the two antigens were not always localized within the same islet cells. We did not find any islets where PDL1 expression occurred in the absence of IRF1. To study these relationships further, 15 insulin-containing islets were selected at random from each of the 6 DiViD type 1 diabetes cases [15] (Table S1) and scored by two independent observers for expression of either PDL1 or IRF1 (Fig. 4L). Triple–staining was employed to label insulin, PDL1 and IRF1 and only insulin-containing islets were scored. IRF1 was present in 58 of 90 (64%) islet cells; most of which were beta cells. In 4 of the cases studied (DiViD 1,3,4 & 5) the majority of the IRF1 immunopositive islets (47 of 60 (78%)) also expressed PDL1, whereas in the remaining 2 cases (DiViD 2 & 6) a lower proportion were positive for both antigens (5 of 30 islets (17%)). Among all 6 cases, only 6 of 90 (6.7%) islets expressed IRF1 alone and none stained only for PDL1. Importantly, there was a clear correlation between insulitis (as judged by infiltration of CD8+ T-cells) and expression of PDL1/IRF1 (Fig. 4M).
4. Discussion
We presently show that PDL1 is expressed in pancreatic islets from T1D patients. Expression is seen most clearly in pancreatic beta cells. PDL1 was present in both young and older individuals with T1D, but was not found in insulin-deficient islets where beta cells had been destroyed. This could imply that PDL1 expression had been lost from such islets in parallel with the loss of beta cells. It cannot be excluded, however, that their early demise may have resulted from a failure to up-regulate PDL1 effectively during the inflammatory assault.
PDL1 up-regulation in beta cells appears to occur as a response to the inflammatory process associated with T1D, as it was not detected in the islets of non-diabetic individuals and it correlated with pancreatic islets CD8+ T-cell infiltration (Fig. 4M). In vitro studies using human islets and human insulin-producing EndoC-βH1 cells indicate that PDL1 is induced by type I and II IFNs, but not by IL-1β. Surprisingly, and in contrast to findings with cancer cells [4], the regulation of IFNα-induced PDL1 expression in beta cells is not dependent of STAT1 or STAT2 alone, but required both STATs (present data) and their downstream transcription factor IRF1. Of note, the present data also indicate that the early steps of IFNα signaling, via JAK activation, are essential for IFNα-induced PDL1 expression in beta cells.
There is probably an active dialog between beta cells and immune cells during insulitis, mediated by the release of pro- and anti-inflammatory cytokines by both immune cells and beta cells and by “danger signals” released from stressed or dying beta cells [26]. This dialog is triggered/modulated by putative environmental signals and the genetic background of the affected individuals [26,27]. It is usually assumed that the dialog has a largely negative outcome for the beta cells, but the present data suggest a more nuanced picture. Thus, two of the cytokines that may be elaborated during insulitis, namely IFNα (that probably plays an important role at the early stages of insulitis, related to local activation of innate immunity) and IFNγ (that probably plays a more important role at later stages, related to adaptive immunity), up-regulate PDL1 expression in human beta cells; these findings were corroborated by a very recent study by another group [28]. This up-regulation of an immune checkpoint inhibitor is probably of importance to delay progression of human T1D, and may explain why beta cell destruction is heterogeneous in the pancreas if, for example, some beta cells express PDL1 to a greater extent than others. This possibility is reinforced by the present findings that some, but not all, beta cells in the IFNα-treated islets or in histology samples from diabetic patients express PDL1. It is thus conceivable that beta cells with high PDL1 expression are able to resist T-cell mediated apoptosis over longer periods and thereby persist in the face of the immune assault, as is the case for PDL1-expressing cancer cells [29]. Indeed, there is an increased expression of PDL1 in long-term surviving beta cells under autoimmune attack in NOD mice [30]. The fact that anticancer therapies that block PD-1-PDL1 trigger T1D in some individuals [11] supports the concept that PDL1 provides protection to beta cells during insulitis, thereby enabling these individuals to evade or at least to delay the onset of diabetes. However, once PDL1 signaling is interrupted, the immune assault is intensified and accelerates beta cell loss. This possibility also bears relevance for the potential use of JAK inhibitors as anti-diabetic agents. Such drugs are currently under evaluation for the therapy of rheumatoid arthritis and various other autoimmune diseases [31,32] and have been suggested by us [19] and others [33] as potential therapies for T1D. The present results, and recent data showing that blockade of type I IFNs can either trigger or prevent diabetes depending on the stage of the autoimmune process in diabetes-prone NOD mice [34], raise concerns about this strategy. Thus, a careful evaluation will be required to determine the appropriate moment in the natural history of human T1D to introduce JAK inhibitors or other agents designed to disrupt IFN signaling. Ideally, new drugs should be developed to prevent IFNα-induced HLA class I up-regulation and ER stress [17,19] without preventing the protective PDL1 up-regulation. The observation that inhibition of STAT2, for instance, prevents IFNα-induced HLA class I up-regulation [17] but not PDL1 up-regulation (present data) implies that this may be feasible.
Funding
Fonds National de la Recherche Scientifique (FNRS), Welbio, Belgium; the Horizon 2020 Program, T2Dsystems; the National Institutes of Health, NIH-NIDDK-HIRN Consortium, Innovative Medicines Initiative 2 Joint (INNODIA).
Acknowledgments
Acknowledgements
The authors are grateful to Isabelle Millard, Anyishaï Musuaya, Nathalie Pachera and Michaël Pangerl of the ULB Center for Diabetes Research for excellent technical support.
This work was supported by grants from the Fonds National de la Recherche Scientifique (FNRS), Welbio CR-2015A-06, Belgium; the Horizon 2020 Program, T2Dsystems (GA667191); the National Institutes of Health, NIH-NIDDK-HIRN Consortium 1UC4DK104166-01. D.L.E and P.M. have received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement No 115797 (INNODIA). This Joint Undertaking receives support from the Union's Horizon 2020 research and innovation programme and “EFPIA”, ‘JDRF” and “The Leona M. and Harry B. Helmsley Charitable Trust”. Additional support was from a JDRF Career Development Award (5-CDA-2014-221-A-N) to S.J.R, a JDRF research grant awarded to the network of Pancreatic Organ Donors – Virus (nPOD-V) consortium (JDRF 25-2012-516); an MRC Project Grant MR/P010695/1 awarded to S.J.R & N.G.M. and a studentship grant from the Norman Family Trust (to S.J.R. & N.G.M.). L.M. was supported by an FNRS postdoctoral fellowship.
Author contributions
M.L.C., N.G.M., S.J.R. and D.L.E. conceived, designed and supervised the experiments. M.L.C., J.L.E.H., L.M., J.C., R.S.D.S., P.L., F.M.M.P., A.O.B., A.C. and S.J.R. acquired data. M.B., L.K., K.D-J. and P.M. contributed with material and reagents. M.L.C., N.G.M., S.J.R. and D.L.E. wrote the manuscript. All authors revised the manuscript. D.L.E. is the guarantor of this work and, as such, have full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of interests
The authors declare that they have no conflicts of interest with the contents of this article.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.ebiom.2018.09.040.
Contributor Information
Maikel L. Colli, Email: mcolli@ulb.ac.be.
Décio L. Eizirik, Email: deizirik@ulb.ac.be.
Appendix A. Supplementary data
Supplementary material
References
- 1.Sharma P., Allison J.P. Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell. 2015;161(2):205–214. doi: 10.1016/j.cell.2015.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ohaegbulam K.C., Assal A., Lazar-Molnar E., Yao Y., Zang X. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol Med. 2015;21(1):24–33. doi: 10.1016/j.molmed.2014.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sun C., Mezzadra R., Schumacher T.N. Regulation and Function of the PD-L1 Checkpoint. Immunity. 2018;48(3):434–452. doi: 10.1016/j.immuni.2018.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Garcia-Diaz A., Shin D.S., Moreno B.H., Saco J., Escuin-Ordinas H., Rodriguez G.A. Interferon Receptor Signaling Pathways Regulating PD-L1 and PD-L2 Expression. Cell Rep. 2017;19(6):1189–1201. doi: 10.1016/j.celrep.2017.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Loke P., Allison J.P. PD-L1 and PD-L2 are differentially regulated by Th1 and Th2 cells. Proc Natl Acad Sci U S A. 2003;100(9):5336–5341. doi: 10.1073/pnas.0931259100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ferreira R.C., Guo H., Coulson R.M., Smyth D.J., Pekalski M.L., Burren O.S. A type I interferon transcriptional signature precedes autoimmunity in children genetically at risk for type 1 diabetes. Diabetes. 2014;63(7):2538–2550. doi: 10.2337/db13-1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang X., Yuang J., Goddard A., Foulis A., James R.F., Lernmark A. Interferon expression in the pancreases of patients with type I diabetes. Diabetes. 1995;44(6):658–664. doi: 10.2337/diab.44.6.658. [DOI] [PubMed] [Google Scholar]
- 8.Martin-Orozco N., Wang Y.H., Yagita H., Dong C. Cutting Edge: Programmed death (PD) ligand-1/PD-1 interaction is required for CD8+ T cell tolerance to tissue antigens. J Immunol. 2006;177(12):8291–8295. doi: 10.4049/jimmunol.177.12.8291. [DOI] [PubMed] [Google Scholar]
- 9.Barroso-Sousa R., Ott P.A., Hodi F.S., Kaiser U.B., Tolaney S.M., Min L. Endocrine dysfunction induced by immune checkpoint inhibitors: Practical recommendations for diagnosis and clinical management. Cancer. 2018;124(6):1111–1121. doi: 10.1002/cncr.31200. [DOI] [PubMed] [Google Scholar]
- 10.Cukier P., Santini F.C., Scaranti M., Hoff A.O. Endocrine side effects of cancer immunotherapy. Endocr Relat Cancer. 2017;24(12) doi: 10.1530/ERC-17-0358. (T331-T47) [DOI] [PubMed] [Google Scholar]
- 11.Stamatouli A.M., Quandt Z., Perdigoto A.L., Clark P.L., Kluger H., Weiss S.A. Collateral damage: Insulin-Dependent Diabetes Induced with Checkpoint Inhibitors. Diabetes. 2018;67(8):1471–1480. doi: 10.2337/dbi18-0002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ansari M.J., Salama A.D., Chitnis T., Smith R.N., Yagita H., Akiba H. The programmed death-1 (PD-1) pathway regulates autoimmune diabetes in nonobese diabetic (NOD) mice. J Exp Med. 2003;198(1):63–69. doi: 10.1084/jem.20022125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.El Khatib M.M., Sakuma T., Tonne J.M., Mohamed M.S., Holditch S.J., Lu B. Beta-cell-targeted blockage of PD1 and CTLA4 pathways prevents development of autoimmune diabetes and acute allogeneic islets rejection. Gene Ther. 2015;22(5):430–438. doi: 10.1038/gt.2015.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li R., Lee J., Kim M.S., Liu V., Moulik M., Li H. PD-L1-driven tolerance protects neurogenin3-induced islet neogenesis to reverse established type 1 diabetes in NOD mice. Diabetes. 2015;64(2):529–540. doi: 10.2337/db13-1737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Krogvold L., Edwin B., Buanes T., Ludvigsson J., Korsgren O., Hyoty H. Pancreatic biopsy by minimal tail resection in live adult patients at the onset of type 1 diabetes: experiences from the DiViD study. Diabetologia. 2014;57(4):841–843. doi: 10.1007/s00125-013-3155-y. [DOI] [PubMed] [Google Scholar]
- 16.Ravassard P., Hazhouz Y., Pechberty S., Bricout-Neveu E., Armanet M., Czernichow P. A genetically engineered human pancreatic beta cell line exhibiting glucose-inducible insulin secretion. J Clin Invest. 2011;121(9):3589–3597. doi: 10.1172/JCI58447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Marroqui L., Dos Santos R.S., Op de Beeck A., Coomans De Brachene A., Marselli L., Marchetti P. Interferon-alpha mediates human beta cell HLA class I overexpression, endoplasmic reticulum stress and apoptosis, three hallmarks of early human type 1 diabetes. Diabetologia. 2017;60(4):656–667. doi: 10.1007/s00125-016-4201-3. [DOI] [PubMed] [Google Scholar]
- 18.Marchetti P., Bugliani M., Lupi R., Marselli L., Masini M., Boggi U. The endoplasmic reticulum in pancreatic beta cells of type 2 diabetes patients. Diabetologia. 2007;50(12):2486–2494. doi: 10.1007/s00125-007-0816-8. [DOI] [PubMed] [Google Scholar]
- 19.Coomans De Brachene A., Dos Santos R.S., Marroqui L., Colli M.L., Marselli L., Mirmira R.G. IFN-alpha induces a preferential long-lasting expression of MHC class I in human pancreatic beta cells. Diabetologia. 2018;61(3):636–640. doi: 10.1007/s00125-017-4536-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mastracci T.L., Turatsinze J.V., Book B.K., Restrepo I.A., Pugia M.J., Wiebke E.A. Distinct gene expression pathways in islets from individuals with short- and long-duration type 1 diabetes. Diabetes Obes Metab. 2018;20(8):1859–1867. doi: 10.1111/dom.13298. [DOI] [PubMed] [Google Scholar]
- 21.Fadista J., Vikman P., Laakso E.O., Mollet I.G., Esguerra J.L., Taneera J. Global genomic and transcriptomic analysis of human pancreatic islets reveals novel genes influencing glucose metabolism. Proc Natl Acad Sci U S A. 2014;111(38):13924–13929. doi: 10.1073/pnas.1402665111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cnop M., Abdulkarim B., Bottu G., Cunha D.A., Igoillo-Esteve M., Masini M. RNA sequencing identifies dysregulation of the human pancreatic islet transcriptome by the saturated fatty acid palmitate. Diabetes. 2014;63(6):1978–1993. doi: 10.2337/db13-1383. [DOI] [PubMed] [Google Scholar]
- 23.Marroqui L., Dos Santos R.S., Floyel T., Grieco F.A., Santin I., Op de Beeck A. TYK2, a Candidate Gene for Type 1 Diabetes, Modulates Apoptosis and the Innate Immune Response in Human Pancreatic beta-Cells. Diabetes. 2015;64(11):3808–3817. doi: 10.2337/db15-0362. [DOI] [PubMed] [Google Scholar]
- 24.Harrison C., Kiladjian J.J., Al-Ali H.K., Gisslinger H., Waltzman R., Stalbovskaya V. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med. 2012;366(9):787–798. doi: 10.1056/NEJMoa1110556. [DOI] [PubMed] [Google Scholar]
- 25.Moore F., Naamane N., Colli M.L., Bouckenooghe T., Ortis F., Gurzov E.N. STAT1 is a master regulator of pancreatic beta-cell apoptosis and islet inflammation. J Biol Chem. 2011;286(2):929–941. doi: 10.1074/jbc.M110.162131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eizirik D.L., Colli M.L., Ortis F. The role of inflammation in insulitis and beta-cell loss in type 1 diabetes. Nat Rev Endocrinol. 2009;5(4):219–226. doi: 10.1038/nrendo.2009.21. [DOI] [PubMed] [Google Scholar]
- 27.Op de Beeck A., Eizirik D.L. Viral infections in type 1 diabetes mellitus--why the beta cells? Nat Rev Endocrinol. 2016;12(5):263–273. doi: 10.1038/nrendo.2016.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Osum K.C., Burrack A.L., Martinov T., Sahli N.L., Mitchell J.S., Tucker C.G. Interferon-gamma drives programmed death-ligand 1 expression on islet beta cells to limit T cell function during autoimmune diabetes. Sci Rep. 2018;8(1):8295. doi: 10.1038/s41598-018-26471-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hughes J., Vudattu N., Sznol M., Gettinger S., Kluger H., Lupsa B. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care. 2015;38(4):e55–e57. doi: 10.2337/dc14-2349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rui J., Deng S., Arazi A., Perdigoto A.L., Liu Z. Herold KC. Beta cells that resist immunological attack develop during progression of autoimmune diabetes in NOD mice. Cell Metab. 2017;25(3):727–738. doi: 10.1016/j.cmet.2017.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fleischmann R., Kremer J., Cush J., Schulze-Koops H., Connell C.A., Bradley J.D. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N Engl J Med. 2012;367(6):495–507. doi: 10.1056/NEJMoa1109071. [DOI] [PubMed] [Google Scholar]
- 32.Gladman D., Rigby W., Azevedo V.F., Behrens F., Blanco R., Kaszuba A. Tofacitinib for psoriatic arthritis in patients with an inadequate response to TNF inhibitors. N Engl J Med. 2017;377(16):1525–1536. doi: 10.1056/NEJMoa1615977. [DOI] [PubMed] [Google Scholar]
- 33.Trivedi P.M., Graham K.L., Scott N.A., Jenkins M.R., Majaw S., Sutherland R.M. Repurposed JAK1/JAK2 inhibitor reverses established autoimmune insulitis in NOD mice. Diabetes. 2017;66(6):1650–1660. doi: 10.2337/db16-1250. [DOI] [PubMed] [Google Scholar]
- 34.Rahman M.J., Rodrigues K.B., Quiel J.A., Liu Y., Bhargava V., Zhao Y. Restoration of the type I IFN-IL-1 balance through targeted blockade of PTGER4 inhibits autoimmunity in NOD mice. JCI Insight. 2018;3(3) doi: 10.1172/jci.insight.97843. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material