Abstract
Background
Diallyl trisulfide (DATS), a garlic-derived organosulfuric compound, has been documented for potential anti-inflammatory effects. However, the mechanism in microglia remains unknown. In this study, we investigated the anti-inflammatory effects of DATS in lipopolysaccharide (LPS)-stimulated BV2 microglial cells.
Methods
The effects of DATS on LPS-induced pro-inflammatory mediators such as nitric oxide (NO) and prostaglandin E2 (PGE2) were assessed under conditions not in the cytotoxicity of DATS. The protein expression of inflammation regulatory genes was measured by Western blot analysis.
Results
DATS significantly inhibited the LPS-induced secretion of NO and PGE2, which was associated with the suppression of their regulatory genes, inducible NO synthase and COX-2. DATS had been shown to inhibit nuclear translocation of NF-κB by destroying the degradation and phosphorylation of IκB-α inhibitors in the cytoplasm. In addition, DATS effectively inhibited the expression of LPS-induced toll-like receptor 4 (TLR4) and myeloid differentiation factor 88. Furthermore, DATS markedly reduced the LPS-induced expression of chemokine (CXC motif) ligand (CXCL) 12 and CXC receptor (CXCR) 4, demonstrating its capacity to block chemo-attractive activity.
Conclusions
These results indicate that DATS inhibits the activation of the CXCL12/CXCR4 axis associated with antagonizing effect on TLR4 and blocks NF-κB signaling, thus demonstrating anti-inflammatory effects against LPS stimulation.
Keywords: Diallyl trisulfide, Anti-inflammation, NF-κB, Toll-like receptor 4, Chemokine
INTRODUCTION
Microglia, considered to be macrophages in the central nervous system, plays a central role in brain development and maintenance. However, microglial dysfunction due to hyperactivity in response to inflammatory signals is closely related to the onset and progression of various neuro-degenerative diseases by damaging brain neurons.1,2 For example, the excessive activation of microglial cells by pathogenic endotoxin, such as lipopolysaccharide (LPS) present in the outer membrane of Gram-negative bacteria, can increase the production of pro-inflammatory mediators and cytokines through toll-like receptor 4 (TLR4) signaling.3,4 TLR activates several downstream signaling pathways and ultimately induce phosphorylation and degradation of IκB, an inhibitor of NF-κB, by recruiting myeloid differentiation gene 88 (MyD88), an adapter molecule of TLRs.5,6 When IκB is degraded, free NF-κB consequently translocates into the nucleus and causes the transcription of a series of inflammatory genes.5–7 Furthermore, the activation of chemokine (CXC motif) receptors (CXCRs) by chemokine (CXC motif) ligands (CXCLs) associated with LPS stimulation, along with TLR4 signaling activated by LPS, is involved in the release of inflammatory mediators and cytokines and exhibits substantial chemo-attractive activity.8,9 Therefore, the regulation of inflammatory response by blocking excessive microglia activation is an important tool that weakens the induction and progression of multiple brain diseases.
Diallyl trisulfide (DATS) is one of the natural organic sulfur compounds derived from the Allium genus, which includes garlic, and is known to have anti-inflammatory potentials.10,11 For example, You et al.12 reported that DATS attenuated the LPS-induced production of pro-inflammatory mediators and cytokines by inhibiting NF-κB and mitogen-activated protein kinases (MAPKs) signaling pathways in the macrophage model. In addition, Wang et al.13 showed that DATS could alleviate endothelial cell inflammation caused by hypoxia and improve endothelial cell proliferation and migration. Although DATS has been reported to inhibit the production of LPS-induced pro-inflammatory factors and monocyte chemoattractant protein-1 induced by inhibiting the NF-κB activation in the microglia model,14 the relevance of TLR4 signaling and chemokine ligand/receptor axis is still poorly documented. Therefore, in this study, the anti-inflammatory effect of DATS was reaffirmed in LPS-stimulated BV2 microglial cells, and the effects of DATS on TLR4 signaling and CXCL12/CXCR4 pathway were investigated in this process.
MATERIALS AND METHODS
1. Cell culture and lipopolysaccharide stimulation
BV2 microglia were cultured in Dulbecco’s modified Eagle’s medium (DMEM; WelGENE Inc., Daegu, Korea) containing 10% FBS, L-glutamine (2 mM), penicillin (100 U/mL), and streptomycin (100 U/mL, WelGENE Inc.) at 37°C in a humidified atmosphere containing 5% CO2 and 95% air. DATS was purchased from LKT Laboratories (St. Paul, MN, USA), dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich Chemical Co., St. Louis, MO, USA), and adjusted to the final concentrations using complete culture medium. To stimulate the cells, the medium was replaced with fresh DMEM, and 100 ng/mL LPS was added in the presence or absence of DATS for the indicated periods.
2. Assessment of cell viability
To evaluate the cytotoxic potential of DATS, BV2 cells were treated with different concentrations of DATS for 1 hour before incubation with or without LPS (100 ng/mL). After culturing for 24 hours, 0.5 mg/mL of MTT (Sigma-Aldrich Chemical Co.) solution was added to each well, followed by incubation at 37°C in dark conditions for 3 hours. The MTT solution was then removed and 5% DMSO was added to dissolve the crystals. The viable cells were detected by reading the absorbance of formazan at 540 nm using an ELISA microplate reader (Dynatech Laboratories, Chantilly, VA, USA). The optical density of formazan formed in control (untreated) cells was used to demonstrate 100% viability.
3. Measurement of nitric oxide and prostaglandin E2 production
The levels of nitric oxide (NO) production were determined indirectly by measuring the stable NO catabolite nitrite in a medium by Griess reaction. Briefly, the conditioned medium was mixed with the same volume of Griess reagent (Sigma-Aldrich Chemical Co.) and incubated at room temperature for 10 minutes. The optical density at 540 nm was measured with an ELISA microplate reader and the concentration of nitrite was calculated according to a standard curve generated from known concentrations of sodium nitrite. Prostaglandin E2 (PGE2) concentrations in conditioned medium were measured using a commercial PGE2 ELISA kit (Cayman Chemical Co., Ann Arbor, MI, USA) according to the manufacturer’s instructions.
4. Protein isolation and Western blot analysis
As described previously,15 cells were harvested and lysed with cell lysis buffer. The cytoplasmic and nuclear extracts were prepared using an NE-PER Nuclear and Cytoplasmic Extraction Reagents kit (Pierce Biotechnology, Rockford, IL, USA) according to the manufacturer’s instructions. For Western blotting, the same amount of protein was electrophoretically transferred onto polyvinylidene difluoride membranes (Schleicher & Schuell, Keene, NH, USA) following separation in SDS gel electrophoresis. The membranes were then blocked for 1 hour with 5% skim milk powder/TBS containing 0.1% Triton X-100 (TBST) and probed with specific primary antibodies at 4°C overnight. After washing the primary antibodies with TBST, the membranes were incubated with appropriate horseradish peroxidase-conjugated secondary antibodies (Amersham Biosciences, Westborough, MA, USA) for 2 hours at room temperature. Protein bands were detected by an enhanced chemiluminescence (ECL) kit (Amersham Biosciences) according to the manufacturer’s instructions.
5. Statistical analysis
All numerical data are expressed as mean ± SD. Student t-test was used to analyze the significant differences between the mean values. All statistical analyses were performed using the SPSS ver. 17.0 software (SPSS Inc., Chicago, IL, USA). Value of P < 0.05 was considered statistically significant.
RESULTS
1. Assessment of Diallyl trisulfide on cell viability in BV2 microglial cells
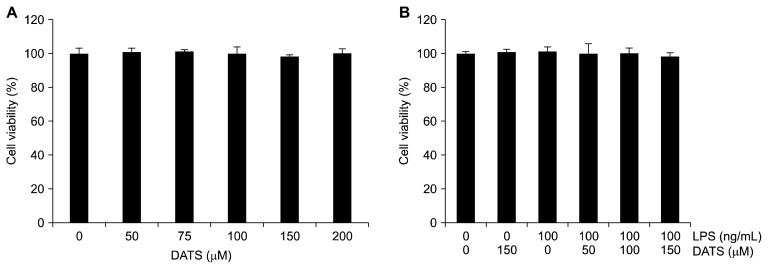
As shown in Figure 1A, the survival rate of BV2 cells was not significantly affected by up to 200 μM of DATS treatment for 24 hours. Also, no significant changes were observed in DATS concentrations up to 150 μM in the presence of 100 ng/mL LPS (Fig. 1B).
Figure 1.
Effects of diallyl trisulfide (DATS) and lipopolysaccharide (LPS) on the cell viability of BV2 microglial cells. The cells were treated with various concentrations of DATS for 24 hours (A) or pre-treated with the indicated concentrations of DATS for 1 hour prior to 100 ng/mL LPS treatment for 24 hours (B). The cell viability was assessed with an MTT reduction assay, and the results were expressed as the percentage of surviving cells over control cells (no addition of DATS and LPS). Values represent the means ± SD of the three independent experiments.
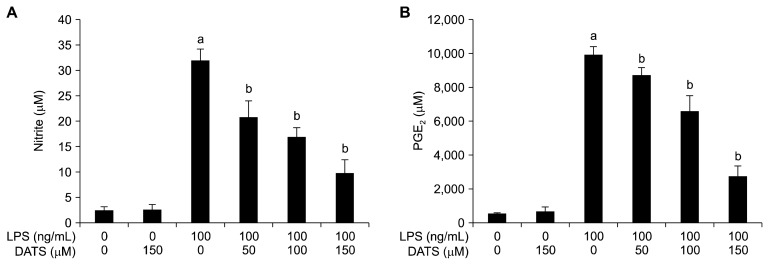
2. Suppression of lipopolysaccharide-induced nitric oxide and prostaglandin E2 production by diallyl trisulfide in BV2 microglial cells
The levels of NO and PGE2 in the culture supernatants were determined by Griess reaction assay and ELISA, respectively. As indicated in Figure 2, when treated with LPS alone, production of NO and PGE2 was greatly increased compared to the untreated control. Conversely, DATS significantly inhibited NO and PGE2 secretion in BV2 cells in a concentration-dependent manner. Thus, inhibition of NO and PGE2 production by DATS is independent of cytotoxicity of DATS and is due to DATS itself.
Figure 2.
Inhibition of nitric oxide (NO) and prostaglandin E2 (PGE2) production by diallyl trisulfide (DATS) in lipopolysaccharide (LPS)-stimulated BV2 microglial cells. The cells were pre-treated with the indicated concentrations of DATS for 1 hour prior to incubation with 100 ng/mL LPS for 24 hours. The levels of NO (A) and PGE2 (B) in the culture media were measured by Griess assay and a commercial ELISA kit, respectively. Each value indicates the mean ± SD and is representative of the results obtained from three independent experiments (aP < 0.05 compared with the control; bP < 0.05 compared with the cells cultured with 100 ng/mL LPS).
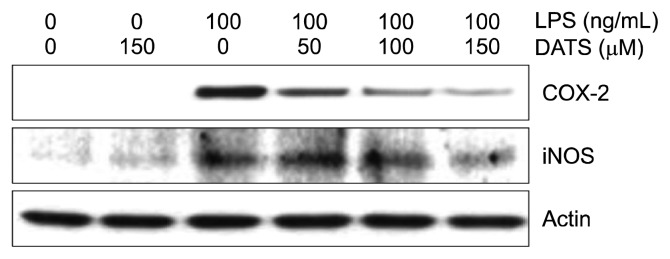
3. Attenuation of lipopolysaccharide-induced inducible nitric oxide synthase and COX-2 expression by diallyl trisulfide in BV2 microglial cells
We next determined whether the inhibitory effect of DATS on NO and PGE2 production was related to the regulation of the expression of their synthesis enzymes, inducible nitric oxide synthase (iNOS) and COX-2. As shown Figure 3, DATS concentration-dependently inhibited the expression of iNOS and COX-2 protein induced by LPS, indicating that DATS inhibits NO and PGE2 production by decreasing the expression of their encoding genes.
Figure 3.
Inhibition of the lipopolysaccharide (LPS)-induced expression of inducible nitric oxide synthase (iNOS) and COX-2 in BV2 microglial cells. BV2 cells were pre-treated with various concentrations of diallyl trisulfide (DATS) for 1 hour, followed by 100 ng/mL LPS treatment for 24 hours. The total proteins were isolated and Western blot analyses were performed using the indicated antibodies and an enhanced chemiluminescence detection system. The experiments were repeated three times, and similar results were obtained. Actin was used as the internal control.
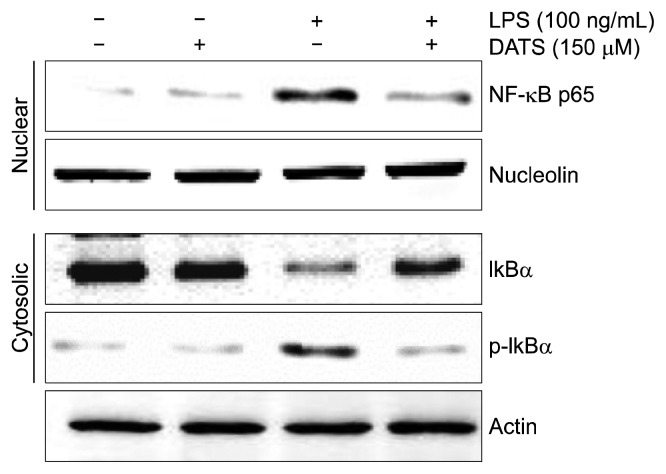
4. Inhibition of lipopolysaccharide-induced activation of NF-κB by diallyl trisulfide in BV2 microglial cells
We further investigated whether DATS could attenuate the activity of NF-κB induced by LPS in BV2 cells. Our data using cytoplasmic and nuclear extracts indicated that DATS pretreatment inhibited the accumulation of NF-κB p65 subunit in nuclei associated with attenuation of IκBα degradation and phosphorylation in LPS-stimulated BV2 cells (Fig. 4), implying that DATS inhibited the transcriptional activation of NF-κB.
Figure 4.
Inhibition of the lipopolysaccharide (LPS)-induced NF-κB activation by diallyl trisulfide (DATS) in BV2 microglial cells. The cells were pre-treated with 150 μM DATS for 1 hour before 100 ng/mL LPS treatment for 1 hour. The nuclear and cytosolic proteins were prepared for Western blot analysis using anti-NF-κB p65 and anti-IκBα antibodies and an enhanced chemiluminescence detection system. Nucleolin and actin were used as the internal controls for the nuclear and cytosolic fractions, respectively.
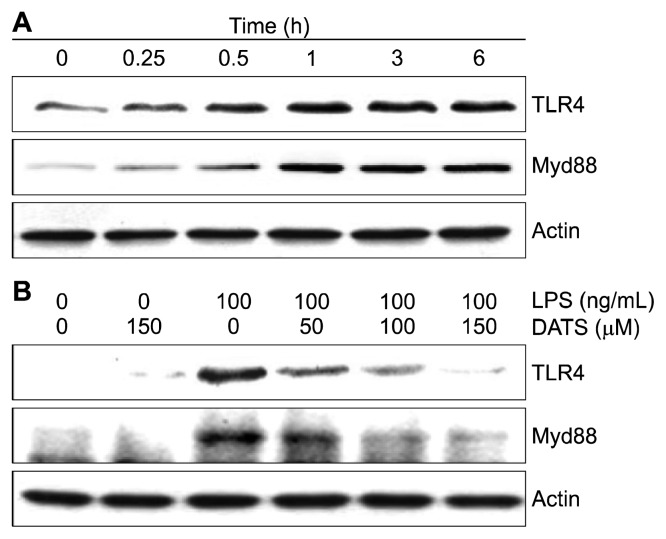
5. Reduction of lipopolysaccharide-induced toll-like receptor 4 and myeloid differentiation gene 88 expressions by diallyl trisulfide in BV2 microglial cells
Expression of TLR4 and Myd88 was investigated to examine whether the anti-inflammatory effect of DATS is associated with blocking of the TLR4 signaling pathway. According to immuno-blotting results, the levels of TLR4 and Myd88 protein were increased by LPS treatment according to treatment time (Fig. 5A). However, LPS-induced expression of the proteins was diminished when DATS was pre-treated (Fig. 5B), suggesting that DATS could inhibit the LPS-activated TLR signaling pathway in BV2 cells.
Figure 5.
Effects of diallyl trisulfide (DATS) on the lip-opolysaccharide (LPS)-induced expression of toll-like receptor 4 (TLR4) and myeloid differentiation gene 88 (Myd88) in BV2 micro-glial cells. The cells were treated with 100 ng/mL LPS for the indicated times (A) or pretreated with the indicated concentrations of DATS for 1 hour prior to LPS treatment for 6 hours (B). The total proteins were prepared for Western blot analysis using anti-TLR4 and anti-Myd88 antibodies and an enhanced chemiluminescence (ECL) detection system. Actin was used as the internal control.
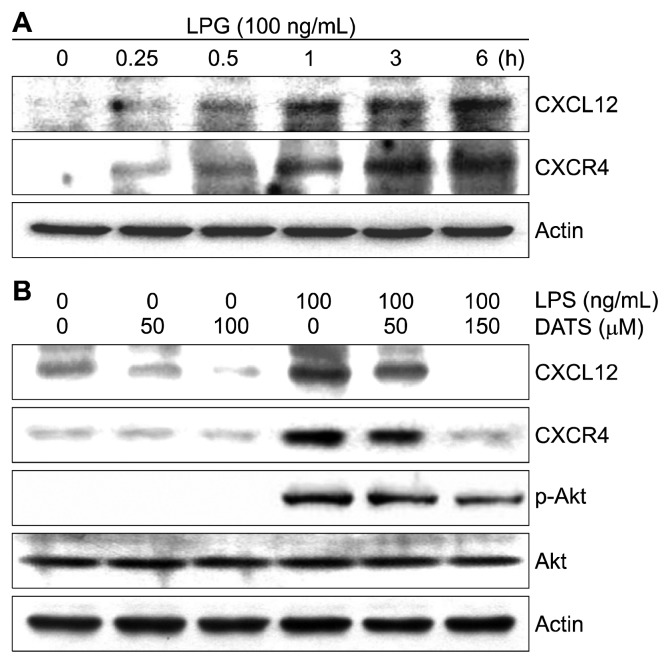
6. Inhibition of lipopolysaccharide-induced CXCL12 and CXCR4 expressions by diallyl trisulfide in BV2 microglial cells
We also investigated the effect of DATS on the CXCL12/CXCR4 axis to determine the association of chemokine networks with anti-inflammatory effects of DATS in BV cells. Our results showed that the expression of CXCL12 and CXCR4 proteins in the LSP-treated BV2 cells was continuously increased in a time- dependent manner (Fig. 6A), but this effect was suppressed in the presence of DATS depending on the DATS treatment concentration (Fig. 6B). Moreover, under the same condition, DATS pretreatment reduced the phosphorylation of LPS-induced Akt without affecting its total protein expression (Fig. 6B).
Figure 6.
Effects of diallyl trisulfide (DATS) on the lip-opolysaccharide (LPS)-induced expression of CXCL12/CXCR4 and phosphorylation of Akt in BV2 microglial cells. The cells were treated with 100 ng/mL LPS for the indicated times (A) or pre-treated with the indicated concentrations of DATS for 1 hour prior to LPS treatment for 6 hours (B). The total proteins were prepared for Western blot analysis using anti-CXCL12, anti-CXCR4, anti-p-Akt and anti-Akt antibodies and an enhanced chemiluminescence detection system. Actin was used as the internal control.
DISCUSSION
Although three enzymes, endothelial NOS, neuronal NOS, and iNOS, are involved in the synthesis of NO, iNOS expression is increased by inflammatory stimuli such as inflammatory and bacterial endotoxins and contributes to excessive NO production.16–19 Among COXs, which is involved in the production of PGE2, COX-1 is expressed in most tissues and serves to maintain normal physiological functions, whereas COX-2 is transiently induced in response to growth factors or inflammatory stimuli.20–22 Various types of inflammatory responses are directly associated with the production of abnormally high levels of NO and PGE2 by upregulation of iNOS and COX-2. Therefore, lowering the production of inflammatory mediators is important for the prevention of inflammatory diseases and the development of therapeutic agents. Similar to previous studies using a macrophage model,15,23 our results showed that DATS significantly inhibited NO and PGE2 production by LPS stimulation without cytotoxicity associated with suppression of iNOS and COX-2 expression, respectively.
NF-κB is a key factor in upregulating the expression of pro-inflammatory genes when it is translocated to the nucleus. NF-κB is generally complexed by IκBα and is located in the cytoplasm. When IκBα is phosphorylated and degraded, NF-κB is separated and transferred to the nucleus.6,7 Our results indicated that DATS effectively blocked the nuclear expression of NF-κB (p65) and the degradation and phosphrylation of IκBα. These results suggest that DATS reduced the expression and production of pro-inflammatory enzymes by inhibiting the NF-κB pathway in LPS-stimulated BV2 microglia. This is also in good agreement with previous results observed in LPS-stimulated macrophages and microglia.14,15,23
Immune cells, such as microglia, recognize pathogen- associated molecular patterns through the TLRs, which are expressed on the cell surface. Among the TLRs, when activated by LPS, TLR4 recruits adapter molecules containing MyD88, LPS-binding protein and a cluster of differentiation 14 (CD14) co-receptors.2,24 Upon activation of TLR4 by LPS, the TLR4- MyD88-mediated signaling can lead to activation of MAPKs, which ultimately promotes activation of NF-κB signaling and produces inflammatory mediators and cytokines.3,4 We have shown here that DATS had an obvious suppressive effect on LPS-stimulated expression of TLR4 and MyD88 in a concentration-dependent manner. This finding demonstrates that DATS inhibits the activation of NFκB and inhibits LPS binding to TLR4, thereby weakening the onset of the LPS- mediated intracellular signaling pathway. According to the results of You et al.,12 DATS significantly inhibited LPS-mediated MAPKs signaling in macrophage cell model. Our previous study have also showed that DATS significantly suppressed inhibits the production of pro-inflammatory mediators and cytokines in macrophages by attenuating the interaction between LPS and TLR4 in macrophages.15 Therefore, DATS may inhibit NF-κB as well as MAPK signaling pathways by showing an antagonistic effect that interferes with the binding of LPS to TLR4 in immune cells, such as macrophages and microglia.
CXCR4 is a multifunctional G-protein coupled receptor and is activated by the natural ligand CXCL12.8,9 In many previous studies on CXCR4 agonists or antagonists, CXCR4, as a member of the LPS-sensing complex, has been shown to modulate TLR4 signaling.24,25 For example, the CXCL12/CXCR4 axis is involved in the production of pro-inflammatory factors and exhibits a substantial chemo-attractive activity on various immune cells, and thus it acts as a cause of the initiation of various inflammatory diseases.26,27 It also plays an important role in the recruitment of neutrophils and macrophages at the early stage of inflammatory disease progression.9,28 However, the basic functions of CXCR4 and CXCL12 in inflammatory diseases remain controversial, and little is known about the precise effect of the CXCL12/CXCR4 axis blockade. Therefore, we further examined the effect of DATS on the expressions of CXCR4 and its ligand, CXCL12. Immunoblotting results indicated that the LPS challenge caused a time-dependent increase in CXCL12 and CXCR4 protein compared to the control. Conversely, in the presence of DATS, the expression of CXCL12 and CXCR4 proteins was significantly reduced, suggesting that the inactivation of the axis were at least involved in the anti-inflammatory effect of DATS in BV2 microglia. In addition, LPS induced prominent phosphorylation of Akt and pretreatment of DATS inhibited phosphorylation of Akt in a concentration-dependent manner, suggesting that down-regulation of the phospho-3-kinase (PI3K)/Akt-mediated signaling pathway is involved in the anti-inflammatory mechanism of DATS. Therefore, subsequent studies are needed to better understand the detailed mechanism underlying the DATS effect on the PI3K/Akt and CXCL12/CXCR4 pathways, including its effects on the migration of neutrophils and macrophages to the inflammation site.
In conclusion, the results obtained in this study demonstrated that DATS has a potent anti-inflammatory effect on BV2 microglia. In BV2 cells stimulated with LPS, DATS was able to reduce inflammation-related NO and PGE2 production associated with the inhibition of iNOS and COX-2 protein expression by inhibiting NF-κB activity. In addition, DATS can block early intracellular signaling cascades by suppressing the TLR4/Myd88 and/or CXCL12/CXCR4 signaling pathways. Although current results may provide a partial understanding of the mechanism of anti-inflammatory effects of DATS, further studies are needed to assess the mechanical role of DATS in a variety of inflammatory diseases.
ACKNOWLEDGMENTS
This study was supported by the Blue-Bio Industry Regional Innovation Center (RIC; grant no. RIC08-06-07) at Dong-Eui University as an RIC program under the Ministry of Trade, Industry and Energy and Busan city and Basic Science Research Program through the National Research Foundation of Korea (NRF) grant funded by the Korea government (2018R1A2B2005705).
Footnotes
CONFLICTS OF INTEREST
No potential conflicts of interest were disclosed.
REFERENCES
- 1.Tremblay MÈ, Stevens B, Sierra A, Wake H, Bessis A, Nimmerjahn A. The role of microglia in the healthy brain. J Neurosci 2011;31:16064–9. 10.1523/JNEUROSCI.4158-11.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gomez-Nicola D, Perry VH. Microglial dynamics and role in the healthy and diseased brain: a paradigm of functional plasticity. Neuroscientist 2015;21:169–84. 10.1177/1073858414530512 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH. Mechanisms underlying inflammation in neurodegeneration. Cell 2010;140: 918–34. 10.1016/j.cell.2010.02.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cherry JD, Olschowka JA, O'Banion MK. Neuroinflammation and M2 microglia: the good, the bad, and the inflamed. J Neuroinflammation 2014;11:98. 10.1186/1742-2094-11-98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rahimifard M, Maqbool F, Moeini-Nodeh S, Niaz K, Abdollahi M, Braidy N, et al. Targeting the TLR4 signaling pathway by poly-phenols: a novel therapeutic strategy for neuroinflammation. Ageing Res Rev 2017;36:11–9. 10.1016/j.arr.2017.02.004 [DOI] [PubMed] [Google Scholar]
- 6.Li Q, Verma IM. NF-kappaB regulation in the immune system. Nat Rev Immunol 2002;2:725–34. 10.1038/nri910 [DOI] [PubMed] [Google Scholar]
- 7.Kopitar-Jerala N. Innate immune response in brain, NF-kappa B signaling and cystatins. Front Mol Neurosci 2015;8:73. 10.3389/fnmol.2015.00073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yang L, Wang M, Guo YY, Sun T, Li YJ, Yang Q, et al. Systemic inflammation induces anxiety disorder through CXCL12/CXCR4 pathway. Brain Behav Immun 2016;56:352–62. 10.1016/j.bbi.2016.03.001 [DOI] [PubMed] [Google Scholar]
- 9.Yaxin W, Shanglong Y, Huaqing S, Hong L, Shiying Y, Xiangdong C, et al. Resolvin D1 attenuates lipopolysaccharide induced acute lung injury through CXCL-12/CXCR4 pathway. J Surg Res 2014; 188:213–21. 10.1016/j.jss.2013.11.1107 [DOI] [PubMed] [Google Scholar]
- 10.Wu CC, Sheen LY, Chen HW, Kuo WW, Tsai SJ, Lii CK. Differential effects of garlic oil and its three major organosulfur components on the hepatic detoxification system in rats. J Agric Food Chem 2002;50:378–83. 10.1021/jf010937z [DOI] [PubMed] [Google Scholar]
- 11.Wang HC, Hsieh SC, Yang JH, Lin SY, Sheen LY. Diallyl trisulfide induces apoptosis of human basal cell carcinoma cells via endoplasmic reticulum stress and the mitochondrial pathway. Nutr Cancer 2012;64:770–80. 10.1080/01635581.2012.676142 [DOI] [PubMed] [Google Scholar]
- 12.You S, Nakanishi E, Kuwata H, Chen J, Nakasone Y, He X, et al. Inhibitory effects and molecular mechanisms of garlic organo-sulfur compounds on the production of inflammatory mediators. Mol Nutr Food Res 2013;57:2049–60. 10.1002/mnfr.201200843 [DOI] [PubMed] [Google Scholar]
- 13.Wang W, Sun X, Zhang H, Yang C, Liu Y, Yang W, et al. Controlled release hydrogen sulfide delivery system based on mesoporous silica nanoparticles protects graft endothelium from ischemia-re-perfusion injury. Int J Nanomedicine 2016;11:3255–63. 10.2147/IJN.S104604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ho SC, Su MS. Evaluating the anti-neuroinflammatory capacity of raw and steamed garlic as well as five organosulfur compounds. Molecules 2014;19:17697–714. 10.3390/molecules191117697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee HH, Han MH, Hwang HJ, Kim GY, Moon SK, Hyun JW, et al. Diallyl trisulfide exerts anti-inflammatory effects in lipopoly-saccharide-stimulated RAW 264.7 macrophages by suppressing the Toll-like receptor 4/nuclear factor-κB pathway. Int J Mol Med 2015;35:487–95. 10.3892/ijmm.2014.2036 [DOI] [PubMed] [Google Scholar]
- 16.Kang HJ, Jeong JS, Park NJ, Go GB, Kim SO, Park C, et al. An ethanol extract of Aster yomena (Kitam.) honda inhibits lipopolysaccharide-induced inflammatory responses in murine RAW 264.7 macrophages. Biosci Trends 2017;11:85–94. 10.5582/bst.2016.01217 [DOI] [PubMed] [Google Scholar]
- 17.Joubert J, Malan SF. Novel nitric oxide synthase inhibitors: a patent review. Expert Opin Ther Pat 2011;21:537–60. 10.1517/13543776.2011.556619 [DOI] [PubMed] [Google Scholar]
- 18.Kanwar JR, Kanwar RK, Burrow H, Baratchi S. Recent advances on the roles of NO in cancer and chronic inflammatory disorders. Curr Med Chem 2009;16:2373–94. 10.2174/092986709788682155 [DOI] [PubMed] [Google Scholar]
- 19.Salerno L, Sorrenti V, Di Giacomo C, Romeo G, Siracusa MA. Progress in the development of selective nitric oxide synthase (NOS) inhibitors. Curr Pharm Des 2002;8:177–200. 10.2174/1381612023396375 [DOI] [PubMed] [Google Scholar]
- 20.Minghetti L, Pocchiari M. Cyclooxygenase-2, prostaglandin E2, and microglial activation in prion diseases. Int Rev Neurobiol 2007;82:265–75. 10.1016/S0074-7742(07)82014-9 [DOI] [PubMed] [Google Scholar]
- 21.Murakami M, Kudo I. Prostaglandin E synthase: a novel drug target for inflammation and cancer. Curr Pharm Des 2006;12: 943–54. 10.2174/138161206776055912 [DOI] [PubMed] [Google Scholar]
- 22.Kudo I, Murakami M. Prostaglandin E synthase, a terminal enzyme for prostaglandin E2 biosynthesis. J Biochem Mol Biol 2005;38:633–8. [DOI] [PubMed] [Google Scholar]
- 23.Liu KL, Chen HW, Wang RY, Lei YP, Sheen LY, Lii CK. DATS reduces LPS-induced iNOS expression, NO production, oxidative stress, and NF-kappaB activation in raw 264.7 macrophages. J Agric Food Chem 2006;54:3472–8. 10.1021/jf060043k [DOI] [PubMed] [Google Scholar]
- 24.Triantafilou M, Lepper PM, Briault CD, Ahmed MA, Dmochowski JM, Schumann C, et al. Chemokine receptor 4 (CXCR4) is part of the lipopolysaccharide “sensing apparatus”. Eur J Immunol 2008; 38:192–203. 10.1002/eji.200636821 [DOI] [PubMed] [Google Scholar]
- 25.Mithal DS, Banisadr G, Miller RJ. CXCL12 signaling in the development of the nervous system. J Neuroimmune Pharmacol 2012;7:820–34. 10.1007/s11481-011-9336-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lu DY, Tang CH, Yeh WL, Wong KL, Lin CP, Chen YH, et al. SDF-1alpha up-regulates interleukin-6 through CXCR4, PI3K/Akt, ERK, and NF-kappaB-dependent pathway in microglia. Eur J Pharmacol 2009;613:146–54. 10.1016/j.ejphar.2009.03.001 [DOI] [PubMed] [Google Scholar]
- 27.Chen HT, Tsou HK, Hsu CJ, Tsai CH, Kao CH, Fong YC, et al. Stromal cell-derived factor-1/CXCR4 promotes IL-6 production in human synovial fibroblasts. J Cell Biochem 2011;112:1219–27. 10.1002/jcb.23043 [DOI] [PubMed] [Google Scholar]
- 28.Peng Y, Zhong Y, Li G. Tubeimoside-1 suppresses breast cancer metastasis through downregulation of CXCR4 chemokine receptor expression. BMB Rep 2016;49:502–7. 10.5483/BMBRep.2016.49.9.030 [DOI] [PMC free article] [PubMed] [Google Scholar]