Abstract
Backgrounds:
Mesenteric tumor deposits are a frequent occurrence in small intestinal neuroendocrine tumors. In many instances, these mesenteric tumor deposits are surrounded by a dense fibrotic stroma and have associated lymphoplasmacytic inflammation. These histologic features seen around mesenteric tumor deposits can be a histologic mimic of IgG4-related mesenteritis, especially when tumor cells are not sampled.
Materials and Methods:
Sixty-six mesenteric tumor deposits from 66 patients with small intestinal neuroendocrine tumor were identified from our archives with blocks available for further studies. Cases were assessed for clinicopathologic features and presence of IgG4-positive and IgG-positive plasma cells by immunohistochemistry. Ratios of IgG4-positive to IgG-positive cells were calculated.
Results:
Seventeen mesenteric tumor deposits (26%) showed greater than 40 IgG4-positive plasma cells per high-power field, and the majority of cases (68%) showed at least some staining of IgG4-positive plasma cells. Mesenteric tumor deposits with >20 IgG4-positve plasma cells per high power field tended to be larger (2.59±1.30 cm versus 1.86±1.58 cm, P=0.07) and had more IgG-positive cells (88±24 versus 36±37; P<0.01) and a higher ratio of IgG4-positive to IgG-positive cells (0.66±0.18 versus 0.17±0.25; P<0.01). All but one mesenteric tumor deposits with >20 IgG4-positve plasma cells had a ratio of >40%.
Conclusions:
IgG4 expression is frequent in mesenteric tumor deposits from small intestinal neuroendocrine tumors. Undersampling of tumor on biopsies of mesenteric tumor deposits could potentially cause diagnostic confusion with IgG4-related sclerosing mesenteritis.
Keywords: small intestinal neuroendocrine tumor, mesenteric tumor deposit, IgG4, sclerosing mesenteritis, plasma cells
INTRODUCTION
Small intestinal neuroendocrine tumors (NETs) are the most commonly encountered malignant neoplasms of the small intestine, with an incidence of 5 per 100,000.1, 2 They are frequently associated with lymph node metastasis and mesenteric tumor deposits (MTDs), both of which alter the overall stage in the 8th edition of the American Joint Committee on Cancer (AJCC) staging system for small intestinal NETs.3–5 MTDs are defined as discrete but irregular mesenteric tumor nodules discontinuous from the primary neoplasm; they are associated with frequent liver metastasis and a poor prognosis.5, 6 In addition, MTDs are frequently associated with dense fibrosis, which can lead to entrapment of adjacent large mesenteric vessels and nerves. Because of this dense fibrotic response to tumor, MTDs can radiographically simulate IgG4-related sclerosing mesenteritis, complicating diagnosis by imaging and necessitating tissue diagnosis.7
IgG4-related sclerosing mesenteritis is characterized by fibrosis, lymphoplasmacytic inflammation, and obliterative thrombophlebitis with prominent IgG4-positive plasma cells.8 Microscopically, the sclerosing fibrotic stromal response associated with MTDs is often accompanied by similar adjacent foci of lymphoplasmacytic inflammation. If these features are present on core biopsy when tumor cells are not sampled, it can histologically mimic sclerosing mesenteritis. This study examined whether MTDs in patients with small intestinal NETs showed histologic and immunophenotypic overlap with IgG4-related sclerosing mesenteritis.
MATERIALS AND METHODS
Seventy patients with MTD(s) arising in small intestinal NET were identified from our pathology archives between 01/01/1996 and 07/01/2017, 66 of which had at least one MTD block available for immunohistochemical (IHC) staining. MTDs were defined as discrete tumor nodules within mesenteric soft tissues with an irregular growth pattern.5, 6 Lesions resulting from direct contiguous spread by the primary lesion or extranodal extension were excluded. Patient electronic medical records were reviewed for demographic information, including age, sex, and clinical history. Pathology reports and slides of the 66 cases were reviewed, and AJCC tumor stage, tumor grade and MTD size were all recorded. This retrospective study was approved by Vanderbilt Institutional Review Board (IRB#: 101735; date: 5/1/2014 to 9/13/2018).
For IHC labeling, 4μm unstained tumor sections from formalin-fixed paraffin-embedded tissue were deparaffinized by routine methods. Antigen retrieval was performed as follows: Slides were heated in a pH 9.0 EDTA buffer at 97°C for 20 minutes in a pressure cooker, followed by a 10-minute cool down to room temperature. All slides were then quenched with 0.02% H2O2 with sodium azide for 5 minutes, and blocked for 20 minutes with serum-free protein block (Dako, Carpinteria, CA), followed by incubation with primary antibody IgG4 (CAT# 05–3800 Mouse Monoclonal; Zymed; Waltham, MA) or IgG (CAT# CMC006 Rabbit Polyclonal; Cell Marque; Rocklin, CA) at 1:500 dilution for 45 minutes at room temperature. Slides were then incubated with Evision+HRP-labeled polymer (DaKo) for 30 minutes. Staining was visualized by 5-minute incubation with diaminobenzidine.
IHC labeling for IgG4 and IgG was performed on one MTD from each of the 66 cases; for cases with multiple MTDs, the staining was performed on the largest. IgG-positive and IgG4-positive plasma cells (PCs) were quantified by finding the highest concentration of positive cells in one high-power field (HPF) in areas of sclerotic stroma adjacent to tumor. The ratio of IgG4-positive to IgG-positive plasma cells was obtained by dividing the number of IgG4-positive cells by the number of IgG-positive cells at one selected HPF. Cases were divided into 2 groups based on the number of IgG4-positive plasma cells per HPF: Group 1 had 0–20 per HPF, and Group 2 had >20 per HPF. Ki67 index was available for all cases and was used to grade each primary tumor based on 2010 World Health Organization (WHO) criteria.9
The data were analyzed using GraphPad Prism Version 5.01 (GraphPad Software, Inc., La Jolla, CA). Fisher’s exact test and Student’s t-test were used to compare clinicopathologic variables and IHC results between two groups. P < 0.05 was considered statistically significant.
RESULTS
Of the 66 patients, there were 34 males and 32 females, with a mean age of 59 years (range: 19–83). Of the primary small intestinal NETs, fifty-one were WHO grade 1 and fifteen were WHO grade 2 (Table 1). Twenty-seven cases presented with AJCC stage II-III tumor (one stage II and twenty-six stage III), and 39 with stage IV tumor.
Table 1:
Clinicopathologic features of mesenteric tumor deposit cases stratified by levels of IgG4-positive plasma cells
| ≤20/HPF (N=46) | >20/HPF (N=20) | All cases (N=66) | P value | ||
|---|---|---|---|---|---|
| Sex | male | 24 | 10 | 34 | 1.00 |
| female | 22 | 10 | 32 | ||
| Age (mean ± SD) | 59±2 | 61±2 | 60±1 | 0.65 | |
| WHO grade | 1 | 37 | 14 | 51 | 0.36 |
| 2 | 9 | 6 | 15 | ||
| AJCC stage | II-III | 16 | 11 | 27 | 0.17 |
| IV | 30 | 9 | 39 | ||
| MTD size (mean ± SD; cm) | 1.86±1.58 | 2.59±1.30 | 2.08±1.53 | 0.07 | |
| IgG+ cells (mean ± SD; cm)* | 36±37 | 88±24 | 51±41 | <0.01 | |
| IgG+/IgG4+ (mean ± SD; cm)* | 0.17±0.25 | 0.66±0.18 | 0.32±0.32 | <0.01 | |
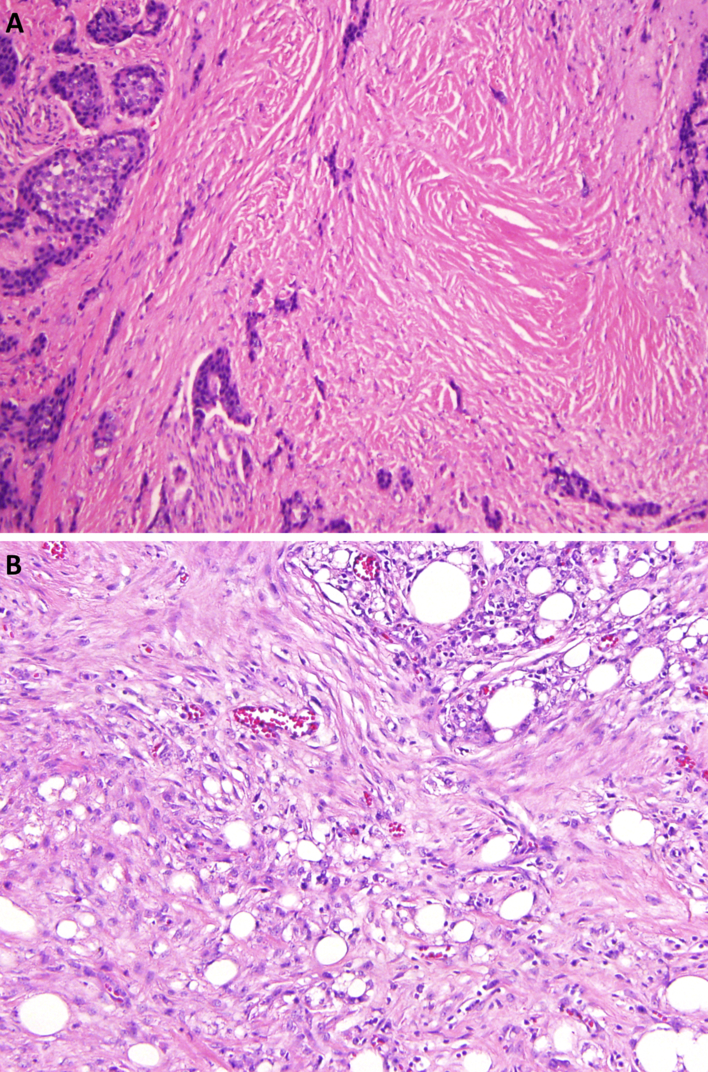
Most patients had one MTD, whereas some (26/66, 40%) had more than one. The median MTD size was 1.8 cm, ranging from 0.2 cm to 7.0 cm. All 66 MTDs showed fibrotic stroma, which was often more prominent at the periphery of the MTDs and was frequently associated with lymphoplasmacytic inflammation (Figure 1A). Significant lymphoplasmacytic inflammation, defined as foci of inflammation easily identified at low power, was seen in 46 (70%) MTDs (Figure 1B). No obliterative thrombophlebitis was seen in any of the cases. The fibrosis associated with MTDs was hypocellular and frequently associated with prominent hyalinization (Figure 2A). Foci of calcification often developed in large hyalinized areas. At the periphery, focal fibrosis in a subtle storiform pattern (Figure 2B) was observed in 18 of 66 (27%) cases, which was frequently associated with lymphoplasmacytic inflammation and/or fat necrosis.
Figure 1.
A representative mesenteric tumor deposit (MTD) from a patient with small intestinal neuroendocrine tumor. A. Portion of a MTD with an irregular contour and dense fibrosis at the periphery (original magnification 20X); B. Lymphoplasmacytic inflammation at the periphery (original magnification 100X); C. Expression of IgG4 by plasma cells surrounding the MTD (original magnification 100X); D. Expression of IgG by plasma cells surrounding the MTD (original magnification 100X).
Figure 2.
Fibrosis in a representative mesenteric tumor deposit (MTD). B. Fibrosis among tumor nests/sheets showing hyalinization (original magnification 100X); B. MTD periphery with focal fibrosis in a subtle storiform pattern associated with lymphoplasmacytic inflammation (original magnification 100X).
Twenty of 66 MTDs (30%) showed greater than 20 IgG4-positive plasma cells per HPF (Group 2), including seventeen (26%) with >40 (Figure 1C) and 3 (4%) with 21–40 per HPF. Forty-six (70%) MTDs had 20 or fewer IgG4-positive plasma cells per HPF (Group 1); among them, 21 (32%) had no IgG4-positive cells, and 25 (38%) had 1 to 20 per HPF. Overall, more than two thirds of MTDs showed at least some staining of IgG4-positive plasma cells.
There were no statistical differences in age, sex, WHO tumor grade, MTD size or AJCC tumor stage between group 1 and group 2 (Table 1). However, MTDs with >20 IgG4-positive plasma cells per HPF tended to be larger compared to those with ≤20 IgG4-positive plasma cells per HPF (P=0.07). In addition, MTDs with >20 IgG4-positive plasma cells per HPF were more often AJCC stage IV, though this was not statistically significant (P=0.17).
IgG staining performed on all MTDs (Figure 1D) showed more IgG-positive plasma cells in Group 2 (with >20 IgG4-positive cells per HPF) compared to Group 1 (P<0.01; Table 1). IgG-positive cells ranged from 0 to 78 per HPF in group 1 and from 60 to 132 per HPF in group 2. The IgG4-positive/IgG-positive plasma cells ratio was also higher in group 2 (P<0.001; Table 1). Ten of 46 (22%) MTDs in group 1 had a ratio of >0.2, whereas all MTDs in group 2 had a ratio of >0.2, and all but one had a ratio of >0.4.
DISCUSSION
MTDs are a frequent occurrence in small intestinal NETs. They are discrete mesenteric tumor nodules with irregular contours. Histologically, most MTDs are associated with extensive fibrosis constricting entrapped large mesenteric vessels and consequently resulting in small bowel ischemia or infarct. We have shown that up to 60% of small intestinal NETs present with MTDs.5 Furthermore, their presence is associated with several poor prognostic factors, including high T-category stage at presentation, increased propensity for lymphovascular invasion, and lymph node and liver metastasis. When present, they are usually larger than primary tumors. Sometimes MTDs may be the only finding on imaging studies, and distinction between other processes such as lymphoma and other mesenteric diseases can be difficult by imaging alone.7,10
IgG4-related disease is a fibroinflammatory tumefactive process characterized by a dense lymphoplasmacytic infiltrate enriched with IgG4-positive plasma cells, storiform fibrosis, and frequent elevations of serum IgG4.11 It first came to be recognized in patients with autoimmune pancreatitis who manifested with extrapancreatic symptoms.12 Since then, IgG4-related disease has been described in many different organ systems, and the diagnostic criteria have been better defined.13,14 The three major histopathologic features noted are dense lymphoplasmacytic inflammation, fibrosis (at least focally storiform), and obliterative thrombophlebitis. The diagnosis can be confidently made when two of these three features are present. IgG and IgG4 immunohistochemical stains can also assist in diagnosis by allowing quantification of the number of IgG4-positive plasma cells per HPF and the overall ratio of IgG4-positive to IgG-positive plasma cells. It has been suggested that greater than 30 IgG4-positive plasma cells per HPF and/or a ratio of 40% of IgG4-positive/IgG-positive plasma cells be used to assist in diagnosis; however, these numbers were mainly generated from one or a small number of studies per site.13,14 Therefore, minimum IgG4 plasma counts for the diagnosis at different sites are still questionable at present. The diagnosis can be difficult in some instances, and IgG4-related disease has been known to mimic a number of other diagnoses, including granulomatosis with polyangiitis and Rosai-Dorfman disease.15
MTDs seen in small intestinal NETs can be an impressive mimic of IgG4-related sclerosing mesenteritis clinically and radiographically. Furthermore, the adjacent fibrosis and lymphoplasmacytic inflammation can cause histologic confusion in the absence of sampled tumor cells. Our study demonstrates that, in addition to these histologic findings, significant numbers of IgG4-positive plasma cells can be associated with these areas of dense sclerotic fibrosis adjacent to MTDs. The majority of cases (68%) showed at least some IgG4-positive plasma cells, and a significant portion (30%) showed more than 20 IgG4-positive plasma cells per HPF. Additionally, all but one of the 20 cases with >20 IgG4-positive plasma cells per HPF showed a ratio of IgG4-positive to IgG-positive plasma cells greater than 40%. In the 25 MTDs containing 1 to 20 IgG4-positive plasma cells per HPF, 7 (28%) had a ratio of IgG4-positive to IgG-positive plasma cells greater than 40%.
Some clinical and radiographic features may help distinguish MTD from sclerosing mesenteritis. Approximately one third of patients with sclerosing mesenteritis have a history of abdominal surgery or abdominal trauma.16, 17 Patients with sclerosing mesenteritis can be asymptomatic, but common signs/symptoms include abdominal pain, palpable abdominal mass, fever, weight loss, and small bowel obstruction. Signs and symptoms of small bowel obstruction/ischemia may also occur in small intestinal NET patients when large MTDs cause kinking of the bowel and encasing large mesenteric vessels. Additionally, patients with MTDs frequently have liver metastasis,5,6 and they can develop carcinoid syndrome when there are extensive liver metastases. Radiographically, both entities appear as a soft-tissue mesenteric mass with radiating fibrotic bands in a stellate pattern; however, MTDs tend to encase and invade mesenteric vessels, whereas sclerosing mesenteritis often spares mesenteric fat immediately surrounding vessels, producing a “fat ring sign”.17 The presence of one or more primary tumors in the small intestine and hypervascular liver masses favor MTDs arising in small intestinal NET.
In summary, MTDs in small intestinal NETs can have extensive areas of fibrosis and chronic inflammation surrounding tumor, which can cause undersampling of the tumor on core biopsy. In addition, IgG4 expression is frequent in MTDs. It is important to be aware of this potential pitfall when facing a biopsy of a mesenteric mass.
Acknowledgement:
This work was partially supported by NIH/NIDDK 5P30 DK058404-13 (CS); NIH/NCI 5P50 CA095103-13 (CS)
Footnotes
Author contributions:
JR reviewed and analyzed immunohistochemical stains and wrote the manuscript
RSG identified the cases and edited the manuscript
FR validated and performed immunohistochemistry
CS designed the study, reviewed and analyzed immunohistochemical stains, and edited the manuscript
Conflict of Interest: None for all authors
REFERENCES
- 1.Bilimoria KY, Bentrem DJ, Wayne JD, Ko CY, Bennett CL, Talamonti MS. Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg 2009; 249: 63–71. [DOI] [PubMed] [Google Scholar]
- 2.Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer 2003; 97: 934–959. [DOI] [PubMed] [Google Scholar]
- 3.Woltering EA, Beyer DT, O’Dorisio TM, et al. In: Amin MB, Edge SB, Greene FL. et al. eds. AJCC Cancer Staging Manual. New York, NY: Springer; 2017. [Google Scholar]
- 4.Rorstad O Prognostic indicators for carcinoid neuroendocrine tumors of the gastrointestinal tract. J Surg Oncol 2005; 89: 151–160. [DOI] [PubMed] [Google Scholar]
- 5.Gonzalez RS, Liu EH, Alvarez JR, Ayers GD, Washington MK, Shi C. Should mesenteric tumor deposits be included in staging of well-differentiated small intestine neuroendocrine tumors? Mod Pathol 2014; 27:1288–1295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fata CR, Gonzalez RS, Liu E, Cates JM, Shi C. Mesenteric Tumor Deposits in Midgut Small Intestinal Neuroendocrine Tumors Are a Stronger Indicator Than Lymph Node Metastasis for Liver Metastasis and Poor Prognosis. Am J Surg Pathol 2017; 41: 128–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Martinez-de-Alegria A, Baleato-Gonzalez S, Garcia-Figueiras R, et al. IgG4-related Disease from Head to Toe. Radiographics 2015; 35: 2007–2025. [DOI] [PubMed] [Google Scholar]
- 8.Lee SJ, Park CK, Yang WI, Kim SK: IgG4-Related Sclerosing Mesenteritis. J Pathol Transl Med 2016; 50: 309–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bosman FT CF, Hruban RH, et al. WHO Classification of Tumours of the Digestive System, 4 edn: Lyon: International Agency for Research on Cancer (IARC); 2010. [Google Scholar]
- 10.Sheth S, Horton KM, Garland MR, Fishman EK. Mesenteric neoplasms: CT appearances of primary and secondary tumors and differential diagnosis. Radiographics 2003; 23: 457–473. [DOI] [PubMed] [Google Scholar]
- 11.Stone JH, Zen Y, Deshpande V. IgG4-related disease. N Engl J Med 2012; 366: 539–551. [DOI] [PubMed] [Google Scholar]
- 12.Kamisawa T, Funata N, Hayashi Y, et al. A new clinicopathological entity of IgG4-related autoimmune disease. J Gastroenterol 2003; 38: 982–984. [DOI] [PubMed] [Google Scholar]
- 13.Deshpande V, Zane NA, Kraft S, Stone JH, Faquin WC. Recurrent Mastoiditis Mimics IgG4 Related Disease: A Potential Diagnostic Pitfall. Head Neck Pathol 2016; 10: 314–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Deshpande V, Zen Y, Chan JK, et al. Consensus statement on the pathology of IgG4-related disease. Mod Pathol 2012; 25: 1181–1192. [DOI] [PubMed] [Google Scholar]
- 15.Ferry JA, Klepeis V, Sohani AR, et al. IgG4-related Orbital Disease and Its Mimics in a Western Population. Am J Surg Pathol 2015; 39:1688–1700. [DOI] [PubMed] [Google Scholar]
- 16.Sharma P, Yadav S, Needham CM, Feuerstadt P. Scleoring mesenteritis: a systematic review of 192 cases. Clin J Gastroenterol 2017; 10:103–111. [DOI] [PubMed] [Google Scholar]
- 17.Winant AJ, Vora A, Ginter PS, Levine MS, Brylka DA. More than just metastases: a practical approach to solid mesenteric masses. Abdom Imaging 2014: 39: 605–621. [DOI] [PubMed] [Google Scholar]