Abstract
Burkholderia lata was isolated from 8 intensive care patients at 2 tertiary hospitals in Australia. Whole-genome sequencing demonstrated that clinical and environmental isolates originated from a batch of contaminated commercial chlorhexidine mouthwash. Genomic analysis identified efflux pump–encoding genes as potential facilitators of bacterial persistence within this biocide.
Keywords: Burkholderia lata, chlorhexidine mouthwash, intensive care unit, genome analysis, bacteria, nosocomial infections, Australia
Burkholderia contaminans and B. lata together form group K of the B. cepacia complex (Bcc). These predominantly environmental species are a major cause of pharmaceutical contamination and have been linked to multiple instances of associated opportunistic infection (1). Although both species are capable of causing serious infections in humans (2,3), only B. contaminans has been associated with infection outbreaks (3,4). We report a healthcare-associated B. lata infection outbreak occurring in intensive care units (ICUs) in 2 tertiary hospitals in Australia.
During May–June 2016, bacterial contamination of chlorhexidine mouthwash (0.2% mg/mL) was associated with an interjurisdictional outbreak in New South Wales and South Australia. Bcc isolates were obtained from blood and tracheal aspirates from 6 ICU patients in hospital A (South Australia) (sample information and isolation protocols detailed in the Technical Appendix). An investigation by the hospital’s infection and prevention control team noted discoloration of a commercial chlorhexidine mouthwash. Bcc isolates were cultured from all 5 tested bottles from the discolored batch but not from 11 bottles tested from 4 other batches. Isolates were further obtained from the surfaces of hand basins in 3 separate ICU rooms, where bottles of mouthwash from the contaminated batch were in use. Separately, a Bcc isolate was obtained from the sputum of an ICU patient in hospital B (New South Wales), where the same batch of mouthwash was in use. After a nationwide recall of the contaminated mouthwash batch, no further cases were reported.
The genomes of 15 Bcc isolates from the 2 hospitals were sequenced (Technical Appendix). A closed genome of isolate A05 was generated as a reference, consisting of 3 circular chromosomes of 3.93, 3.71, and 1.15 Mbp (National Center for Biotechnology Information BioProject accession no. PRJNA419071).
Genome analysis identified an Australasian sublineage of B. lata as the cause of the outbreak. Specifically, all isolates were sequence type 103 (ST103), which sits within a subclade of B. lata isolates from Australia and New Zealand (Technical Appendix Figure 1). Two isolates from hand basins (A07 and A08) and 1 from a hospital bench (A10) were of unknown sequence type.
Single-nucleotide polymorphism (SNP)–based phylogenetic analysis identified the outbreak strain as a distinct clonal lineage (0–1 SNPs) within the group K clade (Figure), separated from other members of the group by a minimum of 167,662 SNPs. SNP variation within the clonal sublineage ranged from 0 to 2 SNPs across isolates from contaminated mouthwash bottles and patients. A single SNP distinguished isolates from patient 4 in room 21 (A04) and the hand basin in the same room (A08) (collected 2 days apart). Three of the taxa isolated from the hospital environment (A07, A08, and A10) had substantial SNP variations compared with contaminated mouthwash and patient isolates (an average of 191,893 SNPs for A07, 655 SNPs for A08, and 1,408 SNPs for A10).
Figure.
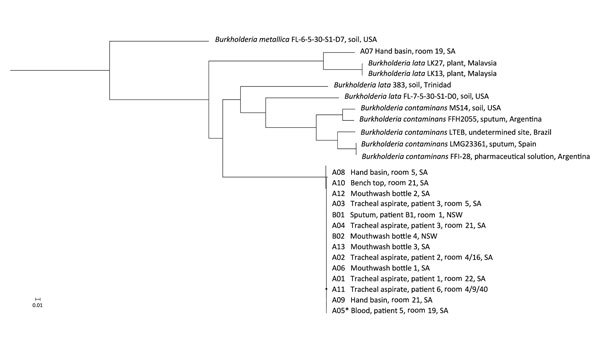
Phylogenetic analysis of isolates implicated in an outbreak Burkholderia lata infection from intrinsically contaminated chlorhexidine mouthwash, Australia, 2016. The maximum-likelihood tree is constructed from core genome single-nucleotide polymorphism alignments (N = 512,480) of the outbreak genomes, bootstrapped 1,000 times, and archival genomes from B. cepacia complex group K, relative to the reference genome B. lata A05 (identified by an asterisk). B. metallica was included as a comparator.
Differences of 0–1 SNPs between isolates from mouthwash in the 2 hospitals, including from unopened bottles, strongly suggests contamination during the manufacturing process. After several previous incidents of contamination of cosmetic, toiletry, and pharmaceutical products by members of Bcc, the US Food and Drug Administration commented that these bacteria very likely are introduced into the manufacturing processes through contaminated water (https://www.fda.gov/Drugs/DrugSafety/ucm559508.htm). Direct patient-to-patient transmission of B. lata would appear unlikely in this instance, given the use of individual ICU rooms with nonshared facilities and incomplete overlap in periods of care.
Chlorhexidine mouthwash is perhaps a surprising source of B. lata infection, given its potent biocidal properties. However, Bcc species have been isolated previously from 0.02% chlorhexidine gluconate (in irrigation apparatuses used for vaginal douching) and from 0.05% preparations (used for storage of suction catheters) (5). The ability of B. lata to remain viable in chlorhexidine appears to be a result of a combination of efflux pump activity, biofilm formation, and cell wall impermeability (1,6,7). These mechanisms probably also contribute to the common inefficacy of combination antibiotic therapy in the treatment of associated infections (8). Prolonged exposure to microbiocides, including chlorhexidine, has been shown to result in a stable increase in the expression of antibiotic-resistance mechanisms (1,6), and elevated chlorhexidine resistance has been reported in multidrug-resistant strains of B. cenocepacia from cystic fibrosis patients (9). Three resistance-nodulation-division (RND) efflux pump genes (RND3, RND4, and RND9) have been shown to be essential for chlorhexidine tolerance in B. cenocepacia (9). Examination of the complete genome of B. lata isolate A05 revealed the presence of RND3, RND4, and RND9 in each strain (>94% sequence identity) (Technical Appendix Figure 2).
B. contaminans is the cause of widespread pharmaceutical product contamination, and infection outbreaks by this species are well-documented (3,10). Our findings suggest that the other member of Bcc group K, B. lata, also represents an important opportunistic pathogen of relevance to infection control, particularly given its intrinsic biocide tolerance.
Materials and methods for investigation of Burkholderia lata infections from intrinsically contaminated chlorhexidine mouthwash, Australia, 2016.
Acknowledgments
The authors gratefully acknowledge Deborah Williamson and Mark Schultz for their helpful discussions, Trang Nguyen for providing the New South Wales isolates, and microbiological laboratory staff from South Australia Pathology, Pathology North of New South Wales, and Pathology West of New South Wales for their technical input.
Biography
Dr. Leong is a research scientist at the South Australian Health and Medical Research Institute. His research interests include pathogen genomic epidemiology and infection outbreaks.
Footnotes
Suggested citation for this article: Leong LEX, Lagana D, Carter GP, Wang Q, Smith K, Stinear TP, et al. Burkholderia lata infections from intrinsically contaminated chlorhexidine mouthwash, Australia, 2016. Emerg Infect Dis. 2018 Nov [date cited]. https://doi.org/10.3201/eid2411.171929
References
- 1.Rushton L, Sass A, Baldwin A, Dowson CG, Donoghue D, Mahenthiralingam E. Key role for efflux in the preservative susceptibility and adaptive resistance of Burkholderia cepacia complex bacteria. Antimicrob Agents Chemother. 2013;57:2972–80. 10.1128/AAC.00140-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Pope CE, Short P, Carter PE. Species distribution of Burkholderia cepacia complex isolates in cystic fibrosis and non-cystic fibrosis patients in New Zealand. J Cyst Fibros. 2010;9:442–6. 10.1016/j.jcf.2010.08.011 [DOI] [PubMed] [Google Scholar]
- 3.Medina-Pascual MJ, Valdezate S, Carrasco G, Villalón P, Garrido N, Saéz-Nieto JA. Increase in isolation of Burkholderia contaminans from Spanish patients with cystic fibrosis. Clin Microbiol Infect. 2015;21:150–6. 10.1016/j.cmi.2014.07.014 [DOI] [PubMed] [Google Scholar]
- 4.Martina P, Bettiol M, Vescina C, Montanaro P, Mannino MC, Prieto CI, et al. Genetic diversity of Burkholderia contaminans isolates from cystic fibrosis patients in Argentina. J Clin Microbiol. 2013;51:339–44. 10.1128/JCM.02500-12 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Oie S, Kamiya A. Microbial contamination of antiseptics and disinfectants. Am J Infect Control. 1996;24:389–95. 10.1016/S0196-6553(96)90027-9 [DOI] [PubMed] [Google Scholar]
- 6.Knapp L, Rushton L, Stapleton H, Sass A, Stewart S, Amezquita A, et al. The effect of cationic microbicide exposure against Burkholderia cepacia complex (Bcc); the use of Burkholderia lata strain 383 as a model bacterium. J Appl Microbiol. 2013;115:1117–26. 10.1111/jam.12320 [DOI] [PubMed] [Google Scholar]
- 7.Drevinek P, Holden MT, Ge Z, Jones AM, Ketchell I, Gill RT, et al. Gene expression changes linked to antimicrobial resistance, oxidative stress, iron depletion and retained motility are observed when Burkholderia cenocepacia grows in cystic fibrosis sputum. BMC Infect Dis. 2008;8:121. 10.1186/1471-2334-8-121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.George AM, Jones PM, Middleton PG. Cystic fibrosis infections: treatment strategies and prospects. FEMS Microbiol Lett. 2009;300:153–64. 10.1111/j.1574-6968.2009.01704.x [DOI] [PubMed] [Google Scholar]
- 9.Coenye T, Van Acker H, Peeters E, Sass A, Buroni S, Riccardi G, et al. Molecular mechanisms of chlorhexidine tolerance in Burkholderia cenocepacia biofilms. Antimicrob Agents Chemother. 2011;55:1912–9. 10.1128/AAC.01571-10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Martin M, Christiansen B, Caspari G, Hogardt M, von Thomsen AJ, Ott E, et al. Hospital-wide outbreak of Burkholderia contaminans caused by prefabricated moist washcloths. J Hosp Infect. 2011;77:267–70. 10.1016/j.jhin.2010.10.004 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Materials and methods for investigation of Burkholderia lata infections from intrinsically contaminated chlorhexidine mouthwash, Australia, 2016.


