Abstract
The response to respiratory viruses varies substantially between individuals, and there are currently no known molecular predictors from the early stages of infection. Here we conduct a community-based analysis to determine whether pre- or early post-exposure molecular factors could predict physiologic responses to viral exposure. Using peripheral blood gene expression profiles collected from healthy subjects prior to exposure to one of four respiratory viruses (H1N1, H3N2, Rhinovirus, and RSV), as well as up to 24 h following exposure, we find that it is possible to construct models predictive of symptomatic response using profiles even prior to viral exposure. Analysis of predictive gene features reveal little overlap among models; however, in aggregate, these genes are enriched for common pathways. Heme metabolism, the most significantly enriched pathway, is associated with a higher risk of developing symptoms following viral exposure. This study demonstrates that pre-exposure molecular predictors can be identified and improves our understanding of the mechanisms of response to respiratory viruses.
The response to respiratory virus exposure can currently not be predicted by pre- or early post-exposure molecular signatures. Here, the authors conduct a community-based analysis of blood gene expression from healthy individuals exposed to respiratory viruses and provide predictive models and biological insight into the physiological response.
Introduction
Acute respiratory viral infections are among the most common reasons for outpatient clinical encounters1. Symptoms of viral infection may range from mild (e.g. sneezing, runny nose) to life-threatening (dehydration, seizures, death), though many individuals exposed to respiratory viruses remain entirely asymptomatic2. Variability in individuals’ responses to exposure has been observed both in natural infections3 and controlled human viral exposure studies. Specifically, some individuals remained asymptomatic despite exposure to respiratory viruses, including human rhinovirus (HRV)4–6, respiratory syncytial virus (RSV)4–6, influenza H3N24–9, and influenza H1N14,5,9. Factors responsible for mediating response to respiratory viral exposure are poorly understood. These individual responses are likely influenced by multiple processes, including the host genetics10, the basal state of the host upon exposure11, and the dynamics of host immune response in the early hours immediately following exposure and throughout the infection12. Many of these processes occur in the peripheral blood through activation and recruitment of circulating immune cells13. However, it remains unknown whether host factors conferring resilience or susceptibility to symptomatic infectious disease can be detected in peripheral blood before infection, or whether they are only apparent in response to pathogen exposure.
In order to identify such gene expression markers of resilience and susceptibility to acute respiratory viral infection, we utilized gene expression data from seven human viral exposure experiments6,7,9. These exposure studies have shown that global gene expression patterns measured in peripheral blood around the time of symptom onset (as early as 36 h after viral exposure) are highly correlated with symptomatic manifestations of illness6,9. However, these later-stage observations do not necessarily reflect the spectrum of early timepoint immune processes that might predict eventual infection. Since transcriptomic signals are weak at these early timepoints, the detection of early predictors of viral response has not yet been possible in any individual study. By combining data collected across these seven studies and leveraging the community to implement state-of-the-art analytical algorithms, the Respiratory Viral DREAM Challenge (www.synapse.org/ViralChallenge) aims to develop early predictors of resilience or susceptibility to symptomatic manifestation based on expression profiles that are collected prior to and at early timepoints following viral exposure and to understand the biological mechanisms underlying those predictors.
Results
Human viral exposure experiments
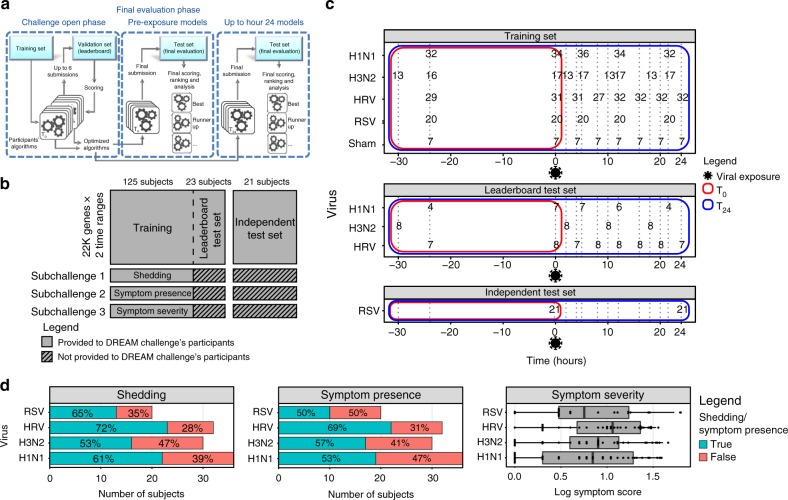
In order to determine whether viral susceptibility could be predicted prior to viral exposure, we collated seven human viral exposure experiments: one RSV, two influenza H1N1, two influenza H3N2, and two HRV studies, in which a combined total of 148 healthy volunteers were exposed to virus (Supplementary Data 1; Fig. 1a−c) or sham (n = 7)6,7,9. Subjects were excluded if pre-existing neutralizing antibodies were detected, except for the RSV study in which neutralizing antibodies were not an exclusion criteria. Each subject in the study was followed for up to 12 days after exposure and serially sampled for peripheral blood gene expression by Affymetrix Human U133A 2.0 GeneChips. Throughout the trial, subjects self-reported clinical symptom scores across 8−10 symptoms (Supplementary Figure 1).These data were used to stratify subjects as either symptomatic or asymptomatic and to quantify symptom severity. Additionally, nasopharyngeal swabs measured viral shedding; these data were used to stratify subjects as either shedders or nonshedders (Fig. 1d). Clinical symptoms were summarized based on a modified Jackson score14 and viral shedding was determined to be present if two or more measurable titers or one elevated titer was observed within 24 h following viral exposure15. Viral shedding and clinical symptoms were provided to the Respiratory Viral DREAM Challenge participating teams only for the training data set (Fig. 1b). An additional, but not previously available, human exposure experiment to the RSV virus (n = 21) was used as an independent test data set (Fig. 1b, c). The study design for this data set was similar to those of the seven original data sets.
Fig. 1.
Respiratory Viral DREAM Challenge overview. a Schematic representation of the Respiratory Viral DREAM Challenge workflow. Participants used feedback from evaluation on the leaderboard test set to optimize their T0 and T24 models, and submitted a single model, per timepoint, for final evaluation on the Independent Test Set. b Schematic representing the data provided to participants. 125 subjects were provided as training data, 23 subjects were provided as a leaderboard test set, and 21 subjects from an independent data set were used for final evaluation. c Challenge data come from seven viral exposure trials with sham or one of four different respiratory viruses (H1N1, H3N2, Rhinovirus, and RSV). In each of these trials, healthy volunteers were followed for 7−9 days following controlled nasal exposure to one respiratory virus. Blood was collected and gene expression of peripheral blood was performed 1 day (24−30 h) prior to exposure, immediately prior to exposure and at regular intervals following exposure. Data were split into a training, leaderboard, and independent test set. Outcome data for the leaderboard and independent test set were not provided to the teams, but instead, teams were asked to predict them based on gene expression pre-exposure (T0) or up to 24 h post-exposure (T24). d Histograms and boxplot of the three outcomes by viruses. Symptom data and nasal lavage samples were collected from each subject on a repeated basis over the course of 7−9 days. Viral infection was quantified by measuring the release of viral particles from viral culture or by qRT-PCR (viral shedding). Symptomatic data were collected through self-report on a repeated basis. Symptoms were quantified using a modified Jackson score, which assessed the severity of eight upper respiratory symptoms (runny nose, cough, headache, malaise, myalgia, sneeze, sore throat, and stuffy nose). On the boxplot, the lower whisker, the lower hinge, the mid hinge, the upper hinge and the upper whisker correspond to −1.5× the interquartile (IQR) from the first quartile, the first quartile, the median, the third quartile and 1.5× IQR from the third quartile of the log symptom score, respectively
Data analysis challenge
Using these data, an open data analysis challenge, the Respiratory Viral DREAM Challenge, was formulated. Teams were asked to predict viral shedding and clinical symptoms based on peripheral blood gene expression data from up to two timepoints: prior to viral exposure (T0) or up to 24 h post viral exposure (T24). Based on gene expression data from the two timepoints, teams were asked to predict at least one of three outcomes: presence of viral shedding (subchallenge 1 (SC1)), presence of symptoms, defined as a modified Jackson score ≥ 6 (subchallenge 2 (SC2)), or symptom severity, defined as the logarithm of the modified Jackson score (subchallenge 3 (SC3)). Teams were asked to submit predictions based on gene expression and basic demographic (age and gender) data from both timepoints to enable cross-timepoint comparison. The seven collated data sets served as a training data set on which teams could build their predictive models. For a subset of subjects (n = 23), phenotypic data were withheld to serve as a leaderboard test set for evaluation with real-time feedback to teams (Fig. 1a).
Teams were asked to submit at least one leaderboard submission at each timepoint to be evaluated on the leaderboard test set. Performance metrics for these models were returned in real time, and teams could update their submissions accordingly up to a maximum of six combined submissions per subchallenge. At the end of this exercise, teams were asked to provide leave-one-out cross-validation-based predictions on the training set (LOOCVs) and predictor lists for each of their best models.
Each team’s best models (one for T0 and one for T24) per subchallenge were ultimately assessed on the held-out human RSV exposure data set that had not been publicly available, previously (Fig. 1a). Predictions for the binary outcomes (shedding and symptoms) were assessed using Area Under the Precision-Recall (AUPR) and Receiver Operating Characteristic (AUROC) curves, and ranked using the mean rank of these two measures. The predictions for the continuous outcome (symptom severity) were assessed using Pearson’s correlation (r) with the observed values. In each case, permutation-based p values were used to identify submissions that performed significantly better than those expected at random. In total, 37 teams participated in some stage of the challenge (Supplementary Table 1).
Challenge results
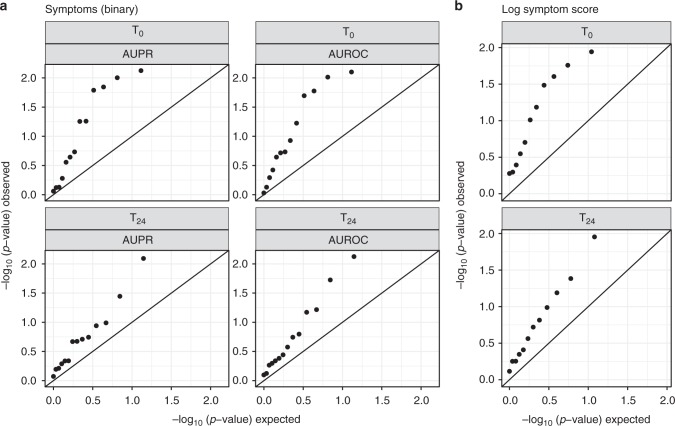
For presence of symptoms (SC2), 27 models were assessed on the independent test data; 13 models were developed using T0 predictors, and 14 models using T24 predictors. Four of the T0 models and three of the T24 models achieved a nominal p value of 0.05 for AUPR or AUROC, with the best scoring models at each timepoint achieving similar scores (AUPR(T0) = 0.958, AUROC(T0) = 0.863, AUPR(T24) = 0.953, AUROC(T24) = 0.863). Team Schrodinger’s Cat was the only team that achieved nominal significance for all measures and timepoints. Despite the few teams achieving statistical significance, the models submitted were overall more predictive than expected at random (one-sided Kolmogorov–Smirnov test for enrichment p values 0.008, 0.002, 0.021, and 0.05 for AUPR(T0), AUROC(T0), AUPR(T24), and AUROC(T24), respectively; Fig. 2a).
Fig. 2.
Models perform better than expected at random. Observed −log10(p value) versus the null expectation for submitted predictions for predicting a presence of symptoms (SC2) and b log symptom score (SC3), where p values were assessed by permutation of the predictions relative to the true values. For both subchallenges significant enrichment of p values (Kolmogorov–Smirnov test for enrichment p value 0.008, 0.002, 0.021, and 0.05 for AUPR(T0), AUROC(T0), AUPR(T24), and AUROC(T24), respectively, for presence of symptoms, and one-sided Kolmogorov–Smirnov test for enrichment p value 0.005 and 0.035 for T0 and T24, respectively, for log symptom score) across submissions demonstrates that pre-exposure and early post-exposure transcriptomic data can predict susceptibility to respiratory viruses
For symptom severity (SC3), 23 models were assessed on the independent test data; 11 models were developed using T0 predictors and 12 models using T24 predictors. Four of the T0 models and two of the T24 models achieved a nominal p value of 0.05 for correlation with the observed log-symptom score, and as above, the best performing models scored similarly at both timepoints (r = 0.490 and 0.495 for T0 and T24, respectively). Teams cwruPatho and Schrodinger’s Cat achieved significant scores at both timepoints. Consistent with SC2, we also saw that the models submitted were overall more predictive than expected at random (one-sided Kolmogorov–Smirnov test for enrichment p values 0.005 and 0.035 for T0 and T24, respectively; Fig. 2b). For both SC2 and SC3, enrichment was more pronounced at T0 compared to T24. Correlation between final scores and leaderboard scores was higher at T0, suggesting T24 predictions may have been subject to a greater degree of overfitting.
For viral shedding (SC1), 30 models were assessed from 16 different teams; 15 models were developed using T0 predictors and 15 models using T24 predictors. No submissions were statistically better than expected by random. In aggregate, these submissions showed no enrichment (one-sided Kolmogorov–Smirnov test for enrichment p values 0.94, 0.95, 0.82, and 0.95, for AUPR(T0), AUROC(T0), AUPR(T24), and AUROC(T24), respectively). In contrast, final scores were negatively correlated with leaderboard scores (r = −0.22, −0.19, −0.65, and −0.54 for AUPR(T0), AUROC(T0), AUPR(T24), and AUROC(T24), respectively) suggesting strong overfitting to the training data or a lack of correspondence to viral shedding as assessed in the independent test data set, relative to the training data sets. The negative correlation was strongest at T24 (Supplementary Figure 2). Accordingly, results based on this subchallenge were excluded from further analysis.
Best performing approaches
The two overall best performing teams were Schrodinger’s Cat and cwruPatho. Team Schrodinger’s Cat used the provided gene expression profiles before the viral exposure to predict shedding and log symptom scores (binary and continuous outcomes, respectively). For the T0 models, arithmetic means over measurements prior to exposure were calculated, whereas for the T24 models, only the latest measurements before viral exposure were used. Epsilon support vector regression (epsilon-SVR)16 with a radial kernel and tenfold cross-validation were used to develop the predictive models. Their work demonstrated that predictive models of symptoms following viral exposure can be built using pre-exposure gene expression.
Team cwruPatho constructed models of infection based on pathway modulation, rather than gene expression, to predict infection outcomes. To do so, they used a sample-level enrichment analysis (SLEA)17 approach to summarize the expression of genes implicated in the Hallmark gene sets18 of the Molecular Signature DataBase (MSigDB)19. They then fitted LASSO regularized regression models, which integrate feature selection with a regression fit20, on the pathways to predict shedding, presence of symptoms and symptom severity following viral exposure. Their work demonstrated that including multiple genes sharing the same biological function results in more robust prediction than using any single surrogate gene.
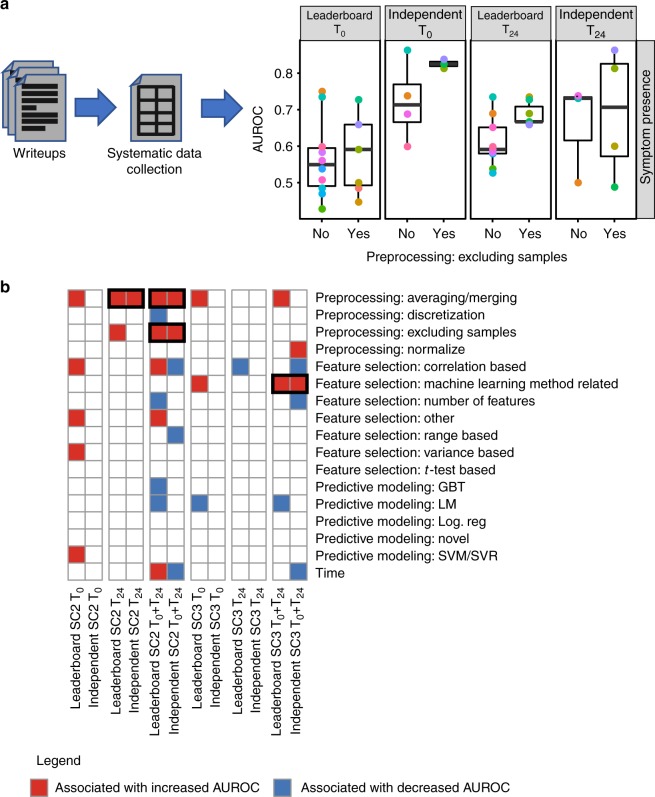
Teams Schrodinger’s Cat and cwruPatho used different feature transformation methods and machine learning approaches, suggesting that multiple approaches can successfully identify pre- or early post-exposure transcriptomic markers of viral infection susceptibility or resilience. To gauge the range of approaches taken, we extended this comparison to all Respiratory Viral DREAM Challenge teams who reported details on the methods they used to develop their submissions. We assessed the range of data preprocessing, feature selection, and predictive modeling approaches employed for the submissions, to determine whether any of these methods were associated with better prediction accuracy. Details of these three analysis steps (preprocessing, feature selection and predictive modeling) were manually extracted from reports of 24 teams (35 separate reports) who submitted predictions either for the leaderboard test set or the independent test set. To more precisely reflect the conceptual variations across employed methodologies, each of these three analysis tasks was broken down into four data preprocessing categories, seven feature selection categories and nine predictive modeling categories (Supplementary Table 2). Twenty of 24 (83.3%) teams employed some version of data preprocessing, the task most significantly associated with predictive ability (Supplementary Figure 3A). Specifically, exclusion of sham-exposed subjects and data normalization associated best with predictive performance (Fig. 3).
Fig. 3.
Preprocessing leads to more accurate predictions. a Schematic representation of the analysis of the participating teams’ writeups to identify methodological steps associated with more accurate prediction of symptoms. First, the writeups were manually inspected to identify the preprocessing, feature selection and predictive modeling method used by each team. Second, the methods were regrouped into general categories across teams. Third, each general method was assessed for its association with predictive model accuracies on the leaderboard test set and the independent test set. On the boxplot, the lower whisker, the lower hinge, the mid hinge, the upper hinge and the upper whisker correspond to −1.5× IQR from the first quartile, the first quartile, the median, the third quartile and 1.5× IQR from the third quartile of the AUROC, respectively. b Heatmap showing the association of each general method with prediction ability (i.e. AUROC for SC2 (prediction of symptom presence) and Pearson’s correlation coefficient for SC3 (prediction of symptom severity)). For each general method, a Wilcoxon rank-sum test was used to assess the association between using the method (coded as a binary variable) and prediction ability
Feature selection and predictive modeling approaches positively associated with predictive ability differed depending on whether the task was classification (presence of symptoms) or regression (symptom severity). Random forest-based predictive models performed slightly better than support vector machine (SVM)/support vector regression (SVR) methods at predicting symptom status (SC2) (Supplementary Figure 3B). However, there was no discernible pattern relating feature selection and improved performance in SC2. Feature selection using machine learning approaches such as cross-validation was associated with improved performance in predicting symptom severity (SC3) (Fig. 3), as were SVM/SVR approaches when compared to linear regression model-based methods (e.g. logistic regression; Supplementary Figure 3C). Of note, SVM/SVR approaches were the most popular among the submissions.
We also sought to compare cross-timepoint predictions to determine the stability of predictions by timepoint. Significant correlation was observed between predictions using T0 and T24 gene expression for symptomatic classification (SC2) (Leaderboard: ρ = 0.608, p value = 1.04e-61; Independent test set: ρ = 0.451, p value = 2.05e-25). Interestingly, we observed that approximately 25% of subjects were difficult to predict based on T0 gene expression profile (inherently difficult; Supplementary Figure 4); similarly, approximately 25% of subjects were correctly predicted by the majority of teams (inherently easy; Supplementary Figure 4). Inherently difficult subjects were also misclassified when T24 gene expression data was used for prediction. Inherently easy subjects were also consistently easy to classify using T24 gene expression data. This suggests ab initio characteristics allow some subjects to be more susceptible or resilient to symptomatic disease and that, within 24 h, those characteristics are not substantially altered in post-exposure peripheral blood expression profiles.
Biological interpretation of predictors
In addition to predictions, each team was asked to submit lists of gene expression features used in their predictive models. Twenty-four teams submitted predictive models with AUROC > 0.5 for SC2 or r > 0 for SC3 (leaderboard test set) for either T0 or T24, among which six teams submitted separate models for each virus and reported virus-specific predictors. The remaining 18 reported models independent of virus, submitting a single model for all viruses. With the exception of the list from cwruPatho, which used pathway information in the selection of features, pathway analysis of individual predictor lists showed no enrichment of pathways from MSigDB19, possibly due to the tendency of most feature selection algorithms to choose one or few features from within correlated sets.
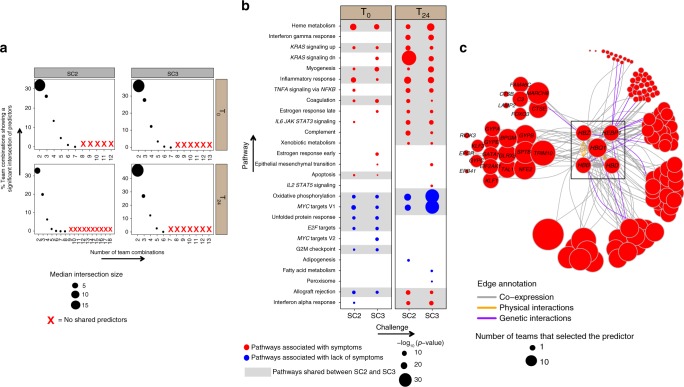
We then assessed whether models showing predictive ability (leaderboard test set AUROC > 0.5 for SC2 or r > 0 for SC3) tended to pick the same gene features, or whether the different gene sets may provide complementary information. Within each subchallenge and timepoint, the significance of the overlap among predictor lists was calculated for every combination of two or more predictor lists across teams. All two-way, three-way, four-way, etc. overlaps were considered. This analysis revealed that there were no genes shared among all teams for any timepoint or subchallenge (Fig. 4a).
Fig. 4.
Overlap and pathway enrichment among predictors of symptoms. a Percent of team combinations showing statistically significant intersections of predictors at T0 and T24. Only teams with AUROC ≥ 0.5 or r ≥ 0 for subchallenges 2 and 3, respectively, were used for this analysis. The x-axis indicates the number of teams included in the combination. For example, the value 2 corresponds to pairwise overlaps, 3 corresponds to 3-way overlaps, etc. The y-axis indicates the percentage of team combinations with a statistically significant (p value < 0.05) predictor intersection. Point size indicates median intersection size of predictors among team combinations with significant predictor intersection; “X” indicates no significant predictor intersection. b Pathway enrichment among predictors of infection for each subchallenge (SC2 and SC3) at T0 and T24. The x-axis indicates subchallenge and each grid indicates timepoint. The y-axis indicates pathways enriched among predictors with a Benjamini−Hochberg-corrected Fisher’s exact test p value < 0.05. Point size represents the Fisher’s exact test enrichment −log10(p value). Point colors indicate whether the pathway was associated with symptoms (red) or lack thereof (blue). Pathways shared between both SC2 and SC3 at each timepoint are highlighted in gray. Pathways are ordered by the decreasing maxP test statistic as determined in Supplementary Figure 5. c GeneMANIA network of the union of predictors involved in the Heme metabolism pathway across timepoints (T0 and T24) and subchallenges (SC2 and SC3). Edges are inferred by GeneMANIA51 corresponding to coexpression (gray), physical interactions (orange), and genetic interactions (purple) among genes. Node size corresponds to the number of teams that selected the predictor
Despite the paucity of overlap among predictor lists, we sought to identify whether genes used in the predictive models were part of the same biological processes or pathways. In other words, we examined whether different teams might have chosen different surrogate genes to represent the same pathway. To test this hypothesis, we performed pathway enrichment analysis of the union of predictors across predictor lists within timepoint and subchallenge. We observed significant enrichments in each case (Fig. 4b), suggesting that predictive gene features are indeed complementary across models. More pathways were enriched among predictors from T24 models (SC2 = 17 pathways and SC3 = 20 pathways) than from T0 models (SC2 = 15 pathways and SC3 = 17 pathways). At T0, genes involved in the metabolism of heme and erythroblast differentiation (heme metabolism), genes specifically upregulated by KRAS activation (KRAS signaling (up)), genes defining an inflammatory response (inflammatory response) and genes mediating cell death by activation of caspases (apoptosis) were associated with presence of symptoms in both SC2 and SC3 (Fig. 4b). At T24, along with heme metabolism, the expression of several inflammatory response pathways like KRAS signaling, inflammatory response, genes upregulated in response to the gamma cytokine IFNg (interferon gamma response), genes upregulated by IL6 via STAT3 (IL6 JAK STAT3 signaling), genes regulated by NF-κB in response to TNF (TNFA signaling via NFKB) and genes encoding components of the complement system (complement) were associated with symptoms in both SC2 and SC3 (Fig. 4b). Additionally, there was a significant overlap in genes across timepoints and subchallenges in each of these enriched pathways (Fisher’s exact test p value ≤ 0.05) (Supplementary Data 2).
A meta-analysis across subchallenges (SC2 and SC3) and timepoints (T0 and T24) was performed in order to identify the most significant pathways associated with outcome. Heme metabolism was the most significantly associated with developing symptoms (susceptibility), while oxidative phosphorylation and MYC targets were the most significantly associated with a lack of symptoms (resilience) (Supplementary Figure 5). This indicates that heme, known to generate inflammatory mediators through the activation of selective inflammatory pathways21 is the best predictor of becoming symptomatic both pre- and early post-exposure to respiratory viruses. Genes in heme metabolism associated with symptoms include genes coding for the hemoglobin subunits (HBB, HBD, HBQ1, and HBZ), the heme binding protein (HEBP1) and genes coding for enzymes important for the synthesis of heme (ALAS2, FECH, HMBS, UROD). It also includes glycophorins, which are the major erythrocyte membrane proteins (GYPA, GYPB, GYPC, and GYPE), which are known receptors for the influenza virus (Fig. 4c)22,23. Genes essential for erythroid maturation and differentiation (NEF2, TAL1, EPOR, and GATA1), including the transcription factor GATA1 and its targets, the hemoglobin subunit genes HBB and HBG1/2, were also part of heme metabolism associated with an increase in symptom frequency and severity.
Discussion
Using an open data analysis challenge framework, this study showed that models based on transcriptomic profiles, even prior to viral exposure, were predictive of infectious symptoms and symptom severity, which has not been previously demonstrated. The best scoring individual models for predicting symptoms and log-symptom score, though statistically significant, fall short of practical significance. However, these outcomes suggest that there is potential to develop models and ultimately, clinically relevant tests, based on the knowledge gained from these results. This would necessitate further efforts to generate more data or identify different biomarker assays which more accurately assess the mechanisms observed in the transcriptomic models. Additionally, since these studies focused on healthy adults, further data generation should extend to a wider range of subjects with respect to age and health status, as well as tracking and modeling these cofactors.
A generally useful exercise in crowdsourcing-based challenges is to construct ensembles from the submissions to assimilate the knowledge contained in them, and boost the overall predictive power of the challenge24. This exercise has yielded useful results in earlier benchmark studies25,26 and the DREAM Rheumatoid Arthritis Challenge27. However, the ensembles constructed for the Respiratory Viral DREAM Challenge did not perform better than the respective best performers among all the individual submissions for the various subchallenges and timepoints. We attribute this shortcoming partly to the relatively small training set (118 subjects), which may incline the ensemble methods to overfit these data, and the assumption of class-conditioned independence of the submissions inherent in SUMMA may not have been appropriate in this challenge28. The relative homogeneity, or lack of diversity, among the submissions for the various subchallenges and timepoints may have been another potential factor behind the diminished performance of the ensembles29.
The relative homogeneity of submissions and observation that the same subjects are misclassified by almost all participating teams suggests there may be a plateau in predictive ability when using gene expression to predict the presence of symptoms or symptom severity. It is possible that an integrative analysis supplementing or replacing the gene expression data with post-transcriptional (such as metabolomic or proteomic) data could further improve accuracy. For example, metabolomic data have been used to differentiate patients with influenza H1N1 from others with bacterial pneumonia or non-infectious conditions as well as differentiate influenza survivors from nonsurvivors30. With respect to proteomics, Burke et al. used four of the viral exposure studies described here to derive and validate a proteomic signature from nasal lavage samples which distinguish, with high accuracy, symptomatic from asymptomatic subjects at the time of maximal symptoms31. Several cytokines have been investigated in a variety of infectious disease conditions. Of particular relevance, cytokine profiling has been performed for one of the influenza H3N2 studies used in this Challenge. In that work, McClain et al. demonstrated that several cytokines were upregulated early after viral exposure (within 24 h in some cases) and differentiated symptomatic from asymptomatic cases32. Baseline differences in cytokine expression were not observed, however, suggesting that cytokine expression is useful for predicting response to viral exposure but not baseline susceptibility. To our knowledge, no study has identified baseline metabolomic or proteomic predictors of resilience or susceptibility to respiratory viral infection. In addition, the combination of these data with transcriptomic predictors has not yet been investigated and may yield robust predictors of susceptibility or resistance to infection.
Our analyses revealed a significant concordance between predictions at T0 and T24 (Supplementary Figure 4), as well as a significant overlap between predictors at each of these timepoints (Supplementary Data 2). Given the stability of predictions and predictors between T0 and T24, it appears that the pre-exposure biological mechanisms conferring susceptibility or resilience to respiratory viral infection may be observable up to 1 day post-exposure. We also observed significant overlap between gene signatures at both T0 and T24 and late stage signatures of viral infection, reported in the literature, and derived from gene expression 48 h or later after viral exposure (Supplementary Data 3)5–9,15,33–38. The overlap between the predictors identified in this study and the later stage signatures was more significant at T24 than T0, suggesting that pre-exposure signatures of susceptibility differ somewhat from post-exposure signatures of active infection, and T24 predictors may reflect some aspects of both. The T0 gene signatures may encompass novel insight into ab initio factors that confer resilience or susceptibility.
Pathway enrichment analysis in our study revealed that the most significantly enriched pathway associated with symptomatic infection was heme metabolism, known to have a direct role in immunity through activation of innate immune receptors on macrophages and neutrophils21. Of note, genes part of heme metabolism were also enriched among late stage signatures of viral infection (ex. Hemoglobin gene HBZ and the iron containing glycoprotein ACP5 in ref.33). Iron (obtained from heme) homeostasis is an important aspect of human health and disease. Viruses require an iron-rich host to survive and grow, and iron accumulation in macrophages has been shown to favor replication and colonization of several viruses (e.g. HIV-1, HCV) and other pathogenic microorganisms39. Furthermore, iron-replete cells have been shown to be better hosts for viral proliferation39. Increased iron loading in macrophages positively correlates with mortality39 and it has been shown that viral infection can cause iron overload which could further exacerbate disease. Additionally, previous evidence suggests counteracting iron accumulation may limit infection21,39. Studies have shown that limiting iron availability to infected cells (by the use of iron chelators) curbed the growth of several infectious viruses and ameliorated disease21,39–41. This important role of iron in the susceptibility and response to infection may be the mechanism by which heme metabolism genes conferred susceptibility to respiratory viral infection. As such, it represents an important biological pathway potentially offering a means by which an individual’s susceptibility or response to infection can be optimized. Such a relationship should be investigated in future studies of infection susceptibility. In addition, Heme-oxygenase (HMOX1), a heme-degrading enzyme that antagonizes heme-induced inflammation and is essential for the clearance of heme from circulation42, was among the predictors from the T0 models. Interestingly, the expression of this gene at baseline was associated with a lack of symptoms (for both SC2 and SC3), in concordance with its reported antiviral role during influenza infection43,44. Augmentation of HMOX1 expression by gene transfer had provided cellular resistance against heme toxicity45. Hence enhancing HMOX1 activity could be an alternative to antagonize heme-induced effects and thereby controlling infection and inflammation.
In addition to heme metabolism, pro-inflammatory pathways such as inflammatory response, KRAS signaling, and apoptosis were also associated with susceptibility to viral infection in our study, while homeostatic pathways, such as oxidative phosphorylation and MYC targets, were associated with resilience, both prior to and post viral exposure (Fig. 4). Enrichment of these pathways among T24 predictors was more significant than among the T0 predictors, suggesting these mechanisms are not only emblematic of baseline system health, but also response to viral invasion. Additional pathways enriched among T24 predictors include interferon gamma response and complement, which are involved in innate and acquired immunity. Several genes among T0 and T24 predictors overlapped with genes positively associated with flu vaccination response46. Among them, FCER1G and STAB1, members of the inflammatory response pathway positively associated with symptoms in this study and were elevated prior to vaccination in young adults who showed good response to vaccination46 (Fisher exact test: p = 0.0338 for T0 and p = 0.000673 for T24). This suggests that individuals predicted at a higher risk of presenting symptoms following influenza exposure may also be the most likely to benefit from vaccination.
The Respiratory Viral DREAM Challenge is to date the largest and most comprehensive analysis of early stage prediction of viral susceptibility. The open data analysis challenge framework is useful for comparing approaches and identifying the most scientifically or clinically relevant model or method in an unbiased fashion24. In this case, we observed few commonalities among the best performing models of symptomatic susceptibility to respiratory viral exposure. Indeed, the overall best performing teams in the challenge used different machine learning techniques to build their models. Interestingly, data preprocessing was the analysis task most significantly associated with model accuracy, suggesting what has often been speculated, that adequate attention to data processing prior to predictive modeling is a crucial first step47.
The open data challenge framework is also useful in arriving at consensus regarding research outcomes that may guide future efforts within a field24. Through this challenge, we have identified ab initio transcriptomic signatures predictive of response to viral exposure, which has provided valuable insight into the biological mechanisms conferring susceptibility to infection. This insight was not evident from any individual model, but became apparent with the meta-analysis of the individual signatures. While development of a diagnostic test of baseline susceptibility is not yet feasible based on these findings, they suggest potential for development in this area.
Methods
Training data
Training data came from seven related viral exposure trials, representing four different respiratory viruses. The data sets are DEE1 RSV, DEE2 H3N2, DEE3 H1N1, DEE4X H1N1, DEE5 H3N2, Rhinovirus Duke, and Rhinovirus UVA6,7,9. In each of these human viral exposure trials, healthy volunteers were followed for 7−9 days following controlled nasal exposure to the specified respiratory virus. Subjects enrolled into these viral exposure experiments had to meet several inclusion and exclusion criteria. Among them was an evaluation of pre-existing neutralizing antibodies to the viral strain. In the case of influenza H3N2 and influenza H1N1, all subjects were screened for such antibodies. Any subject with pre-existing antibodies to the viral strain was excluded. For the rhinovirus studies, subjects with a serum neutralizing antibody titer to RV39 > 1:4 at prescreening were excluded. For the RSV study, subjects were prescreened for neutralizing antibodies, although the presence of such antibodies was not an exclusion criterion.
Symptom data and nasal lavage samples were collected from each subject on a repeated basis over the course of 7−9 days. Viral infection was quantified by measuring release of viral particles from nasal passages (viral shedding), as assessed from nasal lavage samples via qualitative viral culture and/or quantitative influenza RT-PCR. Symptom data were collected through self-report on a repeated basis. Symptoms were quantified using a modified Jackson score14, which assessed the severity of eight upper respiratory symptoms (runny nose, cough, headache, malaise, myalgia, sneeze, sore throat, and stuffy nose) rated 0−4, with 4 being most severe. Scores were integrated daily over 5-day windows.
Blood was collected and gene expression of peripheral blood was performed 1 day (24−30 h) prior to exposure, immediately prior to exposure, and at regular intervals following exposure. These peripheral blood samples were gene expression profiled on the Affy Human Genome U133A 2.0 array.
All subjects exposed to influenza (H1N1 or H3N2) received oseltamivir 5 days post-exposure. However, 14 (of 21) subjects in the DEE5 H3N2 cohort received early treatment (24 h post-exposure) regardless of symptoms or shedding. Rhinovirus Duke additionally included seven volunteers who were exposed to sham rather than active virus.
All subjects provided written consents, and each of the seven trials was reviewed and approved by the appropriate governing IRB.
RSV test data
Healthy nonsmoking adults aged 18−45 were eligible for inclusion after screening to exclude underlying immunodeficiencies. A total of 21 subjects (10 female) were inoculated with 104 plaque-forming units of RSV A Memphis 37 (RSV M37) by intranasal drops and quarantined from 1 day before inoculation to the 12th day after. Peripheral blood samples were taken immediately before inoculation and regularly for the next 7 days and profiled on the Affy Human Genome U133A 2.0 array. Subjects were discharged after study day 12, provided no or mild respiratory symptoms and a negative RSV antigen respiratory secretions test. Shedding was determined by polymerase chain reaction (PCR) in nasal lavage and defined as detectable virus for ≥2 days between day +2 and day +10 to avoid false-positives from the viral inoculum and to align case definitions with the other seven studies. Subjects filled a diary of upper respiratory tract symptoms from day −1 to day +12, which was summarized using a modified Jackson score. All subjects returned for further nasal and blood sampling on day +28 for safety purposes. All subjects provided written informed consent and the study was approved by the UK National Research Ethics Service (London-Fulham Research Ethics Committee ref. 11/LO/1826).
Gene expression normalization
Both raw (CEL files) and normalized versions of the gene expression data were made available to teams in the Challenge. Both versions contained only profiles that pass QC metrics including those for RNA Degradation, scale factors, percent genes present, β-actin 3′ to 5′ ratio and GAPDH 3′ to 5′ ratio in the Affy Bioconductor package. Normalization via RMA was performed on all expression data across all timepoints for the training and leaderboard data sets. The RSV data were later normalized together with the training and leaderboard data, and teams were free to further QC and normalize the data in the way they deemed appropriate.
Analysis challenge design
The training data studies were split into training and leaderboard sets, where the leaderboard subjects were chosen randomly from three of the trials: DEE4X H1N1, DEE5 H3N2, and Rhinovirus Duke, which were not publicly available at the time of challenge launch. Outcome data for the leaderboard set were not provided to the teams, but instead, teams were able to test predictions in these individuals using the leaderboard, with a maximum of six submissions per subchallenge, the purpose of which was to allow teams to optimize their models prior to assessment on the independent test data. Of these, at least one submission was required to use only data prior to viral exposure and at least one using data up to 24 h post-exposure.
For the training data, teams had access to clinical and demographic variables: age, sex, whether the subject received early oseltamivir treatment (DEE5 H3N2 only) and whether the subject received sham exposure rather than virus (Rhinovirus Duke only), as well as gene expression data for the entire time-course of the studies. They also received data for the three outcomes used in the data analysis challenge:
Subchallenge 1: SHEDDING_SC1, a binary variable indicating the presence of virus in nasal swab following exposure;
Subchallenge 2: SYMPTOMATIC_SC2, a binary variable indicating post-exposure maximum 5-day integrated symptom score ≥6;
Subchallenge 3: LOGSYMPTSCORE_SC3, a continuous variable indicating the log of the maximum 5-day integrated symptom score +1
as well as the granular symptom data by day and symptom category. For the leaderboard test data, they were supplied with the clinical and demographic variables and gene expression data up to 24 h post-exposure.
Final assessment of optimized models was performed in the RSV Test Data (i.e. the independent test set), and outcomes for these subjects were withheld from teams. In order to assure that predictions were limited to data from the appropriate time window, the gene expression data were released in two phases corresponding to data prior to viral exposure, and data up to 24 h post exposure. Teams were also supplied with age and sex information for these subjects.
The Challenge was launched and training data were released on May 15, 2016 for participants to use to begin analyzing the data and building their models. In total 38 teams registered for the challenge and 37 participated (Supplementary Table 1). The leaderboards opened approximately 2 months later, and were open for approximately 3 months (July to September) to allow participants to optimize their models with feedback from the scores on the leaderboard data. At the close of this round on September 30, participating teams were also required to submit code, methodological writeups, predictor lists, and LOOCVs, and doing so qualified participants to be included as authors (either Consortium or by-line) on this manuscript. Participating teams could opt to evaluate their optimized models in the independent test data, which occurred from January to February 2017. At the close of the challenge, participating teams were invited to collaborate with the Challenge Organizers to analyze the results. Prior to the launch of the challenge, substantial effort was put forth by the Challenge organizers to collate and vet the data, to determine the feasibility of the Challenge and define the Challenge objectives. For further details on the organizational efforts required to prepare for a challenge, see Saez-Rodriguez et al.24.
Submission scoring
Team predictions were compared to true values using AUPR and AUROC for subchallenges 1 and 2, and Pearson correlation for subchallenge 3. For each submission, a p value, estimating the probability of observing the score under the null hypothesis that the predicted labels are random, was computed by 10,000 permutations of the predictions relative to the true values.
Enrichment of p values of the submitted models was assessed via 1-sided Kolmogorov–Smirnov test with a null hypothesis that the p values follow a U[0,1] distribution, and an alternative hypothesis that they follow a distribution that is stochastically smaller than U[0,1].
Heterogeneity of the predictions
T0 and T24 predictions for each outcome and team were collected to assess whether they were correlated. Three teams provided predictions as binary values while 12 teams provided predictions as continuous values on different scales. In order to compare binary and continuous predictions, we first transformed them into ranks (with ties given the same average rank) and then ordered subjects increasingly by their mean rank across outcomes (mean-rank). The lower the mean-rank, the more likely a subject was predicted by the teams as not showing symptoms, whereas a higher mean-rank means a subject was predicted by most of the teams as showing symptoms. Distribution of the mean-rank (Supplementary Figure 4) revealed three groups of subjects: (1) ~25% of subjects correctly predicted by most of the teams (i.e. inherently easy), (2) ~25% of subjects incorrectly predicted by most of the teams (i.e. inherently difficult) and (3) ~50% of subjects who were predicted differently by the teams.
Ensemble prediction
We constructed a variety of ensembles from the teams’ submissions to the various subchallenges as a part of the collaborative phase of the Respiratory Viral DREAM Challenge. To enable a comparative analysis between individual and ensemble models in the collaborative phase, the teams were requested to submit LOOCV-derived predictions on the training examples using the same methods used to generate leaderboard and/or test set predictions in the competitive phase. The LOOCV setup, which does not involve random subsetting of the training data, was chosen to avoid potential overfitting that can otherwise occur from training and testing on predictions made on the same set of examples25. We used three types of approaches for learning ensembles, namely stacking and its clustering-based variants25, Reinforcement Learning-based ensemble selection26 methods, as well as SUMMA, an unsupervised method for the aggregation of predictions28. Consistent with the process followed by the individual teams, we learned all the ensembles using the training set LOOCV-derived predictions described above, and used the leaderboard data to select the final models to be evaluated on the test data.
Combined gene sets
Statistical significance of the overlap among predictor lists was calculated using the multiset intersection probability method implemented in the SuperExactTest R package48. A first set of analysis was performed with teams whose leaderboard AUROC > 0.5. A second set of analysis aimed at identifying genes that overlap virus-specific, subchallenge-specific and timepoint-specific predictive models, was restricted to teams that provided virus-specific (Nautilus, aydin, SSN_Dream_Team, Txsolo, cwruPatho and Aganita), subchallenge-specific (aydin, SSN_Dream_Team, cwruPatho, jhou) and timepoint-specific predictors (aydin, SSN_Dream_Team, cwruPatho, Espoir, jdn, jhou, burkhajo) and participated in the leaderboard phase of the challenge, respectively. For both analyses, overlapping predictors associated with p values less than or equal to 0.005 were considered significant49.
Pathway enrichment analysis
To assess pathway enrichment among predictors of infection, we considered predictors from teams with leaderboard AUROC > 0.5 (SC2) or Pearson correlation, r > 0 (SC3). Affymetrix Human U133A 2.0 GeneChip probe identifiers were mapped to gene symbols. We removed probes matching multiple genes, and when multiple probes matched a single gene, we retained the probe with the maximum median intensity across subjects.
For the list of predictors of presence of symptoms (SC2), we calculated the log2 fold-change of features (symptomatic(1)/asymptomatic(0)) at T0 and T24, and for prediction of the symptom scores (SC3), we calculated the Spearman’s correlation coefficient of the features, at T0 and T24, with the outcome. Pathway enrichment was then performed on the union of all predictors (across the teams) that were associated with presence/increase severity of symptoms (SC2: log2 fold-change > 0 and SC3: Spearman’s correlation > 0), as well as, for the union of all predictors (across teams) that were associated with lack of symptoms/lower symptoms severity (SC2: log2 fold-change < 0 and SC3: Spearman’s correlation < 0), separately by timepoint and subchallenge. We used the Hallmark gene sets (version 6.0)18 of the Molecular Signature DataBase (MSigDB)19 for the enrichment analysis, and calculated the significance using Fisher’s exact test. The resulting p values were corrected for multiple comparisons using the Benjamini and Hochberg algorithm. Only significantly enriched pathways (corrected p value < 0.05) were reported. Meta-analyses across subchallenges and timepoints were performed using the maxP test statistic50.
Code availability
Code for individual models are available through www.synapse.org/ViralChallenge.
Electronic supplementary material
Description of Additional Supplementary Files
Acknowledgements
This work was supported by Defense Advanced Research Projects Agency and the Army Research Office through Grant W911NF-15-1-0107. The views expressed are those of the authors and do not reflect the official policy or position of the Department of Defense or the U.S. Government. J.G.B. was supported by a training grant from the National Institutes of Health, USA (NIH grant 4T15LM007088-25). G.P. and A.S.’s work was supported by NIH grant # R01GM114434 and an IBM faculty award to G.P. T.E.M.N. was supported by the Ministry of Science and Technology of Taiwan grants MOST 105-2218-E-006-016-MY2 and 107-2634-F-006-009. K.Y.Y. was supported by NIH grants U54 HL127624 and R01GM126019. M.S. was supported by Grants-in-Aid for Scientific Research JP16H02866 from the Japan Society for the Promotion of Science. We wish to thank the DARPA Biochronicity program and its program manager, Dr. Jim Gimlett, for generously offering to share gene expression data generated as part of that program and Rafick P. Sekaly (Case Western Reserve University) for his critical feedback during the writing process.
Author contributions
R.H., C.C., M.T.M., C.W.W., G.S.G., and E.L.T. devised and performed the viral exposure experiments. R.H., T.Y., G.S.G., E.L.T., L.M.M., and S.K.S. designed and ran the data analysis challenge. S.F., A.T., M.M., J.G.B., R.K., Z.A., K.Y.Y., R.A., S.J., X.L., T.E.M.N., M.S., L.L.E., and The Respiratory Viral DREAM Challenge Consortium members participated in the Challenge and S.F., A.T., M.M., J.G.B., R.K., R.H., Z.A., K.Y.Y., M.E.A., R.A., S.J., X.L., T.E.M.N., M.S., A.S., R.V., G.P., L.L.E., and S.K.S. analyzed the data.
Data availability
Data are available through GEO GSE73072. Challenge results and methods and code for individual models are available through www.synapse.org/ViralChallenge. The authors declare that all other data supporting the findings of this study are available within the article and its Supplementary Information files, or are available from the authors upon request.
Competing interests
E.L.T. reports personal fees from bioMerieux. E.L.T., C.W.W., R.H. and M.T.M. report grants from NIAID. E.L.T., C.W.W., R.H., M.T.M., and G.S.G. report grants from DARPA. E.L.T., C.W.W., and G.S.G. are founders and have equity in Predigen, Inc. E.L.T., M.T.M., C.W.W., R.H., and G.S.G. have a patent pending for methods to diagnose and treat acute respiratory infections. The remaining authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Slim Fourati, Aarthi Talla, Mehrad Mahmoudian, Joshua G. Burkhart, Riku Klén.
Contributor Information
Lara M. Mangravite, Email: lara.mangravite@sagebase.org
Solveig K. Sieberts, Email: solly.sieberts@sagebase.org
The Respiratory Viral DREAM Challenge Consortium:
Emna Ben Abdallah, Farnoosh Abbas Aghababazadeh, Alicia Amadoz, Sherry Bhalla, Kevin Bleakley, Erika Bongen, Domenico Borzacchielo, Philipp Bucher, Jose Carbonell-Caballero, Kumardeep Chaudhary, Francisco Chinesta, Prasad Chodavarapu, Ryan D Chow, Thomas Cokelaer, Cankut Cubuk, Sandeep Kumar Dhanda, Joaquin Dopazo, Thomas Faux, Yang Feng, Christofer Flinta, Carito Guziolowski, Di He, Marta R. Hidalgo, Jiayi Hou, Katsumi Inoue, Maria K Jaakkola, Jiadong Ji, Ritesh Kumar, Sunil Kumar, Miron Bartosz Kursa, Qian Li, Michał Łopuszyński, Pengcheng Lu, Morgan Magnin, Weiguang Mao, Bertrand Miannay, Iryna Nikolayeva, Zoran Obradovic, Chi Pak, Mohammad M. Rahman, Misbah Razzaq, Tony Ribeiro, Olivier Roux, Ehsan Saghapour, Harsh Saini, Shamim Sarhadi, Hiroki Sato, Benno Schwikowski, Alok Sharma, Ronesh Sharma, Deepak Singla, Ivan Stojkovic, Tomi Suomi, Maria Suprun, Chengzhe Tian, Lewis E. Tomalin, Lei Xie, and Xiang Yu
Electronic supplementary material
Supplementary Information accompanies this paper at 10.1038/s41467-018-06735-8.
References
- 1.Lee GC, et al. Outpatient antibiotic prescribing in the United States: 2000 to 2010. BMC Med. 2014;12:96. doi: 10.1186/1741-7015-12-96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Byington CL, et al. Community surveillance of respiratory viruses among families in the Utah better identification of Germs-Longitudinal Viral Epidemiology (BIG-LoVE) Study. Clin. Infect. Dis. 2015;61:1217–1224. doi: 10.1093/cid/civ486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.To KKW, Zhou J, Chan JFW, Yuen KY. Host genes and influenza pathogenesis in humans: an emerging paradigm. Curr. Opin. Virol. 2015;14:7–15. doi: 10.1016/j.coviro.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carin L, et al. High-dimensional longitudinal genomic data: an analysis used for monitoring viral infections. IEEE Signal Process. Mag. 2012;29:108–123. doi: 10.1109/MSP.2011.943009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen M, et al. Detection of viruses via statistical gene expression analysis. IEEE Trans. Biomed. Eng. 2011;58:468–479. doi: 10.1109/TBME.2010.2059702. [DOI] [PubMed] [Google Scholar]
- 6.Zaas AK, et al. Gene expression signatures diagnose influenza and other symptomatic respiratory viral infections in humans. Cell Host Microbe. 2009;6:207–217. doi: 10.1016/j.chom.2009.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang Y, et al. Temporal dynamics of host molecular responses differentiate symptomatic and asymptomatic influenza a infection. PLoS Genet. 2011;7:e1002234. doi: 10.1371/journal.pgen.1002234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McClain MT, et al. A genomic signature of influenza infection shows potential for presymptomatic detection, guiding early therapy, and monitoring clinical responses. Open Forum Infect. Dis. 2016;3:ofw007. doi: 10.1093/ofid/ofw007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Woods CW, et al. A host transcriptional signature for presymptomatic detection of infection in humans exposed to influenza H1N1 or H3N2. PLoS ONE. 2013;8:e52198. doi: 10.1371/journal.pone.0052198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Everitt AR, et al. IFITM3 restricts the morbidity and mortality associated with influenza. Nature. 2012;484:519–523. doi: 10.1038/nature10921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pichon M, Lina B, Josset L. Impact of the respiratory microbiome on host responses to respiratory viral infection. Vaccines. 2017;5:40. doi: 10.3390/vaccines5040040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Iwasaki A, Pillai PS. Innate immunity to influenza virus infection. Nat. Rev. Immunol. 2014;14:315–328. doi: 10.1038/nri3665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Heidema J, et al. Dynamics of human respiratory virus-specific CD8+ T cell responses in blood and airways during episodes of common cold. J. Immunol. 2008;181:5551–5559. doi: 10.4049/jimmunol.181.8.5551. [DOI] [PubMed] [Google Scholar]
- 14.Carrat F, et al. Time lines of infection and disease in human influenza: a review of volunteer challenge studies. Am. J. Epidemiol. 2008;167:775–785. doi: 10.1093/aje/kwm375. [DOI] [PubMed] [Google Scholar]
- 15.Liu TY, et al. An individualized predictor of health and disease using paired reference and target samples. BMC Bioinforma. 2016;17:47. doi: 10.1186/s12859-016-0889-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chang C, Lin C. LIBSVM: a library for support vector machines. ACM Trans. Intell. Syst. Technol. 2013;2:1–39. doi: 10.1145/1961189.1961199. [DOI] [Google Scholar]
- 17.Lopez-Bigas N, De S, Teichmann SA. Functional protein divergence in the evolution of Homo sapiens. Genome Biol. 2008;9:R33. doi: 10.1186/gb-2008-9-2-r33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liberzon A, et al. The molecular signatures database hallmark gene set collection. Cell Syst. 2015;1:417–425. doi: 10.1016/j.cels.2015.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Subramanian A, et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA. 2005;102:15545–15550. doi: 10.1073/pnas.0506580102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tibshirani R. Regression shrinkage and selection via the Lasso. J. R. Stat. Soc. Ser. B. 1996;58:267–288. [Google Scholar]
- 21.Dutra FF, Bozza MT. Heme on innate immunity and inflammation. Front. Pharmacol. 2014;5:115. doi: 10.3389/fphar.2014.00115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ohyama K, Yamauchi S, Endo T, Ohkuma S. Presence of influenza virus-reactive glycophorins other than glycophorin A in human erythrocyte membranes. Biochem. Biophys. Res. Commun. 1991;178:79–84. doi: 10.1016/0006-291X(91)91782-8. [DOI] [PubMed] [Google Scholar]
- 23.Ohyama K, Endo T, Ohkuma S, Yamakawa T. Isolation and influenza virus receptor activity of glycophorins B, C and D from human erythrocyte membranes. Biochim. Biophys. Acta. 1993;1148:133–138. doi: 10.1016/0005-2736(93)90170-5. [DOI] [PubMed] [Google Scholar]
- 24.Saez-Rodriguez J, et al. Crowdsourcing biomedical research: leveraging communities as innovation engines. Nat. Rev. Genet. 2016;17:470–486. doi: 10.1038/nrg.2016.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Whalen S, Pandey OP, Pandey G. Predicting protein function and other biomedical characteristics with heterogeneous ensembles. Methods. 2016;93:92–102. doi: 10.1016/j.ymeth.2015.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Stanescu A, Pandey G. Learning parsimonious ensembles for unbalanced computational genomics problems. Pac. Symp. Biocomput. 2017;22:288–299. doi: 10.1142/9789813207813_0028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sieberts SK, et al. Crowdsourced assessment of common genetic contribution to predicting anti-TNF treatment response in rheumatoid arthritis. Nat. Commun. 2016;7:12460. doi: 10.1038/ncomms12460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ahsen, M. E., Vogel, R. & Stolovitzky, G. Unsupervised evaluation and weighted aggregation of ranked predictions. Preprint at http://arxiv.org/abs/1802.04684) (2018).
- 29.Kuncheva LI, Whitaker CJ. Measures of diversity in classifier ensembles and their relationship with the ensemble accuracy. Mach. Learn. 2003;51:181–207. doi: 10.1023/A:1022859003006. [DOI] [Google Scholar]
- 30.Banoei MM, et al. Plasma metabolomics for the diagnosis and prognosis of H1N1 influenza pneumonia. Crit. Care. 2017;21:97. doi: 10.1186/s13054-017-1672-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Burke TW, et al. Nasopharyngeal protein biomarkers of acute respiratory virus infection. EBioMedicine. 2017;17:172–181. doi: 10.1016/j.ebiom.2017.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McClain MT, et al. Differential evolution of peripheral cytokine levels in symptomatic and asymptomatic responses to experimental influenza virus challenge. Clin. Exp. Immunol. 2016;183:441–451. doi: 10.1111/cei.12736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen M, et al. Predicting viral infection from high-dimensional biomarker trajectories. J. Am. Stat. Assoc. 2011;106:1259–1279. doi: 10.1198/jasa.2011.ap10611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zaas AK, et al. A host-based RT-PCR gene expression signature to identify acute respiratory viral infection. Sci. Transl. Med. 2013;5:203ra126. doi: 10.1126/scitranslmed.3006280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Proud D, et al. Gene expression profiles during in vivo human rhinovirus infection: insights into the host response. Am. J. Respir. Crit. Care Med. 2008;178:962–968. doi: 10.1164/rccm.200805-670OC. [DOI] [PubMed] [Google Scholar]
- 36.Chen B, et al. Bayesian inference of the number of factors in gene-expression analysis: application to human virus challenge studies. BMC Bioinforma. 2010;11:552. doi: 10.1186/1471-2105-11-552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Muller J, et al. Development of an objective gene expression panel as an alternative to self-reported symptom scores in human influenza challenge trials. J. Transl. Med. 2017;15:134. doi: 10.1186/s12967-017-1235-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Davenport EE, Antrobus RD, Lillie PJ, Gilbert S, Knight JC. Transcriptomic profiling facilitates classification of response to influenza challenge. J. Mol. Med. (Berl.). 2015;93:105–114. doi: 10.1007/s00109-014-1212-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Drakesmith H, Prentice A. Viral infection and iron metabolism. Nat. Rev. Microbiol. 2008;6:541–552. doi: 10.1038/nrmicro1930. [DOI] [PubMed] [Google Scholar]
- 40.Weinberg ED. Roles of metallic ions in host-parasite interactions. Bacteriol. Rev. 1966;30:136–151. doi: 10.1128/br.30.1.136-151.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Weinberg ED. Iron and susceptibility to infectious disease. Science (80-.). 1974;184:952–956. doi: 10.1126/science.184.4140.952. [DOI] [PubMed] [Google Scholar]
- 42.Wagener FADTG, et al. Heme is a potent inducer of inflammation in mice and is counteracted by heme oxygenase. Blood. 2001;98:1802–1811. doi: 10.1182/blood.V98.6.1802. [DOI] [PubMed] [Google Scholar]
- 43.Cummins NW, et al. Heme oxygenase-1 regulates the immune response to influenza virus infection and vaccination in aged mice. Faseb J. 2012;26:2911–2918. doi: 10.1096/fj.11-190017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Qi X, et al. Down-regulation of cellular protein heme oxygenase-1 inhibits proliferation of avian influenza virus H9N2 in chicken oviduct epithelial cells. J. Gen. Virol. 2018;99:36–43. doi: 10.1099/jgv.0.000986. [DOI] [PubMed] [Google Scholar]
- 45.Abraham NG, et al. Transfection of the human heme oxygenase gene into rabbit coronary microvessel endothelial cells: protective effect against heme and hemoglobin toxicity. Proc. Natl Acad. Sci. USA. 1995;92:6798–6802. doi: 10.1073/pnas.92.15.6798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.HIPC-CHI Signatures Project Team & HIPC-I Consortium. Multicohort analysis reveals baseline transcriptional predictors of influenza vaccination responses. Sci. Immunol. 2, eaal4656 (2017).. [DOI] [PMC free article] [PubMed]
- 47.Bilal E, et al. Improving breast cancer survival analysis through competition-based multidimensional modeling. PLoS Comput. Biol. 2013;9:e1003047. doi: 10.1371/journal.pcbi.1003047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang M, Zhao Y, Zhang B. Efficient test and visualization of multi-set intersections. Sci. Rep. 2015;5:16923. doi: 10.1038/srep16923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Benjamin DJ, et al. Redefine statistical significance. Nat. Hum. Behav. 2018;2:6–10. doi: 10.1038/s41562-017-0189-z. [DOI] [PubMed] [Google Scholar]
- 50.Wilkinson B. A statistical consideration in psychological research. Psychol. Bull. 1951;48:156–158. doi: 10.1037/h0059111. [DOI] [PubMed] [Google Scholar]
- 51.Warde-Farley D, et al. The GeneMANIA prediction server: biological network integration for gene prioritization and predicting gene function. Nucleic Acids Res. 2010;38:W214–W220. doi: 10.1093/nar/gkq537. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Description of Additional Supplementary Files
Data Availability Statement
Data are available through GEO GSE73072. Challenge results and methods and code for individual models are available through www.synapse.org/ViralChallenge. The authors declare that all other data supporting the findings of this study are available within the article and its Supplementary Information files, or are available from the authors upon request.