Abstract
There are few reports on enterostomy closure (EC) timing for acute abdomen in extremely low birth weight (ELBW) patients. We retrospectively reviewed ELBW patients who underwent enterostomy formation (EF) and subsequent EC. We investigated baseline characteristics, surgical outcomes, and follow-up data of 55 patients and analyzed optimal timing by age at EC, enterostomy duration, and body weight (Bwt) at EC. The minimum p-value approach (MPA) using the Chi-squared test was used to determine each cut-off value. Mean gestational age was 25+3 weeks, while mean age and Bwt at EF were 10 days and 660 g. Enterostomy duration and Bwt at EC were 102 days and 2400 g. Fourteen surgical complications were related to EC. The MPA identified a cut-off of 2100 g (p = 0.039) at EC but no significant cut-off age or enterostomy duration. The 18 patients <2100 g had more enterostomy-related problems at EC than the >2100 g group (66.7% vs 10.8%, p < 0.001). No other characteristics were significantly different. Operation time, ventilator period, hospital stay, parenteral nutrition duration, and full feeding day were significantly longer in <2100 g patients. Follow-up Bwt did not differ (11.55 kg vs 13.95 kg, p = 0.324). Our findings suggest EC can be safely performed when Bwt is over 2100 g.
Introduction
The development of perinatal management has made it possible for low birth weight (LBW) infants to survive. Accordingly, abdominal operations have increased due to an increased incidence of acute abdomen conditions such as necrotizing enterocolitis (NEC) and spontaneous intestinal perforation (SIP) in these babies1,2. Treatment in cases of acute abdomen can be medical or surgical according to disease severity or patient condition. Surgical options include primary closure, primary anastomosis with intestinal resection, enterostomy with or without intestinal resection, and peritoneal drainage. Although controversy exists about what is the best treatment for each patient, enterostomy is a generally accepted safe treatment option for LBW patients with acute abdomen3–6.
There are a few studies of the timing of enterostomy closure (EC), but patients, variables, and criteria of each study for identifying the optimal timing of EC are diverse. Lee J. et al. showed body weight (Bwt) at EC is a significant factor for the development of complications after EC in patients with NEC7, while Kang M. et al. showed a similar result using the corrected age at EC for preterm patients with NEC8. Other reports used enterostomy duration to determine timing, but the criteria (4–10 weeks) and patient groups (NEC or acute abdomen patients) varied9–11.
The incidence of extremely LBW (ELBW) is increasing in Korea (Fig. 1). Some studies have included preterm or LBW patients, but few reports have included only ELBW patients7,10–16. Thus, it is undetermined whether previously published are applicable to ELBW patients, so we conducted this study to investigate the optimal timing of EC in ELBW patients with acute abdomen.
Figure 1.
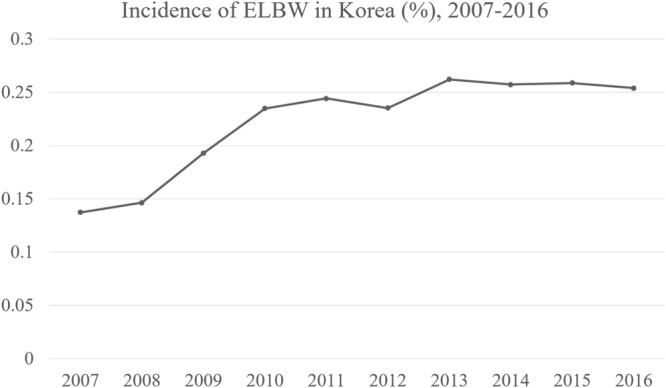
Incidence of extremely low birth weight (ELBW) infants in Korea (total ELBW newborns/total newborns yearly), 2007–2016, raw data from Statistics Korea.
Methods
This retrospective single center study was conducted at Seoul National University Children’s Hospital in South Korea. We reviewed the data of ELBW infants who underwent laparotomy for acute abdomen, including NEC, SIP, meconium-related ileus (MRI), and meconium non-related ileus (MNRI) between January 2003 and April 2016. Children who weighed >1000 g at the time of the first ileostomy formation were excluded. Patients who did not undergo EC were also excluded. Patients’ characteristics, mode of delivery, cause of preterm birth, multiple pregnancy, Apgar score, preterm complications, enterostomy formation (EF)- and closure-related characteristics, EC outcomes, and follow-up data were investigated. This study was approved by the institutional review board of Seoul National University Hospital. We were waived the informed consent by the institutional review board because of the nature of this retrospective study. All methods used in this study were performed in accordance with the relevant guidelines and regulations.
The clinical criteria for closing enterostomy in our institution is body weight for patients otherwise stable without high output enterstomy or prolapse. 2.0 kg of body weight was considered as safe for performing enterostomy closure. Other procedures such as drainage or segmental resection were considered for SIP, but enterostomy formation was procedure of choice in our institution for SIP occurring in ELBW infants.
Acute abdomen was classified into four categories according to the intraoperative findings and pathologic results: NEC, SIP, MRI, or MNRI. Midgut volvulus and congenital anomaly such as intestinal atresia were not considered to enroll in this study. NEC is defined as necrosis of the mucosal and submucosal layers of the bowel, while SIP is an isolated perforation of the bowel with no obvious inflammation in the rest of the bowel17,18. MRI is characterized by a functional ileus caused by impaired meconium excretion not associated with cystic fibrosis and is usually diagnosed with a dilated proximal ileum filled with sticky meconium19, whereas MNRI is an ileus without a definite sticky meconium.
Z-score is the number of standard deviations from the mean of the general population. The World Health Organization AnthroPlus program was used to calculate z-score of Bwt and height for postmenstrual age >40-week patients and PediTools (http://peditools.org/fenton2013) for <40-week patients. Total parenteral nutrition (TPN)–related cholestasis was defined as a direct bilirubin level >2 mg/dL with TPN use. Patients with an already elevated direct bilirubin level before TPN use were excluded. In cases of double-barrel enterostomy, the proximal and distal bowel may differ. Enterostomy type was defined as proximal bowel regardless of distal bowel. Three routine blood tests (white blood cells, hemoglobin [Hb], and albumin) performed within 2 weeks prior to the operation were used to assess the nutritional status of ELBW patients at the time of EC. The full enteral feeding day was defined as the day the patient reached a feeding amount of 100 mL/kg/day. Follow-up Bwt is the last Bwt measurement at the outpatient department (OPD).
The minimum p-value approach (MPA) was used to determine the optimal cut-off for several variables including age, corrected age, Bwt at EC, and enterostomy duration. Chi-squared tests were used to test for significant intergroup differences (low vs high) for EC-related complications at each threshold. The optimal cut-off was chosen as the value that yielded the maximum Chi-squared or minimal p-value on the Chi-squared test. The p-value for the optimal cut-off point by MPA was corrected using the two-fold cross-validation technique to control type I errors.
SPSS version 23 (IBM Corporation, Armonk, NY, USA) was used for the statistical analysis. All continuous variables are presented as median values and range. The Mann-Whitney U test was used to examine continuous data, while the Chi-squared test was used to analyze categorical data. Multivariable logistic regression was performed to adjust for confounding covariates and to examine the result of MPA method on the complication, which included covariates of p-value < 0.05 in difference between groups. The comparison between pre-closure and follow-up Bwt z-score was performed using a paired t-test. P-values < 0.05 were considered statistically significant.
Results
A total of 801 ELBW infants were admitted during study duration; of them, 728 who did not undergo laparotomy for acute abdomen were excluded, 10 were excluded due to not meeting Bwt criteria at EC (<1000 g), seven died before the EC, and one who underwent peritoneal drainage was also excluded. Thus, a total of 55 ELBW patients were analyzed in this study (Fig. 2). The characteristics of 7 patients who died before EC were shown in Table 1.
Figure 2.
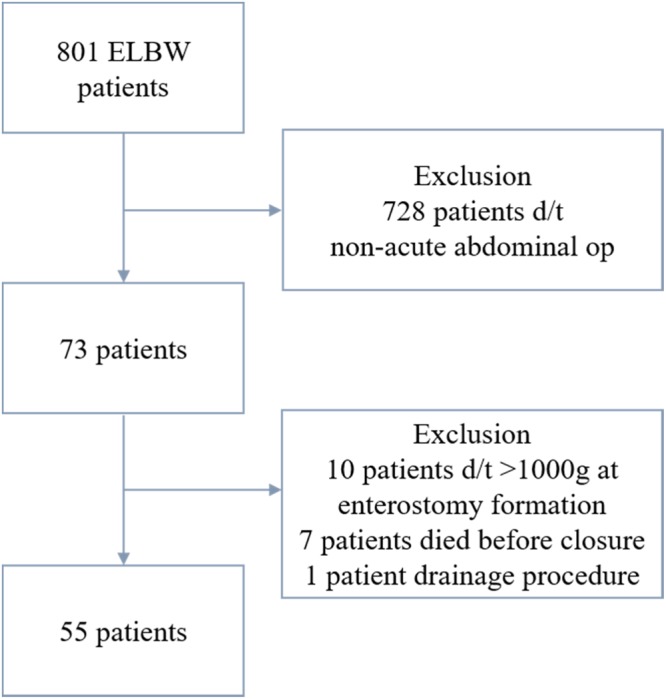
Flow chart of patient inclusion process.
Table 1.
Patients who underwent enterostomy and died before enterostomy closure.
| Case | Sex | GA (week + day) | Birth weight (g) | Age at op (day) | Body weight at op (g) | Dx | Op | Cause of death | Periods from op to death (day) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 25 + 1 | 560 | 24 | 570 | NEC | Jejunostomy | Sepsis | 1 |
| 2 | M | 23 + 1 | 540 | 10 | 580 | NEC | Ileostomy | Intraop cardiac arrest | 0 |
| 3 | F | 22 + 3 | 480 | 17 | 490 | NEC | Ileostomy | Adrenal insufficiency | 14 |
| 4 | M | 27 + 2 | 610 | 34 | 870 | NEC | SB resection and ileostomy | Respiratory failure due to BPD | 128 |
| 5 | M | 30 + 4 | 770 | 9 | 850 | MRI | Jejunostomy and ileostomy | Sepsis | 28 |
| 6 | M | 27 + 1 | 670 | 15 | 700 | NEC | SB resection and jejunostomy | Sepsis | 48 |
| 7 | F | 23 + 2 | 440 | 29 | 640 | SIP | Jejunostomy | Sepsis | 21 |
GA: Gestational age, Op: Operation, Dx: Diagnosis, F: Female, M: Male, NEC: Necrotizing enterocolitis, MRI: Meconium related ileus, SIP: Spontaneous intestinal perforation, BPD: Bronchopulmonary dysplasia.
Patient characteristics and outcomes
Thirty-seven of our patients were male. Median GA was 25+3 weeks (range, 23+1 to 32+1 weeks). Median birth weight was 710 g (range, 430–990 g). A total of 19, 17, 14, and five patients had NEC, SIP, MRI, and MNRI, respectively. A total of 37 patients were born by Cesarean section (C/S). Causes of preterm birth included premature rupture of membrane in 17 cases, spontaneous labor in 12 cases, and labor induction or C/S for fetal (n = 17) or maternal (n = 9) indications. The other characteristics are shown in Table 2.
Table 2.
Patients’ characteristics.
| N = 55 | |
|---|---|
| Male | 37 |
| Gestational age (w) | 25 + 3 (23 + 1–32 + 1) |
| Birth weight (g) | 710 (430–990) |
| z-score | −0.49 |
| Small for gestational age | 20 |
| Diagnosis | |
| NEC | 19 |
| SIP | 17 |
| MRI | 14 |
| MNRI | 5 |
| Mode of delivery | |
| Cesarean section (C/S) | 37 |
| Vaginal delivery | 18 |
| Cause of preterm birth | |
| PPROM | 17 |
| Spontaneous labor | 12 |
| Labor induction or C/S | |
| Fetal indications | 17 |
| Maternal indications | 9 |
| Multiple pregnancy | |
| No | 28 |
| Twin | 22 |
| Triple | 5 |
| Apgar score | |
| 1 min | 2 (0–7) |
| 5 min | 5 (0–8) |
| Preterm complications | |
| RDS | 45 |
| PDA | 50 |
| Ligation operation | 25 |
| ROP | 32 |
| Brain lesion | |
| Hemorrhage | 29 |
| PVE | 19 |
| Normal | 7 |
| Mechanical ventilator after birth (d) | 31 (0–385) |
| Maternal age at delivery (y) | 32 (24–43) |
NEC: necrotizing enterocolitis, SIP: spontaneous intestinal perforation, MRI: meconium-related ileus, MNRI: meconium non-related ileus, PPROM: preterm premature rupture of membrane, RDS: respiratory distress syndrome, PDA: patent ductus arteriosus, ROP: retinopathy of prematurity, PVE: periventricular enlargement.
Median age at EF was 10 days (range, 2–43 days). Median Bwt at EF was 660 g (range, 430–940 g). A total of 19 patients had TPN-related cholestasis before EC. 15 patients underwent 2 or more abdominal operations before EC. A total of 47 patients underwent ileostomy, while eight underwent jejunostomy. Median age at EC was 117 days (range, 65–984 days). Median enterostomy duration was 102 days (range, 38–965 days). Median Bwt at EC was 2400 g (range, 1370–12050 g). Sixteen patients had stoma-related problems at EC. The other EF and EC characteristics are shown in Table 3.
Table 3.
Clinical characteristics with enterostomy formation and closure.
| N = 55 | |
|---|---|
| Enterostomy formation | |
| Age (d) | 10 (2–43) |
| Body weight (g) | 660 (430–940) |
| TPN-related cholestasis before EC | |
| Yes | 19 |
| No | 32 |
| Unknown | 4 |
| Multiple abdominal op before EC | 15* |
| PN stopped before EC | 51 |
| Enterostomy refeeding | 10 |
| Type of enterostomy | |
| Ileostomy | 47 |
| Jejunostomy | 8 |
| Enterostomy closure | |
| Age (d) | 117 (65–984) |
| Corrected age (d) | 15 (−32–882) |
| Enterostomy duration (d) | 102 (38–965) |
| Body weight (g) | 2400 (1370–12050) |
| z-score | −2.86 (−5.72–0.02) |
| Height (cm) | 44.0 (38–87.6) |
| z-score | −3.49 (−8.39–0.6) |
| Enterostomy-related problem at closure | 16† |
| Same hospitalization with EF | 36 |
| Lab | |
| WBC (103/μL) | 10.27 (5.03–22.25) |
| Hb (g/dL) | 11.1 (8.0–14.5) |
| Albumin (g/dL) | 3.7 (2.4–4.6) |
EF: enterostomy formation, EC: enterostomy closure, TPN: total parenteral nutrition, PN: parenteral nutrition, WBC: white blood cell, Hb: hemoglobin.
*Multiple abdominal op: 7 for complications after enterostomy, 3 for correction after primary closure or anastomosis, 1 for definite op after drainage procedure, 1 for 2nd look op, 1 for intestinal biopsy d/t persistent delayed evacuation for contrast study, 1 for perforation during contrast study, 1 for transformation from jejunostomy to ileostomy.
†Enterostomy -related problem: 6 for high output enterostomy, 3 for enterostomy prolapse, 3 for failure to thrive, 1 for both high output and prolapse, 1 for accidental, 1 for peristoma skin problem, 1 for parents’ require.
Median operation time was 75 minutes (range, 25–155 minutes). Twelve patients received a transfusion at EC. Median hospital stay, EN start day, and full enteral feeding days were 18, 6.5, and 9 days, respectively. A total of 14 complications occurred: ileus (n = 7), wound complications (n = 3), incisional hernia (n = 3), and fluid collection identified with sonography (n = 1). Three cases of incisional hernia and two cases of ileus required reoperation. The median follow-up period was 1883 days (range, 42–4992 days). The median follow-up Bwt was 12.7 kg (range, 2.7–25.6 kg). Two deaths occurred. The other outcome and follow-up data are shown in Table 4.
Table 4.
Outcome of enterostomy closure.
| N = 55 | |
|---|---|
| Enterostomy closure outcome | |
| Op time | 75 (25–155) |
| EBL | 0 (0–130) |
| Intraop transfusion | 12 |
| Transfusion within 3 days | 29 |
| Mechanical ventilator (d) | 0 (0–77) |
| Hospital stay (d) | 18 (6–345) |
| PN duration (d) | 8 (0–38) |
| EN start (d) | 6.5 (4–26) |
| Full enteral feeding day | 9 (5–37) |
| Complication | 14 |
| Postop ileus | 7 |
| Wound complication | 3 |
| Incisional hernia | 3 |
| Fluid collection | 1 |
| Reoperation | 5 |
| Incisional hernia | 3 |
| Postop ileus | 2 |
| Follow up | |
| Follow up period (d) | 1883 (42–4992) |
| Corrected age at anthropometric measurement (d) | 1247 (42–3649) |
| Body weight (kg) | 12.7 (2.7–25.6) |
| z-score | −1.52 (−4.88–0.84) |
| Height (cm) | 95.05 (47.3–134.1) |
| z-score | −1.16 (−6.82–1.52) |
| Death | 2 |
EBL: estimated blood loss, PN: parenteral nutrition, EN: enteral nutrition.
MPA analysis
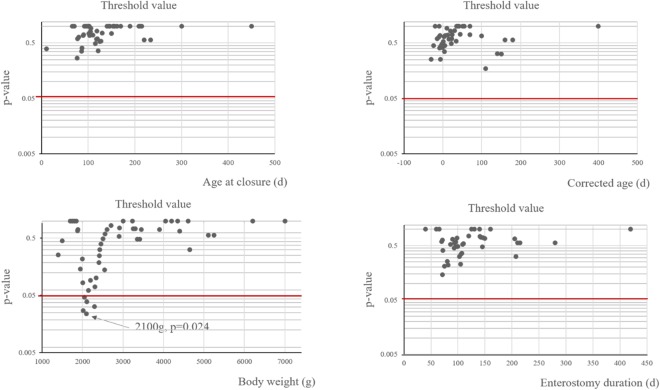
We analyzed the optimal timing of EC with respect to Bwt at EC, age at EC, corrected age at EC, and enterostomy duration. As previously explained, we performed MPA using the Chi-squared test (Fig. 3).
Figure 3.
Results of minimum p-value analysis with respect to age, corrected age, body weight at enterostomy closure (EC), and enterostomy duration for EC-related complication. Only body weight at EC showed significant cut-off values. Among them, 2100 g yielded the minimum p-value in the Chi-squared test.
There were no significant cut-off values for age at EC, corrected age at EC, or enterostomy duration. In fact, there was a significant cut-off value only in Bwt at EC. Among several values showing significance, a Bwt of 2100 g was the value showing minimum p-value (p = 0.024). That is, the complication rate was different most significantly at the value of 2100 g at EC. This result was still significant after use of the two-fold cross-validation technique to control type I error (p = 0.039).
Comparison of patients according to MPA analysis
We divided the patients into two groups with the cut-off value of 2100 g. Group 1 (n = 18) included patients with a Bwt < 2100 g at EC, while group 2 (n = 37) included those with a Bwt > 2100 g at EC. Detailed comparisons are shown in Table 5.
Table 5.
Comparison according to body weight.
| <2100 g (N = 18) | ≥2100 g (N = 37) | p-value | |
|---|---|---|---|
| Patients’ characteristics | |||
| Male | 12 (66.7%) | 25 (67.6%) | 0.947 |
| Gestational age (w) | 27 + 0 | 25 + 3 | 0.07 |
| Birth weight (g) | 650 | 710 | 0.212 |
| z-score | −1.17 | −0.36 | 0.074 |
| Small for gestational age | 9 (50.0%) | 11 (29.7%) | 0.143 |
| Diagnosis | 0.884 | ||
| NEC | 7 (38.9%) | 12 (32.4%) | |
| SIP | 5 (27.8%) | 12 (32.4%) | |
| MRI | 5 (27.8%) | 9 (24.3%) | |
| MNRI | 1 (5.6%) | 4 (10.8%) | |
| C/S | 15 (83.3%) | 22 (59.4%) | 0.077 |
| Cause of preterm birth | 0.130 | ||
| Maternal factor | 10 (55.6%) | 28 (75.7%) | |
| Fetal factor | 8 (44.4%) | 9 (24.3%) | |
| Multiple pregnancy | 8 (44.4%) | 19 (51.4%) | 0.631 |
| Apgar score | |||
| 1 min | 3 | 2 | 0.222 |
| 5 min | 6 | 5 | 0.151 |
| RDS | 14 (77.8%) | 31 (83.8%) | 0.713 |
| PDA operation | 7 (38.9%) | 18 (48.6%) | 0.495 |
| ROP | 9 (50.0%) | 23 (62.2%) | 0.391 |
| Brain hemorrhage | 10 (55.6%) | 19 (51.4%) | 0.769 |
| Mechanical ventilator after birth (d) | 10.5 | 39 | 0.175 |
| Maternal age at delivery (y) | 33 | 31 | 0.076 |
| Enterostomy formation | |||
| Age (d) | 8.5 | 10 | 0.699 |
| Body weight (g) | 660 | 700 | 0.740 |
| Multiple op before closure | 6 (33.3%) | 9 (24.3%) | 0.481 |
| PN stopped before closure | 15 (83.3%) | 36 (97.3%) | 0.061 |
| TPN related cholestasis | 7 (41.2%) | 12 (35.3%) | 0.682 |
| Enterostomy refeeding | 2 (11.1%) | 8 (21.6%) | 0.470 |
| Type of enterostomy | 0.416 | ||
| Ileostomy | 14 (77.8%) | 33 (89.2%) | |
| Jejunostomy | 4 (22.2%) | 4 (10.8%) | |
| Enterostomy closure | |||
| Age (d) | 96 | 136 | <0.001 |
| Corrected age (d) | −4 | 34 | <0.001 |
| Enterostomy duration (d) | 76.5 | 123 | <0.001 |
| Body weight (g) | 1875 | 2920 | <0.001 |
| z-score | −3.49 | −2.61 | 0.002 |
| Height (cm) | 40.75 | 46.8 | <0.001 |
| z-score | −4.38 | −2.94 | 0.014 |
| Enterostomy-related problem at closure | 12 (66.7%) | 4 (10.8%) | <0.001 |
| Same hospitalization with EF | 18 (100%) | 18 (48.6%) | <0.001 |
| Lab | |||
| WBC (103/μL) | 11.51 | 10.12 | 0.101 |
| Hb (g/dL) | 10.1 | 11.4 | 0.006 |
| Albumin (g/dL) | 3.3 | 3.8 | 0.002 |
| Enterostomy closure outcome | |||
| Op time (min) | 88 | 69 | 0.028 |
| EBL (cc) | 0 | 0 | 0.809 |
| Intraop transfusion | 6 (33.3%) | 6 (16.2%) | 0.149 |
| Transfusion within 3 days | 13 (72.2%) | 16 (43.2%) | 0.043 |
| Ventilator period | 1 | 0 | 0.048 |
| Hospital stay | 26.5 | 13 | <0.001 |
| PN duration | 12.5 | 7 | <0.001 |
| EN start day | 7.5 | 6 | 0.02 |
| Full feeding day | 11.5 | 9 | 0.007 |
| Complication | 8 (44.4%) | 6 (16.2%) | 0.024 |
| Reoperation | 3 (16.7%) | 2 (5.4%) | 0.317 |
| Follow up | |||
| Follow up period (d) | 1919 | 1796 | 0.733 |
| Corrected age at anthropometric measurement (d) | 1439 | 1144 | 0.774 |
| Body weight (kg) | 11.55 | 13.95 | 0.324 |
| z-score | −1.76 | −1.27 | 0.132 |
| Height (cm) | 94.25 | 95.25 | 0.582 |
| z-score | −1.28 | −0.98 | 0.179 |
| Death | 0 | 2 | >0.99 |
NEC: necrotizing enterocolitis, SIP: spontaneous intestinal perforation, MRI: meconium-related ileus, MNRI: meconium non-related ileus, RDS: respiratory distress syndrome, PDA: patent ductus arteriosus, ROP: retinopathy of prematurity, PN: parenteral nutrition, TPN: total parenteral nutrition, EF: enterostomy formation, WBC: white blood cell, Hb: hemoglobin, EBL: estimated blood loss, EN: enteral nutrition.
There were no significant differences in any demographic variables except enterostomy-related problems at EC (66.7% in group 1 vs 10.8% in group 2) (p < 0.001).
As shown above the MPA, p-value for complication was 0.024. Other EC outcomes also showed significant intergroup differences. Operation time, mechanical ventilator period, and hospital stay after EC were significantly longer in group 1 (p = 0.028, 0.048, <0.001, respectively). Nutritional factors like PN duration and full feeding day also showed significant differences. More patients in group 1 than in group 2 required transfusion within 3 days after EC (72.2% vs 43.2%, p = 0.043).
The follow-up period did not differ significantly between groups (1919 days vs 1796 days, p = 0.733). Median Bwt and z-score were slightly lower in group 1 than in group 2 (11.55 vs 13.95 kg and −1.76 vs −1.27, respectively), but the difference was not statistically significant (p = 0.324, 0.132).
Multivariable Analysis
Logistic regression test was performed with covariates in Table 5 excluding EC outcome and follow up variables. Age, enterostomy duration, enterostomy-related problems at closure were excluded for covariates due to high correlation with body weight at EC. Body weight group according to MPA method, Hb, and albumin at EC were included. After adjusting potential confounding factors, body weight group was still statistically significant (odds ratios (OR) = 5.124, 95% confidence interval (CI) 1.076–24.390, p = 0.040) (Table 6).
Table 6.
Multivariable Analysis.
| Odds ratio | 95% confidence interval | p-value | |
|---|---|---|---|
| Body weight group (<2100 g, >2100 g) | 5.124 | 1.076–24.390 | 0.04 |
| Hb | 1.394 | 0.857–2.269 | 0.180 |
| Albumin | 0.545 | 0.112–2.643 | 0.451 |
Discussion
Several studies have reported on EC timing in various patient groups. However, it is difficult to apply those results to ELBW patients due to the wide distribution of patient age and Bwt. The present study included only ELBW infants weighing <1000 g who underwent EF to control patients’ characteristics. Thus, this report may help clinicians decide the timing of EC in ELBW patients.
Although many studies have examined EC timing, it is still difficult to make a definite conclusion about when to perform EC. One report analyzed 89 patients who were <6 months of age at the time of EC13. The patients were stratified into four groups based on their Bwt at EC (<2 kg, 2–2.5 kg, 2.5–3.5 kg, and ≥3.5 kg). There was no significant intergroup difference in major morbidities, and only incisional hernia was significantly different in the <2 kg group. However, in that study, patients were arbitrarily stratified by age as older or younger than 6 months. Veenstra M et al. analyzed the patients who underwent EC for NEC by dividing them into enterostomy duration of 8 weeks, 8–12 weeks, or 12 weeks16. They reported no significant differences in PN duration, ventilator period, complication, and mortality. However, Banerjee DB et al. reported that NEC patients who underwent earlier EC with an enterostomy duration <10 weeks had a higher morbidity rate than those who underwent EC later with an enterostomy duration >10 weeks10. Bethell G et al. divided patients according to complications or no complications14 and reported that 24 patients with complications weighed less than 34 patients without complications (3655 g vs 5185 g, respectively). Older for GA and older corrected age at EF was seen in the no complications group (35 weeks vs 28.5 weeks, p = 0.01; and 36.3 weeks vs 32.6 weeks, p = 0.011).
In our study, we found no significant age-based factors related to complications. In contrast, a study of patients who were <37 weeks of GA in 20138 showed that corrected age was a significant factor for complications. They divided patients into groups younger than or older than 40 weeks. The method differed from that of our study, making comparisons difficult; however, we found it interesting that there was a significant difference in complications by age.
The median values for the characteristics of our patients are as follows: GA, 25+3 weeks; birth weight, 710 g; age at EF, 10 days; Bwt at EF, 660 g; age at EC, 117 days; Bwt at EC, 2400 g; and postmenstrual age at EC, 42 weeks. The postoperative hospital stay was 18 days; 25.5% of the patients had complications. At the time of OPD, the patients were a mean 3.4 years of age, had a mean height of 95.5 cm, and had a mean weight of 12.7 kg (z-scores, −1.16 and −1.52, respectively). The z-score of birth weight was −0.49, which was not so small considering GA. However, it decreased to −2.86 at EC and increased to −1.52 at OPD. Long-term follow-up anthropometric results of ELBW infants excluding those with NEC, syndromes, or disabilities were published in 201720. The z-score of Bwt was −0.88 at birth, −2.16 at 40 weeks of corrected age, and −1.29 at 4 years of age. A direct comparison with our study was difficult, but the z-score of Bwt for our patients was higher at birth and slightly lower at similar follow-up times. There might be a growth retardation in relation to acute abdomen or EF. The difference in z-score of Bwt between the pre-closure and follow-up times was statistically significant in our study (Table 7).
Table 7.
Growth difference after enterostomy closure.
| Pre-closure | Follow up | p-value | |
|---|---|---|---|
| Mean z-score | −2.850 | −1.549 | <0.001 |
| <5 percentile | 45 | 25 | 0.015 |
| >5 percentile | 10 | 30 |
The C/S rate (performed for maternal or female indications) was nearly twice as high as that of vaginal delivery. Although there is inconsistent conclusion whether delivery method is a risk factor for NEC or SIP21,22, several articles reported that delivery method affects the intestinal microbiome; thus, it may be a factor in the development of NEC23,24. The numbers of singleton and multiple pregnancies were similar between groups. The proportion of ELBW infants in multiple pregnancies was high considering the fact that only 3.9% of all newborns were born as multiples according to a 2016 survey released by Statistics Korea.
Fifteen patients underwent more than two operations before EC: seven due to stenosis or perforation, three after primary closure or anastomosis due to perforation, one who underwent jejunostomy and then ileostomy, one for a perforation during loopogram, one for a second-look operation, one for a for definite operation after a drainage procedure, and one for intestinal biopsy due to a consistently delayed evacuation. Ganglion cells were present at the sigmoid colon and appendix. Eighteen months after biopsy, EC was performed safely and there were no significant evacuation delays. Sixteen patients (29.1%) had enterostomy-related problems at EC: six with high output enterostomy, three with enterostomy prolapse, three with failure-to-thrive, one with high output and prolapse, one with skin problems around the enterostomy, one at parent request, and one due to an accident. One patient had combined high output and enterostomy prolapse. The accidental case consisted of bowel perforation during the loopogram and was closed immediately. One emergency EC was performed due to rapidly progressing enterostomy prolapse.
Complications occurred in 14 of 55 patients (25.5%). The incidence of postoperative complications after EC in infants was reportedly 24–68%25,26. All three patients with incisional hernia underwent closure operations. The postoperative ileus patient who underwent reoperation was the same patient above who underwent an emergent operation due to perforation during the loopogram. The second reoperation patient with ileus vomited 5 weeks after EC. The patient did not improve with 10 days of conservative care, so the reoperation was performed. Two patients died during follow-up of causes unrelated to surgery. One patient was discharged due to viral pneumonia and died 2 weeks later due to ongoing dyspnea. One patient died of idiopathic dyspnea.
The MPA used in this study to determine the cut-off value differs from the receiver operating characteristic (ROC) curve, which examines test accuracy based on sensitivity and specificity. MPA considers the number of data differing from ROC. It also implies p-value significance, which is used to identify the optimal cut-off value for predictive factors. One study reported that MPA was a stronger predictor than ROC based on a logistic regression analysis in 2016 for predicting the occurrence of major adverse cardiac events27. In our study, MPA was used to identify the significant factors for complications and the relevant cut-off values. We found no significant cut-off values for age, corrected age, or enterostomy duration. We found significance only in Bwt at EC, and the most significant value was 2100 g. This value was still significant after adjusting Hb and albumin level. Enterostomy-related problem at EC was highly associated with body weight at EC because of tendency of early EC to solve the problem although not reaching the relevant clinical status for EC. This factor was excluded for multivariable analysis for potential multicollinearity.
Although not statistically significant, median GA was 11 days lower in group 2 than in group 1. Birth weight was nearly same between the two groups, so more patients were small for gestational age in group 1 (50%) than in group 2 (29.7%). Groups were divided according to Bwt at EC, so it is natural that most EC-related characteristics were significantly different. Hb and albumin levels differed between the two groups. This finding implied that group 1 patients not only had a lower median Bwt, they had nutrition-related problems. This speculation is supported by the fact that there is a significant Bwt z-score difference at EC but not at birth. Another significant different factor was enterostomy-related problems at EC: Group 1 had more enterostomy-related problem, and 10 of 16 patients with this problem were diagnosed with high output or failure-to-thrive. This may lead to nutrition differences between the two groups. This study analyzed complications to determine the optimal timing and found that other surgical outcomes were also significant. In group 1, operation time was longer, transfusion within 3 days after EC was more common, mechanical ventilator period was longer, PN period was longer, full feeding day was longer, and hospitalization was longer.
The results of the previous studies on EC timing after acute abdomen in infants can be divided into four categories: 1) early closure is needed for some patients; 2) early closure yields fine results; 3) early closure yields bad results compared to late closure; and 4) no significant difference between early and late closure. Rothstein emphasized the need for early closure in patients with diarrhea and dehydration after EF in 198228. In 1987, the results of early closure were reportedly acceptable in some studies11,29,30. However, those studies did not compare early and late closure, nor did they use proper statistical methods. One study of early closure would not be considered early closure in the current study due to differences in definition. Since 2009, one paper each on categories 3) and 4) above has been published. This study yielded results similar to those previously published, i.e. complications and other outcomes were significantly higher in the early versus late closure groups. However, this study is different from the previous studies in that we did not compare the two groups according to values based on our own discretion. This study is the first to find the cut-off value of early and late closure with respect to complications rather than just comparing the two groups. Other outcomes were also significant, showing that the cut-off value was well-determined. One study also suggested the cut-off value for the optimal timing of EC, but the study population and methods were different from our study with significantly different value, 2660 g7.
The main limitation of this study is that it was retrospective and included a small number of patients. Although we performed multivariable analysis on the result of MPA method, there may be still confounding factors affecting the occurrence of complications after EC. Further prospective randomized studies that control for patient characteristics are needed.
Body weight at EC was significant factor for complications in ELBW patients. The determined cut-off value was 2100 g. Other outcomes like operation time, hospital stay, mechanical ventilator period, and PN duration were also lower when EC was performed after the patient reached a Bwt of 2100 g at EC.
Acknowledgements
The authors appreciate the statistical advice received from the Medical Research Collaborating Center at Seoul National University Hospital and Seoul National University College of Medicine.
Author Contributions
H.B. Yang wrote the main manuscript text. C. O.h. and J.K. Youn prepared tables and figures and contributed to critical revision. J.W. Han contributed to acquisition, analysis of data. S.E. Jung and H.Y. Kim contributed to conception and design of the study and critical revision. All authors reviewed the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371:75–84. doi: 10.1016/S0140-6736(08)60074-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Moon JY, Hahn WH, Shim KS, Chang JY, Bae CW. Changes of Maternal Age Distribution in Live Births and Incidence of Low Birth Weight Infants in Advanced Maternal Age Group in Korea. Korean J Perinatol. 2011;22:30–36. [Google Scholar]
- 3.Singh M, Owen A, Gull S, Morabito A, Bianchi A. Surgery for intestinal perforation in preterm neonates: anastomosis vs stoma. J Pediatr Surg. 2006;41:725–729. doi: 10.1016/j.jpedsurg.2005.12.017. [DOI] [PubMed] [Google Scholar]
- 4.Sola JE, Tepas JJ, 3rd, Koniaris LG. Peritoneal drainage versus laparotomy for necrotizing enterocolitis and intestinal perforation: a meta-analysis. J Surg Res. 2010;161:95–100. doi: 10.1016/j.jss.2009.05.007. [DOI] [PubMed] [Google Scholar]
- 5.Moss RL, et al. Laparotomy versus peritoneal drainage for necrotizing enterocolitis and perforation. N Engl J Med. 2006;354:2225–2234. doi: 10.1056/NEJMoa054605. [DOI] [PubMed] [Google Scholar]
- 6.Rees CM, et al. Peritoneal drainage or laparotomy for neonatal bowel perforation? A randomized controlled trial. Ann Surg. 2008;248:44–51. doi: 10.1097/SLA.0b013e318176bf81. [DOI] [PubMed] [Google Scholar]
- 7.Lee J, et al. Enterostomy closure timing for minimizing postoperative complications in premature infants. Pediatrics and neonatology. 2014;55:363–368. doi: 10.1016/j.pedneo.2014.01.001. [DOI] [PubMed] [Google Scholar]
- 8.Kang MJ, Lee J, Kim HS, Ko JS, Park KW. Determining the timing for the enterostomy repair using age-based analysis. Korean J Perinatol. 2013;24:251–258. doi: 10.14734/kjp.2013.24.4.251. [DOI] [Google Scholar]
- 9.Struijs MC, Sloots CE, Hop WC, Tibboel D, Wijnen RM. The timing of ostomy closure in infants with necrotizing enterocolitis: a systematic review. Pediatr Surg Int. 2012;28:667–672. doi: 10.1007/s00383-012-3091-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Banerjee DB, Vithana H, Sharma S, Tsang TTM. Outcome of stoma closure in babies with necrotising enterocolitis: early vs late closure. Pediatr Surg Int. 2017;33:783–786. doi: 10.1007/s00383-017-4084-5. [DOI] [PubMed] [Google Scholar]
- 11.Festen C, Severijnen RS, vd Staak FH. Early closure of enterostomy after exteriorization of the small intestine for abdominal catastrophies. J Pediatr Surg. 1987;22:144–145. doi: 10.1016/S0022-3468(87)80433-5. [DOI] [PubMed] [Google Scholar]
- 12.Mochizuki Kyoko, Hayakawa Masahiro, Urushihara Naoto, Miyake Hiromu, Yokoi Akiko, Shiraishi Jun, Fujinaga Hideshi, Ohashi Kensuke, Esumi Genshiro, Ohfuji Satoko, Amae Shintaro, Yanai Toshihiro, Furukawa Taizo, Tazuke Yuko, Minagawa Kyoko, Okuyama Hiroomi. Timing and outcome of stoma closure in very low birth weight infants with surgical intestinal disorders. Surgery Today. 2017;47(8):1001–1006. doi: 10.1007/s00595-017-1498-6. [DOI] [PubMed] [Google Scholar]
- 13.Talbot LJ, et al. Influence of weight at enterostomy reversal on surgical outcomes in infants after emergent neonatal stoma creation. J Pediatr Surg. 2017;52:35–39. doi: 10.1016/j.jpedsurg.2016.10.015. [DOI] [PubMed] [Google Scholar]
- 14.Bethell G, Kenny S, Corbett H. Enterostomy-related complications and growth following reversal in infants. Arch Dis Child Fetal Neonatal Ed. 2017;102:F230–F234. doi: 10.1136/archdischild-2016-311126. [DOI] [PubMed] [Google Scholar]
- 15.Al-Hudhaif J, Phillips S, Gholum S, Puligandla PP, Flageole H. The timing of enterostomy reversal after necrotizing enterocolitis. J Pediatr Surg. 2009;44:924–927. doi: 10.1016/j.jpedsurg.2009.01.028. [DOI] [PubMed] [Google Scholar]
- 16.Veenstra M, Nagappala K, Danielson L, Klein M. Timing of ostomy reversal in neonates with necrotizing enterocolitis. Eur J Pediatr Surg. 2015;25:231–235. doi: 10.1055/s-0034-1372460. [DOI] [PubMed] [Google Scholar]
- 17.Okuyama H, et al. Risk factors for surgical intestinal disorders in VLBW infants: Case-control study. Pediatr Int. 2016;58:34–39. doi: 10.1111/ped.12815. [DOI] [PubMed] [Google Scholar]
- 18.Hayakawa M, et al. Outcome in VLBW infants with surgical intestinal disorder at 18 months of corrected age. Pediatr Int. 2015;57:633–638. doi: 10.1111/ped.12594. [DOI] [PubMed] [Google Scholar]
- 19.Kubota A, et al. Meconium-related ileus in extremely low-birthweight neonates: etiological considerations from histology and radiology. Pediatr Int. 2011;53:887–891. doi: 10.1111/j.1442-200X.2011.03381.x. [DOI] [PubMed] [Google Scholar]
- 20.Stutte Sonja, Gohlke Bettina, Peiler Annika, Schreiner Felix, Born Mark, Bartmann Peter, Woelfle Joachim. Impact of Early Nutrition on Body Composition in Children Aged 9.5 Years Born with Extremely Low Birth Weight. Nutrients. 2017;9(2):124. doi: 10.3390/nu9020124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Son M, Grobman WA, Miller ES. Is mode of delivery associated with the risk of necrotizing enterocolitis? Am J Obstet Gynecol. 2016;215:389 e381–384. doi: 10.1016/j.ajog.2016.04.058. [DOI] [PubMed] [Google Scholar]
- 22.Shah J, et al. Intestinal perforation in very preterm neonates: risk factors and outcomes. Journal of Perinatology. 2015;35:595–600. doi: 10.1038/jp.2015.41. [DOI] [PubMed] [Google Scholar]
- 23.Goulet O. Potential role of the intestinal microbiota in programming health and disease. Nutr Rev. 2015;73(Suppl 1):32–40. doi: 10.1093/nutrit/nuv039. [DOI] [PubMed] [Google Scholar]
- 24.Collado MC, et al. Factors influencing gastrointestinal tract and microbiota immune interaction in preterm infants. Pediatr Res. 2015;77:726–731. doi: 10.1038/pr.2015.54. [DOI] [PubMed] [Google Scholar]
- 25.O’Connor A, Sawin RS. High morbidity of enterostomy and its closure in premature infants with necrotizing enterocolitis. Arch Surg. 1998;133:875–880. doi: 10.1001/archsurg.133.8.875. [DOI] [PubMed] [Google Scholar]
- 26.Weber TR, Tracy TF, Jr., Silen ML, Powell MA. Enterostomy and its closure in newborns. Arch Surg. 1995;130:534–537. doi: 10.1001/archsurg.1995.01430050084014. [DOI] [PubMed] [Google Scholar]
- 27.Vanniyasingam T, et al. Predicting the occurrence of major adverse cardiac events within 30 days of a vascular surgery: an empirical comparison of the minimum p value method and ROC curve approach using individual patient data meta-analysis. Springerplus. 2016;5:304. doi: 10.1186/s40064-016-1936-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rothstein FC, Halpin TC, Jr., Kliegman RJ, Izant RJ., Jr. Importance of early ileostomy closure to prevent chronic salt and water losses after necrotizing enterocolitis. Pediatrics. 1982;70:249–253. [PubMed] [Google Scholar]
- 29.Gertler JP, Seashore JH, Touloukian RJ. Early ileostomy closure in necrotizing enterocolitis. J Pediatr Surg. 1987;22:140–143. doi: 10.1016/S0022-3468(87)80432-3. [DOI] [PubMed] [Google Scholar]
- 30.Musemeche CA, Kosloske AM, Ricketts RR. Enterostomy in necrotizing enterocolitis: an analysis of techniques and timing of closure. J Pediatr Surg. 1987;22:479–483. doi: 10.1016/S0022-3468(87)80200-2. [DOI] [PubMed] [Google Scholar]