Abstract
The positive correlation between the number of M2 phenotype TAMs (M2-TAMs) and tumour development suggests a supportive role of M2-TAMs in glioma progression. In the present study, the molecular link between glioma cells and M2-TAMs was investigated and it was demonstrated that transforming growth factor-β1 (TGF-β1) secreted by M2-TAMs is key in facilitating the stemness and migration of glioma cells. Cluster of differentiation (CD)133 and CD44, markers for the M2 phenotype, were assessed by western blotting. A sphere formation assay and trans-well assay were applied to test the stemness and migration abilities of glioma cells following co-cultured with M2-TAMs. Stemness markers CD133 and CD44, epithelial-mesenchymal transition-associated markers and mothers against decapentaplegic homolog (SMAD)2/3 and sex determining region Y-box 4/2 (SOX4/2) levels were also evaluated by western blotting. A xenograft tumor mouse model was used to demonstrate the tumor forming ability of glioma cells. The results showed that the U251 glioma cells co-cultured with M2-TAMs exhibited high level of sphere formation, stemness and migration ability. Recombinant TGF-β1 protein treatment was able to achieve the same effects on U251 cells, whereas a TGF-β pathway inhibitor reversed the stemness and migration abilities of the glioma cells induced by M2-TAMs. It was also demonstrated that TGF-β1 secreted by M2-TAMs upregulated the phosphorylation of SMAD2/3 and the expression of SOX4/2 in glioma cells. In a mouse xenograft model, solid tumours formed by U251 cells co-cultured with M2-TAMs or pre-treated with TGF-β1 were larger in size and had a higher growth rate. Taken together, results of the present study demonstrated that M2-TAMs promoted the stemness and migration abilities of glioma cells by secreting TGF-β1, which activated the SMAD2/3 pathway and induced the expression of SOX4 and SOX2. These results highlight the mechanism by which M2-TAMs and glioma interact and demonstrate potential therapeutic strategies for glioma treatment.
Keywords: M2 phenotype tumour-associated macrophages, U251 glioma cell, transforming growth factor-β1, small mothers against decapentaplegic 2/3, sex determining region Y-box 2, stemness and migration
Introduction
Malignant glioma is the most prevalent primary brain tumour in the world, with >350,000 patients diagnosed with glioma worldwide each year (1). Despite the development in glioma therapy, the overall survival rate following the diagnosis of patients remains poor (2,3). In previous years, emerging reports have suggested that the migration and self-renewal abilities of glioma cells contribute substantially to tumour development, including tumour initiation, metastasis, drug resistance and recurrence (4-8). However, the influence of the tumour microenvironment on stemness and the migration of glioma cells remains to be fully elucidated.
Increasing evidence has indicated that the tumour microenvironment is key in tumour development, including glioma (9,10). The tumour microenvironment comprises tumour cells, tumour stroma, blood vessels, infiltrating inflammatory cells and various associated tissue cells. Tumour-infiltrating inflammatory cells are mobilised and recruited by tumour-derived factors, which contribute to the tumour microenvironment. Macrophages are derived from monocytic precursors and can undergo various differentiation or polarization processes in tissues (11,12). Macrophage polarization is the process of expressing different functional programmes in response to microenvironmental signals (13). There are multiple polarization statuses based on functional states that can acquire specific phenotypes (14). Tumour-associated macrophages (TAMs) can be divided into M1 and M2 subtypes, which have different roles in tumours, based on their polarization status (15). Studies have demonstrated that M1 and M2 subtype macrophages exhibit tumour-suppressive and tumour-promoting functions, respectively (16). M2 phenotype TAMs (M2-TAMs) serve as the primary contributors to tumour-infiltrating leukocytes and can be identified by several surface markers, including CD163, CD206, Fizz1 and Arg1 (17,18). M2-TAMs directly facilitate tumour initiation, progression and metastasis through the secretion of proteolytic molecules to promote extracellular matrix remodelling (19) or nonproteolytic proteins to stimulate tumour cell proliferation, migration and invasion (20,21). In addition, M2-TAMs interfere with the antitumour functions of other immune cells (22). Consequently, investigating the specific mechanism underlying the tumour-supportive role of M2-TAMs in glioma progression is essential.
Furthermore, the high activity of the transforming growth factor-β (TGF-β) pathway in human glioma is associated with a poor prognosis (23). M2-TAMs have been demonstrated to contribute to the accumulation of TGF-β1 in glioma tissues (24). The role of TGF-β1 in tumour development, including cell proliferation, invasion, immune suppression and microenvi-ronment modification, has been well researched (24-28). In addition, reports have demonstrated that TGF-β1 can promote the self-renewal ability of glioma cells (29). However, how the TGF-β pathway affects the biological properties of glioma cells, including stemness and migration abilities, remains to be fully elucidated.
The TGF-β pathway is induced by binding to pairs of receptor serine/threonine kinases, known as type I and type II receptors, and then activates and phosphorylates the intracellular effectors mothers against decapentaplegic homolog (SMAD)2/SMAD3, which form a complex with SMAD4 and enters the nucleus for target gene recognition and transcriptional regulation (30,31). Sex-determining region Y-box (SOX) factors are a family of transcriptional regulators that comprise 20 members. SOX2 is important in glioma progression (32). The downregulation of SOX2 via RNA interference in glioma cells impairs their proliferation and tumour formation ability in vivo (33), whereas the ectopic elevation of SOX2 increases cell proliferation and self-renewal activity (33,34). SOX2, mediated by other members of the SOX family, including SOX4, which functions downstream of the TGF-β pathway (26), is one of the crucial factors for the maintenance cancer cell stemness (29). The inhibition of TGF-β has been demonstrated to suppress the expression of SOX4, leading to a decrease in the level of SOX2 and impairment of glioma tumourigenicity (26). However, the effects of M2-TAMs on the expression of SOX family members to mediate stemness and migration abilities in glioma cells remain to be fully elucidated.
The present study aimed to elucidate the effects and specific mechanisms of M2-TAMs on the stemness and migration of glioma cells. It was demonstrated that M2-TAMs induced the stemness and migration abilities of glioma cells via secreting TGF-β1, leading to activation of the SMAD2/3 pathway and the upregulation of SOX4 and SOX2, whereas the TGF-β pathway inhibitor SB431542 was shown to eliminate their interaction. Furthermore, implanted tumours in a mouse model, formed by glioma cells pre-treated with TGF-β1 protein or co-cultured with M2-TAMs, exhibited an increase in tumour size and growth rate compared with those formed by glioma cells exposed to TGF-β inhibitor or no treatment. Taken together, the results provided novel insights and strategies for the treatment of gliomas.
Materials and methods
Cell culture and reagents
The U251 human glioma cell line and the THP-1 human monocytic cell line were obtained from the American Type Culture Collection (Manassas, VA, USA). The U251 cells were cultured in Dulbecco's modified Eagle's medium (DMEM; Thermo Fisher Scientific, Inc., Waltham, MA, USA) with 10% foetal bovine serum (FBS; HyClone, GE Healthcare Life Sciences, Logan, UT, USA) and 1% penicillin/streptomycin (Thermo Fisher Scientific, Inc.). The THP-1 cells were cultured in RPMI-1640 (Thermo Fisher Scientific, Inc.) supplemented with 10% FBS and 1% penicillin/streptomycin. The cells were cultured at 37°C in a humidified incubator with 5% CO2. Recombinant human TGF-β1 protein (cat. no. ab50036) was purchased from Abcam (Cambridge, MA, USA), and the TGF-β inhibitor SB431542 (cat. no. HY-10431) was purchased from Medchem Express (Monmouth Junction, NJ, USA). Phorbol myristate acetate (PMA; cat. no. P1585) was purchased from EMD Millipore (Billerica, MA, USA). Interleukin (IL)-4 (cat. no. ab222347) and IL-13 (cat. no. ab221410) were purchased from Abcam.
Preparation of M2 phenotype TAMs and co-culture
The M2-polarised macrophages were generated as previously described (35). Briefly, the THP-1 cells (1×106 cells/ml) were seeded into the upper insert of a six-well Transwell plate (Corning Inc., Corning, MA, USA) and were treated with 320 nM PMA for 6 h at 37°C, followed by incubation with PMA and IL-4 (20 ng/ml) and IL-13 (20 ng/ml) for an addition 18 h at 37°C. The samples were then washed with PBS to remove all PMA, and the M2-TAMs were co-cultured with U251 cells (2×105 cells per well) without direct contact for 48 h at 37°C. The co-cultured U251 cells were then washed and harvested for subsequent experiments.
ELISA
The supernatants of the THP-1 cells and polarised M2-polarised macrophages were centrifuged at 1,000 × g for 5 min under 4°C prior to ELISA. The levels of TGF-β1 (cat. no. DB-100B), epidermal growth factor (EGF; cat. no. DEG00), and IL-10 (cat. no. D1000B) were measured using commercial ELISA kits (R&D Systems, Inc., Minneapolis, MN, USA) according to the manufacturer's protocol. Each sample was evaluated in triplicate.
Sphere formation assay of U251 cells
The U251 cells with or without co-culture were plated on ultralow attachment plates (Corning Inc.) at a density of 20,000 cells/ml in serum-free DMEM, supplemented with B27 (Invitrogen; Thermo Fisher Scientific, Inc.) and 10 ng/ml EGF (BD Biosciences, San Jose, CA, USA). Images of the spheres were captured using a light microscope (Eclipse; Nikon Corporation, Tokyo, Japan) and the spheres were quantified following 10 days of culture.
Transwell assay
A total of 5×105 U251 cells were suspended in serum-free DMEM and plated into the upper insert of a six-well Transwell plate (Corning Inc.), and serum-containing medium was added to the lower chamber. The cells were incubated at 37°C for 8 h. The non-migratory cells in the upper insert were gently removed using cotton swabs, and the migratory cells were fixed with 4% paraformaldehyde in room temperature for 10 min, followed by staining with crystal violet solution. Images were captured using a light microscope (Eclipse; Nikon Corporation) and quantified by counting cell numbers of five randomly picked fields of view for each well.
Western blot analysis
Western blot analysis was performed as described previously (35). Briefly, the total proteins of U251 cells and M2-TAMs were extracted using RIPA lysis buffer (Abcam) containing the protease inhibitor PMSF (EMD Millipore). The concentration of proteins was determined by Pierce BCA Protein Assay kit according to manufacture's protocol (Thermo Fisher Scientific, Inc.). A total of 20 µg of proteins were subjected to 10% SDS-PAGE and were transferred onto polyvinylidene fluoride membranes (EMD Millipore). The membranes were blocked with 5% milk in room temperature for 1 h and then were incubated with rabbit anti-CD133 (cat. no. 86781), rabbit anti-CD44 (cat. no. 37259, 1:2,000), rabbit anti-CD206 (cat. no. 91992, 1:500), rabbit anti-CD163 (cat. no. 93498), rabbit anti-SOX2 (cat. no. 3579), mouse anti-SMAD2 (cat. no. 3103), rabbit anti-SMAD3 (cat. no. 9523), rabbit anti-p-SMAD2 (cat. no. 18338, 1:500), rabbit anti-p-SMAD3 (cat. no. 9520, 1:500) from Cell Signaling Technology, Inc. (Danvers, MA, USA), and rabbit anti-SOX4 (cat. no. ab86809), mouse anti-vimentin (cat. no. ab8978, 1:2,000), mouse anti-N-cadherin (cat. no. ab98952), rabbit anti-E-cadherin (cat. no. ab40772), rabbit anti-matrix metal-loproteinase (MMP)-2 (cat. no. ab37150), mouse anti-MMP-9 (cat. no. ab58803, 1:3,000) from Abcam, and mouse anti-GAPDH (cat. no. SC-32233; Santa Cruz Biotechnology, Inc., Dallas, TX, USA) antibodies at a dilution of 1:1,000 unless indicated otherwise overnight at 4°C. The membranes were then washed by PBS with 0.05% Tween-20 and incubated with the appropriate HRP-conjugated secondary antibodies (cat. no. 7074 for rabbit and 7076 for mouse) from Cell Signalling Technology, Inc., at a dilution of 1:5,000 for 2 h at room temperature. The proteins were detected using the enhanced chemiluminescence detection reagent (Bio-Rad, Laboratories, Inc., Hercules, CA, USA). GAPDH was used as a normalised control. Relative protein levels were quantified by the comparison of grey values by ImageJ 1.51 (National Institutes of Health, Bethesda, MD, USA).
Tumour implantation
All experimental procedures involving animals were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Xiangya Hospital, Central South University (Changsha, China). In total 40 female BALB/c nude mice of 7-8 weeks old and 16-18 g body weight were purchased from SJA Laboratory Animal Company (Changsha, China) and maintained under 27°C in a HEPA-filtered environment with cages in a 12-h light/dark cycle room, with food and water sterilized by autoclaving. To generate a subcutaneous xenograft mouse model, the mice were injected subcutaneously with 2×106 U251 cells into the right flanks following the indicated treatment for 48 h (n=8 mice per group). The tumour sizes were measured every 5 days. After 30 days, the mice were sacrificed by cervical dislocation, and solid tumours were harvested.
Statistical analysis
All experiments were performed at least three times. The data are expressed as the mean ± standard deviation. The statistical significance between the specific group and control was analysed using SPSS 13.0 statistical software (IBM Corps., Armonk, NY, USA). Statistical evaluation was performed using Student's t-test (two-tailed) between two groups or one-way analysis of variance followed by Tukey's post hoc test for multiple comparisons. P<0.05 was considered to indicate a statistically significant difference.
Results
M2-polarised macrophage identification and detection of TGF-β1 secretion
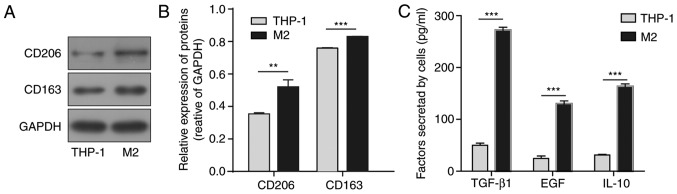
M2-polarised macrophages were obtained from THP-1 human monocyte cell line polarization by PMA, IL-4, and IL-13 treatment. Markers for the M2 phenotype, CD206 and CD163, were assessed by western blot analysis. As presented in Fig. 1A and B, the protein levels of CD206 and CD163 were significantly increased following stimulation compared with the levels in THP-1 cells. The relative growth factors and cytokines secreted from the M2-polarised macrophages were assessed by ELISA. Compared with the original THP-1 cells, M2-polarised macrophages exhibited significant increases in the concentrations of secreted TGF-β1, EGF and IL-10 (Fig. 1C). Due to the crucial role of TGF-β1 in tumour development, TGF-β1 was selected for further investigation.
Figure 1.
Identification of M2-TAMs derived from THP-1 cells and detection of TGF-β1 secretion. (A) Representative western blot images and (B) statistical results of M2-TAM markers CD206 and CD163. GAPDH was used as the control. (C) Secreted cytokines, including TGF-β1, EGF and IL-10, were measured by enzyme-linked immunosorbent assay from M2-TAMs. The results are representative of three independent experiments. The error bars represent the mean + standard deviation. P-values were determined by an unpaired two-tailed t-test. **P<0.01 and ***P<0.001, vs. THP-1 group. M2-TAMs, M2 phenotype tumour-associated macrophages; TGF-β1, transforming growth factor-β1; EGF, epidermal growth factor; IL-10, interleukin-10.
M2-TAMs enhance the stemness and migration abilities of glioma cells
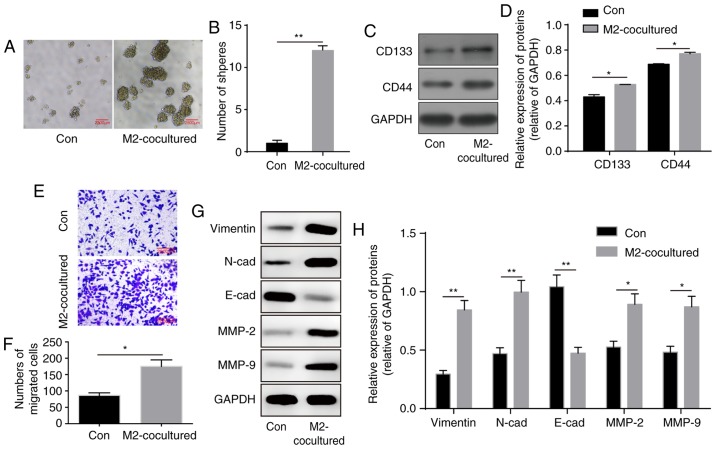
To demonstrate the influence of M2-TAMs on glioma cells, the co-culture system was prepared. Following co-culture for 48 h, the U251 cells were harvested, and tumour formation capacity was assessed by the sphere formation assay. Compared with the U251 cells co-cultured with THP-1 cells, the U251 cells co-cultured with M2-TAMs exhibited a notable increase in sphere formation ability (Fig. 2A and B). Similarly, the expression levels of the stemness markers CD133 and CD44 were significantly enhanced in the U251 cells (Fig. 2C and D), indicating a significant increase in the stemness ability of glioma cells.
Figure 2.
M2-TAMs increase the stemness and migration abilities of glioma cells. (A) Co-culture with M2-TAMs elevated the sphere formation ability of glioma cells. Scale bar=2,500 µm. (B) Results of statistical analysis of the sphere formation ability of glioma cells. (C) Representative western blot images and (D) statistical results of stemness markers CD133 and CD44 following co-culture with M2-TAMs. (E) Migration ability of glioma cells was enhanced following M2-TAM co-culture. Scale bar=2,000 µm. (F) Statistical analysis of migration ability in E. (G) Representative western blot images and (H) results of statistical analysis of EMT-associated proteins in glioma cells following M2-TAM co-culture. The results are representative of three independent experiments. The error bars represent the mean + standard deviation. P-values were determined by an unpaired two-tailed t-test. *P<0.05 and **P<0.01, vs. Con group. M2-TAMs, M2 phenotype tumour-associated macrophages; N-cad, N-cadherin; E-cad, E-cadherin; MMP, matrix metalloproteinase; ns, not significant; Con, control.
The tumour microenvironment is important in facilitating tumour migration. In the Transwell migration assay, co-culture with M2 phenotype macrophages significantly promoted the migration ability of the glioma cells (Fig. 2E and F). Additionally, the protein levels of epithelial-mesen-chymal transition (EMT)-associated markers were altered, with increased levels of mesenchymal markers vimentin, N-cadherin, MMP-2 and MMP-9, and decreased levels of the epithelial marker E-cadherin (Fig. 2G and H). In summary, these results indicated that M2-TAMs facilitated the stemness and migration abilities of the glioma cells.
TGF-β1 derived from M2-TAMs directly increases the stemness and migration abilities of glioma cells
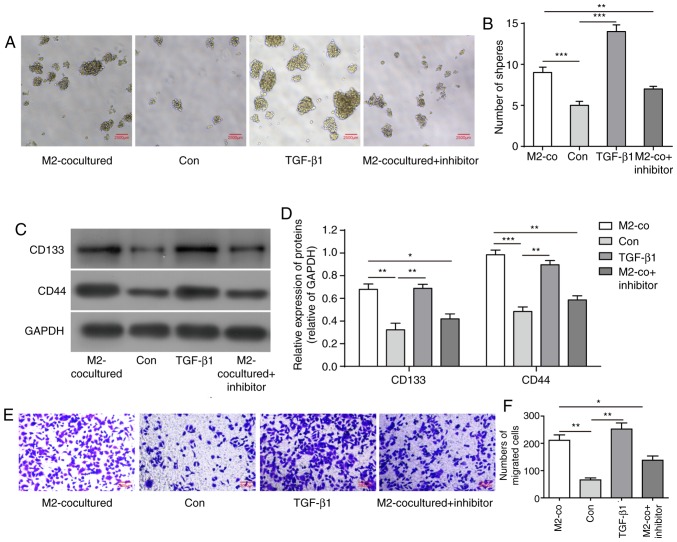
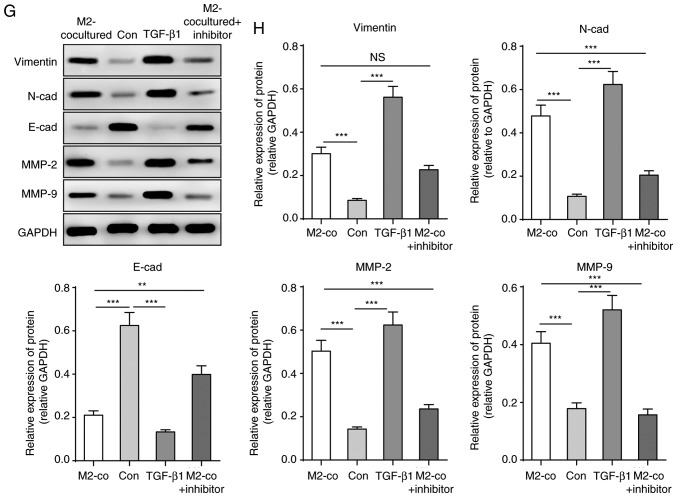
Due to the upregulation of TGF-β1 levels secreted from M2-TAMs and its importance in tumour development, it was hypothesised that M2-TAMs affecting the stemness and migration abilities of glioma cells was achieved by TGF-β1 function. To determine the contribution of TGF-β1 to glioma cells, the U251 cells were treated with human recombinant protein TGF-β1 (200 nM) or TGF-β pathway inhibitor SB431542 (2 µM). Similar to co-culture with M2-TAMs, TGF-β1 treatment increased U251 cell sphere formation and the expression levels of stemness markers CD133 and CD44, whereas the TGF-β inhibitor significantly reversed its effects (Fig. 3A-D). Similarly, TGF-β1 treatment directly elevated the migration ability of the U251 cells and promoted EMT, whereas its inhibitor led to inhibition of these effects (Fig. 3E-H). These results indicated that TGF-β1 derived from M2-TAMs is important in the regulation of the stemness and migration abilities of glioma cells.
Figure 3.
TGF-β1 derived from M2-TAMs directly influence the stemness and migration of glioma cells. (A) Glioma cell sphere formation ability and (B) results of statistical analysis in the indicated groups. Scale bar=2,500 µm. (C) Representative western blot images and (D) statistical results of stemness markers CD133 and CD44 in the indicated groups. (E) Glioma cell migration ability was measured using a Transwell assay in the indicated groups. Scale bar=2,000 µm. (F) Statistical analysis of migration ability in E. (G) Representative western blot images and (H) results of statistical analysis of EMT-associated proteins of glioma cells in the indicated groups. The results are representative of three independent experiments. The error bars represent the mean + standard deviation. P-values were determined by one-way analysis of variance followed by Tukey's post hoc test. *P<0.05, **P<0.01 and ***P<0.001; M2-TAMs, M2 phenotype tumour-associated macrophages; co, co-culture; TGF-β1, transforming growth factor-β1; N-cad, N-cadherin; E-cad, E-cadherin; MMP, matrix metalloproteinase; ns, not significant; Con, control.
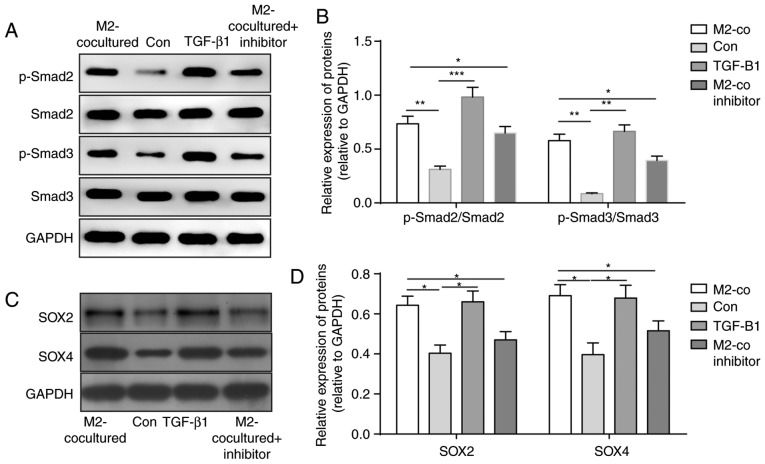
TGF-β1 derived from M2-TAMs induces the stemness and migration abilities of glioma cells via the SMAD2/3 pathway
To determine the mechanism of the increased stemness and migration abilities of glioma cells induced by M2-TAMs, downstream signals of the TGF-β pathway in U251 cells were detected. As shown in Fig. 4A and B, the levels of p-SMAD2 and p-SMAD3 were significantly upregulated following M2 coculture and TGF-β1 treatment, whereas the TGF-β pathway inhibitor eliminated these effects (Fig. 4A and B). As the stemness of glioma cells was increased, the levels of stemness-associated proteins SOX2 and SOX4 were determined and demonstrated to be elevated in the U251 cells in the co-culture system and those treated with TGF-β1 (Fig. 4C and D). These results suggested that TGF-β1 secreted from M2-TAMs activated the TGF-β-SMAD2/3 pathway to induce the expression of SOX4 and SOX2 and promote the stemness and migration abilities of glioma cells.
Figure 4.
TGF-β pathway in glioma cells is activated following M2-TAM stimulation. (A) Representative western blot images and (B) results of statistical analysis of the phosphorylation levels of SMAD2 and SMAD3 following the indicated treatment. (C) Representative western blot images and (D) results of statistical analysis of protein levels of SOX4 and SOX2 following the indicated treatment. The results are representative of three independent experiments. The error bars represent the mean + standard deviation. P-values were determined by one-way analysis of variance followed by Tukey's post hoc test. *P<0.05, **P<0.01 and ***P<0.001. M2-TAMs, M2 phenotype tumour-associated macrophages; Con, control; co, co-culture; TGF-β1, transforming growth factor-β1; SMAD, mothers against decapentaplegic homolog; p-, phosphorylated; SOX, sex determining region Y-box.
M2-TAMs facilitate solid tumour formation in a mouse model
To investigate whether M2-TAMs can affect glioma cell capacity for solid tumour formation in vivo, U251 cells were subcutaneously implanted following different treatments. As shown in Fig. 5A and B, the final tumour sizes formed by cells co-cultured with M2-TAMs or those treated with TGF-β1 were increased compared with those of the control group. Additionally, TGF-β inhibitor treatment decreased the size of the tumours, which were induced by co-culture with M2-TAMs. The growth rate of tumours formed by U251 cells was also accelerated by M2-TAM co-culture or TGF-β1 treatment, and the TGF-β inhibitor reversed these effects. Therefore, these data demonstrated that M2-TAMs facilitated solid tumour formation of glioma cells in vivo through secreting TGF-β1 protein.
Figure 5.
M2-TAMs promote glioma cell tumour formation ability in vivo via secreting TGF-β1. (A) Image of tumours harvested from mice 30 days following xenograft of glioma cells exposed to different treatments. (B) Statistical results of tumour growth formed by glioma cells with the indicated treatment. The results are representative of three independent experiments. The error bars represent the mean ± standard deviation. P-values were determined by one-way analysis of variance followed by Tukey's post hoc test. *P<0.05, **P<0.01 and ***P<0.001, vs. Con. M2 phenotype tumour-associated macrophages; Con, control; co, co-culture; TGF-β1, transforming growth factor-β1.
Discussion
The tumour-associated microenvironment provides cues to cancer cells that regulate their self-renewal and metastatic potential (36,37). The tumour microenvironment features inflammation, which is the major contributing factor to tumour formation and metastasis (38). Among tumour-infiltrating immune cells, TAMs are the major constituents of the inflammation-associated microenvironment in tumours (39). Previous studies have demonstrated a positive correlation between the number of M2-TAMs and glioma development, particularly glioma stem-like cells (21). However, the present study investigated the function and mechanism of M2-TAM regarding the stemness and migration abilities of glioma cells and the associated specific pathways involved in this process. The results of the present study demonstrated that M2-TAMs increased the stemness and migration abilities of glioma cells via secreting TGF-β1, leading to activation of the SMAD2/3 pathway and the upregulation of SOX4 and SOX2. These findings provide novel insights, strategies and therapeutic targets for the treatment of glioma progression.
The initial results of the present study showed that M2-TAMs derived from the THP-1 human monocyte cell line upregulated M2 subtype surface markers CD206 and CD163 (Fig. 1A and B). Compared with THP-1 cells, M2-TAMs exhibited notably higher levels of TGF-β1, EGF and IL-10, indicating the successful transition to M2 subtype macrophages. Subsequently, to determine the cross-talk between M2-TAMs and glioma cells, a co-culture system was established, enabling communication through secreting factors from M2-TAMs. M2-TAMs have been demonstrated to promote cell invasion in pancreatic islets cancer (19) and facilitate the stemness of cancer stem cells in breast cancer (40). In the present study, the glioma cells exhibited a higher tumour sphere formation capacity and increased expression of stemness markers following co-cultured with M2-TAMs, compared with those co-cultured with THP-1 cells, indicating enhancement in their stemness and tumour development ability induced by M2-TAMs. The M2-TAMs were also shown to promote the migration ability of glioma cells and facilitate EMT transition.
Previous reports have demonstrated that the level of TGF-β receptor 2 (TGFBR2), as a specific receptor for TGF-β1, is higher in glioma stem-like cells (21). Repression of the expression of TGFBR2 in glioma notably decreased their invasion ability, even when co-cultured with M2-TAMs, indicating that the TGF-β pathway contributed the major function in the interaction between M2-TAMs and glioma stem-like cells. The results demonstrated that direct TGF-β1 protein treatment was able to achieve similar effects to those induced by M2-TAMs in glioma cell performance, including sphere formation capacity, self-renewal and migration abilities. However, the TGF-β inhibitor significantly eliminated the effects that were induced by M2-TAMs.
The TGF-β signalling cascade is initiated by binding and activating its receptors (TGFBR1 and TGFBR2), leading to the phosphorylation of intracellular effectors SMAD2/SMAD3 (30). To demonstrate the TGF-β pathway activity in glioma cells, the protein levels of pSMAD2 and pSMAD3 were detected. The resulting data indicated that co-culture with M2-TAMs or TGF-β1 protein stimulation significantly increased SMAD2/3 phosphorylation activity, whereas the TGF-β pathway inhibitor was shown to repress these effects (Fig. 4A and B). These results suggested that the TGF-β1-SMAD2/3 pathway is key in the cross-talk between M2-TAMs and glioma cells. Although Ye et al (21) also investigated M2-TAMs in glioma, the study focused on the association between M2-TAMs and glioma stem-like cells, and did not examine the specific associated pathways downstream of TGF-β1. The present study demonstrated for the first time, to the best of our knowledge, that M2-TAMs increase the stemness and migration abilities of glioma cells via the TGF-β1-SMAD2/3 pathway.
SOX2 has been shown to be important in the maintenance of stem cell activity, particularly for cancer stem cells (32). SOX2 has been identified as a frequently amplified gene in small cell lung cancer (41). In breast cancer, SOX has been reported to be upregulated in cancer stem cells (42). The downregulation of SOX2 in glioma stem cells impairs their proliferation and tumour formation ability (33). SOX4 acts downstream of the TGF-β pathway and regulates the expression of SOX2 (26). The inhibition of TGF-β signalling is able to suppress the expression of SOX2 through inhibiting SOX4 (26). SOX4 can also regulate the TGF-β-induced EMT process (43). The results of the present study showed that, when co-cultured with M2-TAMs or treated with TGF-β1 protein, the glioma cells exhibited increased expression of SOX4, leading to elevated SOX2 at the same time. Therefore, these results suggested that TGF-β1 activated the SMAD2/3 pathway to induce the expression of SOX4 and SOX2, promoting the stemness and migration abilities of the glioma cells. These results are the first, to the best of our knowledge, to suggest that the SOX4/SOX2 axis is involved in the regulation of M2-TAMs in the stemness and migration abilities of glioma cells.
The in vivo tumour graft assay further supported the above conclusion. The size and growth rate of tumours formed by U251 cells were significantly increased by co-culture with M2-TAMs and TGF-β1 protein treatment. Additionally, TGF-β inhibitor treatment eliminated these effects induced by M2-TAMs. From these results in vivo, it was concluded that M2-TAMs accelerated the growth of solid tumours by the TGF-β1 pathway, which may result from the increased stemness and migration abilities of glioma cells that were observed in the in vitro results.
In conclusion, the present study investigated the contribution of M2-TAMs to the stemness and migration abilities of glioma cells. It was demonstrated that TGF-β1 in the tumour microenvironment secreted from M2-TAMs activated the SMAD2/3 pathway and then increased the expression levels of SOX4 and SOX2. This resulted in elevation of the stemness and migration abilities of the cells in vitro, by altering the gene expression pattern associated with stemness and the EMT process, and increased solid tumour sizes in vivo. The development of therapeutic strategies against the communication among M2-TAMs and glioma cells may be a potential approach to monitor glioma initiation and progression.
Acknowledgments
Not applicable.
Funding
The present study was supported by Beijing Medical Health Public Welfare Foundation (grant no. YWJKJJHKYJJ-B17468).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
ZL guarantees the integrity of the entire study and was responsible for the design of the study and clarified associated intellectual content. ZL also performed experimental studies, data acquisition and data analysis and then edited the whole manuscript and reviewed it. WK performed experimental studies and manuscript editing. QZ performed data analysis and manuscript preparation. YZ performed data acquisition and reviewed the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All experimental procedures involving animals were performed in accordance with the Guidelines for the Care and Use of Laboratory Animals of Xiangya Hospital, Central South University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing interests.
References
- 1.Ostrom QT, Bauchet L, Davis FG, Deltour I, Fisher JL, Langer CE, Pekmezci M, Schwartzbaum JA, Turner MC, Walsh KM, et al. The epidemiology of glioma in adults: A ‘state of the science’ review. Neuro Oncol. 2014;16:896–913. doi: 10.1093/neuonc/nou087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zagzag D, Salnikow K, Chiriboga L, Yee H, Lan L, Ali MA, Garcia R, Demaria S, Newcomb EW. Downregulation of major histocompatibility complex antigens in invading glioma cells: Stealth invasion of the brain. Lab Invest. 2005;85:328–341. doi: 10.1038/labinvest.3700233. [DOI] [PubMed] [Google Scholar]
- 3.Li J, Yuan J, Yuan X, Zhao J, Zhang Z, Weng L, Liu J. MicroRNA-200b inhibits the growth and metastasis of glioma cells via targeting ZEB2. Int J Oncol. 2016;48:541–550. doi: 10.3892/ijo.2015.3267. [DOI] [PubMed] [Google Scholar]
- 4.Noy R, Pollard JW. Tumor-associated macrophages: From mechanisms to therapy. Immunity. 2014;41:49–61. doi: 10.1016/j.immuni.2014.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sainz B, Jr, Alcala S, Garcia E, Sanchez-Ripoll Y, Azevedo MM, Cioffi M, Tatari M, Miranda-Lorenzo I, Hidalgo M, Gomez-Lopez G, et al. Microenvironmental hCAP-18/LL-37 promotes pancreatic ductal adenocarcinoma by activating its cancer stem cell compartment. Gut. 2015;64:1921–1935. doi: 10.1136/gutjnl-2014-308935. [DOI] [PubMed] [Google Scholar]
- 6.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 7.Molina JR, Hayashi Y, Stephens C, Georgescu MM. Invasive glioblastoma cells acquire stemness and increased Akt activation. Neoplasia. 2010;12:453–463. doi: 10.1593/neo.10126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bao S, Wu Q, McLendon RE, Hao Y, Shi Q, Hjelmeland AB, Dewhirst MW, Bigner DD, Rich JN. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature. 2006;444:756–760. doi: 10.1038/nature05236. [DOI] [PubMed] [Google Scholar]
- 9.Hwang RF, Moore T, Arumugam T, Ramachandran V, Amos KD, Rivera A, Ji B, Evans DB, Logsdon CD. Cancer-associated stromal fibroblasts promote pancreatic tumor progression. Cancer Res. 2008;68:918–926. doi: 10.1158/0008-5472.CAN-07-5714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hanahan D, Weinberg RA. Hallmarks of cancer: The next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 11.Heusinkveld M, van der Burg SH. Identification and manipulation of tumor associated macrophages in human cancers. J Transl Med. 2011;9:216. doi: 10.1186/1479-5876-9-216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Steinman RM, Idoyaga J. Features of the dendritic cell lineage. Immunol Rev. 2010;234:5–17. doi: 10.1111/j.0105-2896.2009.00888.x. [DOI] [PubMed] [Google Scholar]
- 13.Sica A, Larghi P, Mancino A, Rubino L, Porta C, Totaro MG, Rimoldi M, Biswas SK, Allavena P, Mantovani A. Macrophage polarization in tumour progression. Semin Cancer Biol. 2008;18:349–355. doi: 10.1016/j.semcancer.2008.03.004. [DOI] [PubMed] [Google Scholar]
- 14.Hamilton TA, Zhao C, Pavicic PG, Jr, Datta S. Myeloid colony-stimulating factors as regulators of macrophage polarization. Front Immunol. 2014;5:554. doi: 10.3389/fimmu.2014.00554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sielska M, Przanowski P, Wylot B, Gabrusiewicz K, Maleszewska M, Kijewska M, Zawadzka M, Kucharska J, Vinnakota K, Kettenmann H, et al. Distinct roles of CSF family cytokines in macrophage infiltration and activation in glioma progression and injury response. J Pathol. 2013;230:310–321. doi: 10.1002/path.4192. [DOI] [PubMed] [Google Scholar]
- 16.Staudt ND, Jo M, Hu J, Bristow JM, Pizzo DP, Gaultier A, VandenBerg SR, Gonias SL. Myeloid cell receptor LRP1/CD91 regulates monocyte recruitment and angiogenesis in tumors. Cancer Res. 2013;73:3902–3912. doi: 10.1158/0008-5472.CAN-12-4233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ling EA, Wong WC. The origin and nature of ramified and amoeboid microglia: A historical review and current concepts. Glia. 1993;7:9–18. doi: 10.1002/glia.440070105. [DOI] [PubMed] [Google Scholar]
- 18.Komohara Y, Ohnishi K, Kuratsu J, Takeya M. Possible involvement of the M2 anti-inflammatory macrophage phenotype in growth of human gliomas. J Pathol. 2008;216:15–24. doi: 10.1002/path.2370. [DOI] [PubMed] [Google Scholar]
- 19.Gocheva V, Wang HW, Gadea BB, Shree T, Hunter KE, Garfall AL, Berman T, Joyce JA. IL-4 induces cathepsin protease activity in tumor-associated macrophages to promote cancer growth and invasion. Genes Dev. 2010;24:241–255. doi: 10.1101/gad.1874010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li D, Wang X, Wu JL, Quan WQ, Ma L, Yang F, Wu KY, Wan HY. Tumor-produced versican V1 enhances hCAP18/LL-37 expression in macrophages through activation of TLR2 and vitamin D3 signaling to promote ovarian cancer progression in vitro. PLoS One. 2013;8:e56616. doi: 10.1371/journal.pone.0056616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ye XZ, Xu SL, Xin YH, Yu SC, Ping YF, Chen L, Xiao HL, Wang B, Yi L, Wang QL, et al. Tumor-associated microglia/macrophages enhance the invasion of glioma stem-like cells via TGF-beta1 signaling pathway. J Immunol. 2012;189:444–453. doi: 10.4049/jimmunol.1103248. [DOI] [PubMed] [Google Scholar]
- 22.Morford LA, Dix AR, Brooks WH, Roszman TL. Apoptotic elimination of peripheral T lymphocytes in patients with primary intracranial tumors. J Neurosurg. 1999;91:935–946. doi: 10.3171/jns.1999.91.6.0935. [DOI] [PubMed] [Google Scholar]
- 23.Bruna A, Darken RS, Rojo F, Ocaña A, Peñuelas S, Arias A, Paris R, Tortosa A, Mora J, Baselga J, Seoane J. High TGFbeta-Smad activity confers poor prognosis in glioma patients and promotes cell proliferation depending on the methylation of the PDGF-B gene. Cancer Cell. 2007;11:147–160. doi: 10.1016/j.ccr.2006.11.023. [DOI] [PubMed] [Google Scholar]
- 24.Massagué J. TGFbeta in cancer. Cell. 2008;134:215–230. doi: 10.1016/j.cell.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bierie B, Moses HL. Transforming growth factor beta (TGF-beta) and inflammation in cancer. Cytokine Growth Factor Rev. 2010;21:49–59. doi: 10.1016/j.cytogfr.2009.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jun F, Hong J, Liu Q, Guo Y, Liao Y, Huang J, Wen S, Shen L. Epithelial membrane protein 3 regulates TGF-β signaling activation in CD44-high glioblastoma. Oncotarget. 2017;8:14343–14358. doi: 10.18632/oncotarget.11102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Araki S, Eitel JA, Batuello CN, Bijangi-Vishehsaraei K, Xie XJ, Danielpour D, Pollok KE, Boothman DA, Mayo LD. TGF-beta1-induced expression of human Mdm2 correlates with late-stage metastatic breast cancer. J Clin Invest. 2010;120:290–302. doi: 10.1172/JCI39194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Donkor MK, Sarkar A, Savage PA, Franklin RA, Johnson LK, Jungbluth AA, Allison JP, Li MO. T cell surveillance of oncogene-induced prostate cancer is impeded by T cell-derived TGF-β1 cytokine. Immunity. 2011;35:123–134. doi: 10.1016/j.immuni.2011.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ikushima H, Todo T, Ino Y, Takahashi M, Miyazawa K, Miyazono K. Autocrine TGF-beta signaling maintains tumori-genicity of glioma-initiating cells through Sry-related HMG-box factors. Cell Stem Cell. 2009;5:504–514. doi: 10.1016/j.stem.2009.08.018. [DOI] [PubMed] [Google Scholar]
- 30.Moustakas A, Heldin CH. The regulation of TGFbeta signal transduction. Development. 2009;136:3699–3714. doi: 10.1242/dev.030338. [DOI] [PubMed] [Google Scholar]
- 31.Feng XH, Derynck R. Specificity and versatility in tgf-beta signaling through Smads. Annu Rev Cell Dev Biol. 2005;21:659–693. doi: 10.1146/annurev.cellbio.21.022404.142018. [DOI] [PubMed] [Google Scholar]
- 32.Garros-Regulez L, Aldaz P, Arrizabalaga O, Moncho-Amor V, Carrasco-Garcia E, Manterola L, Moreno-Cugnon L, Barrena C, Villanua J, Ruiz I, et al. mTOR inhibition decreases SOX2-SOX9 mediated glioma stem cell activity and temozolomide resistance. Expert Opin Ther Targets. 2016;20:393–405. doi: 10.1517/14728222.2016.1151002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Alonso MM, Diez-Valle R, Manterola L, Rubio A, Liu D, Cortes-Santiago N, Urquiza L, Jauregi P, Lopez de Munain A, Sampron N, et al. Genetic and epigenetic modifications of Sox2 contribute to the invasive phenotype of malignant gliomas. PLoS One. 2011;6:e26740. doi: 10.1371/journal.pone.0026740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garros-Regulez L, Garcia I, Carrasco-Garcia E, Lantero A, Aldaz P, Moreno-Cugnon L, Arrizabalaga O, Undabeitia J, Torres-Bayona S, Villanua J, et al. Targeting SOX2 as a therapeutic strategy in glioblastoma. Front Oncol. 2016;6:222. doi: 10.3389/fonc.2016.00222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Huang WC, Chan ML, Chen MJ, Tsai TH, Chen YJ. Modulation of macrophage polarization and lung cancer cell stemness by MUC1 and development of a related small-molecule inhibitor pterostilbene. Oncotarget. 2016;7:39363–39375. doi: 10.18632/oncotarget.8101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lonardo E, Hermann PC, Mueller MT, Huber S, Balic A, Miranda-Lorenzo I, Zagorac S, Alcala S, Rodriguez-Arabaolaza I, Ramirez JC, et al. Nodal/Activin signaling drives self-renewal and tumorigenicity of pancreatic cancer stem cells and provides a target for combined drug therapy. Cell Stem Cell. 2011;9:433–446. doi: 10.1016/j.stem.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 37.Lonardo E, Frias-Aldeguer J, Hermann PC, Heeschen C. Pancreatic stellate cells form a niche for cancer stem cells and promote their self-renewal and invasiveness. Cell Cycle. 2012;11:1282–1290. doi: 10.4161/cc.19679. [DOI] [PubMed] [Google Scholar]
- 38.Cabarcas SM, Mathews LA, Farrar WL. The cancer stem cell niche-there goes the neighborhood. Int J Cancer. 2011;129:2315–2327. doi: 10.1002/ijc.26312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Solinas G, Schiarea S, Liguori M, Fabbri M, Pesce S, Zammataro L, Pasqualini F, Nebuloni M, Chiabrando C, Mantovani A, Allavena P. Tumor-conditioned macrophages secrete migration-stimulating factor: A new marker for M2-polarization, influencing tumor cell motility. J Immunol. 2010;185:642–652. doi: 10.4049/jimmunol.1000413. [DOI] [PubMed] [Google Scholar]
- 40.Yang J, Liao D, Chen C, Liu Y, Chuang TH, Xiang R, Markowitz D, Reisfeld RA, Luo Y. Tumor-associated macrophages regulate murine breast cancer stem cells through a novel paracrine EGFR/Stat3/Sox-2 signaling pathway. Stem Cells. 2013;31:248–258. doi: 10.1002/stem.1281. [DOI] [PubMed] [Google Scholar]
- 41.Rudin CM, Durinck S, Stawiski EW, Poirier JT, Modrusan Z, Shames DS, Bergbower EA, Guan Y, Shin J, Guillory J, et al. Comprehensive genomic analysis identifies SOX2 as a frequently amplified gene in small-cell lung cancer. Nat Genet. 2012;44:1111–1116. doi: 10.1038/ng.2405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Leis O, Eguiara A, Lopez-Arribillaga E, Alberdi MJ, Hernandez-Garcia S, Elorriaga K, Pandiella A, Rezola R, Martin AG. Sox2 expression in breast tumours and activation in breast cancer stem cells. Oncogene. 2012;31:1354–1365. doi: 10.1038/onc.2011.338. [DOI] [PubMed] [Google Scholar]
- 43.Vervoort SJ, Lourenço AR, van Boxtel R, Coffer PJ. SOX4 mediates TGF-β-induced expression of mesenchymal markers during mammary cell epithelial to mesenchymal transition. PLoS One. 2013;8:e53238. doi: 10.1371/journal.pone.0053238. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.