Abstract
The aim of this study is to create a computational model of the human ureteral system that accurately replicates the peristaltic movement of the ureter for a variety of physiological and pathological functions. The objectives of this research are met using our in-house fluid-structural dynamics code (CgLes–Y code). A realistic peristaltic motion of the ureter is modelled using a novel piecewise linear force model. The urodynamic responses are investigated under two conditions of a healthy and a depressed contraction force. A ureteral pressure during the contraction shows a very good agreement with corresponding clinical data. The results also show a dependency of the wall shear stresses on the contraction velocity and it confirms the presence of a high shear stress at the proximal part of the ureter. Additionally, it is shown that an inefficient lumen contraction can increase the possibility of a continuous reflux during the propagation of peristalsis.
Keywords: CFD, Ureter, Reflux, PUJ, VUJ, VUR
Introduction
The ureter is a muscular tube with non-linear mechanical properties which conveys urine from the kidneys to the bladder [1]. In the urinary system, the peristaltic motion is caused by a muscular contraction of the ureteral wall initiated by pacemakers. This drives urine from the kidney to the bladder through the ureter. Ureteral peristaltic motions are the result of a complicated movement of various differently aligned muscle fibres in the ureteral wall [2, 3]. These are difficult to study experimentally since it is difficult to analyse an individual muscle cell in isolation from its neighbours.
The mechanical properties of ureteral walls have been studied in many biomechanics research of soft biological tissues [1, 4, 5]. Yin et al. [4] showed that the ureteral wall has viscoelastic material properties and its stress–strain relationship is nonlinear. Their results also showed that stress does not only depend on the strain but also its strain history, which is related to hysteresis, stress relaxation and creep.
It is worthy of mention that the biomechanical properties and composition of the human ureter are affected by age and region [6]. According to the study by Sokolis et al. [6], despite a non-significant difference between the left and right ureters, regional differences were established by the displacement of the stress–strain curves from the upper to lower ureter. The different distribution of properties may be related to the difference in the functions. The stiffness, which increases distally, may constitute an adaptation of the ureter to its functional demands, namely storage of urine proximally where it is wider and distensible, and transfer of urine distally where the ureter is more resistant by being narrower and stiffer. Moreover, the collagen content of the upper ureter is increased by aging and it is distributed regional uniformly.
There is an increasing number of patients suffering from ureter diseases such as dysfunctional Pelvis Ureteric Junction (PUJ), VesicoUreteric Reflux (VUR), and intrinsic and extrinsic obstruction of the urinary tract infection. There has been a wide range of clinical studies on the ureteral system with the purpose of improving our understanding of urodynamic responses under different pathological conditions [7–11]. In prior clinical studies, several experimental techniques including X-ray screening, dynamic scintigraphy and Doppler ultrasonography were used to investigate the urodynamic and ureteric peristalsis [12–20]. Kiil [20], for instance, utilized an invasive technique using the strain-gauge pressure transducer attached to a catheter mechanism to measure the ureteral pressure.
Although the clinical data is usually considered to be more reliable, the non-controllable environment of an in vivo study as well as the invasive nature of the measurement procedure has encouraged many research groups to concentrate on numerical modelling and computational simulation [21–27]. Recent advances in computational simulation and an increase in computing power make a realistic computational model of a ureter and peristaltic flow possible. Kumar et al. [25] used a computational method to investigate the effect of the peristalsis amplitude and frequency on the urine flow for different Reynolds number. Their results indicated that progressive sinusoidal waves with high amplitude and low wave frequency caused peristaltic flows with high wall shear stress variations.
In the majority of previous studies [21–26], a number of key parameters such as the actual geometrical model of the ureter, biomechanical properties of the ureteral wall, the origin of contractions and the multi-dimensional ureteral movements were not considered in the simulations. In general, incorporating too many basic assumptions into a simulation may lead to less reliable results. So, developing a computational platform which can incorporate more realistic parameters into the model will provide physicians with a better understanding of the exact mechanisms behind a variety of clinical complications, leading to a better diagnosis and treatment.
A recent computational simulation of the ureter was introduced by Vahidi et al. [27]. A cylindrical geometry with nonlinear material properties was simulated assuming a rigid contact surface in order to model the peristalsis. Their results showed that recirculation regions formed against the jet flow neighbouring the bolus peak. Through the wave propagation, a separation occurred behind the moving bolus on the wall and ureteropelvic reflux began from that location and extended upstream to the ureteral inlet. The maximum luminal pressure consistently occurred behind the urine bolus during the peristalsis. Their studies indicate that the function of ureteropelvic junction in the prevention of reflux was significant.
Measuring the pressure pulses in the ureter is a key diagnostic tool in understanding peristaltic activity. Intra-ureteral pressure has typically been studied using fluid filed catheters connected to a displacement type pressure transducer [10, 19, 20]. These pressure measurements rely on the movement of the urine in and out of a transducer chamber. However, in the absence of a catheter, the muscle contraction completely closes the cross-sectional area, so the pressure distribution over time is only a function of the muscle action and there will not be any fluid inside the closed lumen. Total contraction pressure is therefore solely a result of contact forces between the ureteral walls [28].
In our previous study, we simulated a simplistic cylindrical model of a ureter [29, 30]. The aim was to introduce a novel technique to simulate a realistic ureteral peristaltic contraction. For the present study, the wall contact pressure in addition to the Intra-abdominal Pressure (IAP) is used to investigate the pressure pulse. The pressure pulse was simulated by using a novel Piecewise Linear Force Model (PLFM) with the purpose of emulating the relaxation and contraction of individual muscles with fixed positions across time. To our knowledge, this technique has not been used in previous studies. This novel technique can be used in the simulation of other organs such as the human gastrointestinal tract which have similar peristaltic movements.
For this study, a computational model of the ureteral system has been accurately replicated and the peristaltic movement of a healthy and a depressed contraction force has been investigated. The simulations are carried out using our in-house Computational Fluid Dynamic (CFD) platform, known as CgLes [31, 32] to model the urine flow, coupled with our in-house structural code, known as the Y code [33] to model the viscoelastic ureteral wall. CgLes is a three-dimensional fluid solver with second order accuracy in both time and space and is based on a finite volume formulation. The capability of CgLes to simulate both laminar and turbulent flows has been extensively verified [32]. The Y code uses the combined finite discrete element method to simulate the deformation of solid material [33].
Methodology
A solver for incompressible viscous flow (urine)
For the fluid phase, urine is modelled as an incompressible viscous flow whose motion is governed by the Navier–Stokes equations:
| 1 |
where ui is fluid velocities, p is the pressure, is dynamic viscosity and f i is a body force term. For time-stepping, the 2nd order Adams-Bash method was used. The Navier–Stokes equations are discretised on a fixed staggered Cartesian grid by the finite volume approach. The spatial derivatives in the diffusion and convection terms are approximated using the second order finite volume method. A conjugate gradient method is used to solve the pressure Poisson equation [32, 34].The fluid domain is created by 213 blocks with the grid resolution of 64 × 448 × 960 points. The grid cell length in the x, y and z directions is 0.0078 × 0.0089 × 0.02 cm, respectively.
A solver for the deformation of the solid (ureter)
For the solid phase, the combined finite-discrete element method is used to simulate the movement and deformation of the ureter under external forces [33]. The equation of motion is solved by an explicit time integration scheme based on a second order central difference method.
The static shape of a healthy human ureter was obtained using CT scans of the urinary system of a female human. The CT scans were imported into the image-processing package Mimics (14.01) and converted into a 3D model as shown in Fig. 1a. The centre line of the 3D geometry was obtained using Mimic software and the resulting 3D model gave a fair approximation of the ureter’s geometry. The length and the average diameter of the ureter are 22 and 0.5 cm respectively.
Fig. 1.
a Cropped 3D model of the right ureter and bladder, b the combined fluid and solid domains of the ureter
For this study, Ansys ICEM 15.0 was used to create the tetrahedral mesh for the structural model. There were 6700 elements, 1891 nodes. The minimum and maximum element sizes were 0.005 and 0.7 cm respectively. Figure 1b shows the combined fluid and solid domains.
Ureteral wall properties
To mimic a realistic computational ureteral wall model, the non linear tensile properties of the ureter have to be considered. To adapt the nonlinear tensile properties of the ureter, a scalar quantity called “Equivalent strain” is introduced into the structural code. This is described in Eq. (2) where is Poisson ratio, ε11, ε22, ε33 are principal strains and ε12, ε23, ε31 are shear strains from the strain tensor E. By implementing the equivalent strain into the structural code, multiple strains in the computational model become equivalent to the uniaxial strain reported in the experimental study [2].
| 2 |
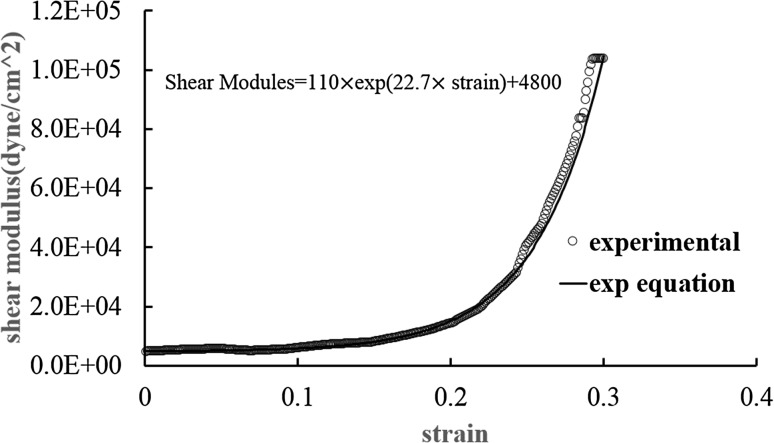
As shown in Fig. 2, an exponential function was obtained for the best-fit function from the stress–strain curve by Yin and Fung [2], and implemented in the structural code.
Fig. 2.
The non-linear stress–strain relationship of the ureteral wall data, extracted from Yin and Fung [2] and an exponential function matched to this data and implemented in the structural code
Immersed boundary
To couple the fluid motion and solid deformation, an Immersed Boundary (IB) method was used to link the interface between the fluid and the solid, both of which have independent meshes. By introducing a force term into the fluid’s momentum equations, the IB points become non-slip boundary points. A spatially second order direct-forcing scheme was used for the implementation of the IB method. The combined code has been verified in previous studies [29, 30, 32, 34] for a range of engineering problems from sediment flow to fluid–structure interaction of pipes.
Piecewise linear force model
The ureteral peristaltic contraction was simulated using the novel Piecewise Linear Force Model (PLFM) with the purpose of emulating the relaxation and contraction of individual muscles with fixed positions across time.
The PLFM equation is shown in Eq. (3) where m is the total number of sections of the contraction. The two other variables are s and t where s is defined as the point in time at which a contraction is transferred from one cross-section to another of the ureter model and t is the time at the beginning of the window for each cross-section. The force function, which acts on each cross-section in this contraction model, is given in Eq. (3).
| 3 |
Figure 3 shows an individual time-window for each cross-section that is exposed to a piecewise linear force. Each time-window is dependent on the current time. Since each section is allocated to an individual time-window with a time span of 2–3 s, the force is applied to the cross-section when the current time reaches a particular time window.
Fig. 3.
The piecewise linear function evaluated at t for different sections in the ureter
Modelling of the intra-abdominal pressure
In order to prevent the ureter from excessive radial expansion, an Intra-Abdominal Pressure (IAP) model was introduced into the Y code. A detailed description of the algorithm is presented in Fig. 4.
Fig. 4.
The IAP algorithm
To mimic IAP in the structural code, a force was applied to all nodes in the ureter’s mesh at each time step. This method keeps the current coordinates of the centre of each cross-section the same as their initial coordinates. The IAP is 4 cm H2O in a healthy ureter. The magnitude of the force of 100 dynes is obtained from Eq. (4) where r is the radius of the ureter, L is its length and N is the number of surface nodes in the computational model.
| 4 |
Figure 5a, b shows a comparison between the two computational models of the peristaltic movement in the presence and absence of the IAP. Figure 5a shows without the IAP; the ureter model is not constrained and starts moving in an eccentric direction. Figure 5b shows that with the presence of the IAP; the ureter model is fully constrained and the applied radial force leads to a centric contraction, similar to an actual human ureter.
Fig. 5.
Comparison between the two simulations in the absence (a) and in the presence (b) of the IAP
Boundary conditions
Urine is an incompressible Newtonian fluid with a dynamic viscosity coefficient of 0.01 Poise and a constant density of 1 g/cm3. For this model, the initial velocity of urine in the ureter is taken as 0.01 cm/s. A no-slip boundary condition is applied between the urine and ureteral wall. Two different pressure differences are used between the inlet and outlet of the ureter to simulate a healthy ureteral contraction and a depressed ureteral contraction under the effect of a relaxation drug. The aim of simulating a depressed ureteral contraction is to assess the possibility of reflux occurring during peristaltic movement in patients with an inadequate contraction force resulting from taking dilators. The pressure differences for the healthy and decreased ureteral contraction are extracted from a study by Shafik [10]. Table 1 shows the boundary conditions, contraction parameters and material properties of the simulations performed in this study.
Table 1.
The boundary conditions, contraction parameters and material properties of the simulations performed in this work
| Case studies | Conditions | Stress–strain relationship | Velocity of Contraction A and B (cm/s) | Pressure difference ()/mmHg | Contraction force (dynes) | IAP equivalent-force (dynes) |
|---|---|---|---|---|---|---|
| 1 | Healthy | Nonlinear | 3.5, 1.75 | 0.5 | 1200 | 100 |
| 2 | Depressed ureteral contraction | Nonlinear | 3.5, 3.5 | 0 | 450 | 100 |
In this study, a simulation of peristaltic movement was carried out by describing two pacemaker contractions: one in the proximal part of ureter (Contraction A) and one in the distal part of the ureter (Contraction B). The sections selected are shown in Fig. 6. For the first case study, according to Kiil [20], the maximum pressure during the contraction was 25 mmHg. This is equivalent to a force with a magnitude of 1200 dynes. The contractions move with a velocity between 1.5 and 3.5 cm/s [10, 20, 35]. The resting pressure difference between the inlet and the outlet is 0.5 mmHg [10]. For the second case study, a depressed ureteral contraction caused by taking dilators was simulated. As shown in previous clinical studies [36], dilators drugs, used in promoting the passage of stones, lead to a highly depressed amplitude of the peristalsis. This results in a significant reduction in the ureteral contraction pressure between 20 and 65% [36]. For this study, to obtain a similar level of the depressed contraction pressure, a force of 450 dynes was applied.
Fig. 6.
Contractions A and B which mimic pacemaker activities in the proximal and distal part of the ureter, Z is the distance along the ureter
In Fig. 6, Z is the distance along the ureter. The total length of the ureter was 22 cm. Z = 22 is the Pelvis Ureteric Junction (PUJ) and Z = 0 is the Vesico Ureteric Junction (VUJ). For the healthy condition, the Contraction A starts from Z = 18.8 cm and propagates towards Z = 14.48 mm with a speed of 3.5 cm/s. Concurrently, Contraction B start at Z = 8.7 mm and propagates towards Z = 4.7 mm with the same speed.
Results and discussion
PLFM results
In order to verify the PLFM, a simulation of the contraction displacement in one ureteral cross–section was performed and the results were compared with experimental data. Figure 7a shows the pressure using the PLFM in comparison with the experimental data extracted from the study by Kiil [20]. The resulting contact pressure from the area in which the contraction force was applied is digitally smoothed using a Savitzky–Golay filter. The maximum contact pressure is obtained by dividing the contact forces on each element by its area. Figure 7a shows that the PLFM produces a contact pressure with an amplitude of 20 mmHg and a pulse duration of 2.3 s indicating a good agreement with the experimental ureteral pressure measurements [10, 20].
Fig. 7.
a The pressure time evolution using the PLFM in comparison with the experimental data extracted from the study by Kiil [20]. b The deformation of a cross-section over the same period of contraction time, where R is the radial displacement of the cross-section in cm
Figure 7b shows the deformation of a cross-section over the same period of contraction time. R is defined as the radial displacement of the ureter in cm. This figure confirms the complete closure of the ureteral cross-section, where the maximum pulse pressure occurs. The pulse duration, shown in Fig. 7, is the time when the contact pressure increases to its maximum value and then gradually drops as the contraction travels away. The computational result from recording contact force is only compared with the part of the experimental result in which the ureteral wall are in contact. The advantage of using the PLFM is to control the timing of contraction and relaxation of each cross-section.
Simulation of a healthy ureteral contraction
A healthy ureter is simulated using the previously described boundary conditions. Figure 8 shows the wall shear stress at t = 0.3, 0.6 and 1.0 s. The results show that, as expected, the shear stress is significantly higher around the contraction regions. It is clear that the highest shear stress occurs at the proximal part of the ureter, in the vicinity of the PUJ. The figure also shows that the maximum value of the shear stress decreases as the peristalsis propagates towards the VUJ.
Fig. 8.
Longitudinal distribution (cm) of wall shear stress (Barye) that was computed with a pressure difference of 0.6 cm H2O (healthy condition)
The results also show that the maximum shear stress on the wall depends on the velocity of the contraction in the ureter. The shear stress upstream of the Contraction B is 75% lower than that upstream of Contraction A, because Contraction B has a lower peristaltic velocity. These results show that the proximal part of the ureter, in the vicinity of the PUJ, can be subject to a high shear stress which may result in wall deformation. The occurrence of high shear stresses at the proximal part of the ureter described by this study support the findings from other computational [23] and experimental studies [6]. This is indicative of large wall deformation and consequent proximal ureter rupture.
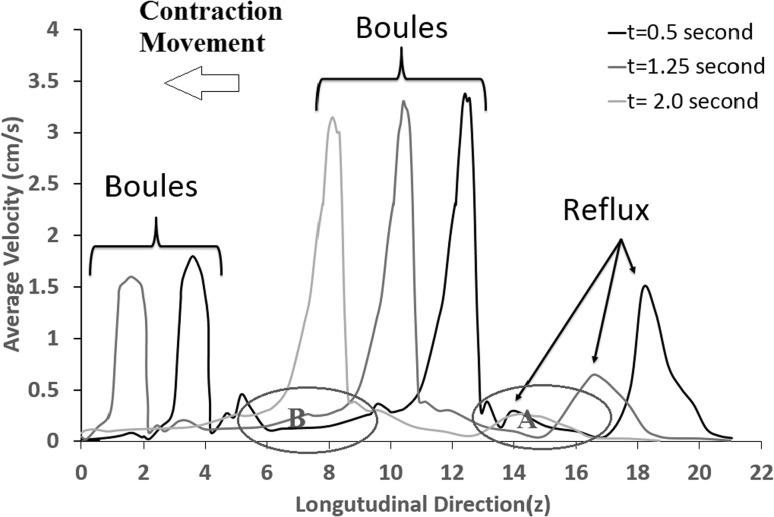
Figure 9 shows the average urine velocity at 0.5, 1.25, and 2.0 s for the healthy pressure difference condition. As the model steps from t = 0.5 to 2 s, the average reflux decreases by 85%. A similar drop in reflux as the contraction progresses has been reported in the study conducted by Vahidi et al. [23]. The maximum speed of urine flow induced by the moving Contractions A and B varies between 3.3–3.5 and 1.5–1.75 cm/s respectively. So, it is concluded that when the complete closure of the cross-section occurs, the urine’s maximum velocity is very close to the contraction speed.
Fig. 9.
The average velocities of urine in the ureter using the model (t = 0.5, 1.25, 2.0 s) in the healthy condition
Figure 10 shows the urine velocity vectors behind the moving Contraction A at t = 0.5 s. It is evident that there is a high backward velocity confirming the occurrence of a reflux upstream of the Contraction A.
Fig. 10.
The urine velocity vectors behind the moving Contraction A at t = 0.5 s
Simulation of a depressed ureteral contraction
Figure 11 shows the instantaneous velocity of the depressed ureteral contraction. The positive velocity represents the reflux and the negative velocity is the (correct) urine velocity in the direction of the contraction movement. The results indicate the presence of a continuous reflux upstream of the contraction during the peristaltic movement at t = 0.3, 1 and 2 s. These results show that a patient taking dilators is more exposed to the risk of ureteric reflux. Although the urine velocity in this model is reduced by 75% compared to the healthy contraction, the reflux velocity almost remains unchanged. Since the average of the ureteral diameter does not exceed than 0.5 cm, it can be concluded that the grade of the potential Vesicoureteric reflux (VUR) can be between 1 and 2. This means that the reflux can be limited to the ureter and up to the renal pelvis [37].
Fig. 11.
Instantaneous velocity profiles in Contractions A and B for the simulation of the unhealthy condition
Conclusion
In this study, a novel peristalsis model is used to investigate the human ureter under two different conditions; a healthy contraction and a depressed ureteral contraction mimicking the effect of a relaxation drug. The realistic peristaltic motion of the ureter is modelled using a novel piecewise linear force function. It has been shown that the piecewise linear force function produces a contact pressure in good agreement with experimental data. The results from the simulation of a depressed ureteral contraction show that the reflux occurring behind the contraction area remains unchanged compared to the healthy condition. It was also found for both conditions that the high level of wall shear stress and the reflux usually occur at the beginning of the lumen closure. This gradually disappears due to the propagation of the peristalsis in which the lumen is completely closed by a contraction force. This result indicates that initial location at which the pacemaker initiates the contraction, is crucial. Since the pacemakers are located at multiple sites in the upper urinary tract, it can be concluded that not all peristalsis movements cause high reflux or high shear stresses in the proximal part of the ureter. This still leaves many questions unresolved. For instance, the nature of the reflux, which is one of the biggest challenges in urodynamic research, is not still clear enough to be simulated. The interaction between the kidney and bladder pressure has hardly received any theoretical or numerical simulations and deserves further investigation.
Acknowledgements
National computing time was provided by UK Turbulence Consortium under EPSRC Grant EP/L000261, Clinical data was provided by Urology Research Innovation at Whipps Cross Hospital.
Conflict of interest
None.
Ethical approval
All scans showing the whole ureter with normal anatomy were anonymised and chosen at random from a larger educational set.
References
- 1.Wein AJ, Kavoussi L, Partin W, Peters CA. Urology, Campbell-Walsh. Vol. 2 of section XIV. 2012.
- 2.Weiss RM, Wagner ML, Hoffman BF. Localization of the pacemaker for peristalsis in the intact canine ureter. Invest Urol. 1967;5(42):1. [Google Scholar]
- 3.Osman F, et al. A novel videomicroscopic technique for studying rat ureteral peristalsis in vivo. World J Urol. 2009;27(2):265–270. doi: 10.1007/s00345-008-0340-6. [DOI] [PubMed] [Google Scholar]
- 4.Yin FC, Fung YC. Mechanical properties of isolated mammalian ureteral segments. Am J Physiol Leg Content. 1971;221(5):1484–1493. doi: 10.1152/ajplegacy.1971.221.5.1484. [DOI] [PubMed] [Google Scholar]
- 5.Watanabe H, Nakagawa Y, Uchida M. Tests to evaluate the mechanical properties of the ureter. In: Kambic HE, Yokobori AT, editors. Biomaterials’ mechanical properties. ASTM International; 1994.
- 6.Sokolis DP, et al. Age- and region-related changes in the biomechanical properties and composition of the human ureter. J Biomech. 2017;51:57–64. doi: 10.1016/j.jbiomech.2016.11.067. [DOI] [PubMed] [Google Scholar]
- 7.Eken Alper, Akbas Tugana, Arpaci Taner. Spontaneous rupture of the ureter. Singap Med J. 2015;56(2):e29. doi: 10.11622/smedj.2015029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Choi S-K, et al. A rare case of upper ureter rupture: ureteral perforation caused by urinary retention. Korean J Urol. 2012;53(2):131–133. doi: 10.4111/kju.2012.53.2.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Woodside JR, Borden TA. The pressure-flow relationship of the normal ureter. Invest Urol. 1980;18(1):82–83. [PubMed] [Google Scholar]
- 10.Shafik Ahmed. Ureteric profilometry: a study of the ureteric pressure profile in the normal and pathologic ureter. Scand J Urol Nephrol. 1998;32(1):14–19. doi: 10.1080/003655998750014620. [DOI] [PubMed] [Google Scholar]
- 11.Irina M, Lubov K. Ureteric function and upper urinary tract urodynamics in patients with stones in kidney and ureter. In: Kommu SS, editor. Evolving trends in urology. InTech; 2012.
- 12.Tillig Bernd, Constantinou Christos E. Videomicroscopic imaging of ureteral peristaltic function in rats during cystometry. J Pharmacol Toxicol Methods. 1996;35(4):191–202. doi: 10.1016/1056-8719(96)00045-7. [DOI] [PubMed] [Google Scholar]
- 13.Lewis CA, et al. Radionuclide imaging of ureteric peristalsis. BJU Int. 1989;63(2):144–148. doi: 10.1111/j.1464-410X.1989.tb05151.x. [DOI] [PubMed] [Google Scholar]
- 14.Patel U, Kellett MJ. Ureteric drainage and peristalsis after stenting studied using colour Doppler ultrasound. BJU Int. 1996;77(4):530–535. doi: 10.1046/j.1464-410X.1996.09298.x. [DOI] [PubMed] [Google Scholar]
- 15.Roshani H, et al. An in vivo endoluminal ultrasonographic study of peristaltic activity in the distal porcine ureter. J Urol. 2000;163(2):602–606. doi: 10.1016/S0022-5347(05)67943-8. [DOI] [PubMed] [Google Scholar]
- 16.Venkatesh R, et al. Impact of a double-pigtail stent on ureteral peristalsis in the porcine model: initial studies using a novel implantable magnetic sensor. J Endourol. 2005;19(2):170–176. doi: 10.1089/end.2005.19.170. [DOI] [PubMed] [Google Scholar]
- 17.Roshani H, et al. A study of ureteric peristalsis using a single catheter to record EMG, impedance, and pressure changes. Tech Urol. 1999;5(1):61–66. [PubMed] [Google Scholar]
- 18.Ulmsten Ulf, Diehl Jan. Investigation of ureteric function with simultaneous intraureteric pressure recordings and ureteropyelography. Radiology. 1975;117(2):283–289. doi: 10.1148/117.2.283. [DOI] [PubMed] [Google Scholar]
- 19.Young AJ, et al. Evaluation of novel technique for studying ureteral function in vivo. J Endourol. 2007;21(1):94–99. doi: 10.1089/end.2006.0161. [DOI] [PubMed] [Google Scholar]
- 20.Kiil Fredrik. Function of the ureter and renal pelvis. Ann Surg. 1958;148(2):280–291. doi: 10.1097/00000658-195808000-00020. [DOI] [Google Scholar]
- 21.Manton MJ. Long-wavelength peristaltic pumping at low Reynolds number. J Fluid Mech. 1975;68(3):467–476. doi: 10.1017/S0022112075001760. [DOI] [Google Scholar]
- 22.Zien T-F, Ostrach S. A long wave approximation to peristaltic motion. J Biomech. 1970;3(1):63–75. doi: 10.1016/0021-9290(70)90051-5. [DOI] [PubMed] [Google Scholar]
- 23.Vahidi B, Fatouraee N. A numerical simulation of peristaltic motion in the ureter using fluid structure interactions. In: Engineering in medicine and biology society, 2007. EMBS 2007. 29th annual international conference of the IEEE. IEEE; 2007. [DOI] [PubMed]
- 24.Xiao Q, Damodaran M. A numerical investigation of peristaltic waves in circular tubes. Int J Comput Fluid Dyn. 2002;16(3):201–216. doi: 10.1080/10618560290034681. [DOI] [Google Scholar]
- 25.Kumar BVR, Naidu KB. A numerical study of peristaltic flows. Comput Fluids. 1995;24(2):161–176. doi: 10.1016/0045-7930(94)00027-V. [DOI] [Google Scholar]
- 26.Vahidi B, et al. A mathematical simulation of the ureter: effects of the model parameters on ureteral pressure/flow relations. J Biomech Eng. 2011;133(3):031004. doi: 10.1115/1.4003316. [DOI] [PubMed] [Google Scholar]
- 27.Vahidi B, et al. A mathematical simulation of the ureter: effects of the model parameters on ureteral pressure/flow relations. J Biomech Eng. 2011;133(3):031004. doi: 10.1115/1.4003316. [DOI] [PubMed] [Google Scholar]
- 28.Woodburne Russell T, Lapides Jack. The ureteral lumen during peristalsis. Dev Dyn. 1972;133(3):255–258. doi: 10.1002/aja.1001330302. [DOI] [PubMed] [Google Scholar]
- 29.Hosseini G, et al. Computational simulation of the urinary system. In: Proceedings of the world congress on engineering and computer science, Vol II; 2012. ISBN: 978-988-19252-4-4.
- 30.Hosseini G. Simulation of the upper urinary system. Crit Rev™ Biomed Eng. 2013;41(3):259–268. doi: 10.1615/CritRevBiomedEng.2013009704. [DOI] [PubMed] [Google Scholar]
- 31.Thomas TG, Williams JJR. Development of a parallel code to simulate skewed flow over a bluff body. J Wind Eng Ind Aerodyn. 1997;67:155–167. doi: 10.1016/S0167-6105(97)00070-6. [DOI] [Google Scholar]
- 32.Ji C, Munjiza A, Williams JJR. A novel iterative direct-forcing immersed boundary method and its finite volume applications. J Comput Phys. 2012;231(4):1797–1821. doi: 10.1016/j.jcp.2011.11.010. [DOI] [Google Scholar]
- 33.Munjiza AA. The combined finite-discrete element method. New York: Wiley; 2004. [Google Scholar]
- 34.Singh KM, et al. On parallel pre-conditioners for pressure Poisson equation in LES of complex geometry flows. Int J Numer Meth Fluids. 2017;83(5):446–464. doi: 10.1002/fld.4277. [DOI] [Google Scholar]
- 35.Boyarsky S, Gottschalk CW, Tanagho EA. Urodynamics: hydrodynamics of the ureter and renal pelvis. London: Academic Press; 2014. [Google Scholar]
- 36.Davenport K, Timoney AG, Keeley Jr. FX. The role of ureteral relaxation in the promotion of stone passage. In: AIP conference proceedings, Vol. 900. No. 1. AIP; 2007.
- 37.Arlen AM, Kirsch AJ, Leong T, Cooper CS. Validation of the ureteral diameter ratio for predicting early spontaneous resolution of primary vesicoureteral reflux. J Pediatr Urol. 2017. 10.1016/j.jpurol.2017.01.012. [DOI] [PubMed]