Abstract
Sphingosine-1-phosphate (S1P) plays an important role in trafficking leukocytes and developing immune disorders including autoimmunity. In the synovium of rheumatoid arthritis (RA) patients, increased expression of S1P was reported, and the interaction between S1P and S1P receptor 1 (S1P1) has been suggested to regulate the expression of inflammatory genes and the proliferation of synovial cells. In this study, we investigated the level of S1P1 mRNA expression in the blood leukocytes of RA patients. In contrast to the previous reports, the expression level of this gene was not correlated to their clinical scores, disease durations and ages. However, S1P1 was transcribed at a significantly lower level in the circulating leukocytes of RA patients when compared to age-, and sex-matched healthy controls. Since these data may suggest the participation of S1P1, further studies are needed to determine the role of this receptor in the pathogenesis of RA.
Keywords: Sphingosine-1-phosphate, Rheumatoid arthritis, S1P receptor, Leukocytes
INTRODUCTION
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disease characterized by inflammation and bone erosion in joints (1,2,3). The joints in hands and feet are the primary targets of the inflammation, but the joints of shoulders, elbows, knees and other organs can also be impaired by this disease. Although the severity of this disease has been well documented, the pathogenesis of this disease is incompletely defined. It has been suggested that CD4 T helper (Th) cells and their secreted cytokines play a crucial role in developing and exacerbating the disease (4,5,6).
Naïve CD4 T cells encounter their cognitive Ag in the secondary lymphoid organs. After Ag-dependent activation, CD4 T cells develop into Th cells and egress from these organs. The egression of these Th cells relies on the tightly regulated mechanism, orchestrated by the chemotaxis of sphingosine-1-phosphate (S1P) and probably other chemokines. The high concentration of S1P in circulation compared to the lymph node is sensed by Th cells using S1P receptor 1 (S1P1), resulting in the egression of Th cells from the lymph node or the spleen (7). Therefore, the regulation of S1P and S1P1 expression is important in mounting immune response including autoimmunity.
High levels of S1P expression in the synovium of RA patients were recently reported (8). In addition, S1P has been shown to promote the expression of receptor activator of NF-κB ligand in an S1P1-dependent fashion, suggesting that this molecule can induce the production of inflammatory cytokines and the erosion of the bone (9). Although the importance of S1P and S1P1 in the synovium became evident for the pathogenesis of RA, it has not been addressed if the expression of S1P1 by the leukocytes in the circulation is compromised in RA patients. Here, we compared the expression of S1P1 mRNA of circulating leukocytes including Th cells between RA patients and healthy controls. The level of S1P1 mRNA expression was decreased in the blood leukocyte of RA patients and loosely correlated with age. However, it did not represent the clinical score of RA. These data suggest that S1P1 expressed by blood leukocytes is important in the pathogenesis of RA.
MATERIALS AND METHODS
Patients
Eighteen RA patients and age and sex matched healthy controls were enrolled. RA was diagnosed according to the 1987 American College of Rheumatology (ACR) criteria or 2010 ACR/European League Against Rheumatism criteria (10,11). Data collected include tender joint count, swollen joint count, patient's global assessment, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), rheumatoid factor or anti-cyclic citrullinated peptide Ab, and current medication. RA patients were grouped by disease activity by DAS28 calculated using CRP (28-joint disease activity score 28): <2.6, remission; ≤3.2, low disease activity; >2 and ≤5.1, moderate disease activity; >5.1, high disease activity (12,13). This study was reviewed and approved by Institutional Review Board at Chungnam National University Hospital (IRB number: 2012-01-024), and that written informed consent for this research project was obtained from all participants.
Extraction RNA from RA patient blood
Peripheral blood mononuclear cells (PBMCs) were purified from blood samples of RA patients and healthy controls by a Ficoll-density gradient method (Ficoll-Paque™ Plus; GE Healthcare, Chicago, IL, USA) as described (14). B cells were depleted from PBMC by negative selection using anti-CD19 MACS Microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany). Total RNAs were extracted from these cells using a Trizol® extraction protocol. Purified RNAs were analyzed immediately or stored at −80°C until use.
Quantitative RT-PCR (qRT-PCR) analyses
RNA was denatured at 65°C for 5 min in the presence of 4.8 μl of random primer, and reverse transcription reactions were performed in a 20 μl volume containing 1μl deoxynucleotide-triphosphates, 1 μl of RNase inhibitor, 2 μl of 0.1 M DTT, 4 μl of 5× first-strand Buffer and 1 μl of Superscript RT-II (Invitrogen, Carlsbad, CA, USA). The real-time qRT-PCR assay was carried out in a final volume of 25 μl as described previously (15). The 5 μl of cDNA was added to 20 μl of reaction mixture prepared using the iTaq Universal SYBRN® Green Supermix kit (Bio-Rad Laboratories, Hercules, CA, USA). Real time qRT-PCR assay was performed on a Stratagene MX3000P (Agilent Technologies, Santa Clara, CA, USA) under the following conditions: DNA polymerase activation at 95°C for 10 min, and 40 amplification cycles including denaturation at 95°C for 30 s and annealing at 60°C for 30 s, extension at 72°C for 30 s. Fluorescence data were recorded at the end of each cycle. The expression levels of S1P1 mRNA were normalized based on GAPDH expression. Ct values obtained from healthy controls were used as calibrator of mRNA expression in RA patients.
Molecular design of qRT-PCR primers
GAPDH; forward primer: 5′-CCACATCGCTCAGACACCAT-3′, reverse primer: 5′-ACCAGGCGCCCAATAGC-3′. S1P1; forward primer: 5′-TTCTGCGGGAAGGGAGTATG-3′, reverse primer: 5′-TATAGCGCTCAATGGCGATG-3′.
Statistical analysis
Association of continuous variables was estimated by Kendall's rank correlation. The difference between the patient group and control group was tested by Wilcoxon rank sum test (16). Box-and-whisker plot was used for visualization. Null hypotheses of no difference were rejected if p-values were less than 0.05. The statistical analysis used SAS 9.3 software package (SAS Institute Inc., Cary, NC, USA).
RESULTS AND DISCUSSION
Since the increased level of S1P expression in the synovium of RA patients was reported, we hypothesized that the expression of S1P1 in the circulating leukocytes of these patients is also increased to facilitate the recruitment of these cells into the joints. In order to test our hypothesis we recruited 18 RA patients and aged 18 and above, sex-matched healthy donors (Table 1). Among the 18 RA patients, 14 were female (77.8%) and the mean age (±standard deviation) was 55.4±12.1 year-old. The median disease duration was 3 (range: 0–51) months, and rheumatoid factor or anti-cyclic citrullinated Ab was positive in 16 patients (88.9%). Eight patients were initially diagnosed as RA as the time of study. Laboratory characteristics as follows: ESR, 66.1±33.2 mm/h; CRP, 2.48±0.75 mg/dl. Disease activity measured by DAS28-ESR was 4.51±0.51, by DAS28-CRP 3.31±0.36. Methotrexate was used in 15 RA patients, prednisolone in 17, or leflunomide in 1 for treatment of RA.
Table 1. Summary of RA patients and healthy controls.
| Variables | Healthy controls | RA patients |
|---|---|---|
| Age (mean±SD) | 55.4±12.1 | 55.4±12.1 |
| Sex (Male/Female) | 4/14 | 4/14 |
| DAS28-ESR (mean±SEM) | NA | 4.51±0.51 |
| DAS28-CRP (mean±SEM) | NA | 3.31±0.36 |
SD, standard deviation; SEM, standard error of the mean.
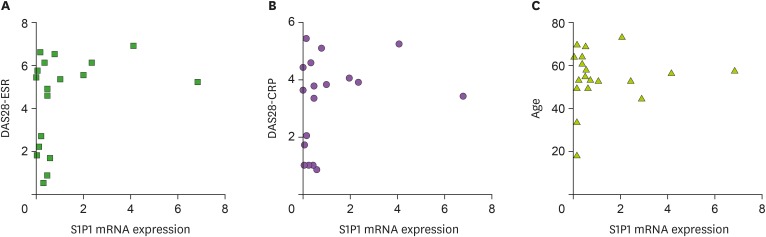
We bled these patients and healthy subjects, and isolated leukocytes from the blood. To focus on the role of S1P1 in T cells and other major inflammatory leukocytes, we depleted B cells from the purified leukocytes. Total RNA from these remaining leukocytes were extracted and analyzed for the expression of S1P1 mRNA by employing qRT-PCR. First, we compared the expressions of S1P1 mRNA to the severity of the disease, because the expressions of this receptor may be related to the progression of the disease. To our surprise, the amount of S1P1 mRNA was not correlated to any clinical scores including DAS28-ESR and DAS28-CRP (Fig. 1A and B). We also found that the expression level of S1P1 mRNA was not correlated with patient's age (Fig. 1C) or the duration of the disease (data not shown). These data may indicate that the expression of S1P1 in the blood leukocytes does not influence the severity of the disease in any age of these patients.
Figure 1. S1P1 mRNA expression in the blood leukocytes of RA patients. The expression of S1P1 mRNA was quantified using qRT-PCR and compared to DAS28-ESR (A), DAS28-CRP (B) and age (C). We obtained similar results in 5 sets of different experiments.
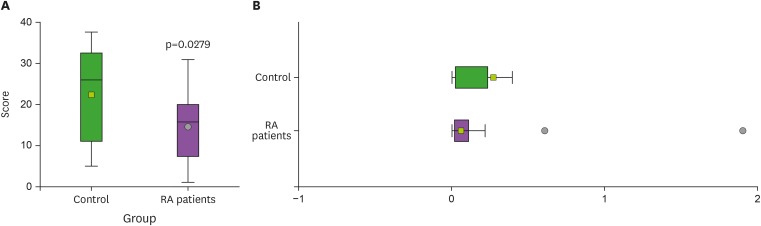
Next, we compared the level of S1P1 mRNA expression in the blood leukocytes between RA patients and healthy controls. As shown in Fig. 2, the amounts of S1P1 mRNA from the leukocytes of the RA patients were significantly less than in controls. Based on these results, we concluded that the expression of this receptor in the blood leukocytes may not promote the development and maintenance of this disease. Instead, S1P1 may be important to regulate the development of the disease.
Figure 2. Differential expressions of the S1P1 mRNA between RA patients and healthy controls. The amounts of S1P1 mRNA in the leukocytes were quantified using qRT-PCR. (A) Comparison between the patient group and the control group was examined by Wilcoxon rank sum test with box-and-whisker plot of rank scores. (B) Box-and-whisker plot was used for visualizing distribution of the S1P1 mRNA. These data are the representative of 3 independent experiments. Shown are mean±SEM.
SEM, standard error of the mean.
In this study, we examined the expressions of S1P1 in the leukocytes of RA patients in the mRNA level. We found that the level of S1P1 mRNA expression was not correlated with the severity and duration of the disease and the age of the patients. In addition, the amount of S1P1 mRNA from the blood samples of RA patients was significantly lower compared to the samples of healthy controls. These data suggest that S1P1 in the blood leukocytes is involved in the development of RA.
This finding is paradoxical considering that the expression of S1P is upregulated in the joints of RA patients (8), and that the S1P1 expression in the synovium is important for the pathogenesis of this disease (9,17). In addition, an antagonist of S1P1 has been shown to ameliorate the severity of the disease, stressing the importance of this receptor in the progress of RA (18). After careful examination of those data, a few explanations can be proposed.
First, it has not been demonstrated that the antagonist of S1P1 affects the function of S1P1 in the blood. If the role of S1P1 in the circulation is not significant, the recruitment of inflammatory leukocytes into the joints may not be dependent on the chemotaxis of S1P. Therefore, it will be interesting to see whether the interaction between S1P and S1P1 is crucial in recruiting inflammatory cells into the joints of RA patients.
Another possible explanation is that the recruitment of inflammatory cells is mediated by S1P receptors other than S1P1. S1P has 5 different receptors (7) and other receptors have not been examined for their roles in the pathogenesis of RA. Therefore, further experiments will be necessary to understand the role of S1P receptors during the pathogenesis of RA.
A third explanation revolves around S1P1's importance in recruiting inflammatory cells to the joints in the onset of RA. Since the patients who participated in our study suffered this disease for a minimum 6 months estimated, it is safe to assume that the disease has been established in these patients. Therefore, pathogenic leukocytes have already been recruited at the time of the experiments so that the remaining blood leukocytes produce lower levels of S1P1 mRNA compared to healthy controls.
In addition to these explanations, we still cannot exclude the possibility that the expressions of S1P1 in the blood leukocytes from RA patients are not regulated normally. Since the regulation of S1P1 expression is poorly understood, it will require additional studies to test this hypothesis.
This study was aimed to examine the expression of S1P1 in the leukocytes of the blood from RA patients. Unexpectedly, the expressions of S1P1 mRNA from these cells were lower in RA patients compared to healthy controls, possibly suggesting that this receptor contribute to inhibit the progression of this disease. Therefore, this finding may lead us to further understand the pathogenesis of this autoimmune disease.
ACKNOWLEDGEMENTS
We thank Hyun Gyung Kim and Seung-min Yeon for their critical comments. This study was supported by Basic Science Research Program (grant No. NRF-2017R1A2B4003376) and Basic Research Lab Program (grant No. NRF-2014R1A4A1007304) of the National Research Foundation of Korea funded by the Korea government.
Abbreviations
- ACR
American College of Rheumatology
- CRP
C-reactive protein
- ESR
erythrocyte sedimentation rate
- PBMC
peripheral blood mononuclear cell
- qRT-PCR
quantitative RT-PCR
- RA
rheumatoid arthritis
- S1P
sphingosine-1-phosphate
- S1P1
sphingosine-1-phosphate receptor 1
- Th
T helper
Footnotes
Conflict of Interest: The authors declare no potential conflicts of interest
Author Contributions: Conceptualization: Choi HS, Kim KH, Kang SW, Jung YW; Formal analysis: Choi HS, Kim KH, Jin S, Yuk SH, Kang SW, Jung YW; Investigation: Choi HS, Kim KH, Ha UH, Kang SW, Jung YW; Resources: Kim J, Yu I, Pack SP, Park TW, Choi SA; Supervision: Ha UH, Kang SW, Jung YW; Writing - original draft: Choi HS, Kim KH, Kang SW, Jung YW; Writing - review & editing: Choi HS, Kim KH, Kang SW, Jung YW.
References
- 1.Guo Q, Wang Y, Xu D, Nossent J, Pavlos NJ, Xu J. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018;6:15. doi: 10.1038/s41413-018-0016-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Combe B. Progression in early rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2009;23:59–69. doi: 10.1016/j.berh.2008.11.006. [DOI] [PubMed] [Google Scholar]
- 3.El Miedany Y, Youssef S, Mehanna AN, El Gaafary M. Development of a scoring system for assessment of outcome of early undifferentiated inflammatory synovitis. Joint Bone Spine. 2008;75:155–162. doi: 10.1016/j.jbspin.2007.04.021. [DOI] [PubMed] [Google Scholar]
- 4.Zhong W, Zhao L, Liu T, Jiang Z. IL-22-producing CD4+T cells in the treatment response of rheumatoid arthritis to combination therapy with methotrexate and leflunomide. Sci Rep. 2017;7:41143. doi: 10.1038/srep41143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mellado M, Martínez-Muñoz L, Cascio G, Lucas P, Pablos JL, Rodríguez-Frade JM. T cell migration in rheumatoid arthritis. Front Immunol. 2015;6:384. doi: 10.3389/fimmu.2015.00384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choy E. Understanding the dynamics: pathways involved in the pathogenesis of rheumatoid arthritis. Rheumatology (Oxford) 2012;51(Suppl 5):v3–v11. doi: 10.1093/rheumatology/kes113. [DOI] [PubMed] [Google Scholar]
- 7.Cyster JG, Schwab SR. Sphingosine-1-phosphate and lymphocyte egress from lymphoid organs. Annu Rev Immunol. 2012;30:69–94. doi: 10.1146/annurev-immunol-020711-075011. [DOI] [PubMed] [Google Scholar]
- 8.Kitano M, Hla T, Sekiguchi M, Kawahito Y, Yoshimura R, Miyazawa K, Iwasaki T, Sano H, Saba JD, Tam YY. Sphingosine 1-phosphate/sphingosine 1-phosphate receptor 1 signaling in rheumatoid synovium: regulation of synovial proliferation and inflammatory gene expression. Arthritis Rheum. 2006;54:742–753. doi: 10.1002/art.21668. [DOI] [PubMed] [Google Scholar]
- 9.Takeshita H, Kitano M, Iwasaki T, Kitano S, Tsunemi S, Sato C, Sekiguchi M, Azuma N, Miyazawa K, Hla T, et al. Sphingosine 1-phosphate (S1P)/S1P receptor 1 signaling regulates receptor activator of NF-κB ligand (RANKL) expression in rheumatoid arthritis. Biochem Biophys Res Commun. 2012;419:154–159. doi: 10.1016/j.bbrc.2012.01.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO, 3rd, Birnbaum NS, Burmester GR, Bykerk VP, Cohen MD, et al. 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010;62:2569–2581. doi: 10.1002/art.27584. [DOI] [PubMed] [Google Scholar]
- 11.Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Fries JF, Cooper NS, Healey LA, Kaplan SR, Liang MH, Luthra HS, et al. The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum. 1988;31:315–324. doi: 10.1002/art.1780310302. [DOI] [PubMed] [Google Scholar]
- 12.Prevoo ML, van't Hof MA, Kuper HH, van Leeuwen MA, van de Putte LB, van Riel PL. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995;38:44–48. doi: 10.1002/art.1780380107. [DOI] [PubMed] [Google Scholar]
- 13.van Riel PL, van Gestel AM. Clinical outcome measures in rheumatoid arthritis. Ann Rheum Dis. 2000;59(Suppl 1):i28–i31. doi: 10.1136/ard.59.suppl_1.i28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chang Y, Kang SY, Kim J, Kang HR, Kim HY. Functional defects in type 3 innate lymphoid cells and classical monocytes in a patient with hyper-ige syndrome. Immune Netw. 2017;17:352–364. doi: 10.4110/in.2017.17.5.352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jung YW, Rutishauser RL, Joshi NS, Haberman AM, Kaech SM. Differential localization of effector and memory CD8 T cell subsets in lymphoid organs during acute viral infection. J Immunol. 2010;185:5315–5325. doi: 10.4049/jimmunol.1001948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wilcoxon F. Individual comparisons of grouped data by ranking methods. J Econ Entomol. 1946;39:269–270. doi: 10.1093/jee/39.2.269. [DOI] [PubMed] [Google Scholar]
- 17.Lai WQ, Melendez AJ, Leung BP. Role of sphingosine kinase and sphingosine-1-phosphate in inflammatory arthritis. World J Biol Chem. 2010;1:321–326. doi: 10.4331/wjbc.v1.i11.321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hu PF, Chen Y, Cai PF, Jiang LF, Wu LD. Sphingosine-1-phosphate: a potential therapeutic target for rheumatoid arthritis. Mol Biol Rep. 2011;38:4225–4230. doi: 10.1007/s11033-010-0545-9. [DOI] [PubMed] [Google Scholar]