Abstract

In the 2000s, nail polish manufacturers started promoting “3-Free” products, phasing out three widely publicized toxic chemicals: toluene, formaldehyde, and dibutyl phthalate (DnBP). However, DnBP was sometimes replaced by another endocrine-disrupting plasticizer, triphenyl phosphate (TPHP). Many new “n-Free” labels have since appeared, without any standardization on which n chemicals are excluded. This study aimed to compare measured plasticizer content against nail polish labels. First, we summarized definitions of labels. Then, we measured 12 phthalate and 10 organophosphate plasticizers in 40 nail polishes from 12 brands selected for popularity and label variety. We found labels ranging from 3- to 13-Free; 10-Free was the most inconsistently defined (six definitions). Our samples contained TPHP and bis(2-ethylhexyl) phthalate (DEHP) at up to 7940 and 331 μg/g, respectively. The 5- to 13-Free samples had lower TPHP levels than unlabeled or 3-Free samples (median <0.002 vs 3730 μg/g, p < 0.001). The samples that did not contain TPHP had higher DEHP levels (median 68.5 vs 1.51 μg/g, p < 0.05). We measured plasticizers above 100 μg/g in five brands that did not disclose them and in two that excluded them in labels. This study highlights inconsistencies in nail polish labels and identifies TPHP and DEHP as ingredient substitutes for DnBP.
Introduction
In the early 2000s in the U.S., many nail polish products were found to list dibutyl phthalate (DnBP) as an ingredient.1,2 DnBP is a plasticizer that improves the flexibility of the nail polish film, prevents the polish from chipping, and holds color.3,4 Concerns grew because animal studies had identified DnBP as a reproductive and developmental toxicant,5−7 and a research study in the U.S. in 2000 had shown that women of reproductive age experience much higher exposures to DnBP than other age or gender groups.8,9 Furthermore, the European Union banned DnBP in cosmetics in 2004.10 By 2006, companies in the U.S. started voluntarily labeling nail polish as “3-Free,” meaning the nail polish does not contain three widely publicized, so-called “toxic trio” chemicals: DnBP, toluene (a developmental and neurological toxicant), and formaldehyde (a human carcinogen).11−15
The reported removal of DnBP has been promising given growing evidence of adverse effects on reproductive health, fetal development, thyroid function, and obesity.16−21 However, the California Environmental Protection Agency (Cal EPA) analyzed seven 3-Free nail polishes and found that one still contained DnBP at 82 000 ppm and four contained toluene at up to 180 000 ppm (2012). In addition, the most common plasticizer ingredient detected in the absence of DnBP was triphenyl phosphate (TPHP),12 an ingredient listed in about half of nail polishes between 2012 and 2015.22,23 Only more recently has TPHP been shown to be an endocrine disruptor that can adversely affect thyroid function and reproductive health in humans.24−28 This new evidence raises concern that one toxic chemical (DnBP) has been replaced by another (TPHP). This phenomenon in public health is called regrettable substitution, in which toxic chemicals are replaced with compounds (sometimes of the same chemical class) that are later found to also be toxic.29,30
Regrettable substitution can occur because the U.S. lacks a regulatory structure to motivate proactive consideration of health risks of replacement chemicals.2 The Food and Drug Administration (FDA) regulates cosmetics but does not require products to be tested for safety or approved before entering the market (except for color additives).31 The FDA holds the burden of proof to show that an ingredient harms health, so less than a dozen ingredients have been prohibited or restricted since 1938.32,33 Labeling and advertising claims such as 3-Free or nontoxic also do not need premarket approval by the FDA or the Federal Trade Commission (FTC). Although these agencies have the authority to take action (e.g., through lawsuits) against products for deceptive claims, the burden again lies with them to prove the deception.31,34 In effect, there is very limited regulation and enforcement of nail polish products.
Although the FDA does require ingredient labels on nail polish sold to consumers (but not salon-only polish), phthalates can be listed as “fragrance” due to trade secret concerns.35,36 The FDA exempts “incidental ingredients” from disclosure requirements, for example when the substance is a component of another ingredient and has no functional effect in the product.37 For nail polish used professionally in nail salons, the Occupational Safety and Health Administration (OSHA) requires chemical hazard information sheets, called Safety Data Sheets (SDS), to be available to workers. However, the SDS only needs to disclose ingredients that have been tested and shown to be hazardous to health above a certain concentration.38
The limited regulation of ingredients in nail polish may leave nail polish users and nail salon workers vulnerable. Consumers that use nail polish mostly consist of women, often of child-bearing age, and even children.39−41 Market research suggests that consumers apply nail polish two to six times per month on average, or as much as once per day.39,40 Two studies of pregnant or lactating women found that urinary concentrations of the DnBP metabolite were significantly higher among recent or frequent nail polish users.42,43 In addition to nail polish users, there are about 400 000 active licensed nail technicians in the U.S., mostly female (97%), Vietnamese (56%), and aged 45 years or younger (54%). At least 45% of the technicians work over 30 h per week and have worked in the field for over eight years,41 so nail salon workers may face higher and more long-term exposures than consumers. Research has shown that nail salon workers have higher urinary levels of the DnBP metabolite cross-shift and compared to the general population,3,44 and that they may have an increased risk of negative birth outcomes and maternal complications.45,46 The main route of exposure for plasticizers like DnBP is dermal absorption from skin contact with the product, settled dust, or contaminated surfaces.3,23 Many plasticizers are semivolatile, and inhalation and dermal uptake from the air have been suggested as additional exposure routes.47−49
A better understanding of nail polish ingredients is needed to effectively inform industry, consumers, and nail salon workers about how to best design and select nail polishes. The objectives of this study are to (1) summarize the types of plasticizer-related labels for nail polish, and (2) measure concentrations of 22 plasticizers in 40 nail polish samples to compare by brand, label, and reported plasticizer ingredient information.
Methods
Nail Polish Labels
To evaluate the different nail polish labels on the market (Objective 1), we conducted a field and Internet investigation of U.S. nail polish products between July 2016 and March 2018. For the field investigation, we identified nail polish brands available at two leading online beauty retailers and at three Greater Boston beauty supply stores. We also observed nail polish brands used at eight Greater Boston nail salons recruited for convenience by our parent exposure assessment study. For each of these brands, we examined the online descriptions of nail polish ingredients and chemical-related labels (e.g., 3-Free). In this manuscript, the term “label” refers to claims listed on the product itself or advertised by the company (e.g., in online product descriptions). When brands had more than one nail polish product line (i.e., a nail polish product category that typically shares one label and ingredient list but consists of several different color options grouped together), we evaluated the product lines individually. Then, we expanded our investigation with a multistep online search of labels. We started with the Google search keywords “nail polish,” “3-Free,” and “nontoxic,” and iteratively expanded our search terms to include each other label type discovered. In the searches, we again identified nail polish product lines with relevant labels.
We verified and defined the identified labels based on online retail product descriptions, brand webpages, or brand social media posts. When the definition of a label was not explained in the descriptions (e.g., if a product line was labeled 3-Free without listing the three excluded chemicals), we determined the definition from conversations with customer service or other online information. When certain brands advertised different labels for different product lines or changed a product line’s label over time, we included each unique label from each brand in the data. Undefined labels were excluded from our assessment.
For each nail polish product line, we recorded the label, the definition of the label, and when possible the associated retail price. Prices were sometimes unable to be identified for labels that product lines had previously used but had since updated. In this manuscript, the term “excluded ingredient” refers to a plasticizer that the label indicated was not used in the nail polish. The term “disclosed ingredient” refers to a plasticizer that was listed as an ingredient in any of the available nail polish information.
Plasticizer Ingredient Levels
We selected 40 nail polishes for the analysis of plasticizers (Objective 2). We employed a multilevel selection strategy to ensure the samples were representative of both the nail polish market and the different label types. First, we selected the most commonly used nail polish brands (regardless of any labels), identified as brands available at online beauty retailers, local beauty supply stores, and local nail salons visited. Then, we selected nine additional brands to ensure our set of samples spanned as many labels identified by early 2017 in our investigation as possible. When a given label was advertised (and equivalently defined) by multiple different brands, we selected the brand that was available on the online retailers and/or in the local nail salons, as a potential indicator of more common use by consumers or workers. When a label was defined differently by different brands, we included two brands in the set of nine. For the most common label, we also included two brands. One of our final selected brands was a UV-cured gel-like nail polish.
For each of the final 12 selected brands, we randomly selected two color coats and one special coat or “finish.” For colors, we chose from red or pink shades, the most popular colors as reported by five local nail salon workers during our nail salon visits. For finishes, we chose from available metallic, shimmer, or glitter coats. In addition, a clear top coat was randomly selected for each of the three most common nail polish brands and for the brand with the reportedly safest label.
The 40 nail polish samples selected were acquired in April 2017 from online retailers or as donated samples. All samples were consumer grade and could be purchased by any consumer. The samples were shipped in sealed, new, and unopened glass bottles to the Wadsworth Center at the New York State Department of Health (Albany, NY) for the analysis of plasticizers.
Each nail polish sample was analyzed for 12 phthalate and 10 organophosphate plasticizers: dimethyl phthalate [DMP], diethyl phthalate [DEP], butyl benzyl phthalate [BBP], di-n-butyl phthalate [DnBP], diisobutyl phthalate [DiBP], bis(2-ethylhexyl) phthalate [DEHP], di-n-hexyl phthalate [DnHP], dicyclohexyl phthalate [DCHP], di-n-octyl phthalate [DnOP], di-isononyl phthalate [DiNP], bis(2-ethylhexyl) adipate [DEHA], bis(2-ethylhexyl) terephthalate [DEHtP], triphenyl phosphate [TPHP], tris(methylphenyl) phosphate [TMPP], triethyl phosphate [TEP], tripropyl phosphate [TPP], tris(2-chloroethyl)phosphate [TCEP], tris(1-chloro-2-propyl)phosphate [TCIPP], tris(1,3-dichloro-2-propyl)phosphate [TDCIPP], p,p′-1,3-phenylene p,p,p′,p′-tetraphenyl ester phosphate [PBDPP], 2-ethylhexyl diphenyl phosphate [EHDPP], and tris(4-butylphenyl)phosphate [TBPhP]. Twelve phthalate diesters and 11 deuterated internal standards were measured in nail polish sample extracts using a gas chromatograph (Agilent Technologies 6890N; Agilent Technologies, Santa Clara, CA) coupled with a mass spectrometer (Agilent Technologies 5973 MSD) in the selected ion monitoring mode. Nail polish sample extracts were also analyzed for 10 organophosphate plasticizers and eight corresponding deuterated compounds using high-performance liquid chromatography (Agilent 1100 series HPLC; Agilent Technologies, Santa Clara, CA) coupled with electrospray triple quadrupole mass spectrometry (API 2000, Applied Biosystems, Foster City, CA). We analyzed two procedural blanks for the target compounds and only detected trace levels of BBP, DEHA, and DEHP (0.16 to 2.47 ng/g). We subtracted the average values in blanks from the sample values. Two matrix spikes (50 ng) were analyzed, which showed recoveries of target chemicals ranging from 85 to 121%. More information on the laboratory methods is detailed in the Supporting Information (SI).
Label definitions, product ingredient lists, and SDSs for the samples were obtained from the online product descriptions, customer service, or a supplier Web site. When differently dated SDSs were located for a product, we conservatively recorded disclosures from either. We classified the samples based on their labels as “old generation,” for polishes without a label or with the original 3-Free label, or as “new generation,” for polishes with n-Free labels beyond 3-Free.
We evaluated nail polish patent information to determine what concentrations of plasticizers are considered functional (thus, likely intentionally added) for nail polish performance. We searched the United States Patent and Trademark Office database in March 2018 for U.S.-based patents with the abstract keywords “nail,” “polish,” and “plasticizer." The five investigated patents listed preferred total plasticizer levels as the following ranges: 0.01–25%,50 1–5%,51 10–30%,52 3–20%,53 and 0.01–25%.54 We chose 0.01% by weight (100 μg/g), the lowest reported level, as our threshold above which concentrations could be suitable for plasticizer performance. Although the lower bound, this may be a conservative estimate given that patent levels are based on total plasticizer compositions, not individual plasticizer concentrations. In addition, we identified other suitable types of plasticizers (according to the patents) that we did not measure but that were disclosed in ingredient lists of our samples.
Statistical Analyses
We calculated descriptive statistics for nail polish labels and for plasticizer concentrations by brand, label, finish, and reported disclosed/excluded ingredients. We substituted all nondetect measured concentrations with half the LOD; in tables, however, if medians or minimums were calculated to be less than the LOD, they were presented as “<[LOD]”. We then compared measured plasticizer levels of nail polishes with old generation labels (unlabeled or 3-Free) to polishes with new generation labels (chemical-related labels different than 3-Free) using Wilcoxon rank sum tests (chosen due to the non-normality of the data).
To evaluate potential replacement chemicals for TPHP specifically, we performed Wilcoxon rank sum tests for concentrations of plasticizers measured in samples with detected levels of TPHP vs samples without detected levels of TPHP. For the Wilcoxon rank sum tests, we only evaluated plasticizers detected above 100 μg/g (0.01% by weight), the cutoff chosen to represent functional concentrations.
Statistical significance was evaluated at the alpha of 0.05 level for the Wilcoxon rank sum tests. All analyses were performed in R (version 3.3.1).
Results
Nail Polish Labels
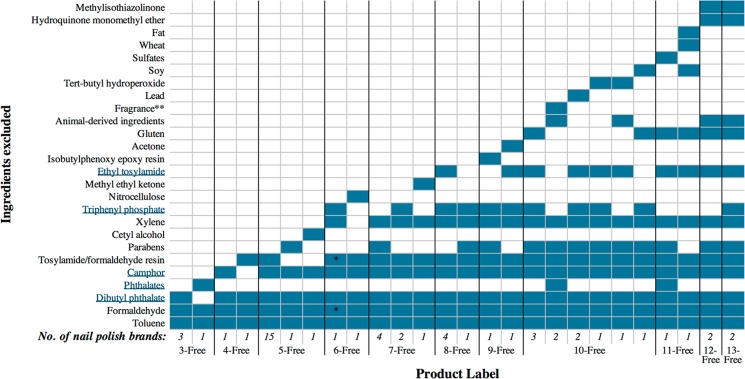
We identified 11 different n-Free labels ranging from 3-Free to 13-Free that reported the exclusion of n ingredients. These labels were used by 55 nail polish product lines comprising 44 unique brands. The definitions of the labels, as reported by the brands, are presented in Figure 1. The 5-Free label was the most commonly used (17 brands), followed by 10-Free (10 brands).
Figure 1.
Product label definitions for investigated nail polish product lines. Note: The blue color represents the ingredients that are removed from the product line, according to the label. Potential plasticizer ingredients are underlined. Nonplasticizer ingredients are not underlined. No. of nail polish brands refers to the number of brands that had a nail polish product line with that particular product label. *These two ingredients were reported to count as one exclusion. **Fragrances can contain plasticizer chemicals.
We identified six different 10-Free label definitions, as well as three different definitions of 5-Free and 7-Free. Although all 10-Free product lines removed seven of the same chemicals (DnBP, toluene, formaldehyde, camphor, tosylamide/formaldehyde resin, parabens, and xylene), the remaining three chemical exclusions varied. For example, TPHP was only excluded in four 10-Free definitions and phthalates were only excluded in one definition. Other exclusions by only some of the 10-Free product lines were ethyl tosylamide, gluten, animal-derived ingredients, fragrance, lead, tert-butyl hydroperoxide, and soy.
Plasticizer-related ingredient exclusions included: DnBP, phthalates, camphor, TPHP, and ethyl tosylamide.50−55 All investigated product lines excluded DnBP, four of which excluded all phthalates. All product lines 5-Free or higher excluded camphor. TPHP began to be excluded from many product lines 6-Free or higher (35% of all labels). Ethyl tosylamide was excluded from 33% of product lines (all 8-Free or higher). Two 10-Free labels also excluded fragrance, an ingredient that can potentially indicate the presence of phthalate plasticizers.35,37 Of the investigated product lines, 20 were advertised as nontoxic in the product description. Five product lines described the polish as safe for kids and/or pregnancy.
Plasticizer Ingredient Levels
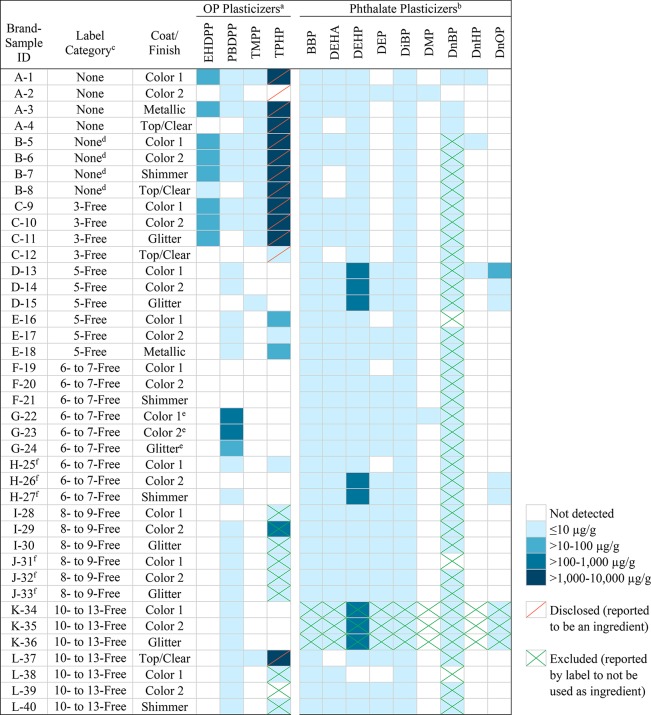
The ranges of measured plasticizer concentrations in the 40 nail polish samples by brand, label category, and coat or finish are summarized in Table 1. Thirteen out of the 22 plasticizers were detected: one plasticizer at levels above 1000 μg/g (TPHP), two at levels above 100 μg/g (DEHP and PBDPP), and two at levels above 10 μg/g (EHDPP and DnOP). The median, range, and percent detected for each detected plasticizer are detailed in Table 2. The maximum concentrations were 7940 μg/g for organophosphates (specifically, TPHP) and 331 μg/g for phthalates (DEHP). TPHP and DEHP were detected in 60% and 98% of samples, respectively. PBDPP was detected above 100 μg/g in samples from just one brand, the UV-cured nail polish. There were no apparent patterns in plasticizer levels by coat or finish type, but we found some variability of the five above-mentioned plasticizers within brands (8–200% coefficients of variation when detected).
Table 1. Product Characteristics, Concentrations of Phthalate and Organophosphate (OP) Plasticizers (μg/g), and Product Information on Chemicals Reported to Be Disclosed or Excluded as Ingredients for 40 Nail Polish Samplesg.
Limits of detection (LODs) for OP plasticizers were 10, 5.0, 0.2, and 2.0 ng/g (in order of appearance).
LODs for phthalate plasticizers were 2.0, 2.0, 1.0, 1.0, 1.0, 0.5, 0.5, 0.5, and 0.5 ng/g (in order of appearance).
Newer labels after 5-Free are described in categories in order to preserve brand anonymity.
Product description reported the exclusion of the toxic trio chemicals without a specific ″3-Free” label.
Semipermanent UV-cured nail polish (but not a full gel polish).
Safety Data Sheet was not acquired.
Note: Plasticizers not detected in any of the samples were TEP, TPP, TCEP, TCIPP, TDCIPP, TBPhP, DCHP, DEHtP, and DiNP.
Table 2. Percent Detected and Median [Range] of Concentrations (μg/g) of Phthalate and Organophosphate (OP) Plasticizers for 40 Nail Polish Samplesa.
| plasticizer | n (%) detected | median [range] | |
|---|---|---|---|
| OPsb | |||
| EHDPP | 9 (22.5) | <0.01 [<0.01, 49.4] | |
| PBDPP | 30 (75) | 0.387 [<0.005, 115] | |
| TMPP | 12 (30) | <0.0002 [<0.0002, 0.379] | |
| TPHP | 24 (60) | 2.73 [<0.002, 7940] | |
| phthalatesc | |||
| BBP | 40 (100) | 1.00 [0.415, 2.13] | |
| DEHA | 35 (87.5) | 0.107 [<0.002, 0.467] | |
| DEHP | 39 (97.5) | 1.65 [<0.001, 331] | |
| DEP | 25 (62.5) | 0.0140 [<0.001, 1.56] | |
| DiBP | 40 (100) | 0.114 [0.00300, 0.778] | |
| DMP | 2 (5) | <0.0005 [<0.0005, 1.57] | |
| DnBP | 36 (90) | 0.021 [<0.0005, 0.138] | |
| DnHP | 3 (7.5) | <0.0005 [<0.0005, 0.918] | |
| DnOP | 8 (20) | <0.0005 [<0.0005, 11.5] | |
Note: < means the calculated value for the median or range fell below the limit of detection.
OP plasticizers not detected in any of the samples were TEP, TPP, TCEP, TCIPP, TDCIPP, and TBPhP.
Phthalate plasticizers not detected in any of the samples were DCHP, DEHtP, and DiNP.
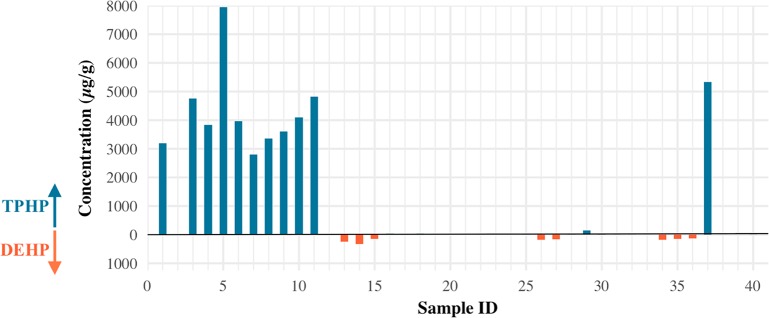
Table 3 presents group comparison test results for concentrations of plasticizers detected above 100 μg/g by label generation and TPHP detection. Concentrations of TPHP were significantly lower in new generation (5- to 13-Free) nail polish samples compared to old generation (unlabeled and 3-Free) samples (median <0.002 vs 3730 μg/g, p < 0.001). Among the nail polish samples without measurable levels of TPHP, concentrations of DEHP were significantly higher compared to samples with measurable TPHP (median 68.5 vs 1.51 μg/g, p = 0.02). Figure 2 highlights the patterns of high DEHP levels only in the absence of TPHP and vice versa.
Table 3. Median [Range] of Concentrations (μg/g) of Phthalate and Organophosphate (OP) Plasticizers Detected above 100 μg/g in at Least One Sample for 40 Nail Polish Samples by Generation of Label and Detection of TPHPa.
| median
[range] |
|||||
|---|---|---|---|---|---|
| plasticizer | type | old generation label (n = 12) | new generation label (n = 28) | detected TPHP (n = 24) | no detected TPHP (n = 16) |
| DEHP | phthalate | 1.61 [0.703, 2.23] | 1.67 [<0.001, 331] | 1.51 [<0.001, 2.50] | 68.5 [0.684, 331]b |
| PBDPP | OP | 0.287 [<0.005, 0.62] | 0.522 [<0.005, 115] | 0.531 [<0.005, 2.27] | 0.151 [<0.005, 115] |
| TPHP | OP | 3730 [<0.002, 7940] | <0.002 [<0.002, 5330]c | ||
Note: Old generation, unlabeled or 3-Free; New generation, 5- to 13-Free; < , calculated value fell below the limit of detection.
Samples with detected TPHP significantly different from samples without detected TPHP at p < 0.05 (Wilcoxon rank sum test).
New generation samples significantly different from old generation samples at p < 0.001 (Wilcoxon rank sum test).
Figure 2.
Side-by-side comparison of concentrations (μg/g) of TPHP (top) vs DEHP (bottom) for 40 nail polish samples.
In these 40 nail polish samples, TPHP was disclosed as an ingredient (in ingredient lists or SDSs) for 13 samples. TPHP was detected in 12 (44.4%) of the 27 nail polishes that did not disclose TPHP as an ingredient (Table 1). These 12 samples composed five unique brands, all 5-Free or higher. Concentrations of TPHP were significantly higher in the nail polishes that disclosed TPHP compared to the polishes that did not (median 3840 vs 3.65 μg/g, p < 0.001). Although SDSs typically did not list plasticizer ingredients, the SDSs that did disclose TPHP described ranges of 0–1%, ≤ 2%, 1–9%, or 5–10% by weight; none of the measured TPHP levels exceeded these reported ranges.
No other phthalates or organophosphates were reported as ingredients in ingredient lists or SDSs for any samples. Overall, nine brands had samples with undisclosed plasticizer levels measured above 10 μg/g. Five of these brands had undisclosed plasticizer levels above 100 μg/g, and none had undisclosed plasticizer levels above 1000 μg/g. In SDSs, DEHP would only have needed to be disclosed at concentrations above 1000 μg/g (0.1% by weight).14,56 In product ingredient lists, plasticizers must be declared if they have a functional effect on the nail polish,37 which we assume to be true above 100 μg/g (based on patents). Fragrance was not disclosed as an ingredient in any sample.
Many samples had labels that reported the exclusion of a phthalate or organophosphate ingredient, which in most cases was consistent with measured levels. Ninety percent (36 out of 40 samples tested) had a label that excluded DnBP, and measured levels did not exceed 0.138 μg/g. One brand’s label listed the exclusion of all phthalates and fragrance; however, the three samples of this brand had levels of DEHP ranging from 134 to 174 μg/g (0.013% to 0.017%, respectively). Three brands (nine nail polish samples) reported the exclusion of TPHP in their labels; however, samples from one of the brands had levels of TPHP up to 153 μg/g (0.015%). The other two brands had lower TPHP levels, at up to 5.20 μg/g and 2.01 μg/g.
We found other suitable plasticizers disclosed in the ingredient lists of the 40 nail polish samples that we did not measure. Of these plasticizers, acetyl tributyl citrate (ATBC) was the most commonly disclosed (in seven of the 12 sampled brands, all of which had new generation labels). Six of these brands also disclosed trimethyl pentanediyl dibenzoate. Other identified plasticizer ingredients in the sampled brands included trimethyl pentanyl diisobutyrate (three brands), ethyl tosylamide (two brands–both conventional), camphor (two brands–both conventional), propylene glycol (one brand), sucrose acetate isobutyrate (one brand), dipropylene glycol dibenzoate (one brand), and propylene carbonate (one brand).
Discussion
Nail Polish Labels
The original practice of promoting nail polishes with a standardized 3-Free label has been replaced with an expanding list of inconsistent n-Free labels. We found that (1) a label can be defined differently by brands, (2) the ingredient exclusions are usually not validated by a third party or justified for their relevance to health, and (3) new label types are often not consistent with preceding labels in regard to the ingredients excluded.
The 5-Free label is now more common than the original 3-Free, and most brands (except for two) use the same definition of 5-Free. The second most common label is 10-Free, but it is inconsistently defined by many brands. Our results showed that the number of ingredient exclusions in n-Free labels does not always translate to the degree of reduced toxicity. For example, the exclusions of animal-derived ingredients, gluten, fat, or soy may not impact nail polish toxicity. Thus, the expanded labels can cause confusion about the meaning of the labels, which originally intended to denote the number of toxic nail polish ingredients removed. Adding to the barriers to understanding labels, in some cases the label definitions were not provided in the product descriptions and had to be acquired from customer service. The rapid expansion of nail polish labels also presents a difficult challenge for research and healthy nail salon recognition programs, as nail polishes free of the toxic trio are largely still the recommendation or certification requirement for salon purchasing decisions.57−62
The available retail prices for nail polish product lines that we evaluated were, on average, $11.70 per fluid ounce for 3-Free brands (range: $2.41–$21.00) and $33.50 per fluid ounce for brands with labels higher than 3-Free (range: $6.00–$60.00). In comparison, the two unlabeled brands we sampled were $16.50 per fluid ounce on average. The higher average price point may be motivating brands to use new generation labels with more exclusions.
Plasticizer Ingredient Levels
Our analysis showed that DnBP was not added as an ingredient in the 40 nail polish samples. The detected levels, all below 1 μg/g, were much lower than had been previously detected by most studies, although our sample selection was weighted toward popularity and label variety (Table 4). In support of this finding, the cosmetics ingredient database (with over 50 nail product brands) by the Environmental Working Group (EWG) only shows one nail polish brand to still disclose DnBP in a few products,22 and that brand actually no longer discloses DnBP in the ingredient lists. Thus, DnBP is likely not currently used in most nail polishes.
Table 4. Maximum Concentrations of Phthalate and Organophosphate (OP) Plasticizers (μg/g) Detected in Nail Polish Products in This Study Compared to Other Studiesa.
| OPs | phthalates |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| country | sample selection | n | TPHP | BBP | DCHP | DEHA | DEHP | DEP | DiBP | DMP | DnBP | DnOP | reference |
| U.S.A. | |||||||||||||
| popularity & labels | 40 | 7900 | 2.1 | ND | 0.47 | 330 | 1.6 | 0.78 | 1.6 | 0.14 | 12 | this study | |
| convenience & TPHP | 10 | 17 000 | Mendelsohnet al (2016) | ||||||||||
| convenience & popularity | 8 | 2.2 | ND | 140 | 9.2 | 59 | 0.22 | 27000 | ND | Guo & Kannan (2013) | |||
| convenience | 4 | ND | ND | ND | 1–100 | ND | 1–100 | ND | ND | Dodson et al (2012) | |||
| convenience & 3-Free | 25 | 25 000 | 88 000 | CalEPA (2012) | |||||||||
| convenience | 24 | ND | ND | ND | ND | 63 000 | Hubinger (2010) | ||||||
| convenience | 6 | 110 | ND | 1100 | 15 000 | 60 000 | Hubinger & Havery (2006) | ||||||
| otherb | |||||||||||||
| Canada | convenience | 20 | ND | ND | 1000 | ND | 0.4 | ND | 24 000 | ND | Koniecki et al (2011) | ||
| China | convenience | 10 | ND | 140 | 80 | 2 | 79 | ND | 5.7 | 5 | Bao et al (2015) | ||
| Korea | convenience | 21 | ND | 25 | 31 | 3900 | Koo & Lee (2004) | ||||||
Note: ND, not detected. Nearly all minimum concentrations in the studies were below the limit of detection (LOD) or not reported. Maximums were selected as metrics due to their reporting across studies.
Only literature in English was searched.
Maximum TPHP levels (7900 μg/g) measured in our 40 samples were also lower than previously reported in other studies. The Cal EPA study found TPHP at 17 000–18 000 μg/g in three of the same brands selected in our study (including the brand with our highest detected TPHP level) (2012). This result suggests that some manufacturers are indeed reducing TPHP content in nail polishes.
The finding that our samples do not contain added DnBP and that TPHP use has decreased demonstrates a public health success in which scientific research about harmful ingredients has successfully reached industry and impacted manufacturing practices. To build on this progress, our study can inform industry attention to replacement chemicals: the fact that certain minimum levels of plasticizers are needed for adequate nail polish performance raises questions about what ingredients are replacing DnBP and TPHP in nail polish. In this study, we detected other phthalate and organophosphate plasticizers in the samples. For example, the nail polishes without measurable TPHP had higher DEHP levels than those with measurable TPHP. Three of those brands contained added DEHP above total plasticizer levels functional for nail polish performance (100 μg/g).50−54 Concentrations of DEHP were higher than had previously been detected in nail polish in the U.S. (Table 4). Of further concern, DEHP was not disclosed as an ingredient at those levels (above 100 μg/g) and in one brand was reported to be excluded in the label. Lower measured levels of DEHP and other plasticizers (i.e., below 100 μg/g) may result from their use as one component in mixtures of multiple plasticizers or due to unintentional impurities from the manufacturing process, contaminated ingredients, or the plastic brush cap.
Similarly to TPHP, DEHP is a semivolatile endocrine-disrupting chemical.16,24−28 DEHP can cause adverse reproductive health effects, impaired development, and metabolic disruption.16 It is also a possible human carcinogen that was banned from cosmetics in the European Union at the same time as DnBP in 2004.10,14 Due to its chemical properties, DEHP may be absorbed more effectively from ingestion or inhalation than through skin contact.63 Dietary intake is considered the primary route of exposure to DEHP for the general population, but inhalation is a concern in occupational settings.63−65 In fact, DEHP has been suggested to persist in indoor environments for years even with ventilation and after removal of the primary source.49 So, for workers in nail salons, inhalation may be a more important route of exposure to DEHP than dermal contact.
Unmeasured plasticizers disclosed in the ingredient lists of our samples included alternatives that have been less studied for toxicity. ATBC, for example, has been found in some animal studies to potentially impact the endocrine system.66−69 We should remain cautious about the understudied toxicity of plasticizers being introduced into nail polish. Overall, our results indicate a possibly recurring pattern of substituting plasticizers without evaluating the replacement for toxicity.
Strengths and Limitations
This study is the first comprehensive evaluation of nail polish labels and their definitions. It is also the first study to assess plasticizers in nail polish by a wide variety of labels. However, there are several limitations to the study. Our results do not provide a comprehensive assessment of plasticizers in nail polishes across the whole nail polish market. For one, it is possible that DnBP is still present in some other products we did not analyze. The Cal EPA investigation conducted in 2011 reported detected DnBP concentrations between 14 000 and 88 000 ppm in nail polish, and these levels were measured in less common brands that we did not sample. We did sample five of the same brands, which did not have detectable DnBP levels in that analysis (possibly because any DnBP had already been removed).12 However, given the lack of DnBP-containing nail polish currently in EWG’s database,22 compared to EWG’s finding of at least 20 nail polish brands with DnBP in the early 2000s,1,2 we can still make inferences that DnBP has largely been phased out in the nail polish market.
Second, during our online research, we investigated nearly 100 different U.S. nail polish brands, so our sampling of a dozen brands may not be generalizable to the large nail polish market. However, we made every effort to include representation by popularity in our study design. The total market share of our sampled brands is not known, but two of the included brands made up about 15% of the nail polish market share in 2015.40 Our study’s measured plasticizer levels may actually be underestimates of the average market levels since we based part of our selection on brands that reported to be less toxic. Even though we did not measure all plasticizer ingredients in the nail polishes studied, we measured 22 chemicals from two different classes, including many organophosphate plasticizers not previously studied in nail polish.12,23
Our label accuracy assessment was limited because we only focused on plasticizers and only four sampled brands reported to exclude TPHP or all phthalates. However, phthalate-free brands are uncommon, and 11 of the 12 selected brands did report to exclude DnBP. Also, our selection strategy was more focused on analyzing nail polish across a variety of label types.
Another limitation is that the samples could contain other plasticizers we did not analyze for, including the alternatives listed in some patents. Finally, we excluded gel polishes (except for one gel-like brand) from the laboratory analysis, and gels may contain different chemical formulations.70 The gel-like nail polish brand in our study was the only brand to contain PBDPP levels above 10 μg/g, which warrants further investigation of gel polishes for plasticizer differences. U.S. nail polish sales have been declining in recent years with a 14% decrease between 2013 and 2015, whereas gel nail polishes have been trending upward.40 Future studies should include other nail product types and analysis of other plasticizer types such as citrates.
In our study, we did not account for potential variability in a single product due to differences in production batches or shipping and storage conditions. However, we analyzed multiple products of different colors and finishes from each selected brand; only two other studies have sampled multiple products (and for only two of their selected brands).12,23 Thus, our study was able to capture the moderate plasticizer variability within some brands. In comparison to other studies, we also analyzed a higher total number of samples. The results of this study could inform larger studies to understand the variability in nail polish plasticizer levels.
Implications
Our study identifies the use of DEHP and TPHP as potential substitutes to the historically common nail polish plasticizer DnBP, even among nail polishes with newer n-Free labels. These n-Free labels seem to be based on a framework of removing chemicals one at a time. As a result, since many brands now report to exclude DnBP, TPHP, and other plasticizers, regrettable substitution of ingredients in nail polish is a growing concern. This is especially true since nail polish formulations may not be able to eliminate all plasticizer additives without sacrifices to product performance. Therefore, nail polish labels would benefit from standardization and validation by a third unbiased party. The labels should exclude ingredients based on a chemical class-based approach (e.g., phthalates and organophosphates as a whole) instead of one ingredient at a time. Then, certified labels could be useful tools for educating nail polish users, nail salon owners, and nail salon workers about toxic chemicals and how to make best purchasing decisions. More broadly, our results reinforce the need for the nail polish industry to remove harmful classes of chemical, test any ingredient substitutes for safety before use, and design safer ingredient alternatives following known validated frameworks.71
Acknowledgments
We would like to acknowledge the Massachusetts Healthy Cosmetology Committee, Nan Pham, Lorri Ducharme, and local nail salons for their help with recruitment and worker interviews, and nail polish manufacturers for some donated samples. We would also like to thank Jose Vallarino, Jaime Hart, Francine Laden, Aaron Specht, and Chensheng Lu for their help. This investigation was made possible by Grant No. T42 OH008416 from the National Institute for Occupational Safety and Health (NIOSH). Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIOSH. This research was also supported by NIH P30ES000002, NIH/NIEHS 2R25ES023635-04, and the Harvard Hoffman Program on Chemicals and Health. D.M.C. is a JPB Environmental Health Fellow at the Harvard Chan School. T.F.W. is supported in part by R01 ES016099 and R01 ES028800.
Supporting Information Available
The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/acs.est.8b04495.
Detailed summary of methods for laboratory analysis (PDF)
The authors declare no competing financial interest.
Supplementary Material
References
- Houlihan J.; Brody C.; Schwan B.. Not Too Pretty: Phthalates, Beauty Products, & the FDA; 2002.
- Houlihan J.; Wiles R.. Beauty Secrets: Does a Common Chemical in Nail Polish Pose Risks to Human Health?; 2000.
- Kwapniewski R.; Kozaczka S.; Hauser R.; Silva M. J.; Calafat A. M.; Duty S. M. Occupational Exposure to Dibutyl Phthalate among Manicurists. J. Occup. Environ. Med. 2008, 50 (6), 705–711. 10.1097/JOM.0b013e3181651571. [DOI] [PubMed] [Google Scholar]
- Schlossman M. L. Modern Nail Enamel Technology. J. Soc. Cosmet. Chem. 1980, 31, 29–36. [Google Scholar]
- Foster P. M. D.; Thomas L. V.; Cook M. W.; Gangolli S. D. Study of the Testicular Effects and Changes in Zinc Excretion Produced by Some N-Alkyl Phthalates in the Rat. Toxicol. Appl. Pharmacol. 1980, 54 (3), 392–398. 10.1016/0041-008X(80)90165-9. [DOI] [PubMed] [Google Scholar]
- Heindel J. J.; Powell C. J. Phthalate Ester Effects on Rat Sertoli Cell Function in Vitro: Effects of Phthalate Side Chain and Age of Animal. Toxicol. Appl. Pharmacol. 1992, 115 (1), 116–123. 10.1016/0041-008X(92)90374-2. [DOI] [PubMed] [Google Scholar]
- Shiota K.; Chou M. J.; Nishimura H. Embryotoxic Effects of Di-2-Ethylhexyl Phthalate (DEHP) and Di-N-Butyl Phthalate (DBP) in Mice. Environ. Res. 1980, 22 (1), 245–253. 10.1016/0013-9351(80)90136-X. [DOI] [PubMed] [Google Scholar]
- Blount B. C.; Silva M. J.; Caudill S. P.; Needham L. L.; Pirkle J. L.; Sampson E. J.; Lucier G. W.; Jackson R. J.; Brock J. W. Levels of Seven Urinary Phthalate Metabolites in a Human Reference Population. Environ. Health Perspect. 2000, 108 (10), 979–982. 10.1289/ehp.00108979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kohn M. C.; Parham F.; Masten S. A.; Portier C. J.; Shelby M. D.; Brock J. W.; Needham L. L. Human Exposure Estimates for Phthalates. Environ. Health Perspect. 2000, 108 (10), A440–2. 10.1289/ehp.108-a440b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EU (European Union). Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products; 2016.
- Breskey J. D. California Again Leading the Way: Cosmetics Safety and Worker Health. Calif. J. Heal. Promot. 2013, 11 (1), vi–viii. [Google Scholar]
- Cal EPA (California Environmental Protection Agency). Summary of Data and Findings from Testing of a Limited Number of Nail Products; 2012.
- EPA (Environmental Protection Agency). Toluene Hazard Summary; 2012.
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans: List of Classifications, Volumes 1–121; 2018.
- Nir S. M.Perfect Nails, Poisoned Workers. New York Times; 2015.
- Casals-Casas C.; Desvergne B. Endocrine Disruptors: From Endocrine to Metabolic Disruption. Annu. Rev. Physiol. 2011, 73, 135–162. 10.1146/annurev-physiol-012110-142200. [DOI] [PubMed] [Google Scholar]
- Ferguson K. K.; McElrath T. F.; Meeker J. D. Environmental Phthalate Exposure and Preterm Birth. JAMA Pediatr. 2014, 168 (1), 61–67. 10.1001/jamapediatrics.2013.3699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hauser R.; Meeker J. D.; Duty S.; Silva M. J.; Calafat A. M. Altered Semen Quality in Relation to Urinary Concentrations of Phthalate Monoester and Oxidative Metabolites. Epidemiology 2006, 17 (6), 682–691. 10.1097/01.ede.0000235996.89953.d7. [DOI] [PubMed] [Google Scholar]
- Huang P.; Kuo P.; Guo Y.; Liao P.; Lee C. Associations between Urinary Phthalate Monoesters and Thyroid Hormones in Pregnant Women. Hum. Reprod. 2007, 22 (10), 2715–2722. 10.1093/humrep/dem205. [DOI] [PubMed] [Google Scholar]
- Schug T. T.; Janesick A.; Blumberg B.; Heindel J. J. Endocrine Disrupting Chemicals and Disease Susceptibility. J. Steroid Biochem. Mol. Biol. 2011, 127, 204–215. 10.1016/j.jsbmb.2011.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swan S. H.; Main K. M.; Liu F.; Stewart S. L.; Kruse R. L.; Calafat A. M.; Mao C. S.; Redmon J. B.; Ternand C. L.; Sullivan S.; Teague J. L. the Study for Future Families Research Team. Decrease in Anogenital Distance among Male Infants with Prenatal Phthalate Exposure. Environ. Health Perspect. 2005, 113 (8), 1056–1061. 10.1289/ehp.8100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EWG (Environmental Working Group). Skin Deep Cosmetic Database http://www.ewg.org/skindeep (accessed Feb 5, 2018).
- Mendelsohn E.; Hagopian A.; Hoffman K.; Butt C. M.; Lorenzo A.; Congleton J.; Webster T. F.; Stapleton H. M. Nail Polish as a Source of Exposure to Triphenyl Phosphate. Environ. Int. 2016, 86, 45–51. 10.1016/j.envint.2015.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carignan C. C.; Mínguez-Alarcón L.; Butt C. M.; Williams P. L.; Meeker J. D.; Stapleton H. M.; Toth T. L.; Ford J. B.; Hauser R. Urinary Concentrations of Organophosphate Flame Retardant Metabolites and Pregnancy Outcomes among Women Undergoing in Vitro Fertilization for the EARTH Study Team. Environ. Health Perspect. 2017, 125 (8), 8. 10.1289/EHP1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu C.; Wang Q.; Liang K.; Liu J.; Zhou B.; Zhang X.; Liu H.; Giesy J. P.; Yu H. Effects of tris(1,3-Dichloro-2-Propyl) Phosphate and Triphenyl Phosphate on Receptor-Associated mRNA Expression in Zebrafish Embryos/larvae. Aquat. Toxicol. 2013, 128–129, 147–157. 10.1016/j.aquatox.2012.12.010. [DOI] [PubMed] [Google Scholar]
- Meeker J. D.; Stapleton H. M. House Dust Concentrations of Organophosphate Flame Retardants in Relation to Hormone Levels and Semen Quality Parameters. Environ. Health Perspect. 2010, 118 (3), 318–323. 10.1289/ehp.0901332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meeker J. D.; Cooper E. M.; Stapleton H. M.; Hauser R. Exploratory Analysis of Urinary Metabolites of Phosphorus-Containing Flame Retardants in Relation to Markers of Male Reproductive Health. Endocr. Disruptors 2013, 1 (1), e26306. 10.4161/endo.26306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preston E. V.; McClean M. D.; Claus Henn B.; Stapleton H. M.; Braverman L. E.; Pearce E. N.; Makey C. M.; Webster T. F. Associations between Urinary Diphenyl Phosphate and Thyroid Function. Environ. Int. 2017, 101, 158–164. 10.1016/j.envint.2017.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherer L. D.; Maynard A.; Dolinoy D. C.; Fagerlin A.; Zikmund-Fisher B. J. The Psychology of “Regrettable Substitutions”: Examining Consumer Judgements of Bisphenol A and Its Alternatives. Health. Risk Soc. 2014, 16 (7–8), 649–666. 10.1080/13698575.2014.969687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmerman J. B.; Anastas P. T. Toward Substitution with No Regrets. Science 2015, 347 (6227), 1198–1199. 10.1126/science.aaa0812. [DOI] [PubMed] [Google Scholar]
- FDA (Food and Drug Administration). Cosmetics: FDA authority over cosmetics: how cosmetics are not FDA-approved, but are FDA-regulated https://www.fda.gov/Cosmetics/GuidanceRegulation/LawsRegulations/ucm074162.htm (accessed Feb 25, 2018).
- FDA. Cosmetics: prohibited & restricted ingredients https://www.fda.gov/Cosmetics/GuidanceRegulation/LawsRegulations/ucm127406.htm (accessed Feb 25, 2018).
- FDA. About FDA: Part II: 1938, Food, Drug, Cosmetic Act https://www.fda.gov/AboutFDA/WhatWeDo/History/FOrgsHistory/EvolvingPowers/ucm054826.htm (accessed Feb 25, 2018).
- FTC (Federal Trade Commission). Rule 16 CFR Part 260: Guides for the use of environmental marketing claims https://www.ecfr.gov/cgi-bin/text-idx?SID=ddcda39ef66f41f82d4b3910f7d5082a&mc=true&node=pt16.1.260&rgn=div5 (accessed Sep 17, 2018).
- FDA. Cosmetics: fragrances in cosmetics https://www.fda.gov/Cosmetics/ProductsIngredients/Ingredients/ucm388821.htm (accessed Feb 25, 2018).
- FDA. Cosmetics: phthalates https://www.fda.gov/Cosmetics/ProductsIngredients/Ingredients/ucm128250.htm (accessed Feb 25, 2018).
- FDA. Cosmetic Labeling Guide https://www.fda.gov/cosmetics/labeling/regulations/ucm126444.htm#clgl (accessed Sep 17, 2018).
- OSHA (Occupational Safety and Health Administration). Hazard communication standard: Safety Data Sheets https://www.osha.gov/Publications/OSHA3514.html (accessed Feb 25, 2018).
- EPA. Exposure Factors Handbook; 2011. [Google Scholar]
- Mintel. Nail Color and Care: US, January 2016; 2016.
- 2017 Big Book Statistics. NAILS Magazine. 2018.
- Buckley J. P.; Palmieri R. T.; Matuszewski J. M.; Herring A. H.; Baird D. D.; Hartmann K. E.; Hoppin J. A. Consumer Product Exposures Associated with Urinary Phthalate Levels in Pregnant Women. J. Exposure Sci. Environ. Epidemiol. 2012, 22 (5), 468–475. 10.1038/jes.2012.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hines E. P.; Calafat A. M.; Silva M. J.; Mendola P.; Fenton S. E. Concentrations of Phthalate Metabolites in Milk, Urine, Saliva, and Serum of Lactating North Carolina Women. Environ. Health Perspect. 2009, 117 (1), 86–92. 10.1289/ehp.11610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hines C. J.; Nilsen Hopf N. B.; Deddens J. A.; Calafat A. M.; Silva M. J.; Grote A. A.; Sammons D. L. Urinary Phthalate Metabolite Concentrations among Workers in Selected Industries: A Pilot Biomonitoring Study. Ann. Occup. Hyg. 2009, 53 (1), 1–17. 10.1093/annhyg/men066. [DOI] [PubMed] [Google Scholar]
- John E. M.; Savitz D. A.; Shy C. M. Spontaneous Abortions among Cosmetologists. Epidemiology 1994, 5 (2), 147–155. 10.1097/00001648-199403000-00004. [DOI] [PubMed] [Google Scholar]
- Quach T.; Von Behren J.; Goldberg D.; Layefsky M.; Reynolds P. Adverse Birth Outcomes and Maternal Complications in Licensed Cosmetologists and Manicurists in California. Int. Arch. Occup. Environ. Health 2015, 88 (7), 823–833. 10.1007/s00420-014-1011-0. [DOI] [PubMed] [Google Scholar]
- Tran T. M.; Kannan K. Occurrence of Phthalate Diesters in Particulate and Vapor Phases in Indoor Air and Implications for Human Exposure in Albany, New York, USA. Arch. Environ. Contam. Toxicol. 2015, 68 (3), 489–499. 10.1007/s00244-015-0140-0. [DOI] [PubMed] [Google Scholar]
- Weschler C. J.; Bekö G.; Koch H. M.; Salthammer T.; Schripp T.; Toftum J.; Clausen G. Transdermal Uptake of Diethyl Phthalate and Di(n-Butyl) Phthalate Directly from Air: Experimental Verification. Environ. Health Perspect. 2015, 123 (10), 928–934. 10.1289/ehp.1409151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weschler C. J.; Nazaroff W. W. Semivolatile Organic Compounds in Indoor Environments. Atmos. Environ. 2008, 42 (40), 9018–9040. 10.1016/j.atmosenv.2008.09.052. [DOI] [Google Scholar]
- Bui H. S.; Li C.; Fairneny S.; Ortega L.; Kanji M.; Hariharan R.. Nitrocellulose-Free Nail Polish Compositions. 9713588, 2011.
- Crescimanno S.Low Haze Film Formers for Top Coat Nail Polish. 9603786, 2016.
- Homma M.; Homma V.. Nail Polish Composition. 9050272, 2014.
- McFadden D.; Steffier L.. Nail Polish Composition and Method of Making a Nail Polish. 9248091, 2014.
- Ortega L.; Bui H. S.; Hariharan R.; Kanji M.. Nitrocellulose-Free Nail Polish Compositions. 9517360, 2010.
- Jefferson J.; Rich P. Update on Nail Cosmetics. Dermatol. Ther. 2012, 25 (6), 481–490. 10.1111/j.1529-8019.2012.01543.x. [DOI] [PubMed] [Google Scholar]
- OSHA. Appendix A to §1910.1200 - health hazard criteria (mandatory) https://www.osha.gov/pls/oshaweb/owadisp.show_document?p_table=STANDARDS&p_id=10100 (accessed Mar 1, 2018).
- BPHC (Boston Public Health Commission). Choose Green & Clean for your next mani-pedi http://www.bphc.org/onlinenewsroom/Blog/Lists/Posts/Post.aspx?List=24ee0d58-2a85-4a4a-855b-f5af9d781627&ID=969&Web=03126e14-4972-4333-b8a3-800cbc1cafce (accessed May 7, 2018).
- Cal EPA. Healthy Nail Salon Recognition Program Guidelines; 2018.
- California Healthy Nail Salon Collaborative. What is a healthy nail salon? https://cahealthynailsalons.org/what-is-a-healthy-salon-1/ (accessed May 7, 2018).
- King County Healthy Nail Salon Recognition Program. Healthy nail salon recognition program: application form 4–5 stars http://www.hazwastehelp.org/health/nail-salons-recog.aspx (accessed May 4, 2018).
- Oregon OSHA. Nail salons http://osha.oregon.gov/OSHAPubs/4783e.pdf (accessed May 4, 2018).
- OSHA. Health hazards in nail salons https://www.osha.gov/SLTC/nailsalons/chemicalhazards.html (accessed Mar 1, 2018).
- ATSDR (Agency for Toxic Substances and Disease Registry). Toxicological profile for di(e-ethylhexyl)phthalate; 2012. [PubMed]
- Fong J. P.; Lee F. J.; Lu I. S.; Uang S. N.; Lee C. C. Estimating the contribution of inhalation exposure to di-2-ethylhexyl phthalate (DEHP) for PVC production workers, using personal air sampling and urinary metabolite monitoring. Int. J. Hyg. Environ. Health 2014, 217 (1), 102–109. 10.1016/j.ijheh.2013.04.002. [DOI] [PubMed] [Google Scholar]
- Kurahashi N.; Kondo T.; Omura M.; Umemura T.; Ma M.; Kishi R. The effects of subacute inhalation of di (2-ethylhexyl) phthalate (DEHP) on the testes of prepubertal Wistar rats. J. Occup. Health 2005, 47 (5), 437–444. 10.1539/joh.47.437. [DOI] [PubMed] [Google Scholar]
- Rasmussen L. M.; Sen N.; Vera J. C.; Liu X.; Craig Z. R. Effects of in Vitro Exposure to Dibutyl Phthalate, Mono-Butyl Phthalate, and Acetyl Tributyl Citrate on Ovarian Antral Follicle Growth and Viability. Biol. Reprod. 2017, 96 (5), 1105–1117. 10.1095/biolreprod.116.144691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rasmussen L. M.; Sen N.; Liu X.; Craig Z. R. Effects of Oral Exposure to the Phthalate Substitute Acetyl Tributyl Citrate on Female Reproduction in Mice. J. Appl. Toxicol. 2017, 37 (6), 668–675. 10.1002/jat.3413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strajhar P.; Tonoli D.; Jeanneret F.; Imhof R. M.; Malagnino V.; Patt M.; Kratschmar D. V.; Boccard J.; Rudaz S.; Odermatt A. Steroid Profiling in H295R Cells to Identify Chemicals Potentially Disrupting the Production of Adrenal Steroids. Toxicology 2017, 381, 51–63. 10.1016/j.tox.2017.02.010. [DOI] [PubMed] [Google Scholar]
- Takeshita A.; Igarashi-Migitaka J.; Nishiyama K.; Takahashi H.; Takeuchi Y.; Koibuchi N. Acetyl Tributyl Citrate, the Most Widely Used Phthalate Substitute Plasticizer, Induces Cytochrome p450 3a through Steroid and Xenobiotic Receptor. Toxicol. Sci. 2011, 123 (2), 460–470. 10.1093/toxsci/kfr178. [DOI] [PubMed] [Google Scholar]
- Baran R. Nail Cosmetics: Allergies and Irritations. Am. J. Clin. Dermatol. 2002, 3 (8), 547–555. 10.2165/00128071-200203080-00005. [DOI] [PubMed] [Google Scholar]
- EPA. Design for the Environment Program Alternatives Assessment Criteria for Hazard Evaluation; 2011.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.