Abstract
Soldiers involved in combat operations worldwide may be subjected to a wide array of tissue-specific injuries of varying degrees, thereby undergoing complicated medical treatments and prolonged rehabilitations. In many cases involving inadequate recovery, soldiers are further mentally traumatized as they can no longer serve their beloved country. In addition, many severe injuries can lead to soldiers being incapacitated for life and unable to perform even the most basic day-to-day activities. Present therapy for combat injuries is majorly aimed at alleviating pain and limiting further tissue damage from secondary infections. Cell-based therapy using stem cells is a promising tissue regenerative source, which will help our soldiers to recuperate from the severe injuries, and in some cases, even continue their service for the country after complete recovery. In this context, we would like to discuss the yet fully untapped potential of induced pluripotent stem cells (iPSCs) in regenerative medicine on the battlefield. In this review, we shall try to explore the rationale behind the use of these cells for military medicine, as well as the conventional and novel approaches to produce them for therapeutic applications. We shall also attempt to elucidate the evolving trends of battlefield injuries throughout history and the ongoing research on regeneration of tissues of specific interest using iPSCs and their potential role in combat medicine in the future. Additionally, we shall also discuss the concept of stem cell bio-banking for military personnel as a personalized safeguard against crippling and traumatic combat injuries.
Keywords: Induced pluripotent stem cells, Military medicine, Cell- and tissue-based therapy, Cellular reprogramming
Introduction
With the further enhancement of human technology and knowledge over the past century, man has invented more sophisticated and devastating weapons to wage warfare against his fellow man. The most obvious outcome of war is the casualty and the death toll it brings. Even today, all around the globe, countless international and civil wars are fought, each resulting in numerous deaths and military personnel left crippled or incapacitated for life. Cell-based therapy involving stem cells provide an attractive approach to revolutionize combat medicine as well as enhance the recovery of soldiers off the battlefield.1 Pluripotent stem cells hold the most potential among all the major types of stem cells, because of their unique ability to differentiate into all somatic cell types of an adult human body. However, therapy using human pluripotent stem cells is still a debatable topic due to the ethical considerations and owing to the difficulty of their acquisition. The remarkable discovery of induced pluripotent stem cells (iPSCs),2 heralded a new era of stem cell research and finally provided a feasible solution to the dilemma of using human pluripotent stem cells for regenerative therapy. This review outlines the major advantages of iPSCs over other major stem cell types as well as the different approaches used for their generation. Also discussed are the wide range of tissue damages in modern warfare and the use of iPSCs for the therapy of specific tissue damage.
iPSCs – definition and potential
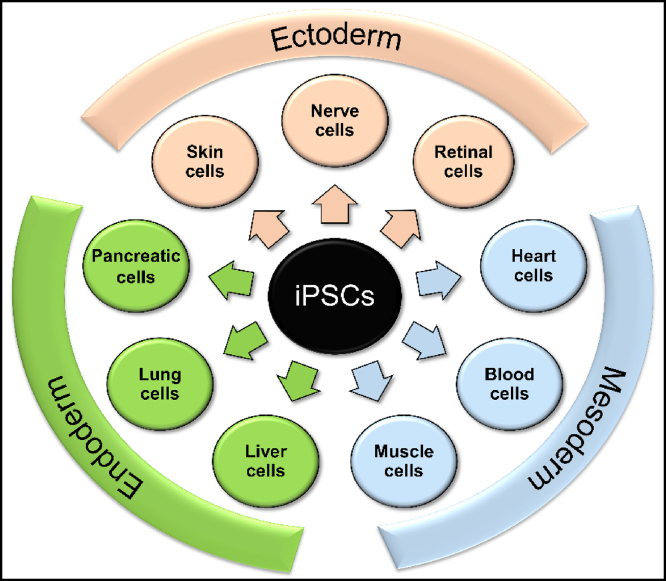
iPSCs are adult somatic cells that are genetically reprogrammed back to an early embryonic stem cell-like state having an active endogenous pluripotent signature program vital for maintaining the defining properties: self-renewal and pluripotency. Though iPSCs apparently appear indistinguishable when compared to embryonic stem cells (ESCs), the two groups can be distinguished by their gene expression signatures. However, like ESCs, iPSCs have the property of self-renewal, i.e., the ability to propagate indefinitely without loss of its undifferentiated state. Also, in a manner similar to ESCs, iPSCs are pluripotent, which is the ability to differentiate into a wide array of different cell types of an adult human body (few representative cell types are shown in Fig. 1). Thus, being able to generate functional, stable, clinical-grade iPSC cell lines may, in theory, give us access to a potentially infinite number of different somatic cell lines for use in cell therapy applications.
Fig. 1.
In vitro differentiation of iPSCs into all three germ layers: ectoderm, mesoderm and endoderm (few cell types are represented for each of this germ layer).
iPSCs vs. other major stem cells
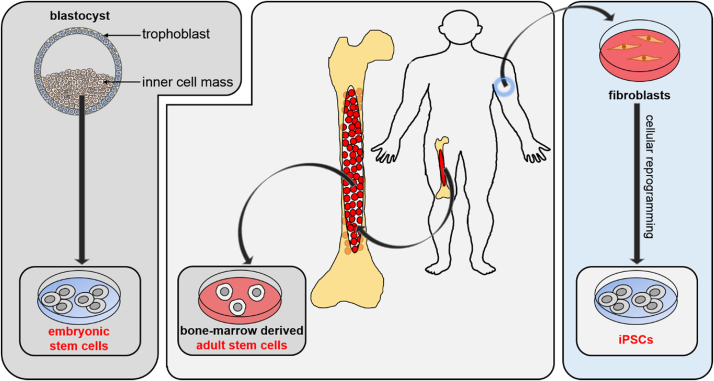
Stem cells are a diverse group of unspecialized cells that include many other cell types besides iPSCs. The major sources of stem cells are illustrated in Fig. 2. Before the discovery of iPSCs, scientists worked primarily with only two major groups of stem cells derived from humans and animals – ESCs and ‘non-embryonic’ Adult Stem Cells (ASCs). Though possessing the two basic features of all stem cells (self-renewal and differentiation potential), these two cell types suffered from major limitations that hindered their widespread application in regenerative medicine.
Fig. 2.
Most widely investigated stem cells and their derivation (embryonic stem cells, adult stem cells and induced pluripotent stem cells).
ESCs are pluripotent cells derived from the inner cell mass of the blastocyst of mouse, human or any other animal. During embryogenesis, the inner cell mass is able to give rise to the primitive ectoderm (epiblast) that will eventually generate the three primary germ layers – endoderm, mesoderm and ectoderm. Thus, these cells isolated from embryonic tissues can self-renew and differentiate into any desired tissue or cells. However, these cells have multiple shortcomings that impair further research into their therapeutic applications. Firstly, these cells are reported to be genetically unstable and prone to chromosomal abnormalities, unless maintained under stringent culture conditions. Secondly, during in vivo regenerative therapy with ESCs, accidental engraftment of partially undifferentiated ESCs into the damaged site poses a risk of teratoma formation. Thirdly, due to the unavailability of natural autologous ESCs for a particular patient, transplantation with allogenic ESCs may result in graft rejection and patient has to undergo prolonged immunosuppression. Last but not the least, there are major ethical considerations regarding the use of oocytes, blastocysts and human embryos for research purposes. This alone has proved to be the major hurdle to ESC research in recent years.
Adult Stem Cells are multipotent cells that are found throughout the body, even after complete development, that play a key role in the natural regeneration of damaged tissue. They are also called somatic stem cells as, unlike their embryonic counterparts, are found in both developing and adult animals and human. However, as opposed to the pluripotency of ESCs, the multipotent ASCs have much lesser plasticity and can only differentiate into cells of a specific lineage. Few extensively investigated major types of ASCs are as follows:
-
a)
Hematopoietic Stem Cells that give rise to all blood cells types, i.e., lymphoid and myeloid lineages.
-
b)
Mesenchymal Stem Cells that can give rise to a diverse range of cell types: chondrocytes (cartilage cells), myocytes (muscle cells), (adipocytes (fat cells) and osteoblasts (bone cells), etc.).
-
c)
Neural Stem Cells that primarily give rise to cells such as oligodendrocytes, astrocytes and neurons.
Though these stem cells have some advantages over ESCs, such as lack of ethical concerns, these cells still suffer from their own limitations. Firstly, the major barrier to widespread ASC therapy is the limited plasticity of ASCs. Since they can differentiate into only specific cell lineages, we can only obtain a handful of specific cell types from a pure population of ASCs. Thus, their therapeutic potential is significantly reduced. Secondly, though most ASCs can be easily isolated, isolation of specific ASC types from their ‘niche’ is quite impractical (e.g. human neural stem cells from the brain). Thirdly, ASCs have a relatively limited self-renewal ability and, as such, cannot be cultured ‘indefinitely’ like ESCs or iPSCs. Lastly, the isolation and propagation of a homogenous or ‘pure’ population of ASCs has proven to be quite an arduous task for even the most experienced researchers.
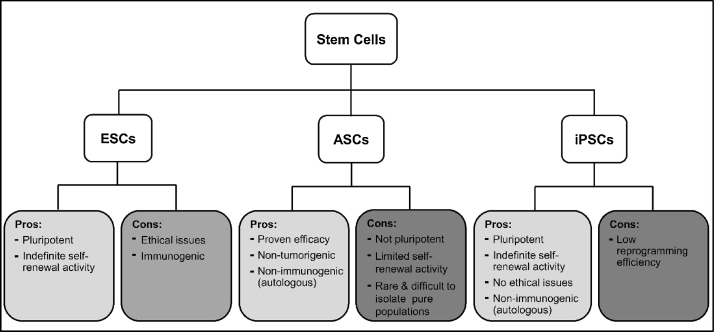
Thus, iPSCs have proven to be quite a lucrative option to researchers working in the field of regenerative medicine as they combine the best features of both ESCs and ASCs. A summary of some of the advantages and disadvantages of each stem cell type has been provided in Fig. 3.
Fig. 3.
An overview of the pros and cons of the three major types of stem cells.
iPSC generation – somatic cell sources
Generating clinical-grade iPSCs for therapeutic purposes also requires careful consideration of the appropriate somatic cell source to be reprogrammed. The molecular properties of the different cell types lead to variations in the reprogramming efficiency.3 Thus, depending on the quality and quantity of iPSCs required, a variety of cell sources can be employed. A few of the human cell types utilized in the recent years for iPSC generation is listed in Table 1.
Table 1.
Comparison of somatic cell sources for iPSC generation.
| Organism | Source cell type | Origin | Invasiveness | Reference |
|---|---|---|---|---|
| Homo sapiens | Dermal fibroblasts | Mesodermal | Invasive | 4 |
| Keratinocytes | Ectodermal | Non-invasive | 5 | |
| Umbilical vein endothelial cells | Mesodermal | Non-invasive | 6 | |
| Renal epithelial cells | Mesodermal | Non-invasive (from urine) | 7 | |
| Cord blood-derived Endothelial cells | Mesodermal | Non-invasive | 8 |
iPSC generation – reprogramming approaches
The core mechanism behind the phenomenon of induced pluripotency is the activation of pluripotency genes coupled with the repression of differentiation genes. In most cases, this is achieved by the ectopic expression of pluripotency related transcription factors such as OCT4, SOX2, KLF4 and c-MYC (referred to as the OSKM cocktail) with or without additional supplementation with chemical modifiers such as valproic acid. Since the generation of the first iPSCs by Shinya Yamanaka and his colleagues in 2006 using retroviral integration of the OSKM cocktail, many novel approaches have been explored to reprogram somatic cells to pluripotency.
Conventional integrating (genetic) approaches using viral (retro- and lenti-viral) vectors with various combinations of transcription factors have been successfully used to generate iPSCs (Table 2). These viral-based gene delivery systems are robust and efficient but they carry an enormous risk of insertional mutagenesis and tumor formation due to random integration of transgenes, thereby eliminating the clinical safety of these cells. To overcome this serious safety issue, tremendous progress has been made in developing non-integrating (non-genetic) techniques (episomal, modified mRNA, micro RNAs, recombinant proteins and small molecules) to generate iPS cells (Table 2). These techniques diminish or exclude the possibility of any alteration in the host genome. However, most of these non-integrative techniques are laborious and are less efficient with slow kinetics and generate large number of partially reprogrammed iPSC-like colonies. Therefore, extensive characterization of iPS clones/cells generated using these techniques is vital to identify “bona fide” iPSCs. Transplantation of “bona fide” iPSCs, generated using safe non-integrative techniques, may still result in tumor formation due to their oncogenic potential. This major problem can be addressed by transplanting desired matured (functional) differentiated derivatives of these iPSCs, instead of iPSCs itself. This coupled with cell sorting technology can separate desired differentiated cells from other differentiated cells or partially differentiated or undifferentiated cells using reliable markers. The other major issue is acquisition of genetic and chromosomal mutations during prolonged iPSC culture due to growth advantage. This can be resolved by thorough examination of the most intact iPS clones, free from any genetic and chromosomal mutations checked by full genome sequencing assays or by high-resolution chips. These crucial steps will safeguard patients from any adverse effects before regenerative therapy applications and will translate iPSC-based treatments routine practices in clinic.
Table 2.
Conventional and novel approaches for iPSC generation.
| Delivery method | Gene cocktail | Somatic cell source | Efficiency | Reference |
|---|---|---|---|---|
| Integrative approachesa | ||||
| Retrovirus | OSKM | Human fibroblasts | Moderate | 4 |
| Lentivirus | OSLN | Human neonatal foreskin fibroblasts | Moderate-high | 9 |
| Non-integrative approachesb | ||||
| Episomal vectors | OSKML + TSV40/N | Human cord blood CD34+ cells | Low | 10 |
| mRNA | OSLN | Human foreskin fibroblasts | Moderate-high | 11 |
| microRNA | None | Mouse adipose stromal cells, human fibroblasts | Moderate-high | 12 |
| Proteins | OSKM | Human neonatal fibroblasts | Low | 13 |
| Small molecules | None | Mouse embryonic fibroblasts | Moderate | 14 |
O, octamer-binding transcription factor 4 (OCT4); S, SRY (sex determining region Y)-box 2 (SOX2); K, Kruppel-like factor 4 (KLF4); M, c-MYC; N, NANOG; L, Lin-28 homolog A (LIN28); TSV40, Simian Virus 40 Large T-antigen.
Clinically unsafe approaches.
Clinically safe approaches but extensive characterization is required before autologous/allogenic transplantation.
Types of tissue-specific injuries in modern combat and their remediation using iPSC therapy
Penetrative ocular injuries
Before the advent of 20th century modern warfare, eye injuries were relatively rare on the battlefield. The incidence of ocular injuries increased to 2.0–2.5% in World War I (1914–18) and World War II (1939–45), 6–10% in the Middle East (1967–73), 5–9% in the Vietnam conflict (1962–72) and as high as 13% in Operation Desert Shield and Storm (1990–91), mainly because of the increasing use of fragment-producing munitions and projectiles.15 In addition to the destructive ocular injuries caused by bullets and shrapnel from hand-grenades, many soldiers in World War I were exposed to chemical agents like mustard gas by the Germans which resulted in episcleritis, keratitis and chemical conjunctivitis. As per the information from the Optometry Consultant for the Army Surgeon General, during Operation Iraqi Freedom and Operation Enduring Freedom, the three destructive approaches (rocket-propelled grenades (RPGs), shrapnel and improvised explosive devices (IEDs)) for causing injuries resulted in approximately 70% of all injuries. Herein, IEDs were the main cause of over half of all eye injuries between March 2003 and December 2004. The globe and orbit are the locations most commonly affected during ocular injuries.
This type of penetrative and traumatic injuries causes irreparable damage to the inner components of the eyes including the retina, choroid and optic nerve as well as the outer sclera. Since retinal photoreceptors and optic neurons cannot spontaneously regenerate, permanent vision loss is the most probable outcome.
However, these retinal cells may be regenerated using iPSC therapy. Of all the generated retinal cells, a significant progress in deriving functional retinal pigment epithelial (RPE) cells is from iPSCs. Many research groups have successfully differentiated iPSCs into RPE cells either by Embryoid Body formation or monolayer culture method.16 These pigmented RPE cells are vital for absorbing scattered light penetrating the eye and retaining visual function. There have also been substantial improvements in generating cone and rod photoreceptors from pluripotent cells, as well integrating these cells in host retina to partially restore vision in mouse models.17 Another important retinal cell type that have been generated from human iPSCs (hiPSCs) are retinal ganglion cells which are a type of neuron that receive visual information from photoreceptors.18 Recently, a clinical trial successfully tested the viability of autologous RPE cells differentiated from iPSCs derived from patients with age-related macular degeneration. A sheet of patient-specific RPE cells were successfully derived from episome-generated iPSCs and transplanted onto their donors without any adverse after-effects.19 The ability to generate such a large variety of crucial cell types provides a great impetus to regenerative therapy for even the most severe ocular injuries.
Auditory damage
According to the Department of Veterans Affairs, auditory injury is the major disability in the war on terror, and few experts say the true toll may possibly take decades to identify. A good numbers of Marines and Soldiers trapped in roadside bombings and firefights in Afghanistan and Iraq are coming back home with ringing in their ears and long-lasting loss in hearing. This type of long-term damage is a result of not only armaments and explosives used by enemies but also by weapons, equipment and vehicles the soldiers use themselves.
According to the National Institute of Health, extensive or recurrent acquaintance to sounds at or above 85 decibels can result in loss in hearing. Auditory loss in the military results from mechanical and biochemical injury to the outer and inner ear, generally in the form of loss of sensory hair. This type hair cell loss can be reversed by regeneration with reprogrammed cochlear hair cells derived from iPSCs. While there are multiple well-established and efficient procedures for stepwise differentiation of retinal photoreceptor cells, there are only a few similar instances for cochlear cells, such as the generation of mechanosensitive hair cell-like cells from iPSCs.20 In other studies, iPSC-derived neural progenitors that were co-cultured with cochlear explants made synaptic connections with the sensory hair cells21 or could be successfully transplanted into the cochlea of neonatal mice.22
Nervous tissue damage
In the context of warfare, there are three major types of nervous tissue injuries – Traumatic Brain Injury (TBI), Spinal Cord Injury (SCI) and Peripheral Nerve Trauma. Owing to the structural complexity of an organ such as the brain, it is very difficult to accurately assess the severity of an external injury on the internal workings, and even more difficult to reverse this damage. As such, damage to inner brain tissue is presently irreversible with long-term side-effects even after prolonged restorative surgery. Exposure to improvised explosive devices may cause mild traumatic brain injury (mTBI) which may result in concussion and/or sub-concussion. In military settings, most traumatic brain injuries (TBIs) are mild TBIs (mTBIs). The occurrence of mTBI among returning service members is estimated to be 15.2–22.8%, impacting as many as 320,000 soldiers. Spinal Cord Injuries (SCIs) are also physically devastating with the cervical spine and lumbar spine being the most commonly affected areas. Mechanistically, TBI and SCI are similarly characterized by a first wave of damage caused by the primary injury itself, and a secondary wave or cascade that is modulated by ongoing inflammation, neuronal cell death, fluid and electrolyte imbalances, free radical damage, and cerebral edema that leads to further persistent damage. Peripheral Nerve Trauma is another common injury type that is sustained during wartime. Traumatic injury to peripheral nerves give rise to widespread disability across the world.
Multiple studies have been conducted to investigate whether iPSCs can be used for peripheral nerve repair and to treat SCIs, however, similar studies have not been reported for injuries to the brain (TBIs). This is probably owing to the difficulty of cell transplantation therapy in an inaccessible organ such as the brain as well as the technical challenges associated with neural induction and expansion of iPSC-derived NSCs. Studies have demonstrated that mouse iPSC cells can give rise to secondary neurospheres (SNSs) in vitro that generates trilineage neural cells, comprising numerous types of neurons. When grafted into an SCI mouse model, these iPSC-derived SNSs differentiate into astrocytes. neurons and oligodendrocytes, without forming teratomas or other tumors, and promote re-myelination, axonal regrowth and tissue sparing, resulting in improved function.23 Further studies with experimental mouse models for peripheral nerve damage also showed that transplantation of iPSC-SNSs24 could promote functional recovery of damaged sciatic nerve.
Musculoskeletal damage
Musculoskeletal injuries (injuries to bone, muscle, cartilage, connective tissue, etc.) is one of the most commonly occurring injury types observed in combat veterans. In the mid-nineteenth century, Artillery shrapnel, Grapeshot and Gunshot were the major reasons of injury, with the Civil War representing the last major American engagement where less than 10% of fatalities happened due to explosive attacks. Starting with World War I, the occurrence of injuries related to explosion has surpassed 30%, with such wounds outdoing those due to gunshot for each encounter since World War II. Explosive devices of injury, including rocket-propelled grenades and landmines, explosively formed projectiles and enhanced explosive devices, have been identified as the major cause of all musculoskeletal injuries incurred in Afghanistan or Iraq. The destructive force of these devices results in skeletal wounds and severely contaminated soft tissues, predominantly comprising the extremities, in dismounted personnel, which, in the long run causes a high susceptibility for wound-related complications like heterotopic ossification, osteomyelitis, and soft-tissue contractures to develop.25
The major cell types in demand for treatment of musculoskeletal damage include skeletal muscle fiber cells, osteoclasts, osteoblasts and chondrocytes, all of which are derived from a mesodermal lineage. In a study conducted in 2012,26 mouse iPSCs were efficiently differentiated into PDGFR-α+/Flk-1-paraxial mesodermal progenitors using a serum-free defined culture condition. The iPS cell-derived paraxial mesodermal progenitors exhibited chondrogenic, myogenic and osteogenic differentiation potential both in vitro as well as in vivo. In the same study, PDGFR-α+/Flk-1-population of human iPSC-derived paraxial mesodermal progenitors showed differentiation potential into chondrocytes, osteocytes, and skeletal myocytes in vitro. These mesodermal lineage cells can promote muscle and bone regeneration when combined with 3D biomaterials. In another study, co-culture of hiPSC-derived MSCs with hiPSC-derived macrophages on a HA-based 3D composite scaffold, not only induced the osteoclastic differentiation of hiPSC-macrophage but also promoted strong osteogenic activity of hiPSC-MSCs, leading to the demonstration of a successful ectopic bone formation model.27
Skin injuries
Damage to skin is present in almost all combat injuries, ranging from destruction of external epidermal tissue to deep dermal injuries leading to formation of scar tissue. In contrast to most other tissue systems, the skin and its appendages are gifted with excellent regenerative capacity facilitating their incessant self-renewal. It is mainly due to the fact that skin has a large reservoir of various stem/progenitor cells predisposed to generate new epidermis during injury. However, deep-seated penetrative injuries, chemical injuries and higher degree burn injuries may cause loss of these progenitor skin cells leading to decreased regeneration capacity and scar formation. In such cases, autologous transplantation with differentiated skin cells derived from patient-specific iPSCs could help in faster healing and improved recovery rates.
Though research on use of iPSCs for dermatological therapy has been slow, there have been reports that validate the feasibility of the approach. In 2011, researchers28 reported the conversion of 3-factor (3F) or 4-factor (4F) derived human iPSCs into melanocytes by EB formation and subsequent culture in a differentiation media. Each set of derived melanocytes tested positive for melanocytes markers like TYRP1, S100, SLIV, TYR and MITF. Additionally, transmission electron micrography also revealed melanosome formation in the pigmented cells of both 3F and 4F-iPSC derived melanocytes. In another study,29 iPS cells derived from both normal subjects and patients with Recessive Dystrophic Epidermolysis Bullosa were differentiated into keratinocytes using an adapted protocol that was previously used for ESC differentiation into epithelial lineage. The derived cells expressed keratinocyte markers and were used for generation of 3D skin equivalents. In a separate study by the same group,30 they reported a procedure for differentiating iPSCs into dermal fibroblasts that produce type VII collagen and can be used for Recessive Dystrophic Epidermolysis Bullosa treatment. The authors used a EB formation method along with a medium supplemented with ascorbic acid and Tumor Growth Factor β2 (TGFβ2) to accelerate mesodermal differentiation. The derived cell populations expressed fibroblast-associated CD surface markers as well as vimentin and P4HB. Using Western blot analysis, they also showed that the fibroblasts secreted mature type VII collagen and they tested the in vivo functional capacity of iPSC-derived fibroblasts using skin reconstitution chamber assays in immune-compromised mice.
Therefore, iPSC-based or its differentiated derivatives can be used for regeneration of variety of cells to restore these cells and thereby lost function.
Stem cell bio-banking
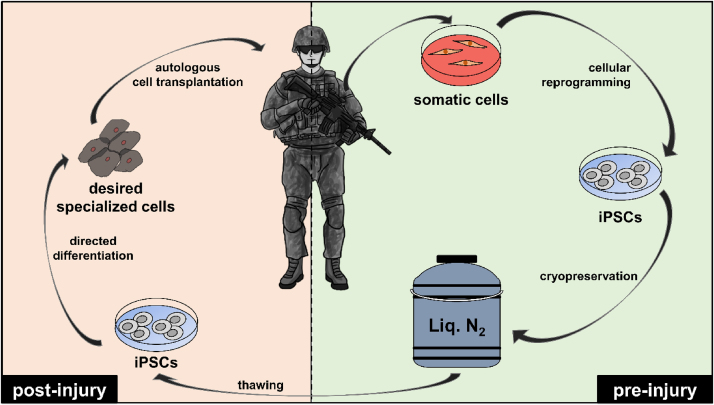
As previously discussed, cell therapy using autologous iPSCs bypass many limitations that are inevitably present in the use of other stem cells types, including immune rejection, ethical concerns and availability. However, application of iPSCs in an on-field combat scenario still presents an unforeseen dilemma, the time limitation required for reprogramming and differentiation. Unlike ESCs and ASCs that only need to be isolated and differentiated before application, human iPSCs require an additional step of pluripotency induction that takes approximately 2–4 weeks. Thus, to circumvent this limitation, a proposed system of iPSC bio-banking may be used. In this system active military personnel, prior to deployment, will provide cell samples (e.g. dermal fibroblasts from a skin biopsy), that will then be reprogrammed to autologous, immune-compatible iPSCs and then cryopreserved for long-term storage. When the need arises, the preserved iPSC samples from the respective donors will be differentiated into appropriate desired cell types and transplanted for regenerative therapy. The various steps involved in the process of iPSC bio-banking are depicted in Fig. 4. As of now, bio-banks are only utilized for storage of infant cord blood and serves as a cell repository for immediate cell therapy, however, this system, in the near future, can be extended to iPSC storage as well. Combined with future developments that may further enhance the efficacy of the system, the present intrinsic potential of therapy-ready, autologous and immune-compatible stem cells makes bio-banking a concept worth serious consideration.
Fig. 4.
Flowchart for the process of iPSC bio-banking for military personnel.
Conclusion
Though originally stem cell therapy was investigated as a possible mode of treatment for certain diseases in civilian population, recent studies into the physiology of combat injuries reveal that regenerative stem cell therapy can provide an efficient and long-lasting treatment for traumatic injuries in military personnel as well.1 Throughout history, the most important advancements in military medicine research have stemmed from the detailed analysis of wartime injuries suffered by on-field personnel. With the development of newer weaponry, the diversity of traumatic and life-threatening combat injuries will only increase, much of which may be benefited by iPSC mediated cell therapies. Though at present, the efficacy and feasibility of these therapies are still a matter of debate, we can hope that in the future, further advancements in stem cell technologies will help direct this revolutionary concept toward the benefit of those who protect our country.
Conflicts of interest
The authors have none to declare.
Acknowledgments
We thank all the members of Laboratory for Stem Cell Engineering and Regenerative Medicine (SCERM) for their excellent support. This work was supported by grants North Eastern Region – Biotechnology Programme Management Cell (NERBPMC), Department of Biotechnology, Government of India (BT/PR16655/NER/95/132/2015) and by IIT Guwahati Institutional Start-Up Grant.
References
- 1.Christopherson G.T., Nesti L.J. Stem cell applications in military medicine. Stem Cell Res Ther. 2011;2(5):40. doi: 10.1186/scrt81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Takahashi K., Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 3.Raab S., Klingenstein M., Liebau S., Linta L.A. Comparative view on human somatic cell sources for iPSC generation. Stem Cells Int. 2014;2014:768391. doi: 10.1155/2014/768391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Takahashi K., Tanabe K., Ohnuki M. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131(5):861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 5.Aasen T., Raya A., Barrero M.J. Efficient and rapid generation of induced pluripotent stem cells from human keratinocytes. Nat Biotechnol. 2008;26(11):1276–1284. doi: 10.1038/nbt.1503. [DOI] [PubMed] [Google Scholar]
- 6.Haile Y., Nakhaei-nejad M., Boakye P.A. Reprogramming of HUVECs into induced pluripotent stem cells (HiPSCs), generation and characterization of HiPSC-derived neurons and astrocytes. PLOS ONE. 2015;10(3):e0119617. doi: 10.1371/journal.pone.0119617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhou T., Benda C., Dunzinger S. Generation of human induced pluripotent stem cells from urine samples. Nat Protoc. 2012;7(12):2080–2089. doi: 10.1038/nprot.2012.115. [DOI] [PubMed] [Google Scholar]
- 8.Haase A., Olmer R., Schwanke K. Generation of induced pluripotent stem cells from human cord blood. Cell Stem Cell. 2009;5(4):434–441. doi: 10.1016/j.stem.2009.08.021. [DOI] [PubMed] [Google Scholar]
- 9.Yu J., Vodyanik M.A., Smuga-otto K. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917–1920. doi: 10.1126/science.1151526. [DOI] [PubMed] [Google Scholar]
- 10.Chou B.K., Mali P., Huang X. Efficient human iPS cell derivation by a non-integrating plasmid from blood cells with unique epigenetic and gene expression signatures. Cell Res. 2011;21(3):518–529. doi: 10.1038/cr.2011.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yakubov E., Rechavi G., Rozenblatt S., Givol D. Reprogramming of human fibroblasts to pluripotent stem cells using mRNA of four transcription factors. Biochem Biophys Res Commun. 2010;394(1):189–193. doi: 10.1016/j.bbrc.2010.02.150. [DOI] [PubMed] [Google Scholar]
- 12.Miyoshi N., Ishii H., Nagano H. Reprogramming of mouse and human cells to pluripotency using mature microRNAs. Cell Stem Cell. 2011;8(6):633–638. doi: 10.1016/j.stem.2011.05.001. [DOI] [PubMed] [Google Scholar]
- 13.Kim D., Kim C.H., Moon J.I. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell. 2009;4(6):472–476. doi: 10.1016/j.stem.2009.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hou P., Li Y., Zhang X. Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science. 2013;341(6146):651–654. doi: 10.1126/science.1239278. [DOI] [PubMed] [Google Scholar]
- 15.Ari A.B. Eye injuries on the battlefields of Iraq and Afghanistan: public health implications. Optometry – J Am Optom Assoc. 2006;41(6):433–459. doi: 10.1016/j.optm.2006.03.015. [DOI] [PubMed] [Google Scholar]
- 16.Kokkinaki M., Sahibzada N., Golestaneh N. Human induced pluripotent stem-derived retinal pigment epithelium (RPE) cells exhibit ion transport, membrane potential, polarized vascular endothelial growth factor secretion, and gene expression pattern similar to native RPE. Stem Cells. 2011;29(5):825–835. doi: 10.1002/stem.635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lamba D.A., Mcusic A., Hirata R.K., Wang P.R., Russell D., Reh T.A. Generation, purification and transplantation of photoreceptors derived from human induced pluripotent stem cells. PLoS ONE. 2010;5(1):e8763. doi: 10.1371/journal.pone.0008763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Parameswaran S., Balasubramanian S., Babai N. Induced pluripotent stem cells generate both retinal ganglion cells and photoreceptors: therapeutic implications in degenerative changes in glaucoma and age-related macular degeneration. Stem Cells. 2010;28(4):695–703. doi: 10.1002/stem.320. [DOI] [PubMed] [Google Scholar]
- 19.Mandai M., Kurimoto Y., Takahashi M. Autologous induced stem-cell-derived retinal cells for macular degeneration. N Engl J Med. 2017;377(8):792–793. doi: 10.1056/NEJMc1706274. [DOI] [PubMed] [Google Scholar]
- 20.Oshima K., Shin K., Diensthuber M., Peng A.W., Ricci A.J., Heller S. Mechanosensitive hair cell-like cells from embryonic and induced pluripotent stem cells. Cell. 2010;141(4):704–716. doi: 10.1016/j.cell.2010.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gunewardene N., Crombie D., Dottori M., Nayagam B.A. Innervation of cochlear hair cells by human induced pluripotent stem cell-derived neurons in vitro. Stem Cells Int. 2016;2016:1781202. doi: 10.1155/2016/1781202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nishimura K., Nakagawa T., Sakamoto T., Ito J. Fates of murine pluripotent stem cell-derived neural progenitors following transplantation into mouse cochleae. Cell Transplant. 2012;21(4):763–771. doi: 10.3727/096368911X623907. [DOI] [PubMed] [Google Scholar]
- 23.Tsuji O., Miura K., Okada Y. Therapeutic potential of appropriately evaluated safe-induced pluripotent stem cells for spinal cord injury. Proc Natl Acad Sci U S A. 2010;107(28):12704–12709. doi: 10.1073/pnas.0910106107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Uemura T., Takamatsu K., Ikeda M. Transplantation of induced pluripotent stem cell-derived neurospheres for peripheral nerve repair. Biochem Biophys Res Commun. 2012;419(1):130–135. doi: 10.1016/j.bbrc.2012.01.154. [DOI] [PubMed] [Google Scholar]
- 25.Schoenfeld A.J., Belmont P.J. Traumatic combat injuries. In: Cameron K.L., Owens B.D., editors. Musculoskeletal injuries in the military. Springer; 2015. [Google Scholar]
- 26.Sakurai H., Sakaguchi Y., Shoji E. In vitro modeling of paraxial mesodermal progenitors derived from induced pluripotent stem cells. PLoS ONE. 2012;7(10):e47078. doi: 10.1371/journal.pone.0047078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jeon O.H., Panicker L.M., Lu Q., Chae J.J., Feldman R.A., Elisseeff J.H. Human iPSC-derived osteoblasts and osteoclasts together promote bone regeneration in 3D biomaterials. Sci Rep. 2016;6:26761. doi: 10.1038/srep26761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ohta S., Imaizumi Y., Okada Y. Generation of human melanocytes from induced pluripotent stem cells. PLoS ONE. 2011;6(1):e16182. doi: 10.1371/journal.pone.0016182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Itoh M., Kiuru M., Cairo M.S., Christiano A.M. Generation of keratinocytes from normal and recessive dystrophic epidermolysis bullosa-induced pluripotent stem cells. Proc Natl Acad Sci U S A. 2011;108(21):8797–8802. doi: 10.1073/pnas.1100332108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Itoh M., Umegaki-arao N., Guo Z., Liu L., Higgins C.A., Christiano A.M. Generation of 3D skin equivalents fully reconstituted from human induced pluripotent stem cells (iPSCs) PLOS ONE. 2013;8(10):e77673. doi: 10.1371/journal.pone.0077673. [DOI] [PMC free article] [PubMed] [Google Scholar]