Abstract
Background
Combination therapies of drugs with distinct mechanisms of action are emerging as ways to achieve strict glycemic control, thus preventing the onset and progression of diabetic complications in type 2 diabetes patients. A rapid-acting insulin secretagog, nateglinide, and a potent dipeptidyl peptidase-4 inhibitor, sitagliptin, meet such criteria.
Methods
A total of 121 patients inadequately controlled with sitagliptin monotherapy received 52-week combination therapy (nateglinide + sitagliptin). The primary endpoint was the safety of the therapy, and its efficacy was also evaluated. A meal tolerance test was performed 4 weeks before the start of combination therapy (week −4) and at week 24 and week 52 after the start of combination therapy.
Results
HbA1c levels were lower at week 52 than at week 0 [−0.42% (95% confidence interval −0.53, −0.31)]. Fasting plasma glucose levels tended to decrease from baseline (week 0) to week 52 [−4.8 mg/dl (−9.4, −0.2)]. In the meal tolerance test, postprandial plasma glucose levels and area under the curve of glucose from before to 2 h after the meal load were lower at week 24 and week 52 than at week −4. In addition, the levels of insulin and active glucagon-like peptide-1 were higher at week 52 than at week −4. Furthermore, the incidence of adverse events in combination therapy with sitagliptin was similar to those previously shown in nateglinide monotherapy.
Conclusion
Compared with sitagliptin monotherapy, the combination therapy of nateglinide plus sitagliptin was more effective in type 2 diabetes patients at improving glycemic control while showing similar safety.
Keywords: Nateglinide, DPP-4 inhibitor, Combination therapy, Diabetes, Safety, Long-term study
Introduction
Nateglinide is classified as a rapid-acting insulin secretagog glinide, and as with sulfonylurea (SU), it acts on the SU receptor on the surface of cellular membranes of pancreatic β-cells and improves the postprandial glycemic control of type 2 diabetes patients by promoting early phase insulin secretion [1]. In addition, because nateglinide has a pharmacokinetic profile characterized by rapid absorption and elimination after administration [2, 3], its immediate preprandial administration suppresses an increase in postprandial plasma glucose (PPG), and it is less likely to cause delayed hypoglycemia compared with SU drugs.
On the other hand, inhibitors of dipeptidyl peptidase-4 (DPP-4) inhibit the degradation of glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic peptide (GIP) and maintain the plasma concentration of these peptides, thus exhibiting a hypoglycemic effect by promoting insulin secretion in a plasma glucose-dependent manner.
In Japan, DPP-4 inhibitor is widely prescribed, but glycemic control with its monotherapy is inadequate in some diabetic patients [4]. For such patients, a combination therapy of DPP-4 inhibitor and an insulin secretagog with a mechanism of action different from that of DPP-4 inhibitor, including SU drugs and glinides, is expected to additively improve the hyposecretion of insulin. The Japan Diabetes Society recommends that the dose of SU drugs be reduced in combination with DPP-4 inhibitors, particularly in elderly patients and patients with mild decline of renal function, since the co-administration of SU drugs and DPP-4 inhibitors promotes serious hypoglycemia [5]. In contrast, glinides are considered to have a lower risk of hypoglycemia than SU drugs in the combined administration with DPP-4 inhibitors since glinides have a short duration of action. Meanwhile, the Diabetes Epidemiology: Collaborative analysis of Diagnostic criteria in Europe (DECODE) study, a meta-analysis of the prospective cohort studies conducted in Europe, revealed that plasma glucose levels at 2 h after glucose load were correlated with the risk of death, regardless of fasting plasma glucose (FPG) levels, and 2-h post-load glucose was an independent risk factor [6]. In the Funagata study, a prospective cohort study in Japan [7], and the Diabetes Epidemiology: Collaborative analysis of Diagnostic criteria in Asia (DECODA) study, a meta-analysis of the prospective cohort studies including the Funagata study and targeting the Asian population [8], it was suggested that plasma glucose levels at 2 h after glucose load were more closely correlated with cardiovascular diseases than fasting glucose levels. Postprandial hyperglycemia, which is closely associated with cardiovascular disease risk, is becoming more important as an independent risk factor. In addition, postprandial hyperglycemia is associated with reduced insulin secretion in the early postprandial period, and East Asians generally have low insulin secretion capacity [9]. For these reasons, combination therapies of DPP-4 inhibitors and glinides are expected to contribute to safe and effective glycemic control after a meal.
Taking these into account, to evaluate safety as the primary endpoint and also to assess the efficacy of long-term co-administration of nateglinide and a DPP-4 inhibitor, we performed a 52-week administration study of nateglinide and sitagliptin in patients who were inadequately controlled by sitagliptin monotherapy. We show here that compared to sitagliptin monotherapy, this combination therapy more effectively improves glycemic control in type 2 diabetes patients while showing similar safety.
Materials and methods
Study design
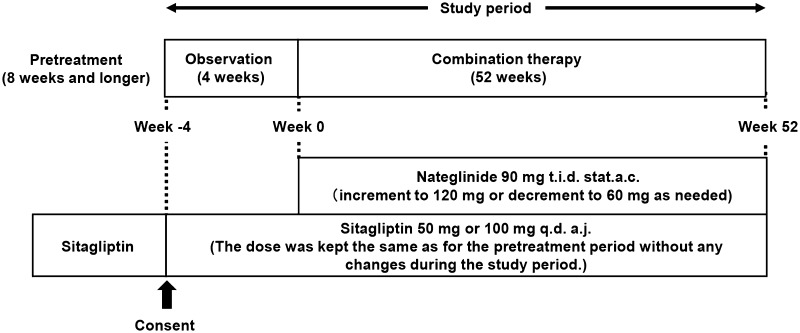
The study design is shown in Fig. 1. The study period included a 4-week observation and 52-week combination therapy period. This was an open-label, multicenter trial with sitagliptin monotherapy during the observation period and combined use of sitagliptin and nateglinide during the combination therapy period.
Fig. 1.
Study design. The study period was composed of the observation (4 weeks) and combination therapy period (52 weeks). The detailed daily doses of nateglinide and sitagliptin are shown. The following abbreviations were used: t.i.d. for three times daily, stat. a.c. for just before each meal, q.d. for once daily, and a.j. for before breakfast
Nateglinide 90 mg was orally administered three times a day just before each meal during the combination therapy period. Depending on the state of plasma glycemic control and the occurrence of adverse events related to plasma glycemic control, the dosage was allowed to decrease to 60 mg or increase to 120 mg three times a day when judged necessary by the investigator. In addition, when dosage reduction to 60 mg three times a day did not yet lead to improvement because of the occurrence of adverse events associated with hypoglycemia, the patients were allowed to stop nateglinide treatment.
Sitagliptin 50 or 100 mg was orally administered once daily before breakfast throughout the observation and combination therapy periods. The dose of sitagliptin was kept the same as in the pretreatment period without any changes during the study period. Throughout the study period, the use of oral hypoglycemic agents other than investigational nateglinide and sitagliptin, insulin preparations, GLP-1 receptor agonists, and other investigational drugs were prohibited. On the other hand, exercise and diet therapies were to be continued with no change throughout the study period in the instructions given at the start of the observation period.
To evaluate efficacy, hemoglobin A1c (HbA1c) (National Glycohemoglobin Standardization Program: NGSP) and FPG as well as plasma glucose, insulin, and active GLP-1 were measured in a meal tolerance test. HbA1c and FPG in the fasting condition in the morning were measured every 4 weeks after week 0 (the start of the combination therapy) and at week −4 (the start of the observation period), respectively. A meal tolerance test was performed at weeks −4, 24, and 52 from the start of combination therapy. Plasma glucose, insulin, and active GLP-1 levels were measured in blood samples collected before meal load and at 0.5, 1, and 2 h after meal load. Both nateglinide and sitagliptin were administered between the time of preprandial blood sampling and breakfast. In particular, nateglinide was administered within 10 min before breakfast. On the other visiting days, the patients took preprandial nateglinide and sitagliptin after laboratory testing. The meal tolerance test was carried out using meals adjusted to approximately 450 kcal at each medical institute. In addition, the contents and start time of meal load in a meal tolerance test at weeks 24 and 52 (or at the time of discontinuation) were set to create the same conditions as at week −4 to the utmost extent.
To evaluate safety, investigations of adverse events, laboratory tests, measurement of vital signs, and 12-lead electrocardiography were performed. We defined unfavorable events occurring in a patient with combination therapy of nateglinide and sitagliptin as adverse events regardless of the causal relationship with these investigational drugs and examined the names of adverse events, the association with hypoglycemia, the date of onset, seriousness, severity, causal relationship with nateglinide, treatment, and outcome (the date in the case of recovery or death). Laboratory tests (hematology, blood biochemistry, and qualitative urinalysis) were performed at the start of the combination therapy (week 0) and at weeks 4, 12, 24, 36, and 52 (or at the time of discontinuation), and blood and urine samples were collected in the fasted state. Vital signs (body weight, blood pressure, and pulse rate) were measured every 4 weeks (or at the time of discontinuation) from the start of the combination therapy (week 0), and 12-lead electrocardiography was performed at the start of the combination therapy (week 0) and at weeks 4, 12, 24, 36, and 52 (or at the time of discontinuation). All laboratory assessments were performed at LSI Medience Corp. (Tokyo, Japan).
This study was conducted in the following ten medical institutes in Japan and was approved in advance by the Sakayori Clinic Institutional Review Board on July 23, 2012 (approval number is not applicable) in compliance with the Declaration of Helsinki and the International Conference on Harmonisation (ICH)-Good Clinical Practice Guideline. The study was performed from July 2012 to December 2013. The participating institutes were Oikawa Medical Clinic, Hirano Medical Clinic, Noritake Clinic, Itabashi Diabetes and Dermatology Clinic, Tokuyama Clinic, KKR Mishuku Hospital, Hino Municipal Hospital, Koukan Clinic, NTT West Takamatsu Hospital, and KKR Takamatsu Hospital. The patients were informed of the study before participating by the use of an informed consent form approved by the Institutional Review Board, and written consent was obtained from the patients themselves. This study was registered at http://www.clinicaltrials.jp as JapicCTI-121927.
Participants
The patients in this study were outpatients aged at least 20 years at the time of consent with type 2 diabetes inadequately controlled with sitagliptin monotherapy who were defined as the patients who had received continuous administration of sitagliptin at a dose of 50 or 100 mg/day for not less than 8 weeks before the start of the observation period (week −4) and whose HbA1c (NGSP) levels were ≥ 6.9 and < 8.9% at week −4 and whose 1- or 2-h PPG levels were ≥ 200 mg/dl.
The main exclusion criteria were as follows: patients who were administered oral hypoglycemic agents other than sitagliptin or insulin preparations within 8 weeks before the start of the observation period (week −4), patients with type 1 diabetes, patients with severe ketosis (ketone level is ≥ 2 + by urinalysis), patients who changed hypertension drug therapies at least three times to control blood pressure in the 6 months before the start of the observation period (week −4), patients with renal dysfunction (creatinine > 1.5 mg/dl for males and > 1.3 mg/dl for females), patients with a medical history of hypersensitivity to sitagliptin and nateglinide, pregnant women, nursing women, and patients who participated in other clinical studies, or patients who had participated in other clinical studies within 12 weeks before participating in this study (written consent was obtained).
Statistical analysis
The sample size was determined in accordance with the Guideline for Clinical Evaluation of Oral Hypoglycemic Agents released in Japan [10]. Taking into account that the risk of hypoglycemia may increase because of the co-administration of insulin secretagogs, we planned to recruit 100 patients to assess 52 weeks of clinical safety regarding combined use of nateglinide and sitagliptin. The planned sample size provided a 95% probability that the population rate of adverse events would be 3%. Considering dropout patients, we determined the need to recruit 120 patients for combination therapy.
Data were analyzed using SAS® 9.3 Software (SAS Institute Inc., Cary, NC, USA).
For the efficacy analyses, the full analysis set, consisting of patients who received at least one dose of nateglinide and had at least one efficacy measurement, was used; multiplicity was not considered.
For HbA1c (NGSP) and FPG, descriptive statistics at each evaluation point and their changes from baseline (week 0) were calculated, and two-sided 95% confidence intervals were constructed based on the t distribution.
Descriptive statistics of plasma glucose, insulin, and active GLP-1 before meal load and 0.5, 1, and 2 h after meal load in the meal tolerance test were calculated at each evaluation point. Descriptive statistics of the area under the curve from before to 2 h after meal load (AUC0–2h) at each evaluation point were calculated. Their changes from baseline (week −4) were calculated, and two-sided 95% confidence intervals were constructed based on the t distribution.
The safety analysis set was defined as all of the patients who received at least one dose of nateglinide. The adverse events, the adverse events for which the causal relationship with nateglinide cannot be ruled out, serious adverse events, and adverse events associated with hypoglycemia were tabulated according to the Medical Dictionary for Regulatory Activities (MedDRA), version 15.0 (MedDRA Japanese Maintenance Organization, Tokyo, Japan).
Results
Patient description and demographic characteristics
The patient disposition is shown in Fig. 2. A total of 122 out of 169 patients who provided informed consent were enrolled. Among them, one patient was discontinued before the start of the combination therapy of sitagliptin and nateglinide, and 121 began to receive the medication. After the start of combination therapy, 13 patients were discontinued and 108 completed the study. The breakdown of the causes of discontinuation of the 13 patients was as follows: 6 patients, aggravation of the target disease; 5 patients, occurrence of adverse events; 1 patient, patient’s inconvenience; 1 patient, judgment by the investigator.
Fig. 2.
Disposition of patients. Disposition of 122 enrolled patients in this study is detailed here
All of the 121 patients who received combination therapy of sitagliptin and nateglinide were included in the safety analysis set and the full analysis set.
The demographic and baseline characteristics are shown in Table 1. The age was 62.9 ± 11.2 years (mean ± standard deviation): 64 patients (52.9%) were less than 65 years of age and 57 patients (47.1%) were at least 65 years of age. Seventy-nine males (65.3%) and 42 females (34.7%) were included, and the body mass index was 25.0 ± 3.7 kg/m2. Duration of type 2 diabetes was 6.5 ± 6.1 years, and the daily dose of sitagliptin was 50 mg for 101 patients (83.5%) and 100 mg for 20 patients (16.5%). At week 0, HbA1c (NGSP) was 7.7 ± 0.6% and the FPG level was 156.0 ± 35.0 mg/dl.
Table 1.
Demographic and baseline characteristics
| Variables | Total (n = 121) |
|---|---|
| Age (years) | 62.9 ± 11.2 |
| < 65 | 64 (52.9) |
| ≥ 65 | 57 (47.1) |
| Sex | |
| Male | 79 (65.3) |
| Female | 42 (34.7) |
| Body weight (kg) | 66.0 ± 14.1 |
| Body mass index (kg/m2) | 25.0 ± 3.7 |
| Duration of type 2 diabetes disease (years) | 6.5 ± 6.1 |
| Sitagliptin, daily dose | |
| 50 mg | 101 (83.5) |
| 100 mg | 20 (16.5) |
| HbA1c (%) | 7.7 ± 0.6 |
| < 6.9 | 6 (5.0) |
| ≥ 6.9 < 7.4 | 38 (31.4) |
| ≥ 7.4 < 7.9 | 39 (32.2) |
| ≥ 7.9 < 8.4 | 22 (18.2) |
| ≥ 8.4 | 16 (13.2) |
| Fasting plasma glucose (mg/dl) | 156.0 ± 35.0 |
Efficacy
Figure 3 shows the changes in HbA1c levels during combination therapy. The HbA1c level decreased from 7.65 ± 0.60% at week 0 to 7.47 ± 0.67% at week 4 [change from baseline (week 0): −0.19% (95% confidence interval −0.24, −0.13)] and 7.22 ± 0.75% at week 52 [−0.42% (−0.53, −0.31)]. FPG levels showed a tendency to decrease from 156.0 ± 35.0 mg/dl at baseline (week 0) to as low as 147.4 ± 30.6 mg/dl at week 24 [change from baseline (week 0): −5.6 mg/dl (−10.7, −0.6)]; thereafter, the same level was maintained, and the level was 148.6 ± 28.6 mg/dl at week 52 [−4.8 mg/dl (−9.4, −0.2)].
Fig. 3.
Changes in HbA1c levels during combination therapy. a HbA1C levels over the duration of combination therapy. Data are presented as the mean ± standard deviation. b Changes in HbA1C level from baseline (week 0). Data are presented as the mean and 95% confidence intervals
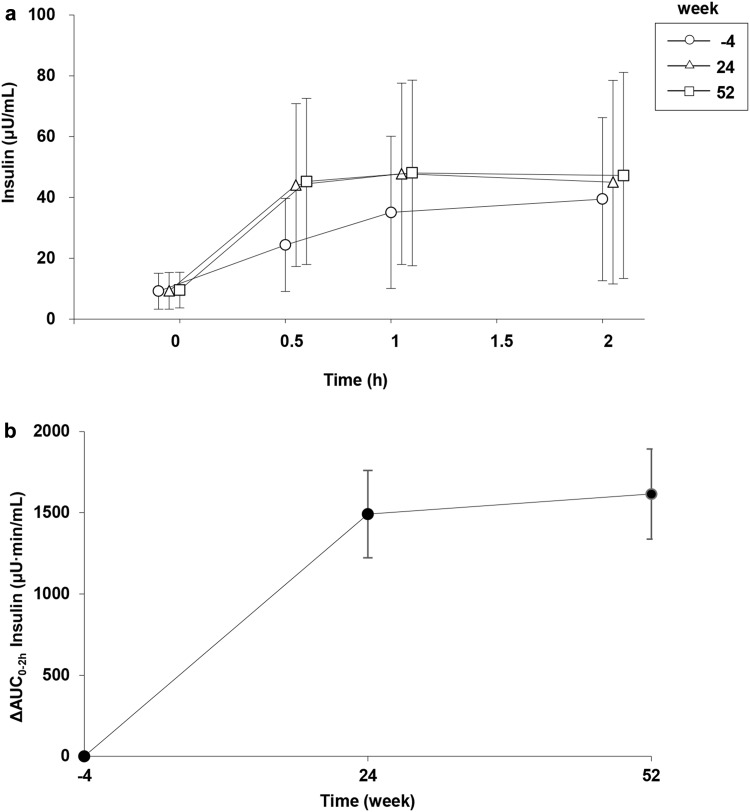
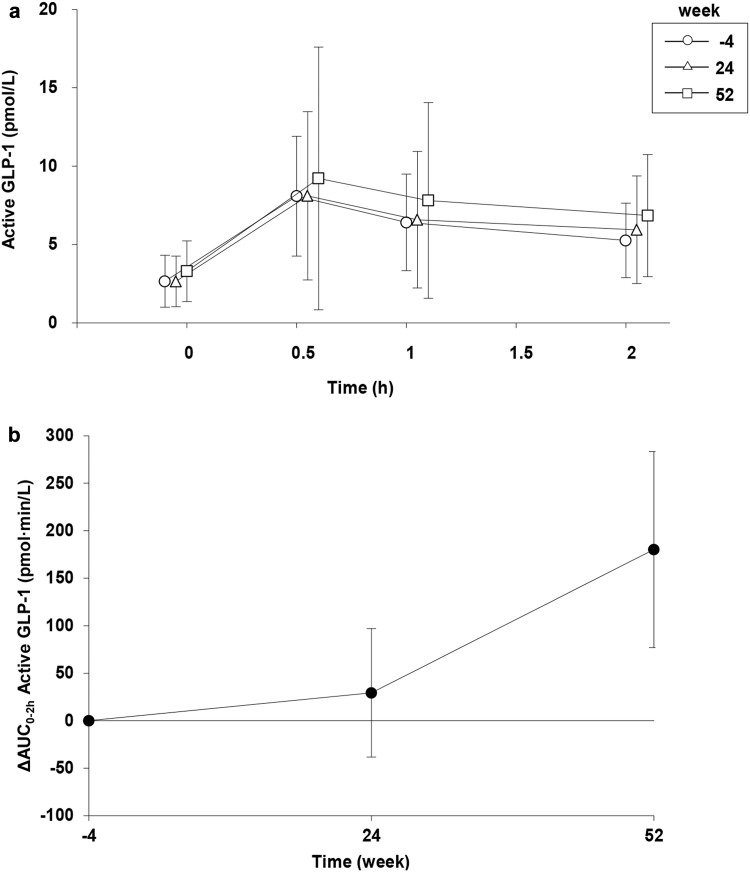
Figures 4, 5, and 6 show the changes in plasma glucose, insulin, and active GLP-1 until 2 h after the start of the meal tolerance test and the changes in AUC0–2h in the observation window from baseline (week −4) to week 52. The PPG level at 1 h was 243.8 ± 32.9, 202.0 ± 40.8, and 206.6 ± 40.5 mg/dl at weeks −4, 24, and 52, respectively. AUC0–2h was 28,352.7 ± 3842.8, 24,377.7 ± 4565.6, and 24,872.3 ± 4515.8 mg min/dl at weeks −4, 24, and 52, respectively, and the value decreased at week 24 [change from baseline (week −4): −4067.6 mg min/dl (−4749.3, −3385.8)] and week 52 [−3430.1 mg min/dl (−4265.5, −2594.7)]. The postprandial insulin level at 0.5 h was 24.397 ± 15.273, 44.105 ± 26.793, and 45.250 ± 27.320 µU/m at weeks −4, 24, and 52, respectively, and the postprandial insulin level at 1 h was 35.079 ± 25.028, 47.784 ± 29.806, and 48.066 ± 30.513 μU/ml at weeks −4, 24, and 52, respectively. AUC0–2h was 3788.163 ± 2415.920, 5212.295 ± 3062.610, and 5351.279 ± 3241.332 µU min/ml at weeks −4, 24, and 52, respectively. The value increased at week 24 [change from baseline (week −4): 1490.878 μU min/ml (1221.998, 1759.759)] and week 52 [1614.992 μU min/ml (1337.750, 1892.233)]. The postprandial active GLP-1 level at 2 h was 5.259 ± 2.379, 5.939 ± 3.435, and 6.844 ± 3.894 pmol/l at weeks −4, 24, and 52, respectively. AUC0–2h was 781.898 ± 302.323, 811.768 ± 430.701, and 945.194 ± 644.687 pmol min/l at weeks −4, 24, and 52, respectively, and the values at week 24 [29.280 pmol min/l (−38.302, 96.862)] and week 52 [180.189 pmol min/l (74.893, 285.484)] were similar to and higher than that at week −4, respectively.
Fig. 4.
Changes in postprandial plasma glucose levels. a Postprandial plasma glucose levels at weeks −4, 24, and 52. The values were obtained from the samples before (0 h) and 0.5, 1, and 2 h after meal load. Data are presented as the mean ± standard deviation. b Changes in AUC0–2h glucose at weeks 24 and 52 from baseline (week −4). Data are presented as the mean and 95% confidence intervals
Fig. 5.
Changes in plasma insulin levels. a Postprandial plasma insulin levels at weeks −4, 24, and 52. The values were obtained from the samples before (0 h) and 0.5, 1, and 2 h after meal load. Data are presented as the mean ± standard deviation. b Changes in AUC0–2h insulin at weeks 24 and 52 from baseline (week −4). Data are presented as the mean and 95% confidence intervals
Fig. 6.
Changes in plasma active GLP-1 levels. a Postprandial plasma active GLP-1 levels at weeks −4, 24, and 52. The values were obtained from the samples before (0 h) and 0.5, 1, and 2 h after meal load. Data are presented as the mean ± standard deviation. b Changes in AUC0–2h active GLP-1 at weeks 24 and 52 from baseline (week −4). Data are presented as the mean and 95% confidence intervals
Safety
The overall incidence of adverse events was 86.0% (104/121 patients) and the incidence of adverse events, of which the causal relationship with nateglinide cannot be ruled out, was 7.4% (9/121 patients). Table 2 shows adverse events occurring in at least 2% of total patients as commonly reported adverse events. The most commonly reported adverse events (incidence, at least 5%) included nasopharyngitis at 43.8% (53/121 patients), upper respiratory tract inflammation at 9.1% (11/121 patients), and pharyngitis at 5.0% (6/121 patients). The causal relationship with nateglinide was ruled out for all of these events. Adverse events, of which the causal relationship with nateglinide cannot be ruled out, included dizziness at 3.3% (4/121 patients), tremor and hunger at 1.7% (2/121 patients), and acute myeloid leukemia, hypoglycemia, feeling cold, malaise, and thirst at 0.8% (1/121 patients), respectively. Except for acute myeloid leukemia and thirst, all of these were adverse events associated with hypoglycemia. Acute myeloid leukemia was less likely caused by nateglinide administration, but there were significant increases in the white blood cell count and decreases in platelet count after the start of nateglinide administration. Therefore, it was very difficult to completely rule out the relationship with nateglinide, which was judged to be unclear by the investigator. Thirst was mild and the patient recovered early. The incidence of serious adverse events was 5.0% (6/121 patients). Pyelonephritis, acute myeloid leukemia, breast cancer, gastric cancer, macular hole, and autoimmune pancreatitis were observed in one patient each, and the causal relationship with nateglinide was ruled out except for acute myeloid leukemia. Adverse events associated with hypoglycemia are shown in Table 3. The incidence of adverse events associated with hypoglycemia was 5.8% (7/121 patients). Although no adverse events associated with hypoglycemia were ruled out concerning a causal relationship with nateglinide, they were non-serious and mild events, and it was possible to continue the study by dose reduction of nateglinide or intake of glucose.
Table 2.
Summary of commonly reported adverse events (incidence: ≥ 2%)
| Dose of sitagliptin | Number of patients (incidence, %) | ||
|---|---|---|---|
| 50 mg | 100 mg | Total | |
| Numbers analyzed | 101 | 20 | 121 |
| Infections and infestations | |||
| Nasopharyngitis | 43 (42.6) | 10 (50.0) | 53 (43.8) |
| Pharyngitis | 5 (5.0) | 1 (5.0) | 6 (5.0) |
| Cystitis | 5 (5.0) | 0 | 5 (4.1) |
| Metabolism and nutrition disorders | |||
| Diabetes mellitus inadequate control | 3 (3.0) | 1 (5.0) | 4 (3.3) |
| Type 2 diabetes mellitus | 2 (2.0) | 1 (5.0) | 3 (2.5) |
| Nervous system disorders | |||
| Dizziness | 4 (4.0) | 1 (5.0) | 5 (4.1) |
| Eye disorders | |||
| Cataract | 4 (4.0) | 0 | 4 (3.3) |
| Respiratory, thoracic, and mediastinal disorders | |||
| Upper respiratory tract inflammation | 9 (8.9) | 2 (10.0) | 11 (9.1) |
| Allergic rhinitis | 5 (5.0) | 0 | 5 (4.1) |
| Gastrointestinal disorders | |||
| Diarrhea | 4 (4.0) | 0 | 4 (3.3) |
| Stomatitis | 3 (3.0) | 0 | 3 (2.5) |
| Skin and subcutaneous tissue disorders | |||
| Eczema | 5 (5.0) | 0 | 5 (4.1) |
| Musculoskeletal and connective tissue disorders | |||
| Back pain | 4 (4.0) | 1 (5.0) | 5 (4.1) |
| Muscle spasms | 3 (3.0) | 0 | 3 (2.5) |
| Myalgia | 1 (1.0) | 2 (10.0) | 3 (2.5) |
| Periarthritis | 3 (3.0) | 0 | 3 (2.5) |
| Investigations | |||
| Blood triglycerides increased | 4 (4.0) | 0 | 4 (3.3) |
| Blood uric acid increased | 4 (4.0) | 0 | 4 (3.3) |
| Liver function test abnormal | 3 (3.0) | 0 | 3 (2.5) |
| Injury, poisoning, and procedural complications | |||
| Contusion | 4 (4.0) | 0 | 4 (3.3) |
Table 3.
Summary of adverse events associated with hypoglycemia
| Dose of sitagliptin | Number of patients (incidence, %) | ||
|---|---|---|---|
| 50 mg | 100 mg | Total | |
| Numbers analyzed | 101 | 20 | 121 |
| All adverse events | 6 (5.9) | 1 (5.0) | 7 (5.8) |
| Metabolism and nutrition disorders | 1 (1.0) | 0 | 1 (0.8) |
| Hypoglycemia | 1 (1.0) | 0 | 1 (0.8) |
| Nervous system disorders | 4 (4.0) | 0 | 4 (3.3) |
| Dizziness | 4 (4.0) | 0 | 4 (3.3) |
| Tremor | 2 (2.0) | 0 | 2 (1.7) |
| General disorders and administration site conditions | 2 (2.0) | 1 (5.0) | 3 (2.5) |
| Hunger | 2 (2.0) | 0 | 2 (1.7) |
| Feeling cold | 1 (1.0) | 0 | 1 (0.8) |
| Malaise | 0 | 1 (5.0) | 1 (0.8) |
There was not much difference in incidences of both common adverse events and adverse events associated with hypoglycemia by sitagliptin dose.
Discussion
In this study, we investigated the efficacy and safety of 52-week long-term co-administration of nateglinide in 121 patients with type 2 diabetes who were inadequately controlled by monotherapy with sitagliptin, which is classified as a DPP-4 inhibitor.
For efficacy, both 0.5- and 1-h postprandial insulin levels increased at week 24 of nateglinide combination therapy compared with baseline (week −4) of sitagliptin monotherapy, and the increases were also maintained until week 52. Both 1- and 2-h PPG levels decreased at week 24 compared with those at baseline (week −4), and these decreases were maintained until week 52. AUC0–2h also decreased at week 24 and week 52 compared with baseline (week −4). Improvement effects on PPG levels as well as the stimulation of insulin secretion were observed throughout a 52-week period. HbA1c was lowered by 0.19% at week 4, by 0.30% at week 24, and by 0.42% at week 52 compared with baseline (week 0), and the decrease in HbA1c was maintained over 52 weeks. FPG levels were also well controlled over 52 weeks. These results reflect the characteristics of nateglinide that promote early-phase insulin secretion and, by making the insulin secretion patterns of the patients similar to those of healthy individuals, improve PPG levels and enable proper glycemic control.
In recent years, in vivo and in vitro study data have been reported, suggesting that GLP-1 secretion is promoted by the inhibition of DPP-4 activity by nateglinide and the accompanying suppression of degradation of GLP-1 [11, 12]. Moreover, nateglinide alone, GLP-1 alone, or the combination of nateglinide and GLP-1 was administered beforehand every week in a three-way crossover design in patients with type 2 diabetes to examine the hypoglycemic effects of nateglinide and exogenous GLP-1 [13]. Specifically, glucose was intravenously administered to raise and adjust the plasma glucose level to 15 mmol/l at 15 min after single or co-administration of nateglinide and GLP-1. The resulting AUC0–180min value for plasma glucose was the lowest in the combination therapy, while that for insulin was the highest in the same treatment. The AUC0–180min value for DPP-4 was lower with combination therapy and nateglinide alone compared with GLP-1 alone. The AUC0–180min value for GLP-1 was the highest with combination therapy. These results suggested that nateglinide exhibited hypoglycemic effects in association with enhanced active GLP-1 level and inhibited DPP-4 activity. On the other hand, it was reported that the active GLP-1 level was significantly increased at week 24 compared with baseline by both the combination therapy with nateglinide plus vildagliptin and the monotherapy with vildagliptin in patients inadequately controlled by nateglinide, whereas a significant difference was not observed between the two therapy groups [14]. It was confirmed that the results differed from those obtained in the previously described single-dose loading test.
In this study, to evaluate the relevance of the corresponding mechanisms to the hypoglycemic effect of nateglinide, the active GLP-1 level was measured by conducting the meal tolerance test. Active GLP-1 levels at week 24 were similar to those at week −4 and their increase was observed at week 52, but the increase in early-phase insulin secretion and the accompanying improvement in PPG levels were observed at week 24 and were similar at week 52. These collectively suggest that an improvement in PPG levels was caused by the already known insulin secretive effect of nateglinide. This study is an open trial and there is a limitation to consider. The clinical significance of the increase in the active GLP-1 level seen at 52 weeks is ill defined, and further investigation is required.
With respect to safety, the overall incidence of adverse events was 86.0% (104/121 patients); the incidence of adverse events, for which the causal relationship with nateglinide cannot be ruled out, was 7.4% (9/121 patients). Most adverse events observed in this study occurred by week 12, and there was no trend for the incidence of adverse events to increase because of long-term administration. Thus far, the safety of nateglinide has been confirmed over a long period of time in monotherapy [15, 16], combined therapy with biguanide-based drugs [17], and combined therapy with pioglitazone hydrochloride [18]. Moreover, similar safety has been confirmed for the combined therapy of nateglinide and sitagliptin in this study.
Recently, the efficacy and safety of an additional DPP-4 inhibitor to rapid-acting insulin secretagog monotherapy were demonstrated in type 2 diabetes patients inadequately controlled by a rapid-acting insulin secretagog alone [19]. In our study, the efficacy and safety of additional nateglinide to DPP-4 inhibitor monotherapy were demonstrated in type 2 diabetes patients inadequately controlled by DPP-4 inhibitor alone. These results suggest that combination therapies of DPP-4 inhibitors and rapid-acting insulin secretagogs are safe and effective, regardless of the order of administration, for glycemic control in type 2 diabetes patients.
Limitations of our study should be noted. First, this study was conducted with no control arm and can be compromised by unknown biases. Thus, we cannot exclude the possibility that, as both patients and investigators were aware of the treatments performed, outcomes in patients may have been evaluated in favor of improvements. However, to reduce this risk, we used objectively measured laboratory values. Second, depending on the state of plasma glycemic control and the occurrence of hypoglycemia-related adverse events, dose changes or temporal discontinuation of nateglinide was allowed at the discretion of investigators during the study period, which makes it difficult to assess how these changes in the regimen influenced our findings regarding safety and efficacy.
In conclusion, we consider that the combined therapy of nateglinide and DPP-4 inhibitors including sitagliptin can be one of the useful combination therapies in performing safe and stable glycemic control over a long period of time.
Acknowledgements
The authors thank all of the patients and investigators who took part in this study. The principal investigators in this study were: Dr. Keiichi Oikawa, Oikawa Medical Clinic; Dr. Shigeru Hirano, Hirano Medical Clinic; Dr. Masayuki Noritake, Noritake Clinic; Dr. Naoki Itabashi, Itabashi Diabetes and Dermatology Clinic; Dr. Takahiko Tokuyama, Tokuyama Clinic; Dr. Munehiro Honda, KKR Mishuku Hospital; Dr. Marohito Murakami, Hino Municipal Hospital; Drs. Yasushi Iwaita and Yoshihiro Takamiya, Koukan Clinic; Dr. Toshiki Fukui, NTT West Takamatsu Hospital; Dr. Satoshi Murao, KKR Takamatsu Hospital. Nateglinide was jointly developed by EA Pharma Co., Ltd., and Astellas Pharma Inc. This study was primarily sponsored and funded by EA Pharma Co., Ltd.
Conflict of interest
T. Hirose received consultancy fees from EA Pharma Co., Ltd.; lecture fees from MSD K.K., Eli Lilly Japan K.K., Takeda Pharmaceutical Co., Ltd., Novartis Pharma K.K., Dainippon Sumitomo Pharma Co., Ltd., Novo Nordisk Pharma Ltd, Sanofi K.K., AstraZeneca K.K., and Daiichi Sankyo Co., Ltd.; and research funds from MSD K.K., Eli Lilly Japan K.K., Takeda Pharmaceutical Co., Ltd., Novo Nordisk Pharma Ltd, Kissei Pharmaceutical Co., Ltd., Boehringer Ingelheim, Mitsubishi Tanabe Pharma Corporation, Dainippon Sumitomo Pharma Co., Ltd., Sanofi K.K., Ono pharmaceutical Co., Ltd., AstraZeneca K.K., and Daiichi Sankyo Co., Ltd. C. Saitoh and I. Oikawa are employees of EA Pharma Co., Ltd. N. Kondo was an employee of EA Pharma Co., Ltd.
Human rights statement
This research involves human participants. The study protocol was approved by the Sakayori Clinic Institutional Review Board on July 23, 2012 (approval number is not applicable).
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Informed consent was obtained from all patients for being included in the study.
References
- 1.Ikenoue T, Akiyoshi M, Fujitani S, Okazaki K, Kondo N, Maki T. Hypoglycaemic and insulinotropic effects of a novel oral antidiabetic agent, (−)-N-(trans-4-isopropylcyclohexanecarbonyl)-d-phenylalanine (A-4166) Br J Pharmacol. 1997;120(1):137–145. doi: 10.1038/sj.bjp.0700875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kosaka K, Kikuchi M, Kuzuya T, Akanuma Y, Ohashi Y. Clinical effect and safety of AY4166 in patients with non-insulin-dependent diabetes mellitus. Yakuri Rinsho. 1997;7:635–651. [Google Scholar]
- 3.Kosaka K, Kikuchi M, Kuzuya T, Akanuma Y, Takiguchi K, Ishihara H, Ohashi Y. Change in postprandial plasma glucose and pharmacodynamics in patients with non-insulin-dependent diabetes mellitus by administration with AY4166. Yakuri Rinsho. 1997;7:653–668. [Google Scholar]
- 4.Kaku K, Inagaki N, Kobayashi N. Long-term effects of mitiglinide in Japanese diabetics inadequately controlled with DPP-4 inhibitor or biguanide monotherapy. Diabetes Ther. 2014;5(1):97–111. doi: 10.1007/s13300-014-0051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Committee on Proper Use of Incretins (GLP-1 Receptor Agonists and DPP-4 Inhibitors). The Japan Diabetes Society. 2011. http://www.fa.kyorin.co.jp/jds/uploads/photos/797.pdf. Accessed 11 Oct 2017 (in Japanese).
- 6.Glucose tolerance and mortality: comparison of WHO and American Diabetes Association diagnostic criteria. The DECODE study group European Diabetes Epidemiology Group. Diabetes epidemiology: collaborative analysis of diagnostic criteria in Europe. Lancet. 1999;354(9179):617–621. doi: 10.1016/S0140-6736(98)12131-1. [DOI] [PubMed] [Google Scholar]
- 7.Tominaga M, Eguchi H, Manaka H, Igarashi K, Kato T, Sekikawa A. Impaired glucose tolerance is a risk factor for cardiovascular disease, but not impaired fasting glucose. The Funagata Diabetes Study. Diabetes Care. 1999;22(6):920–924. doi: 10.2337/diacare.22.6.920. [DOI] [PubMed] [Google Scholar]
- 8.Nakagami T, Qiao Q, Tuomilehto J, Balkau B, Tajima N, Hu G, Borch-Johnsen K. Screen-detected diabetes, hypertension and hypercholesterolemia as predictors of cardiovascular mortality in five populations of Asian origin: the DECODA study. Eur J Cardiovasc Prev Rehabil. 2006;13(4):555–561. doi: 10.1097/01.hjr.0000183916.28354.69. [DOI] [PubMed] [Google Scholar]
- 9.Kodama K, Tojjar D, Yamada S, Toda K, Patel CJ, Butte AJ. Ethnic differences in the relationship between insulin sensitivity and insulin response: a systematic review and meta-analysis. Diabetes Care. 2013;36(6):1789–1796. doi: 10.2337/dc12-1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pharmaceutical and Food Safety Bureau. Guideline for clinical evaluation of oral hypoglycemic agents. 2010. http://www.pmda.go.jp/files/000208194.pdf. Accessed 22 Feb 2017.
- 11.Duffy NA, Green BD, Irwin N, Gault VA, McKillop AM, O’Harte FP, Flatt PR. Effects of antidiabetic drugs on dipeptidyl peptidase IV activity: nateglinide is an inhibitor of DPP IV and augments the antidiabetic activity of glucagon-like peptide-1. Eur J Pharmacol. 2007;568(1–3):278–286. doi: 10.1016/j.ejphar.2007.05.010. [DOI] [PubMed] [Google Scholar]
- 12.Kitahara Y, Miura K, Yasuda R, Kawanabe H, Ogawa S, Eto Y. Nateglinide stimulates glucagon-like peptide-1 release by human intestinal L cells via a K(ATP) channel-independent mechanism. Biol Pharm Bull. 2011;34(5):671–676. doi: 10.1248/bpb.34.671. [DOI] [PubMed] [Google Scholar]
- 13.Bell PM, Cuthbertson J, Patterson S, O’Harte FP. Additive hypoglycaemic effect of nateglinide and exogenous glucagon-like peptide-1 in type 2 diabetes. Diabetes Res Clin Pract. 2011;91(3):e68–e70. doi: 10.1016/j.diabres.2010.11.033. [DOI] [PubMed] [Google Scholar]
- 14.Kudo-Fujimaki K, Hirose T, Yoshihara T, Sato F, Someya Y, Ohmura C, Kanazawa A, Fujitani Y, Watada H. Efficacy and safety of nateglinide plus vildagliptin combination therapy compared with switching to vildagliptin in type 2 diabetes patients inadequately controlled with nateglinide. J Diabetes Investig. 2014;5(4):400–409. doi: 10.1111/jdi.12160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kosaka K, Kikuchi M, Tarui S, Shigeta Y, Kuzuya T, Akanuma Y, Toyota T, Ohashi Y. Long-term administration of fast and short-acting insulin secretagogue AY4166 to non-insulin-dependent diabetes mellitus. Yakuri Rinsho. 1997;7:797–818. [Google Scholar]
- 16.Kuzuya T, Oka Y, Yaga K, Inoue Y, Matsutani A, Kosaka K. Utility of long-term administration of fast and short-acting insulin secretagogue AY4166 to non-insulin-dependent diabetes mellitus. Yakuri Rinsho. 1997;7:819–832. [Google Scholar]
- 17.Kikuchi M. Safety and efficacy of long-term combined therapy of nateglinide and metformin hydrochloride in type 2 diabetes patients inadequately controlled with monotherapy of metformin hydrochloride: multicenter, open-label trial. Rinsho Iyaku. 2008;24:741–760. [Google Scholar]
- 18.Kikuchi M. Utility of long-term combined therapy of nateglinide and pioglitazone hydrochloride in type 2 diabetes patients inadequately controlled with monotherapy of pioglitazone hydrochloride (extended administration from phase II/phase III double blind parallel group comparison trials): multicenter, open-label trial. Rinsho Iyaku. 2009;25:57–75. [Google Scholar]
- 19.Tajima N, Kadowaki T, Odawara M, Minamide T, Seki A, Oki K, Nagayasu R, Arjona Ferreira JC. Safety and efficacy of addition of sitagliptin to rapid-acting insulin secretagogues for glycemic control, including post-prandial hyperglycemia, among Japanese with type 2 diabetes mellitus. Diabetol Int. 2016;7(2):155–166. doi: 10.1007/s13340-015-0230-2. [DOI] [PMC free article] [PubMed] [Google Scholar]