Abstract
Objectives:
Bacterial biofilm-dependent infections (e.g., cystic fibrosis, surgical sites, and medical implants) are associated with enhanced drug-resistance and thus difficult to eradicate. The goal of this study was to systematically compare three distinct classes of antimicrobial peptides (AMPs) that include the clinically used antibiotic colistin, the natural AMP LL37, the engineered cationic-AMP WLBU2, and four commonly used antibiotics with different bactericidal mechanisms (tobramycin, ciprofloxacin, ceftazidime and vancomycin) for biofilm prevention properties.
Methods:
Using biofilm-prevention assays, we detected bacterial biomass post-attachment in subinhibitory concentrations (1/3 of the MIC) for each AMP, by the crystal violet method, to distinguish the commonly known bactericidal from potentially distinct mechanisms of biofilm prevention. Biofilm regulatory gene expression was assessed using RT-qPCR for correlation with biofilm growth inhibition.
Results:
Commonly used antibiotics at 1× MIC showed modest ESKAPE biofilm prevention while 1/3 MIC of AMPs demonstrated up to 90% of biofilm prevention. WLBU2 was generally more effective in preventing bacterial attachment than colistin and LL37. Changes in expression of bacterial genes known to affect biofilm regulation were consistent with biofilm prevention.
Conclusion:
The data warrant further exploration of AMPs with optimized structures to fill a knowledge gap on the potential application of AMPs to difficult-to-cure bacterial biofilm-related infections
Keywords: antimicrobial peptide, biofilm, eCAP, ESKAPE pathogen
Graphical abstract
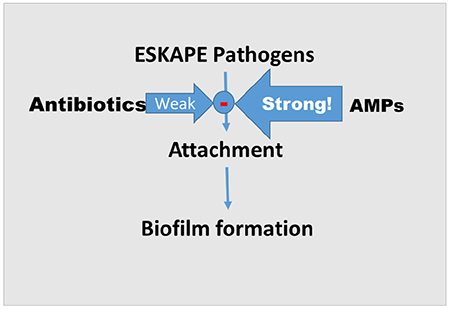
1. Introduction
ESKAPE pathogens (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, andEnterobacter species), represent a group of multidrug-resistant (MDR) bacteria that demonstrate the ability to escape antimicrobial treatments. These organisms are highly responsible for nosocomial and community-acquired infections, such as ventilator-associated pneumonia and S. aureus induced diabetic foot infection. Notably, P. aeruginosa contributes directly to the high mortality rate of cystic fibrosis (CF) chronic lung infections. Respiratory P. aeruginosa biofilm colonization affects more than 80% of late stage CF patients, and it is rarely successfully eradicated by standard antibiotic treatment. ESKAPE pathogens share the ability to attach to tissues and form biofilm (e.g., surgical sites, medical implants, and cystic fibrosis), which makes ESKAPE infections difficult to prevent and treat [1].
In contrast to the floating planktonic growth of bacteria, biofilms are communities of bacteria enclosed by a self-produced exopolysaccharide matrix (EPM), which serves as a shield when bacteria attach to solid surfaces, including human tissues such as teeth, skin, trachea and urinary tract. In addition to protection from immune effector molecules from the host, the EPM tends to protect bacteria from antibiotics. It is commonly accepted that once biofilm is formed, it could develop up to 1,000-fold or more antibiotic resistance, compared to planktonic mode of growth. While biofilm formation is usually more common than the counterpart planktonic growth in infectious diseases [2], more concerning are infections initiated exclusively through biofilm formation by attachment to solid surfaces such as medical implants, trauma and surgical sites. Such infections are very difficult to treat with current antibiotics and often require surgical debridement. Thus, prevention of early-stage biofilm formation is imperative in eradicating these infections.
Antimicrobial peptides (AMPs) are usually short cationic polypeptides (<60 amino acids) that exhibit antimicrobial activity. While natural AMPs (nAMP) play an important role in the prevention of bacterial colonization in the immunocompetent host, certain pathophysiologic changes (e.g., acidic pH and high salt concentration in the CF airway) may impair their effectiveness [3]. Previous studies showed that the de novo-engineered cationic antimicrobial peptide WLBU2 retained activity in acidic environments, PBS, and divalent cations against both gram-positive and -negative bacteria [4]. More recently, we and others have shown that WLBU2 can inhibit the growth of a variety of MDR ESKAPE pathogens [5] including those that are resistant to the human nAMP LL37 and the membrane-active cationic lipopeptide colistin. WLBU2 both inhibits biofilm growth, disrupts biofilm formation [6], and displays efficacy in respiratory infection when delivered by intratracheal instillation in the mouse airway [7]. However, it is unclear whether prevention of biofilm growth is a specific activity or due to the killing of planktonic bacteria prior to attachment, the critical initial step in biofilm formation. Because AMPs typically recognize bacterial cells by electrostatic interactions with negatively charged lipids (e.g. LPS) on their surfaces, we postulated that these interactions alone would interfere with the ability of bacteria to attach to surfaces and would, therefore, prevent biofilm formation at concentrations that are lower than their planktonic minimum inhibitory concentrations or MIC. Therefore, in this study, we used subinhibitory concentrations (1/3MIC) to examine biofilm prevention activity of WLBU2 compared to two other well-studied peptides: LL37, one of the well-known natural AMPs, and colistin, a clinically used antibiotic of last resort against 36 test strains of ESKAPE pathogens.
2. Materials and methods
2.1. Bacteria
All ESKAPE pathogens were isolated from pediatric CF patients with chronic pulmonary infections at Seattle Children’s Hospital or adult patients at the University of Pittsburgh Medical Center.
2.2. Peptide synthesis
Colistin sulfate was purchased from Sigma. Engineered AMP WLBU2 (RRWVRRVRRVWRRVVRVVRRWVRR) and natural AMP LL37 (LLGDFFRKSKEKIGKEFKRIVQRIKDFLRNLVPRTES) were synthesized using standard Fmoc (9-fluorenylmethoxy carbonyl) synthesis protocols as previously described [8] and purification achieved by reversed-phase high-pressure liquid chromatography on Vydac C18 or C4 columns (The Separations Group). The identity of each peptide was established by MS (Electrospray Quatro II triple quadrupole mass spectrometer).
2.3. Minimum Inhibitory Concentration (MIC)
MIC (Supplemental Table 1 and 2) was determined [5] by following the standard procedures established by Clinical Laboratory Standards Institute (CLSI) with some modifications [9], using cation-adjusted Muller Hinton broth (MHB2).
2.4. Biofilm assay
We used a slightly modified version of the microtiter plate assay developed by O’Toole [10], Log-phase bacteria were diluted in DMEM to 108 CFU/mL using a spectrophotometer. A 50μL volume of AMP or antibiotic dilutions (in PBS), at different concentrations, was added to 50μL of bacterial suspension in a sterile 96-well polystyrene plate. The final bacterial concentration of the mixture is 5×107 CFU/mL, 50-fold compared to 106 CFU/mL in MIC assays for adequate attachment. After 3 hours of bacterial attachment at 37°C (no shaking), the supernatant was discarded. The plate was washed with PBS prior to staining with 125μL of 0.5% Crystal Violet (in 20% Ethanol) for 15 minutes. Excess stain was removed by washing twice with distilled water [11], Crystal violet-stained biomass was dissolved in 150μL of 95% ethanol and measured using a plate reader at 620nm. Untreated bacteria (100% bacterial attachment), served as positive control. Wells with a mixture of sterile DMEM and PBS were used to control for possible contamination. Biomass in different treatment groups was quantified as percent OD of the positive controls.
2.5. Gene expression
For each treatment group, P. aeruginosa PAOl (accession number: AE004091.2) was incubated in a mixture of 50% DMEM and 50% PBS with 1/3 MICs of AMPs, consistent with the bacterial attachment/biofilm assays. Total bacterial RNA was extracted using TRI Reagent (Sigma). The cDNA was synthesized by High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems). Fast SYBR Green Master Mix (Applied Biosystems) and 7900HT Fast Real-Time PCR System (Applied Biosystems) were used for quantification. Fold change was calculated by ΔΔCT values. Constitutively expressed rplU gene encoding 50S ribosomal protein was used as housekeeping gene to normalize qPCR data. Primer sequences are listed in web-only Supplemental Table 3.
2.6. Statistical analysis
Statistical analyses were performed by one-way analysis of variance and Dunnett’s multiple comparisons test. Statistically significance is achieved at p<0.05.
3. Results
3.1. Biofilm prevention activity of clinically used antibiotics
Many ESKAPE strains are resistant to commonly used antibiotics (Supplemental Table 1). We chose 2 strains of each ESKAPE pathogen, including both susceptible and resistant strains, to test our hypothesis that AMP recognition of bacterial membrane interfere with bacterial attachment, the first step in bacterial biofilm formation. When bacteria were exposed to antibiotics at 1× MIC for 3h to allow bacterial attachment (Fig.1a and b), all antibiotics showed negligible biofilm prevention activity against gram-positive bacteria E. faecium and S. aureus with one exception: ceftazidime against S. aureus (approximately 25%-50% prevention). Fig.1c to e, antibiotics sometimes stimulated more bacterial attachment at 1× MIC. Antibiotics showed moderate but less than 50% of biofilm prevention activities against Enterobacter aerogenes 518 and Enterobacter cloacae 470 (Fig.1f).
Figure 1. Biofilm prevention activity of clinically used antibiotics and AMPs at 1× MICs against two of each strain of ESKAPE bacteria.

Biofilm formation on 96-well microtiter plates was quantified by the crystal violet staining method [10], Bacteria that are susceptible to antibiotics and AMPs were treated with their respective MIC. Bacteria that are resistant to antibiotics and AMPs were treated with the highest tested MIC (20μg/mL for tobramycin, 100μg/mL for ceftazidime, 10μg/mL for ciprofloxacin, 50μg/mL for vancomycin and 32 μM for all AMPs). Dashed lines represent 100% biofilm formation (untreated positive control). Data are mean ± SEM (n=4). Statistical analyses were performed by one-way analysis of variance (ANOVA) and Dunnett’s multiple comparisons test, p < 0.05 is considered statistically significant. p < 0.05 (*), p < 0.01 (**), p < 0.001 (***), p < 0.0001 (****) and NS=non-significant compared to untreated positive controls. R denotes drug-resistant strain.
3.2. Biofilm prevention activity of AMPs
As illustrated in Supplemental Table 1 and 2, we defined resistant bacteria as strains exhibiting an MIC of 32μM or more for a particular antimicrobial agent in our standard growth inhibition assay (GIA). Of the 36 ESKAPE strains, 18 different strains were resistant to colistin, including all 12 gram-positive strains as expected. A total of 20 strains were resistant to LL37 whereas 7 were resistant to WLBU2. We adopted 1/3 MIC, as planktonic bacteria in broth cultures are able to grow normally in the presence of AMPs at that concentration. Thus, we compared colistin, LL37, and WLBU2 against six different MDR clinical isolates of each ESKAPE organism for broad-spectrum biofilm prevention activities.
3.2.1. Gram-positive biofilm prevention.
Colistin is typically not effective against gram-positive bacteria, and resistance is represented by the letter “R” (Fig.2). As a gram-positive organism, E.faecium (a) is resistant to colistin (MIC≥32) but remains susceptible to LL37. In addition, two of six strains of E.faecium are resistant to WLBU2. An interesting observation is that susceptibility to AMPs does not correlate with biofilm prevention activity (Fig.2), as the two WLBU2-resistant E. faecium strains (a) demonstrated lowest levels of biomass (>95% reduction in attachment); colistin demonstrated higher level of attachment than LL37 while all E. faecium strains are susceptible to LL37 (Fig.2a); LL37 displayed highest biofilm prevention activities against two S. aureus-resistant strains and was similar to WLBU2 in biofilm prevention despite that all the S. aureus strains were resistant to LL37 (Fig.2b).
Figure 2. Biofilm prevention activity of antimicrobial peptides at 1/3 of the MICs against gram-positive bacteria: (a) Enterococcus faecium, (b) Staphylococcus aureus and methicillin-resistant Staphylococcus aureus (MRSA) and gram-negative bacteria: (c) Klebsiella pneumoniae, (d) Acinetobacter baumannii, (e) Pseudomonas aeruginosa and (f) Enterobacter species.

Biofilm assay was described in Fig. 1. Results are from three independent experiments. Dashed lines represent 100% biofilm formation (untreated positive control). Data are mean ± SEM (n=3). Statistical analyses were performed by one-way ANOVA and Dunnett’s multiple comparisons test. p < .05 is considered statistically significant. p < 0.05 (*), p < 0.01 (**), p < 0.001 (***) and p < 0.0001 (****) compared to untreated positive controls (otherwise non-significant). R denotes resistant strain (MIC≥32μM).
3.2.2. Gram-negative biofilm prevention.
Upon AMPs treatment, gram-negative bacteria displayed similar biofilm prevention pattern, exemplified by the lack of correlation between susceptibility profiles and biofilm prevention activities, as shown in figure 2. WLBU2 generally displayed highest levels of biofilm prevention (up to 95% reduction in bacterial attachment) with the exception of two strains (D5 and F9) of A. baumannii (Fig.2d). Biofilm prevention activities of LL37 and colistin were similar against about 50% of the gram-negative strains, while LL37 reduced bacterial attachment more effectively than colistin in 8 of the remaining half of the test strains. This lack of correlation between MIC and biofilm prevention activities led us to look at biofilm-associated gene expression to explain the biofilm-prevention activities, with a focus on P. aeruginosa PAOl (Fig.3).
Figure 3. P. aeruginosa PAO1 gene expression.

10 mL of 108/mL planktonic PAO1 was treated by 1/3 of the MICs of colistin (0.33μM), LL37 (5.33μM) and WLBU2 (2.67μM) for 3 hours in an orbital shaker at 37°C. Total RNA was extracted immediately after the incubation. Results are representative of three independent experimental trials. Data are shown as mean ± SEM (n=2). Statistical analyses were performed by one-way ANOVA and Dunnett’s multiple comparisons test. p < .05 is considered statistically significant. p < 0.01(**), p < 0.001 (***) and p < 0.0001(****) compared to untreated positive controls (otherwise non-significant)
3.3. Biofilm-related gene expression after treatment of AMPs
Among all six species of ESKAPE pathogens, P. aeruginosa is one of the strongest biofilm producers and has been extensively studied. Therefore, we examined gene expression of P. aeruginosa with a broad-spectrum list of well-established biofilm/virulence-related genes, to further test our hypothesis.
In P. aeruginosa (PAO1), the fleN gene is involved in regulating the number of flagella and provides the motility required for initial bacterial attachment [12]. All AMPs showed significant inhibition of this gene (colistin, 40% inhibition; LL37, 60%; WLBU2, 70%), which is consistent with the results of inhibited PAO1 biofilm attachment in Fig.2e. The mucB gene is a negative regulator of the sigma factor AlgU [13], which upregulates alginate production toward a more mucoid phenotype. Interestingly, WLBU2 was the only antimicrobial agent to enhance mucB expression by more than 2-fold, with the concomitant reduction in biofilm attachment (Fig.2e).
P. aeruginosa utilizes a type III secretion system (T3SS), which injects cytotoxins (e.g. exoS and exoT) into host cells via the bacterial pilus [14]. T3SS is required at the early stage of biofilm formation [15]. As shown in Fig.3, LL37 and WLBU2 displayed a 50% reduction in exoS and exoT mRNA levels at 1/3 MICs suggesting that, in the presence of AMPs, PAO1 may become less virulent. Both fimT and fimX are responsible for the function of pseudomonal type IV pili and twitching motility [16]. Twitching motility is necessary for biofilm formation [12]. The expression was not affected except for a minor increase in the colistin group (Fig.3), indicating that these two genes are not the main targets for AMPs LL37 and WLBU2.
These results indicated AMPs may modulate bacterial gene expression at sub-inhibitory concentrations, suggesting a potentially novel mechanism of biofilm prevention by these AMPs.
4. Discussion and conclusions
It is widely accepted that AMPs kill bacteria or inhibit bacterial growth mainly by membrane-perturbing mechanisms [17], although other mechanisms of AMPs are also well known (e.g., lipopolysaccharide binding, bacterial stress response, cell penetration, motility inhibition, and inhibition of intracellular processes) [18]. The premise of this study is that membrane recognition by AMPs alone (without bacterial killing) can interfere with bacterial attachment to solid surfaces (e.g. medical implants or surgical sites), thereby preventing biofilm formation. Thus, it was imperative to test whether AMPs could prevent biofilm formation at subinhibitory concentrations using both susceptible and resistant strains, which distinguishes the direct killing of planktonic bacteria prior to attachment from the inhibition of biofilm formation. This was the rationale for using 1/3 MIC and 50× higher bacterial number (5×107 CFU/mL) than the suspension (106 CFU/mL) normally used in the standard planktonic GIA, with DMEM (instead of a typically nutrient rich broth) facilitating biofilm over the planktonic mode of growth. As other AMPs (e.g., LL37) have demonstrated anti-biofilm activities as well, the membranetargeting property of AMPs is likely to play a role in affecting the ability of the bacteria to initially attach to a surface. Specifically, WLBU2 is a highly cationic AMP with a charge of 13+ on the hydrophilic face and LPS-binding property, which may interfere with bacterial attachment [19], the initial step in biofilm formation. By using only 3h of AMP treatment, we mainly addressed the inhibition of this first critical step (bacterial attachment or early biofilm formation), which was consistent with the ability of AMPs to decrease or increase (in the case of mucB) the expression of biofilm-related genes. Of note, the level of housekeeping gene indicates that the effect on gene expression was not due to differences in bacterial growth, but rather specifically targeted AMP-dependent gene expression.
We have already demonstrated that WLBU2 was effective at eradicating P. aeruginosa sepsis in mice at a systemic dose of 4mg/kg, a rare demonstration of efficacy among thousands of AMPs, with a maximum tolerated dose of 12-15mg/kg) [20]. It is also much more difficult for P. aeruginosa to develop sub-MIC treatment-induced selection of resistance against WLBU2 (28 days) than colistin (15 days), LL37 (13 days), or rifampicin (3 days) [5]. This differential drug resistance development and enhanced anti-biofilm activity suggest that WLBU2 may work by more than one mechanism, depending on bacterial biofilm or planktonic mode of growth. Our gene expression study clearly showed that the effects of WLBU2 were specific, as exoS, exoT and fleN expressions were all decreased at different levels by all three cationic AMPs whereas mucB expression distinctively increased only in response to WLBU2 treatment.
As the patterns of MDR pathogens are rapidly emerging, a new approach to antimicrobial therapy is urgently needed, especially when common antibiotics do not generally prevent biofilm formation at MIC levels (Fig.1). The data suggest that AMPs with optimized structures (certainly including WLBU2) warrant further consideration for potential in vivo efficacy specifically in biofilm-associated infections.
Supplementary Material
Highlights.
WLBU2 inhibits biofilm growth by preventing bacterial attachment
WLBU2 affects gene expression consistent with reduced biofilm formation
WLBU2 activity is remarkably higher than that of colistin, LL37
AMPs prevents more biofilm at sub-MIC concentrations than antibiotics
Acknowledgments
Funding: This work was supported by the National Institutes of Health (R01 HL-125128 and AI-133351 to Y.P.D.) and the Flight Attendant Medical Research Institute (CIA-123062 to Y.P.D.). The funding agencies had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
DECLARATIONS
Competing Interests: R.C.M. holds stock in and serves on an advisory board for Peptilogics. Although a financial conflict of interest was identified based on the author’s relationship with Peptilogics, the research findings included in this publication may not necessarily be related to the interests of Peptilogics. All other authors: none to declare.
Ethical Approval: Not required.
References
- [1].Bales PM, Renke EM, May SL, Shen Y, Nelson DC. Purification and Characterization of Biofilm-Associated EPS Exopolysaccharides from ESKAPE Organisms and Other Pathogens. PLoS One. 2013;8:e67950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Bjarnsholt T. The role of bacterial biofilms in chronic infections. APMIS Suppl 2013:1–51. [DOI] [PubMed] [Google Scholar]
- [3].Ko YH, Delannoy M, Pedersen PL. Cystic fibrosis, lung infections, and a human tracheal antimicrobial peptide (hTAP). FEBS letters. 1997;405:200–8. [DOI] [PubMed] [Google Scholar]
- [4].Deslouches B, Islam K, Craigo JK, Paranjape SM, Montelaro RC, Mietzner TA. Activity of the de novo engineered antimicrobial peptide WLBU2 against Pseudomonas aeruginosa in human serum and whole blood: implications for systemic applications. Antimicrob Agents Chemother 2005;49:3208–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Deslouches B, Steckbeck JD, Craigo JK, Doi Y, Burns JL, Montelaro RC. Engineered cationic antimicrobial peptides to overcome multidrug resistance by ESKAPE pathogens. Antimicrob Agents Chemother 2015;59:1329–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Lashua LP, Melvin JA, Deslouches B, Pilewski JM, Montelaro RC, Bomberger JM. Engineered cationic antimicrobial peptide (eCAP) prevents Pseudomonas aeruginosa biofilm growth on airway epithelial cells. J Antimicrob Chemother 2016;71:2200–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Chen C, Deslouches B, Montelaro RC, Di YP. Enhanced efficacy of the engineered antimicrobial peptide WLBU2 via direct airway delivery in a murine model of Pseudomonas aeruginosa pneumonia. Clin Microbiol Infect. 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Deslouches B, Phadke SM, Lazarevic V, Cascio M, Islam K, Montelaro RC, et al. De novo generation of cationic antimicrobial peptides: influence of length and tryptophan substitution on antimicrobial activity. Antimicrob Agents Chemother 2005;49:316–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Wiegand I, Hilpert K, Hancock RE. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc 2008;3:163–75. [DOI] [PubMed] [Google Scholar]
- [10].O’Toole GA. Microtiter dish biofilm formation assay. J Vis Exp 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Liu Y, Di ME, Chu HW, Liu X, Wang L, Wenzel S, et al. Increased susceptibility to pulmonary Pseudomonas infection in Splunc1 knockout mice. J Immunol 2013;191:4259–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].O’Toole GA, Kolter R. Flagellar and twitching motility are necessary for Pseudomonas aeruginosa biofilm development. Mol Microbiol 1998;30:295–304. [DOI] [PubMed] [Google Scholar]
- [13].Cezairliyan BO, Sauer RT. Control of Pseudomonas aeruginosa AlgW protease cleavage of MucA by peptide signals and MucB. Mol Microbiol 2009;72:368–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Barbieri JT, Sun J. Pseudomonas aeruginosa ExoS and ExoT. Rev Physiol Biochem Pharmacol 2004;152:79–92. [DOI] [PubMed] [Google Scholar]
- [15].Hauser AR. The type III secretion system of Pseudomonas aeruginosa: infection by injection. Nat Rev Microbiol 2009;7:654–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Huang B, Whitchurch CB, Mattick JS. FimX, a multidomain protein connecting environmental signals to twitching motility in Pseudomonas aeruginosa. J Bacteriol 2003;185:7068–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Epand RF, Maloy WL, Ramamoorthy A, Epand RM. Probing the “charge cluster mechanism” in amphipathic helical cationic antimicrobial peptides. Biochemistry. 2010;49:4076–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Lam SJ, O’Brien-Simpson NM, Pantarat N, Sulistio A, Wong EH, Chen YY, et al. Combating multidrug-resistant Gram-negative bacteria with structurally nanoengineered antimicrobial peptide polymers. Nat Microbiol 2016;1:16162. [DOI] [PubMed] [Google Scholar]
- [19].Ryder MP, Wu X, McKelvey GR, McGuire J, Schilke KF. Binding interactions of bacterial lipopolysaccharide and the cationic amphiphilic peptides polymyxin B and WLBU2. Colloids Surf B Biointerfaces. 2014;120:81–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Deslouches B, Gonzalez IA, DeAlmeida D, Islam K, Steele C, Montelaro RC, et al. De novo-derived cationic antimicrobial peptide activity in a murine model of Pseudomonas aeruginosa bacteraemia. J Antimicrob Chemother 2007;60:669–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


