Abstract
Thermometers play an important role to study the biological significance of temperature. Fluorescent nanodiamonds (FNDs) with negatively-charged nitrogen-vacancy centers, a novel type of fluorescence-based temperature sensor, have physicochemical inertness, low cytotoxicity, extremely stable fluorescence, and unique magneto-optical properties that allow us to measure the temperature at the nanoscale level inside single cells. Here, we demonstrate that the thermosensing ability of FNDs is hardly influenced by environmental factors, such as pH, ion concentration, viscosity, molecular interaction, and organic solvent. This robustness renders FNDs reliable thermometers even under complex biological cellular environment. Moreover, the simple protocol developed here for measuring the absolute temperature inside a single cell using a single FND enables successful temperature measurement in a cell with an accuracy better than ±1°C.
Keywords: fluorescent nanodiamond, temperature sensing, nitrogen-vacancy center, magnetic resonance, cellular imaging
Significance.
Temperature sensing ability of commonly used fluorescence-based sensors is susceptible to environmental factors, which is the major concern in the practical bioapplications. We demonstrated here that the thermosensing ability of fluorescent nanodiamonds (FNDs), a novel type of temperature sensor, is hardly influenced by environmental factors, such as pH, ion concentration, viscosity, molecular interaction, and organic solvent. Furthermore, we developed a simple protocol to measure the absolute temperature inside a single cell using a single FND.
Thermal sensing is a key technique for understanding heat production in cells; thus, many kinds of fluorescence-based nanothermometers, including quantum dots, rare-earth metal complexes, polymers, and genetically encoded proteins, have been developed [1,2]. Indeed, using these thermometers, several research groups have discovered markedly high temperature inhomogeneity (>1°C) inside single cells [3–9]. Because this inhomogeneity may serve as a torque for certain biological reactions, scientists have started to unveil the biological significance of the inhomogeneity of temperature within a cell. However, Baffou et al. challenged some of these results regarding temperature distribution in a cell, who claimed that, based on standard thermodynamic considerations and scaling laws, a single cell cannot substantially raise its temperature by more than 1°C via endogenous thermogenesis [10,11]. They also claimed that biological factors, such as pH, ion concentration, and/or microviscosity, could readily affect these fluorescence-based thermometers, resulting in inaccurate temperature reading. Currently, the inhomogeneous temperature distribution within a single cell remains controversial. Hence, for understanding intrinsic heat production, the development of thermometers free from these artifacts in temperature measurement is anticipated.
Fluorescent nanodiamonds (FNDs) containing negatively-charged nitrogen-vacancy centers (NV−s) have recently been reported as a novel class of thermometers [12,13]. In Figure 1a, the energy level diagram of NV− is displayed. The ground state is a spin-triplet, and the spin sublevels, ms = 0 and ms = ±1, are split by a temperature-dependent zero-field splitting D. By monitoring the temperature-dependent shift of D using the optically detected magnetic resonance (ODMR) technique, the measurement of the temperature around FND is possible within an accuracy of ±1°C [12,14]. In addition, FNDs are well known to be highly bio-compatible and physicochemically inert, with a fluorescence that shows neither photoblinking nor photobleaching [15,16]. FNDs have considerable potential for precisely and quantitatively analyzing temperatures inside single cells because of these favorable properties.
Figure 1.
(a) Energy diagram of the NV− spin sublevels in nanodiamonds. (b) Schematic representation of the experimental setup. A typical fluorescence image is shown (24 μm × 24 μm). (c) ODMR spectra of the FND marked by a yellow circle in (b), measured at 27.0, 32.0, and 37.0°C. Close-up spectra inside the black square are shown on the right.
Nevertheless, two issues remain as obstacles for practical thermosensing inside a cell using FNDs. First, because NV−s are deeply embedded inside the diamond crystal structure, FNDs are expected to serve as robust thermometers inside cells without being influenced by biological factors. However, to date, the robustness of their thermosensing ability against environmental factors exists has not been experimentally evidenced. Second, for understanding intrinsic heat production in cells, it is important to measure absolute temperature. To achieve absolute temperature measurement, as with other temperature sensors, an in vitro calibration line must be obtained. However, the characteristics of the FND particles, such as the size, shape, impurity content, and the number of NV−s in FND, differ from particle to particle. Accordingly, each FND has a different calibration line that limits the absolute temperature measurement because achievement of both in vitro detection of calibration lines and temperature sensing in cells using identical FNDs is technically difficult. Relative temperature changes induced by exogenous heat sources in cells have been measured, which do not require calibration lines [14,17]. However, the absolute temperature has not been measured in cells using FNDs to the best of our knowledge.
Here we first reported that the thermosensing ability of FNDs is hardly influenced by pH, ion concentration, viscosity, molecular interaction, and organic solvent, having experimentally proved, for the first time, that its robustness against environmental factors renders FND as an ideal temperature sensor inside a single cell. We further present a protocol for measuring the absolute temperature inside a cell, while simple, enables absolute temperature measurement in a single cell.
Materials and Methods
Reagents and chemicals
He+ ion irradiated nanodiamond particles were donated by Prof. Huan-Cheng Chang, Academia Sinica. The nanodiamonds were annealed in vacuum at 800°C to form NV− centers. Then, the particles were oxidized in air at 450°C for 2 h to remove graphitic carbon on the surface and yield FNDs. HCl, NaOH, NaCl, glycerol and polyethylenimine (Mw: 50,000–100,000) were purchased from Wako Pure Chemical Industries, Ltd. (Osaka, Japan) and ethanol was purchased from Nacalai Tesque, Inc (Kyoto, Japan). Milli-Q water was used for all experiments. Dulbecco’s modified Eagle’s medium (DMEM: Sigma-Aldrich Co., MO, USA), with addition of penicillin-streptomycin (Life Technologies Co., NY, USA) and 10% fetal bovine serum (Biowest, MO, USA), was used for cell culture.
Surface coating of FND with polyethyleneimine
FNDs (1 mg) and polyethyleneimine (5 mg) were dissolved in Milli-Q water (1 ml), sonicated, and gently mixed for 1 h. Unbound polymers were removed from the FND-polyethyleneimine conjugates by repeated centrifugation/ wash processes with Milli-Q water.
Optically detected magnetic resonance microscope system
We configured the system on an inverted microscope system (Nikon, Ti–E) capable of measuring single-molecule fluorescence. A continuous Nd:YAG laser at 532 nm illuminated the nanodiamonds to initialize and readout the spin state of NVCs. Fluorescence was captured by an oil immersion objective lens [×100, numerical aperture (NA) = 1.49], passed through a long-wavelength pass filter so as to cut off the excitation light, and imaged by an electron-multiplying charge-coupled device camera (EMCCD, Andor, iXon860). A two-turn copper coil with a diameter of approximately1 mm was placed just above the cover glass to irradiate the sample with microwave (MW) for magnetic resonance of NVCs. A gate pulse given by a pulser (Berkley Nucleonics Corporation, 565) activated a MW generator (Agilent, E8257D) to output a MW pulse followed by amplification with linear MW power amplifiers (Mini-Circuits, PAN35-5A and ZHL-16W-43+) and transmission through a coaxial cable to the MW coil. A computer controlled the pulser and the EMCCD camera for synchronous acquisition. Fluorescence images were stored in the EMCCD camera and transferred to the computer for performing real-time data analysis. Optically detected magnetic resonance (ODMR) spectrum for a swept range of the MW frequency was obtained as the difference in fluorescence intensity between the microwave on and off. The microscope was equipped with a computer-controlled moving stage and temperature control system (TOKAI HIT, TPi-108RH26). The temperature of the sample stage was monitored using a thermocouple sensor (TOA ELECTRIC, L-TN-SP-K).
Temperature measurement
The FND samples were scattered on a glass-bottomed dish, and a solution was added (Fig. 1b). Fluorescence images were measured while the MW frequency was digitally swept across the resonant frequency ranging from 2820 to 2920 MHz in increments of 1 MHz. By this process, ODMR spectra were obtained from a single FND particle. The exposure time of the camera, the number of accumulations per MW frequency, and the number of scans were 10 ms, 64, and 4, respectively. It takes ca. 5 min to obtain the whole ODMR spectra of a single field of view. Each ODMR spectrum was then fitted to a sum of two Lorentzian functions to determine the zero-field splitting, D. We note that D is defined as the central value of the two resonances for ms = 0 to 1 and ms = 0 to −1. Temperature dependence of D, i.e., ΔD/ΔT, was determined for each FND in each environmental condition by measuring D at three temperatures (27, 32, and 37°C) and subsequent fitting to a linear function of T. The accuracy of the temperature measurements was calculated on the basis of fitting errors between the model curves and the observed data.
Cell culture and labeling
HeLa cells were cultured in DMEM in a 5% CO2 incubator at 37°C. Ten-thousand cells were plated on a glass-bottomed dish and cultured for 24 h. FNDs in Milli-Q water were added to the culture plate to obtain a final concentration of 1 μg/ml and incubated for 1 h. The medium was discarded and changed to fresh DMEM, and the cells were kept for further 3 h to ensure internalization of FNDs. Cell fixation was performed using 70% ethanol on the microscope after ODMR measurement of the live cell.
Results and Discussion
We present our experimental setup for ODMR-based thermosensing in Figure 1b. Briefly, we used an inverted fluorescence microscope equipped with a green laser for NV excitation, an EMCCD camera for field view, an MW coil for magnetic resonance, and a computer-controlled moving stage with a temperature control system. The exact temperature on the sample stage was monitored using a thermocouple. The accuracy test of our temperature controlling system is shown in Supplementary Figure S1. The temperature on the stage can be controlled with an accuracy of 0.1°C. FND samples were scattered on a glass dish and soaked in a certain solution. A cover slip was placed on the FND samples to avoid evaporation of the solution. Figure 1c shows the ODMR spectra of the FND indicated by a yellow circle in the fluorescence image in Figure 1b, measured at different temperatures (27.0, 32.0, and 37.0°C). The peak in the ODMR spectrum shifted to a lower frequency with increasing temperature. To precisely estimate the peak shift of the ODMR spectrum, we fitted the spectrum to a sum of two Lorentzian distributions.
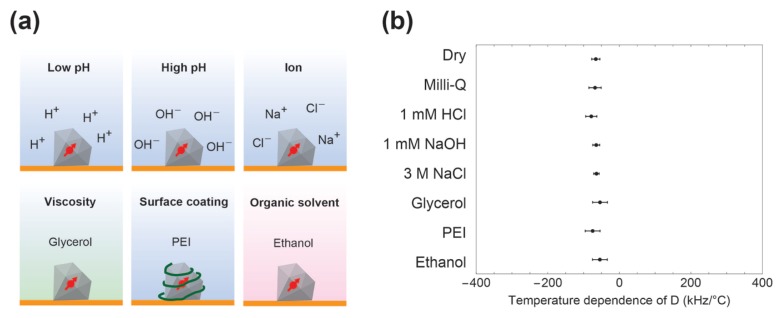
For demonstrating the robustness of FNDs as temperature sensors, we investigated the temperature dependence of D (ΔD/ΔT) under various conditions: low-pH and high-pH solutions, high ion concentration, high viscosity, surface polymer coating, and organic solvent (Fig. 2a). In the experiment, we measured ODMR spectra of the samples at 27.0, 32.0, and 37.0°C and obtained D of a single FND at each temperature (Supplementary Fig. S2a). We then determined ΔD/ΔT in various conditions: dry, Milli-Q water, 1 mM HCl aq (pH ca. 3.0), 1 mM NaOH aq (pH ca. 11.0), 3 M NaCl aq, glycerol, Milli-Q water with surface polyethylenimine coating, and ethanol to be −66 ± 11, −69 ± 17, −66 ± 10, −78 ± 15, −65 ± 10, −64 ± 7, −54 ± 20, −75 ± 21, and −54 ± 15 kHz/°C, respectively (Fig. 2b). The result showed that ΔD/ΔT was hardly affected by environmental factors, indicating that inside cells, FNDs are robust temperature sensors without interference from artifacts. The robustness against the environmental factors was not obtained from fluorescent dye molecule (Supplementary Fig. S3).
Figure 2.
(a) Illustration of our experimental conditions: FNDs and environmental factors. (b) ΔD/ΔT for the samples from at least five FNDs in each condition. Error bars indicate the standard deviations.
We found the values of ΔD/ΔT to be similar among FND particles; however, the offsets were significantly different (Supplementary Fig. S2a). We noted that the variation in the offsets was surmised to be caused not by environmental factors but rather by the characteristics of the particles (Supplementary Fig. S2b, c, and Supplementary Table S1). Therefore, we needed to obtain both a calibration line in vitro and an ODMR spectrum in a live cell from an identical FND to determine the absolute temperature, which is technically difficult. To overcome this difficulty, we developed a divergent strategy of obtaining calibration lines after cell fixation (Fig. 3a). The strategy is specifically applicable to FNDs because the structure of FND is not affected by the chemical treatments during cell fixation, unlike those of polymer and protein thermometers. In Figure 3b, we show merged images (bright field and fluorescence) of live and fixed states of a single HeLa cell that was fed FNDs. These FNDs might be internalized via endocytosis [18,19]. For the FND that is indicated by a blue circle, we first measured ODMR spectra and determined the D value in the live cell to be 2868.55 MHz (Fig. 3c). The cell was then fixed, and a calibration line was obtained for this FND particle (Fig. 3d). With reference of the calibration line, we successfully obtained the absolute temperature of 33.5°C ± 0.5°C for this FND, which is 1.5°C higher than the temperature of the culture medium.
Figure 3.
(a) Schematic illustration of temperature sensing inside a cell. (b) Merged bright field and fluorescence image of a HeLa cell before and after fixation. (c) ODMR spectrum of the FND in the live cell that are indicated by the circle in (b). The whole spectrum was obtained in 5 min and subsequently fitted to a sum of two Lorentzian functions. (d) D values from the FND sample in the fixed cell at 27.0, 32.0, and 37.0°C, with a calibration line obtained by linear fitting. From the calibration line, the temperature of the FND in the live cell was determined (dashed line), while the temperature of the medium was maintained at 32.0°C.
To find a link between biological phenomena and temperature, it is important that the thermometer used is not influenced by other biological factors. We have demonstrated the thermosensing ability of FNDs to be independent of environmental factors. The employed conditions of our thermosensing ability test were extreme and could not have been directly observed in cells, namely, FNDs would serve as a reliable thermometer even in a complex biological environment. Because conventionally employed thermometers cannot deliver a comparable level of robustness, we believe that FNDs have the potential to give a definitive answer to the controversial argument over the temperature distribution inside a single cell. To realize absolute temperature measurement in cells, we developed a novel protocol that obtains ODMR spectra in a live cell and the calibration line in a fixed cell using an identical FND. This simple protocol is suitable for FNDs because they are chemically inert and their crystal structure is not destroyed by organic solvents. The protocol might be also applicable to dye molecules [20], but cannot be applied to nano-gel polymers or protein thermometers due to the leak out of the sensors. Here in this work, we observed a significantly higher temperature (>1°C) in a single cell compared to the temperature of the culture medium. Nevertheless, in order to address the biological significance of this temperature, improvement in temporal resolution and imaging quality of our ODMR microscope to precisely determine the localization of FNDs in a single cell is required. This robust temperature sensor and our protocol open the way for temperature-related biology.
Supplementary Information
Acknowledgments
We would like to thank Dr. Huan-Cheng Chang for providing fluorescent nanodiamonds. We also gratefully acknowledge discussions with Dr. Hisashi Tadakuma and Dr. Madoka Suzuki. This study was supported in part by Grant-in-Aid for Scientific Research (KAKENHI) (26220602 to Y. H.) from Japan Society for the Promotion of Science (JSPS); Grant-in-Aid for Scientific Research (KAKENHI) (15H05931 to Y. H.) from Ministry of Education, Culture, Sports, Science and Technology (MEXT) and by Grant-in-Aid for JSPS Research Fellow (A18J002870 to S. S.) from Japan Society for the Promotion of Science (JSPS).
Footnotes
Conflicts of Interest
The authors declare no conflicts of interest.
Author Contribution
Y. H. conceived and designed the experiments. T. S. introduced the temperature control system into ODMR microscope. S. S. carried out the ODMR measurements. T. S. and S. S. wrote the manuscript. Y. H. supervised the project.
References
- 1.Okabe K, Sakaguchi R, Shi B, Kiyonaka S. Intracellular thermometry with fluorescent sensors for thermal biology. Pflügers Arch Eur. 2018;470:717–731. doi: 10.1007/s00424-018-2113-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Nakano M, Nagai T. Thermometers for monitoring cellular temperature. J Photochem Photobiol C Photochem Rev. 2017;30:2–9. [Google Scholar]
- 3.Yang J, Yang H, Lin L. Quantum dot nano thermometers reveal heterogeneous local thermogenesis in living cells. ACS Nano. 2011;5:5067–5071. doi: 10.1021/nn201142f. [DOI] [PubMed] [Google Scholar]
- 4.Okabe K, Inada N, Gota C, Harada Y, Funatsu T, Uchiyama S. Intracellular temperature mapping with a fluorescent polymeric thermometer and fluorescence lifetime imaging microscopy. Nat Commun. 2012;3:705. doi: 10.1038/ncomms1714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kiyonaka S, Kajimoto T, Sakaguchi R, Shinmi D, Omatsu-Kanbe M, Matsuura H, et al. Genetically encoded fluorescent thermosensors visualize subcellular thermoregulation in living cells. Nat Methods. 2013;10:1232–1238. doi: 10.1038/nmeth.2690. [DOI] [PubMed] [Google Scholar]
- 6.Nakano M, Arai Y, Kotera I, Okabe K, Kamei Y, Nagai T. Genetically encoded ratiometric fluorescent thermometer with wide range and rapid response. PLoS One. 2017;12:e0172344. doi: 10.1371/journal.pone.0172344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chrétien D, Bénit P, Ha HH, Keipert S, El-Khoury R, Chang YT, et al. Mitochondria are physiologically maintained at close to 50°C. PLoS Biol. 2018;16:e2003992. doi: 10.1371/journal.pbio.2003992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kiyonaka S, Sakaguchi R, Hamachi I, Yoshizaki T, Mori Y. Validating subcellular thermal changes revealed by fluorescent thermosensors. Nat Methods. 2015;12:801–802. doi: 10.1038/nmeth.3548. [DOI] [PubMed] [Google Scholar]
- 9.Suzuki M, Zeeb V, Arai S, Oyama K, Ishiwata S. The 10(5) gap issue between calculation and measurement in single-cell thermometry. Nat Methods. 2015;12:802–803. doi: 10.1038/nmeth.3551. [DOI] [PubMed] [Google Scholar]
- 10.Baffou G, Rigneault H, Marguet D, Jullien L. A critique of methods for temperature imaging in single cells. Nat Methods. 2014;11:899–901. doi: 10.1038/nmeth.3073. [DOI] [PubMed] [Google Scholar]
- 11.Baffou G, Rigneault H, Marguet D, Jullien L. Reply to: “Validating subcellular thermal changes revealed by fluorescent thermosensors” and “The 10(5) gap issue between calculation and measurement in single-cell thermometry”. Nat Methods. 2015;12:803. doi: 10.1038/nmeth.3552. [DOI] [PubMed] [Google Scholar]
- 12.Acosta VM, Bauch E, Ledbetter MP, Waxman A, Bouchard LS, Budker D. Temperature dependence of the nitrogen-vacancy magnetic resonance in diamond. Phys Rev Lett. 2010;104:070801. doi: 10.1103/PhysRevLett.104.070801. [DOI] [PubMed] [Google Scholar]
- 13.Sotoma S, Epperla CP, Chang HC. Diamond nanothermometry. ChemNanoMat. 2018;4:15–27. [Google Scholar]
- 14.Kucsko G, Maure PC, Yao NY, Kubo M, No HJ, Lo PK, et al. Nanometre-scale thermometry in a living cell. Nature. 2013;500:54–58. doi: 10.1038/nature12373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yu S-J, Kang MW, Chang HC, Chen KM, Yu YC. Bright fluorescent nanodiamonds: no photobleaching and low cytotoxicity. J Am Chem Soc. 2005;127:17604–17605. doi: 10.1021/ja0567081. [DOI] [PubMed] [Google Scholar]
- 16.Chen X, Zhang W. Diamond nanostructures for drug delivery, bioimaging, and biosensing. Chem Soc Rev. 2017;46:734–760. doi: 10.1039/c6cs00109b. [DOI] [PubMed] [Google Scholar]
- 17.Simpson DA, Morrisroe E, McCoey JM, Lombard AH, Mendios DC, Treussart F, et al. Non-neurotoxic nanodiamond probes for intraneuronal temperature mapping. ACS Nano. 2017;11:12077–12086. doi: 10.1021/acsnano.7b04850. [DOI] [PubMed] [Google Scholar]
- 18.Vaijayanthimala V, Tzeng YK, Chang HC, Li CH. The biocompatibility of fluorescent nanodiamonds and their mechanism of cellular uptake. Nanotechnology. 2009;20:425103. doi: 10.1088/0957-4484/20/42/425103. [DOI] [PubMed] [Google Scholar]
- 19.Faklaris O, Joshi V, Irinopoulou T, Tauc P, Sennour M, Girard H, et al. Nanoparticles for cell labeling: study of the uptake mechanism in mammalian cells. ACS Nano. 2009;3:3955–3962. doi: 10.1021/nn901014j. [DOI] [PubMed] [Google Scholar]
- 20.Arai S, Lee S, Zhai D, Suzuki M, Chang Y. A molecular fluorescent probe for targeted visualization of temperature at the endoplasmic reticulum. Sci Rep. 2014;4:6701. doi: 10.1038/srep06701. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.