Abstract
Scanning electrochemical microscopy (SECM) is a powerful and versatile technique for visualizing the local electrochemical activity of a surface as an ultramicroelectrode tip is moved towards or over a sample of interest using precise positioning systems. In comparison with other scanning probe techniques, SECM not only enables topographical surface mapping but also gathers chemical information with high spatial resolution. Considerable progress has been made in the analysis of biological samples, including living cells and immobilized biomacromolecules such as enzymes, antibodies and DNA fragments. Moreover, combinations of SECM with complementary analytical tools broadened its applicability and facilitated multi-functional analysis with extended life science capabilities. The aim of this review is to present a brief topical overview on recent applications of biological SECM, with particular emphasis on important technical improvements of this surface imaging technique, recommended applications and future trends.
Keywords: scanning electrochemical microscopy, microelectrodes, nanoelectrodes, chemical imaging, bio-SECM, microelectrochemistry
1. Introduction
The analytical operation of miniaturized voltammetric sensors above, at, and inside cultured or isolated living cells, living cell clusters and macro- or microstructures of immobilized bio-(macro-)molecules for sensitive local determination of biological activity is a unique strategy across disciplines. The aim is to inspect, on a fundamental level, physiological, pathological and molecular life science phenomena with high spatial and temporal resolution. The output of the bioanalysis using micro- and nanoelectrode voltammetry is data and knowledge generation in support of advancements in modern personal healthcare, drug development and nutrition, all issues that are more than ever before acute for an increasingly ageing global society. Examples of foundational explorations of the tailored bioelectroanalytical methodology are the 1969 analyses of oxygen profiles in and above microbial slime [1] and the 1973 initiation of voltammetry in brain tissue [2] with the sensitive tips made of metal and carbon, respectively. The two decades later, the move to, for instance, electrochemical detection of distinct biological processes of chemical neurotransmission at the level of single living cells and single-vesicle exocytosis [3,4] was the logical result of the accomplishment of continuous sensor and apparatus improvements. This was driven by the scientific desire and methodical necessity to extend investigations into the function of biological entities to actions within or at the smallest complete building blocks of living organisms. Selection of target cells and sensor tip positioning at or in close proximity to their membranes were initially performed in conventional electrophysiology set-ups with inverted microscopes employed for cell visualization and manually operated three-dimensional micropositioning devices used for voltammetric sensor and/or culture plate movement. However, at approximately the same time as the progression of single cell voltammetry, the invention of scanning electrochemical microscopy (SECM) occurred and the novel scanning probe system became rather quickly a favoured complementary analytical platform for the inspection and visualization of cellular activities. In a scanning electrochemical microscope, a scanning electrochemical probe (SECM tip) is a sensitive collector of redox species as the basis of a faradaic current signal that depends on both the distance to the sample and the absence or presence of sample surface redox activity. Careful computer-controlled movement across a sample of interest and acquisition of localized SECM tip currents is performed as a function of lateral (x, y) tip positions. In the optimal case, the collected data can be used to generate an interpretable real-time replication of sample topography and/or a genuine image of spatially resolved concentration profiles of redox active species in close vicinity to the evaluated sample and with a lateral resolution that is governed roughly by the tip dimension. A few tens of review articles are at present available on the broad topic ‘SECM’ and they are together a good primary source of information about the theory and practice of SECM experimentation, the technical accomplishments that helped advance SECM instrumentation and, last but not least, the many special applications in the fields of materials, energy, natural and life sciences. With special focus on biological applications of SECM several reviews are recommended [5–25].
Not considering mergers with other scanning microscopy techniques such as, among others, atomic force microscopy (AFM), scanning ion conductance microscopy (SICM) and near-field optical scanning microscopy (NSOM), the most frequently applied operation modes of SECM are the amperometric feedback and the generator/collector (G/C) modes, with the SECM tip moved at either constant z height or at constant tip-to-sample distance across the sample surface. Here, a summary of adaptations of the various technical SECM solutions for biological, single-cell and single-molecule studies is followed by the classifications of possible applications. A careful selection of cutting-edge articles from about the last 5 years will then be summarized with the aim of highlighting the enormous level of sophistication at which SECM works on living cells and immobilized bio-macromolecules. A discussion of the promising potentials of state-of-the-art ‘biological’ SECM-based detection and imaging and recommendations for near- and far-term future goals of applications of scanning miniaturized electrochemical sensors on biological cells and molecular biological entities are provided as part of the concluding section.
2. Scanning electrochemical microscopy-based live cell imaging: technical aspects, targets and potentials
Living cells are lipid double-layer membrane-confined three-dimensional small objects with heights and diameters in the low micrometre scale. They exist in primary cell cultures freshly isolated from the body tissue of interest or in secondary cell cultures as passaged constituents of cell lines. Usually, cultured living cells are used for electrophysiology experiments as seeds on thin glass coverslips or the bottom of plastic Petri dishes, with or without cell adhesion-enhancing surface layers of, for instance, poly-l-lysine. For SECM work on individual cells, careful maintenance of a suitable density of the cell population is important just to have enough healthy but distant choices. Figure 1 is a graphical illustration of the principles of SECM studies on single living cells. Selections of SECM tips for the voltammetric detection of biological cell activity include bare or chemically modified micro- or nanometre diameter disc-shaped noble metal or carbon electrodes. Independent of their size, scanning SECM tips should traverse gently over the cell to be screened and with appropriate precautions. The required contactless movement in the x/y plane is possible in computer-controlled constant-height or constant-distance modes. A typical ‘bio-SECM’ is mounted on the base plate of an inverted microscope with reasonable magnification to aid the visual assessment of cell quality before approach and to facilitate coarse alignment between a healthy cell of choice and the SECM tip prior to operation.
Figure 1.
Options for the scanning operation on single biological cells. Disc-shaped SECM tips of micrometre (a) or nanometre (b) dimensions can be used as electrochemical probes. (Online version in colour.)
(a). Constant-height mode cellular scanning electrochemical microscopy
This is the simplest procedural approach for SECM-based cell visualization and the execution of SECM assessments of cellular activity. The related workstations are fairly cheap and technically undemanding. Inexpensiveness together with the ease of device assembly makes constant-height SECM set-ups a decent entrance tool for newcomers to the field of cellular electrochemical probe microscopy. In a conventional constant-height mode of SECM the control of the vertical tip position over a cell and/or a cell-coated coverslip/Petri dish surface is the result of the amperometric recording of SECM tip currents, it, as a function of the distance to the cell-keeping solid plane, (z). In bulk electrolyte, the diffusion-limited electrochemical faradaic probe response (e.g. a dissolved oxygen reduction current) is obviously unobstructed, while close to a cell or in the vicinity of glass/plastic mass transport is hindered because of the manifestation of a gap. it is measurably suppressed due to the occurrence of the ‘negative amperometric feedback’. Information from microdisc electrode approach curves (it versus z) in positions adjacent to a selected cell together with estimates of cell heights and meaningful tip retractions are the means for adjustments of tip-to-cell distances at which the soft biological objects can be safely scanned without physical contact. Horizontal scans at the ‘working distance’ with simultaneous SECM tip current acquisition provide coarse images of cell topography which may guide placement of the tip at a detection position right above the cell membrane for subsequent stationary G/C type detection of (stimulated) release or uptake activity. Device and assay simplicity and speed of completion of full cell scans are the strengths of SECM in the constant-height mode for cell applications. The drawbacks are evident from figure 1. Cells are of irregular three-dimensional (3D) shape with ups and downs on the micrometre scale and a gradual flattening to their grounded side. Current sampling throughout constant-height activity scans thus will suffer from the impact of tip-to-cell distance variations. The micrometre-diameter active electrode discs of standard tips for constant-height SECM on cells are, on the other hand, not much smaller in diameter than the cross-sections of typical target cells and, though they are comparatively easy to fabricate and use, their spatial resolution is limited (figure 1a). Nevertheless, with clever experimental design, execution of considerate controls and careful data analysis and interpretation the constant-height SECM mode on cells is capable of revealing meaningful initial insight into the redox chemistry around metabolizing cells of various sorts. Apart from advancing the understanding of cellular physiology and pathology processes this mode may also provide methodical guidance for users of the more complex constant-distance mode. Some representative early examples of cases of successfully adapted and executed constant-height cellular SECM are the detection of redox activities of non-metastatic and metastatic human breast cells [26], heavy metal-induced plant stress visualization [27], recording the glucose uptake of individual fibroblasts and lactate production by single cancer cells by means of selective amperometric glucose or lactate micro-biosensors [28], imaging of epidermal growth factor receptors on Chinese hamster ovary and human epidermoid carcinoma A431 cell lines [29], viability assessments on human cervical cancer (HeLa) cells [30], imaging of glycan expression of human gastric carcinoma (BGC-823) cells [31] and topography and respiration activity of rat pheochromocytoma (PC12) cells [32].
(b). Constant-distance mode cellular scanning electrochemical microscopy
SECM with nanometre-sized electrochemical sensors moving at constant distance over the membranes of selected cells (figure 1b) creates images of cell topography and offers the chance for improved resolution of cell activity at neighbouring membrane locations. Moreover, it is able to explore, for instance, the spatial heterogeneity of a cell response to external stimuli. Suitable nanodisc electrodes are obviously more difficult to fabricate and handle. A collection of recent comprehensive reviews as the source of details on nanoelectrode fabrication, their electrochemical behaviour and potential applications can be found in [33–37]. The operation of nanodisc electrodes on single cells requires the preservation of a nanometre tip-to-sample working distance for the entire duration of lateral tip displacement relative to the fixed biological sample. Special feedback loop-regulated tip-to-sample distance control units were developed that use the magnitude of shear force-based dampening of intentionally induced vibrations of the nano-SECM tip as the input signal. Corresponding practical bio-SECM set-ups were realized with optical [38,39], piezoelectric [40,41] or tuning fork-based [42–44] readouts of tip vibration dampening. Exemplary accomplishments of shear force-supported constant-distance cellular bio-SECM prior to the actual period of this review are, for instance, the spatially resolved detection of neurotransmitter secretion from single pheochromocytoma (PC12) cells [38], localized measurement of stimulated NO release from adherently growing endothelial cells [45] and simultaneous detection of the cellular expression activity of secreted alkaline phosphatase outside and green fluorescent protein inside of gene-transfected single HeLa cells [44]. Though, currently, shear force-based feedback is the leading methodology for constant-distance SECM work on cells, the specific positioning task has also been accomplished with alternative distance-dependent signal inputs for the regulating control units. Examples include responsive circuitries using DC [46,47] and AC [48] SECM tip currents or probe impedance [46]. Problematic for all currently available solutions for constant-distance cellular SECM is the technical complexity of the necessary apparatus, the need for high user skills for successful operation and the relatively long duration for the completion of full living cell scans. These hurdles are at the moment against the use of bio-SECM as a regular electrophysiology tool that could valuably supplement the patch-clamp single channel current assay [49] and conventional single cell carbon-fibre amperometry [50]. Nevertheless, efforts with the SECM approach are undoubtedly justified by the remarkable gain in resolution for localized cell inspection and the feasible increase in investigational opportunities.
(c). Prospective targets for cellular scanning electrochemical microscopy
Living organisms, humans included, are complex functional assemblies of myriads of cells of all sorts. In a highly coordinated manner the various cell types work together as the building blocks of the nervous and endocrine systems, central organs such as the heart, lung, kidney and liver, components of the sensory systems including eye, ear, tongue and skin, and also the bones of the rigid skeletal framework of the body. Virtually any of the many different vertebrate cells could be a meaningful study object of constant-height or constant-distance bio-SECM. The target of investigations can be single cell processes such as respiration (oxygen consumption due to internal metabolism), chemical communication (release of neurotransmitters or hormones), development (division, migration, formation and branching of dendrites), apoptosis, etc. Rational methodical prerequisites are the availability of cells as healthy primary or secondary cultures and a relation of the cellular process of interest to dynamic measurable local changes in the concentrations of natively present or intentionally supplemented electrochemically active species in the cell membrane vicinity. The motivation for a focused electrochemical look at particular cell models may be interest in the details of the pathways of internal cellular metabolism, curiosity about the phenomena controlling cellular respiration or the desire to learn about the mechanisms behind the flux of nutrients, cytosolic waste and drugs across the cell membrane and the fundamentals of cellular chemical communication.
Figure 2 shows a selection of organs of the human body that are relevant for neurophysiology, neuropathology, endocrinology and cancer research. Cells, usually derived from laboratory animals and not humans, can be valued models for thoughtful inspections with bio-SECM. Feasible candidates for local studies of stimulated vesicular and non-vesicular hormone and neurotransmitter release are, for instance, particular brain neurons and endocrine secretory cells from laboratory rats and mice or from the secretory glands of slaughtered farmed pigs and cattle while cultured cells of the various known cancers across the body provide the prototypes for physiological assessments of tumour cell behaviour.
Figure 2.
(a) Functional elements of the nervous and endocrine systems. (b) Parts of the human body most commonly affected by cancer appearance and progression. With a variety of analytical assays individual model nerve, endocrine and cancer cells are routine objects of fundamental pathological and (electro-) physiological research for mechanistic studies regarding chemical cell communication, tumour development and oncology. Accordingly, the cellular archetypes of common medical and life science research are also ideal candidates for diagnostic inspections via bio-SECM, targeting localized electrochemical detection of the structural and functional cell features with high spatial resolution. (Online version in colour.)
In general, living cells develop distinct changes in their physical appearance and/or clear abnormalities of their physiological and biochemical functions in the course of a transformation from healthy to unhealthy or even morbid states. The normally soft and semi-permeable cellular membrane may become, for instance, stiffer and the predictable harm to chemical permeability will make membranous nutrient and metabolic waste flux in and out of the cytosol more difficult. Interdisciplinary work between electrochemists and life science researchers and strategic design of cellular SECM experiments with and without treatments by cell toxic or protective culture medium supplements is a unique chance to provide insight into the fate of healthy cells in terms of growth/proliferation and to learn about the causes that ultimately trigger cell death/apoptosis.
3. Scanning electrochemical microscopy-based live cell imaging: examples of accomplishments from 2012 to date
Within the specified period, publications related to SECM on single cells ranged from methodology advancements to the cautious use of adapted schemes for the study of particular cellular systems. As an improved strategy for scanned topographical and electrochemical cell inspections, for example, the so-called voltage switching mode of SECM with a carbon nanoelectrode as the scanning sensor was proposed (VSM-SECM, [51]). In brief, the nanoelectrode tip was positioned at individual imaging grid points via control of the probe current for the reduction of a freely diffusing redox active species ([Ru(NH3)6]3+ or O2) and brought into surface proximity using the distance-dependent negative amperometric feedback effect. A short-range probe retraction and collection of a reference current datum point (iref) in the adjusted top vertical position was performed. During a slow pre-approach at nm s−1 speed the current-regulated distance control unit of the system continuously compares the stored iref with the current recorded at the actual height and stops the downward movement upon reaching a user-defined set-point value. Here, the tip potential is changed from that required for negative feedback positioning to an anodic one for the local detection of the oxidation of redox species related to cell activity. Evidence for the ability of VSM-SECM to image cell topography with good quality was a set of detailed SECM micrographs of boar sperm cells, differentiated PC12 cells, cardiac myocytes, hair cells, hippocampal neurons and individual members of the human cell line A431, which is cultured from epidermoid carcinomas. For the last sample, the successful electrochemical visualization of the occurrence of membranous epidermal growth factor (EGFR) on the outer cell surface was also possible (figure 3). The target membrane protein was deliberately labelled with an alkaline phosphatase-tagged antibody and inspected via VSM-SECM in solutions containing p-aminophenol phosphate (PAPP) as the substrate and at nanoelectrode detection potentials suitably anodic for oxidation of the enzymatically produced p-aminophenol (PAP). The uneven distribution of EGRF on A431 lipid membranes and the absence of a link with microvilli and lamellipodia sites could be visualized, demonstrating well the potential of the approach for the creation of nanoscale maps of cellular surface features.
Figure 3.
Application of the voltage switching mode of SECM (VSM-SECM) with carbon nanoelectrodes for living A431 cell imaging. The cell topography from SECM scans in the VSM mode is shown in (a) while an electrochemical image of the presence of membranous epidermal growth factor (EGFR) on the outer cell surface is shown in (b). EGRF protein entities on A431 membranes were labelled with alkaline phosphatase prior to SECM experiments to facilitate electrochemical visualization by means of p-aminophenol phosphate (PAPP) substrate exposure and anodic p-aminophenol (PAP) detection. The carbon SECM tip with an active radius of 721.5 nm was held at −500 mV for negative feedback topography imaging and + 350 mV for PAP detection (versus Ag/AgCl), in HEPES buffer containing 10 mM [Ru(NH3)6]Cl3 and 4.7 mM PAPP. Reprinted with permission from Takahashi et al. [51] (Copyright 2012 National Academy of Sciences). (Online version in colour.)
Nebel et al. on the other hand, reported in two correlated publications the four-dimensional shear force-based constant-distance redox competition mode of SECM (4D-SF/CD-RC-SECM) as advanced routine for measurements of single cell topography and localized respiration [52]. The technique is, in fact, a sophisticated combination of the previously established 4D-SF/CD [53] and RC-SECM [54] variants, with the positioning and measuring procedures optimized for the rejection of experimental artefacts that otherwise originate from the competition between the oxygen-detecting SECM tip and the neighbouring breathing cell for the same redox species, namely dissolved molecular oxygen. The task of the ‘4D-SF/CD’ utility of 4D-SF/CD-RC-SECM is to initiate at individual x,y sample grid points a slow downward tip movement until the appearance of a hydrodynamic shear force between the SECM tip and the sample surface, invoking the discontinuation of the z approach. This closest tip-to-sample distance is the starting point for a series of stepwise tip retractions and pairs of tip current (i) and positions are collected for x, y, i plots in increasing but constant z increments. The fourth dimension of 4D-SF/CD is, therefore, the vertical position of a particular current map above the reference level corresponding to the closest approach with shear force contact. The actual sample topography is, of course, an x,y-display of the z-coordinates corresponding to the localizations of shear force induction. The ‘RC’ function of 4D-SF/CD-RC-SECM was designed to collect current data not continuously as in the constant potential amperometric mode, but instead via short potential pulses. This strategy in combination with a relatively low negative potential applied for O2 detection at the Pt tip allowed the impact of probe-induced analyte conversion to be minimized in the tiny volume between the sensing electrode and the cellular membrane. This demonstrated that the position of the microelectrode actively influences the measurements that are performed [52]. A fine-tuning of the parameters for the pulsed SECM tip detection potential and optimization of the duration of data acquisition after applying the oxygen-consuming potential steps and the working distance for the scanning probe operation actually delivered convincing duplets of SECM images of cell topography and cellular breathing activity for human embryonic kidney (HEK 293) cells as a model system (figure 4).
Figure 4.
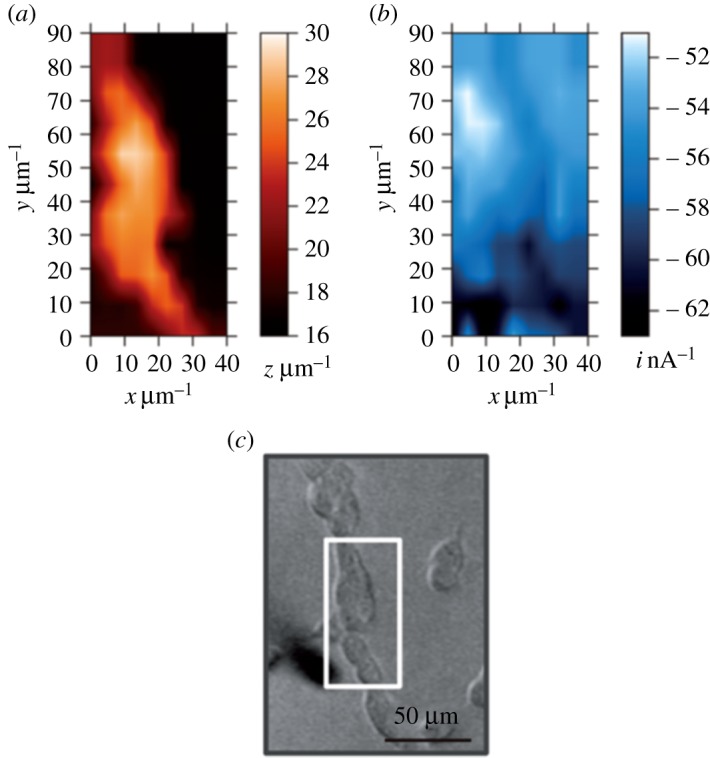
Application of the 4D-SF/CD-RC mode of SECM for the visualization of the topography and respiration activity of HEK 293 model cells. The topography image (a) is the result of the acquisition of a shear force-based distance-dependent signal while high and low SECM tip currents for oxygen reduction at the point of closest approach (b) are representative of cellular respiration. Parameters for the pulsed electrochemical oxygen monitoring were: Etip,base = 500 mV, Etip,pulse1 = 500 mV, tpulse1 = 0.2 s, Etip,pulse2 = −350 mV, tpulse2 = 0.5 s; image taken at t = 15 ms (all potentials versus Ag/AgCl/3 M KCl). An optical image of the investigated cell area is shown in (c). Reprinted with permission from Nebel et al. [52] (Copyright 2013 Wiley-VCH Verlag GmbH & Co. KGaA). (Online version in colour.)
The strength of the protocol for 4D-SF/CD-RC-SECM is the merger of distance-dependent activity profiling (slicing) with oxygen monitoring that is minimally invasive to cellular oxygen consumption and thus well adapted for true capture of the process of cell respiration. Not all reports with contributions to methodical advancements of bio-SECM can be listed here in full detail. The time-lapse mode has been reported as a tool with applications for live cell movement and morphology change tracking [55] and the imaging of the periodical evolution of reactive oxygen species in living cells [56]. It is worth mentioning that a hybrid SECM/SICM system with an ionic current-based feedback distance regulation was established with tailored dual-functional nanopipette/nanoring electrode tips for combined topographical and activity cell imaging in 2010, and the evaluation of permeation properties of the membranes of rat cardiac myocytes as was a pioneering application as a biological model [57]. A more recent technical adaptation of hyphenated SECM/SICM was optimized for 3D electrochemical and ion current imaging, which facilitated the visualization of local variations in cell membrane ion permeability and supported the creation of 3D profiles of ion concentration around cells, for single fixed adipocyte cells as the model system [58]. Quantitative recognition of molecular membrane transfer activity of living cells was realized via self-referencing SECM/SICM with specially adapted multi-functional dual-channel scanning nanopipettes [59]. A merger of AFM and SECM obtained by nanoelectrode integration into AFM cantilever tips had a similar early appearance, and proof-of-principle experiments demonstrated the feasibility of the technique for, for instance, energy conversion activity of single algae cells and chloroplasts [60]. The obvious potential of SECM/SICM and AFM/SECM combinations for fundamental and applied live cell studies has not yet been fully exploited, which may be a sign of the high procedural demand in probe fabrication and the technical hitches for their use with delicate biological objects. Recent relevant suggestions for SECM studies of cultured living cells include gentle but firm formaldehyde-based cell fixation and surfactant-induced permeabilization of the membrane of fixed cells [61]. Moreover, the use of forced convection during SECM cell imaging [62] and contrast enhancement of SECM cell images by means of conductive cell culture plate surfaces [63] were also introduced. Finally, an SICM-based solution for surface charge visualization at viable living cell membranes was also presented in joint SECM/SICM measurements [64].
Over the past half-decade, a large variety of cellular systems have been investigated with bio-SECM. Though most of the work dealt with inspections of eukaryotic cells from vertebrates as the more common biological model case, bio-SECM on fungal eukaryotic cells and bacterial prokaryotic cells has been carried out as well. In the context of the development of biofuel cells, the transmembrane charge transfer ability of eukaryotic yeast cells was, for instance, assessed by means of G/C SECM [65,66] while the biofilm formation ability of Shewanella oneidensis colonies [67], quorum sensing of aggregates of Pseudomonas aeruginosa [68] and metabolite production of Pseudomonas aeruginosa during proliferation [69] are examples of G/C-SECM studies with bacterial prokaryotes. Exotic targets for biological SECM studies were salt glands on the leaf of the halophyte Cynodon dactylon (L.) [70] and chloroplasts and their thylakoid membranes for which local photosynthetic flux measurements have been attempted [71]. SECM study entries into the literature databases that underline well the wide applicability of scanning electrochemical probe-based cellular analysis are examinations of the differentiation status of embryonic stem cells [72], evaluation of the respiration activity of blastocysts [73], monitoring intracellular enzyme activity of HeLa cells [74], analysis of beat fluctuations and oxygen consumption of cardiomyocytes [75], characterization of Ag+ toxicity on fibroblast cells [76], inspection of the multi-drug resistance of adenocarcinoma cervical cancer cells [77], the detection of reactive oxygen species in prostate cancer cells [78], the creation of insulin release profiles of single pancreatic islets [79] and the disclosure of Cd2+-induced variability in the permeation properties of human bladder cancer cells [80,81]. Recent activities also addressed topics such as SECM-based inspections of heavy metal (chromium) cell toxicity [82,83], quantitative detection of protein markers for lung cancer [84], real-time monitoring of metabolic crosstalk between neighbouring bacterial cells [85], spatial mapping of molecular uptake of chemical species at living cells [59] and the pH dependence of yeast cell viability and metabolism [66].
Interestingly, systematic SECM studies of the mechanism of hormone and neurotransmitter release from isolated endocrine and neuronal cells via single-vesicle exocytosis were not published in the reviewed period of 2012 to date even though the subject is of high importance. A large number of original studies employing conventional carbon fibre amperometry and/or microelectrode array platforms and the accounts of three recent reviews [86–88] and a book chapter on the subject [89] highlight on the other hand the relevance of the phenomena of vesicular/cellular chemical communication to the field of medical sciences. Also, an exciting move has been made by application of conventional amperometric detection of single-vesicle exocytosis towards measurements with finite conical carbon nanoelectrodes inside the very small structures of single synapses of neuronal cells [90,91]. Apparently, there is currently a lag of success with SECM-based amperometry of exocytosis behind secretory cell amperometry with manually positioned carbon nano- or microelectrodes. The reason for this may be that the SECM instrument developers are not neuroscientists. Their focus is not primarily on applications for secretory cell/neuron studies. The high device and protocol complexity of single cell SECM, in particular of the constant-distant mode employing voltammetric nanoelectrode SECM tips, is an inherent weakness and a barrier to the rapid and routine incorporation of these methods in chemical release studies at the cellular level.
The focus above was predominantly on representative examples of recent SECM work that exploited voltammetric microelectrodes as miniaturized detectors of localized cell activity. However, it is also worth briefly mentioning past research efforts addressing scanning potentiometric sensor tips for SECM-type electrochemical cell mapping. Applications of needle-type ion-selective microelectrodes for cytosolic ion concentration measurements have been common for several decades in specialized electrophysiology laboratories [92–98]. The development of ion-selective potentiometric microelectrodes for so-called biological scanning potentiometric microscopy and especially the details of potentiometric probe fabrication can be found in [99–105] and some recent reviews [7,106,107]. Ion-selective electrochemical probe microscopy for biological imaging was used for the visualization of pH profiles of discs of yeast cells embedded in agarose gels [100], measurements of the extracellular pH value of various adherently growing mammalian cells [108], the visualization of K+ ion transfer through gramicidin channels in a horizontal cell membrane-like bilayer lipid membrane [109], the acquisition of topography and ion flux images of the membrane of human embryonic kidney 293 cells (HEK 293) [110], and finally lateral mapping of ion transport through membranes of seeded epithelial cell monolayers [111,112]. This representative work is clearly demonstrating the potential of life cell inspections with scanning ion-selective potentiometric probe tips. Accordingly, the strategy is valuable as an alternative to amperometric SECM tests in proximity to heterogeneously active cell membranes and can contribute well to multi-functional cellular SECM developments.
4. Scanning electrochemical microscopy imaging of immobilized biomolecules: from proteins to DNA
Applications of SECM in the investigation of biological systems have also been directed towards the measurement of cellular components or small molecules that will allow better insight into the overall function of living organisms. As SECM is focused on small and localized measurement areas, not only high spatial but also high temporal resolution is required in many experiments. In addition to cells, several species of biological origin have been the target in SECM measurements, including enzymes, antibodies, DNA and different cellular components. The earliest SECM images of biological macromolecules included samples of DNA, haemocyanin, monoclonal IgG and glucose oxidase (GOx) adsorbed on a mica substrate using a tungsten tip. By working under high relative humidity conditions, the analysed sample was embedded in a thin layer of liquid, allowing the electrochemical reactions to take place at the tip and producing a faradaic current. Therefore, purely topographic images were obtained in the constant-current mode but with very high resolution because only the end of the tip was in contact with the water layer adsorbed on the sample [113,114]. Since then, SECM has been widely applied for the analysis of biological processes at the molecular level. The characterization of proteins allows, for example, antigen–antibody interactions to be recognized and the imaging of enzymatic processes. Moreover, interesting experiments were developed for imaging immobilized DNA, which was first investigated by SECM more than one decade ago. Advanced studies now allow the sensitive identification of basepair mismatches in double-stranded DNA (ds-DNA). In addition, the development of miniaturized analytical systems for enzyme sensors, immunoassays and DNA analysis make the application of SECM attractive for the optimization and further characterization of sensing platforms as well as the evaluation of detection strategies. The high electrochemical resolution achievable with SECM is ideally suited to determining the distribution of electroactive species in the different reaction layers of in operando devices and to resolve and detect problems associated with crosstalk between sensing elements, making SECM a powerful quality control technique [9]. SECM has thus been widely exploited in the characterization of a variety of different sensors and systems.
(a). Imaging of proteins and enzymatic activity
Electrochemical imaging of proteins requires the availability of electrochemically active compounds. In many cases, the activity of a particular protein induces addressable variations in the local concentration of ions or redox molecules that are easily measurable with an electrochemical probe, such as, among others, molecular oxygen, hydrogen peroxide, molecular hydrogen or protons. In the case of oxidoreductases, artificial electrochemically reversible redox mediators can be used, allowing a higher reproducibility and sensitivity [115].
SECM has been used mainly to detect the products resulting from electron transfer reactions by an investigated enzyme immobilized on a variety of surfaces. A summary and overview about previous works dealing with imaging of enzymatic activity can be found in earlier reviews [6,9]. Although to date a wide range of proteins remain to be characterized by SECM, the basic ideas shown in the past have been expanded for the analysis of a considerable number of immobilized proteins. As summarized in figure 5, mainly two different SECM modes are used in the analysis of enzymes, either applying different redox mediators in the feedback mode or detecting a generated product in the G/C mode of SECM. Enzyme-mediated feedback imaging (figure 5a) involves the replacement of the enzyme's cosubstrate by a redox mediator that is regenerated at the tip. Here, the progress of the biochemical reaction is dependent on the presence of the tip [9]. A positive feedback is obtained, with the magnitude of the signal directly related to the activity of the investigated enzyme [116]. Upon removal of the enzyme substrate, the feedback signal stops. Since the redox reaction can only occur in the vicinity of the tip, the feedback mode has an inherently higher spatial resolution. In this mode, it is of significant importance that the components of the mediator solution are not oxidized or reduced at the tip and, equally important, they should not interfere with the activity of the evaluated protein [6]. On the other hand, the G/C mode (figure 5b) is based on the direct oxidation or reduction of an enzyme-generated product at the SECM tip. Since the background current is very low, any change in current can be attributed to the enzymatic activity. Thus, the G/C mode offers a higher sensitivity. The tip can be scanned to generate an image of the local concentration of a molecule generated or consumed by the enzyme. However, the diffusion of the generated products away from the source of their formation prevents high-resolution analysis and an accurate location of the evaluated protein.
Figure 5.

Schematic diagram comparing the feedback and generator/collector (G/C) modes of SECM used for enzyme imaging. (a) Enzyme-mediated positive feedback. The reaction only occurs when the tip is present. (b) G/C mode in the presence of a redox enzyme. A product of the enzymatic reaction is collected at the tip, either (i) the oxidized form of a redox mediator in solution or (ii) the enzymatically converted substrate. (Online version in colour.)
Novel advances in protein imaging include the application of SECM in the feedback mode using ferrocyanide as the redox mediator for the study of the activity of laccase and bilirubin oxidase [117]. By enclosing the entire SECM measurement system, it was possible to work under a controlled atmosphere. The redox activity of laccase was evaluated under argon and oxygen, demonstrating a significant increase in the measured cathodic current when oxygen was present. In addition, by comparing the activity of laccase and bilirubin oxidase in solutions of different pH values, bilirubin oxidase was found to exhibit higher activity especially at neutral pH values. The versatility of the proposed methodology allows in-depth characterization of enzymatic activity under different and controlled conditions. Moreover, by using soft carbon microelectrodes as SECM tips, it was possible to evaluate the antioxidant activity of apple peel under a contact-scanning mode [118]. The use of soft probes made of flexible polymeric materials with built-in microelectrodes provides the possibility for a simple analysis of corrugated biological surfaces by working under constant gentle contact with the sample. Thus, a constant working distance is ensured without causing damage to the investigated surface or the SECM tip.
Recently, an increased number of miniaturized biosensors capable of being used as SECM probes has been reported. Interesting examples include the development of a microbiosensor for hydrogen detection at photosystem I-Pt nanoparticle bio-assemblies [119] capable of improving the detection capabilities of a previously used unmodified SECM tip [120]. Moreover, the implementation of microbiosensors for the detection of adenosine-5′-triphosphate (ATP) and glucose by co-entrapment of the enzymes GOx and hexokinase in a poly(benzoxazine) derivative [121] and a poly(metaphenylendiamine)-modified SECM tip incorporating the enzyme d-amino acid oxidase for the detection of d-serine [122] were demonstrated.
SECM imaging of enzymes has also been applied for the evaluation of advanced enzymatic assemblies at surface-confined enzyme-based logic gates. An insulating glass surface was modified with arrays of GOx, invertase (INV) and peroxidase (HRP) as a basis for two different AND and XOR logic gates, each of them consisting of two enzymes as Boolean operators. An AND gate was assembled consisting of GOx and INV using ferrocenemethanol (FMA) as the redox mediator in solution. Sucrose was catalytically cleaved by INV, while the immobilized GOx oxidized the generated glucose with an associated reduction of oxidized FMA, which in turn was re-oxidized at the tip, thus creating a positive feedback signal only if both inputs are positive. Similarly, a XOR logic gate was fabricated with a GOx/HRP patterned surface. While GOx catalyses the reduction of oxidized FMA, HRP catalyses the oxidation of FMA upon reduction of H2O2. Therefore, a positive output is obtained if only one of the inputs is positive [123].
SECM was also coupled to a continuous nanoflow system [124]. The specially designed tip consisted of a capillary injector surrounded by a microring electrode (r = 11.5 µm). The assembly was achieved by embedding a thin gold film between a pulled fused-silica capillary and a borosilicate glass capillary. The apex of the tip was later bevelled while applying pressurized air from the back of the capillary to keep the central opening unobstructed. The system was used for imaging of surfaces in the G/C mode with important advantages over conventional SECM experiments, including higher imaging resolution, immunity from interference by species in the bulk solution or at other sides of the substrate, elimination of the feedback current and versatility of initiating surface reactions by introducing different reactants into the flowing stream. Although of increased complexity due to the need for a precision pump capable of delivering solutions at a flux of a few nl min−1 to achieve an adequate collection efficiency, the applicability of the system was demonstrated by analysing HRP microarrays. By flowing H2O2 and FMA through the injector, the oxidized form of the redox mediator generated at the modified surface was detected (figure 6).
Figure 6.
(a) Illustration of the developed continuous nanoflow (CNF)-SECM system for imaging of enzyme spots. (b) Comparison of the resolution obtained with the conventional generator/collector mode (SG-TC) and the implemented CNF-SECM mode. Reprinted with permission from Kai et al. [124] (Copyright 2015 American Chemical Society). (Online version in colour.)
As has been recently addressed in a critical review on SECM-based multi-functional imaging [35], with the development of hybrid scanning probe microscopy techniques complementary information about the evaluation of a sample is easily and simultaneously obtained. In this regard, concurrent imaging of enzyme activity by detection of enzymatic products during AFM examination of a surface was demonstrated [8,125]. The coupling of these two techniques provides tremendous benefits, allowing nanometre-resolution topographic mapping of the evaluated surface with the concomitant acquisition of electrochemical activity from the enzyme. Using bifunctional SECM-AFM probes and working in the G/C mode of SECM and the contact mode of AFM, the approach was used for the evaluation of GOx and HRP activity [126]. A hybrid system of SECM and SICM was also developed, using concentric probes comprising an SICM tip surrounded by a nanoring electrode for SECM measurements [57]. Simultaneous imaging of the topography and spatial distribution of electrochemical species was possible in the evaluation of different enzymes, i.e. HRP and GOx, and single live cells.
Immunoassay strategies have proved to be particularly interesting for the specific recognition of target proteins or even smaller analytes. As recently exemplified in the detection of tyrosinase, three different SECM strategies for imaging of protein-modified surfaces were compared [127]. An indirect detection of tyrosinase adsorbed on polymeric membranes is possible by using the SECM tip for the reduction of O2, mapping the differential pore oxygen concentrations induced by the local hydrophobic changes caused by the adsorption of the enzyme on the porous membrane. Although simple, the method is poorly selective. Direct monitoring of enzymatic activity was realized by detecting the reaction products from the enzymatic conversion of l-3,4-dihydroxyphenylalanine (l-DOPA). However, the low specificity in the collection of enzymatic products due to the high applied microelectrode potential and fouling of the tip surface by reaction products are the main disadvantages of this strategy. Specific recognition of tyrosinase spots was then realized by implementing an immunoassay strategy applying a secondary antibody labelled with HRP and using the G/C mode of SECM for detection.
In a similar manner, AFM-SECM has been used in combination with redox immunolabelling for mapping the protein distribution on individual virus particles [128]. While the obtained topographic information allows the identification of isolated particles, the electrochemical current response permits the effective imaging of specifically labelled proteins.
Recent advances in the electrochemical detection of enzyme-based immunoassays became possible by minimization of non-specific adsorptions [129]. The concept was shown in an electrochemical immunoassay against the Epstein–Barr virus with SECM readout in the G/C mode. Using secondary antibodies marked with alkaline phosphatase, the enzymatic substrate PAPP was converted to PAP, which in turn was oxidized at the SECM tip. The assay incorporated an innovative method for the removal of non-specific adsorption of immunoreagents. A previously deposited self-assembled monolayer (SAM) was locally modified by means of an electrochemically induced pH modulation at a positioned SECM tip. Local ester cleavage led to the availability of carboxylic groups, which after activation can bind the antigen. After the sequential steps of the immunoassay were performed, a final step was introduced for global cleavage of the remaining ester groups by incubation in acetic acid and leading to chemically induced lift-off with the consequent removal of any non-specifically bound proteins.
Moreover, recent advances in SECM imaging of antigen–antibody interactions include examples for the determination of small molecular weight analytes. The G/C mode of SECM was used for imaging of the immunological event in a direct competitive immunoassay against antibiotics using selective capture antibodies and an HRP-labelled analogue [130]. Furthermore, the same assay was imaged after enzyme-catalysed deposition of Ag nanoparticles [131]. In this case, visualization of deposited Ag nanoparticle clusters related to the immunological event was enabled by a competition between the tip and the modified substrate for the oxidation of ferrocyanide using a bipotentiostatic SECM approach that allowed high contrast visualization.
Imaging of antigen–antibody interactions with SECM was predominantly based on labelling with a reporter molecule, e.g. a redox active molecule or an enzyme, capable of providing an electrochemical readout. Nevertheless, label-free imaging of antibody–antigen interactions using SECM has also been suggested. Using ferrocenecarboxylic acid as the redox mediator, differences between the binding of complementary and non-complementary antigens to specifically immobilized antibodies were obtained, attributed to different electrostatic interactions of the anionic redox mediator and the free or bound antibodies [132,133].
(b). DNA imaging
The study of DNA modified surfaces and hybridization events with SECM is particularly attractive for applications including gene expression [9], the detection and quantification of hybridization events [134] and the development and characterization of DNA chips [135]. Similarly to immunological assays, ds-DNA has been detected by monitoring the hybridization event using electrochemically active labels (figure 7a). Recent reports include SECM imaging of hybridization between target DNA and an immobilized hairpin probe with enzymatic amplification. A streptavidin–HRP conjugate is captured by the immobilized probe DNA modified with biotin at its 3′ end after hybridization with target DNA. In the presence of H2O2, hydroquinone is oxidized to benzoquinone through the HRP-catalysed reaction and the generated benzoquinone corresponding to the amount of target DNA is reduced at the SECM tip [136]. HRP-modified SiO2 nanoparticles have been used in a sandwich DNA assay where the capture probe immobilized on a gold surface is hybridized with target DNA and a biotinylated indicator probe. The enzymatic label linked to the sandwich structure through a biotin–streptavidin interaction allowed SECM imaging of hybridization [137,138]. Signal amplification was also reported by using long DNA concatemers. The target DNA is hybridized with thiol-tethered DNA capture probes. After addition of DNA auxiliary probes, continuous self-assembly with alternating long DNA led to the formation of concatemers. Finally, streptavidin–HRP was linked to the long concatemers through biotin–streptavidin interaction. This assay was demonstrated with the simultaneous detection of four kinds of target DNA through SECM imaging [139]. Moreover, SECM has been also used as a valuable tool for the evaluation of a potential-pulsed assisted protocol for DNA surface modification and thiol passivation leading to minimized non-specific adsorptions [140].
Figure 7.
SECM strategies for imaging of DNA modified surfaces and the detection of the hybridization event. (a) Use of labelled DNA strands for providing a redox signal measurable with the SECM tip. (b) Use of redox active intercalators capable of binding to virtually any dsDNA. (c) Repelling mode of SECM. While full recycling of the redox mediator in solution occurs at the electrode surface, the [Fe(CN)6]4− ions generated at the tip experience coulombic repulsions above a DNA modified spot, therefore leading to a decrease in tip current. (Online version in colour.)
To overcome the use of labelled DNA strands, more universal methods for DNA detection are required. One promising strategy is the use of redox intercalators that interact and form a stable complex with the ds-DNA and permit the electrochemical detection of the hybridization event. In this way, SECM imaging of DNA microarrays on gold surfaces was reported using redox-active intercalators, e.g. methylene blue [141] or Nile blue [142], in combination with ferrocyanide as the redox mediator in solution (figure 7b). By applying a negative potential to the substrate, the intercalator is electrochemically reduced by electron transfer from the underlying electrode surface. The product is then re-oxidized in solution by ferricyanide generated at the SECM tip with the concomitant generation of a feedback current. Microarrays of DNA were successfully visualized with this method, discriminating between single-stranded DNA (ss-DNA) and ds-DNA and the presence of basepair mismatches [141].
One of the most interesting improvements in the imaging of immobilized ss-DNA and DNA hybridization was done by the implementation of the repelling mode of SECM [143,144]. Taking advantage of the negatively charged phosphate backbone of DNA strands, a straightforward electrostatic approach was developed to visualize surface-bound DNA probes. The method is based on the feedback mode of SECM and the presence of coulombic interactions between the negatively charged DNA and a freely diffusing redox mediator. The SECM tip was scanned over a gold electrode modified with spots of immobilized DNA, at a constant tip-to-sample distance and using the negatively charged species [Fe(CN)6]3− as the redox mediator. Consequently, a significant decrease in the feedback current recorded at the tip for the regeneration of the redox mediator was observed over DNA modified regions because of electrostatic repulsion that hampers the diffusion of the mediator to the gold surface and prevents the recycling of the redox probe (figure 7c). The assumption was confirmed by using a positively charged redox mediator, i.e. [Ru(NH3)6]3+, which provided a negligible decrease in the recorded current [144]. Since after hybridization of ss-DNA with a complementary strand the net negative charge on the DNA modified surface increases, an additional increase in the electrostatic repulsion of the redox probe also allows the detection of hybridization events.
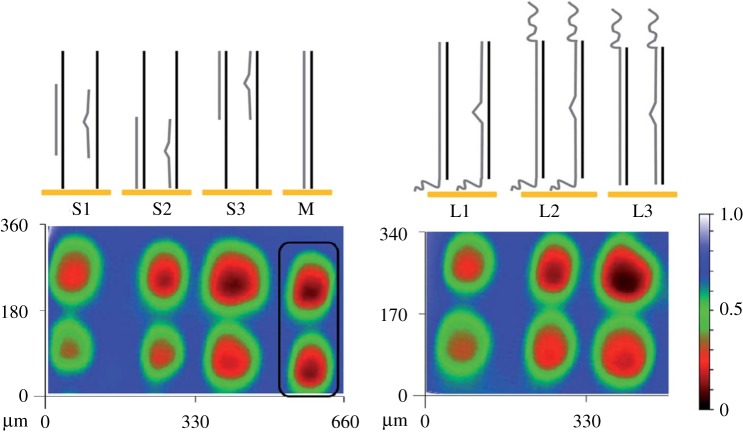
Based on this strategy, later reports have shown the possibility of detecting nucleotide mismatches in ds-DNA (figure 8). An increased sensitivity of the implemented method was obtained in the presence of bivalent ions, such as Zn2+. The presence of Zn2+ ions reduces the electrostatic repulsion between the negatively charged DNA and the redox mediator, allowing for facilitated diffusion of the latter to the substrate, with the associated increase in feedback current. By this, an increased sensitivity in the detection of single-nucleotide mismatches was achieved because the change in tip current upon addition of Zn2+ was much higher in the case of fully matched DNA than in the presence of mismatches [146–148]. This approach allowed differentiation between mitochondrial DNA fragments of the cytochrome c1 oxidase of three related species in a platform that could be used multiple times by dehybridization and rehybridization of capture strands [149]. Interestingly, the method was also applied for the evaluation of the target sequence size and the impact of overhangs in probe and target strands on single nucleotide mismatch detection in ds-DNA films (figure 8). After hybridization and analysis of fully complementary and single nucleotide mismatched probe and target strands of different size, it was concluded that the presence of overhangs in ds-DNA films affects the film order, still allowing the detection of mismatches in the ds-DNA films. Moreover, while for longer complementary strands, the position of the overhang plays an important role, the position of hybridization of the complementary strand along the probe sequence is more important in the case of short complementary strands [145].
Figure 8.
Schematics of ds-DNA (matched and mismatched) films evaluated and corresponding SECM images obtained. S1–S3: short complementary strands; L1–L3: long complementary strands; M: fully matched ds-DNA. The scale bar shows the intensity of the current. Adapted with permission from Shamsi & Kraatz [145] (Copyright 2013 Royal Society of Chemistry). (Online version in colour.)
5. Concluding remarks
(a). Challenges and future aspects of single cell scanning electrochemical microscopy
The most imminent challenge for cellular SECM is the ability to undergo maturation from a high-technology approach restricted to applications in expert electrochemical laboratories to a robust standard methodology with potential for widespread usage in life and medical science research facilities. Scanned electrochemical measurements at single living cell levels are not yet really explored for the many existing solutions of chemically modified electrodes including enzyme biosensors and immunosensors, the wide range of ion-selective potentiometric micro- and nanoelectrodes and electrochemical scanning probe detectors made of advanced sensor materials such as diamond [150]. Miniaturization of sensor structures to suitable geometries for SECM and use of completely novel tip types in multi-functional biological SECM devices are requisite steps towards possible cell inspections. Last but not least the plethora of feasible living cell targets (refer to figure 2) has not yet been approached with biological SECM and, so far, un-inspected healthy and defect cell choices offer plenty of options for the design of useful experiments for joint SECM-based topography and local chemical activity measurements.
(b). Challenges and future aspects of scanning electrochemical microscopy studies of immobilized biomolecules
Although SECM has proven to be extremely useful and adequate for imaging of immobilized biomolecules, considerable advances in the technique should be implemented to overcome current limitations and meet future challenges. The combination of SECM with techniques allowing a high spatial resolution, e.g. AFM-SECM or SICM-SECM [35], showed an important advance in high-resolution electrochemical imaging and large-scale mapping. Still, research has to evolve from the proof of concept of the experimental set-up and methodology to the implementation of interesting applications pushing the limits of the technique. Until now, only a limited amount of proteins have been investigated with SECM and more advanced methodologies are required for the fast and reliable detection of the DNA hybridization event. The development of multi-functional and selective tips will lead to a better understanding of many biological phenomena. This will also require not only an improvement in achievable spatial resolution but also the possibility of attaining higher imaging speed, because the temporal resolution is crucial to evaluate many biological investigated processes. Single-molecule detection has proven to be possible [151], yet new experimental platforms and analytical methodologies must be developed to reach the possibility of a comprehensive study of single biocatalytic systems.
Acknowledgements
We gratefully acknowledge the support of our funders.
Data accessibility
This work does not include any additional experimental data.
Authors' contributions
The manuscript was written with contributions from all authors. All the authors gave their final approval for publication.
Competing interests
The authors have no competing interests.
Funding
A.S. is grateful to the Thailand Research Fund (TRF) and the Suranaree University of Technology in Nakhon Ratchsima for kind joint financial support through Basic Research grant no.~BRG 568000013. F.C. and W.S. acknowledge the Cluster of Excellence RESOLV (EXC 1069) funded by the Deutsche Forschungsgemeinschaft (DFG), and the Deutsch-Israelische Projektkooperation (DIP) in the framework of the project ‘Nanoengineered Optobioelectronics with Biomaterials and Bioinspired Assemblies'.
References
- 1.Whalen WJ, Bungay HR, Sanders WM. 1969. Microelectrode determination of oxygen profiles in microbial slime systems. Environ. Sci. Technol. 3, 1297–1298. ( 10.1021/es60035a004) [DOI] [Google Scholar]
- 2.Kissinger PT, Hart JB, Adams RN. 1973. Voltammetry in brain tissue: a new neurophysiological measurement. Brain Res. 55, 209–213. ( 10.1016/0006-8993(73)90503-9) [DOI] [PubMed] [Google Scholar]
- 3.Wightman RM, Jankowski JA, Kennedy RT, Kawagoe KT, Schroeder TJ, Leszczyszyn DJ, Near JA, Diliberto EJ, Viveros OH. 1991. Temporally resolved catecholamine spikes correspond to single vesicle release from individual chromaffin cells. Proc. Natl Acad. Sci. USA 88, 10 754–10 758. ( 10.1073/pnas.88.23.10754) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chow RH, Ruden L, Neher E. 1992. Delay in vesicle fusion revealed by electrochemical monitoring of single secretory events in adrenal chromaffin cells. Nature 356, 60–63. ( 10.1038/356060a0) [DOI] [PubMed] [Google Scholar]
- 5.Yasukawa T, Kaya T, Matsue T. 2000. Characterization and imaging of single cells with scanning electrochemical microscopy. Electroanalysis 12, 653–659. [Google Scholar]
- 6.Wittstock G. 2001. Modification and characterization of artificially patterned enzymatically active surfaces by scanning electrochemical microscopy. Fresen. J. Anal. Chem. 370, 303–315. ( 10.1007/s002160100795) [DOI] [PubMed] [Google Scholar]
- 7.Bard AJ, Li X, Zhan W. 2006. Chemically imaging living cells by scanning electrochemical microscopy. Biosens. Bioelectron. 22, 461–472. ( 10.1016/j.bios.2006.04.028) [DOI] [PubMed] [Google Scholar]
- 8.Edwards MA, Martin S, Whitworth AL, Macpherson JV, Unwin PR. 2006. Scanning electrochemical microscopy. Principles and applications to biophysical systems. Physiol. Meas. 27, R63–R108. ( 10.1088/0967-3334/27/12/R01) [DOI] [PubMed] [Google Scholar]
- 9.Roberts WS, Lonsdale DJ, Griffiths J, Higson SPJ. 2007. Advances in the application of scanning electrochemical microscopy to bioanalytical systems. Biosens. Bioelectron. 23, 301–318. ( 10.1016/j.bios.2007.06.020) [DOI] [PubMed] [Google Scholar]
- 10.Schulte A, Schuhmann W. 2007. Single-cell microelectrochemistry. Angew. Chem. Int. Ed. 46, 8760–8777. ( 10.1002/anie.200604851) [DOI] [PubMed] [Google Scholar]
- 11.Zhang M, Wittstock G, Shao Y, Girault HH. 2007. Scanning electrochemical microscopy as a readout tool for protein electrophoresis. Anal. Chem. 79, 4833–4839. ( 10.1021/ac062356k) [DOI] [PubMed] [Google Scholar]
- 12.Amemiya S, Bard AJ, Fan F-RF, Mirkin MV, Unwin PR. 2008. Scanning electrochemical microscopy. Annu. Rev. Phys. Chem. 1, 95–131. ( 10.1146/annurev.anchem.1.031207.112938) [DOI] [PubMed] [Google Scholar]
- 13.Schulte A, Nebel M, Schuhmann W. 2010. Scanning electrochemical microscopy in neuroscience. Annu. Rev. Anal. Chem. 3, 299–318. ( 10.1146/annurev.anchem.111808.073651) [DOI] [PubMed] [Google Scholar]
- 14.Schulte A, Nebel M, Schuhmann W. 2012. Single live cell topography and activity imaging with the shear-force-based constant-distance scanning electrochemical microscope. Methods Enzymol. 504, 237–254. ( 10.1016/B978-0-12-391857-4.00012-4) [DOI] [PubMed] [Google Scholar]
- 15.Beaulieu I, Kuss S, Mauzeroll J, Geissler M. 2011. Biological scanning electrochemical microscopy and its application to live cell studies. Anal. Chem. 83, 1485–1492. ( 10.1021/ac101906a) [DOI] [PubMed] [Google Scholar]
- 16.Bergner S, Vatsyayan P, Matysik F-M. 2013. Recent advances in high resolution scanning electrochemical microscopy of living cells: a review. Anal. Chim. Acta 775, 1–13. ( 10.1016/j.aca.2012.12.042) [DOI] [PubMed] [Google Scholar]
- 17.Matsue T. 2013. Bioimaging with micro/nanoelectrode systems. Anal. Sci. 29, 171–179. ( 10.2116/analsci.29.171) [DOI] [PubMed] [Google Scholar]
- 18.Takahashi Y, Shiku H, Matsue T. 2015. Scanning electrochemical microscopy for imaging single cells and biomolecules. In Nanobiosensors and nanobioanalyses (eds Vestergaard MDC, Kerman K, Hsing I-M, Tamiya E), pp. 335–352. Tokyo, Japan: Springer. [Google Scholar]
- 19.Holzinger A, Steinbach C, Kranz C. 2015. Chapter 4. Scanning electrochemical microscopy (SECM): fundamentals and applications in life sciences. In Electrochemical strategies in detection science (ed. Arrigan DWM.), pp. 125–169. Cambridge, UK: Royal Society of Chemistry. [Google Scholar]
- 20.Nagy L, Nagy G. 2017. Application of scanning electrochemical microscopy in bioanalytical chemistry. In Trends in bioelectroanalysis (ed. Matysik F-M.), vol. 6, pp. 281–339. Cham, Switzerland: Springer International Publishing. [Google Scholar]
- 21.Zoski CG. 2017. Nanoscale scanning electrochemical microscopy. Emerging advances in applications and theory. Curr. Opin. Electrochem. 1, 46–52. ( 10.1016/j.coelec.2017.01.002) [DOI] [Google Scholar]
- 22.Takahashi Y, Kumatani A, Shiku H, Matsue T. 2017. Scanning probe microscopy for nanoscale electrochemical imaging. Anal. Chem. 89, 342–357. ( 10.1021/acs.analchem.6b04355) [DOI] [PubMed] [Google Scholar]
- 23.Bentley CL, Kang M, Unwin PR. 2017. Scanning electrochemical cell microscopy. New perspectives on electrode processes in action. Curr. Opin. Electrochem. 6, 23–30. ( 10.1016/j.coelec.2017.06.011) [DOI] [Google Scholar]
- 24.Izquierdo J, Knittel P, Kranz C. 2018. Scanning electrochemical microscopy: an analytical perspective. Anal. Bioanal. Chem. 410, 307–324. ( 10.1007/s00216-017-0742-7) [DOI] [PubMed] [Google Scholar]
- 25.Kai T, Zoski CG, Bard AJ. 2018. Scanning electrochemical microscopy at the nanometer level. Chem. Commun. 54, 1934–1947. ( 10.1039/c7cc09777h) [DOI] [PubMed] [Google Scholar]
- 26.Liu B, Rotenberg SA, Mirkin MV. 2000. Scanning electrochemical microscopy of living cells. Different redox activities of nonmetastatic and metastatic human breast cells. Proc. Natl Acad. Sci. USA 97, 9855–9860. ( 10.1073/pnas.97.18.9855) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhu R, Macfie SM, Ding Z. 2005. Cadmium-induced plant stress investigated by scanning electrochemical microscopy. J. Exp. Bot. 56, 2831–2838. ( 10.1093/jxb/eri275) [DOI] [PubMed] [Google Scholar]
- 28.Ciobanu M, Taylor DE Jr, Wilburn JP, Cliffel DE. 2008. Glucose and lactate biosensors for scanning electrochemical microscopy imaging of single live cells. Anal. Chem. 80, 2717–2727. ( 10.1021/ac7021184) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Takahashi Y, Miyamoto T, Shiku H, Asano R, Yasukawa T, Kumagai I, Matsue T. 2009. Electrochemical detection of epidermal growth factor receptors on a single living cell surface by scanning electrochemical microscopy. Anal. Chem. 81, 2785–2790. ( 10.1021/ac900195m) [DOI] [PubMed] [Google Scholar]
- 30.Li X, Bard AJ. 2009. Scanning electrochemical microscopy of HeLa cells: effects of ferrocene methanol and silver ion. J. Electroanal. Chem. 628, 35–42. ( 10.1016/j.jelechem.2009.01.002) [DOI] [Google Scholar]
- 31.Xue Y, Ding L, Lei J, Yan F, Ju H. 2010. In situ electrochemical imaging of membrane glycan expression on micropatterned adherent single cells. Anal. Chem. 82, 7112–7118. ( 10.1021/ac101688p) [DOI] [PubMed] [Google Scholar]
- 32.Koch JA, Baur MB, Woodall EL, Baur JE. 2012. Alternating current scanning electrochemical microscopy with simultaneous fast-scan cyclic voltammetry. Anal. Chem. 84, 9537–9543. ( 10.1021/ac302402p) [DOI] [PubMed] [Google Scholar]
- 33.Fan Y, Han C, Zhang B. 2016. Recent advances in the development and application of nanoelectrodes. Analyst 141, 5474–5487. ( 10.1039/c6an01285j) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Clausmeyer J, Schuhmann W. 2016. Nanoelectrodes. Applications in electrocatalysis, single-cell analysis and high-resolution electrochemical imaging. TrAC Trends Anal. Chem. 79, 46–59. ( 10.1016/j.trac.2016.01.018) [DOI] [Google Scholar]
- 35.Kranz C. 2014. Recent advancements in nanoelectrodes and nanopipettes used in combined scanning electrochemical microscopy techniques. Analyst 139, 336–352. ( 10.1039/c3an01651j) [DOI] [PubMed] [Google Scholar]
- 36.Cox JT, Zhang B. 2012. Nanoelectrodes: recent advances and new directions. Annu. Rev. Anal. Chem. 5, 253–272. ( 10.1146/annurev-anchem-062011-143124) [DOI] [PubMed] [Google Scholar]
- 37.Li MSM, Filice FP, Ding Z. 2018. Submicron probes for scanning electrochemical microscopy. Can. J. Chem. 96, 328–335. ( 10.1139/cjc-2017-0611) [DOI] [Google Scholar]
- 38.Hengstenberg A, Blochl A, Dietzel ID, Schuhmann W. 2001. Spatially resolved detection of neurotransmitter secretion from individual cells by means of scanning electrochemical microscopy. Angew. Chem. Int. Ed. 40, 905–908. ( 10.1002/1521-3773(20010302)40:5%3C905::AID-ANIE905%3E3.0.CO;2-%23) [DOI] [PubMed] [Google Scholar]
- 39.Pitta BL, Schuhmann W, Schulte A. 2004. An advanced biological scanning electrochemical microscope (Bio-SECM) for studying individual living cells. Phys. Chem. Chem. Phys. 6, 4003 ( 10.1039/b405233a) [DOI] [Google Scholar]
- 40.Ballesteros KB, Schulte A, Schuhmann W. 2003. Constant-distance mode scanning electrochemical microscopy (SECM). Part I. Adaptation of a non-optical shear-force-based positioning mode for SECM tips. Chem. Eur. J. 9, 2025–2033. ( 10.1002/chem.200204267) [DOI] [PubMed] [Google Scholar]
- 41.Ballesteros KB, Schulte A, Schuhmann W. 2004. Constant-distance mode scanning electrochemical microscopy. Part II. High-resolution SECM imaging employing Pt nanoelectrodes as miniaturized scanning probes. Electroanalysis 16, 60–65. ( 10.1002/elan.200302918) [DOI] [Google Scholar]
- 42.Lee Y, Ding Z, Bard AJ. 2002. Combined scanning electrochemical/optical microscopy with shear force and current feedback. Anal. Chem. 74, 3634–3643. ( 10.1021/ac015713u) [DOI] [PubMed] [Google Scholar]
- 43.Takahashi Y, Hirano Y, Yasukawa T, Shiku H, Yamada H, Matsue T. 2006. Topographic, electrochemical, and optical images captured using standing approach mode scanning electrochemical/optical microscopy. Langmuir 22, 10 299–10 306. ( 10.1021/la0611763) [DOI] [PubMed] [Google Scholar]
- 44.Takahashi Y, Shiku H, Murata T, Yasukawa T, Matsue T. 2009. Transfected single-cell imaging by scanning electrochemical optical microscopy with shear force feedback regulation. Anal. Chem. 81, 9674–9681. ( 10.1021/ac901796r) [DOI] [PubMed] [Google Scholar]
- 45.Isik S, Schuhmann W. 2006. Detection of nitric oxide release from single cells by using constant-distance-mode scanning electrochemical microscopy. Angew. Chem. Int. Ed. 45, 7451–7454. ( 10.1002/anie.200601708) [DOI] [PubMed] [Google Scholar]
- 46.Kurulugama RT, Wipf DO, Takacs SA, Pongmayteegul S, Garris PA, Baur JE. 2005. Scanning electrochemical microscopy of model neurons: constant distance imaging. Anal. Chem. 77, 1111–1117. ( 10.1021/ac048571n) [DOI] [PubMed] [Google Scholar]
- 47.Takahashi Y. 2016. Development of high-resolution scanning electrochemical microscopy for nanoscale topography and electrochemical simultaneous imaging. Electrochemistry 84, 662–666. ( 10.5796/electrochemistry.84.662) [DOI] [Google Scholar]
- 48.Zhao X, Diakowski PM, Ding Z. 2010. Deconvoluting topography and spatial physiological activity of live macrophage cells by scanning electrochemical microscopy in constant-distance mode. Anal. Chem. 82, 8371–8373. ( 10.1021/ac101524v) [DOI] [PubMed] [Google Scholar]
- 49.Cahalan M, Neher E. 1992. Patch clamp techniques. An overview. Methods Enzymol. 207, 3–14. ( 10.1016/0076-6879(92)07003-7) [DOI] [PubMed] [Google Scholar]
- 50.Fathali H, Cans A-S. 2018. Amperometry methods for monitoring vesicular quantal size and regulation of exocytosis release. Pflug. Arch. Eur. J. Physiol. 470, 125–134. ( 10.1007/s00424-017-2069-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Takahashi Y, et al. 2012. Topographical and electrochemical nanoscale imaging of living cells using voltage-switching mode scanning electrochemical microscopy. Proc. Natl Acad. Sci. USA 109, 11 540–11 545. ( 10.1073/pnas.1203570109) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nebel M, Grützke S, Diab N, Schulte A, Schuhmann W. 2013. Visualization of oxygen consumption of single living cells by scanning electrochemical microscopy: the influence of the faradaic tip reaction. Angew. Chem. Int. Ed. 52, 6335–6338. ( 10.1002/anie.201301098) [DOI] [PubMed] [Google Scholar]
- 53.Nebel M, Eckhard K, Erichsen T, Schulte A, Schuhmann W. 2010. 4D shearforce-based constant-distance mode scanning electrochemical microscopy. Anal. Chem. 82, 7842–7848. ( 10.1021/ac1008805) [DOI] [PubMed] [Google Scholar]
- 54.Eckhard K, Chen X, Turcu F, Schuhmann W. 2006. Redox competition mode of scanning electrochemical microscopy (RC-SECM) for visualisation of local catalytic activity. Phys. Chem. Chem. Phys. 8, 5359 ( 10.1039/b609511a) [DOI] [PubMed] [Google Scholar]
- 55.Zhang MM-N, Long Y-T, Ding Z. 2012. Filming a live cell by scanning electrochemical microscopy: label-free imaging of the dynamic morphology in real time. Chem. Cent. J. 6, 20 ( 10.1186/1752-153X-6-20) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zhang MMN, Long Y-T, Ding Z. 2012. Cisplatin effects on evolution of reactive oxygen species from single human bladder cancer cells investigated by scanning electrochemical microscopy. J. Inorg. Biochem. 108, 115–122. ( 10.1016/j.jinorgbio.2011.11.010) [DOI] [PubMed] [Google Scholar]
- 57.Takahashi Y, Shevchuk AI, Novak P, Murakami Y, Shiku H, Korchev YE, Matsue T. 2010. Simultaneous noncontact topography and electrochemical imaging by SECM/SICM featuring ion current feedback regulation. J. Am. Chem. Soc. 132, 10 118–10 126. ( 10.1021/ja1029478) [DOI] [PubMed] [Google Scholar]
- 58.Takahashi Y, Ida H, Matsumae Y, Komaki H, Zhou Y, Kumatani A, Kanzaki M, Shiku H, Matsue T. 2017. 3D electrochemical and ion current imaging using scanning electrochemical-scanning ion conductance microscopy. Phys. Chem. Chem. Phys. 19, 26 728–26 733. ( 10.1039/c7cp05157c) [DOI] [PubMed] [Google Scholar]
- 59.Page A, Kang M, Armitstead A, Perry D, Unwin PR. 2017. Quantitative visualization of molecular delivery and uptake at living cells with self-referencing scanning ion conductance microscopy-scanning electrochemical microscopy. Anal. Chem. 89, 3021–3028. ( 10.1021/acs.analchem.6b04629) [DOI] [PubMed] [Google Scholar]
- 60.Fasching R, Ryu W-H, Bai S-J, Park J-S, Fabian T, Moseley J, Grossman A, Prinz F. 2007. AFM/EC nano probing of single cells and organelles. In Proc. Sensors 2007, Atlanta, GA, 28–31 October 2007, pp. 699–702. New York, NY: IEEE; ( 10.1109/ICSENS.2007.4388495) [DOI] [Google Scholar]
- 61.Bondarenko A, Lin T-E, Stupar P, Lesch A, Cortes-Salazar F, Girault HH, Pick H. 2016. Fixation and permeabilization approaches for scanning electrochemical microscopy of living cells. Anal. Chem. 88, 11 436–11 443. ( 10.1021/acs.analchem.6b02379) [DOI] [PubMed] [Google Scholar]
- 62.Kuss S, Kuss C, Trinh D, Schougaard SB, Mauzeroll J. 2013. Forced convection during scanning electrochemical microscopy imaging over living cells. Effect of topographies and kinetics on the microelectrode current. Electrochim. Acta 110, 42–48. ( 10.1016/j.electacta.2013.03.149) [DOI] [Google Scholar]
- 63.Razzaghi F, Seguin J, Amar A, Griveau S, Bedioui F. 2015. Biological cell morphology studies by scanning electrochemical microscopy imagery at constant height. Contrast enhancement using biocompatible conductive substrates. Electrochim. Acta 157, 95–100. ( 10.1016/j.electacta.2015.01.033) [DOI] [Google Scholar]
- 64.Perry D, Paulose Nadappuram B, Momotenko D, Voyias PD, Page A, Tripathi G, Frenguelli BG, Unwin PR. 2016. Surface charge visualization at viable living cells. J. Am. Chem. Soc. 138, 3152–3160. ( 10.1021/jacs.5b13153) [DOI] [PubMed] [Google Scholar]
- 65.Morkvenaite-Vilkonciene I, Genys P, Ramanaviciene A, Ramanavicius A. 2015. Scanning electrochemical impedance microscopy for investigation of glucose oxidase catalyzed reaction. Colloids Surf. B Biointerfaces 126, 598–602. ( 10.1016/j.colsurfb.2015.01.007) [DOI] [PubMed] [Google Scholar]
- 66.Ramanavicius A, Morkvenaite-Vilkonciene I, Kisieliute A, Petroniene J, Ramanaviciene A. 2017. Scanning electrochemical microscopy based evaluation of influence of pH on bioelectrochemical activity of yeast cells—Saccharomyces cerevisiae. Colloids Surf. B Biointerfaces 149, 1–6. ( 10.1016/j.colsurfb.2016.09.039) [DOI] [PubMed] [Google Scholar]
- 67.Zhang W, Wu H, Hsing I-M. 2015. Real-time label-free monitoring of Shewanella oneidensis MR-1 biofilm formation on electrode during bacterial electrogenesis using scanning electrochemical microscopy. Electroanalysis 27, 648–655. ( 10.1002/elan.201400578) [DOI] [Google Scholar]
- 68.Connell JL, Kim J, Shear JB, Bard AJ, Whiteley M. 2014. Real-time monitoring of quorum sensing in 3D-printed bacterial aggregates using scanning electrochemical microscopy. Proc. Natl Acad. Sci. USA 111, 18 255–18 260. ( 10.1073/pnas.1421211111) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Connell J, Kim J, Shear JB, Bard AJ, Whiteley M. 2014. Analyzing secondary metabolite production by 3D printed bacterial populations using scanning electrochemical microscopy. Microsc. Microanal. 20, 1182–1183. ( 10.1017/S1431927614007648) [DOI] [Google Scholar]
- 70.Parthasarathy M, Pemaiah B, Natesan R, Padmavathy SR, Pachiappan J. 2015. Real-time mapping of salt glands on the leaf surface of Cynodon dactylon L. using scanning electrochemical microscopy. Bioelectrochemistry 101, 159–164. ( 10.1016/j.bioelechem.2014.10.004) [DOI] [PubMed] [Google Scholar]
- 71.McKelvey K, Martin S, Robinson C, Unwin PR. 2013. Quantitative local photosynthetic flux measurements at isolated chloroplasts and thylakoid membranes using scanning electrochemical microscopy (SECM). J. Phys. Chem. B 117, 7878–7888. ( 10.1021/jp403048f) [DOI] [PubMed] [Google Scholar]
- 72.Arai T, Nishijo T, Matsumae Y, Zhou Y, Ino K, Shiku H, Matsue T. 2013. Noninvasive measurement of alkaline phosphatase activity in embryoid bodies and coculture spheroids with scanning electrochemical microscopy. Anal. Chem. 85, 9647–9654. ( 10.1021/ac401824q) [DOI] [PubMed] [Google Scholar]
- 73.Kumasako Y, Goto K, Koike M, Araki Y, Abe H, Utsunomiya T. 2013. Respiratory activity of single blastocysts measured by scanning electrochemical microscopy. The relationship between pre-freezing and post-warming. J. Mamm. Ova. Res. 30, 30–35. ( 10.1274/jmor.30.30) [DOI] [Google Scholar]
- 74.Matsumae Y, Takahashi Y, Ino K, Shiku H, Matsue T. 2014. Electrochemical monitoring of intracellular enzyme activity of single living mammalian cells by using a double-mediator system. Anal. Chim. Acta 842, 20–26. ( 10.1016/j.aca.2014.06.047) [DOI] [PubMed] [Google Scholar]
- 75.Hirano Y, Kodama M, Shibuya M, Maki Y, Komatsu Y. 2014. Analysis of beat fluctuations and oxygen consumption in cardiomyocytes by scanning electrochemical microscopy. Anal. Biochem. 447, 39–42. ( 10.1016/j.ab.2013.11.008) [DOI] [PubMed] [Google Scholar]
- 76.Zhan D, Li X, Nepomnyashchii AB, Alpuche-Aviles MA, Fan F-RF, Bard AJ. 2013. Characterization of Ag+ toxicity on living fibroblast cells by the ferrocenemethanol and oxygen response with the scanning electrochemical microscope. J. Electroanal. Chem. 688, 61–68. ( 10.1016/j.jelechem.2012.07.008) [DOI] [Google Scholar]
- 77.Kuss S, Cornut R, Beaulieu I, Mezour MA, Annabi B, Mauzeroll J. 2011. Assessing multidrug resistance protein 1-mediated function in cancer cell multidrug resistance by scanning electrochemical microscopy and flow cytometry. Bioelectrochemistry 82, 29–37. ( 10.1016/j.bioelechem.2011.04.008) [DOI] [PubMed] [Google Scholar]
- 78.Salamifar SE, Lai RY. 2013. Use of combined scanning electrochemical and fluorescence microscopy for detection of reactive oxygen species in prostate cancer cells. Anal. Chem. 85, 9417–9421. ( 10.1021/ac402367f) [DOI] [PubMed] [Google Scholar]
- 79.Wilburn JP, Ciobanu M, Cliffel DE. 2015. Scanning electrochemical microscopy of individual pancreatic islets. J. Electrochem. Soc. 163, H3077–H3082. ( 10.1149/2.0111604jes) [DOI] [Google Scholar]
- 80.Filice FP, Li MSM, Henderson JD, Ding Z. 2016. Mapping Cd2+-induced membrane permeability changes of single live cells by means of scanning electrochemical microscopy. Anal. Chim. Acta 908, 85–94. ( 10.1016/j.aca.2015.12.027) [DOI] [PubMed] [Google Scholar]
- 81.Li MSM, Filice FP, Ding Z. 2014. A time course study of cadmium effect on membrane permeability of single human bladder cancer cells using scanning electrochemical microscopy. J. Inorg. Biochem. 136, 177–183. ( 10.1016/j.jinorgbio.2014.02.009) [DOI] [PubMed] [Google Scholar]
- 82.Henderson JD, Filice FP, Li MSM, Ding Z. 2017. Tracking live-cell response to hexavalent chromium toxicity by using scanning electrochemical microscopy. ChemElectroChem 4, 856–863. ( 10.1002/celc.201600783) [DOI] [Google Scholar]
- 83.Filice FP, Li MSM, Wong JM, Ding Z. 2018. The effects of long duration chronic exposure to hexavalent chromium on single live cells interrogated by scanning electrochemical microscopy. J. Inorg. Biochem. 182, 222–229. ( 10.1016/j.jinorgbio.2018.02.009) [DOI] [PubMed] [Google Scholar]
- 84.Ning X, Xiong Q, Zhang F, He P. 2018. Simultaneous detection of tumor markers in lung cancer using scanning electrochemical microscopy. J. Electroanal. Chem. 812, 101–106. ( 10.1016/j.jelechem.2018.01.061) [DOI] [Google Scholar]
- 85.Joshi VS, Sheet PS, Cullin N, Kreth J, Koley D. 2017. Real-time metabolic interactions between two bacterial species using a carbon-based pH microsensor as a scanning electrochemical microscopy probe. Anal. Chem. 89, 11 044–11 052. ( 10.1021/acs.analchem.7b03050) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bucher ES, Wightman RM. 2015. Electrochemical analysis of neurotransmitters. Annu. Rev. Anal. Chem. 8, 239–261. ( 10.1146/annurev-anchem-071114-040426) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lemaître F, Guille Collignon M, Amatore C. 2014. Recent advances in electrochemical detection of exocytosis. Electrochim. Acta 140, 457–466. ( 10.1016/j.electacta.2014.02.059) [DOI] [Google Scholar]
- 88.Trouillon R, Passarelli MK, Wang J, Kurczy ME, Ewing AG. 2013. Chemical analysis of single cells. Anal. Chem. 85, 522–542. ( 10.1021/ac303290s) [DOI] [PubMed] [Google Scholar]
- 89.Amatore C, Collignon MG, Lemaître F. 2013. Electrochemical detection of exocytosis: a survey from the earliest amperometry at carbon fiber ultramicroelectrodes to recent integrated systems. In Electrochemical biosensors (ed. Cosnier S.), pp. 1–52. Boca Raton, FL: Taylor & Francis Group, LLC. [Google Scholar]
- 90.Li Y-T, Zhang S-H, Wang L, Xiao R-R, Liu W, Zhang X-W, Zhou Z, Amatore C, Huang W-H. 2014. Nanoelectrode for amperometric monitoring of individual vesicular exocytosis inside single synapses. Angew. Chem. Int. Ed. 53, 12 456–12 460. ( 10.1002/anie.201404744) [DOI] [PubMed] [Google Scholar]
- 91.Li Y-T, Zhang S-H, Wang X-Y, Zhang X-W, Oleinick AI, Svir I, Amatore C, Huang W-H. 2015. Real-time monitoring of discrete synaptic release events and excitatory potentials within self-reconstructed neuromuscular junctions. Angew. Chem. Int. Ed. 54, 9313–9318. ( 10.1002/anie.201503801) [DOI] [PubMed] [Google Scholar]
- 92.Steiner RA, Oehme M, Ammann D, Simon W. 1979. Neutral carrier sodium ion-selective microelectrode for intracellular studies. Anal. Chem. 51, 351–353. ( 10.1021/ac50039a008) [DOI] [PubMed] [Google Scholar]
- 93.Haemmerli A, Janata J, Brown HM. 1980. Ion-selective electrode for intracellular potassium measurements. Anal. Chem. 52, 1179–1182. ( 10.1021/ac50058a006) [DOI] [PubMed] [Google Scholar]
- 94.Lanter F, Erne D, Ammann D, Simon W. 1980. Neutral carrier based ion-selective electrode for intracellular magnesium activity studies. Anal. Chem. 52, 2400–2402. ( 10.1021/ac50064a037) [DOI] [Google Scholar]
- 95.Miller AJ. 1995. Ion-selective microelectrodes for measurement of intracellular ion concentrations. Methods Cell Biol. 49, 275–291. ( 10.1016/S0091-679X(08)61460-0) [DOI] [PubMed] [Google Scholar]
- 96.Adlimoghaddam A, Weihrauch D, O'Donnell MJ. 2014. Localization of K+, H+, Na+ and Ca2+ fluxes to the excretory pore in Caenorhabditis elegans. Application of scanning ion-selective microelectrodes. J. Exp. Biol. 217, 4119–4122. ( 10.1242/jeb.112441) [DOI] [PubMed] [Google Scholar]
- 97.Ruan L, Wei K, Wang L, Cheng H, Zhang F, Wu L, Bai P, Zhang C. 2016. Characteristics of NH4+ and NO3− fluxes in tea (Camellia sinensis) roots measured by scanning ion-selective electrode technique. Sci. Rep. 6, 38370 ( 10.1038/srep38370) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Li L-Z, Yu S-Y, Peijnenburg WJGM, Luo Y-M. 2017. Determining the fluxes of ions (Pb2+, Cu2+ and Cd2+) at the root surface of wetland plants using the scanning ion-selective electrode technique. Plant Soil 414, 1–12. ( 10.1007/s11104-016-3109-5) [DOI] [Google Scholar]
- 99.Denuault G, Troise Frank MH, Peter LM. 1992. Scanning electrochemical microscopy. Potentiometric probing of ion fluxes. Faraday Disc. 94, 23 ( 10.1039/fd9929400023) [DOI] [Google Scholar]
- 100.Horrocks BR, Mirkin MV, Pierce DT, Bard AJ, Nagy G, Toth K. 1993. Scanning electrochemical microscopy. 19. Ion-selective potentiometric microscopy. Anal. Chem. 65, 1213–1224. ( 10.1021/ac00057a019) [DOI] [Google Scholar]
- 101.Tóth K, Nagy G, Wei C, Bard AJ. 1995. Novel application of potentiometric microelectrodes. Scanning potentiometric microscopy. Electroanalysis 7, 801–810. ( 10.1002/elan.1140070904) [DOI] [Google Scholar]
- 102.Gray NJ, Unwin PR. 2000. Simple procedure for the fabrication of silver/silver chloride potentiometric electrodes with micrometre and smaller dimensions. Application to scanning electrochemical microscopy. Analyst 125, 889–893. ( 10.1039/b001659o) [DOI] [Google Scholar]
- 103.Etienne M, Schulte A, Mann S, Jordan G, Dietzel ID, Schuhmann W. 2004. Constant-distance mode scanning potentiometry. 1. Visualization of calcium carbonate dissolution in aqueous solution. Anal. Chem. 76, 3682–3688. ( 10.1021/ac0349227) [DOI] [PubMed] [Google Scholar]
- 104.Etienne M, Dierkes P, Erichsen T, Schuhmann W, Fritsch I. 2007. Constant-distance mode scanning potentiometry. High resolution pH measurements in three-dimensions. Electroanalysis 19, 318–323. ( 10.1002/elan.200603735) [DOI] [Google Scholar]
- 105.Yamada H, Ikuta Y, Koike T, Matsue T. 2008. Fabrication of a shear force-based ion-selective capillary probe for scanning electrochemical microscopy. Chem. Lett. 37, 392–393. ( 10.1246/cl.2008.392) [DOI] [Google Scholar]
- 106.Takami T, Park BH, Kawai T. 2014. Nanopipette exploring nanoworld. Nano Converg. 1, 17 ( 10.1186/s40580-014-0017-3) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lazenby R, White R. 2018. Advances and perspectives in chemical imaging in cellular environments using electrochemical methods. Chemosensors 6, 24 ( 10.3390/chemosensors6020024) [DOI] [Google Scholar]
- 108.Munteanu R-E, Stănică L, Gheorghiu M, Gáspár S. 2018. Measurement of the extracellular pH of adherently growing mammalian cells with high spatial resolution using a voltammetric pH microsensor. Anal. Chem. 90, 6899–6905. ( 10.1021/acs.analchem.8b01124) [DOI] [PubMed] [Google Scholar]
- 109.Amemiya S, Bard AJ. 2000. Scanning electrochemical microscopy. 40. Voltammetric ion-selective micropipet electrodes for probing ion transfer at bilayer lipid membranes. Anal. Chem. 72, 4940–4948. ( 10.1021/ac0004207) [DOI] [PubMed] [Google Scholar]
- 110.Yamada H, Haraguchi D, Yasunaga K. 2014. Fabrication and characterization of a K+-selective nanoelectrode and simultaneous imaging of topography and local K+ flux using scanning electrochemical microscopy. Anal. Chem. 86, 8547–8552. ( 10.1021/ac502444y) [DOI] [PubMed] [Google Scholar]
- 111.Zhou L, Gong Y, Sunq A, Hou J, Baker LA. 2016. Capturing rare conductance in epithelia with potentiometric-scanning ion conductance microscopy. Anal. Chem. 88, 9630–9637. ( 10.1021/acs.analchem.6b02392) [DOI] [PubMed] [Google Scholar]
- 112.Zhou L, Gong Y, Hou J, Baker LA. 2017. Quantitative visualization of nanoscale ion transport. Anal. Chem. 89, 13 603–13 609. ( 10.1021/acs.analchem.7b04139) [DOI] [PubMed] [Google Scholar]
- 113.Fan F-RF, Bard AJ. 1999. Imaging of biological macromolecules on mica in humid air by scanning electrochemical microscopy. Proc. Natl Acad. Sci. USA 96, 14 222–14 227. ( 10.1073/pnas.96.25.14222) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zoski CG. 2015. Review—advances in scanning electrochemical microscopy (SECM). J. Electrochem. Soc. 163, H3088–H3100. ( 10.1149/2.0141604jes) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Gyurcsanyi RE, Jagerszki G, Kiss G, Toth K. 2004. Chemical imaging of biological systems with the scanning electrochemical microscope. Bioelectrochemistry 63, 207–215. ( 10.1016/j.bioelechem.2003.12.011) [DOI] [PubMed] [Google Scholar]
- 116.Polcari D, Dauphin-Ducharme P, Mauzeroll J. 2016. Scanning electrochemical microscopy: a comprehensive review of experimental parameters from 1989 to 2015. Chem. Rev. 116, 13 234–13 278. ( 10.1021/acs.chemrev.6b00067) [DOI] [PubMed] [Google Scholar]
- 117.Nogala W, Szot K, Burchardt M, Roelfs F, Rogalski J, Opallo M, Wittstock G. 2010. Feedback mode SECM study of laccase and bilirubin oxidase immobilised in a sol-gel processed silicate film. Analyst 135, 2051–2058. ( 10.1039/c0an00068j) [DOI] [PubMed] [Google Scholar]
- 118.Lin T-E, Lesch A, Li C-L, Girault HH. 2017. Mapping the antioxidant activity of apple peels with soft probe scanning electrochemical microscopy. J. Electroanal. Chem. 786, 120–128. ( 10.1016/j.jelechem.2017.01.015) [DOI] [Google Scholar]
- 119.Zhao F, et al. 2017. A novel versatile microbiosensor for local hydrogen detection by means of scanning photoelectrochemical microscopy. Biosens. Bioelectron. 94, 433–437. ( 10.1016/j.bios.2017.03.037) [DOI] [PubMed] [Google Scholar]
- 120.Zhao F, Conzuelo F, Hartmann V, Li H, Nowaczyk MM, Plumeré N, Rögner M, Schuhmann W. 2015. Light induced H2 evolution from a biophotocathode based on photosystem 1 - Pt nanoparticles complexes integrated in solvated redox polymers films. J. Phys. Chem. B 119, 13 726–13 731. ( 10.1021/acs.jpcb.5b03511) [DOI] [PubMed] [Google Scholar]
- 121.Ziller C, Lin J, Knittel P, Friedrich L, Andronescu C, Pöller S, Schuhmann W, Kranz C. 2017. Poly(benzoxazine) as an immobilization matrix for miniaturized ATP and glucose biosensors. ChemElectroChem 4, 864–871. ( 10.1002/celc.201600765) [DOI] [Google Scholar]
- 122.Polcari D, Perry SC, Pollegioni L, Geissler M, Mauzeroll J. 2017. Localized detection of D-serine by using an enzymatic amperometric biosensor and scanning electrochemical microscopy. ChemElectroChem 4, 920–926. ( 10.1002/celc.201600766) [DOI] [Google Scholar]
- 123.Gdor E, Shemesh S, Magdassi S, Mandler D. 2015. Multienzyme inkjet printed 2D arrays. ACS Appl. Mater. Interfaces 7, 17 985–17 992. ( 10.1021/acsami.5b04726) [DOI] [PubMed] [Google Scholar]
- 124.Kai T, Chen S, Monterroso E, Zhou F. 2015. Continuous nanoflow-scanning electrochemical microscopy: voltammetric characterization and application for accurate and reproducible imaging of enzyme-labeled protein microarrays. Anal. Chem. 87, 4523–4529. ( 10.1021/acs.analchem.5b00625) [DOI] [PubMed] [Google Scholar]
- 125.Eifert A, Kranz C. 2014. Hyphenating atomic force microscopy. Anal. Chem. 86, 5190–5200. ( 10.1021/ac5008128) [DOI] [PubMed] [Google Scholar]
- 126.Kranz C, Kueng A, Lugstein A, Bertagnolli E, Mizaikoff B. 2004. Mapping of enzyme activity by detection of enzymatic products during AFM imaging with integrated SECM-AFM probes. Ultramicroscopy 100, 127–134. ( 10.1016/j.ultramic.2003.10.004) [DOI] [PubMed] [Google Scholar]
- 127.Lin T-E, Cortés-Salazar F, Lesch A, Qiao L, Bondarenko A, Girault HH. 2015. Multiple scanning electrochemical microscopy mapping of tyrosinase in micro-contact printed fruit samples on polyvinylidene fluoride membrane. Electrochim. Acta 179, 57–64. ( 10.1016/j.electacta.2015.03.224) [DOI] [Google Scholar]
- 128.Nault L, et al. 2015. Electrochemical atomic force microscopy imaging of redox-immunomarked proteins on native potyviruses. From subparticle to single-protein resolution. ACS Nano 9, 4911–4924. ( 10.1021/acsnano.5b00952) [DOI] [PubMed] [Google Scholar]
- 129.Stratmann L, Gebala M, Schuhmann W. 2013. A chemical lift-off process: removing non-specific adsorption in an electrochemical Epstein-Barr virus immunoassay. Chemphyschem 14, 2198–2207. ( 10.1002/cphc.201300029) [DOI] [PubMed] [Google Scholar]
- 130.Conzuelo F, Stratmann L, Grützke S, Pingarrón JM, Schuhmann W. 2014. Detection and quantification of sulfonamide antibiotic residues in milk using scanning electrochemical microscopy. Electroanalysis 26, 481–487. ( 10.1002/elan.201300577) [DOI] [Google Scholar]
- 131.Conzuelo F, Grützke S, Stratmann L, Pingarrón JM, Schuhmann W. 2016. Interrogation of immunoassay platforms by SERS and SECM after enzyme-catalyzed deposition of silver nanoparticles. Microchim. Acta 183, 281–287. ( 10.1007/s00604-015-1654-x) [DOI] [Google Scholar]
- 132.Holmes JL, Davis F, Collyer SD, Higson SPJ. 2011. A new application of scanning electrochemical microscopy for the label-free interrogation of antibody-antigen interactions. Anal. Chim. Acta 689, 206–211. ( 10.1016/j.aca.2011.01.033) [DOI] [PubMed] [Google Scholar]
- 133.Holmes JL, Davis F, Collyer SD, Higson SPJ. 2012. A new application of scanning electrochemical microscopy for the label-free interrogation of antibody-antigen interactions: Part 2. Anal. Chim. Acta 741, 1–8. ( 10.1016/j.aca.2012.07.004) [DOI] [PubMed] [Google Scholar]
- 134.Nunes KC, Szunerits S, Wittstock G. 2007. Scanning electrochemical microscopy (SECM) based detection of oligonucleotide hybridization and simultaneous determination of the surface concentration of immobilized oligonucleotides on gold. Electroanalysis 19, 1258–1267. ( 10.1002/elan.200703862) [DOI] [Google Scholar]
- 135.Neugebauer S, Zimdars A, Liepold P, Gębala M, Schuhmann W, Hartwich G. 2009. Optimization of an electrochemical DNA assay by using a 48-electrode array and redox amplification studies by means of scanning electrochemical microscopy. Chembiochem 10, 1193–1199. ( 10.1002/cbic.200800767) [DOI] [PubMed] [Google Scholar]
- 136.Zhang Z, Zhou J, Tang A, Wu Z, Shen G, Yu R. 2010. Scanning electrochemical microscopy assay of DNA based on hairpin probe and enzymatic amplification biosensor. Biosens. Bioelectron. 25, 1953–1957. ( 10.1016/j.bios.2010.01.013) [DOI] [PubMed] [Google Scholar]
- 137.Fan H, Jiao F, Chen H, Zhang F, Wang Q, He P, Fang Y. 2013. Qualitative and quantitative detection of DNA amplified with HRP-modified SiO2 nanoparticles using scanning electrochemical microscopy. Biosens. Bioelectron. 47, 373–378. ( 10.1016/j.bios.2013.03.027) [DOI] [PubMed] [Google Scholar]
- 138.Fan H, Wang X, Jiao F, Zhang F, Wang Q, He P, Fang Y. 2013. Scanning electrochemical microscopy of DNA hybridization on DNA microarrays enhanced by HRP-modified SiO2 nanoparticles. Anal. Chem. 85, 6511–6517. ( 10.1021/ac4011155) [DOI] [PubMed] [Google Scholar]
- 139.Chen B, Hu Q, Xiong Q, Zhang F, He P. 2016. An ultrasensitive scanning electrochemical microscopy (SECM)-based DNA biosensing platform amplified with the long self-assembled DNA concatemers. Electrochim. Acta 192, 127–132. ( 10.1016/j.electacta.2015.12.102) [DOI] [Google Scholar]
- 140.Jambrec D, Conzuelo F, Zhao B, Schuhmann W. 2018. Potential-pulse assisted thiol chemisorption minimizes non-specific adsorptions in DNA assays. Electrochim. Acta 276, 233–239. ( 10.1016/j.electacta.2018.04.180) [DOI] [Google Scholar]
- 141.Wain AJ, Zhou F. 2008. Scanning electrochemical microscopy imaging of DNA microarrays using methylene blue as a redox-active intercalator. Langmuir 24, 5155–5160. ( 10.1021/la703922v) [DOI] [PubMed] [Google Scholar]
- 142.Gorodetsky AA, Hammond WJ, Hill MG, Slowinski K, Barton JK. 2008. Scanning electrochemical microscopy of DNA monolayers modified with Nile Blue. Langmuir 24, 14 282–14 288. ( 10.1021/la8029243) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Turcu F, Schulte A, Hartwich G, Schuhmann W. 2004. Imaging immobilised ssDNA and detecting DNA hybridisation by means of the repelling mode of scanning electrochemical microscopy (SECM). Biosens. Bioelectron. 20, 925–932. ( 10.1016/j.bios.2004.06.011) [DOI] [PubMed] [Google Scholar]
- 144.Turcu F, Schulte A, Hartwich G, Schuhmann W. 2004. Label-free electrochemical recognition of DNA hybridization by means of modulation of the feedback current in SECM. Angew. Chem. Int. Ed. 43, 3482–3485. ( 10.1002/anie.200454228) [DOI] [PubMed] [Google Scholar]
- 145.Shamsi MH, Kraatz H-B. 2013. Electrochemical signature of mismatch in overhang DNA films: a scanning electrochemical microscopic study. Analyst 138, 3538–3543. ( 10.1039/c3an36810f) [DOI] [PubMed] [Google Scholar]
- 146.Diakowski PM, Kraatz H-B. 2009. Detection of single-nucleotide mismatches using scanning electrochemical microscopy. Chem. Commun. 0, 1189–1191. ( 10.1039/b819876d) [DOI] [PubMed] [Google Scholar]
- 147.Shamsi MH, Kraatz H-B. 2010. Probing nucleobase mismatch variations by electrochemical techniques: exploring the effects of position and nature of the single-nucleotide mismatch. Analyst 135, 2280–2285. ( 10.1039/c0an00184h) [DOI] [PubMed] [Google Scholar]
- 148.Diakowski PM, Kraatz H-B. 2011. Towards the electrochemical identification of species. Chem. Commun. 47, 1431–1433. ( 10.1039/c0cc03940c) [DOI] [PubMed] [Google Scholar]
- 149.Shamsi MH, Kraatz H-B. 2011. Electrochemical identification of artificial oligonucleotides related to bovine species. Potential for identification of species based on mismatches in the mitochondrial cytochrome C1 oxidase gene. Analyst 136, 4724–4731. ( 10.1039/c1an15414a) [DOI] [PubMed] [Google Scholar]
- 150.Kranz C. 2016. Diamond as advanced material for scanning probe microscopy tips. Electroanalysis 28, 35–45. ( 10.1002/elan.201500630) [DOI] [Google Scholar]
- 151.Lemay SG, Kang S, Mathwig K, Singh PS. 2013. Single-molecule electrochemistry: present status and outlook. Acc. Chem. Res. 46, 369–377. ( 10.1021/ar300169d) [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This work does not include any additional experimental data.